β-Cryptoxanthin Reduces Body Fat and Increases Oxidative Stress Response in Caenorhabditis elegans Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Carotenoid Standards and Extracts
2.2. Quantification of Individual Carotenoids in Extracts
2.3. C. elegans Strain and Treatments
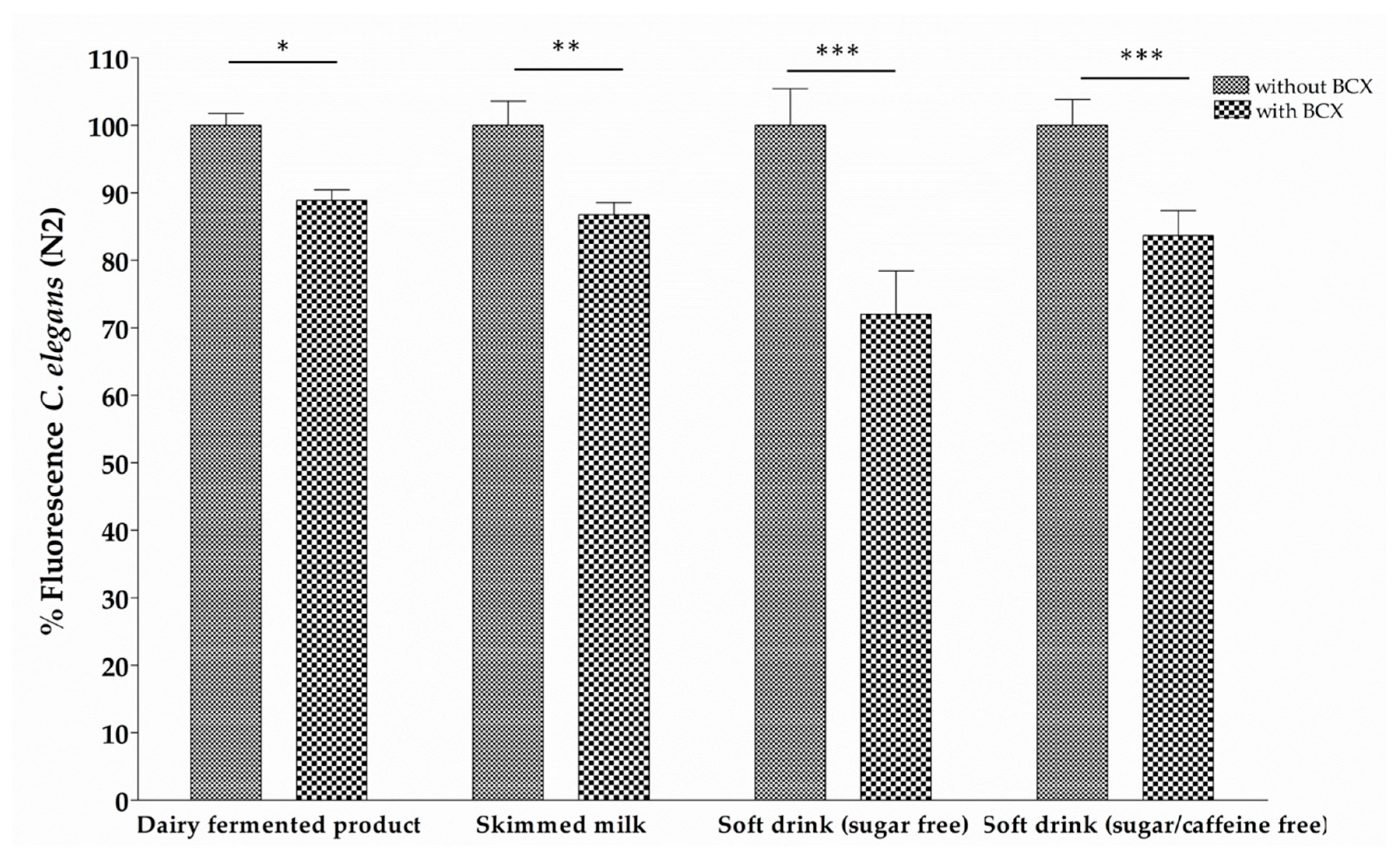
2.4. Bioassimilation Assay in C. elegans
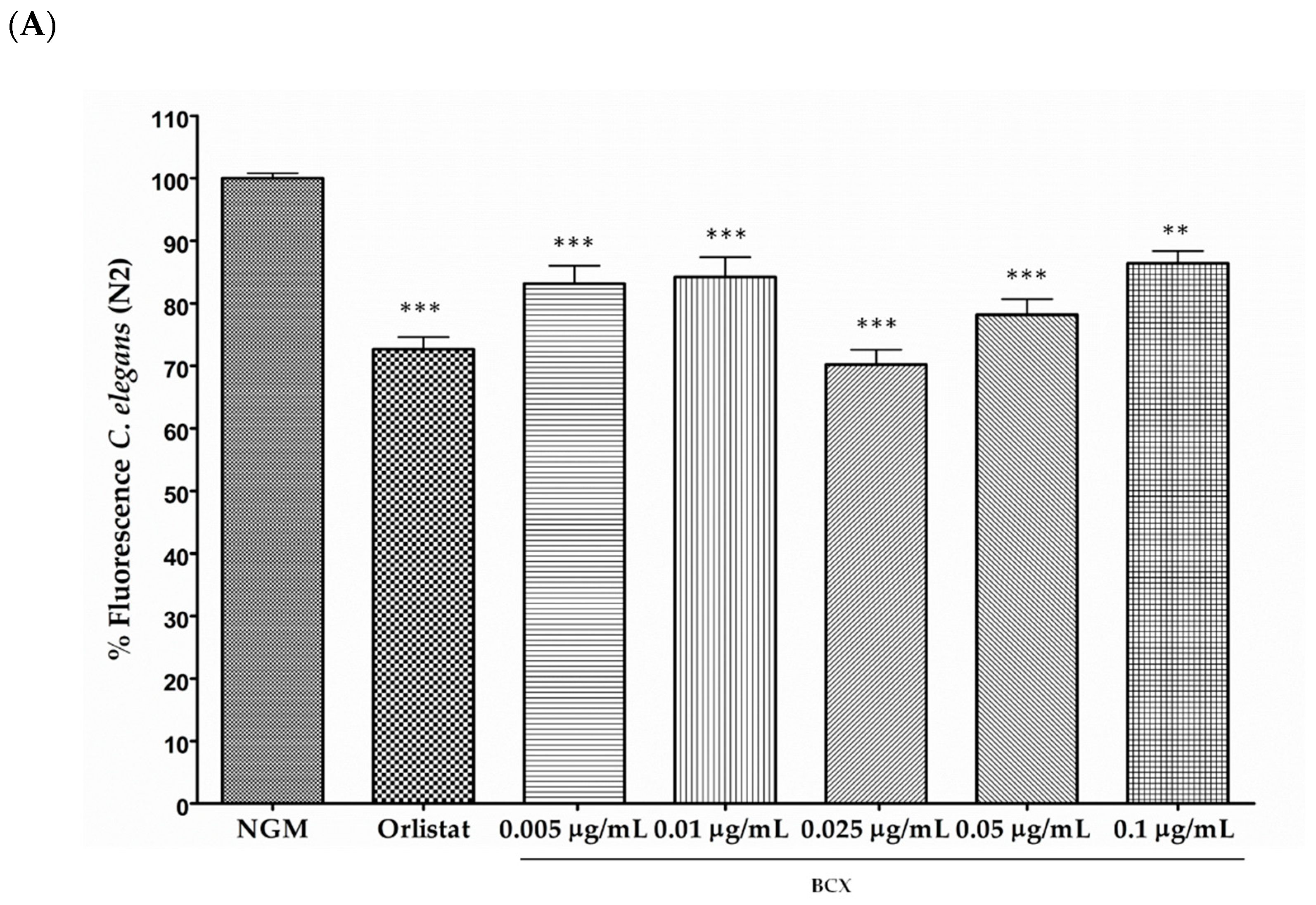
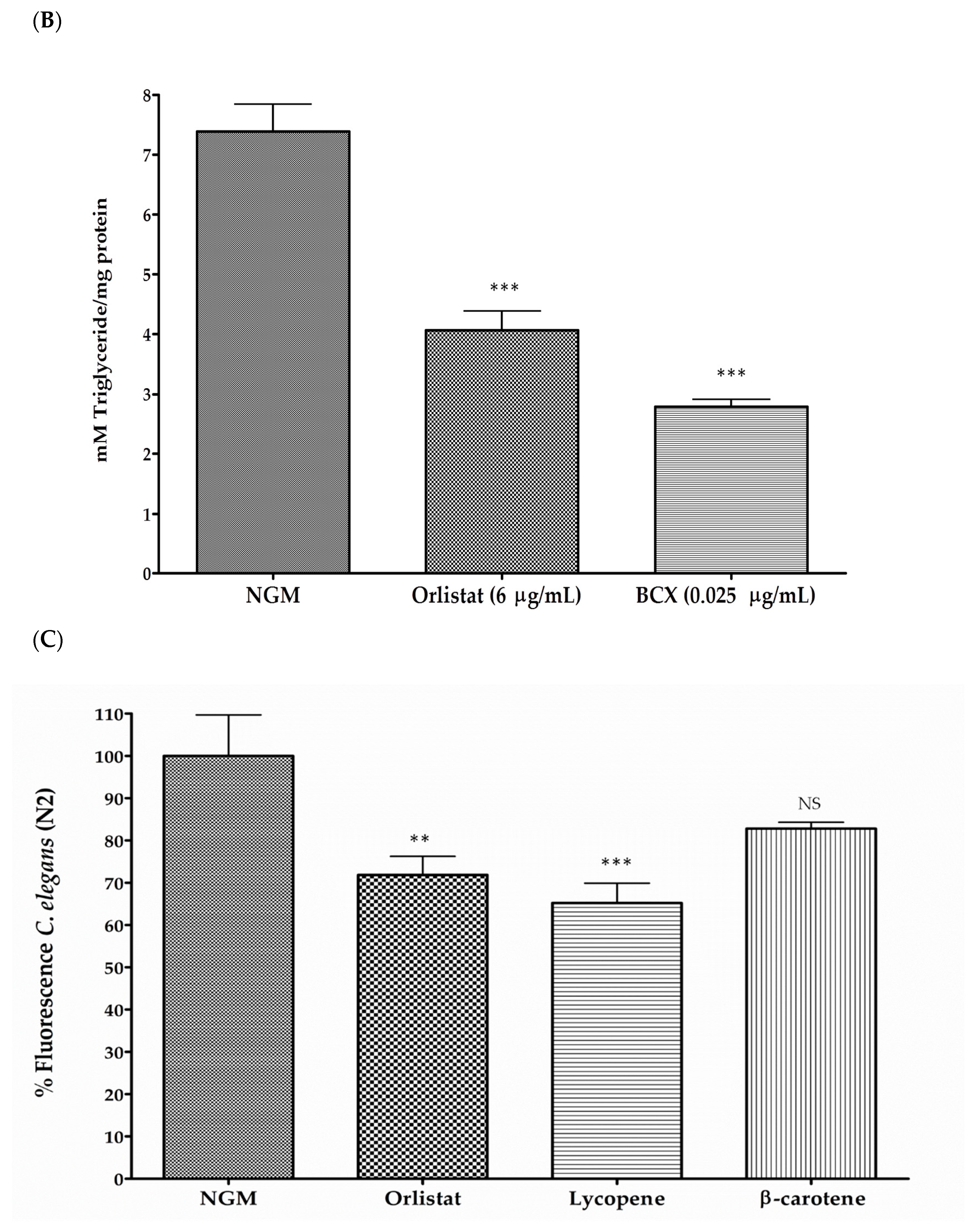
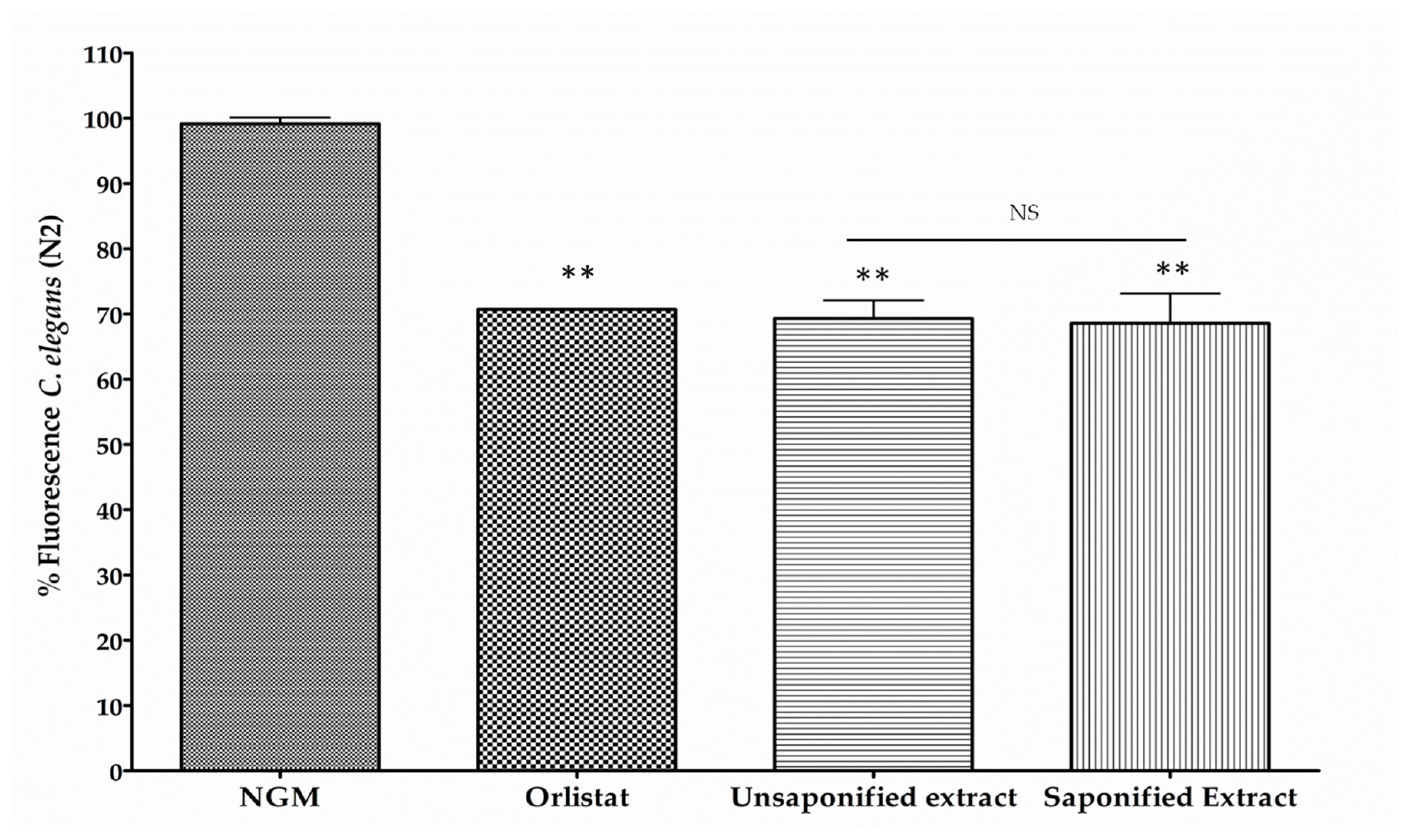
2.5. Body Fat Reduction Assays in C. elegans
2.6. Oxidative Stress Assays in C. elegans
2.7. Transcriptomic Analysis in C. elegans
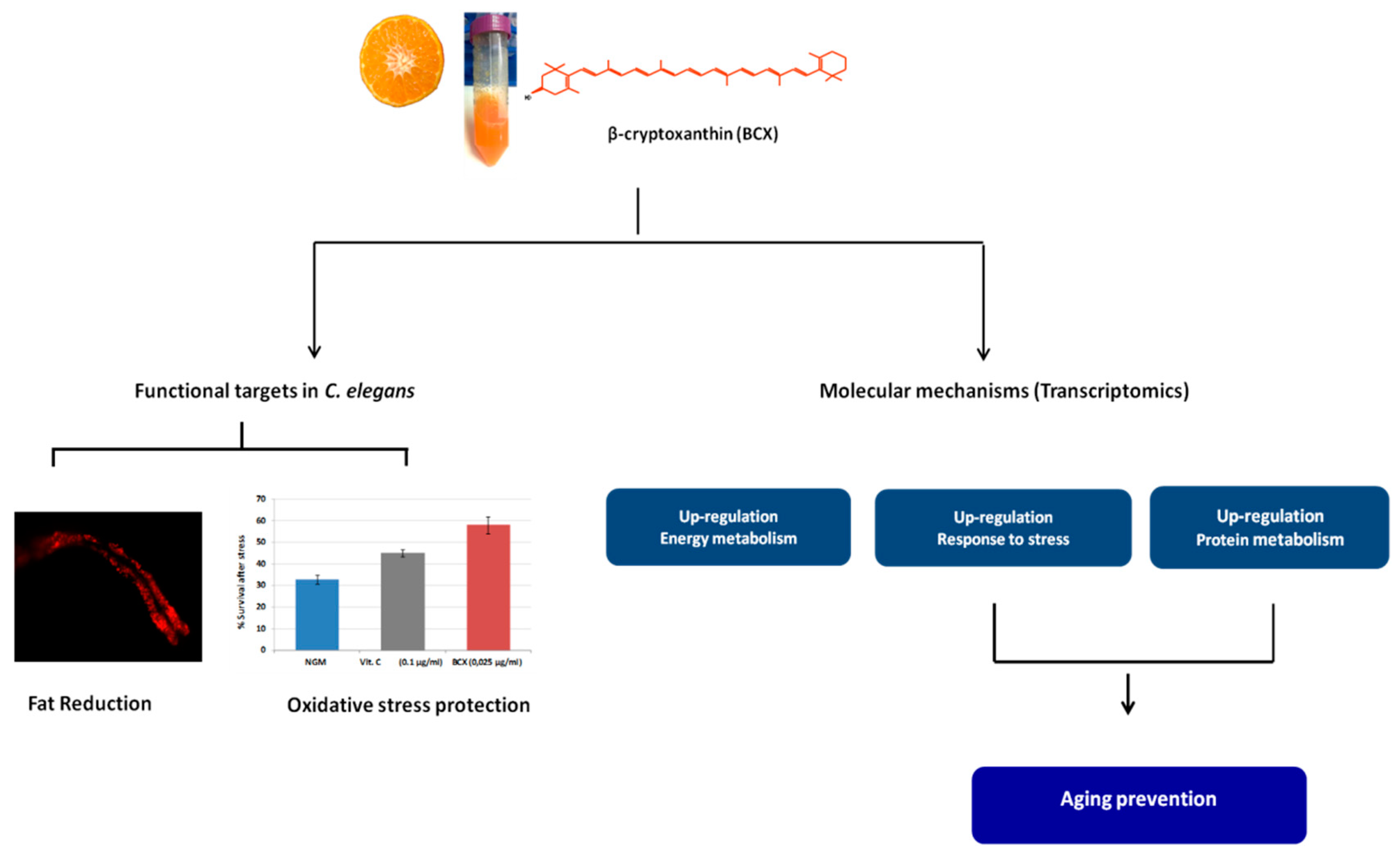
3. Results
3.1. β-Cryptoxanthin Reduces Fat Content in C. elegans N2 in a Dose-Dependent Manner
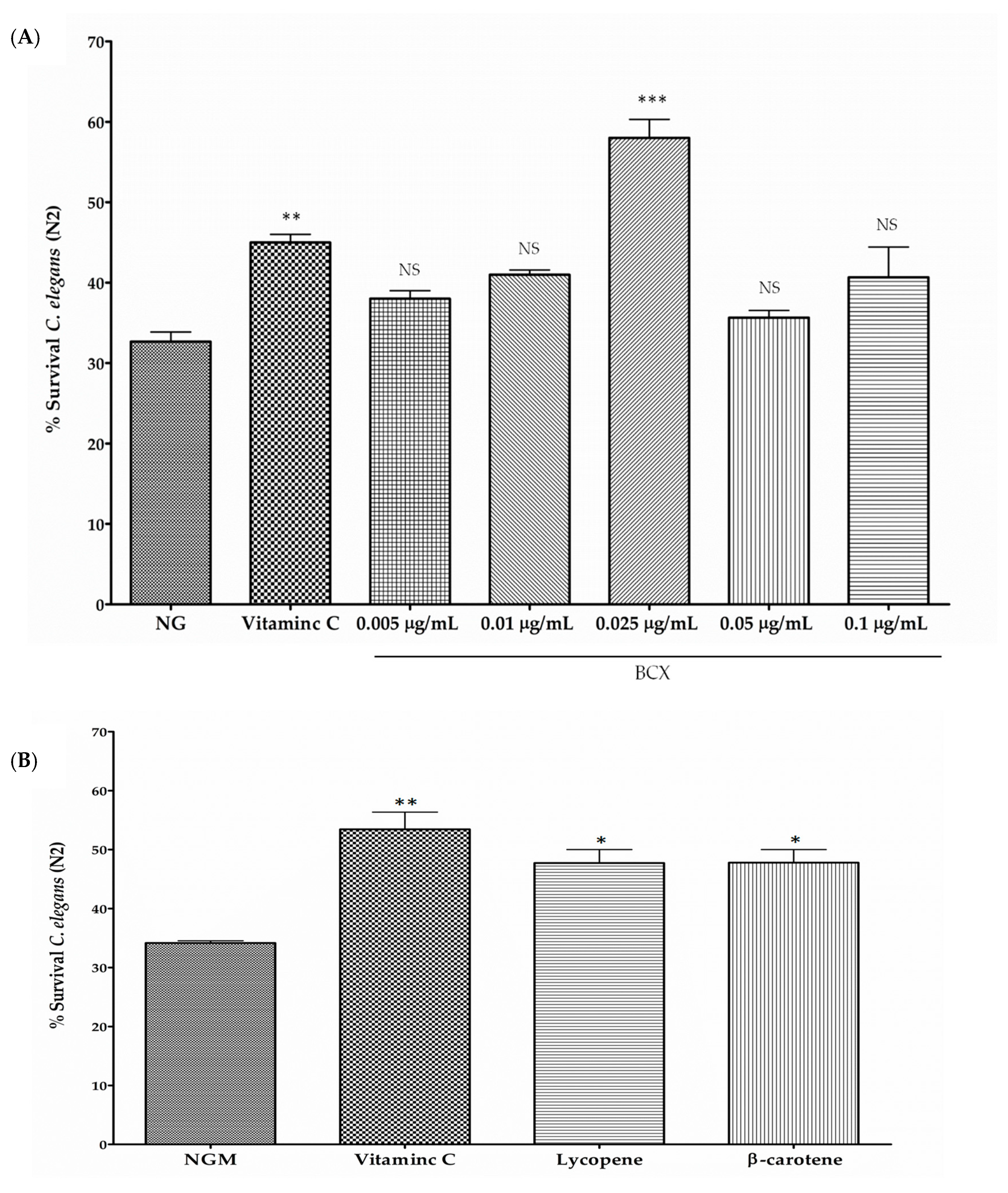
3.2. BCX has Antioxidant Activity in C. elegans N2
3.3. BCX Modulates Energy Metabolism, Antioxidant Response, and Protein Homeostasis in C. elegans
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Rodriguez-Concepcion, M.; Avalos, J.; Bonet, M.L.; Boronat, A.; Gomez-Gomez, L.; Hornero-Mendez, D.; Limon, M.C.; Meléndez-Martínez, A.J.; Olmedilla-Alonso, B.; Palou, A.; et al. A global perspective on carotenoids: Metabolism, biotechnology and benefits for nutrition and health. Prog. Lipid Res. 2018, 70, 62–93. [Google Scholar] [CrossRef] [PubMed]
- Nisar, N.; Li, L.; Lu, S.; Khin, N.C.; Pogson, B.J. Carotenoid metabolism in plants. Mol. Plant 2015, 8, 68–82. [Google Scholar] [CrossRef] [PubMed]
- Burri, B.J. BCX as a source of vitamin A. J. Sci. Food Agric. 2015, 95, 1786–1794. [Google Scholar] [CrossRef] [PubMed]
- Beltrán-de-Miguel, B.; Estévez-Santiago, R.; Olmedilla-Alonso, B. Assessment of dietary vitamin A intake (retinol, α-carotene, β-carotene, β-cryptoxanthin) and its sources in the National Survey of Dietary Intake in Spain (2009–2010). Int. J. Food Sci. Nutr. 2015, 66, 706–712. [Google Scholar] [CrossRef]
- Meléndez-Martínez, A.J.; Mapelli-Brahm, P.; Benítez-González, A.; Stinco, C.M. A comprehensive review on the colorless carotenoids phytoene and phytofluene. Arch. Biochem. Biophys. 2015, 572, 188–200. [Google Scholar] [CrossRef] [PubMed]
- Burri, B.J.; La Frano, M.R.; Zhu, C. Absorption, metabolism, and functions of β-cryptoxanthin. Nutr. Rev. 2016, 74, 69–82. [Google Scholar] [CrossRef] [PubMed]
- Estévez-Santiago, R.; Olmedilla-Alonso, B.; Fernández-Jalao, I. Bioaccessibility of provitamin A carotenoids from fruits: Application of a standardised static in vitro digestion method. Food Funct. 2016, 7, 1354–1366. [Google Scholar] [CrossRef] [PubMed]
- Breithaupt, D.E.; Bamedi, A. Carotenoid esters in vegetables and fruits: A screening with emphasis on beta-cryptoxanthinesters. J. Agric. Food Chem. 2001, 49, 2064–2070. [Google Scholar] [CrossRef] [PubMed]
- Granado, F.; Olmedilla, B.; Blanco, I.; Rojas-Hidalgo, E. Major fruit and vegetable contributors to the main serum carotenoids in the Spanish diet. Eur. J. Clin. Nutr. 1996, 50, 246–250. [Google Scholar] [PubMed]
- Sugiura, M.; Kato, M.; Matsumoto, H.; Nagao, A.; Yano, M. Serum concentration of β-Cryptoxanthin in Japan reflects the frequency of Satsuma mandarin (Citrus unshiu Marc.) consumption. J. Health Sci. 2002, 48, 350–353. [Google Scholar] [CrossRef]
- Mercadante, A.Z.; Rodrigues, D.B.; Petry, F.C.; Mariutti, L.R.B. Carotenoid esters in foods—A review and practical directions on analysis and occurrence. Food Res. Int. 2017, 99, 830–850. [Google Scholar] [CrossRef] [PubMed]
- Ma, G.; Zhang, L.; Iida, K.; Madono, Y.; Yungyuen, W.; Yahata, M.; Yamawaki, K.; Kato, M. Identification and quantitative analysis of β-cryptoxanthin and β-citraurin esters in Satsuma mandarin fruit during the ripening process. Food Chem. 2017, 234, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Burri, B.J.; Chang, J.S.; Neidlinger, T.R. β-Cryptoxanthin and α-carotene-rich foods have greater apparent bioavailability than β-carotene-rich foods in Western diets. Br. J. Nutr. 2011, 105, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Stahl, W.; Sies, H. Bioactivity and protective effects of natural carotenoids. Biochim. Biophys. Acta 2005, 1740, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Katsuura, S.; Imamura, T.; Bando, N.; Yamanishi, R. β-carotene and BCX but not lutein evoke redox and immune changes in RAW264 murine macrophages. Mol. Nutr. Food Res. 2009, 53, 1396–1405. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Sugiura, M.; Ogawa, K.; Takabayashi, F.; Toda, M.; Sakuma, M.; Maeda, K.; Fujitani, K.; Miyazaki, H.; Yamamoto, H.; et al. Beta-cryptoxanthin, plentiful in Japanese mandarin orange, prevents age-related cognitive dysfunction and oxidative damage in senescence-accelerated mouse brain. Biol. Pharm. Bull. 2011, 34, 311–317. [Google Scholar] [CrossRef]
- Iribarren, C.; Folsom, A.R.; Jacobs, D.R.; Gross, M.D.; Belcher, J.D.; Eckfeldt, J.H. Association of serum vitamin levels, LDL susceptibility to oxidation and autoantibodies aginst MDA-LDL with carotid atherosclerosis: A case-control study. Artheroscler. Thromb. Vasc. Biol. 1997, 17, 1171–1177. [Google Scholar] [CrossRef]
- Dwyer, J.H.; Paul-Labrador, M.J.; Fan, J.; Shircore, A.M.; Merz, C.N.; Dwyer, K.M. Progression of carotid-intima media thickness and plasma antioxidants: The Los Angeles Atherosclerosis Study. Artheroscler. Thromb. Vasc. Biol. 2004, 24, 231–239. [Google Scholar] [CrossRef]
- Nakamura, M.; Sugiura, M.; Aoki, N. High β-carotene and BCX are associated with low pulse wave velocity. Atherosclerosis 2006, 184, 363–369. [Google Scholar] [CrossRef]
- Gammone, M.A. Carotenoids, ROS, and cardiovascular health. In Reactive Oxyen Species in Biology and Human Health; Ahmad, S.I., Ed.; CRC Press: Boca Raton, FL, USA, 2016; pp. 325–331. ISBN 9781498735452. [Google Scholar]
- Miyazawa, K.; Miyamoto, S.; Suzuki, R.; Yasuu, Y.; Ikeda, R.; Kohno, H.; Yano, M.; Tanaka, T.; Hata, K.; Suzuki, K. Dietary beta-cryptoxanthin inhibits N-butyl-N-(4-hydroxybutyl)nitrosamine-induced urinary bladder carcinogenesis in male ICR mice. Oncol. Rep. 2007, 17, 297–304. [Google Scholar] [CrossRef]
- Tanaka, T.; Shnimizu, M.; Moriwaki, H. Cancer chemoprevention by carotenoids. Molecules 2012, 17, 3202–3242. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M. Role of carotenoid β-cryptoxanthin in bone homeostasis. J. Biomed. Sci. 2012, 2, 19–36. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Weitzmann, M.N. The bone anabolic carotenoid beta-cryptoxanthin enhances transforming growth factor-beta1-induced SMAD activation in MC3T3 preosteoblasts. Int. J. Mol. Med. 2009, 24, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, M.; Nakamura, M.; Ogawa, K.; Ikoma, Y.; Ando, F.; Shimokata, H.; Yano, M. Dietary patterns of antioxidant vitamin and carotenoid intake associated with bone mineral density: Findings from post-menopausal Japanese female subjects. Osteoporos. Int. 2011, 22, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Kabagambe, E.K.; Furtado, J.; Baylin, A.; Campos, H. Some dietary and adipose tissue carotenoids are associated with the risk of nonfatal acute myocardial infarction in Costa Rica. J. Nutr. 2005, 135, 1763–1769. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.F.; Jacobs, D.R., Jr.; Gross, M.D.; Schreiner, P.J.; Williams, O.D.; Lee, D.H. Longitudinal associations between body mass index and serum carotenoids: The CARDIA study. Br. J. Nutr. 2006, 95, 358–365. [Google Scholar] [CrossRef]
- Iwamoto, M.; Imai, K.; Ohta, H.; Shirouchi, B.; Sato, M. Supplementation of highly concentrated BCX in a satsuma mandarin beverage improves adipocytokine profiles in obese Japanese women. Lipids Health Dis. 2012, 11, 52–55. [Google Scholar] [CrossRef]
- Tsuchida, T.; Minagawa, K.; Mukai, K.; Mizuno, Y.; Mashiko, K.; Minagawa, K. The comparative study of BCX derived from Satsuma mandarin for fat of human body. Jpn. Pharmacol. Ther. 2008, 36, 247–253. [Google Scholar]
- Iwata, A.; Matsubara, S.; Miyazaki, K. Beneficial effects of a beta-cryptoxanthin-containing beverage on body mass index and visceral fat in pre-obese men: Double-blind, placebo-controlled parallel trials. J. Funct. Foods 2018, 41, 250–257. [Google Scholar] [CrossRef]
- Takayanagi, K.; Morimoto, S.; Shirakura, Y.; Mukai, K.; Sugiyama, T.; Tokuji, Y.; Ohnishi, M. Mechanism of visceral fat reduction in Tsumura Suzuki obese, diabetes (TSOD) mice orally administered BCX from Satsuma mandarin oranges (Citrus unshiu Marc). J. Agric. Food Chem. 2011, 59, 12342–12351. [Google Scholar] [CrossRef]
- Sugiura, M.; Nakamura, M.; Ogawa, K.; Ikoma, Y.; Matsumoto, H.; Ando, F.; Shimokata, H.; Yano, M. Associations of serum carotenoid concentrations with the metabolic syndrome: Interaction with smoking. Br. J. Nutr. 2008, 100, 1297–1306. [Google Scholar] [CrossRef]
- Ni, Y.; Nagashimada, M.; Zhan, L.; Nagata, N.; Kobori, M.; Sugiura, M.; Ogawa, K.; Kaneko, S.; Ota, T. Prevention and reversal of lipotoxicity-induced hepatic insulin resistance and steatohepatitis in mice by an antioxidant carotenoid, BCX. Endocrinology 2015, 156, 987–999. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, M.; Nakamura, M.; Ogawa, K.; Ikoma, Y.; Yano, M. High serum carotenoids associated with lower risk for the metabolic syndrome and its components among Japanese subjects: Mikkabi cohort study. Br. J. Nutr. 2015, 114, 1674–1682. [Google Scholar] [CrossRef] [PubMed]
- Shirakura, Y.; Takayanagi, K.; Mukai, K.; Tanabe, H.; Inoue, M. BCX suppresses the adipogenesis of 3T3-L1 cells via RAR activation. J. Nutr. Sci. Vitaminol. 2011, 57, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Ashrafi, K.; Chang, F.Y.; Watts, J.L.; Fraser, A.G.; Kamath, R.S.; Ahringer, J.; Ruvkun, G. Genome wide RNAi analysis of Caenorhabditis elegans fat regulatory genes. Nature 2003, 421, 268–272. [Google Scholar] [CrossRef]
- Jones, K.T.; Ashrafi, K. Caenorhabditis elegans as an emerging model for studying the basic biology of obesity. Dis. Models Mech. 2009, 2, 224–229. [Google Scholar] [CrossRef]
- Zheng, J.; Greenway, F.L. Caenorhabditis elegans as a model for obesity research. Int. J. Obes. 2012, 36, 186–194. [Google Scholar] [CrossRef]
- Chiang, S.H.; MacDougald, O.A. Will fatty worms help cure human obesity? Trends Genet. 2003, 19, 523–525. [Google Scholar] [CrossRef]
- Martorell, P.; Llopis, S.; González, N.; Chenoll, E.; López-Carreras, N.; Aleixandre, A.; Chen, Y.; Karoly, E.D.; Ramón, D.; Genovés, S. Probiotic strain Bifidobacterium animalis subsp. lactis CECT 8145 reduces fat content and modulates lipid metabolism and antioxidant response in Caenorhabditis elegans. J. Agric. Food Chem. 2016, 64, 3462–3472. [Google Scholar] [CrossRef]
- Lemieux, G.A.; Ashrafi, K. Insights and challenges in using C. elegans for investigation of fat metabolism. Crit. Rev. Biochem. Mol. Biol. 2015, 50, 69–84. [Google Scholar] [CrossRef]
- Kim, H.M.; Do, C.H.; Lee, D.H. Characterization of taurine as anti-obesity agent in C. elegans. J. Biomed. Sci. 2010, 17, S33. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Enright, F.; Keenan, M.; Finley, J.; Zhou, J.; Ye, J.; Greenway, F.; Senevirathne, N.; Gissendanner, C.R.; Manaois, R.; et al. Resistant starch, fermented resistant starch, and short-chain fatty acids reduce intestinal fat deposition in Caenorhabditis elegans. J. Agric. Food Chem. 2010, 58, 4744–4748. [Google Scholar] [CrossRef]
- Zarse, K.; Bossecker, A.; Müller-Kuhrt, L.; Siems, K.; Hernandez, M.A.; Berendsohn, W.G.; Birringer, M.; Ristow, M. The phytochemical glaucarubinone promotes mitochondrial metabolism, reduces body fat, and extends lifespan of Caenorhabditis elegans. Horm. Metab. Res. 2011, 43, 241–243. [Google Scholar] [CrossRef]
- Martorell, P.; Llopis, S.; González, N.; Montón, F.; Ortiz, P.; Genovés, S.; Ramón, D. Caenorhabditis elegans as a model to study the effectiveness and metabolic targets of dietary supplements used for obesity treatment: The specific case of a conjugated linoleic acid mixture (Tonalin). J. Agric. Food Chem. 2012, 60, 11071–11709. [Google Scholar] [CrossRef] [PubMed]
- Yazaki, K.; Yoshikoshi, C.; Oshiro, S.; Yanase, S. Supplemental cellular protection by a carotenoid extends lifespan via Ins/IGF-1 signaling in Caenorhabditis elegans. Oxid. Med. Cell. Longev. 2011. [Google Scholar] [CrossRef] [PubMed]
- Pons, E.; Alquézar, B.; Rodríguez, A.; Martorell, P.; Genovés, S.; Ramón, D.; Rodrigo, M.J.; Zacarías, L.; Peña, L. Metabolic engineering of β-carotene in orange fruit increases its in vivo antioxidant properties. Plant Biotechnol. J. 2014, 12, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Lashmanova, E.; Proshkina, E.; Zhikrivetskaya, S.; Shevchenko, O.; Marusich, E.; Leonov, S.; Melerzanov, A.; Zhavoronkov, A.; Moskalev, A. Fucoxanthin increases lifespan of Drosophila melanogaster and Caenorhabditis elegans. Pharmacol. Res. 2015, 100, 228–241. [Google Scholar] [CrossRef]
- Chen, W.; Mao, L.; Xing, H.; Xu, L.; Fu, X.; Huang, L.; Huang, D.; Pu, Z.; Li, Q. Lycopene attenuates Aβ1-42 secretion and its toxicity in human cell and Caenorhabditis elegans models of Alzheimer disease. Neurosci. Lett. 2015, 608, 28–33. [Google Scholar] [CrossRef]
- You, J.S.; Jeon, S.; Byun, Y.J.; Koo, S.; Choi, S.S. Enhanced biological activity of carotenoids stabilized by phenyl groups. Food Chem. 2015, 177, 339–345. [Google Scholar] [CrossRef]
- Liu, X.; Luo, Q.; Cao, Y.; Goulette, T.; Liu, X.; Xiao, H. Mechanism of different stereoisomeric astaxanthin in resistance to oxidative stress in Caenorhabditis elegans. J. Food Sci. 2016, 81, H2280–H2287. [Google Scholar] [CrossRef]
- Harrison, E.H.; Quadro, L. Apocarotenoids: Emerging roles in mammals. Annu. Rev. Nutr. 2018, 38, 153–172. [Google Scholar] [CrossRef]
- Cui, Y.; Freedman, J.H. Cadmium induces retinoic acid signaling by regulating retinoic acid metabolic gene expression. J. Biol. Chem. 2009, 284, 24925–24932. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, M.J.; Cilla, A.; Barberá, R.; Zacarías, L. Carotenoid bioaccessibility in pulp and fresh juice from carotenoid-rich sweet oranges and mandarins. Food Funct. 2015, 6, 1950–1959. [Google Scholar] [CrossRef] [PubMed]
- Petry, F.C.; Mercadante, A.Z. Composition by LC-MS/MS of new carotenoid esters in mango and citrus. J. Agric. Food Chem. 2016, 64, 8207–8224. [Google Scholar] [CrossRef] [PubMed]
- Carmona, L.; Zacarías, L.; Rodrigo, M.J. Stimulation of coloration and carotenoid biosynthesis during postharvest storage of ‘Navelina’ orange fruit at 12 °C. Postharvest Biol. Technol. 2012, 74, 108–117. [Google Scholar] [CrossRef]
- Martorell, P.; Forment, J.V.; de Llanos, R.; Montón, F.; Llopis, S.; González, N.; Genovés, S.; Cienfuegos, E.; Monzó, H.; Ramón, D. Use of Saccharomyces cerevisiae and Caenorhabditis elegans as model organisms to study the effect of cocoa polyphenols in the resistance to oxidative stress. J. Agric. Food Chem. 2011, 59, 2077–2085. [Google Scholar] [CrossRef] [PubMed]
- Irizarry, R.A.; Hobbs, B.; Collin, F.; Beazer-Barclay, Y.D.; Antonellis, K.J.; Scherf, U.; Speed, T.P. Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics 2003, 4, 249–264. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B (Methodol.) 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Montaner, D.; Dopazo, J. Multidimensional gene set analysis of genomic data. PLoS ONE 2010, 5, e10348. [Google Scholar] [CrossRef]
- Barros, R.G.C.; Andrade, J.K.S.; Denadai, M.; Nunes, M.L.; Narain, N. Evaluation of bioactive compounds potential and antioxidant activity in some Brazilian exotic fruit residues. Food Res. Int. 2017, 102, 84–92. [Google Scholar] [CrossRef]
- Wu, C.; Han, L.; Riaz, H.; Wang, S.; Cai, K.; Yang, L. The chemopreventive effect of β-cryptoxanthin from mandarin on human stomach cells (BGC-823). Food Chem. 2013, 136, 1122–1129. [Google Scholar] [CrossRef] [PubMed]
- Eroglu, A.; Schulze, K.J.; Yager, J.; Cole, R.N.; Christian, P.; Nonyane, B.A.S.; Lee, S.E.; Wu, L.S.F.; Khatry, S.; Groopman, J.; et al. Plasma proteins associated with circulating carotenoids in Nepalese school- aged children. Arch. Biochem. Biophys. 2018, 646, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Ciccone, M.M.; Cortese, F.; Gesualdo, M.; Carbonara, S.; Zito, A.; Ricci, G.; De Pascalis, F.; Scicchitano, P.; Riccioni, G. Dietary intake of carotenoids and their antioxidant and anti-inflammatory effects in cardiovascular care. Mediat. Inflamm. 2013. [Google Scholar] [CrossRef] [PubMed]
- Kaletsky, R.; Murphy, C.T. The role of insulin/IGF-like signaling in C. elegans longevity and aging. Dis. Models Mech. 2010, 3, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.I.; Pincus, Z.; Slack, F.J. Longevity and stress in Caenorhabditis elegans. Aging 2011, 3, 733–753. [Google Scholar] [CrossRef] [PubMed]
- Martorell, P.; Llopis, S.; González, N.; Ramón, D.; Serrano, G.; Torrens, A.; Serrano, J.M.; Navarro, M.; Genovés, S. A nutritional supplement containing lactoferrin stimulates the immune system, extends lifespan, and reduces amyloid β peptide toxicity in Caenorhabditis elegans. Food Sci. Nutr. 2017, 5, 255–265. [Google Scholar] [CrossRef]
- Gammone, M.A.; Riccioni, G.; D’Orazio, N. Carotenoids: Potential allies of cardiovascular health? Food Nutr. Res. 2015, 59, 26762. [Google Scholar]
- Phelps, M.E.; Barrio, J.R. Correlation of brain amyloid with “aerobic glycolysis”: A question of assumptions? Proc. Natl. Acad. Sci. USA 2010, 107, 17459–17460. [Google Scholar] [CrossRef]
- Sugiura, M.; Nakamura, M.; Ikoma, Y.; Yano, M.; Ogawa, K.; Matsumoto, H.; Kato, M.; Ohshima, M.; Nagao, A. The homeostasis model assessment-insulin resistance index is inversely associated with serum carotenoids in non-diabetic subjects. J. Epidemiol. 2006, 16, 71–78. [Google Scholar] [CrossRef]
- Sahin, K.; Orhan, C.; Akdemir, F.; Tuzcu, M.; Sahin, N.; Yilmaz, I.; Juturu, V. BCX ameliorates metabolic risk factors by regulating NF-κB and Nrf2 pathways in insulin resistance induced by high-fat diet in rodents. Food Chem. Toxicol. 2017, 107, 270–279. [Google Scholar] [CrossRef]
- Kaur, P.; Kaur, J. Potentyal role of lycopene as antioxidant and implications for human health and disease. In Lycopene Food Sources, Potential Role in Human Health and Antioxidant Effects; Bailey, J.R., Ed.; Nova Science Publishers, Inc.: New York, NY, USA, 2015; pp. 1–38. ISBN 1631179276. [Google Scholar]
- Fu, H.; Xie, B.; Fan, G.; Ma, S.; Zhu, X.; Pan, S.Y. Effect of esterification with fatty acid of β-cryptoxanthin on its thermal stability and antioxidant activity by chemiluminescence method. Food Chem. 2010, 122, 602–609. [Google Scholar] [CrossRef]
- Breithaupt, D.E.; Weller, P.; Wolters, M.; Hahn, A. Plasma response to a single dose of dietary BCX esters from papaya (Carica papaya L.) or non-esterified BCX in adult human subjects: A comparative study. Br. J. Nutr. 2003, 90, 795–801. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, Y.; Azqueta, A.; Luna, L.; Bonilla, F.; Domínguez, G.; Collins, A.R. The carotenoid BCX stimulates the repair of DNA oxidation damage in addition to acting as an antioxidant in human cells. Carcinogenesis 2009, 30, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Dandona, P.; Aljada, A.; Chaudhuri, A.; Mohanty, P.; Garg, R. Metabolic syndrome. A comprehensive perspective based on interactions between obesity, diabetes, and inflammation. Circulation 2005, 111, 1448–1454. [Google Scholar] [CrossRef] [PubMed]
- Haegele, A.D.; Gillette, C.; O’Neill, C.; Wolfe, P.; Heimendinger, J.; Sedlacek, S.; Thompson, H.J. Plasma xanthophyll carotenoids correlate inversely with indices of oxidative DNA damage and lipid peroxidation. Cancer Epidemiol. Biomark. Prev. 2000, 9, 421–425. [Google Scholar]
- Alavez, S.; Vantipalli, M.C.; Zucker, D.J.; Klang, I.M.; Lithgow, G.J. Amyloid binding compounds maintain protein homeostasis during ageing and extend lifespan. Nature 2011, 472, 226–229. [Google Scholar] [CrossRef] [PubMed]
- Regitz, C.; Dußling, L.M.; Wenzel, U. Amyloid-beta (Aβ(1-42))-induced paralysis in Caenorhabditis elegans is inhibited by the polyphenol quercetin through activation of protein degradation pathways. Mol. Nutr. Food Res. 2014, 58, 1931–1940. [Google Scholar] [CrossRef] [PubMed]
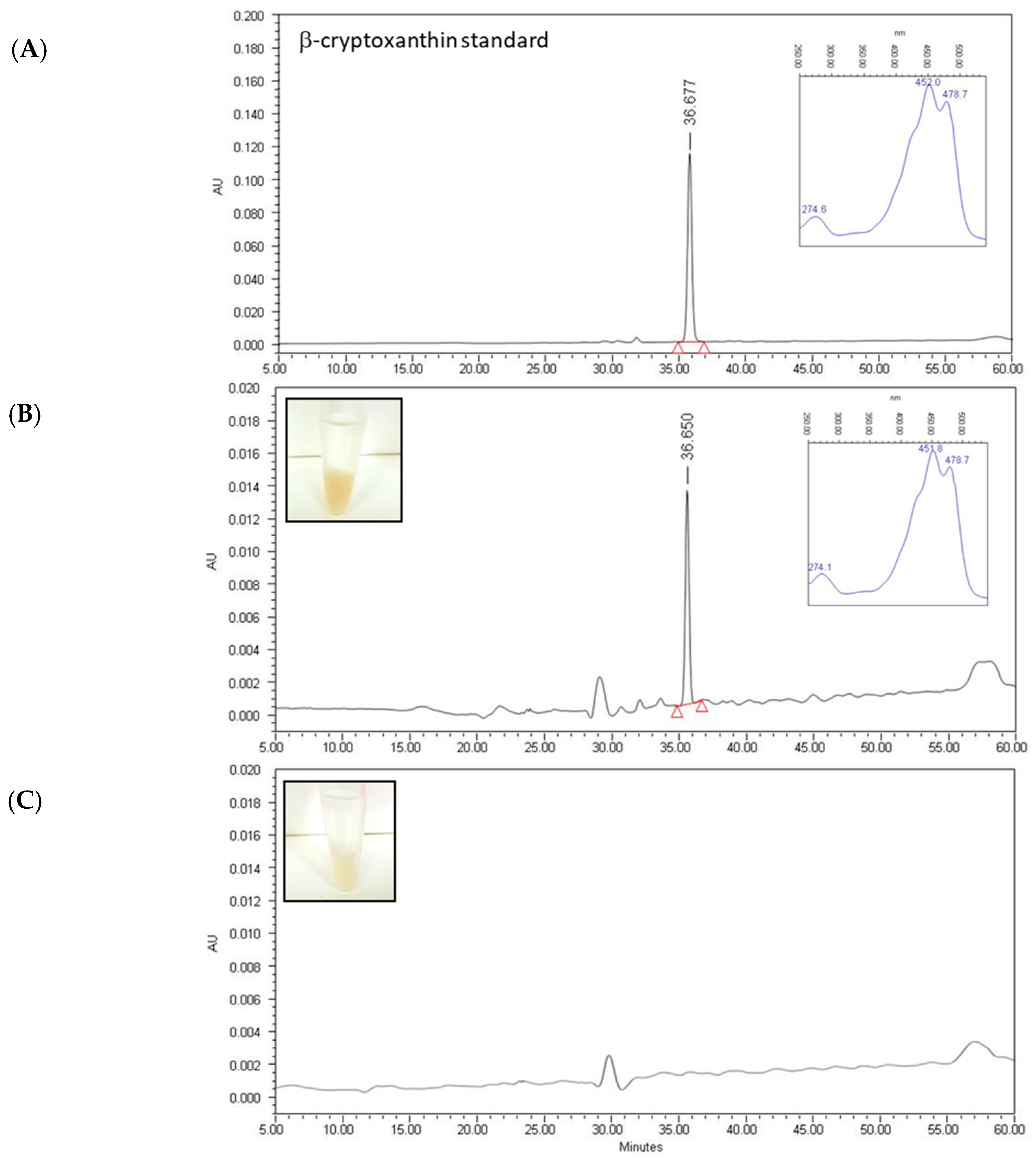
| Carotenoid | Concentration (µg/mL) in Mandarin Juice Extract 1 | |
|---|---|---|
| Saponified | Non-saponified | |
| Phytoene | 0.80 ± 0.05 | 0.86 ± 0.03 |
| Phytofluene | 0.37 ± 0.09 | 0.45 ± 0.02 |
| ζ-Carotene | 0.19 ± 0.04 | 0.25 ± 0.03 |
| β-Carotene | 0.28 ± 0.05 | 0.35 ± 0.02 |
| β-Cryptoxanthin | 4.86 ± 0.23 (61%) | 0.30 ± 0.02 (3%) |
| Zeaxanthin | 0.11 ± 0.02 | N.D. |
| Anteraxanthin | 0.52 ± 0.01 | N.D. |
| Violaxanthin 2 | 0.75 ± 0.01 | N.D. |
| Esters (mono- and diesters) | ||
| β-Crytoxanthin | N.D. | 5.35 ± 0.25 (57%) |
| Zeaxanthin | N.D. | 0.20 ± 0.01 |
| Antherxanthin | N.D. | 0.70 ± 0.03 |
| Violaxanthin 2 | N.D. | 0.92 ± 0.04 |
| Total carotenoids | 7.88 ± 0.62 | 9.38 ± 0.48 |
| Upregulated KEGGSs Pathways (BCX vs. Control) | |||
|---|---|---|---|
| ID | Name | Size a | p-Value |
| 04141 | Protein processing in endoplasmic reticulum | 170 | 1.26 × 109 |
| 00020 | Citrate cycle (TCA cycle) | 42 | 0.0005 |
| 03040 | Spliceosome | 123 | 0.0025 |
| 00010 | Glycolysis/gluconeogenesis | 38 | 0.0067 |
| 03030 | DNA replication | 39 | 0.0088 |
| 00970 | Aminoacyl-tRNA biosynthesis | 49 | 0.0118 |
| 03050 | Proteasome | 45 | 0.0180 |
| 00030 | Pentose phosphate pathway | 26 | 0.0180 |
| 04144 | Endocytosis | 109 | 0.0231 |
| 00520 | Amino sugar and nucleotide sugar metabolism | 32 | 0.0232 |
| 04130 | SNARE interactions in vesicular transport | 23 | 0.0232 |
| 00240 | Pyrimidine metabolism | 71 | 0.0232 |
| 00052 | Galactose metabolism | 13 | 0.0247 |
| 00051 | Fructose and mannose metabolism | 31 | 0.0247 |
| 00564 | Glycerophospholipid metabolism | 54 | 0.0323 |
| 04330 | Notch signaling pathway | 28 | 0.0393 |
| 00190 | Oxidative phosphorylation | 127 | 0.0393 |
| 04120 | Ubiquitin mediated proteolysis | 111 | 0.0393 |
| Non-Redundant Upregulated GOs (BCX vs. Control) | |
|---|---|
| ID | Name |
| 0000910 | Cytokinesis |
| 0048598 | Embryonic morphogenesis |
| 0006886 | Intracellular protein transport |
| 0006400 | tRNA modification |
| 0006479 | Protein amino acid methylation |
| 0006099 | Tricarboxylic acid cycle |
| 0006412 | Translation |
| 0019751 | Polyol metabolic process |
| 0006261 | DNA-dependent DNA replication |
| 0006457 | Protein folding |
| 0007346 | Regulation of mitotic cell cycle |
| 0030261 | Chromosome condensation |
| 0008593 | Regulation of Notch signaling pathway |
| 0008284 | Positive regulation of cell proliferation |
| 0040028 | Regulation of vulval development |
| 0000075 | Cell cycle checkpoint |
| 0001708 | Cell fate specification |
| 0040019 | Positive regulation of embryonic development |
| 0007067 | Mitosis |
| 0008356 | Asymmetric cell division |
| 0006096 | Glycolysis |
| 0040015 | Negative regulation of multicellular organism growth |
| 0007283 | Spermatogenesis |
| 0051246 | Regulation of protein metabolic process |
| 0040039 | Inductive cell migration |
| 0033554 | Cellular response to stress |
| 0000132 | Stablishment of mitotic spindle orientation |
| 0045132 | Meiotic chromosome segregation |
| 0045137 | Development of primary sexual characteristics |
| 0035194 | Posttranscriptional gene silencing by RNA |
| 0007143 | Female meiosis |
| 0046580 | Negative regulation of Ras protein signal transduction |
| 0000184 | Nuclear-transcribed mRNA catabolic process, nonsense-mediated decay |
| 0022613 | Ribonucleoprotein complex biogenesis |
| 0051302 | Regulation of cell division |
| Non-Redundant Downregulated GOs (BCX vs. Control) | |
|---|---|
| ID | Name |
| 0006813 | Potassium ion transport |
| 0007606 | Sensory perception of chemical stimulus |
| 0007186 | G-protein coupled receptor protein signaling pathway |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Llopis, S.; Rodrigo, M.J.; González, N.; Genovés, S.; Zacarías, L.; Ramón, D.; Martorell, P. β-Cryptoxanthin Reduces Body Fat and Increases Oxidative Stress Response in Caenorhabditis elegans Model. Nutrients 2019, 11, 232. https://doi.org/10.3390/nu11020232
Llopis S, Rodrigo MJ, González N, Genovés S, Zacarías L, Ramón D, Martorell P. β-Cryptoxanthin Reduces Body Fat and Increases Oxidative Stress Response in Caenorhabditis elegans Model. Nutrients. 2019; 11(2):232. https://doi.org/10.3390/nu11020232
Chicago/Turabian StyleLlopis, Silvia, María Jesús Rodrigo, Nuria González, Salvador Genovés, Lorenzo Zacarías, Daniel Ramón, and Patricia Martorell. 2019. "β-Cryptoxanthin Reduces Body Fat and Increases Oxidative Stress Response in Caenorhabditis elegans Model" Nutrients 11, no. 2: 232. https://doi.org/10.3390/nu11020232
APA StyleLlopis, S., Rodrigo, M. J., González, N., Genovés, S., Zacarías, L., Ramón, D., & Martorell, P. (2019). β-Cryptoxanthin Reduces Body Fat and Increases Oxidative Stress Response in Caenorhabditis elegans Model. Nutrients, 11(2), 232. https://doi.org/10.3390/nu11020232





