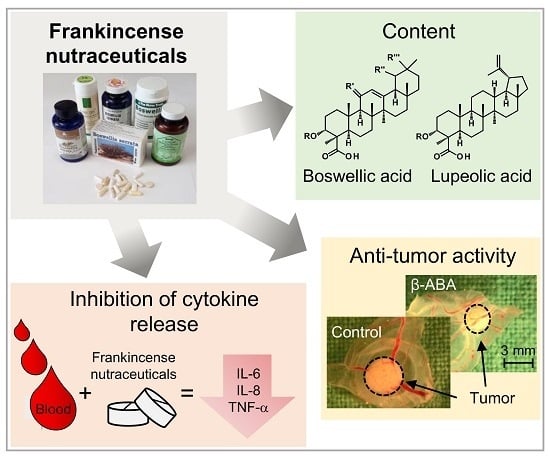
Comparative Investigation of Frankincense Nutraceuticals: Correlation of Boswellic and Lupeolic Acid Contents with Cytokine Release Inhibition and Toxicity against Triple-Negative Breast Cancer Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Quantification of Boswellic and Lupeolic Acids by HPLC-MS/MS Analysis
2.3. Analysis of Cytokine Release
2.4. Analysis of Antiproliferative and Cytotoxic Effects In Vitro
2.5. Human Tumor Xenografts on the Chick Chorioallantoic Membrane
2.6. Statistical Analysis
3. Results
3.1. Compositions of Boswellic and Lupeolic Acids in Frankincense Nutraceuticals
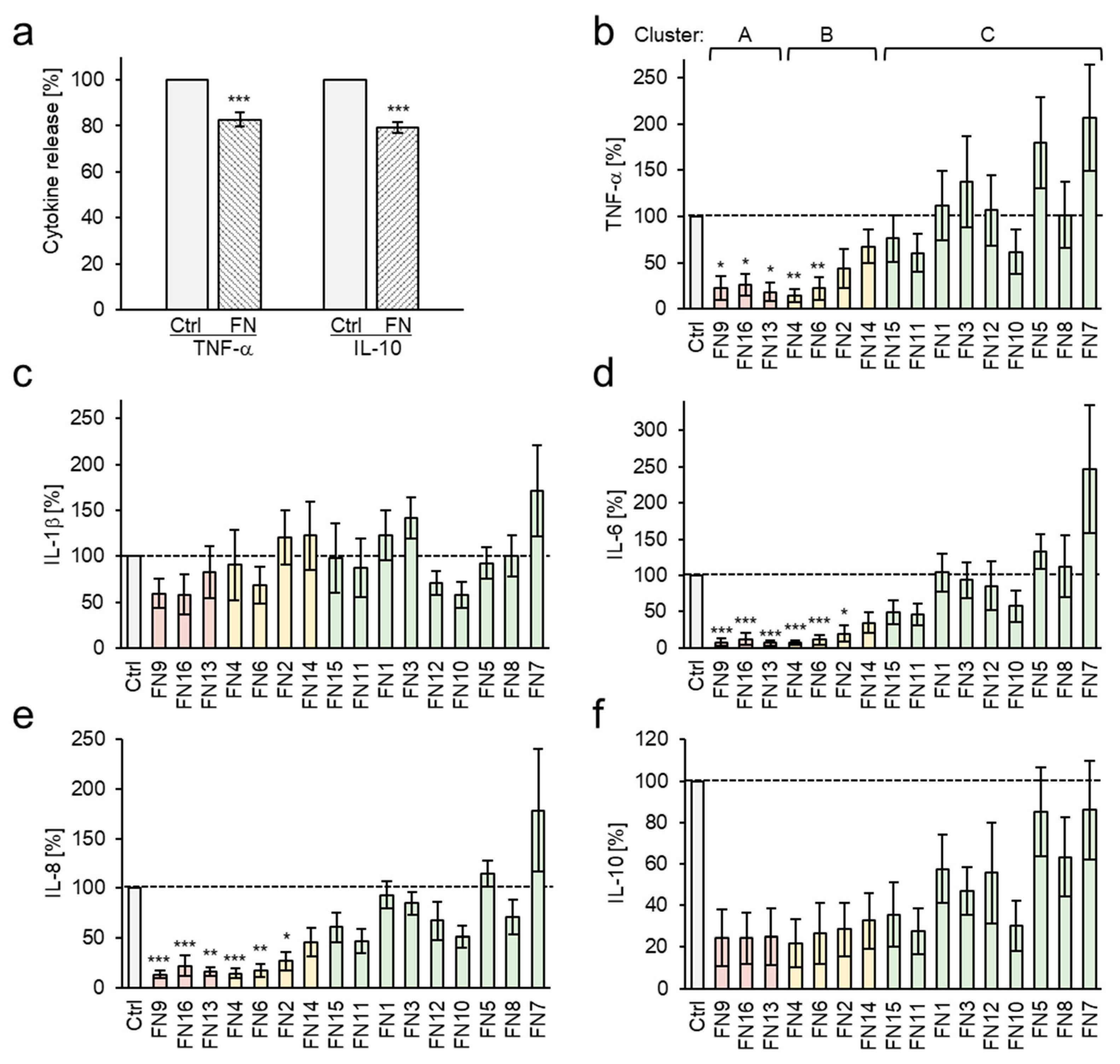
3.2. Inhibition of Proinflammatory Cytokine Release by Frankincense Nutraceuticals
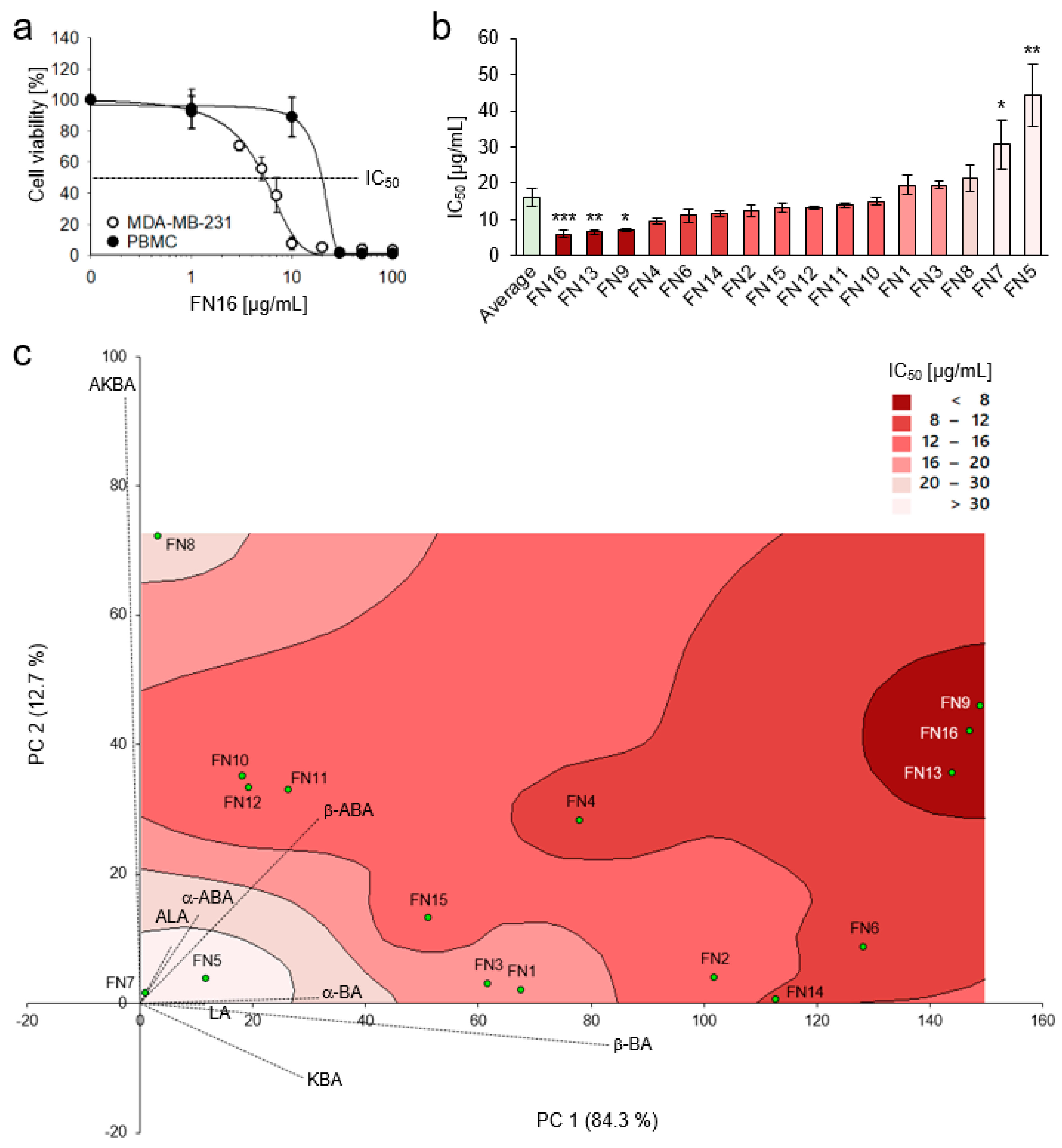
3.3. Cytotoxic Efficacy of Frankincense Nutraceuticals Against Triple-Negative Breast Cancer Cells In Vitro
3.4. Boswellic and Lupeolic Acids Inhibit Cytokine Release and Are Cytotoxic for TNBC Cells
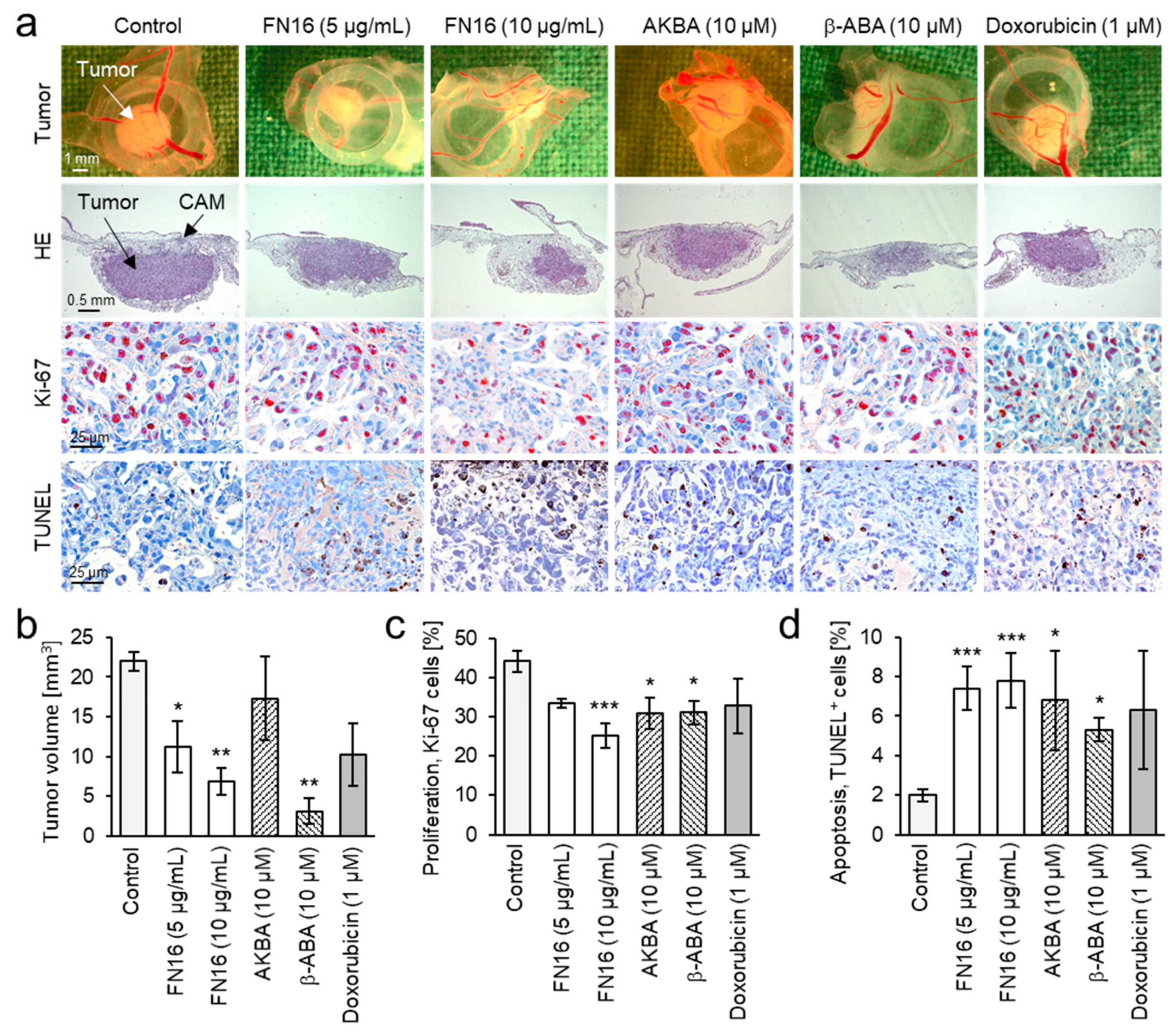
3.5. Inhibition of Proliferation and Induction of Apoptosis in Breast Cancer Xenografts
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| FN Sample | Manufacturer/Distributor | Manufacturer’s Specifications | |||
|---|---|---|---|---|---|
| Product | Batch | Stated Content | RDA 1 | ||
| FN1 | Gall Pharma (Judenburg, Austria) | Boswellia serrata 200 mg GPH Kapseln | 5260.53 | 200 mg B. serrata extract | 2 |
| FN2 | Hecht Pharma (Bremervörde, Germany) | H15® Weihrauch Boswellia serrata 350 mg | 2260.52 | 350 mg B. serrata extract | 1 × 1 |
| FN3 | Hecht Pharma (Bremervörde, Germany) | H15® Weihrauch Kapseln 200 mg | 5260.53 | 200 mg B. serrata extract | 1 × 2 |
| FN4 | Heidelberg Pharmacy (Bisingen, Germany) | Weihrauch-Extrakt-Kapseln | HBMI07 | 300 mg B. serrata extract | 3 × 1–2 |
| FN5 | The Nutri Store (Mauren, Liechtenstein) | Boswellia carterii Extrakt Kapseln | #7779529-1612 | 400 mg B. carterii extract | 1 × 1 |
| FN6 | Wellnest Nutrazeutika (East Grinstead, England) | Boswellia Weihrauchextrakt | NB/BOS-1215189 | 400 mg B. serrata extract; 200 mg B. carterii extract | 2 |
| FN7 | Vitabay (Masstricht, Netherlands) | Weihrauch Extrakt 400 mg | C17030104 | 400 mg B. serrata extract | 2 |
| FN8 | Vitacost (Boca Raton, Florida) | Synergy 5-Loxin® Boswellia serrata Extract | #703657 | 150 mg B. serrata extract | 2 |
| FN9 | Schloss Pharmacy (Koblenz, Germany) | Indische Weihrauchkapseln | 611142B | 400 mg B. serrata extract | |
| FN10 | Heilsteine Methusalem (Neu-Ulm, Germany) | Sacra Weihrauch Gold | WS/102017/NT | 430 mg B. sacra gum resin | 2 × 2 |
| FN11 | Heilsteine Methusalem (Neu-Ulm, Germany) | Weihrauch Boswellia serrata | WBS/2425/12AS | 500 mg B. serrata gum resin | 2 × 1 |
| FN12 | Olibanum B.V. (Kerkrade, Netherlands) | Boscari®, original afrikanischer Weihrauch | B0417 | 400 mg B. carterii gum resin | 3 × 1 |
| FN13 | Biotikon (Gorxheimertal, Germany) | 85 Premium Boswellia serrata | BOSW-200917 | 400 mg B. serrata extract | 1 |
| FN14 | Gall Pharma (Judenburg, Austria) | Boswellia serrata Tabletten 400 mg | 261.29 | 400 mg B. serrata extract | 3 × 1 |
| FN15 | Gufic Biosciences Ltd. (Mumbai, India) | Sallaki® Tablets 400 mg | AB17019 | 400 mg B. serrata extract | |
| FN16 | Delphin Pharmacy (Langenau, Germany) | Weihrauch-Kapseln | BBD 2 05.04.2018 | 400 mg B. serrata extract | |
References
- Moussaieff, A.; Mechoulam, R. Boswellia resin: From religious ceremonies to medical uses; A review of in-vitro, in-vivo and clinical trials. J. Pharm. Pharmacol. 2009, 61, 1281–1293. [Google Scholar] [CrossRef]
- Schrott, E. Weihrauch; Mosaik: Munich, Germany, 1998. [Google Scholar]
- Ammon, H.P. Boswellic acids and their role in chronic inflammatory diseases. Adv. Exp. Med. Biol. 2016, 928, 291–327. [Google Scholar] [CrossRef]
- Kluge, H.; Fernando, C.; Winking, M. Weihrauch und Seine Heilende WIRKUNG; Karl, F., Ed.; Haug Verlag: Heidelberg, Germany, 1998. [Google Scholar]
- Roy, N.K.; Deka, A.; Bordoloi, D.; Mishra, S.; Kumar, A.P.; Sethi, G.; Kunnumakkara, A.B. The potential role of boswellic acids in cancer prevention and treatment. Cancer Lett. 2016, 377, 74–86. [Google Scholar] [CrossRef]
- Bongers, F.; Groenendijk, P.; Bekele, T.; Birhane, E.; Damtew, A.; Decuyper, M.; Eshete, A.; Gezahgne, A.; Girma, A.; Khamis, M.A.; et al. Frankincense in peril. Nat. Sustain. 2019, 2, 602–610. [Google Scholar] [CrossRef]
- Huber, G. Weihrauch; Ansata: Munich, Germany, 2018. [Google Scholar]
- Martinetz, D.; Lohs, K.; Janzen, J. Weihrauch und Myrrhe: Kulturgeschichte und wirtschaftliche Bedeutung, Botanik, Chemie, Medizin; Wissenschaftliche Verlangsgesellschaft mbH Stuttgart: Stuttgart, Germany, 1988. [Google Scholar]
- Belsner, K.; Büchele, B.; Werz, U.; Syrovets, T.; Simmet, T. Structural analysis of pentacyclic triterpenes from the gum resin of Boswellia serrata by NMR spectroscopy. Magn. Reson. Chem. 2003, 41, 115–122. [Google Scholar] [CrossRef]
- Safayhi, H.; Mack, T.H.; Sabieraj, J.O.; Anazodo, M.I.; Subramanian, L.R.; Ammon, H.P. Boswellic acids: Novel, specific, nonredox inhibitors of 5-lipoxygenase. J. Pharmacol. Exp. Ther. 1992, 261, 1143–1146. [Google Scholar] [PubMed]
- Syrovets, T.; Büchele, B.; Krauss, C.; Laumonnier, Y.; Simmet, T. Acetyl-boswellic acids inhibit lipopolysaccharide-mediated TNF-α induction in monocytes by direct interaction with IκB kinases. J. Immunol. 2005, 174, 498–506. [Google Scholar] [CrossRef] [PubMed]
- Ernst, E. Frankincense: Systematic review. BMJ 2008, 337, a2813. [Google Scholar] [CrossRef] [PubMed]
- Gupta, I.; Parihar, A.; Malhotra, P.; Singh, G.B.; Ludtke, R.; Safayhi, H.; Ammon, H.P. Effects of Boswellia serrata gum resin in patients with ulcerative colitis. Eur. J. Med. Res. 1997, 2, 37–43. [Google Scholar] [PubMed]
- Glaser, T.; Winter, S.; Groscurth, P.; Safayhi, H.; Sailer, E.R.; Ammon, H.P.T.; Schabet, M.; Weller, M. Boswellic acids and malignant glioma: Induction of apoptosis but no modulation of drug sensitivity. Br. J. Cancer 1999, 80, 756–765. [Google Scholar] [CrossRef]
- Hoernlein, R.F.; Orlikowsky, T.; Zehrer, C.; Niethammer, D.; Sailer, E.R.; Simmet, T.; Dannecker, G.E.; Ammon, H.P.T. Acetyl-11-keto-β-boswellic acid induces apoptosis in HL-60 and CCRF-CEM cells and inhibits topoisomerase I. J. Pharmacol. Exp. Ther. 1999, 288, 613–619. [Google Scholar] [PubMed]
- Schmidt, C.; Loos, C.; Jin, L.; Schmiech, M.; Schmidt, C.Q.; El Gaafary, M.; Syrovets, T.; Simmet, T. Acetyl-lupeolic acid inhibits Akt signaling and induces apoptosis in chemoresistant prostate cancer cells in vitro and in vivo. Oncotarget 2017, 8, 55147–55161. [Google Scholar] [CrossRef] [PubMed]
- Syrovets, T.; Gschwend, J.; Büchele, B.; Laumonnier, Y.; Zugmaier, W.; Genze, F.; Simmet, T. Inhibition of IκB kinase activity by acetyl-boswellic acids promotes apoptosis in androgen-independent PC-3 prostate cancer cells in vitro and in vivo. J. Biol. Chem. 2005, 280, 6170–6180. [Google Scholar] [CrossRef] [PubMed]
- Schmiech, M.; Lang, S.J.; Werner, K.; Rashan, L.J.; Syrovets, T.; Simmet, T. Comparative analysis of pentacyclic triterpenic acid compositions in oleogum resins of different Boswellia species and their in vitro cytotoxicity against treatment-resistant human breast cancer cells. Molecules 2019, 24, 2153. [Google Scholar] [CrossRef]
- Suhail, M.M.; Wu, W.; Cao, A.; Mondalek, F.G.; Fung, K.M.; Shih, P.T.; Fang, Y.T.; Woolley, C.; Young, G.; Lin, H.K. Boswellia sacra essential oil induces tumor cell-specific apoptosis and suppresses tumor aggressiveness in cultured human breast cancer cells. BMC Complement. Altern. Med. 2011, 11. [Google Scholar] [CrossRef]
- Der Weihrauch Blog. Available online: https://weihrauch-blog.de/ (accessed on 29 May 2019).
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Carey, L.; Winer, E.; Viale, G.; Cameron, D.; Gianni, L. Triple-negative breast cancer: Disease entity or title of convenience? Nat. Rev. Clin. Oncol. 2010, 7, 683. [Google Scholar] [CrossRef]
- Grand View Research. Dietary Supplements Market Size Analysis Report by Ingredient (Botanicals, Vitamins), by form, by Application (Immunity, Cardiac Health), by End User, by Distribution Channel, and Segment Forecasts, 2019–2025; Grand View Research: San Francisco, CA, USA, 2019. [Google Scholar]
- Yates, A.A.; Erdman, J.W., Jr.; Shao, A.; Dolan, L.C.; Griffiths, J.C. Bioactive nutrients–Time for tolerable upper intake levels to address safety. Regul. Toxicol. Pharmacol. 2017, 84, 94–101. [Google Scholar] [CrossRef]
- Belsner, K.; Büchele, B.; Werz, U.; Simmet, T. Structural analysis of 3-α-acetyl-20(29)-lupene-24-oic acid, a novel pentacyclic triterpene isolated from the gum resin of Boswellia serrata, by NMR spectroscopy. Magn. Reson. Chem. 2003, 41, 629–632. [Google Scholar] [CrossRef]
- Lang, S.J.; Schmiech, M.; Hafner, S.; Paetz, C.; Steinborn, C.; Huber, R.; Gaafary, M.E.; Werner, K.; Schmidt, C.Q.; Syrovets, T.; et al. Antitumor activity of an Artemisia annua herbal preparation and identification of active ingredients. Phytomedicine 2019, 62, 152962. [Google Scholar] [CrossRef]
- Rajpal, V. Standardization of Botanicals, 2nd ed.; Business Horizons: New Delhi, India, 2011; Volume 1. [Google Scholar]
- Meins, J.; Artaria, C.; Riva, A.; Morazzoni, P.; Schubert-Zsilavecz, M.; Abdel-Tawab, M. Survey on the quality of the top-selling european and american botanical dietary supplements containing boswellic acids. Planta Med. 2016, 82, 573–579. [Google Scholar] [CrossRef]
- Mathe, C.; Culioli, G.; Archier, P.; Vieillescazese, C. High-performance liquid chromatographic analysis of triterpenoids in commercial frankincense. Chromatographia 2004, 60, 493–499. [Google Scholar] [CrossRef]
- Iyer, S.S.; Ghaffari, A.A.; Cheng, G. Lipopolysaccharide-mediated IL-10 transcriptional regulation requires sequential induction of type I IFNs and IL-27 in macrophages. J. Immunol. 2010, 185, 6599–6607. [Google Scholar] [CrossRef] [PubMed]
- De Waal Malefyt, R.; Abrams, J.; Bennett, B.; Figdor, C.G.; de Vries, J.E. Interleukin 10 (IL-10) inhibits cytokine synthesis by human monocytes: An autoregulatory role of IL-10 produced by monocytes. J. Exp. Med. 1991, 174, 1209–1220. [Google Scholar] [CrossRef] [PubMed]
- Wanidworanun, C.; Strober, W. Predominant role of tumor necrosis factor-alpha in human monocyte IL-10 synthesis. J. Immunol. 1993, 151, 6853–6861. [Google Scholar]
- Siemoneit, U.; Pergola, C.; Jazzar, B.; Northoff, H.; Skarke, C.; Jauch, J.; Werz, O. On the interference of boswellic acids with 5-lipoxygenase: Mechanistic studies in vitro and pharmacological relevance. Eur. J. Pharmacol. 2009, 606, 246–254. [Google Scholar] [CrossRef]
- Belge, K.U.; Dayyani, F.; Horelt, A.; Siedlar, M.; Frankenberger, M.; Frankenberger, B.; Espevik, T.; Ziegler-Heitbrock, L. The proinflammatory CD14+CD16+DR++ monocytes are a major source of TNF. J. Immunol. 2002, 168, 3536–3542. [Google Scholar] [CrossRef]
- Swanson, K.V.; Deng, M.; Ting, J.P. The NLRP3 inflammasome: Molecular activation and regulation to therapeutics. Nat. Rev. Immunol. 2019, 19, 477–489. [Google Scholar] [CrossRef]
- Ayla, S.; Seckin, I.; Tanriverdi, G.; Cengiz, M.; Eser, M.; Soner, B.C.; Oktem, G. Doxorubicin induced nephrotoxicity: Protective effect of nicotinamide. Int. J. Cell Biol. 2011, 2011, 390238. [Google Scholar] [CrossRef]
- Swain, S.M.; Whaley, F.S.; Ewer, M.S. Congestive heart failure in patients treated with doxorubicin: A retrospective analysis of three trials. Cancer 2003, 97, 2869–2879. [Google Scholar] [CrossRef]
- Gupta, I.; Parihar, A.; Malhotra, P.; Gupta, S.; Ludtke, R.; Safayhi, H.; Ammon, H.P. Effects of gum resin of Boswellia serrata in patients with chronic colitis. Planta Med. 2001, 67, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Syrovets, T.; Büchele, B.; Gedik, E.; Slupsky, J.R.; Simmet, T. Acetyl-boswellic acids are novel catalytic inhibitors of human topoisomerases I and IIa. Mol. Pharmacol. 2000, 58, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Karin, M.; Cao, Y.; Greten, F.R.; Li, Z.W. NF-κB in cancer: From innocent bystander to major culprit. Nat. Rev. Cancer 2002, 2, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Grivennikov, S.I.; Greten, F.R.; Karin, M. Immunity, Inflammation, and Cancer. Cell 2010, 140, 883–899. [Google Scholar] [CrossRef] [PubMed]
- Balkwill, F.; Charles, K.A.; Mantovani, A. Smoldering and polarized inflammation in the initiation and promotion of malignant disease. Cancer Cell 2005, 7, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, H.H.; Abd-Rabou, A.A.; Hassan, A.Z.; Kotob, S.E. Phytochemical analysis and anti-cancer investigation of boswellia serrata bioactive constituents in vitro. Asian Pac. J. Cancer Prev. 2015, 16, 7179–7188. [Google Scholar] [CrossRef] [PubMed]
- Van Hoogevest, P.; Wendel, A. The use of natural and synthetic phospholipids as pharmaceutical excipients. Eur. J. Lipid. Sci. Technol. 2014, 116, 1088–1107. [Google Scholar] [CrossRef]
- Sterk, V.; Buchele, B.; Simmet, T. Effect of food intake on the bioavailability of boswellic acids from a herbal preparation in healthy volunteers. Planta Med. 2004, 70, 1155–1160. [Google Scholar] [CrossRef]
- Riva, A.; Morazzoni, P.; Artaria, C.; Allegrini, P.; Meins, J.; Savio, D.; Appendino, G.; Schubert-Zsilavecz, M.; Abdel-Tawab, M. A single-dose, randomized, cross-over, two-way, open-label study for comparing the absorption of boswellic acids and its lecithin formulation. Phytomedicine 2016, 23, 1375–1382. [Google Scholar] [CrossRef]
- Wang, H.; Syrovets, T.; Kess, D.; Büchele, B.; Hainzl, H.; Lunov, O.; Weiss, J.M.; Scharffetter-Kochanek, K.; Simmet, T. Targeting NF-κB with a natural triterpenoid alleviates skin inflammation in a mouse model of psoriasis. J. Immunol. 2009, 183, 4755–4763. [Google Scholar] [CrossRef]
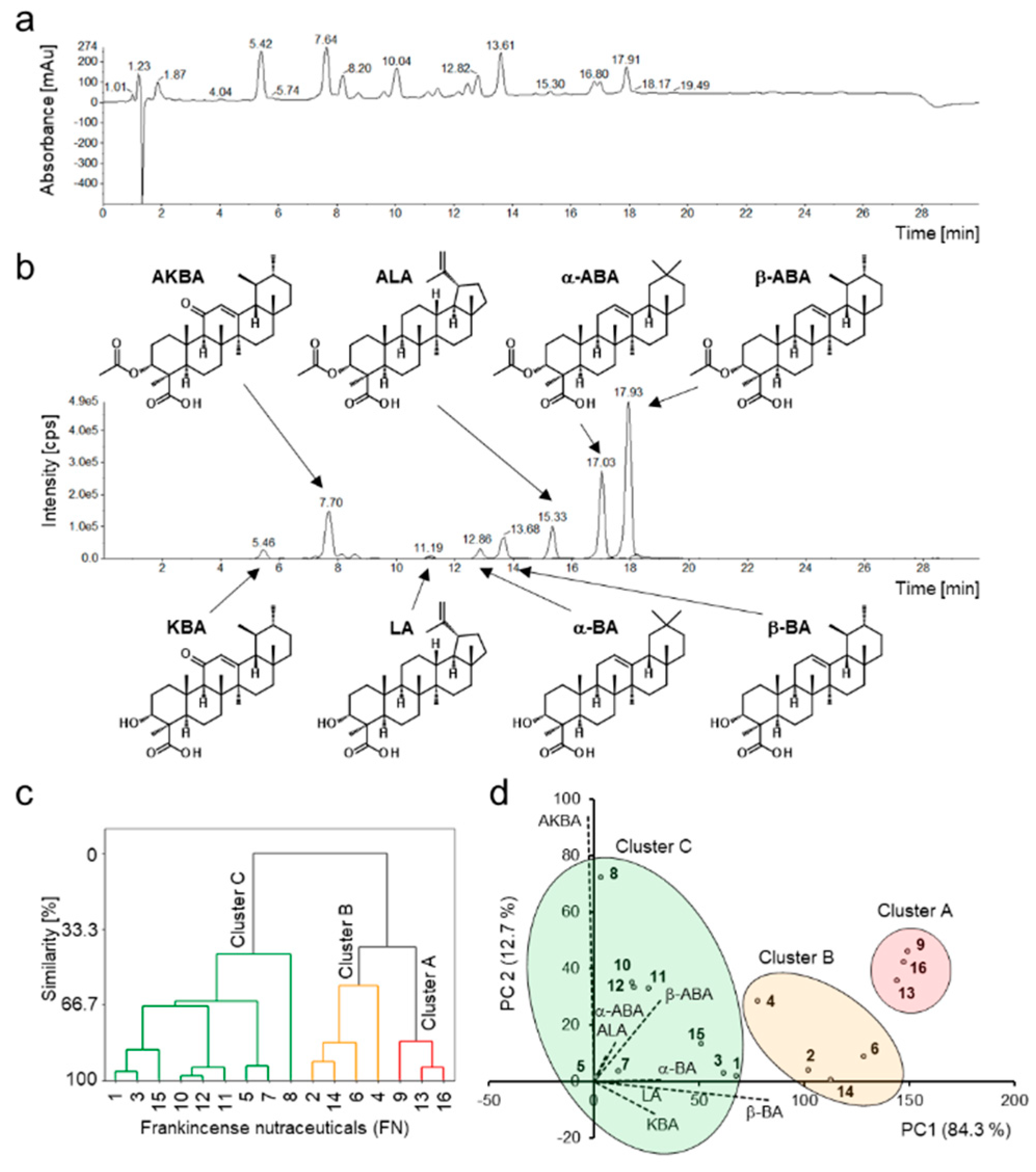
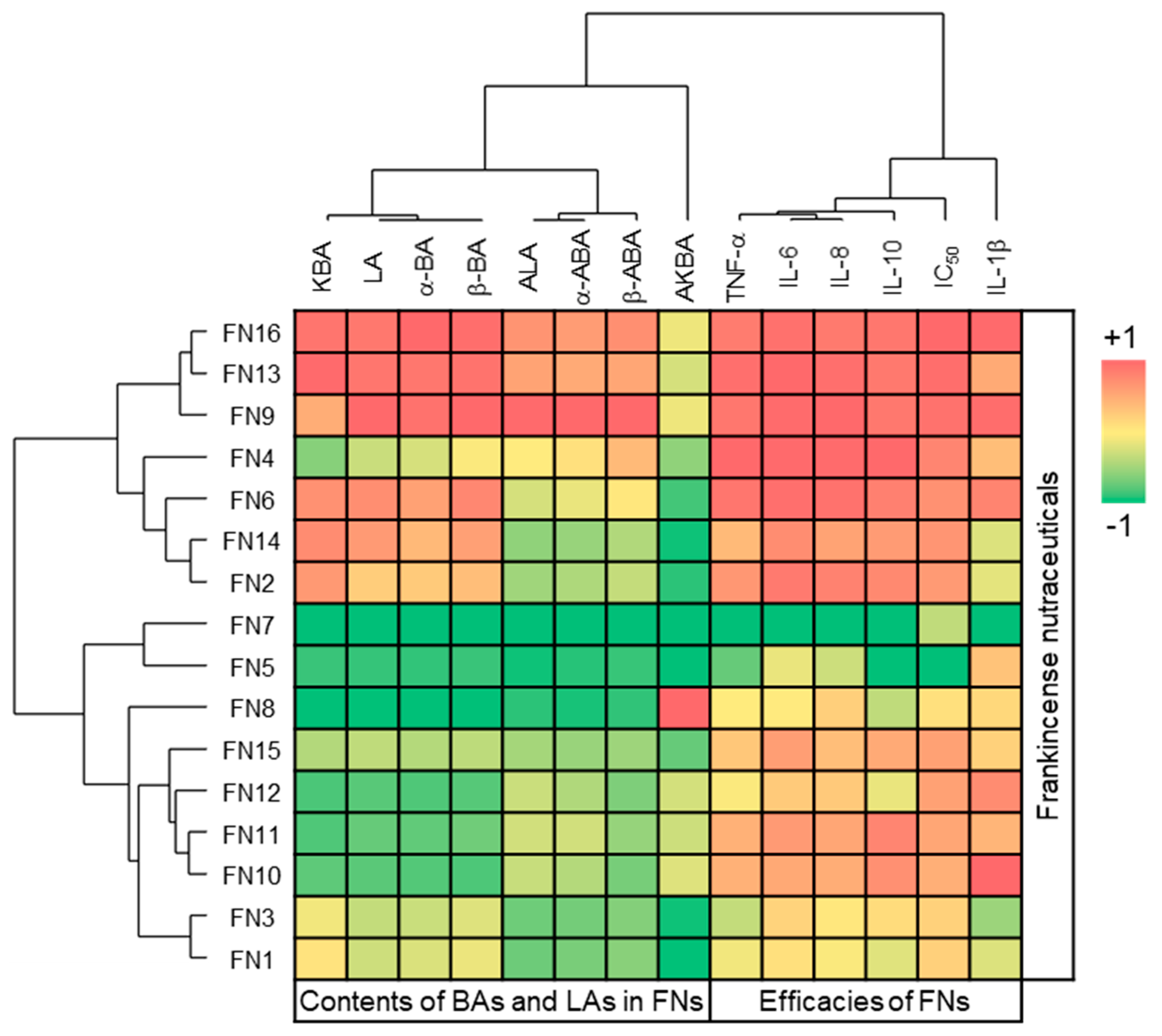
| FN Sample | Concentrations of Boswellic and Lupeolic acids | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Deacetylated Compounds (µg/mg) | Acetylated Compounds (µg/mg) | Σ (%) (w/w) | |||||||
| KBA | LA | α-BA | β-BA | AKBA | ALA | α-ABA | β-ABA | ||
| FN9 | 34.1 | 17.2 | 46.5 | 120.7 | 36.9 | 13.8 | 24.4 | 63.6 | 35.7 |
| FN16 | 42.3 | 16.4 | 47.7 | 118.8 | 36.9 | 11.9 | 20.3 | 55.7 | 35.0 |
| FN13 | 43.9 | 16.5 | 45.6 | 117.2 | 31.8 | 11.2 | 19.2 | 50.7 | 33.6 |
| FN6 | 38.1 | 15.0 | 38.8 | 109.0 | 7.7 | 5.9 | 11.8 | 36.7 | 26.3 |
| FN14 | 38.9 | 14.4 | 34.9 | 98.3 | 4.2 | 3.5 | 6.6 | 20.8 | 22.1 |
| FN2 | 36.9 | 11.3 | 32.0 | 85.9 | 5.6 | 4.0 | 7.9 | 24.0 | 20.8 |
| FN4 | 9.7 | 6.6 | 19.9 | 62.9 | 18.6 | 7.8 | 14.7 | 46.3 | 18.6 |
| FN1 | 25.7 | 6.8 | 20.5 | 57.6 | 3.6 | 2.4 | 4.9 | 14.8 | 13.6 |
| FN3 | 21.8 | 6.3 | 18.3 | 53.1 | 4.3 | 2.4 | 4.6 | 13.9 | 12.5 |
| FN15 | 14.2 | 6.1 | 15.6 | 42.4 | 11.8 | 4.1 | 6.7 | 17.7 | 11.9 |
| FN11 | 4.8 | 2.5 | 6.8 | 20.1 | 30.0 | 5.7 | 10.1 | 16.7 | 9.7 |
| FN8 | 1.3 | 0.5 | 1.1 | 3.2 | 75.5 | 1.1 | 1.6 | 4.6 | 8.9 |
| FN10 | 5.9 | 2.2 | 5.6 | 12.7 | 33.6 | 5.3 | 8.2 | 12.2 | 8.6 |
| FN12 | 4.5 | 2.1 | 5.3 | 14.2 | 31.4 | 5.4 | 8.0 | 13.2 | 8.4 |
| FN5 | 3.4 | 1.3 | 3.5 | 9.2 | 2.9 | 0.8 | 1.9 | 5.7 | 2.9 |
| FN7 | 0.3 | 0.1 | 0.3 | 0.5 | 1.2 | 0.3 | 0.6 | 1.1 | 0.4 |
| Compound | TNF-α | IL-1β | IL-6 | IL-8 | IL-10 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| R | p | R | p | R | p | R | p | R | p | |
| KBA | −0.621 | * | −0.241 | > 0.05 | −0.744 | *** | −0.665 | ** | −0.656 | ** |
| LA | −0.721 | ** | −0.341 | > 0.05 | −0.832 | *** | −0.791 | *** | −0.765 | *** |
| α-BA | −0.709 | ** | −0.356 | > 0.05 | −0.815 | *** | −0.774 | *** | −0.774 | *** |
| β-BA | −0.735 | *** | −0.359 | > 0.05 | −0.850 | *** | −0.815 | *** | −0.794 | *** |
| AKBA | −0.537 | * | −0.714 | ** | −0.430 | > 0.05 | −0.530 | * | −0.511 | * |
| ALA | −0.867 | *** | −0.784 | *** | −0.884 | *** | −0.890 | *** | −0.918 | *** |
| α-ABA | −0.879 | *** | −0.797 | *** | −0.891 | *** | −0.897 | *** | −0.932 | *** |
| β-ABA | −0.847 | *** | −0.447 | > 0.05 | −0.932 | *** | −0.903 | *** | −0.900 | *** |
| Σ BAs and LAs | −0.753 | *** | −0.318 | > 0.05 | −0.835 | *** | −0.818 | *** | −0.785 | *** |
| TNF-α | 0.612 | * | 0.959 | *** | 0.968 | *** | 0.944 | *** | ||
| IL-1β | 0.535 | * | 0.600 | * | 0.624 | ** | ||||
| IL-6 | 0.976 | *** | 0.947 | *** | ||||||
| IL-8 | 0.947 | *** | ||||||||
| FN Sample | XTT: IC50 (µg/mL) | Regression: IC50 (µg/mL) | Absolute Residue (µg/mL) | ||
|---|---|---|---|---|---|
| Mean | SEM | Value | SE | ||
| FN16 | 6.0 | 0.9 | 6.7 | 0.9 | 0.7 |
| FN13 | 6.6 | 0.6 | 5.7 | 0.8 | 1.0 |
| FN9 | 7.0 | 0.4 | 7.5 | 1.0 | 0.5 |
| FN4 | 9.5 | 0.9 | 8.4 | 0.9 | 1.1 |
| FN6 | 11.0 | 1.8 | 9.2 | 1.1 | 1.8 |
| FN14 | 11.6 | 0.7 | 13.7 | 0.9 | 2.0 |
| FN2 | 12.2 | 1.6 | 13.1 | 0.7 | 0.9 |
| FN15 | 13.2 | 1.1 | 15.4 | 0.7 | 2.2 |
| FN12 | 13.2 | 0.6 | 14.5 | 0.8 | 1.3 |
| FN11 | 13.9 | 0.5 | 13.6 | 0.8 | 0.3 |
| FN10 | 15.0 | 0.9 | 14.4 | 0.9 | 0.6 |
| FN1 | 19.4 | 2.7 | 18.1 | 0.8 | 1.3 |
| FN3 | 19.5 | 1.0 | 18.4 | 0.8 | 1.1 |
| FN8 | 21.5 | 3.6 | 21.4 | 1.5 | 0.2 |
| FN7 | 30.7 | 6.7 | 30.4 | 1.4 | 0.3 |
| FN5 | 44.3 | 8.5 | / | / | / |
| FN Sample | IC50 (µg/mL) | |||
|---|---|---|---|---|
| MDA-MB-453 | Cal-51 | |||
| Mean | SEM | Mean | SEM | |
| FN9 | 14.5 | 0.9 | 3.8 | 0.5 |
| FN13 | 12.9 | 0.6 | 3.9 | 0.6 |
| FN16 | 17.3 | 0.5 | 4.0 | 0.3 |
| Compound | IC50 | |||
|---|---|---|---|---|
| µg/mL | µM | |||
| Mean | SEM | Mean | SEM | |
| KBA | 12.0 | 0.4 | 25.4 | 0.9 |
| LA | 5.6 | 0.1 | 12.3 | 0.2 |
| α-BA | 5.0 | 0.4 | 10.8 | 0.9 |
| β-BA | 4.2 | 0.1 | 9.3 | 0.2 |
| AKBA | 3.4 | 0.1 | 6.6 | 0.2 |
| ALA | 13.4 | 0.5 | 26.9 | 1.0 |
| α-ABA | 3.6 | 0.5 | 7.2 | 1.0 |
| β-ABA | 2.9 | 0.4 | 5.9 | 0.8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schmiech, M.; Lang, S.J.; Ulrich, J.; Werner, K.; Rashan, L.J.; Syrovets, T.; Simmet, T. Comparative Investigation of Frankincense Nutraceuticals: Correlation of Boswellic and Lupeolic Acid Contents with Cytokine Release Inhibition and Toxicity against Triple-Negative Breast Cancer Cells. Nutrients 2019, 11, 2341. https://doi.org/10.3390/nu11102341
Schmiech M, Lang SJ, Ulrich J, Werner K, Rashan LJ, Syrovets T, Simmet T. Comparative Investigation of Frankincense Nutraceuticals: Correlation of Boswellic and Lupeolic Acid Contents with Cytokine Release Inhibition and Toxicity against Triple-Negative Breast Cancer Cells. Nutrients. 2019; 11(10):2341. https://doi.org/10.3390/nu11102341
Chicago/Turabian StyleSchmiech, Michael, Sophia J. Lang, Judith Ulrich, Katharina Werner, Luay J. Rashan, Tatiana Syrovets, and Thomas Simmet. 2019. "Comparative Investigation of Frankincense Nutraceuticals: Correlation of Boswellic and Lupeolic Acid Contents with Cytokine Release Inhibition and Toxicity against Triple-Negative Breast Cancer Cells" Nutrients 11, no. 10: 2341. https://doi.org/10.3390/nu11102341
APA StyleSchmiech, M., Lang, S. J., Ulrich, J., Werner, K., Rashan, L. J., Syrovets, T., & Simmet, T. (2019). Comparative Investigation of Frankincense Nutraceuticals: Correlation of Boswellic and Lupeolic Acid Contents with Cytokine Release Inhibition and Toxicity against Triple-Negative Breast Cancer Cells. Nutrients, 11(10), 2341. https://doi.org/10.3390/nu11102341