β2 Adrenergic Regulation of the Phagocytic and Microbicide Capacity of Macrophages from Obese and Lean Mice: Effects of Exercise
Abstract
:1. Introduction
2. Methods
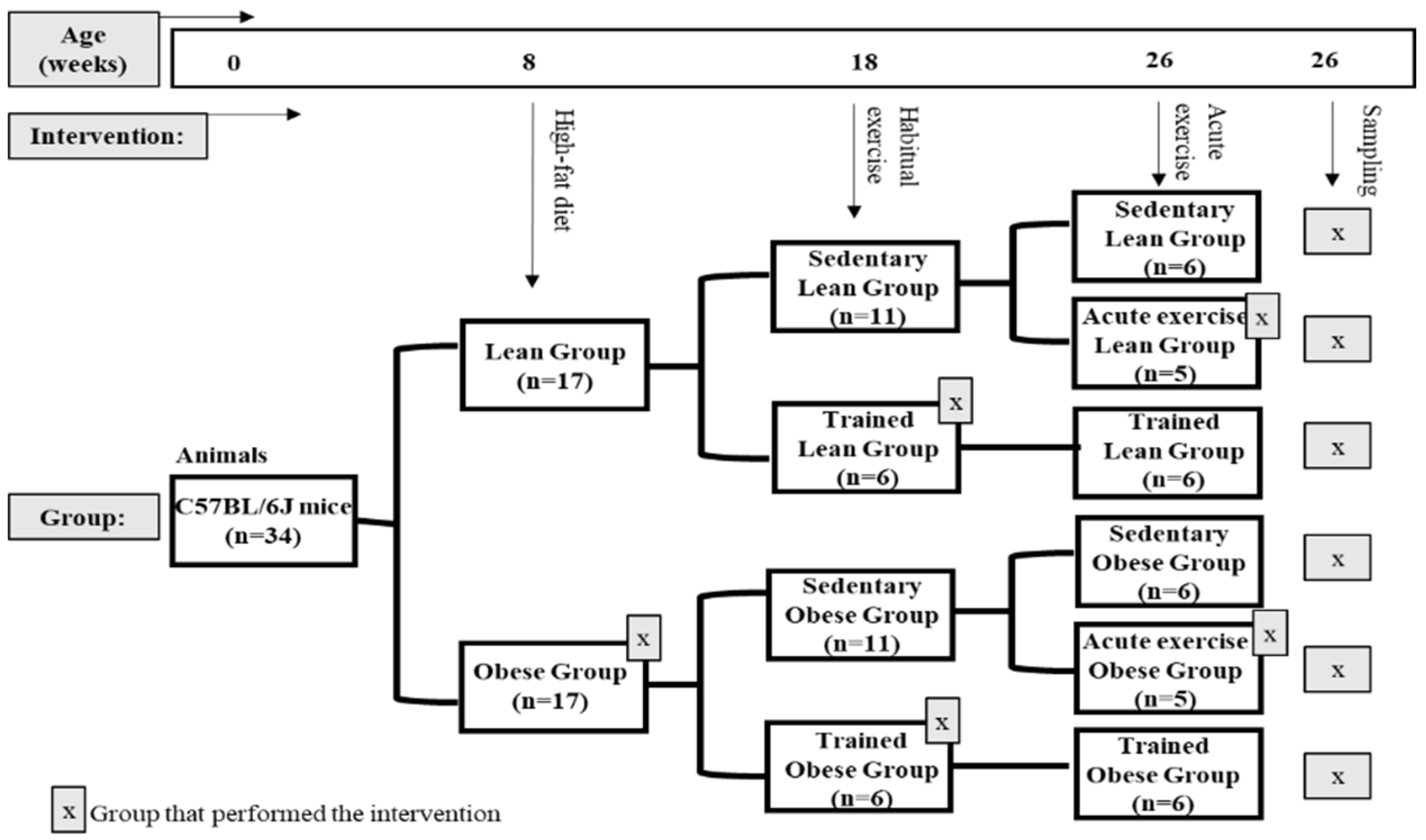
2.1. Animals and Experimental Design
2.2. Exercise
2.3. Anaesthesia
2.4. Collection of Biological Samples, Cell Culture, and Incubation
2.5. Phagocytic and Oxidative Burst Assays via Flow Cytometer
2.6. β2-Adrenergic Receptor Expression Assays via Flow Cytometer
2.7. Flow Cytometry Assays
2.8. Statistical Analysis
3. Results
3.1. Body Weight, Dietary Intake, and Fasting Blood Glucose and Lipid Profile. Effect of Exercise
3.2. Effect of β2-Adrenergic Activation by Terbutaline on the Phagocytic and Microbicide Capacities of Peritoneal Macrophages from Lean and Obese Mice
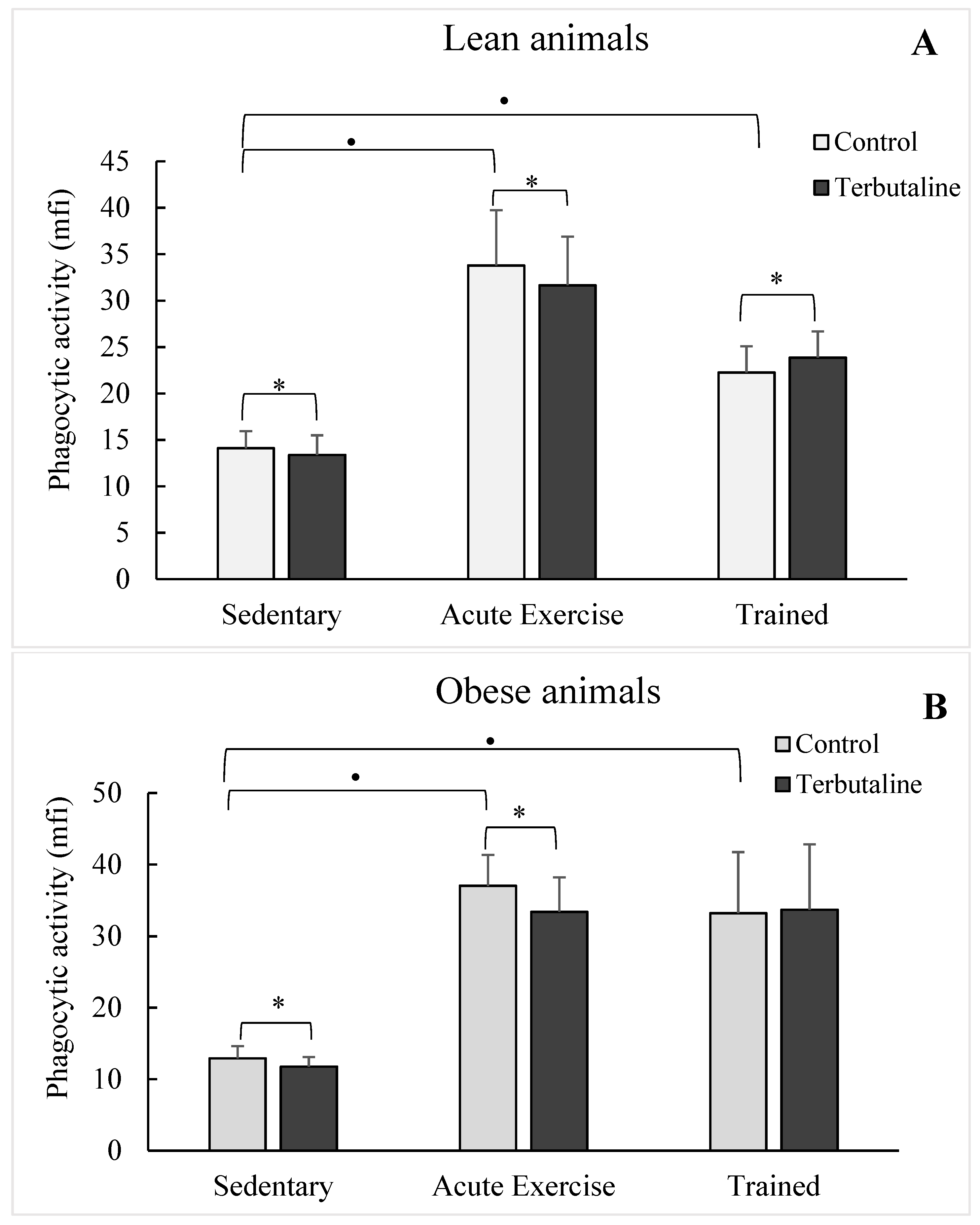
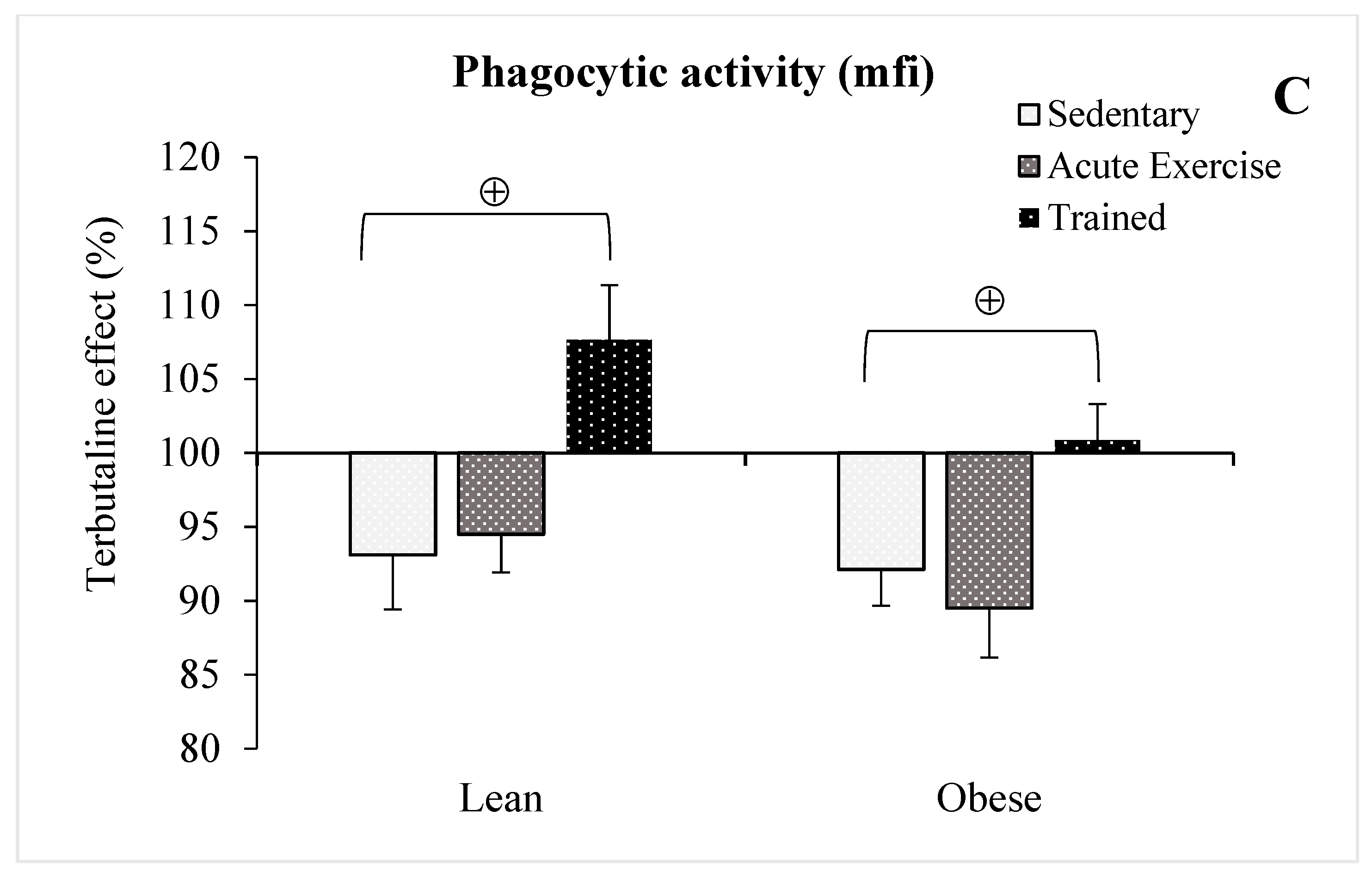
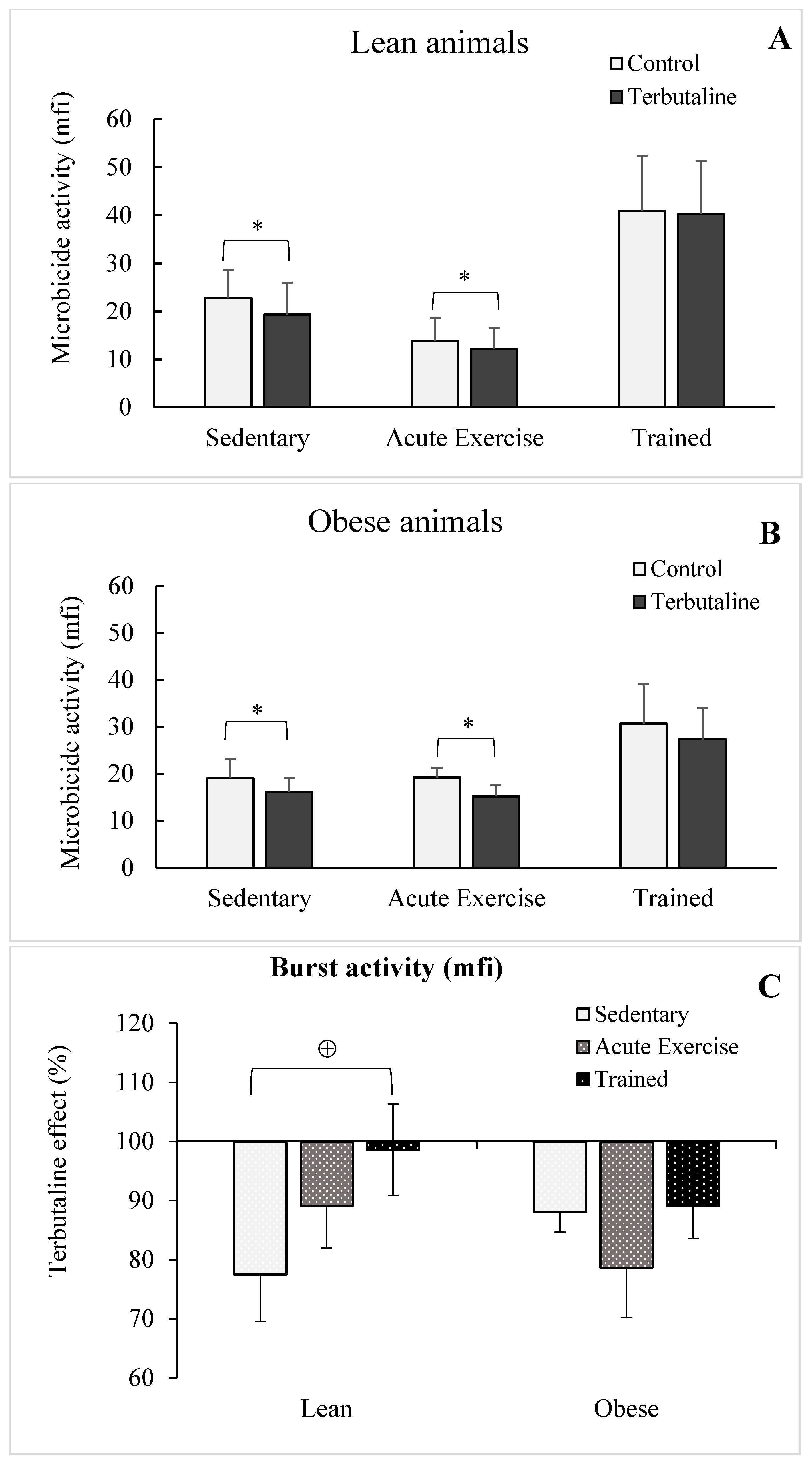
3.3. Effect of Exercise in the β2-Adrenergic Regulation of the Phagocytic Capacity of Peritoneal Macrophages from Obese and Lean Mice
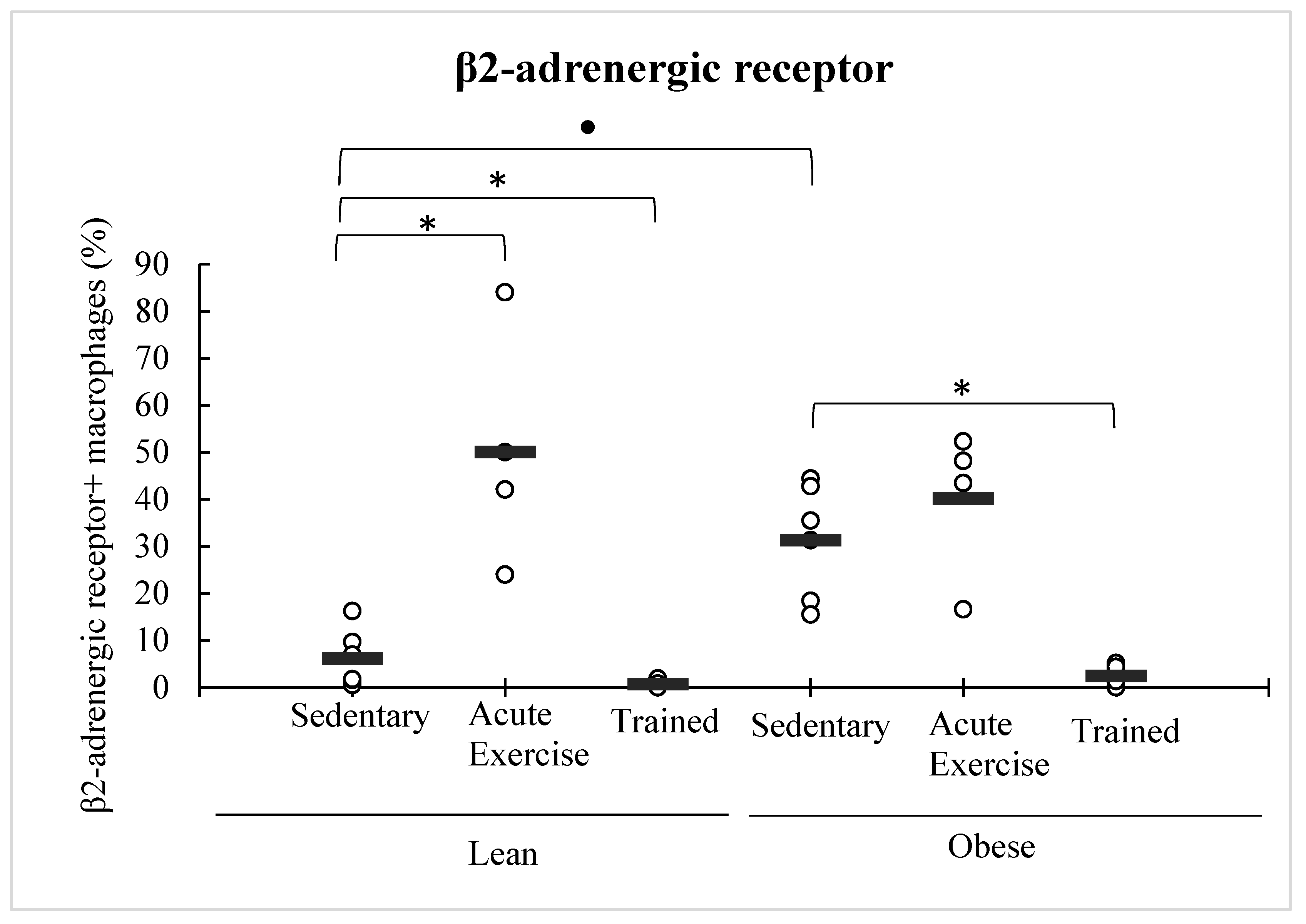
3.4. Effect of Obesity and Exercise in the β2-Adrenergic Receptor Expression in Peritoneal Macrophages
4. Discussion
5. Conclusions
- The β2-AR agonist terbutaline decreases the phagocytic and microbicide capacities of peritoneal macrophages in sedentary animals, and obesity does not alter this β2-AR neuroimmunomodulation of macrophages.
- Regular exercise causes an adaptation in the response of peritoneal macrophages to the β2-AR agonist terbutaline, by inducing a stimulatory effect in the innate response of macrophages. Obesity seems to hinder this immunophysiological adaptation to regular exercise. This effect is contrary to those produced in both sedentary and acute exercise lean and obese animals.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Medzhitov, R. Origin and Physiological Roles of Inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Li, J.; Zhu, Y.; Sun, H.; Zhao, H.; Shao, B.; Li, Y. Effects of Aluminum Exposure on the Adherence, Chemotaxis, and Phagocytosis Capacity of Peritoneal Macrophages in Rats. Biol. Trace Elem. Res. 2011, 144, 1032–1038. [Google Scholar] [CrossRef] [PubMed]
- Martín-Cordero, L.; García, J.; Ortega, E. Noradrenaline-Mediated Inhibition of Inflammatory Cytokines Is Altered in Macrophages from Obese Zucker Rats: Effect of Habitual Exercise. Endocrine Metab. Immune Disord. Targets 2013, 13, 234–239. [Google Scholar] [CrossRef]
- Ortega, E. Neuroendocrine Mediators in the Modulation of Phagocytosis by Exercise: Physiological Implications. Exerc. Immunol. Rev. 2003, 9, 70–93. [Google Scholar]
- Ortega, E. The “Bioregulatory Effect of Exercise” on the Innate/Inflammatory Responses. J. Physiol. Biochem. 2016, 72, 361–369. [Google Scholar] [CrossRef]
- Ortega, E.; Giraldo, E.; Hinchado, M.D.; Martín, L.; García, J.J.; De La Fuente, M. Neuroimmunomodulation during Exercise: Role of Catecholamines as “stress Mediator” and/or “Danger Signal” for the Innate Immune Response. Neuroimmunomodulation 2007, 14, 206–212. [Google Scholar] [CrossRef]
- Martín-Cordero, L.; García, J.J.; Hinchado, M.D.; Ortega, E. The Interleukin-6 and Noradrenaline Mediated Inflammation-Stress Feedback Mechanism Is Dysregulated in Metabolic Syndrome: Effect of Exercise. Cardiovasc. Diabetol. 2011, 10, 42. [Google Scholar] [CrossRef]
- Gálvez, I.; Martín-Cordero, L.; Hinchado, M.D.; Álvarez-Barrientos, A.; Ortega, E. Anti-Inflammatory Effect of Β2 Adrenergic Stimulation on Circulating Monocytes with a pro-Inflammatory State in High-Fat Diet-Induced Obesity. Brain. Behav. Immun. 2019, 80, 564–572. [Google Scholar] [CrossRef]
- Ortega, E.; Galvez, I.; Martin-Cordero, L. Adrenergic Regulation of Macrophage-Mediated Innate/Inflammatory Responses in Obesity and Exercise in This Condition: Role of B2 Adrenergic Receptors. Endocrine Metab. Immune Disord. Drug Targets 2019, 19. [Google Scholar] [CrossRef]
- Johnson, A.R.; Milner, J.J.; Makowski, L. The Inflammation Highway: Metabolism Accelerates Inflammatory Traffic in Obesity. Immunol. Rev. 2012, 249, 218–238. [Google Scholar] [CrossRef]
- Lumeng, C.N. Innate Immune Activation in Obesity. Mol. Aspects Med. 2013, 34, 12–29. [Google Scholar] [CrossRef] [PubMed]
- Straznicky, N.E.; Lambert, E.A.; Nestel, P.J.; McGrane, M.T.; Dawood, T.; Schlaich, M.P.; Masuo, K.; Eikelis, N.; de Courten, B.; Mariani, J.A.; et al. Sympathetic Neural Adaptation to Hypocaloric Diet with or without Exercise Training in Obese Metabolic Syndrome Subjects. Diabetes 2010, 59, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Straznicky, N.E.; Lambert, G.W.; Lambert, E.A. Neuroadrenergic Dysfunction in Obesity: An Overview of the Effects of Weight Loss. Curr. Opin. Lipidol. 2010, 21, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Martín-Cordero, L.; García, J.J.; Hinchado, M.D.; Bote, E.; Ortega, E. Influence of Exercise on NA- and Hsp72-Induced Release of IFNγ by the Peritoneal Suspension of Macrophages and Lymphocytes from Genetically Obese Zucker Rats. J. Physiol. Biochem. 2013, 69, 125–131. [Google Scholar] [CrossRef]
- Martín-Cordero, L.; Reis, F.; García, J.J.; Teixeira, F.; Ortega, E. Effect of exercise without diet on functional capacity of peritoneal macrophages and TNF-a levels in blood and in adipose tissue in the obese Zucker rat model of the metabolic syndrome. Proc. Nutr. Soc. 2013, 72, E76. [Google Scholar] [CrossRef]
- Chawla, A.; Nguyen, K.D.; Goh, Y.P.S. Macrophage-Mediated Inflammation in Metabolic Disease. Nat. Rev. Immunol. 2011, 11, 738–749. [Google Scholar] [CrossRef]
- Mraz, M.; Haluzik, M. The Role of Adipose Tissue Immune Cells in Obesity and Low-Grade Inflammation. J. Endocrinol. 2014, 222, R113–R127. [Google Scholar] [CrossRef]
- Gleeson, M.; Bishop, N.C.; Stensel, D.J.; Lindley, M.R.; Mastana, S.S.; Nimmo, M.A. The Anti-Inflammatory Effects of Exercise: Mechanisms and Implications for the Prevention and Treatment of Disease. Nat. Rev. Immunol. 2011, 11, 607–615. [Google Scholar] [CrossRef]
- Ortega, E.; Martín-Cordero, L.; Garcia-Roves, P.M.; Chicco, A.J.; Gonzalez-Franquesa, A.; Marado, D. Diabetes Mellitus and Metabolic Syndrome. In Biomarkers of Cardiometabolic Risk, Inflammation and Disease; Palavra, F., Reis, F., Marado, D., Sena, A., Eds.; Springer International Publishing Switzerland: Basel, Switzerland, 2015; pp. 55–80. [Google Scholar]
- Ortega, E.; García, J.J.; Sáez, M.C.; De la Fuente, M. Changes with Aging in the Modulation of Macrophages by Norepinephrine. Mech. Ageing Dev. 2000, 118, 103–114. [Google Scholar] [CrossRef]
- Elenkov, I.J.; Iezzoni, D.G.; Daly, A.; Harris, A.G.; Chrousos, G.P. Cytokine Dysregulation, Inflammation and Well-Being. Neuroimmunomodulation 2005, 12, 255–269. [Google Scholar] [CrossRef]
- Khan, M.M.; Sansoni, P.; Silverman, E.D.; Engleman, E.G.; Melmon, K.L. Beta-Adrenergic Receptors on Human Suppressor, Helper, and Cytolytic Lymphocytes. Biochem. Pharmacol. 1986, 35, 1137–1142. [Google Scholar] [CrossRef]
- Bosmann, M.; Grailer, J.J.; Zhu, K.; Matthay, M.A.; Sarma, J.V.; Zetoune, F.S.; Ward, P.A. Anti-Inflammatory Effects of Β2 Adrenergic Receptor Agonists in Experimental Acute Lung Injury. FASEB J. 2012, 26, 2137–2144. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Su, H.; Zhang, Q.; Tang, H.; Wang, C.; Zhou, Q.; Wei, W.; Zhu, H.; Wang, Y. Melatonin Attenuates Aortic Endothelial Permeability and Arteriosclerosis in Streptozotocin-Induced Diabetic Rats. J. Cardiovasc. Pharmacol. Ther. 2016, 21, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Assis de Brito, T.L.; Monte-Alto-Costa, A.; Romana-Souza, B. Propranolol Impairs the Closure of Pressure Ulcers in Mice. Life Sci. 2014, 100, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Ağaç, D.; Estrada, L.D.; Maples, R.; Hooper, L.V.; Farrar, J.D. The Β2-Adrenergic Receptor Controls Inflammation by Driving Rapid IL-10 Secretion. Brain. Behav. Immun. 2018, 74, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.-W.; Choi, H.-H.; Chung, Y.-R. Treadmill Exercise Alleviates Impairment of Cognitive Function by Enhancing Hippocampal Neuroplasticity in the High-Fat Diet-Induced Obese Mice. J. Exerc. Rehabil. 2016, 12, 156–162. [Google Scholar] [CrossRef]
- Petrosino, J.M.; Heiss, V.J.; Maurya, S.K.; Kalyanasundaram, A.; Periasamy, M.; LaFountain, R.A.; Wilson, J.M.; Simonetti, O.P.; Ziouzenkova, O. Graded Maximal Exercise Testing to Assess Mouse Cardio-Metabolic Phenotypes. PLoS ONE 2016, 11, e0148010. [Google Scholar] [CrossRef]
- Strandberg, L.; Verdrengh, M.; Enge, M.; Andersson, N.; Amu, S.; Onnheim, K.; Benrick, A.; Brisslert, M.; Bylund, J.; Bokarewa, M.; et al. Mice Chronically Fed High-Fat Diet Have Increased Mortality and Disturbed Immune Response in Sepsis. PLoS ONE 2009, 4, e7605. [Google Scholar] [CrossRef]
- Amar, S.; Zhou, Q.; Shaik-Dasthagirisaheb, Y.; Leeman, S. Diet-Induced Obesity in Mice Causes Changes in Immune Responses and Bone Loss Manifested by Bacterial Challenge. Proc. Natl. Acad. Sci. USA 2007, 104, 20466–20471. [Google Scholar] [CrossRef]
- Galvan, D.L.; Danesh, F.R. Β2-Adrenergic Receptors in Inflammation and Vascular Complications of Diabetes. Kidney Int. 2017, 92, 14–16. [Google Scholar] [CrossRef]
- Noh, H.; Yu, M.R.; Kim, H.J.; Lee, J.H.; Park, B.-W.; Wu, I.-H.; Matsumoto, M.; King, G.L. Beta 2-Adrenergic Receptor Agonists Are Novel Regulators of Macrophage Activation in Diabetic Renal and Cardiovascular Complications. Kidney Int. 2017, 92, 101–113. [Google Scholar] [CrossRef] [PubMed]
- García, J.J.; del Carmen Sáez, M.; De la Fuente, M.; Ortega, E. Regulation of Phagocytic Process of Macrophages by Noradrenaline and Its End Metabolite 4-Hydroxy-3-Metoxyphenyl-Glycol. Role of Alpha- and Beta-Adrenoreceptors. Mol. Cell. Biochem. 2003, 254, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Ortega, E.; Giraldo, E.; Hinchado, M.D.; Martín-Cordero, L.; García, J.J. 72 KDa Extracellular Heat Shock Protein (EHsp72), Norepinephrine (NE), and the Innate Immune Response Following Moderate Exercise. In Heat Shock Proteins and Whole Body Physiology; Asea, A., Pederser, B.-K., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 327–350. [Google Scholar]
- Ortega, E.; Collazos, M.E.; Barriga, C.; de la Fuente, M. Effect of Physical Activity Stress on the Phagocytic Process of Peritoneal Macrophages from Old Guinea Pigs. Mech. Ageing Dev. 1992, 65, 157–165. [Google Scholar] [CrossRef]
- Ortega, E.; Forner, M.A.; Barriga, C.; De La Fuente, M. Effect of Age and of Swimming-Induced Stress on the Phagocytic Capacity of Peritoneal Macrophages from Mice. Mech. Ageing Dev. 1993, 70, 53–63. [Google Scholar] [CrossRef]
- Ortega Rincón, E. Physiology and Biochemistry: Influence of Exercise on Phagocytosis. Int. J. Sports Med. 1994, 15 (Suppl. 3), S172–S178. [Google Scholar] [CrossRef]
- Walsh, N.P.; Gleeson, M.; Shephard, R.J.; Gleeson, M.; Woods, J.A.; Bishop, N.C.; Fleshner, M.; Green, C.; Pedersen, B.K.; Hoffman-Goetz, L.; et al. Position statement. Part one: Immune function and exercise. Exerc. Immunol. Rev. 2011, 17, 6–63. [Google Scholar]
- Leite, F.; Lima, M.; Marino, F.; Cosentino, M.; Ribeiro, L. Β2 Adrenoceptors Are Underexpressed in Peripheral Blood Mononuclear Cells and Associated with a Better Metabolic Profile in Central Obesity. Int. J. Med. Sci. 2017, 14, 853–861. [Google Scholar] [CrossRef]
- Landmann, R.; Portenier, M.; Staehelin, M.; Wesp, M.; Box, R. Changes in Beta-Adrenoceptors and Leucocyte Subpopulations after Physical Exercise in Normal Subjects. Naunyn. Schmiedebergs. Arch. Pharmacol. 1988, 337, 261–266. [Google Scholar] [CrossRef]
- Burman, K.D.; Ferguson, E.W.; Djuh, Y.-Y.; Wartofsky, L.; Latham, K. Beta Receptors in Peripheral Mononuclear Cells Increase Acutely during Exercise. Acta Endocrinol. (Copenh.) 1985, 109, 563–568. [Google Scholar] [CrossRef]
- Hunsche, C.; Hernandez, O.; De la Fuente, M. Impaired Immune Response in Old Mice Suffering from Obesity and Premature Immunosenescence in Adulthood. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2016, 71, 983–991. [Google Scholar] [CrossRef]
- Saez, M.C.; Garcia, J.J.; De la Fuente, M.; Ortega, E. Modulation of Superoxide Anion Levels of Macrophages from Young–Adult and Old Mice by the Norepinephrine Metabolite, 4-Hydroxy-3-Methoxyphenyl-Glycol. Exp. Gerontol. 2002, 37, 395–400. [Google Scholar] [CrossRef]
- Marino, F.; Cosentino, M. Adrenergic Modulation of Immune Cells: An Update. Amino Acids 2013, 45, 55–71. [Google Scholar] [CrossRef] [PubMed]

| Lean | Obese | |||||
|---|---|---|---|---|---|---|
| Sedentary | Acute Exercise | Trained | Sedentary | Acute Exercise | Trained | |
| Body Weight (g) | 29.3 ± 1.2 | 30.5 ± 2 | 25.6 ± 1 * | 42.3 ± 1.15 • | 39.8 ± 2 | 36.0 ± 3.0 * |
| Dietary Intake (g/day) | 4.0 ± 0.1 | 4.2 ± 0.1 | 4.1 ± 0.1 | 2.7 ± 0.1 • | 2.6 ± 0.1 | 2.5 ± 0.03 * |
| Energy Intake (Kj/day) | 55.32 ± 3.1 | 58.2 ± 1.4 | 56.6 ± 0.9 | 62.01 ± 2.7 • | 60.5 ± 1.9 | 57.6 ± 0.7 * |
| Glucose (mg/dL) | 218.9 ± 13.2 | 17 4± 32 * | 196.4 ± 25 | 311 ± 31 • | 222.7 ± 23 * | 282.5 ± 28 |
| Cholesterol (mg/dL) | ||||||
| Total Cholesterol | 103.7 ± 2.2 | <99 † | 106.7 ± 3 | 172.7 ± 19 • | 175 ± 42 | 178.1 ± 25 |
| HDL-C | 42.1 ± 2.9 | 51.7 ± 4 * | 59.7 ± 5.7 • | 55.5 ± 3 | 75.3 ± 4 * | |
| cLDL-C | 50.7 ± 3.5 | 88.0 ± 0 | 39.4 ± 2 * | 88.8 ± 16 • | 78 ± 12 | 38.5 ± 1 * |
| Triglycerides (mg/dL) | 86.8 ± 1.9 | 88.0 ± 0 | 76.6 ± 1 * | 91.5 ± 2 • | 98.7 ± 7 | 80 ± 1 * |
| Control | Terbutaline | Terbutaline + Butaxamine | ||
|---|---|---|---|---|
| Phagocytic percentage | lean | 85.3 ± 4 | 79.6 ± 3 * | 80.2 ± 4 |
| obese | 77.6 ± 5 | 71.9 ± 3 * | 78.3 ± 4 • | |
| Microbicide percentage | lean | 65.8 ± 7 | 64.9 ± 7 | 64.2 ± 8 |
| obese | 73.1 ± 8 | 67.6 ± 8 * | 67.7 ± 8 | |
| Sedentary | Acute Exercise | Trained | |||||
|---|---|---|---|---|---|---|---|
| Control | Terbutaline | Control | Terbutaline | Control | Terbutaline | ||
| Phagocytic percentage | lean | 85.3 ± 4 | 79.6 ± 3 * | 61.9 ± 5 • | 57.4 ± 6 * | 68.6 ± 11 | 67.9 ± 10 |
| obese | 77.6 ± 5 | 71.9 ± 3 * | 64.9 ± 1 • | 61.6 ± 2 * | 65.4 ± 1 | 65.7 ± 11 | |
| Microbicide percentage | lean | 65.8 ± 7 | 64.9 ± 7 | 57.7 ± 2 | 54.3 ± 15 | 71.7 ± 5 | 71.8 ± 6 |
| obese | 73.1 ± 8 | 67.6 ± 8 * | 74.8 ± 7 | 65.1 ± 5 * | 62.1 ± 6 | 61.9 ± 7 | |
| Without Terbutaline | With Terbutaline | ||||
|---|---|---|---|---|---|
| Sedentary | Acute Exercise | Trained | |||
| Phagocytic percentage | lean | 100 | 93.5 ± 2 | 92.4 ± 2 | 99.1 ± 3 • |
| obese | 100 | 93.3 ± 3 | 94.9 ± 3 | 100.5 ± 2 • | |
| Microbicide percentage | lean | 100 | 98.2 ± 4 | 93.3 ± 3 | 100.5 ± 4 |
| obese | 100 | 91.1 ± 4 | 87.5 ± 4 | 99.2 ± 3 • | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martín-Cordero, L.; Gálvez, I.; Hinchado, M.D.; Ortega, E. β2 Adrenergic Regulation of the Phagocytic and Microbicide Capacity of Macrophages from Obese and Lean Mice: Effects of Exercise. Nutrients 2019, 11, 2721. https://doi.org/10.3390/nu11112721
Martín-Cordero L, Gálvez I, Hinchado MD, Ortega E. β2 Adrenergic Regulation of the Phagocytic and Microbicide Capacity of Macrophages from Obese and Lean Mice: Effects of Exercise. Nutrients. 2019; 11(11):2721. https://doi.org/10.3390/nu11112721
Chicago/Turabian StyleMartín-Cordero, Leticia, Isabel Gálvez, María Dolores Hinchado, and Eduardo Ortega. 2019. "β2 Adrenergic Regulation of the Phagocytic and Microbicide Capacity of Macrophages from Obese and Lean Mice: Effects of Exercise" Nutrients 11, no. 11: 2721. https://doi.org/10.3390/nu11112721
APA StyleMartín-Cordero, L., Gálvez, I., Hinchado, M. D., & Ortega, E. (2019). β2 Adrenergic Regulation of the Phagocytic and Microbicide Capacity of Macrophages from Obese and Lean Mice: Effects of Exercise. Nutrients, 11(11), 2721. https://doi.org/10.3390/nu11112721






