The Impact of Epicatechin on Human Cognition: The Role of Cerebral Blood Flow
Abstract
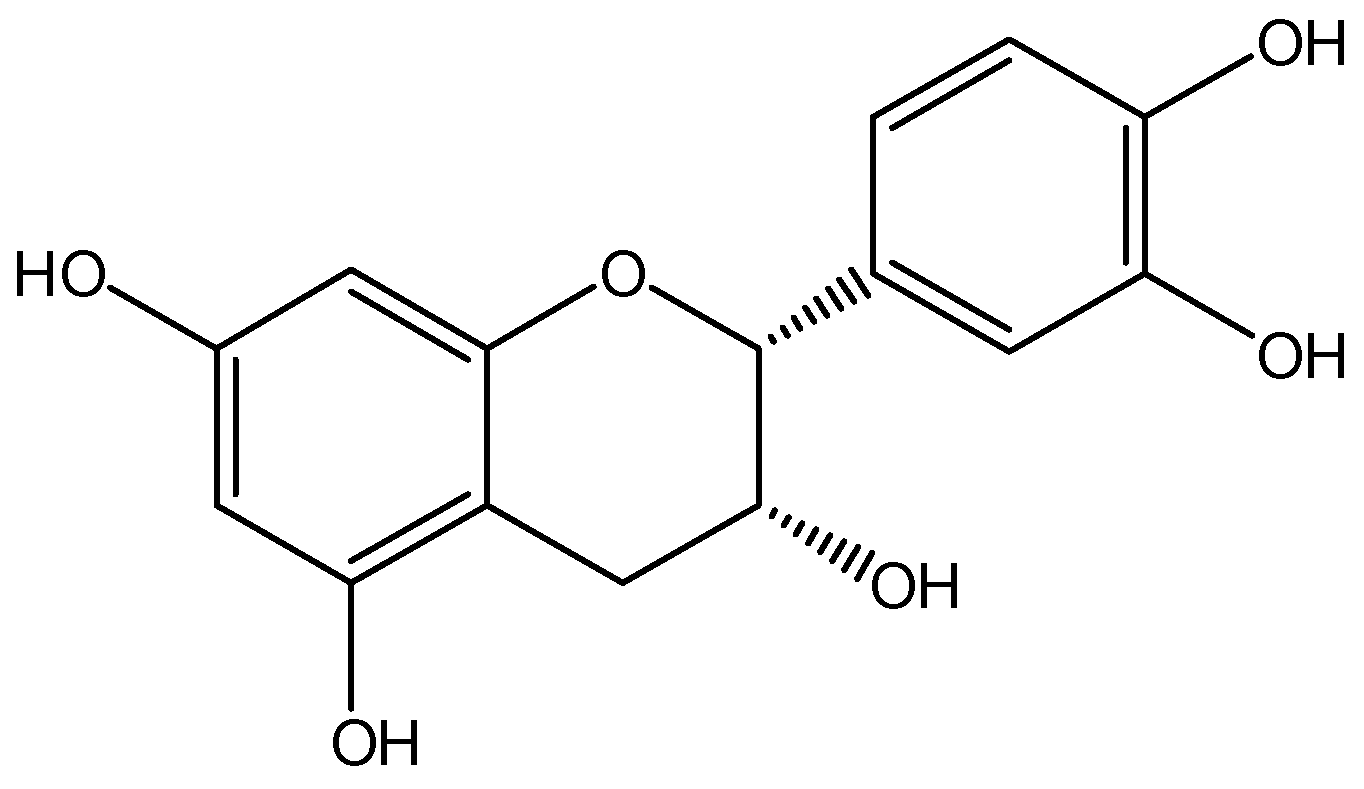
1. Introduction
2. Cognition Intervention Studies
3. Potential Mechanisms and Bioavailability
3.1. Potential Mechanisms
3.2. Caveats for Result Interpretation
3.3. Blood-Brain Barrier
4. Peripheral and Cerebral Blood Flow Intervention Studies
5. Summary of Evidence for Cognitive Effects and Their Relationship to Blood Flow
6. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Commenges, D.; Scotet, V.; Renaud, S.; Jacqmin-Gadda, H.; Barberger-Gateau, P.; Dartigues, J.F. Intake of flavonoids and risk of dementia. Eur. J. Epidemiol. 2000, 16, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Letenneur, L.; Proust-Lima, C.; Le Gouge, A.; Dartigues, J.F.; Barberger-Gateau, P. Flavonoid intake and cognitive decline over a 10-year period. Am. J. Epidemiol. 2007, 165, 1364–1371. [Google Scholar] [CrossRef] [PubMed]
- Kuriyama, S.; Hozawa, A.; Ohmori, K.; Shimazu, T.; Matsui, T.; Ebihara, S.; Awata, S.; Nagatomi, R.; Arai, H.; Tsuji, I. Green tea consumption and cognitive function: A cross-sectional study from the Tsurugaya Project. Am. J. Clin. Nutr. 2006, 83, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.P.; Feng, L.; Niti, M.; Kua, E.H.; Yap, K.B. Tea consumption and cognitive impairment and decline in older Chinese adults. Am. J. Clin. Nutr. 2008, 88, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Gwee, X.; Kua, E.H.; Ng, T.P. Cognitive function and tea consumption in community dwelling older Chinese in Singapore. J. Nutr. Health Aging 2010, 14, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Nurk, E.; Refsum, H.; Drevon, C.A.; Tell, G.S.; Nygaard, H.A.; Engedal, K.; Smith, A.D. Intake of flavonoid-rich wine, tea, and chocolate by elderly men and women is associated with better cognitive test performance. J. Nutr. 2009, 139, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Neafsey, E.J.; Collins, M.A. Moderate alcohol consumption and cognitive risk. Neuropsychiatr. Dis. Treat. 2011, 7, 465–484. [Google Scholar] [CrossRef] [PubMed]
- Francis, S.T.; Head, K.; Morris, P.G.; Macdonald, I.A. The effect of flavanol-rich cocoa on the fMRI response to a cognitive task in healthy young people. J. Cardiovasc. Pharmacol. 2006, 47, S215–S220. [Google Scholar] [CrossRef] [PubMed]
- Scholey, A.B.; French, S.J.; Morris, P.J.; Kennedy, D.O.; Milne, A.L.; Haskell, C.F. Consumption of cocoa flavanols results in acute improvements in mood and cognitive performance during sustained mental effort. J. Psychopharmacol. 2010, 24, 1505–1514. [Google Scholar] [CrossRef] [PubMed]
- Boolani, A.; Lindheimer, J.B.; Loy, B.D.; Crozier, S.; O’Connor, P.J. Acute effects of brewed cocoa consumption on attention, motivation to perform cognitive work and feelings of anxiety, energy and fatigue: A randomized, placebo-controlled crossover experiment. BMC Nutr. 2017, 3, 8. [Google Scholar] [CrossRef]
- Tsukamoto, H.; Suga, T.; Ishibashi, A.; Takenaka, S.; Tanaka, D.; Hirano, Y.; Hamaoka, T.; Goto, K.; Ebi, K.; Isaka, T.; et al. Flavanol-rich cocoa consumption enhances exercise-induced executive function improvements in humans. Nutrition 2018, 46, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Decroix, L.; Tonoli, C.; Soares, D.D.; Tagougui, S.; Heyman, E.; Meeusen, R. Acute cocoa flavanol improves cerebral oxygenation without enhancing executive function at rest or after exercise. Appl. Physiol. Nutr. Metab. 2016, 41, 1225–1232. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Socci, V.; Tempesta, D.; Ferri, C.; De Gennaro, L.; Desideri, G.; Michele, F. Flavanol-rich chocolate acutely improves arterial function and working memory performance counteracting the effects of sleep deprivation in healthy individuals. J. Hypertens. 2016, 34, 1298–1308. [Google Scholar] [CrossRef] [PubMed]
- Field, D.T.; Williams, C.M.; Butler, L.T. Consumption of cocoa flavanols results in an acute improvement in visual and cognitive functions. Physiol. Behav. 2011, 103, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Massee, L.A.; Ried, K.; Pase, M.; Travica, N.; Yoganathan, J.; Scholey, A.; Scholey, A.; Macpherson, H.; Kennedy, G.; Sali, A.; et al. The acute and sub-chronic effects of cocoa flavanols on mood, cognitive and cardiovascular health in young healthy adults: A randomized, controlled trial. Front. Pharmacol. 2015, 6, 93. [Google Scholar] [CrossRef] [PubMed]
- Pase, M.P.; Scholey, A.B.; Pipingas, A.; Kras, M.; Nolidin, K.; Gibbs, A.; Wesnes, K.; Stough, C. Cocoa polyphenols enhance positive mood states but not cognitive performance: A randomized, placebo-controlled trial. J. Psychopharmacol. 2013, 27, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Scholey, A.B.; Tildesley, N.T.J.; Ballard, C.G.; Wesnes, K.A.; Tasker, A.; Perry, E.K.; Kennedy, D.O. An extract of Salvia (sage) with anticholinesterase properties improves memory and attention in healthy older volunteers. Psychopharmacology 2008, 198, 127–139. [Google Scholar] [CrossRef] [PubMed]
- Haskell, C.F.; Scholey, A.B.; Jackson, P.A.; Elliott, J.M.; Defeyter, M.A.; Greer, J.; Robertson, B.C.; Buchanan, T.; Tiplady, B.; Kennedy, D.O. Cognitive and mood effects in healthy children during 12 weeks' supplementation with multi-vitamin/minerals. Br. J. Nutr. 2008, 100, 1086–1096. [Google Scholar] [CrossRef] [PubMed]
- Bond, A.; Lader, M. Use of analog scales in rating subjective feelings. Br. J. Med. Psychol. 1974, 47, 211–218. [Google Scholar] [CrossRef]
- Camfield, D.A.; Scholey, A.; Pipingas, A.; Silberstein, R.; Kras, M.; Nolidin, K.; Wesnes, K.; Pase, M.; Stougha, C. Steady state visually evoked potential (SSVEP) topography changes associated with cocoa flavanol consumption. Physiol. Behav. 2012, 105, 948–957. [Google Scholar] [CrossRef] [PubMed]
- Silberstein, R.B.; Nunez, P.L.; Pipingas, A.; Harris, P.; Danieli, F. Steady state visually evoked potential (SSVEP) topography in a graded working memory task. Int. J. Psychophysiol. 2001, 42, 219–232. [Google Scholar] [CrossRef]
- Brickman, A.M.; Khan, U.A.; Provenzano, F.A.; Yeung, L.K.; Suzuki, W.; Schroeter, H.; Wall, M.; Sloan, R.P.; Small, S.A. Enhancing dentate gyrus function with dietary flavanols improves cognition in older adults. Nat. Neurosci. 2014, 17, 1798–1803. [Google Scholar] [CrossRef] [PubMed]
- Crews, W.D., Jr.; Harrison, D.W.; Wright, J.W. A double-blind, placebo-controlled, randomized trial of the effects of dark chocolate and cocoa on variables associated with neuropsychological functioning and cardiovascular health: Clinical findings from a sample of healthy, cognitively intact older adults. Am. J. Clin. Nutr. 2008, 87, 872–880. [Google Scholar] [PubMed]
- Sorond, F.A.; Hurwitz, S.; Salat, D.H.; Greve, D.N.; Fisher, N.D. Neurovascular coupling, cerebral white matter integrity, and response to cocoa in older people. Neurology 2013, 81, 904–909. [Google Scholar] [CrossRef] [PubMed]
- Neshatdoust, S.; Saunders, C.; Castle, S.M.; Vauzour, D.; Williams, C.; Butler, L.; Lovegrove, J.A.; Spencer, J.P.E. High-flavonoid intake induces cognitive improvements linked to changes in serum brain-derived neurotrophic factor: Two randomised, controlled trials. Nutr. Healthy Aging 2016, 4, 81–93. [Google Scholar] [CrossRef] [PubMed]
- Desideri, G.; Kwik-Uribe, C.; Grassi, D.; Necozione, S.; Ghiadoni, L.; Mastroiacovo, D.; Raffaele, A.; Ferri, L.; Bocale, R.; Lechiara, M.C.; et al. Benefits in cognitive function, blood pressure, and insulin resistance through cocoa flavanol consumption in elderly subjects with mild cognitive impairment: The Cocoa, Cognition, and Aging (CoCoA) study. Hypertension 2012, 60, 794–801. [Google Scholar] [CrossRef] [PubMed]
- Mastroiacovo, D.; Kwik-Uribe, C.; Grassi, D.; Necozione, S.; Raffaele, A.; Pistacchio, L.; Righetti, R.; Bocale, R.; Lechiara, M.C.; Marini, C.; et al. Cocoa flavanol consumption improves cognitive function, blood pressure control, and metabolic profile in elderly subjects: The Cocoa, Cognition, and Aging (CoCoA) Study—A randomized controlled trial. Am. J. Clin. Nutr. 2015, 101, 538–548. [Google Scholar] [CrossRef] [PubMed]
- Marsh, C.E.; Carter, H.H.; Guelfi, K.J.; Smith, K.J.; Pike, K.E.; Naylor, L.H.; Green, D.J. Brachial and Cerebrovascular Functions Are Enhanced in Postmenopausal Women after Ingestion of Chocolate with a High Concentration of Cocoa. J. Nutr. 2017, 147, 1686–1692. [Google Scholar] [CrossRef] [PubMed]
- Spencer, J. The interactions of flavonoids within neuronal signalling pathways. Genes Nutr. 2007, 2, 257–273. [Google Scholar] [CrossRef] [PubMed]
- Spencer, J.P. Flavonoids and brain health: Multiple effects underpinned by common mechanisms. Genes Nutr. 2009, 4, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.J.; Spencer, J.P. Flavonoids, cognition, and dementia: Actions, mechanisms, and potential therapeutic utility for Alzheimer disease. Free Radic. Biol. Med. 2012, 52, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Schroeter, H.; Bahia, P.; Spencer, J.P.E.; Sheppard, O.; Rattray, M.; Cadenas, E.; Rice-Evans, C.; Williams, R.J. (−) Epicatechin stimulates ERK-dependent cyclic AMP response element activity and up-regulates GluR2 in cortical neurons. J. Neurochem. 2007, 101, 1596–1606. [Google Scholar] [CrossRef] [PubMed]
- Heo, H.J.; Lee, C.Y. Epicatechin and catechin in cocoa inhibit amyloid 6 protein induced apoptosis. J. Agric. Food. Chem. 2005, 53, 1445–1448. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.S.; Lee, K.W.; Lee, H.J. Cocoa procyanidins protect PC12 cells from hydrogen-peroxide-induced apoptosis by inhibiting activation of p38 MAPK and JNK. Mutat. Res. 2008, 640, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.S.; Jang, Y.J.; Kang, N.J.; Hwang, M.K.; Kim, Y.T.; Lee, K.W.; Lee, H.J. Cocoa procyanidins attenuate 4-hydroxynonenal-induced apoptosis of PC12 cells by directly inhibiting mitogen-activated protein kinase kinase 4 activity. Free Radic. Biol. Med. 2009, 46, 1319–1327. [Google Scholar] [CrossRef] [PubMed]
- Ramiro-Puig, E.; Casadesús, G.; Lee, H.-G.; Zhu, X.; McShea, A.; Perry, G.; Pérez-Cano, F.J.; Smith, M.A.; Castell, M. Neuroprotective effect of cocoa flavonids on in vitro oxidative stress. Eur. J. Nutr. 2009, 48, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Donovan, J.L. Pharmacokinetics and metabolism of dietary flavonoids in humans. Free Radic. Res. 2004, 38, 771–785. [Google Scholar] [CrossRef] [PubMed]
- Rein, M.J.; Renouf, M.; Cruz-Hernandez, C.; Actis-Goretta, L.; Thakkar, S.K.; Pinto, M.D.S. Bioavailability of bioactive food compounds: A challenging journey to bioefficacy. Br. J. Clin. Pharmacol. 2013, 75, 588–602. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, J.I.; Kwik-Uribe, C.; Keen, C.L.; Schroeter, H. Intake of dietary procyanidins does not contribute to the pool of circulating flavanols in humans. Am. J. Clin. Nutr. 2012, 95, 851–858. [Google Scholar] [CrossRef] [PubMed]
- Schroeter, H.; Heiss, C.; Balzer, J.; Kleinbongard, P.; Keen, C.L.; Hollenberg, N.K.; Sies, H.; Kwik-Uribe, C.; Schmitz, H.H.; Kelm, M. (−)-Epicatechin mediates beneficial effects of flavanol-rich cocoa on vascular function in humans. Proc. Natl. Acad. Sci. USA 2006, 103, 1024–1029. [Google Scholar] [CrossRef] [PubMed]
- Actis-Goretta, L.; Leveques, A.; Rein, M.; Teml, A.; Schafer, C.; Hofmann, U.; Li, H.; Schwab, M.; Eichelbaum, M.; Williamson, G. Intestinal absorption, metabolism, and excretion of (−)-epicatechin in healthy humans assessed by using an intestinal perfusion technique. Am. J. Clin. Nutr. 2013, 98, 924–933. [Google Scholar] [CrossRef] [PubMed]
- Actis-Goretta, L.; Leveques, A.; Giuffrida, F.; Romanov-Michailidis, F.; Viton, F.; Barron, D.; Duenas-Paton, M.; Gonzalez-Manzano, S.; Santos-Buelga, C.; Williamsonac, G. Elucidation of (−)-epicatechin metabolites after ingestion of chocolate by healthy humans. Free Radic. Biol. Med. 2012, 53, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, J.I.; Momma, T.Y.; Kuhnle, G.K.; Keen, C.L.; Schroeter, H. Structurally related (−)-epicatechin metabolites in humans: Assessment using de novo chemically synthesized authentic standards. Free Radic. Biol. Med. 2012, 15, 1403–1412. [Google Scholar] [CrossRef] [PubMed]
- Barnett, C.F.; Moreno-Ulloa, A.; Shiva, S.; Ramirez-Sanchez, I.; Taub, P.R.; Su, Y.; Ceballos, G.; Dugar, S.; Schreinerf, G.; Villarreal, F. Pharmacokinetic, partial pharmacodynamic and initial safety analysis of (−)-epicatechin in healthy volunteers. Food Funct. 2015, 6, 824–833. [Google Scholar] [CrossRef] [PubMed]
- El Aidy, S.; van den Bogert, B.; Kleerebezem, M. The small intestine microbiota, nutritional modulation and relevance for health. Curr. Opin. Biotechnol. 2015, 32, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Tzounis, X.; Rodriguez-Mateos, A.; Vulevic, J.; Gibson, G.R.; Kwik-Uribe, C.; Spencer, J.P.E. Prebiotic evaluation of cocoa-derived flavanols in healthy humans by using a randomized, controlled, double-blind, crossover intervention study. Am. J. Clin. Nutr. 2011, 93, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Faria, A.; Pestana, D.; Teixeira, D.; Couraud, P.-O.; Romero, I.; Weksler, B.; de Freitas, V.; Mateus, N.; Calhau, C. Insights into the putative catechin and epicatechin transport across blood-brain barrier. Food Funct. 2011, 2, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ferruzzi, M.G.; Ho, L.; Blount, J.; Janle, E.M.; Gong, B.; Pan, Y.; Gowda, G.N.; Raftery, D.; Arrieta-Cruz, I.; et al. Brain-targeted proanthocyanidin metabolites for alzheimer’s disease treatment. J. Neurosci. 2012, 32, 5144–5150. [Google Scholar] [CrossRef] [PubMed]
- Van Praag, H.; Lucero, M.J.; Yeo, G.W.; Stecker, K.; Heivand, N.; Zhao, C.; Yip, E.; Afanador, M.; Schroeter, H.; Hammerstone, J.; et al. Plant-derived flavanol (−) epicatechin enhances angiogenesis and retention of spatial memory in mice. J. Neurosci. 2007, 27, 5869–5878. [Google Scholar] [CrossRef] [PubMed]
- Youdim, K.A.; Dobbie, M.S.; Kuhnle, G.; Proteggente, A.R.; Abbott, N.J.; Rice-Evans, C. Interaction between flavonoids and the blood–brain barrier: In vitro studies. J. Neurochem. 2003, 85, 180–192. [Google Scholar] [CrossRef] [PubMed]
- Bengtsson, J.; Ederoth, P.; Ley, D.; Hansson, S.; Amer-Wåhlin, I.; Hellström-Westas, L.; Marsal, K.; Nordström, C.H.; Hammarlund-Udenaes, M. The influence of age on the distribution of morphine and morphine-3-glucuronide across the blood-brain barrier in sheep. Br. J. Pharmacol. 2009, 157, 1085–1096. [Google Scholar] [CrossRef] [PubMed]
- Aasmundstad, T.A.; Morland, J.; Paulsen, R.E. Distribution of morphine 6-glucuronide and morphine across the blood-brain-barrier in awake, freely moving rats investigated by in-vivo microdialysis sampling. J. Pharmacol. Exp. Ther. 1995, 275, 435–441. [Google Scholar] [PubMed]
- Buijsse, B.; Feskens, E.J.M.; Kok, F.J.; Kromhout, D. Cocoa intake, blood pressure, and cardiovascular mortality—The Zutphen Elderly Study. Arch. Intern. Med. 2006, 166, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Buijsse, B.; Weikert, C.; Drogan, D.; Bergmann, M.; Boeing, H. Chocolate consumption in relation to blood pressure and risk of cardiovascular disease in German adults. Eur. Heart J. 2010, 31, 1616–1623. [Google Scholar] [CrossRef] [PubMed]
- Djousse, L.; Hopkins, P.N.; North, K.E.; Pankow, J.S.; Arnett, D.K.; Ellison, R.C. Chocolate consumption is inversely associated with prevalent coronary heart disease: The National Heart, Lung, and Blood Institute Family Heart Study. Clin. Nutr. 2011, 30, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Janszky, I.; Mukamal, K.J.; Ljung, R.; Ahnve, S.; Ahlbom, A.; Hallqvist, J. Chocolate consumption and mortality following a first acute myocardial infarction: The Stockholm Heart Epidemiology Program. J. Intern. Med. 2009, 266, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Mostofsky, E.; Levitan, E.B.; Wolk, A.; Mittleman, M.A. Chocolate intake and incidence of heart failure a population-based prospective study of middle-aged and elderly women. Circ. Heart Fail. 2010, 3, 612–616. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.R.; Prince, R.L.; Zhu, K.; Devine, A.; Thompson, P.L.; Hodgson, J.M. Habitual chocolate intake and vascular disease: A prospective study of clinical outcomes in older women. Arch. Intern. Med. 2010, 170, 1857–1858. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Virtamo, J.; Wolk, A. Chocolate consumption and risk of stroke in women. J. Am. Coll. Cardiol. 2011, 58, 1828–1829. [Google Scholar] [CrossRef] [PubMed]
- Bayard, V.; Chamorro, F.; Motta, J.; Hollenberg, N.K. Does flavanol intake influence mortality from nitric oxide-dependent processes? Ischemic heart disease, stroke, diabetes mellitus, and cancer in Panama. Int. J. Med. Sci. 2007, 4, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Fraga, C.G.; Actis-Goretta, L.; Ottaviani, J.I.; Carrasquedo, F.; Lotito, S.B.; Lazarus, S.; Schmitz, H.H.; Keen, C.L. Regular consumption of a flavanol-rich chocolate can improve oxidant stress in young soccer players. Clin. Dev. Immunol. 2005, 12, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Necozione, S.; Lippi, C.; Croce, G.; Valeri, L.; Pasqualetti, P.; Desideri, G.; Blumberg, J.B.; Ferri, C. Cocoa reduces blood pressure and insulin resistance and improves endothelium-dependent vasodilation in hypertensives. Hypertension 2005, 46, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Lippi, C.; Necozione, S.; Desideri, G.; Ferri, C. Short-term administration of dark chocolate is followed by a significant increase in insulin sensitivity and a decrease in blood pressure in healthy persons. Am. J. Clin. Nutr. 2005, 81, 611–614. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Desideri, G.; Necozione, S.; Lippi, C.; Casale, R.; Properzi, G.; Blumberg, J.B.; Ferri, C. Blood pressure is reduced and insulin sensitivity increased in glucose-intolerant, hypertensive subjects after 15 days of consuming high-polyphenol dark chocolate. J. Nutr. 2008, 138, 1671–1676. [Google Scholar] [CrossRef] [PubMed]
- Faridi, Z.; Njike, V.Y.; Dutta, S.; Ali, A.; Katz, D.L. Acute dark chocolate and cocoa ingestion and endothelial function: A randomized controlled crossover trial. Am. J. Clin. Nutr. 2008, 88, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Taubert, D.; Berkels, R.; Roesen, R.; Klaus, W. Chocolate and blood pressure in elderly individuals with isolated systolic hypertension. JAMA 2003, 290, 1029–1030. [Google Scholar] [CrossRef] [PubMed]
- Taubert, D.; Roesen, R.; Lehmann, C.; Jung, N.; Schoemig, E. Effects of low habitual cocoa intake on blood pressure and bioactive nitric oxide—A randomized controlled trial. JAMA 2007, 298, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Heiss, C.; Jahn, S.; Taylor, M.; Real, W.M.; Angeli, F.S.; Wong, M.L.; Amabile, N.; Prasad, M.; Rassaf, T.; Ottaviani, J.I.; et al. Improvement of endothelial function with dietary flavanols is associated with mobilization of circulating angiogenic cells in patients with coronary artery disease. J. Am. Coll. Cardiol. 2010, 56, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.J.; Chronopoulos, A.K.; Singh, I.; Francis, M.A.; Moriarty, H.; Pike, M.J.; Turner, A.H.; Mann, N.J.; Sinclair, A.J. Dietary flavanols and procyanidin oligomers from cocoa (Theobroma cacao) inhibit platelet function. Am. J. Clin. Nutr. 2003, 77, 1466–1473. [Google Scholar] [CrossRef] [PubMed]
- Hamed, M.S.; Gambert, S.; Bliden, K.P.; Bailon, O.; Singla, A.; Antonino, M.J.; Hamed, F.; Tantry, U.S.; Gurbel, P.A. Dark chocolate effect on platelet activity, C-reactive protein and lipid profile: A pilot study. South. Med. J. 2008, 101, 1203–1208. [Google Scholar] [CrossRef] [PubMed]
- Monagas, M.; Khan, N.; Andres-Lacueva, C.; Casas, R.; Urpi-Sarda, M.; Llorach, R.; Lamuela-Raventos, R.M.; Estruch, R. Effect of cocoa powder on the modulation of inflammatory biomarkers in patients at high risk of cardiovascular disease. Am. J. Clin. Nutr. 2009, 90, 1144–1150. [Google Scholar] [CrossRef] [PubMed]
- Flammer, A.J.; Hermann, F.; Sudano, I.; Spieker, L.; Hermann, M.; Cooper, K.A.; Serafini, M.; Lüscher, T.F.; Ruschitzka, F.; Noll, G.; et al. Dark chocolate improves coronary vasomotion and reduces platelet reactivity. Circulation 2007, 116, 2376–2382. [Google Scholar] [CrossRef] [PubMed]
- Hermann, F.; Spieker, L.E.; Ruschitzka, F.; Sudano, I.; Hermann, M.; Binggeli, C.; Lüscher, T.F.; Riesen, W.; Noll, G.; Corti, R. Dark chocolate improves endothelial and platelet function. Heart 2006, 92, 119–120. [Google Scholar] [CrossRef] [PubMed]
- Engler, M.B.; Engler, M.M.; Chen, C.Y.; Malloy, M.J.; Browne, A.; Chiu, E.Y.; Kwak, H.K.; Milbury, P.; Paul, S.M.; Blumberg, J.; et al. Flavonoid-rich dark chocolate improves endothelial function and increases plasma epicatechin concentrations in healthy adults. J. Am. Coll. Nutr. 2004, 23, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Davison, K.; Coates, A.M.; Buckley, J.D.; Howe, P.R.C. Effect of cocoa flavanols and exercise on cardiometabolic risk factors in overweight and obese subjects. Int. J. Obes. 2008, 32, 1289–1296. [Google Scholar] [CrossRef] [PubMed]
- Heiss, C.; Dejam, A.; Kleinbongard, P.; Schewe, T.; Sies, H.; Kelm, M. Vascular effects of cocoa rich in flavan-3-ols. JAMA 2003, 290, 1030–1031. [Google Scholar] [CrossRef] [PubMed]
- Heiss, C.; Kleinbongard, P.; Dejam, A.; Perré, S.; Schroeter, H.; Sies, H.; Kelm, M. Acute consumption of flavanol-rich cocoa and the reversal of endothelial dysfunction in smokers. J. Am. Coll. Cardiol. 2005, 46, 1276–1283. [Google Scholar] [CrossRef] [PubMed]
- Vlachopoulos, C.; Aznaouridis, K.; Alexopoulos, N.; Economou, E.; Andreadou, I.; Stefanadis, C. Effect of dark chocolate on arterial function in healthy individuals. Am. J. Hypertens. 2005, 18, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Njike, V.Y.; Faridi, Z.; Shuval, K.; Dutta, S.; Kay, C.D.; West, S.G.; Kris-Etherton, P.M.; Katz, D.L. Effects of sugar-sweetened and sugar-free cocoa on endothelial function in overweight adults. Int. J. Cardiol. 2011, 149, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Heiss, C.; Finis, D.; Kleinbongard, P.; Hoffmann, A.; Rassaf, T.; Kelm, M.; Sies, H. Sustained increase in flow-mediated dilation after daily intake of high-flavanol cocoa drink over 1 week. J. Cardiovasc. Pharmacol. 2007, 49, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Balzer, J.; Rassaf, T.; Heiss, C.; Kleinbongard, P.; Lauer, T.; Merx, M.; Heussen, N.; Gross, H.B.; Keen, C.L.; Schroeter, H.; et al. Sustained benefits in vascular function through flavanol-containing cocoa in medicated diabetic patients a double-masked, randomized, controlled trial. J. Am. Coll. Cardiol. 2008, 51, 2141–2149. [Google Scholar] [CrossRef] [PubMed]
- Mills, C.E.; Flury, A.; Marmet, C.; Poquet, L.; Rimoldi, S.F.; Sartori, C.; Rexhaj, E.; Brenner, R.; Allemann, Y.; Zimmermann, D.; et al. Mediation of coffee-induced improvements in human vascular function by chlorogenic acids and its metabolites: Two randomized, controlled, crossover intervention trials. Clin. Nutr. 2017, 36, 1520–1529. [Google Scholar] [CrossRef] [PubMed]
- Dower, J.I.; Geleijnse, J.M.; Gijsbers, L.; Zock, P.L.; Kromhout, D.; Hollman, P.C. Effects of the pure flavonoids epicatechin and quercetin on vascular function and cardiometabolic health: A randomized, double-blind, placebo-controlled, crossover trial. Am. J. Clin. Nutr. 2015, 101, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Heiss, C.; Sansone, R.; Karimi, H.; Krabbe, M.; Schuler, D.; Rodriguez-Mateos, A.; Kraemer, T.; Cortese-Krott, M.M.; Kuhnle, G.G.; Spencer, J.P.; et al. Impact of cocoa flavanol intake on age-dependent vascular stiffness in healthy men: A randomized, controlled, double-masked trial. Age 2015, 37, 9794. [Google Scholar] [CrossRef] [PubMed]
- Lamport, D.J.; Pal, D.; Moutsiana, C.; Field, D.T.; Williams, C.M.; Spencer, J.P.; Butler, L.T. The effect of flavanol-rich cocoa on cerebral perfusion in healthy older adults during conscious resting state: A placebo controlled, crossover, acute trial. Psychopharmacology 2015, 232, 3227–3234. [Google Scholar] [CrossRef] [PubMed]
- Sorond, F.A.; Lipsitz, L.A.; Hollenberg, N.K.; Fisher, N.D. Cerebral blood flow response to flavanol-rich cocoa in healthy elderly humans. Neuropsychiatr. Dis. Treat. 2008, 4, 433–440. [Google Scholar] [PubMed]
- Desjardins, M.B.A.; Desautels, A.; Marquis, L.; Soucy, J.; Montplaisir, J.; Zadra, A. Effects of sleep deprivation on brain perfusion patterns in sleepwalkers’ wakefulness and slow wave sleep. J. Sleep Sleep Disord. Res. 2017, 40, A273–A274. [Google Scholar]
- Asllani, I.H.C.; Borogovac, A.; Tatarina, O.; Brown, T.R.; Basner, R.C.; Stern, Y. Effects of 48hr sleep deprivation on cerebral blood flow measured with arterial spin labeling MRI. Proc. Intl. Soc. Mag. Reson. 2007, 15, 507. [Google Scholar]
- Grolimund, P.; Seiler, R.W. Age dependence of the flow velocity in the basal cerebral-arteries—A transcranial doppler ultrasound study. Ultrasound Med. Biol. 1988, 14, 191–198. [Google Scholar] [CrossRef]
- Fabiani, M.; Gordon, B.A.; Maclin, E.L.; Pearson, M.A.; Brumback-Peltz, C.R.; Low, K.A.; McAuley, E.; Sutton, B.P.; Kramer, A.F.; Gratton, G. Neurovascular coupling in normal aging: A combined optical, ERP and fMRI study. Neuroimage 2014, 85, 592–607. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, A.; Macklin, E.A.; Betensky, R.; Hyman, B.; Smith, E.E.; Blacker, D. The Influence of Vascular Risk Factors and Stroke on Cognition in Late Life: Analysis of the NACC Cohort. Alzheimer Dis. Assoc. Disord. 2015, 29, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Fisher, N.D.; Hollenberg, N.K. Aging and vascular responses to flavanol-rich cocoa. J. Hypertens. 2006, 24, 1575–1580. [Google Scholar] [CrossRef] [PubMed]
- Moncada, S.; Palmer, R.M.J.; Higgs, E.A. Nitric-oxide—Physiology, pathophysiology, and pharmacology. Pharmacol. Rev. 1991, 43, 109–142. [Google Scholar] [PubMed]
- Loke, W.M.; Hodgson, J.M.; Proudfoot, J.M.; McKinley, A.J.; Puddey, I.B.; Croft, K.D. Pure dietary flavonoids quercetin and (−)-epicatechin augment nitric oxide products and reduce endothelin-1 acutely in healthy men. Am. J. Clin. Nutr. 2008, 88, 1018–1025. [Google Scholar] [CrossRef] [PubMed]
- Ward, M.R.; Thompson, K.A.; Isaac, K.; Vecchiarelli, J.; Zhang, Q.; Stewart, D.J.; Kutryk, M.J. Nitric oxide synthase gene transfer restores activity of circulating angiogenic cells from patients with coronary artery disease. Mol. Ther. 2011, 19, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Iadecola, C. Nitric oxide: Roles in neurovascular regulation and ischemic brain injury. Nitric Oxide Biol. Chem. 2012, 27, S3. [Google Scholar] [CrossRef]
- Gallo, E.F.; Iadecola, C. Neuronal nitric oxide contributes to neuroplasticity-associated protein expression through cGMP, protein kinase G, and extracellular signal-regulated kinase. J. Neurosci. 2011, 31, 6947–6955. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, M.; Etani, H.; Yagita, Y.; Kinoshita, N.; Nukada, T. Diminished reserve for cerebral vasomotor response to l-arginine in the elderly: Evaluation by transcranial Doppler sonography. Gerontology 2001, 47, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Kamper, A.M.; Spilt, A.; de Craen, A.J.; van Buchem, M.A.; Westendorp, R.G.; Blauw, G.J. Basal cerebral blood flow is dependent on the nitric oxide pathway in elderly but not in young healthy men. Exp. Gerontol. 2004, 39, 1245–1248. [Google Scholar] [CrossRef] [PubMed]
- Toda, N. Age-related changes in endothelial function and blood flow regulation. Pharmacol. Ther. 2012, 133, 159–176. [Google Scholar] [CrossRef] [PubMed]
- Lamport, D.J.; Lawton, C.L.; Merat, N.; Jamson, H.; Myrissa, K.; Hofman, D.; Chadwick, H.K.; Quadt, F.; Wightman, J.D.; Dye, L. Concord grape juice, cognitive function, and driving performance: A 12-wk, placebo-controlled, randomized crossover trial in mothers of preteen children. Am. J. Clin. Nutr. 2016, 103, 775–783. [Google Scholar] [CrossRef] [PubMed]
- Haskell-Ramsay, C.F.; Stuart, R.C.; Okello, E.J.; Watson, A.W. Cognitive and mood improvements following acute supplementation with purple grape juice in healthy young adults. Eur. J. Nutr. 2017, 56, 2621–2631. [Google Scholar] [CrossRef] [PubMed]
- Fisher, N.D.; Hughes, M.; Gerhard-Herman, M.; Hollenberg, N.K. Flavanol-rich cocoa induces nitric-oxide-dependent vasodilation in healthy humans. J. Hypertens. 2003, 21, 2281–2286. [Google Scholar] [CrossRef] [PubMed]
- Toda, N.; Nakanishi-Toda, M. How mental stress affects endothelial function. Pflugers Arch. Eur. J. Physiol. 2011, 462, 779–794. [Google Scholar] [CrossRef] [PubMed]
| Reference | Sample | Design | Dose, Duration | Methods | Effects of Epicatechin |
|---|---|---|---|---|---|
| Francis et al. (2006) [8] | n = 16 females (18–30 years) | RDBPC crossover | 172 mg CF (31 mg epicatechin). Five days | Switch task; fMRI; HR (1.5 h PD) | Increased BOLD response in the dorsolateral prefrontal cortex, parietal cortex and ACC. No effects on cognition. |
| Scholey et al. (2010) [9] | n = 30 (18–35 years; mean 22) | RDBPC crossover | 520 mg CF; 994 mg CF (94/184 mg epicatechin). Acute | 2 serial subtraction tasks (3 and 7 s); RVIP; mental fatigue; STAI-Y1 (1.5 h PD) | Increased correct serial 3 subtractions; 94 mg attenuated mental fatigue; 184 mg improved RVIP RT but increased serial 7 subtraction errors. |
| Boolani et al. (2017) [10] | n = 23 (17 male) (mean 20 years) | RDBPC crossover | 499 mg CF (epicatechin NK); 499 mg CF+70 mg caff; 66 mg caff. Acute | Serial subtractions (3 and 7 s); Bakan; CPT; motivation; mood; salivary methylxanthines (baseline and 22, 60 and 98 min PD) | CF vs placebo: decreased Bakan RT and FA. CF vs. CF + caff: decreased Bakan correct and increased omission errors. CF + caff vs. caff: decreased anxiety. |
| Tsukamoto et al. (2018) [11] | n = 10 males (mean 23 years) | RSBPC crossover | 563 mg CF (epicatechin NK) | 2-part Stroop; Face-name matching; FAS; mental fatigue; concentration; motivation: HR; MAP; glucose; lactate (baseline and 30 and 60 min PD at rest and 100, 130 and 160 min PD after exercise) | Improved Stroop interference. |
| Decroix et al. (2016) [12] | n = 12 male (mean 30 years) | RDBPC crossover | 900 mg CF (185 mg epicatechin). Acute | 2-part Stroop; NIRS; BDNF (baseline and 95 min PD at rest and 145 min PD after exercise) | Increased ΔHbO2 during word-colour Stroop at rest. |
| Grassi et al. (2016) [13] | n = 32 (16 male) (mean 25 years) | RDBPC crossover | 520 mg CF (447 mg epicatechin). Acute | KSS; PVT; 2-back; FMD; BP; PWV (baseline ‘sleep’ condition and 90 min PD in ‘deprivation’ condition following one-night total sleep deprivation) | Preserved 2-back accuracy in women after ‘deprivation’. SBP, DBP and pulse pressure lower after CF vs. control. Negative effects of ‘deprivation’ on FMD and PWV counteracted by CF. FMD correlated with 2-back accuracy in ‘sleep’. |
| Field et al. (2011) [14] | n = 30 (18–25 years) | RSBPC crossover | 773 mg CF (epicatechin NK). Acute | CS; motion coherence threshold; motion integration time threshold; visual SWM; CRT (2 h PD) | Improved CS, improved WM accuracy and speeded motion integration and CRT. |
| Massee et al. (2015) [15] | n = 40 (18–40 years, mean 24) | RDBPC parallel groups | 250 mg CF (epicatechin NK). Acute/30 days | TCD of CCA, SUCCAB, CDB × 3, mental fatigue and stress before and after CDB, (baseline and 2 h acutely and at 30 days) | Decrease in fatigue pre-CDB and increase in sevens correct during the first repetition acutely. Stress lower at 30 days in the placebo group. |
| Pase et al. (2013) [16] | n = 72 (40–65 years) | RDBPC parallel groups | 250 mg CF; 500 mg (25/50 mg epicatechin). Acute/30 days | Immediate WR; Simple RT; DV; Choice RT; Tracking; Spatial WM; Numeric WM; Delayed WR; Word Recognition; Picture Recognition; Bond-Lader VAS (baseline, 1, 2.5 and 4 h acutely and at 30 days) | Increased calm and content at 30 days. |
| Camfield et al. (2011) [20] | n = 63 (40–65 years; mean 52) | RDBPC parallel groups | 250 mg CF; 500 mg CF (25/50 mg epicatechin). Thirty days | SST-SSVEP; SWM (baseline and PD) | Decreased SSVEP amplitude (25 mg) and increased latency (25 and 50 mg) in posterior parietal regions. |
| Brickman et al. (2014) [22] | n = 37 (50–69 years) | RDBPC parallel groups | 900 mg CF (138 mg epicatechin). Twelve weeks | ModBent; CBV dentate gyrus (fMRI) (baseline and PD) | ModBent RT 630 ms faster in CF vs. control. CBV enhanced in CF; a correlation between change in cognition and CBV. |
| Crews et al. (2008) [23] | n = 90 (≥60 years; mean 69) MCI (≥24 MMSE) | RDBPC parallel groups | 755 mg CF (epicatechin NK). Six weeks | BSRT; WMS-III Faces I and II; TMT; Stroop; WAIS-III DSST; total cholesterol (HDL, LDL, VLDL); triacylglycerol; C-reactive protein (baseline and 6 weeks). A-DACL General Activation; SBP; DBP; HR (baseline, and at 2 h PD at 3 and 6 weeks) | No positive effects. CF increased HR (3 and 6 weeks). |
| Sorond et al. (2013) [24] | n = 60 (mean 73 years) hypertension and/or T2D | RDBPC parallel groups | 1218 mg CF (219 mg epicatechin). A duration of 24 h/30 days | MMSE; TMT-A and B; TCD of MCA to assess neurovascular coupling (baseline and PD) | No effects in the primary analysis. |
| Neshatdoust et al. (2016) [25] | n = 40 (22 male) (62–75 years, mean 68) | RDBPC crossover | 494 mg CF (89 mg epicatechin). Twenty-eight days | Go-No-Go; Stroop; plus-minus; TMT; letter memory; free and delayed WR; word and face recognition; serial sevens; spatial delayed recall; virtual 3D radial arm maze; word stem completion; DSST; RVIP (baseline and PD) | Significant increase in global cognition and BDNF. |
| Desideri et al. (2012) [26] | n = 90 (65–82 years) MCI | RDBPC parallel groups | 520 mg CF; 993 mg CF (95/185 mg epicatechin). Eight weeks | MMSE; TMT-A and B; VF (baseline and PD) | Increased speed of TMT-A and TMT-B. A total of 185 mg improved VF. Reduced IR, BP and LP, with IR explaining ~40% of composite z score variability |
| Mastroiacovo et al. (2015) [26] | n = 90 (~69 years) | RDBPC parallel groups | 520 mg CF; 993 mg CF (95/185 mg epicatechin). Eight weeks | MMSE; TMT-A; TMT-B; VF (baseline and PD) | Increased speed of TMT-A and TMT-B. A total of 185 mg improved VF. Reduced IR, BP and LP, with IR explaining ~17% of composite z score variability. |
| Marsh et al. (2017) [28] | n = 12 (post-menopausal women) (77 years) | RSBPC crossover | 200 mg CF; 395 mg CF (25 mg/49 mg epicatechin). Acute | Detection task; n-back (1 and 2 back); list learning and recall; continuous paired-association learning (separate day baseline and 60 min PD). FMD; MAP; HR; TCD of MCA (baseline and 60 min PD). | Milk and dark chocolate increased FMD, decreased CBV at rest and during tasks with no effects on cognition or MAP. |
| Age | 18–40 | 40–65 | >50 | References | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Duration | ||||||||||||||||||||
| Acute | [8] |  | [9,10] |  | [11,13,14] |  | [12] |  | [15] |  | [16] |  | [28] |  | [85] |  | Francis et al. (2006) [8]; Scholey et al. (2010) [9]; Boolani et al. (2017) [10]; Tsukamoto et al. (2018) [11]; Decroix et al. (2016) [12]; Grassi et al. (2016) [13]; Field et al. (2011) [14]; Massee et al. (2015) [15]; Pase et al. (2013) [16]; Marsh et al. (2017) [28]; Lamport et al. (2015) [85] | |||
 |  |  |  |  | ||||||||||||||||
 | ||||||||||||||||||||
| 1–14 days | [8] |  | [24] |  | [86] |  | Francis et al. (2006) [8]; Sorond et al. (2013) [24]; Sorond et al. (2008) [86] | |||||||||||||
 |  | |||||||||||||||||||
| 28–30 days | [15] |  | [16] |  | [20] |  | [25] |  | Massee et al. (2015) [15]; Pase et al. (2013) [16]; Camfield et al. (2011) [20]; Neshatdoust et al. (2016) [25] | |||||||||||
 |  |  | ||||||||||||||||||
 | ||||||||||||||||||||
| 6 weeks | [23] |  | Crews et al. (2008) [23] | |||||||||||||||||
| 8 weeks | [26] |  | [27] |  | Desideri et al. (2012) * [26]; Mastroiacovo et al. (2015) [27] | |||||||||||||||
| 12 weeks | [22] |  | Brickman et al. (2014) [22] | |||||||||||||||||
 | ||||||||||||||||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haskell-Ramsay, C.F.; Schmitt, J.; Actis-Goretta, L. The Impact of Epicatechin on Human Cognition: The Role of Cerebral Blood Flow. Nutrients 2018, 10, 986. https://doi.org/10.3390/nu10080986
Haskell-Ramsay CF, Schmitt J, Actis-Goretta L. The Impact of Epicatechin on Human Cognition: The Role of Cerebral Blood Flow. Nutrients. 2018; 10(8):986. https://doi.org/10.3390/nu10080986
Chicago/Turabian StyleHaskell-Ramsay, Crystal F., Jeroen Schmitt, and Lucas Actis-Goretta. 2018. "The Impact of Epicatechin on Human Cognition: The Role of Cerebral Blood Flow" Nutrients 10, no. 8: 986. https://doi.org/10.3390/nu10080986
APA StyleHaskell-Ramsay, C. F., Schmitt, J., & Actis-Goretta, L. (2018). The Impact of Epicatechin on Human Cognition: The Role of Cerebral Blood Flow. Nutrients, 10(8), 986. https://doi.org/10.3390/nu10080986





