Effect of a Euglena gracilis Fermentate on Immune Function in Healthy, Active Adults: A Randomized, Double-Blind, Placebo-Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Investigational Product
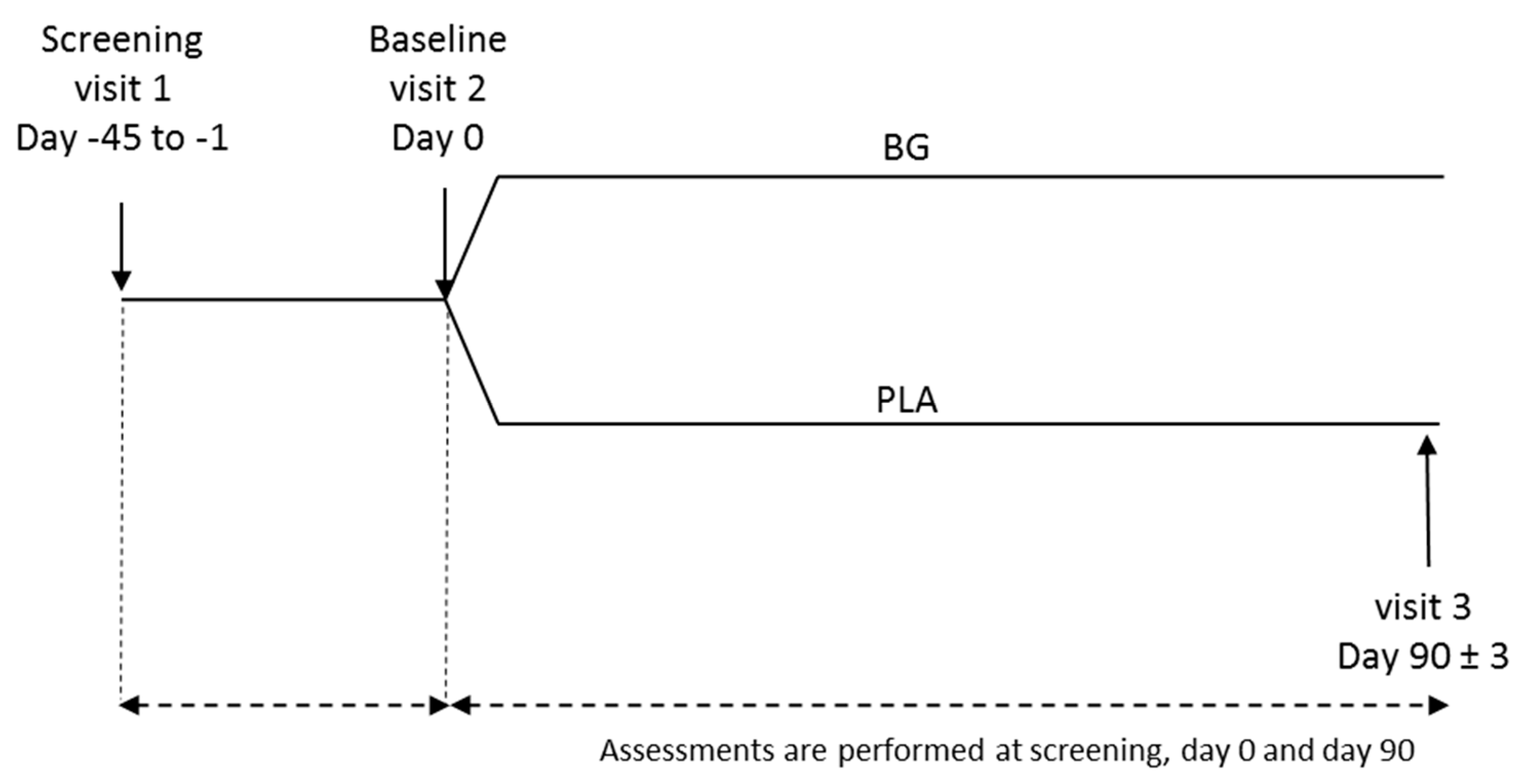
2.2. Study Design
2.3. Study Population
2.4. Study Procedure
2.5. Wisconsin Upper Respiratory Symptom Survey-24 (WURSS-24)
2.6. Outcomes
2.7. Safety
2.8. Statistical Analyses
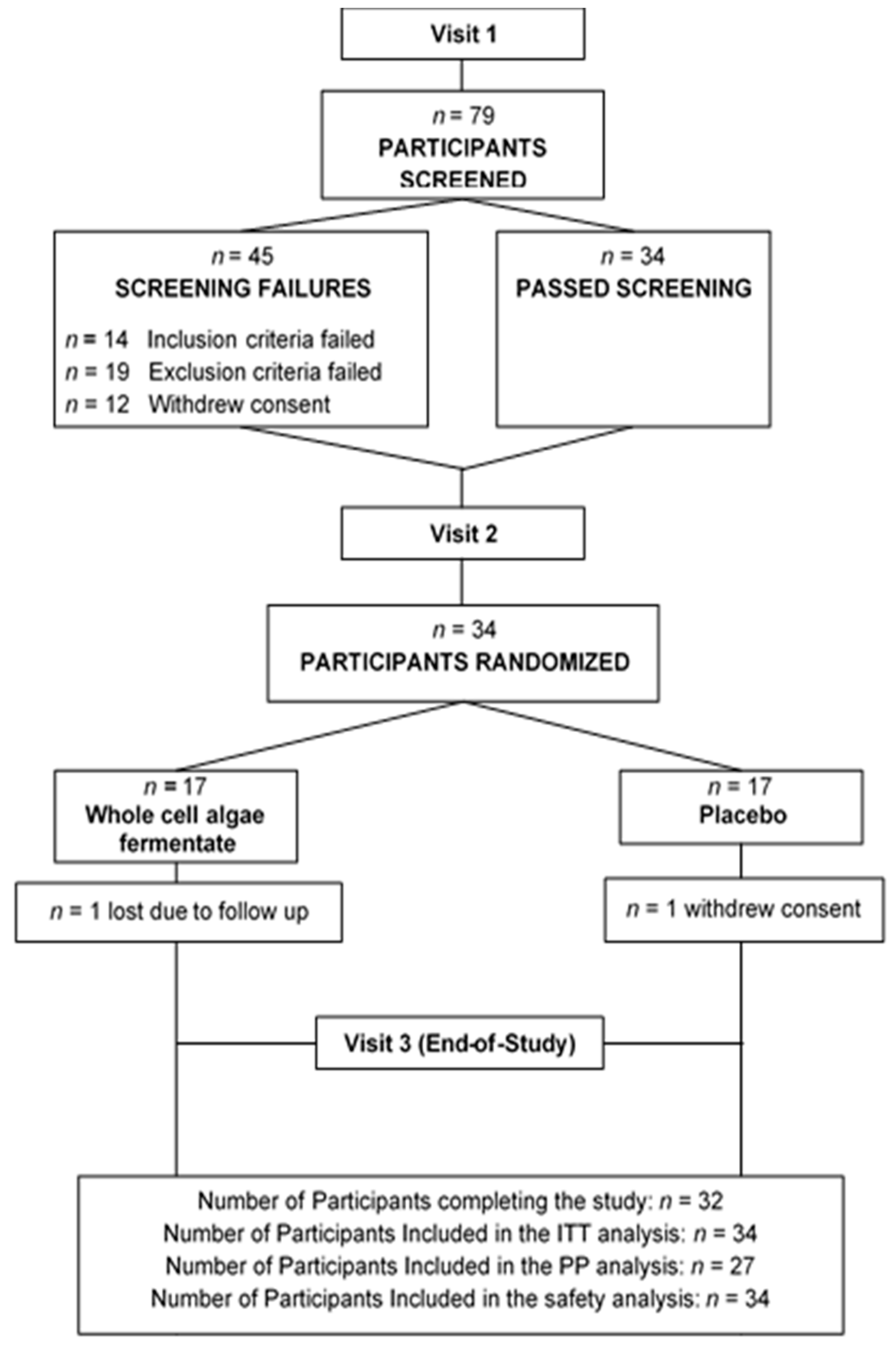
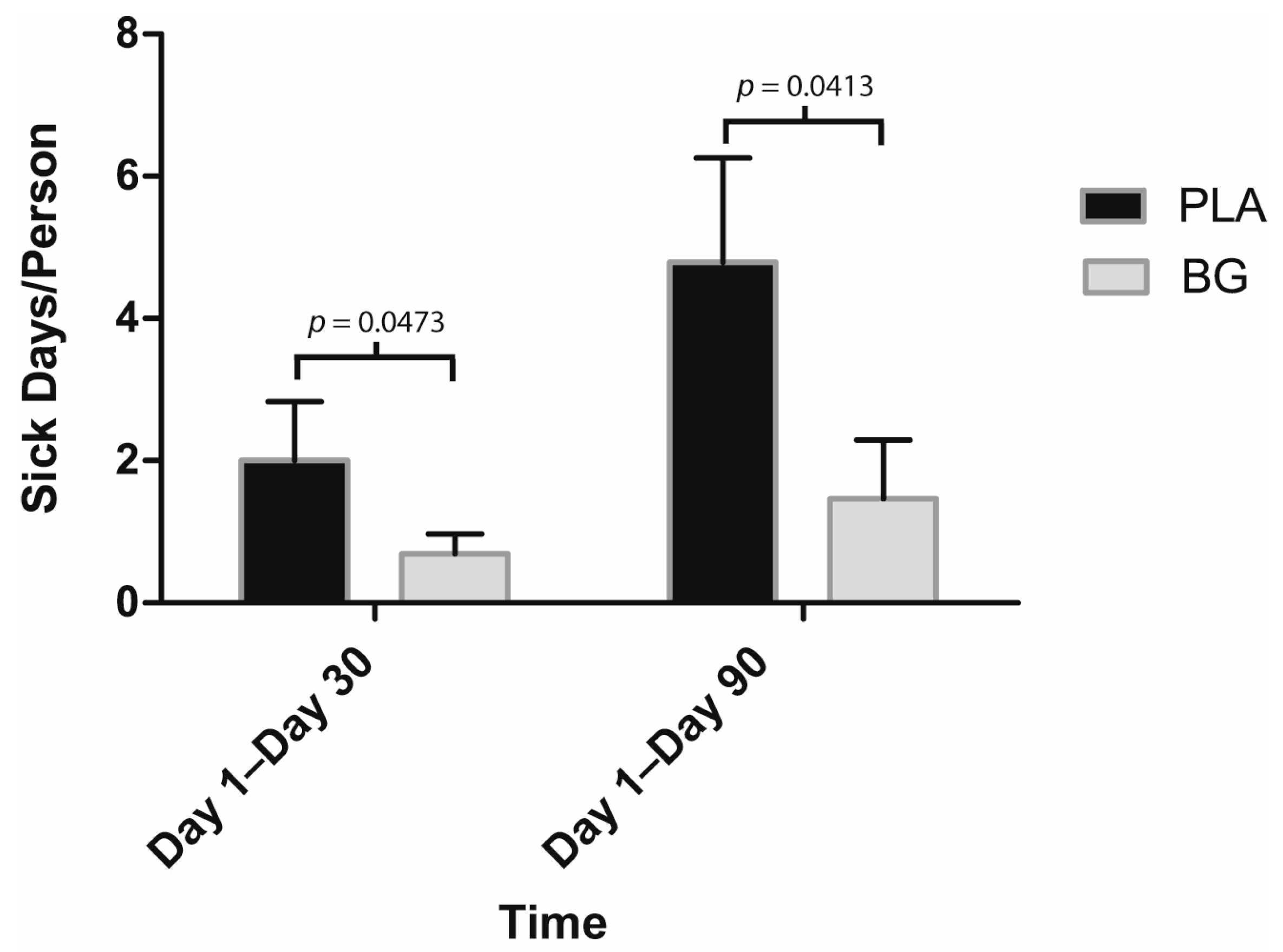
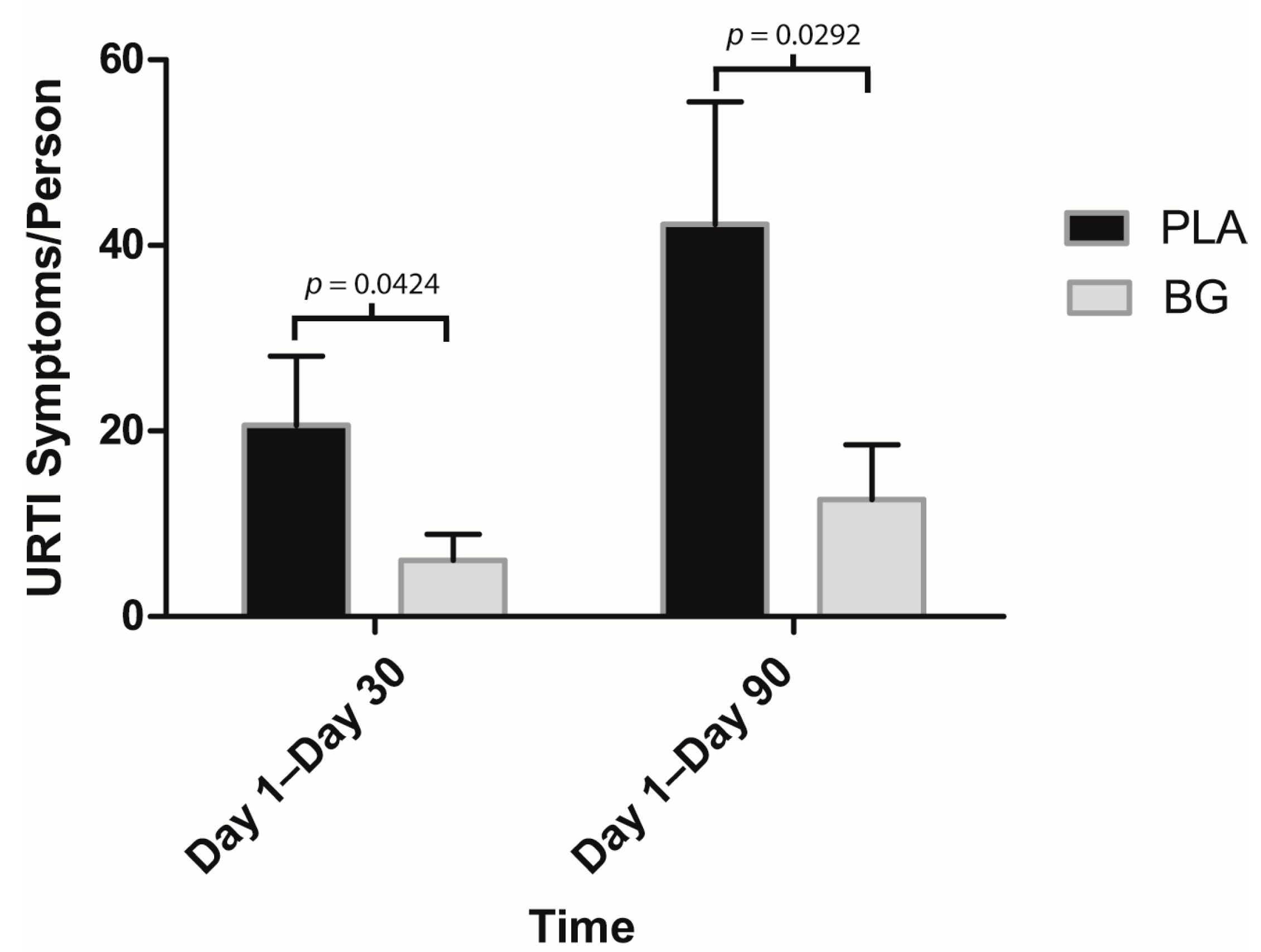
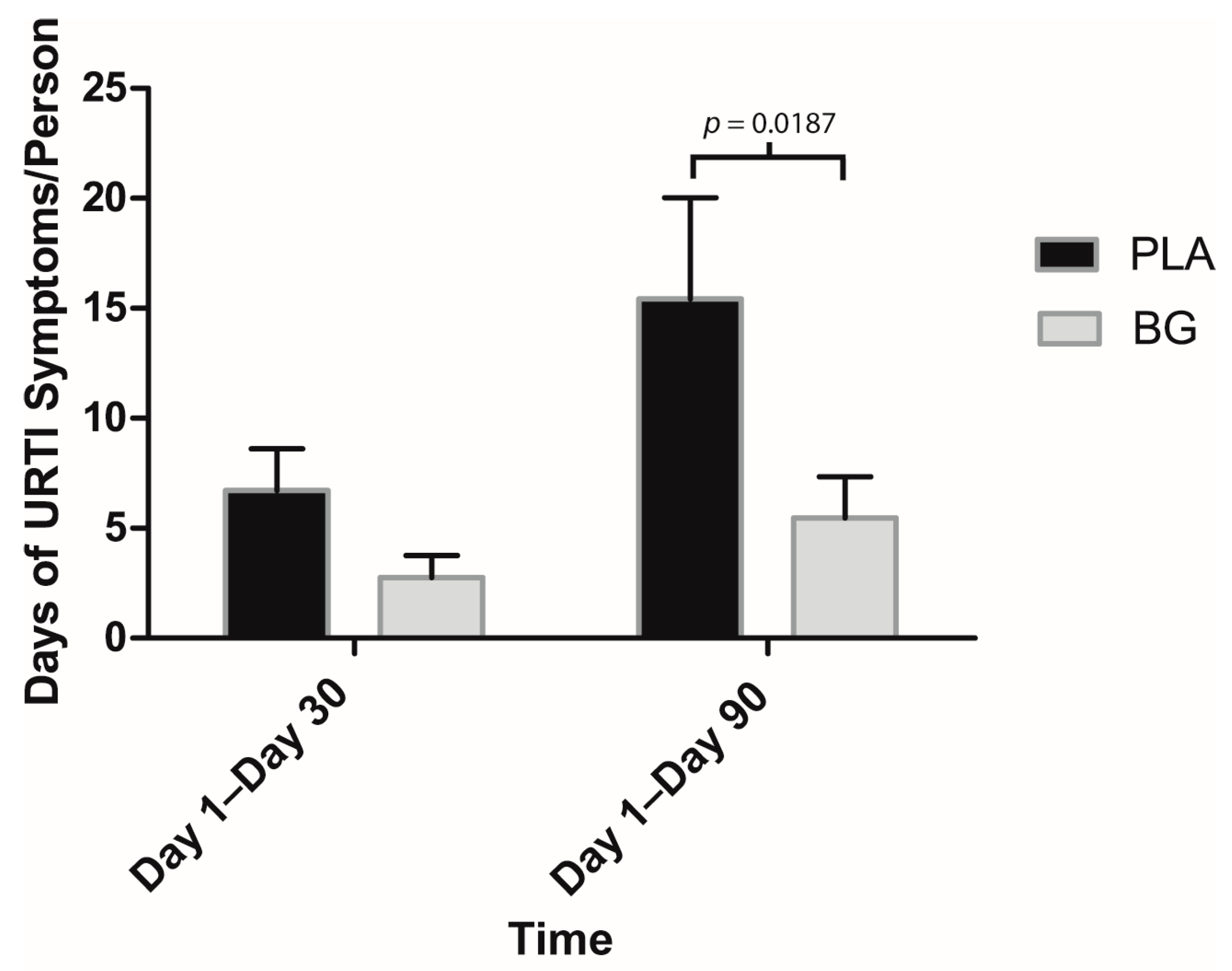
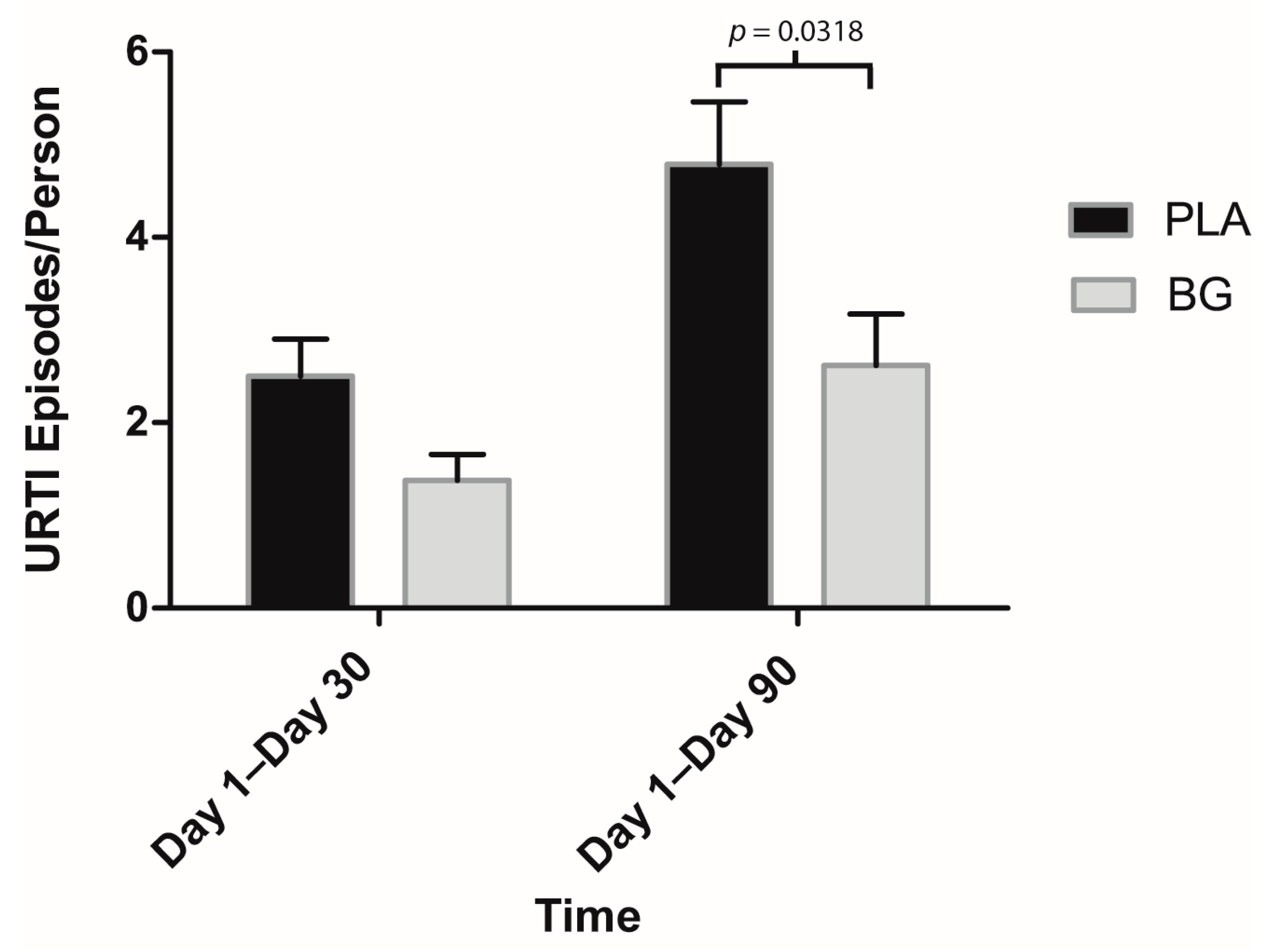
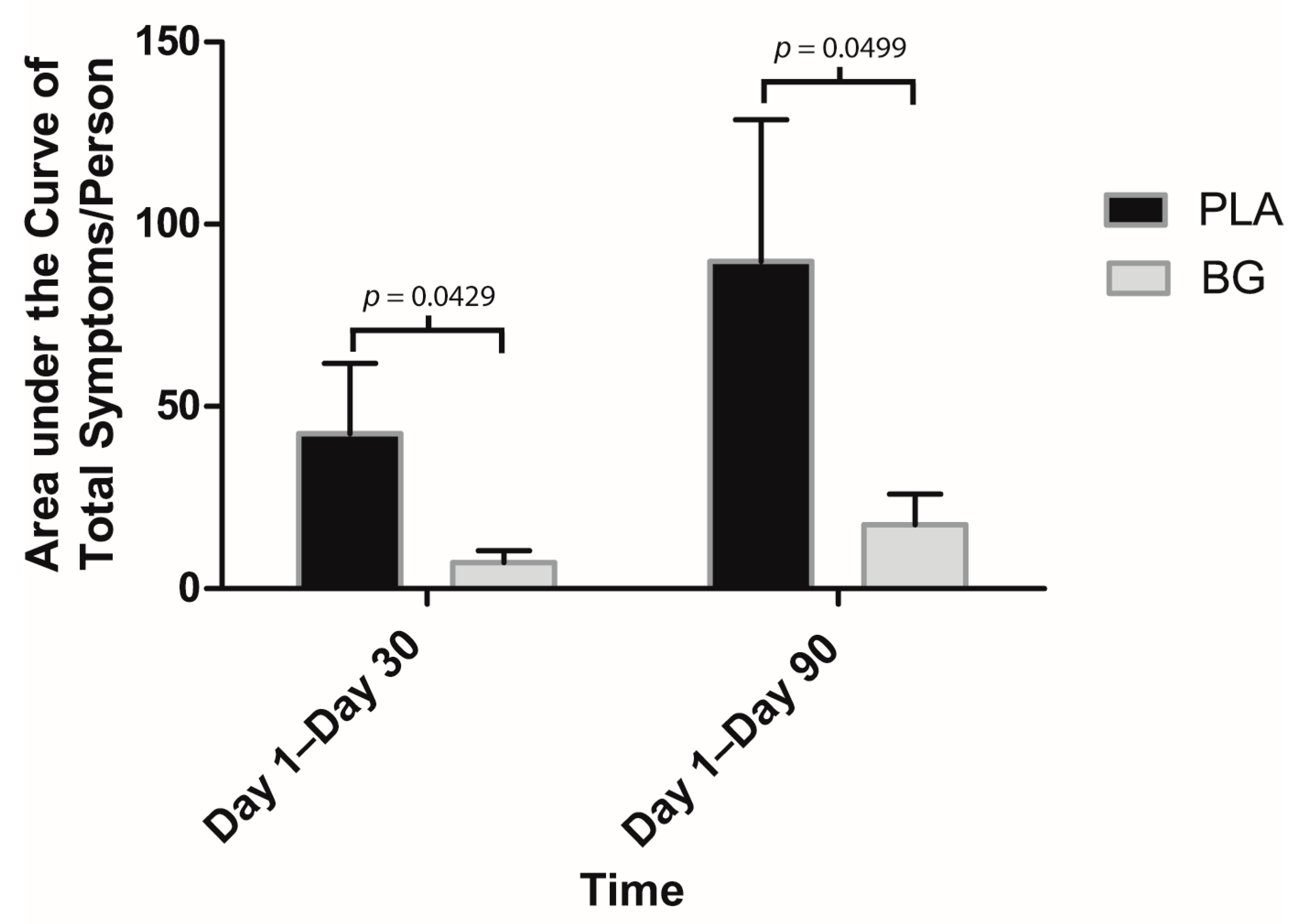
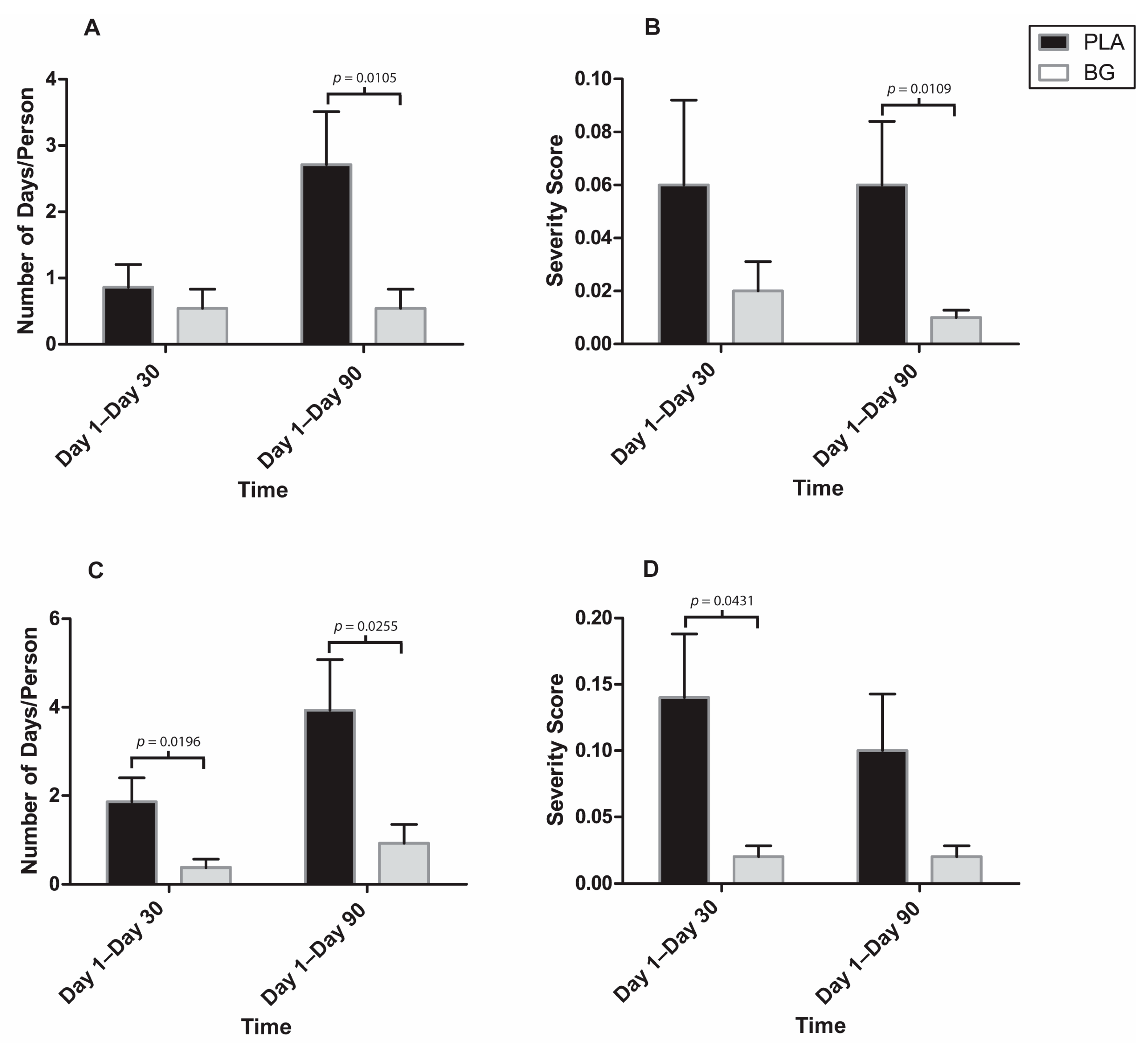
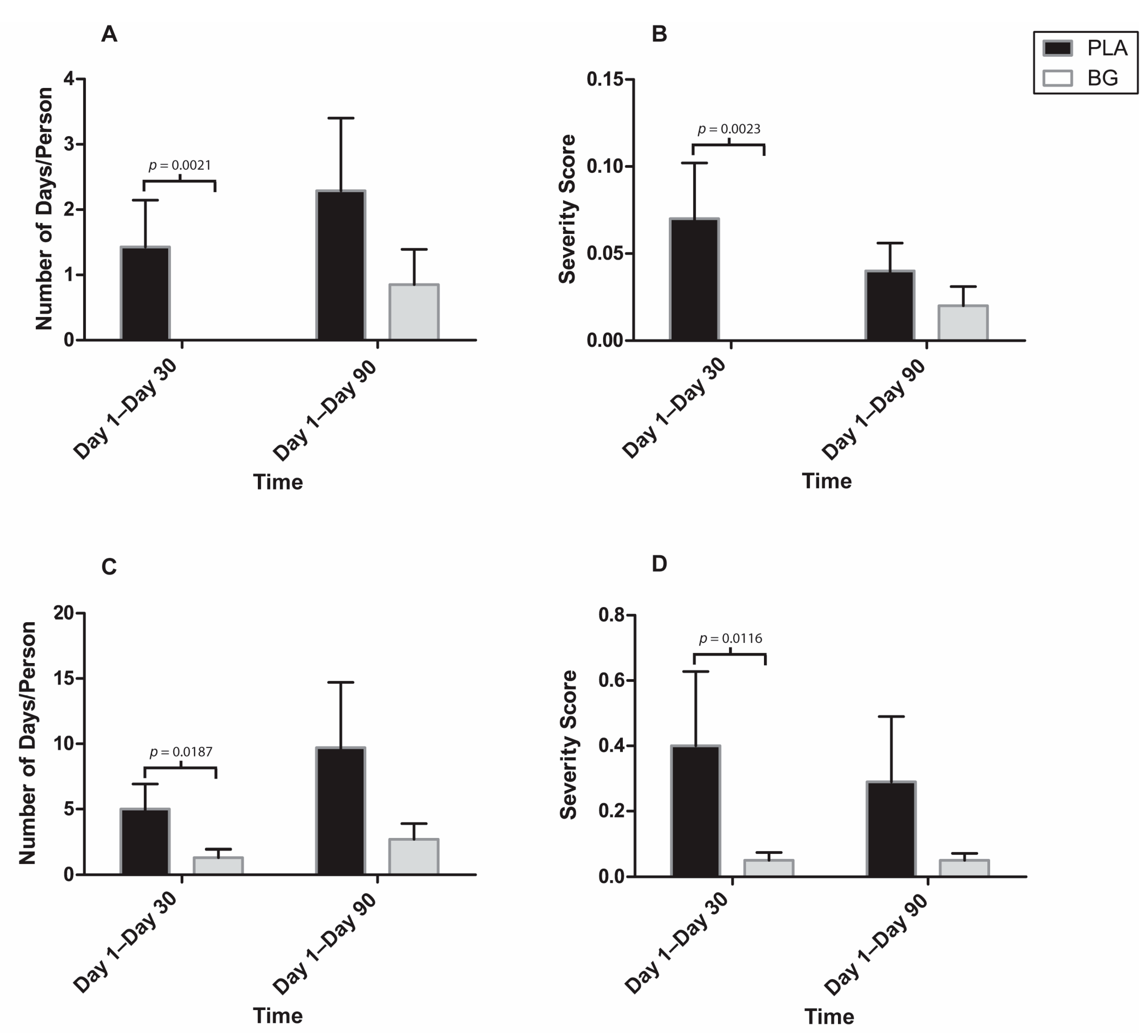
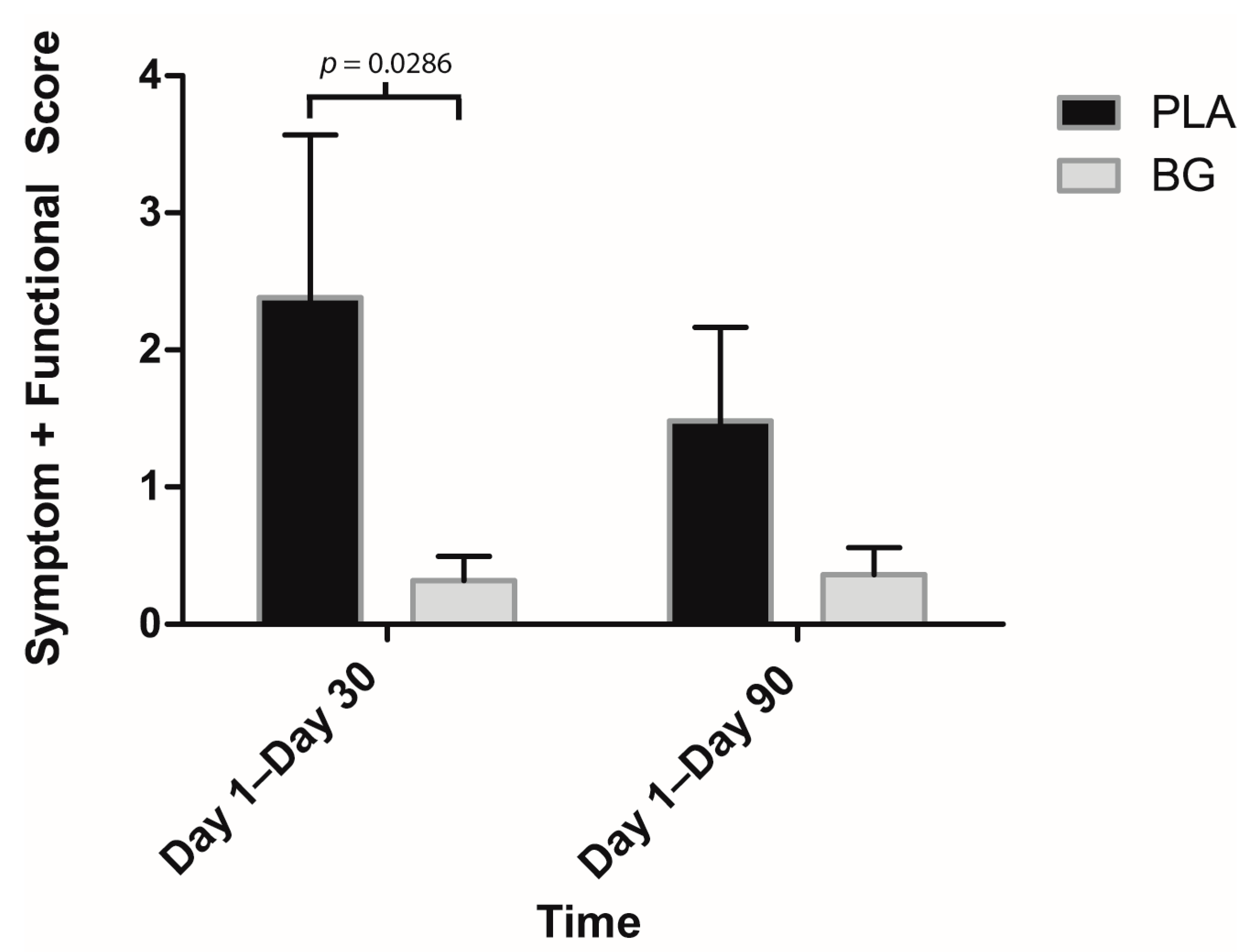
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barrett, B.; Brown, R.L.; Mundt, M.P.; Thomas, G.R.; Barlow, S.K.; Highstrom, A.D.; Bahrainian, M. Validation of a short form Wisconsin Upper Respiratory Symptom Survey (WURSS-21). Health Qual. Life Outcomes 2009, 7, 76. [Google Scholar] [CrossRef] [PubMed]
- Douglas, R.M. Respiratory tract infections as a public health challenge. Clin. Infect. Dis. 1999, 28, 192–194. [Google Scholar] [CrossRef] [PubMed]
- Gwaltney, J.M., Jr.; Hendley, J.O.; Simon, G.; Jordan, W.S., Jr. Rhinovirus infections in an industrial population: The occurrence of illness. New Engl. J. Med. 1966, 275, 1261–1268. [Google Scholar] [CrossRef]
- Monto, A.S.; Ullman, B.M. Acute respiratory illness in an American community: The Tecumseh study. JAMA. 1974, 227, 164–169. [Google Scholar] [CrossRef]
- Dingle, J.H.; Badger, G.F.; Jordan, W.S., Jr. Illness in the Home. A Study of 25,000 Illnesses in a Group of Cleveland Families; The Press of Western Reserve University: Cleveland, OH, USA, 1964; p. 398. [Google Scholar]
- Fendrick, A.M.; Monto, A.S.; Nightengale, B.; Sarnes, M. The economic burden of non-influenza-related viral respiratory tract infection in the United States. Arch. Intern. Med. 2003, 163, 487–494. [Google Scholar] [CrossRef]
- Wu, D.; Lewis, E.D.; Pae, M.; Meydani, S.N. Nutritional Modulation of Immune Function: Analysis of Evidence, Mechanisms, and Clinical Relevance. Front. Immunol. 2018, 9, 3160. [Google Scholar] [CrossRef]
- Krajcovic, J.; Matej, V.; Schwartzbach, S.D. Euglenoid flagellates: a multifaceted biotechnology platform. J Biotechnol 2015, 202, 135–145. [Google Scholar] [CrossRef]
- Watanabe, F.; Yoshimura, K.; Shigeoka, S. Biochemistry and Physiology of Vitamins in Euglena. Euglena: Biochem. Cell Mol. Biol. 2017, 979, 65–90. [Google Scholar] [CrossRef]
- Lewis, E.D.; Meydani, S.N.; Wu, D. Regulatory role of vitamin E in the immune system and inflammation. IUBMB Life 2019, 71, 487–494. [Google Scholar] [CrossRef]
- Carr, A.C.; Maggini, S. Vitamin C and Immune Function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef]
- Russo, R.; Barsanti, L.; Evangelista, V.; Frassanito, A.M.; Longo, V.; Pucci, L.; Penno, G.; Gualtieri, P. Euglena gracilis paramylon activates human lymphocytes by upregulating pro-inflammatory factors. Food Sci. Nutr. 2017, 5, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Monfils, A.K.; Triemer, R.E.; Bellairs, E.F. Characterization of paramylon morphological diversity in photosynthetic euglenoids (Euglenales, Euglenophyta). Phycologia 2011, 50, 156–169. [Google Scholar] [CrossRef]
- Barsanti, L. Paramylon ([beta]-1,3-glucan) content in wild type and WZSL mutant of Euglena gracilis. Effects of growth conditions. J. Appl. Phycol. 2001, 13, 59–65. [Google Scholar] [CrossRef]
- Raina MacIntyre, C.; Chughtai, A.A.; Zhang, Y.; Seale, H.; Yang, P.; Chen, J.; Pan, Y.; Zhang, D.; Wang, Q. Viral and bacterial upper respiratory tract infection in hospital health care workers over time and association with symptoms. BMC Infect. Dis. 2017, 17, 553. [Google Scholar] [CrossRef] [PubMed]
- Harger-Domitrovich, S.G. Effects of an Immunomodulating Supplement on Upper Respiratory Tract Infection Symptoms in Wildland Firefighters 2001. Med. Sci. Sports Exerc. 2008, 40. [Google Scholar] [CrossRef]
- Gleeson, M.; Pyne, D.B. Respiratory inflammation and infections in high-performance athletes. Immunol. Cell Biol. 2016, 94, 124–131. [Google Scholar] [CrossRef]
- Moraes, H.; Aoki, M.S.; Freitas, C.G.; Arruda, A.; Drago, G.; Moreira, A. SIgA response and incidence of upper respiratory tract infections during intensified training in youth basketball players. Biol. Sport 2017, 34, 49–55. [Google Scholar] [CrossRef]
- Bergendiova, K.; Tibenska, E.; Majtan, J. Pleuran (beta-glucan from Pleurotus ostreatus) supplementation, cellular immune response and respiratory tract infections in athletes. Eur. J. Appl. Physiol 2011, 111, 2033–2040. [Google Scholar] [CrossRef]
- Woods, J.A.; Davis, J.M.; Smith, J.A.; Nieman, D.C. Exercise and cellular innate immune function. Med. Sci. Sports Exerc. 1999, 31, 57–66. [Google Scholar] [CrossRef]
- Carpenter, K.C.; Breslin, W.L.; Davidson, T.; Adams, A.; McFarlin, B.K. Baker’s yeast beta-glucan supplementation increases monocytes and cytokines post-exercise: Implications for infection risk? Br. J. Nutr. 2013, 109, 478–486. [Google Scholar] [CrossRef]
- Peters, E.M.; Bateman, E.D. Ultramarathon running and upper respiratory tract infections. An epidemiological survey. South. Afr. Med. J. 1983, 64, 582–584. [Google Scholar]
- Moher, D.; Hopewell, S.; Schulz, K.F.; Montori, V.; Gotzsche, P.C.; Devereaux, P.J.; Elbourne, D.; Egger, M.; Altman, D.G. CONSORT 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials. Int. J. Surg. 2012, 10, 28–55. [Google Scholar] [CrossRef]
- Ensley, H.E.; Tobias, B.; Pretus, H.A.; McNamee, R.B.; Jones, E.L.; William Browder, I.; Williams, D.L. NMR spectral analysis of a water-insoluble (1 → 3)-β-d-glucan isolated from Saccharomyces cerevisiae. Carbohydr. Res. 1994, 258, 307–311. [Google Scholar] [CrossRef]
- Kim, Y.T.; Kim, E.H.; Cheong, C.; Williams, D.L.; Kim, C.W.; Lim, S.T. Structural characterization of β-d-(1 → 3, 1 → 6)-linked glucans using NMR spectroscopy. Carbohydr. Res. 2000, 328, 331–341. [Google Scholar] [CrossRef]
- Simon, R.R.; Vo, T.D.; Levine, R. Genotoxicity and subchronic toxicity evaluation of dried Euglena gracilis ATCC PTA-123017. Regul. Toxicol. Pharmacol. 2016, 80, 71–81. [Google Scholar] [CrossRef]
- Longmier, E.; Barrett, B.; Brown, R. Can patients or clinicians predict the severity or duration of an acute upper respiratory infection? Fam. Pract. 2013, 30, 379–385. [Google Scholar] [CrossRef]
- Johane, G.; Pierre, C.; Pascal, G.; Joerg, G. The Efficacy and Safety of a Patent Pending Combination of Ginger and Goldenrod Extracts on the Management of Cold Symptoms: A Randomized, Double-Blind Controlled Trial. Food Nutr. Sci. 2012, 3, 1651–1657. [Google Scholar]
- Nieman, D.C. Risk of upper respiratory tract infection in athletes: An epidemiologic and immunologic perspective. J. Athl. Train. 1997, 32, 344–349. [Google Scholar]
- 2017–2018 Community Influenza Surveillance Report. Available online: https://www.google.ch/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&ved=2ahUKEwi1kKmTo5bmAhXMQN4KHa6tBpoQFjAAegQIARAC&url=https%3A%2F%2Fwww.healthunit.com%2Fuploads%2F2017-11-22-community-influenza-surveillance.pdf&usg=AOvVaw3LqkulaqiPzaEdsHrh8LnX (accessed on 1 May 2019).
- McFarlin, B.K.; Carpenter, K.C.; Davidson, T.; McFarlin, M.A. Baker’s yeast beta glucan supplementation increases salivary IgA and decreases cold/flu symptomatic days after intense exercise. J. Diet. Suppl. 2013, 10, 171–183. [Google Scholar] [CrossRef]
- Talbott, S.; Talbott, J. Effect of Beta 1, 3/1, 6 Glucan on Upper Respiratory Tract Infection Symptoms and Mood State in Marathon Athletes. J. Sports Sci. Med. 2009, 8, 509–515. [Google Scholar]
- Fuller, R.; Moore, M.V.; Lewith, G.; Stuart, B.L.; Ormiston, R.V.; Fisk, H.L.; Noakes, P.S.; Calder, P.C. Yeast-derived beta-1,3/1,6 glucan, upper respiratory tract infection and innate immunity in older adults. Nutrition 2017, 39–40, 30–35. [Google Scholar] [CrossRef]
- Talbott, S.M.; Talbott, J.A. Baker’s yeast beta-glucan supplement reduces upper respiratory symptoms and improves mood state in stressed women. J. Am. Coll. Nutr. 2012, 31, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Mah, E.; Kaden, V.N.; Kelley, K.M.; Liska, D.J. Beverage Containing Dispersible Yeast beta-Glucan Decreases Cold/Flu Symptomatic Days After Intense Exercise: A Randomized Controlled Trial. J. Diet. Suppl. 2018, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Feldman, S.; Schwartz, H.I.; Kalman, D.S.; Mayers, A.; Kohrman, H.M.; Clemens, R.; Krieger, D.R. Randomized phase II clinical trials of Wellmune WGP® for immune support during cold and flu season. J. Appl. Res. 2009, 9, 30–42. [Google Scholar]
- Kondo, Y.; Kato, A.; Hojo, H.; Nozoe, S.; Takeuchi, M.; Ochi, K. Cytokine-related immunopotentiating activities of paramylon, a beta-(1 → 3)- D-glucan from Euglena gracilis. J. Pharm. 1992, 15, 617–621. [Google Scholar] [CrossRef] [PubMed]
- Barsanti, L.; Passarelli, V.; Evangelista, V.; Frassanito, A.M.; Gualtieri, P. Chemistry, physico-chemistry and applications linked to biological activities of beta-glucans. Nat. Prod. Rep. 2011, 28, 457–466. [Google Scholar] [CrossRef]
- Brown, G.D.; Taylor, P.R.; Reid, D.M.; Willment, J.A.; Williams, D.L.; Martinez-Pomares, L.; Wong, S.Y.; Gordon, S. Dectin-1 is a major beta-glucan receptor on macrophages. J. Exp. Med. 2002, 196, 407–412. [Google Scholar] [CrossRef]
- Taylor, P.R.; Brown, G.D.; Reid, D.M.; Willment, J.A.; Martinez-Pomares, L.; Gordon, S.; Wong, S.Y. The beta-glucan receptor, dectin-1, is predominantly expressed on the surface of cells of the monocyte/macrophage and neutrophil lineages. J. Immunol. 2002, 169, 3876–3882. [Google Scholar] [CrossRef]
- Adams, E.L.; Rice, P.J.; Graves, B.; Ensley, H.E.; Yu, H.; Brown, G.D.; Gordon, S.; Monteiro, M.A.; Papp-Szabo, E.; Lowman, D.W.; et al. Differential high-affinity interaction of dectin-1 with natural or synthetic glucans is dependent upon primary structure and is influenced by polymer chain length and side-chain branching. J. Pharmacol. Exp. Ther. 2008, 325, 115–123. [Google Scholar] [CrossRef]
- Taylor, P.R. Dectin-1 is required for beta-glucan recognition and control of fungal infection. Nat. Immunol. 2007, 8, 31–38. [Google Scholar] [CrossRef]
- Goodridge, H.S.; Wolf, A.J.; Underhill, D.M. Beta-glucan recognition by the innate immune system. Immunol. Rev. 2009, 230, 38–50. [Google Scholar] [CrossRef]
- Kim, H.S.; Hong, J.T.; Kim, Y.; Han, S.B. Stimulatory Effect of beta-glucans on Immune Cells. Immune Netw. 2011, 11, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Goodridge, H.S.; Reyes, C.N.; Becker, C.A.; Katsumoto, T.R.; Ma, J.; Wolf, A.J.; Bose, N.; Chan, A.S.; Magee, A.S.; Danielson, M.E.; et al. Activation of the innate immune receptor Dectin-1 upon formation of a ‘phagocytic synapse’. Nature 2011, 472, 471–475. [Google Scholar] [CrossRef] [PubMed]
- Auinger, A.; Riede, L.; Bothe, G.; Busch, R.; Gruenwald, J. Yeast (1, 3)-(1, 6)-beta-glucan helps to maintain the body’s defence against pathogens: A double-blind, randomized, placebo-controlled, multicentric study in healthy subjects. Eur. J. Nutr. 2013, 52, 1913–1918. [Google Scholar] [CrossRef]
| Inclusion Criteria |
| 1. Males and females 21 to 65 years of age |
| 2. BMI > 18 kg/m2 to <35 kg/m2 |
| 3. Wash-out period for supplements affecting immune function |
| 4. Females agree to use of appropriate birth control |
| 5. Consistent diet and lifestyle routine |
| 6. Abstain from exercising, tobacco use, and supplements at study visits |
| 7. Abstain from music, computer/cell phone use at study visits |
| 8. Abstain from consuming candy, chewing gum at study visits |
| 9. Abstain from caffeine for 1 h prior to and at study visits |
| 10. Participated in an endurance exercise for 1.5–3 h/day for 5–6 days per week |
| 11. Healthy as determined by laboratory results and medical history |
| 12. Willingness to perform study procedures and to complete all clinic visits. |
| 13. Has given voluntary, written, informed consent |
| Exclusion Criteria |
| 1. Women who were pregnant or breastfeeding |
| 2. Previous major gastrointestinal surgery or digestive disorder a. inflammatory bowel disease, irritable bowel syndrome, chronic constipation, and history of chronic diarrhea; history of surgery for weight loss, gastroparesis, or clinically important lactose intolerance; and chronic GI illness. |
| 3. Consumed beta-glucan supplements and unwilling to wash out for 4 weeks |
| 4. Consumption of anti-inflammatory medications (81 mg aspirin is acceptable) |
| 5. Upper respiratory tract infection at baseline |
| 6. Antibiotics within 4 weeks of screening |
| 7. Chronic inflammatory condition |
| 8. Type I or Type II diabetes or clinically important renal, hepatic, cardiac, pulmonary, pancreatic, neurologic, or biliary disorder, or a recent history of cancer other than non-melanoma skin cancer. |
| 9. Current use of antipsychotic medications |
| 10. Allergies, chronic bronchitis, asthma, or wheezing |
| 11. Auto-immune disorders |
| 12. Unusual sleep routine |
| 13. Use of immunomodulators |
| 14. Consumed supplements affecting immune function |
| 15. Chronic use of Antacids and Proton Pump Inhibitors |
| 16. Prebiotics and Probiotics |
| 17. Unwilling to have blood drawn |
| 18. Diagnosed depression in the 2 years prior to screening |
| 19. Eating disorders or extreme dietary habits |
| 20. Use of marijuana |
| 21. Active infection or signs/symptoms of an acute infection at study visits. |
| 22. Heavy use of tobacco |
| 23. Consumption of ≥ 14 drinks per week |
| 24. Alcohol or drug abuse within the last 2 years |
| 25. Blood donation during the study or within 30 days of completing the study |
| 26. Unwilling to comply with study procedures and study product consumption |
| 27. Known allergy to the test material’s active or inactive ingredients |
| 28. Unstable medical conditions as assessed by the QI |
| 29. Clinically significant abnormal laboratory results at screening |
| 30. Participation in a clinical research trial within 30 days prior to randomization |
| Parameter | BG n = 13 | PLA n = 14 | Between Group p-Value * |
|---|---|---|---|
| Age (years) | |||
| Mean +/− SEM | 46.77 ± 3.67 (13) | 42.93 ± 3.29 (14) | 0.4346 |
| Gender (n (%)) | 0.8632 | ||
| Female | 7 (53.8%) | 8 (57.1%) | |
| Male | 6 (46.2%) | 6 (42.9%) | |
| Ethnicity (n (%)) | |||
| Eastern European White | 2 (15.4%) | 1 (7.1%) | 0.3933 |
| South Asian | 1 (7.7%) | 0 (0%) | |
| Western European White | 10 (76.9%) | 13 (92.9%) |
| Symptom | Interval | BG n = 13 n (%) | PLA n = 14 n (%) | Between Group p-Value * |
|---|---|---|---|---|
| Very Mild | Days 1–30 | 53.8% (7) | 85.7% (12) | 0.1032 |
| Days 1–90 | 76.9% (10) | 100.0% (14) | 0.0978 | |
| Mild | Days 1–30 | 30.8% (4) | 57.1% (8) | 0.2519 |
| Days 1–90 | 46.2% (6) | 64.3% (9) | 0.4495 | |
| Moderate | Days 1–30 | 0.0% (0) | 21.4% (3) | 0.2222 |
| Days 1–90 | 15.4% (2) | 28.6% (4) | 0.6483 | |
| Severe | Days 1–30 | 7.7% (1) | 14.3% (2) | 1.0000 |
| Days 1–90 | 15.4% (2) | 14.3% (2) | 1.0000 | |
| Any | Days 1–30 | 69.2% (9) | 85.7% (12) | 0.3845 |
| Days 1–90 | 92.3% (12) | 100.0% (14) | 0.4815 |
| Parameter | Study Day | BG n = 17 Mean ± SEM (n) | PLA n = 17 Mean ± SEM (n) | Between Group p-Value * | Reference Value |
|---|---|---|---|---|---|
| Creatinine (µmol/L) | Screening | 82.82 ± 4.43 (17) | 80.00 ± 3.41 (17) | 0.6169 | 44–97 |
| Day 90 | 78.81 ± 4.32 (16) | 79.59 ± 3.26 (17) | |||
| Change from Screening to Day 90 | −2.69 ± 1.82 (16) | −0.41 ± 1.33 (17) | 0.3510 | ||
| eGFR (mL/min/1.73) | Screening | 86.59 ± 4.05 (17) | 89.24 ± 3.33 (17) | 0.6172 | 60–120 |
| Day 90 | 89.56 ± 4.51 (16) | 87.24 ± 3.18 (17) | |||
| Change from Screening to Day 90 | 2.25 ± 1.91 (16) | −2.00 ± 1.57 (17) | 0.1075 | ||
| Sodium (mmol/L) | Screening | 142.00 ± 0.57 (17) | 142.35 ± 0.50 (17) | 0.6442 | 133–148 |
| Day 90 | 142.06 ± 0.66 (16) | 141.76 ± 0.41 (17) | |||
| Change from Screening to Day 90 | 0.19 ± 0.73 (16) | −0.59 ± 0.56 (17) | 0.5635 | ||
| Potassium (mmol/L) | Screening | 4.79 ± 0.09 (17) | 4.62 ± 0.13 (15) | 0.2944 | 3.3–5.7 |
| Day 90 | 4.81 ± 0.09 (16) | 4.36 ± 0.08 (17) | |||
| Change from Screening to Day 90 | −0.01 ± 0.16 (16) | −0.27 ± 0.10 (15) | 0.0038 | ||
| Chloride (mmol/L) | Screening | 101.88 ± 0.62 (17) | 102.82 ± 0.46 (17) | 0.2319 | 98–115 |
| Day 90 | 101.56 ± 0.57 (16) | 102.35 ± 0.36 (17) | |||
| Change from Screening to Day 90 | −0.31 ± 0.77 (16) | −0.47 ± 0.39 (17) | 0.4314 | ||
| Total Bilirubin (µmol/L) | Screening | 7.64 ± 0.92 (17) | 10.00 ± 1.67 (17) | 0.5192 (r) | ≤25 |
| Day 90 | 9.31 ± 4.67 (16) | 11.24 ± 1.65 (17) | |||
| Change from Screening to Day 90 | 1.57 ± 1.14 (16) | 1.24 ± 0.68 (17) | 0.9531 | ||
| Aspartate Transaminase (AST) (U/L) | Screening | 25.41 ± 1.62 (17) | 19.40 ± 1.32 (15) | 0.0081 | 7–70 |
| Day 90 | 21.75 ± 1.82 (16) | 28.18 ± 9.01 (17) | |||
| Change from Screening to Day 90 | −3.19 ± 1.90 (16) | 9.87 ± 9.47 (15) | 0.1107 (r) | ||
| Alanine Transaminase (ALT) (U/L) | Screening | 25.82 ± 0.77 (17) | 17.65 ± 1.18 (17) | 0.0246 (r) | 12–90 |
| Day 90 | 19.49 ± 2.27 (16) | 20.76 ± 2.64 (17) | |||
| Change from Screening to Day 90 | −3.76 ± 2.06 (16) | 3.12 ± 2.35 (17) | 0.0990 (r) | ||
| Calcium (mmol/L) | Screening | 2.36 ± 0.01 (17) | 2.35 ± 0.02 (17) | 0.9277 | 1.8–3.0 |
| Day 90 | 2.41 ± 0.02 (16) | 2.38 ± 0.02 (17) | |||
| Change from Screening to Day 90 | 0.06 ± 0.015 (16) | 0.02 ± 0.02 (17) | 0.1735 |
| Parameter | Study Day | BG n = 17 Mean ± SEM (n) | PLA n = 17 Mean ± SEM (n) | Between Group p-Value * | Reference Value |
|---|---|---|---|---|---|
| Hemoglobin (g/L) | Screening | 134.94 ± 2.54 (17) | 139.53 ± 2.96 (17) | 0.2482 | 135–175 (M), 120–160 (F) |
| Day 90 | 137.75 ± 2.12 (16) | 141.12 ± 2.66 (17) | |||
| Change from Screening to Day 90 | 4.13 ± 1.97 (16) | 1.59 ± 1.59 (17) | 0.7404 | ||
| Hematocrit (L/L) | Screening | 0.40 ± 0.007 (17) | 0.42 ± 0.009 (17) | 0.1809 | 0.4–0.5 (M), 0.35–0.45 (F) |
| Day 90 | 0.41 ± 0.005 (16) | 0.42 ± 0.007 (17) | |||
| Change from Screening to Day 90 | 0.01 ± 0.005 (16) | 0.01 ± 0.005 (17) | 0.9021 | ||
| White Blood Cell Count (×109/L) | Screening | 5.91 ± 0.33 (17) | 5.59 ± 0.30 (17) | 0.4787 | 4.0–10.0 |
| Day 90 | 5.44 ± 0.29 (16) | 5.12 ± 0.31 (17) | |||
| Change from Screening to Day 90 | −0.49 ± 0.28 (16) | −0.48 ± 0.27 (17) | 0.6987 | ||
| Red Blood Cell Count (×1012/L) | Screening | 4.50 ± 0.09 (17) | 4.69 ± 0.10 (17) | 0.1797 | 4.5–6.0 (M), 4.0–5.1 (F) |
| Day 90 | 4.62 ± 0.065 (16) | 4.72 ± 0.09 (17) | |||
| Change from Screening to Day 90 | 0.13 ± 0.065 (16) | 0.04 ± 0.053 (17) | 0.7215 | ||
| MCV (fl) | Screening | 89.53 ± 0.71 (17) | 89.59 ± 1.02 (17) | 0.9626 | 80–100 |
| Day 90 | 89.19 ± 0.79 (16) | 89.82 ± 0.96 (17) | |||
| Change from Screening to Day 90 | −0.06 ± 0.335 (16) | 0.24 ± 0.36 (17) | 0.5145 | ||
| MCH (pg) | Screening | 30.01 ± 0.35 (17) | 29.81 ± 0.33 (17) | 0.6814 | 27.5–33.0 |
| Day 90 | 29.83 ± 0.28 (16) | 29.90 ± 0.34 (17) | |||
| Change from Screening to Day 90 | 0.01 ± 0.14 (16) | 0.09 ± 0.01 (17) | 0.5950 | ||
| MCHC (g/L) | Screening | 334.71 ± 2.07 (17) | 333.59 ± 1.92 (17) | 0.6945 | 305–360 |
| Day 90 | 334.38 ± 1.58 (16) | 333.29 ± 1.67 (17) | |||
| Change from Screening to Day 90 | 0.63 ± 1.85 (16) | −0.29 ± 1.71 (17) | 0.6190 | ||
| RDW (%) | Screening | 13.44 ± 0.59 (17) | 13.61 ± 0.56 (17) | 0.4088 | 11.5–14.5 |
| Day 90 | 13.41 ± 0.80 (16) | 13.58 ± 0.54 (17) | |||
| Change from Screening to Day 90 | −0.01 ± 0.15 (16) | −0.03 ± 0.075 (17) | 0.3380 (r) | ||
| Platelets (×109/L) | Screening | 262.53 ± 14.94 (17) | 243.18 ± 11.69 (17) | 0.3153 | 150–00 |
| Day 90 | 244.63 ± 11.70 (16) | 242.71 ± 13.08 (17) | |||
| Change from Screening to Day 90 | −21.94 ± 11.56 (16) | −0.47 ± 6.76 (17) | 0.2571 | ||
| Absolute: Neutrophils (×109/L) | Screening | 3.28 ± 0.27 (17) | 3.29 ± 0.24 (17) | 0.9738 | 2.0–7.5 |
| Day 90 | 3.07 ± 0.24 (16) | 2.93 ± 0.24 (17) | |||
| Change from Screening to Day 90 | −0.23 ± 0.26 (16) | −0.36 ± 0.22 (17) | 0.6352 | ||
| Absolute: Lymphocytes (×109/L) | Screening | 1.96 ± 0.12 (17) | 1.73 ± 0.09 (17) | 0.1303 | 1.0–3.5 |
| Day 90 | 1.78 ± 0.11 (16) | 1.63 ± 0.11 (17) | |||
| Change from Screening to Day 90 | −0.20 ± 0.01 (16) | −0.10 ± 0.08 (17) | 0.8927 | ||
| Absolute: Monocytes (×109/L) | Screening | 0.52 ± 0.05 (17) | 0.41 ± 0.03 (17) | 0.1665 (r) | 0.2–1.0 |
| Day 90 | 0.44 ± 0.04 (16) | 0.41 ± 0.03 (17) | |||
| Change from Screening to Day 90 | −0.07 ± 0.04 (16) | −0.01 ± 0.03 (17) | 0.7287 | ||
| Absolute: Eosinophils (×109/L) | Screening | 0.15 ± 0.03 (17) | 0.14 ± 0.02 (17) | 1.0000 (r) | 0.0–0.5 |
| Day 90 | 0.13 ± 0.02 (16) | 0.13 ± 0.02 (17) | |||
| Change from Screening to Day 90 | −0.01 ± 0.02 (16) | −0.01 ± 0.02 (17) | 0.7022 (r) | ||
| Absolute: Basophils (×109/L) | Screening | 0.01 ± 0.007 (17) | 0.01 ± 0.004 (17) | 0.5595 (r) | 0.0–0.2 |
| Day 90 | 0.01 ± 0.008 (16) | 0.01 ± 0.004 (17) | |||
| Change from Screening to Day 90 | 0.00 ± 0.01 (16) | 0.00 ± 0.01 (17) | 0.4850 (r) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Evans, M.; Falcone, P.H.; Crowley, D.C.; Sulley, A.M.; Campbell, M.; Zakaria, N.; Lasrado, J.A.; Fritz, E.P.; Herrlinger, K.A. Effect of a Euglena gracilis Fermentate on Immune Function in Healthy, Active Adults: A Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients 2019, 11, 2926. https://doi.org/10.3390/nu11122926
Evans M, Falcone PH, Crowley DC, Sulley AM, Campbell M, Zakaria N, Lasrado JA, Fritz EP, Herrlinger KA. Effect of a Euglena gracilis Fermentate on Immune Function in Healthy, Active Adults: A Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients. 2019; 11(12):2926. https://doi.org/10.3390/nu11122926
Chicago/Turabian StyleEvans, Malkanthi, Paul H. Falcone, David C. Crowley, Abdul M. Sulley, Marybelle Campbell, Nisrine Zakaria, Joanne A. Lasrado, Emily Pankow Fritz, and Kelli A. Herrlinger. 2019. "Effect of a Euglena gracilis Fermentate on Immune Function in Healthy, Active Adults: A Randomized, Double-Blind, Placebo-Controlled Trial" Nutrients 11, no. 12: 2926. https://doi.org/10.3390/nu11122926
APA StyleEvans, M., Falcone, P. H., Crowley, D. C., Sulley, A. M., Campbell, M., Zakaria, N., Lasrado, J. A., Fritz, E. P., & Herrlinger, K. A. (2019). Effect of a Euglena gracilis Fermentate on Immune Function in Healthy, Active Adults: A Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients, 11(12), 2926. https://doi.org/10.3390/nu11122926





