Associations of Probiotic Fermented Milk (PFM) and Yogurt Consumption with Bifidobacterium and Lactobacillus Components of the Gut Microbiota in Healthy Adults
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Experimental Design
2.3. Microbiota Analysis
2.4. Statistical Analysis
3. Results
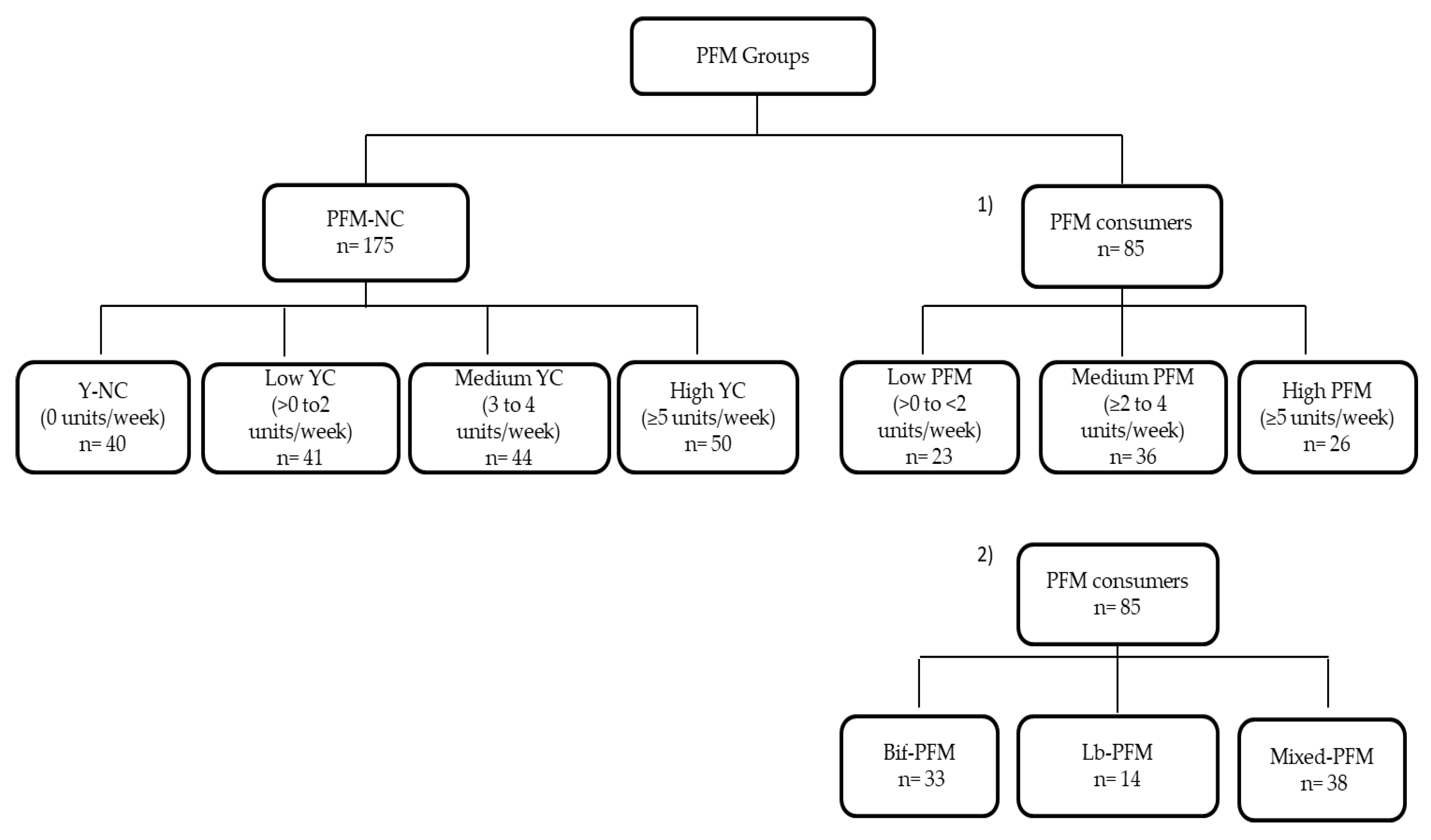
3.1. Subjects’ Distribution and Demographics, and Anthropometric and Lifestyle Characteristics of PFM Groups
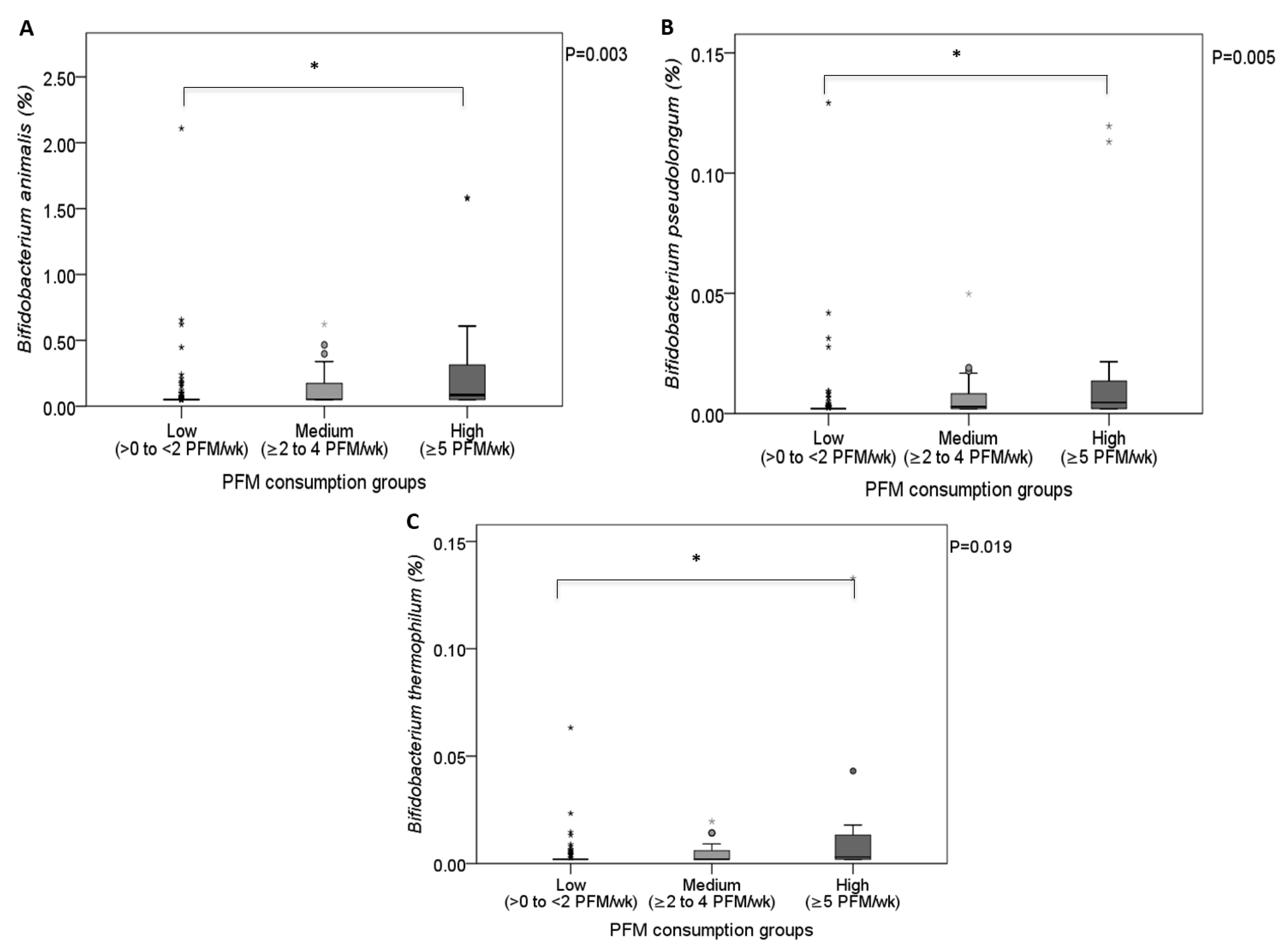
3.2. Relationship of PFM Consumption with Lactobacillus and Bifidobacterium Levels
3.3. Relationship of YC with Lactobacillus and Streptococcus Levels
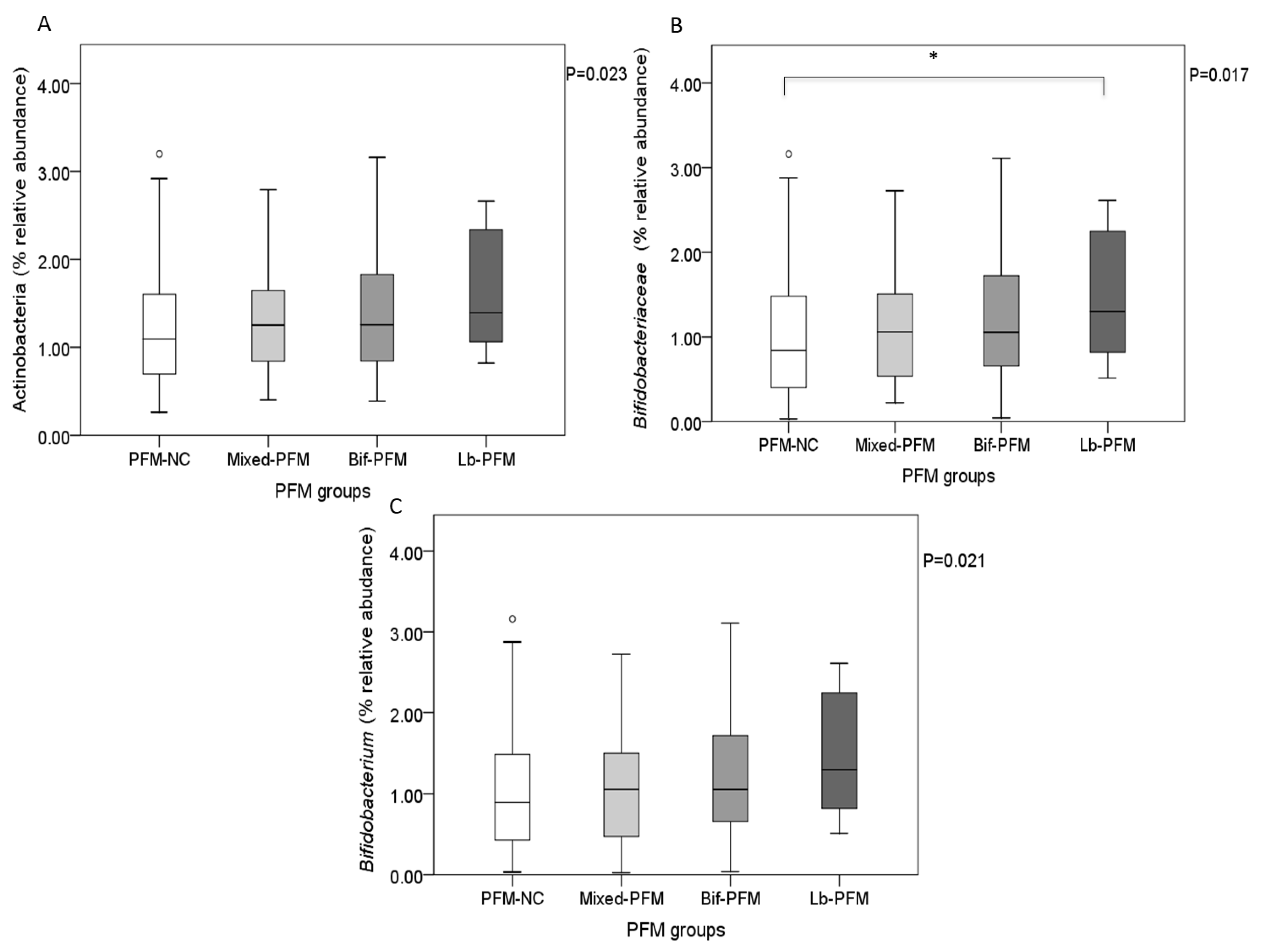
3.4. Relationship of PFM and Yogurt Consumption with the Overall Gut Microbiota Composition
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef]
- Lozupone, C.A.; Stombaugh, J.I.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 2012, 489, 220–230. [Google Scholar] [CrossRef]
- Tap, J.; Mondot, S.; Levenez, F.; Pelletier, E.; Caron, C.; Furet, J.P.; Ugarte, E.; Muñoz-Tamayo, R.; Paslier, D.L.; Nalin, R.; et al. Towards the human intestinal microbiota phylogenetic core. Environ. Microbiol. 2009, 11, 2574–2584. [Google Scholar] [CrossRef]
- Reuter, G. The Lactobacillus and Bifidobacterium microflora of the human intestine: Composition and succession. Curr. Issues Intest. Microbiol. 2001, 2, 43–53. [Google Scholar]
- Kok, C.R.; Hutkins, R. Yogurt and other fermented foods as sources of health-promoting bacteria. Nutr. Rev. 2018, 76, 4–15. [Google Scholar] [CrossRef]
- Derrien, M.; van Hylckama Vlieg, J.E. Fate, activity, and impact of ingested bacteria within the human gut microbiota. Trends Microbiol. 2015, 23, 354–366. [Google Scholar] [CrossRef]
- McNulty, N.P.; Yatsunenko, T.; Hsiao, A.; Faith, J.J.; Muegge, B.D.; Goodman, A.L.; Henrissat, B.; Oozeer, R.; Cools-Portier, S.; Gobert, G.; et al. The impact of a consortium of fermented milk strains on the gut microbiome of gnotobiotic mice and monozygotic twins. Sci. Transl. Med. 2011, 3, 106ra106. [Google Scholar] [CrossRef]
- Bezkorovainy, A. Probiotics: Determinants of survival and growth in the gut. Am. J. Clin. Nutr. 2001, 73, 399S–405S. [Google Scholar] [CrossRef]
- Uyeno, Y.; Sekiguchi, Y.; Kamagata, Y. Impact of consumption of probiotic lactobacilli-containing yogurt on microbial composition in human feces. Int. J. Food Microbiol. 2008, 122, 16–22. [Google Scholar] [CrossRef]
- Del Campo, R.; Bravo, D.; Cantón, R.; Ruiz-Garbajosa, P.; García-Albiach, R.; Montesi-Libois, A.; Yuste, F.J.; Abraira, V.; Baquero, F. Scarce Evidence of Yogurt Lactic Acid Bacteria in Human Feces after Daily Yogurt Consumption by Healthy Volunteers. Appl. Environ. Microb. 2005, 71, 547–549. [Google Scholar] [CrossRef]
- García-Albiach, R.; Pozuelo de Felipe, M.; Angulo, S.; Morosini, M.I.; Bravo, D.; Baquero, F.; del Campo, R. Molecular analysis of yogurt containing Lactobacillus delbrueckii subsp. bulgaricus and Streptococcus thermophilus in human intestinal microbiota. Am. J. Clin. Nutr. 2008, 87, 91–96. [Google Scholar] [CrossRef]
- Elli, M.; Callegari, M.L.; Ferrari, S.; Bessi, E.; Cattivelli, D.; Soldi, S.; Morelli, L.; Goupil Feuillerat, N.; Antoine, J.M. Survival of yogurt bacteria in the human gut. Appl. Environ. Microbiol. 2006, 72, 5113–5117. [Google Scholar] [CrossRef]
- Mater, D.D.; Bretigny, L.; Firmesse, O.; Flores, M.J.; Mogenet, A.; Bresson, J.L.; Corthier, G. Streptococcus thermophilus and Lactobacillus delbrueckii subsp. bulgaricus survive gastrointestinal transit of healthy volunteers consuming yogurt. FEMS Microbiol. Lett. 2005, 250, 185–187. [Google Scholar] [CrossRef]
- Morelli, L. Yogurt, living cultures, and gut health. Am. J. Clin. Nutr. 2014, 99, 1248S–1250S. [Google Scholar] [CrossRef]
- Lisko, D.J.; Johnston, G.P.; Johnston, C.G. Effects of Dietary Yogurt on the Healthy Human Gastrointestinal (GI) Microbiome. Microorganisms 2017, 5, 6. [Google Scholar] [CrossRef]
- Redondo, N.; García-González, N.; Diaz-Prieto, L.E.; Olmedilla-Alonso, B.; Martín-Diana, A.B.; Asensio-Vegas, C.; Nova, E. Effects of ewe’s milk yogurt (whole and semi-skimmed) and cow’s milk yogurt on inflammation markers and gut microbiota of subjects with borderline-high plasma cholesterol levels: A crossover study. Eur. J. Nutr. 2018. [Google Scholar] [CrossRef]
- Farhana Fazilaha, N.; Ariffac, A.; Khayatbc, M.E.; Rios-Solis, L.; Hali, D. Influence of probiotics, prebiotics, synbiotics and bioactive phytochemicals on the formulation of functional yogurt. J. Funct. Foods 2018, 48, 387–399. [Google Scholar] [CrossRef]
- Filteau, M.; Matamoros, S.; Savard, P.; Roy, D. Molecular monitoring of fecal microbiota in healthy adults following probiotic yogurt intake. Pharma Nutr. 2013, 1, 123–129. [Google Scholar] [CrossRef]
- Kim, S.W.; Suda, W.; Kim, S.; Oshima, K.; Fukuda, S.; Ohno, H.; Morita, H.; Hattori, M. Robustness of gut microbiota of healthy adults in response to probiotic intervention revealed by high-throughput pyrosequencing. DNA Res. 2013, 20, 241–253. [Google Scholar] [CrossRef]
- Burton, K.J.; Rosikiewicz, M.; Pimentel, G.; Bütikofer, U.; von Ah, U.; Voirol, M.J.; Croxatto, A.; Aeby, S.; Drai, J.; McTernan, P.G.; et al. Probiotic yogurt and acidified milk similarly reduce postprandial inflammation and both alter the gut microbiota of healthy, young men. Br. J. Nutr. 2017, 117, 1312–1322. [Google Scholar] [CrossRef]
- Tanaka, Y.; Takami, K.; Nishijima, T.; Aoki, R.; Mawatari, T.; Ikeda, T. Short- and long-term dynamics in the intestinal microbiota following ingestion of Bifidobacterium animalis subsp. lactis GCL2505. BMFH 2015, 34, 77–85. [Google Scholar] [CrossRef]
- Matsumoto, K.; Takada, T.; Shimizu, K.; Moriyama, K.; Kawakami, K.; Hirano, K.; Kajimoto, O.; Nomoto, K. Effects of a probiotic fermented milk beverage containing Lactobacillus casei strain Shirota on defecation frequency, intestinal microbiota, and the intestinal environment of healthy individuals with soft stools. J. Biosci. Bioeng. 2010, 110, 547–552. [Google Scholar] [CrossRef]
- Savard, P.; Lamarche, B.; Paradis, M.E.; Thiboutot, H.; Laurin, É.; Roy, D. Impact of Bifidobacterium animalis subsp. lactis BB-12 and Lactobacillus acidophilus LA-5-containing yoghurt, on fecal bacterial counts of healthy adults. Int. J. Food Microbiol. 2011, 149, 50–57. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, L.; Guo, Z.; Sun, Z.; Gesudu, Q.; Kwok, L.; Menghebilige; Zhang, H. 454 pyrosequencing reveals changes in the faecal microbiota of adults consuming Lactobacillus casei Zhang. FEMS Microbiol. Ecol. 2014, 88, 612–622. [Google Scholar] [CrossRef]
- Fooks, L.J.; Gibson, G.R. Probiotics as modulators of the gut flora. Br. J. Nutr. 2002, 88, S39–S49. [Google Scholar] [CrossRef]
- Eales, J.; Gibson, P.; Whorwell, P.; Kellow, J.; Yellowlees, A.; Perry, R.H.; Edwards, M.; King, S.; Wood, H.; Glanville, J. Systematic review and meta-analysis: The effects of fermented milk with Bifidobacterium lactis CNCM I-2494 and lactic acid bacteria on gastrointestinal discomfort in the general adult population. Ther. Adv. Gastroenterol. 2017, 10, 74–88. [Google Scholar] [CrossRef]
- Delcenserie, V.; Martel, D.; Lamoureux, M.; Amiot, J.; Boutin, Y.; Roy, D. Immunomodulatory effects of probiotics in the intestinal tract. Curr. Issues Mol. Biol. 2008, 10, 37–54. [Google Scholar]
- Lemon, K.P.; Armitage, G.C.; Relman, D.A.; Fischbach, M.A. Microbiota-targeted therapies: An ecological perspective. Sci. Transl. Med. 2002, 4, 137rv5. [Google Scholar] [CrossRef]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Global Strategy on Diet, Physical Activity and Health, 57th World Health Assembly (WHA); WHO: Geneva, Switzerland, 2004. [Google Scholar]
- Vulevic, J.; Juric, A.; Tzortzis, G.; Gibson, G.R. A mixture of trans-galactooligosaccharides reduces markers of metabolic syndrome and modulates the fecal microbiota and immune function of overweight adults. J. Nutr. 2013, 143, 324–331. [Google Scholar] [CrossRef]
- Revicki, D.A.; Wood, M.; Wiklund, I.; Crawley, J. Reliability and validity of the Gastrointestinal Symptom Rating Scale in patients with gastroesophageal reflux disease. Qual. Life Res. 1998, 7, 75–83. [Google Scholar] [CrossRef]
- Martin-Moreno, J.M.; Boyle, P.; Gorgojo, L.; Maisonneuve, P.; Fernandez-Rodriguez, J.C.; Salvini, S.; Willett, W.C. Development and validation of a food frequency questionnaire in Spain. Int. J. Epidemiol. 1993, 22, 512–519. [Google Scholar] [CrossRef]
- Dore, J.; Ehrlich, S.D.; Levenez, F.; Pelletier, E.; Alberti, A.; Bertrand, L.; Bork, P.; Costea, P.I.; Sunagawa, S.; Guarner, F.; et al. IHMS_SOP 06 V1: Standard operating procedure for faecal samples DNA extraction, Protocol, Q. International Human Microbiome Standards. 2015. Available online: http://www.microbiome-standards.org (accessed on 13 May 2016).
- Illumina. Available online: https://support.illumina.com/help/BaseSpace_App_16S_Metagenomics_help/16S_Metagenomics_App_Help.htm#Vault/Informatics/Sequencing_Analysis/Apps/swSEQ_mAPP_16S_Read_Classification.htm%3FToc (accessed on 20 December 2018).
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate-a practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B Meth. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Khalesi, S.; Bellissimo, N.; Vandelanotte, C.; Williams, S.; Stanley, D.; Irwin, C. A review of probiotic supplementation in healthy adults: Helpful or hype? Eur. J. Clin. Nutr. 2019, 73, 24–37. [Google Scholar] [CrossRef]
- Ouwehand, A.C. A review of dose-responses of probiotics in human studies. Benef. Microbes 2017, 8, 143–151. [Google Scholar] [CrossRef]
- Larsen, C.N.; Nielsen, S.; Kaestel, P.; Brockmann, E.; Bennedsen, M.; Christensen, H.R.; Eskesen, D.C.; Jacobsen, B.L.; Michaelsen, K.F. Dose-response study of probiotic bacteria Bifidobacterium animalis subsp. lactis BB-12 and Lactobacillus paracasei subsp paracasei CRL-341 in healthy young adults. Eur. J. Clin. Nutr. 2006, 60, 1284–1293. [Google Scholar] [CrossRef]
- Kristensen, N.B.; Bryrup, T.; Allin, K.H.; Nielsen, T.; Hansen, T.H.; Pedersen, O. Alterations in fecal microbiota composition by probiotic supplementation in healthy adults: A systematic review of randomized controlled trials. Genome Med. 2016, 8, 52. [Google Scholar] [CrossRef]
- Turroni, F.; Foroni, E.; Pizzetti, P.; Giubellini, V.; Ribbera, A.; Merusi, P.; Cagnasso, P.; Bizzarri, B.; de’Angelis, G.L.; Shanahan, F.; et al. Exploring the diversity of the bifidobacterial population in the human intestinal tract. Appl. Environ. Microbiol. 2009, 75, 1534–1545. [Google Scholar] [CrossRef]
- Ventura, M.; Turroni, F.; van Sinderen, D. Bifidobacteria: General Overview on Ecology, Taxonomy, and Genomics. In Lactic Acid Bacteria. Microbiological and Functional Aspects, 4th ed.; Lahtinen, S., Ouwehand, A.C., Salminen, S., Von wright, A., Eds.; CRC Press: New York, NY, USA, 2012; p. 149. [Google Scholar]
- Moens, F.; Verce, M.; De Vuyst, L. Lactate- and acetate-based cross-feeding interactions between selected strains of lactobacilli, bifidobacteria and colon bacteria in the presence of inulin-type fructans. Int. J. Food Microbiol. 2017, 241, 225–236. [Google Scholar] [CrossRef]
- Mack, D.R.; Michail, S.; Wei, S.; McDougall, L.; Hollingsworth, M.A. Probiotics inhibit enteropathogenic E. coli adherence in vitro by inducing intestinal mucin gene expression. Am. J. Physiol. 1999, 276, G941–G950. [Google Scholar]
- Turroni, F.; Bottacini, F.; Foroni, E.; Mulder, I.; Kim, J.H.; Zomer, A.; Sánchez, B.; Bidossi, A.; Ferrarini, A.; Giubellini, V.; et al. Genome analysis of Bifidobacterium bifidum PRL2010 reveals metabolic pathways for host-derived glycan foraging. Proc. Natl. Acad. Sci. USA 2010, 107, 19514–19519. [Google Scholar] [CrossRef]
- Marteau, P.; Cuillerier, E.; Meance, S.; Gerhardt, M.F.; Myara, A.; Bouvier, M.; Bouley, C.; Tondu, F.; Bommelaer, G.; Grimaud, J.C. Bifidobacterium animalis strain DN-173 010 shortens the colonic transit time in healthy women: A double-blind, randomized, controlled study. Aliment. Pharmacol. Ther. 2002, 16, 587–593. [Google Scholar] [CrossRef]
- Meng, H.; Ba, Z.; Lee, Y.; Peng, J.; Lin, J.; Fleming, J.A.; Furumoto, E.J.; Roberts, R.F.; Kris-Etherton, P.M.; Rogers, C.J. Consumption of Bifidobacterium animalis subsp. lactis BB-12 in yogurt reduced expression of TLR-2 on peripheral blood-derived monocytes and pro-inflammatory cytokine secretion in young adults. Eur. J. Nutr. 2017, 56, 649–661. [Google Scholar] [CrossRef]
- Lugli, G.A.; Milani, C.; Turroni, F.; Duranti, S.; Ferrario, C.; Viappiani, A.; Mancabelli, L.; Mangifesta, M.; Taminiau, B.; Delcenserie, V.; et al. Investigation of the evolutionary development of the genus Bifidobacterium by comparative genomics. Appl. Environ. Microbiol. 2014, 80, 6383–6394. [Google Scholar] [CrossRef]
- Schirmer, M.; Ijaz, U.Z.; D’Amore, R.; Hall, N.; Sloan, W.T.; Quince, C. Insight into biases and sequencing errors for amplicon sequencing with the Illumina MiSeq platform. Nucleic Acids Res. 2015, 43, e37. [Google Scholar] [CrossRef]
- Rizkalla, S.W.; Luo, J.; Kabir, M.; Chevalier, A.; Pacher, N.; Slama, G. Chronic consumption of fresh but not heated yogurt improves breath hydrogen status and short-chain fatty acid profiles: A controlled study in healthy men with or without lactose maldigestion. Am. J. Clin. Nutr. 2000, 72, 1474–1479. [Google Scholar] [CrossRef]
- Guarner, F.; Perdigon, G.; Corthier, G.; Salminen, S.; Koletzko, B.; Morelli, L. Should yoghurt cultures be considered probiotic? Br. J. Nutr. 2005, 93, 783–786. [Google Scholar] [CrossRef]
- Plaza-Díaz, J.; Fernández-Caballero, J.Á.; Chueca, N.; García, F.; Gómez-Llorente, C.; Sáez-Lara, M.J.; Fontana, L.; Gil, Á. Pyrosequencing analysis reveals changes in intestinal microbiota of healthy adults who received a daily dose of immunomodulatory probiotic strains. Nutrients 2015, 7, 3999–4015. [Google Scholar] [CrossRef]
- Stolaki, M.; De Vos, W.M.; Kleerebezem, M.; Zoetendal, E.G. Lactic Acid Bacteria in the gut. In Lactic Acid Bacteria. Microbiological and Functional Aspects, 4th ed.; Lahtinen, S., Ouwehand, A.C., Salminen, S., Von wright, A., Eds.; CRC Press: New York, NY, USA, 2012; pp. 385–390. [Google Scholar]
- Lahti, L.; Salonen, A.; Kekkonen, R.A.; Salojärvi, J.; Jalanka-Tuovinen, J.; Palva, A.; Orešič, M.; de Vos, W.M. Associations between the human intestinal microbiota, Lactobacillus rhamnosus GG and serum lipids indicated by integrated analysis of high-throughput profiling data. PeerJ 2013, 1, e32. [Google Scholar] [CrossRef]
- Ferrario, C.; Taverniti, V.; Milani, C.; Fiore, W.; Laureati, M.; De Noni, I.; Stuknyte, M.; Chouaia, B.; Riso, P.; Guglielmetti, S. Modulation of fecal Clostridiales bacteria and butyrate by probiotic intervention with Lactobacillus paracasei DG varies among healthy adults. J. Nutr. 2014, 144, 1787–1796. [Google Scholar] [CrossRef]
- Koeth, R.A.; Wang, Z.; Levison, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L.; et al. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 2013, 19, 576–585. [Google Scholar] [CrossRef]
- Fan, Y.; Ya, E.Z.; Ji-Dong, W.; Yu-Fan, L.; Ying, Z.; Ya-Lun, S.; Meng-Yu, M.; Rui-Ling, Z. Comparison of Microbial Diversity and Composition in Jejunum and Colon of the Alcohol-dependent Rats. J. Microbiol. Biotechnol. 2018, 28, 1883–1895. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Claesson, M.J.; O’Sullivan, O.; Wang, Q.; Nikkilä, J.; Marchesi, J.R.; Smidt, H.; de Vos, W.M.; Ross, R.P.; O’Toole, P.W. Comparative analysis of pyrosequencing and a phylogenetic microarray for exploring microbial community structures in the human distal intestine. PLoS ONE 2009, 4, e6669. [Google Scholar] [CrossRef]
- Pokusaeva, K.; Fitzgerald, G.F.; van Sinderen, D. Carbohydrate metabolism in Bifidobacteria. Genes Nutr. 2011, 6, 285–306. [Google Scholar] [CrossRef]
- Haro, C.; Rangel-Zúñiga, O.A.; Alcalá-Díaz, J.F.; Gómez-Delgado, F.; Pérez-Martínez, P.; Delgado-Lista, J.; Quintana-Navarro, G.M.; Landa, B.B.; Navas-Cortés, J.A.; Tena-Sempere, M.; et al. Intestinal microbiota is influenced by gender and body mass index. PLoS ONE 2016, 11, e0154090. [Google Scholar] [CrossRef]
- Fransen, F.; van Beek, A.A.; Borghuis, T.; Meijer, B.; Hugenholtz, F.; van der Gaast-de Jongh, C.; Savelkoul, H.F.; de Jonge, M.I.; Faas, M.M.; Boekschoten, M.V.; et al. The impact of gut microbiota on gender-specific differences in immunity. Front. Immunol. 2017, 8, 754. [Google Scholar] [CrossRef]
- Wahlström, A.; Sayin, S.I.; Marschall, H.U.; Bäckhed, F. Intestinal crosstalk between bile acids and microbiota and its impact on host metabolism. Cell Metab. 2016, 24, 41–50. [Google Scholar] [CrossRef]
- Hyland, N.P.; Cryan, J.F. Microbe-host interactions: Influence of the gut microbiota on the enteric nervous system. Dev. Biol. 2016, 417, 182–187. [Google Scholar] [CrossRef]
| Nonconsumers | PFM Consumers | ||||
|---|---|---|---|---|---|
| Men | n = 101 | n = 33 | p * | p# | p† |
| Age (years) | 37.31 (5.895) | 35.84 (5.957) | – | – | – |
| Energy (kcal/day) | 2204 (547.4) | 2268 (619.6) | – | 0.779 | – |
| Carbohydrates (%) | 40.3 (5.810) | 39.53 (5.570) | 0.371 | – | – |
| Proteins (%) | 17.44 (2.661) | 17.89 (3.119) | 0.370 | – | – |
| Fat (%) | 39.37 (5.068) | 39.56 (4.26) | 0.759 | – | – |
| Fiber (g/day) Ʇ | 23.73 (10.15) | 22.55 (6.520) | 0.361 | – | – |
| Sleep hours | 7.285 (0.827) | 7.355 (0.755) | – | 0.820 | – |
| BMI (kg/m2) | 25.91 (3.201) | 25.43 (3.570) | – | 0.733 | – |
| Body fat (%) | 19.55 (5.616) | 18.10 (5.914) | – | 0.373 | |
| Waist circumference (cm) | 88.12 (8.838) | 86.80 (8.451) | – | 0.815 | – |
| Visceral fat index | 6.0 (4.0–9.0) | 6.0 (4.0–7.5) | – | – | 0.284 |
| Total score of intestinal symptoms | 7.0 (4.0–11.0) | 9.0 (6.0–11.0) | – | – | 0.345 |
| Smoking habits (%) | |||||
| Current smokers | 77.8 | 22.2 | – | – | |
| Former smokers | 83.3 | 16.7 | – | – | 0.634 |
| Nonsmokers | 73.0 | 27.0 | – | – | |
| Income (%) | |||||
| Low (<50,000 euros/year) | 34.7 | 30.3 | – | – | |
| Medium (50,000–200,000 euros/year) | 45.5 | 45.5 | – | – | 0.807 |
| High (>200,000 euros/year) | 19.8 | 24.2 | – | – | |
| Women | n = 74 | n = 52 | |||
| Age (years) | 34.73 (6.623) | 34.54 (6.592) | – | – | – |
| Energy (kcal/day) | 2018 (470.9) | 1992 (506.3) | – | 0.721 | – |
| Carbohydrates (%) | 39.82 (7.853) | 40.77 (6.750) | 0.946 | – | – |
| Proteins (%) | 16.90 (2.941) | 16.89 (2.618) | 0.802 | – | – |
| Fat (%) | 41.01 (6.457) | 40.26 (5.747) | 0.913 | – | – |
| Fiber (g/day) Ʇ | 23.03 (10.64) | 22.58 (8.845) | 0.623 | – | – |
| Sleep hours | 7.517 (0.841) | 7.566 (0.624) | – | 0.737 | – |
| BMI (kg/m2) | 23.41 (3.312) | 22.02 (2.279) | – | 0.009 | – |
| Body fat (%) | 28.85 (7.261) | 25.04 (4.770) | – | 0.001 | – |
| Waist circumference (cm) | 74.87 (8.442) | 72.44 (5.475) | – | 0.062 | |
| Visceral fat index | 3.0 (2.0–4.13) | 2.5 (1.0–3.0) | – | – | 0.032 |
| Total score of intestinal symptoms | 11.0 (7.0–15.0) | 12.0 (6.0–16.0) | – | – | 0.546 |
| Smoking habits (%) | |||||
| Current smokers | 66.7 | 33.3 | – | – | |
| Former smokers | 45.5 | 54.5 | – | – | 0.194 |
| Nonsmokers | 61.9 | 38.1 | – | – | |
| Income (%) | |||||
| Low (<50,000 euros/year) | 64.9 | 55.8 | – | – | |
| Medium (50,000–200,000 euros/year) | 32.4 | 32.7 | – | – | 0.120 |
| High (>200,000 euros/year) | 2.70 | 11.5 | – | – | |
| PFM Groups | |||
|---|---|---|---|
| % Relative Abundance | Nonconsumers (n = 175) | Consumers (n = 85) | p Value * |
| Actinobacteria # | 2.945 (2.984) | 4.089 (4.090) | 0.011 |
| Bifidobacteriaceae# | 2.368 (2.885) | 3.358 (3.794) | 0.012 |
| Bifidobacterium# | 2.399 (2.869) | 3.307 (3.773) | 0.027 |
| PFM Groups | |||||||
|---|---|---|---|---|---|---|---|
| Nonconsumers (n = 175) | Consumers (n = 85) | ||||||
| % Relative Abundance | Occurrence (n, %) | Median (IQR) | Occurrence (n, %) | Median (IQR) | p Value † Occurrence | p Value * Abundance | FDR Critical Value |
| Bifidobacterium animalis | 41 (23.4) | 0.000 (0.000–0.000) | 49 (66.2) | 0.011 (0.000–0.187) | <0.001 | <0.001 | 0.027 |
| Bifidobacterium pseudolongum | 29 (16.6) | 0.000 (0.000–0.000) | 48 (64.2) | 0.001 (0.000–0.007) | <0.001 | <0.001 | 0.009 |
| Bifidobacterium thermophilum | 15 (8.6) | 0.000 (0.000–0.000) | 36 (50.7) | 0.000 (0.000–0.007) | <0.001 | <0.001 | 0.018 |
| Bifidobacterium merycicum | 102 (58.3) | 0.001 (0.000–0.002) | 66 (77.5) | 0.005 (0.001–0.030) | 0.002 | <0.001 | 0.036 |
| Bifidobacterium kashiwanohense | 160 (91.4) | 0.012 (0.002–0.043) | 83 (97.6) | 0.017 (0.005–0.061) | 0.057 | 0.016 | 0.045 |
| Bifidobacterium dentium | 71 (40.6) | 0.000 (0.000–0.001) | 44 (52.1) | 0.001 (0.000–0.001) | 0.088 | 0.045 | 0.054 |
| Bifidobacterium longum | 170 (97.1) | 0.175 (0.043–0.356) | 79 (92.9) | 0.176 (0.050–0.658) | 0.114 | 0.050 | 0.063 |
| Bifidobacterium bombi | 170 (97.1) | 0.009 (0.003–0.018) | 84 (98.8) | 0.012 (0.004–0.022) | 0.397 | 0.059 | 0.072 |
| Bifidobacterium magnum# | |||||||
| Men (n = 134: 101 NC/33 C) | 55 (54.5) | 0.001 (0.000–0.001) | 26 (78.8) | 0.001 (0.001–0.007) | 0.013 | <0.001 | 0.010 |
| Women (n = 126: 74 NC/52 C) | 49 (66.2) | 0.001 (0.000–0.002) | 42 (80.8) | 0.002 (0.001–0.007) | 0.073 | <0.001 | 0.005 |
| PFM Groups | |||||
|---|---|---|---|---|---|
| % Relative Abundance | PFM-NC n = 175 | Bif-PFM n = 33 | Lb-PFM n = 14 | Mixed-PFM n = 38 | p Value * |
| Bifidobacterium animalis | 0.000 a (0.000–0.000) | 0.018 b (0.001–0.241) | 0.000 a (0.000–0.000) | 0.002 b (0.000–0.157) | <0.001 |
| Bifidobacterium pseudolongum | 0.000 a (0.000–0.000) | 0.001 b (0.000–0.012) | 0.000 a (0.000–0.000) | 0.001 b (0.000–0.006) | <0.001 |
| Bifidobacterium thermophilum | 0.000 a (0.000–0.000) | 0.001 b (0.000–0.011) | 0.000 a (0.000–0.000) | 0.000 b (0.000–0.006) | <0.001 |
| Bifidobacterium merycicum | 0.001 a (0.000–0.002) | 0.005 b (0.001–0.034) | 0.001 ab (0.001–0.425) | 0.004 b (0.001–0.028) | <0.001 |
| Bifidobacterium kashiwanohense | 0.012 a (0.003–0.043) | 0.018 ab (0.006–0.049) | 0.067 b (0.029–0.143) | 0.016 ab (0.004–0.069) | 0.009 |
| Bifidobacterium dentium | 0.000 (0.000–0.001) | 0.001 (0.000–0.004) | 0.000 (0.000–0.003) | 0.001 (0.000–0.001) | 0.184 |
| Bifidobacterium longum | 0.175 (0.043–0.357) | 0.194 (0.042–0.982) | 0.467 (0.141–1.300) | 0.169 (0.069–0.515) | 0.071 |
| Bifidobacterium bombi | 0.009 (0.003–0.018) | 0.011 (0.005–0.027) | 0.016 (0.008–0.036) | 0.012 (0.004–0.018) | 0.213 |
| Bifidobacterium magnum# | |||||
| Men | 0.001 a (0.000–0.001) | 0.002 b (0.001–0.011) | 0.001 ab (0.000–0.001) | 0.003 b (0.001–0.007) | <0.001 |
| Women | 0.001 a (0.000–0.002) | 0.002 b (0.001–0.009) | 0.001 ab (0.001–0.003) | 0.004 b (0.000–0.009) | 0.004 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Redondo-Useros, N.; Gheorghe, A.; Díaz-Prieto, L.E.; Villavisencio, B.; Marcos, A.; Nova, E. Associations of Probiotic Fermented Milk (PFM) and Yogurt Consumption with Bifidobacterium and Lactobacillus Components of the Gut Microbiota in Healthy Adults. Nutrients 2019, 11, 651. https://doi.org/10.3390/nu11030651
Redondo-Useros N, Gheorghe A, Díaz-Prieto LE, Villavisencio B, Marcos A, Nova E. Associations of Probiotic Fermented Milk (PFM) and Yogurt Consumption with Bifidobacterium and Lactobacillus Components of the Gut Microbiota in Healthy Adults. Nutrients. 2019; 11(3):651. https://doi.org/10.3390/nu11030651
Chicago/Turabian StyleRedondo-Useros, Noemí, Alina Gheorghe, Ligia E. Díaz-Prieto, Brenda Villavisencio, Ascensión Marcos, and Esther Nova. 2019. "Associations of Probiotic Fermented Milk (PFM) and Yogurt Consumption with Bifidobacterium and Lactobacillus Components of the Gut Microbiota in Healthy Adults" Nutrients 11, no. 3: 651. https://doi.org/10.3390/nu11030651
APA StyleRedondo-Useros, N., Gheorghe, A., Díaz-Prieto, L. E., Villavisencio, B., Marcos, A., & Nova, E. (2019). Associations of Probiotic Fermented Milk (PFM) and Yogurt Consumption with Bifidobacterium and Lactobacillus Components of the Gut Microbiota in Healthy Adults. Nutrients, 11(3), 651. https://doi.org/10.3390/nu11030651






