Postprandial Glycaemic, Hormonal and Satiety Responses to Rice and Kiwifruit Preloads in Chinese Adults: A Randomised Controlled Crossover Trial
Abstract
1. Introduction
2. Materials and Methods
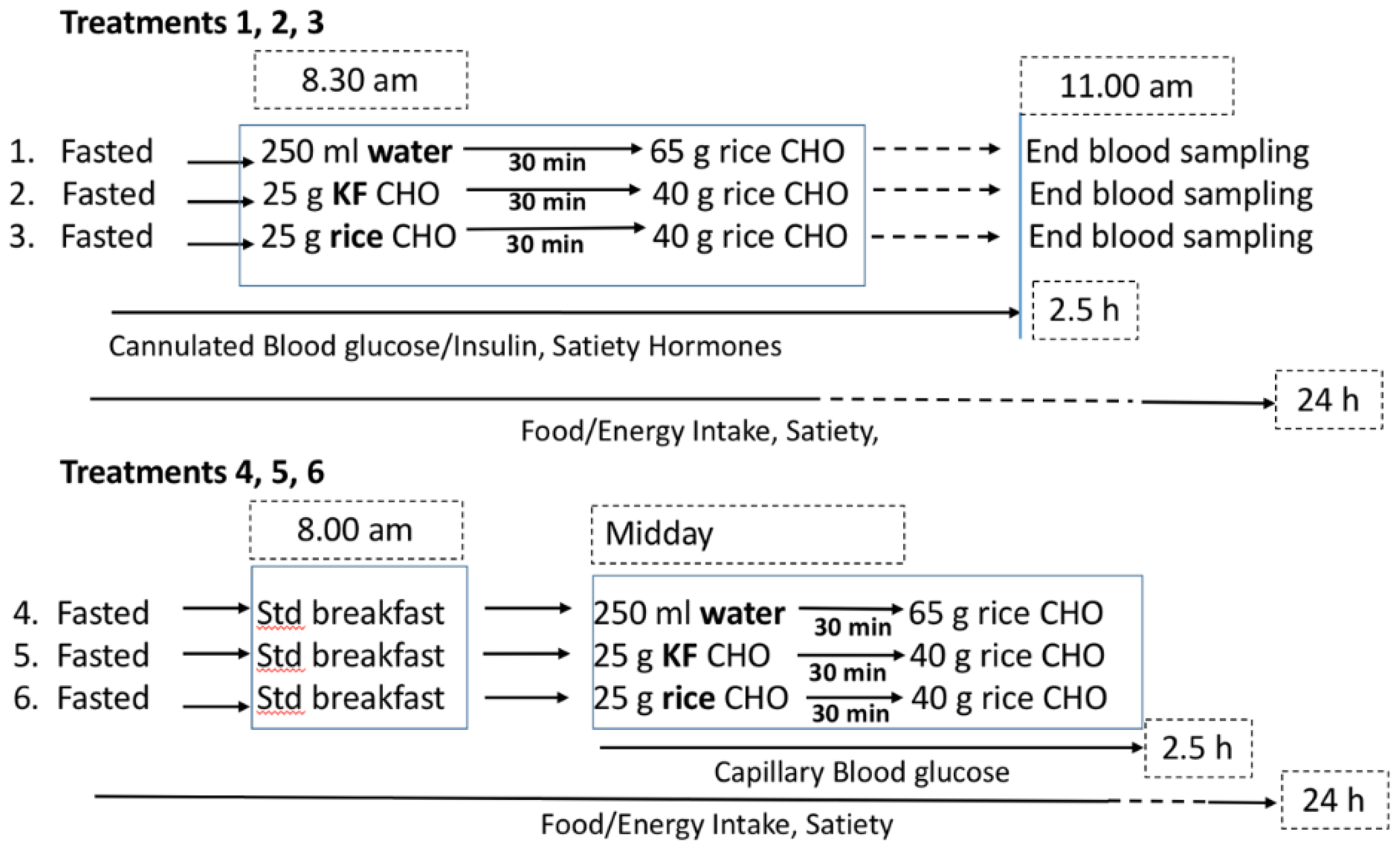
2.1. Ethics and Design
2.2. Meal Components
2.3. Participants
2.4. Blood Sampling
2.4.1. Breakfast Test
- How hungry do you feel at this moment? (Not at all hungry ------ Extremely hungry)
- How full does your stomach feel at this moment? (Not at all full ------ Extremely full)
- How strong is your desire to eat at this moment? (Very weak ------ Very strong)
- How much food do you think you could eat at this moment? (Nothing at all ------ A very large amount).
2.4.2. Lunch Test
2.5. Blood Analysis
2.6. Energy Intake
2.7. Statistical Methods
3. Results
3.1. Blood Glucose
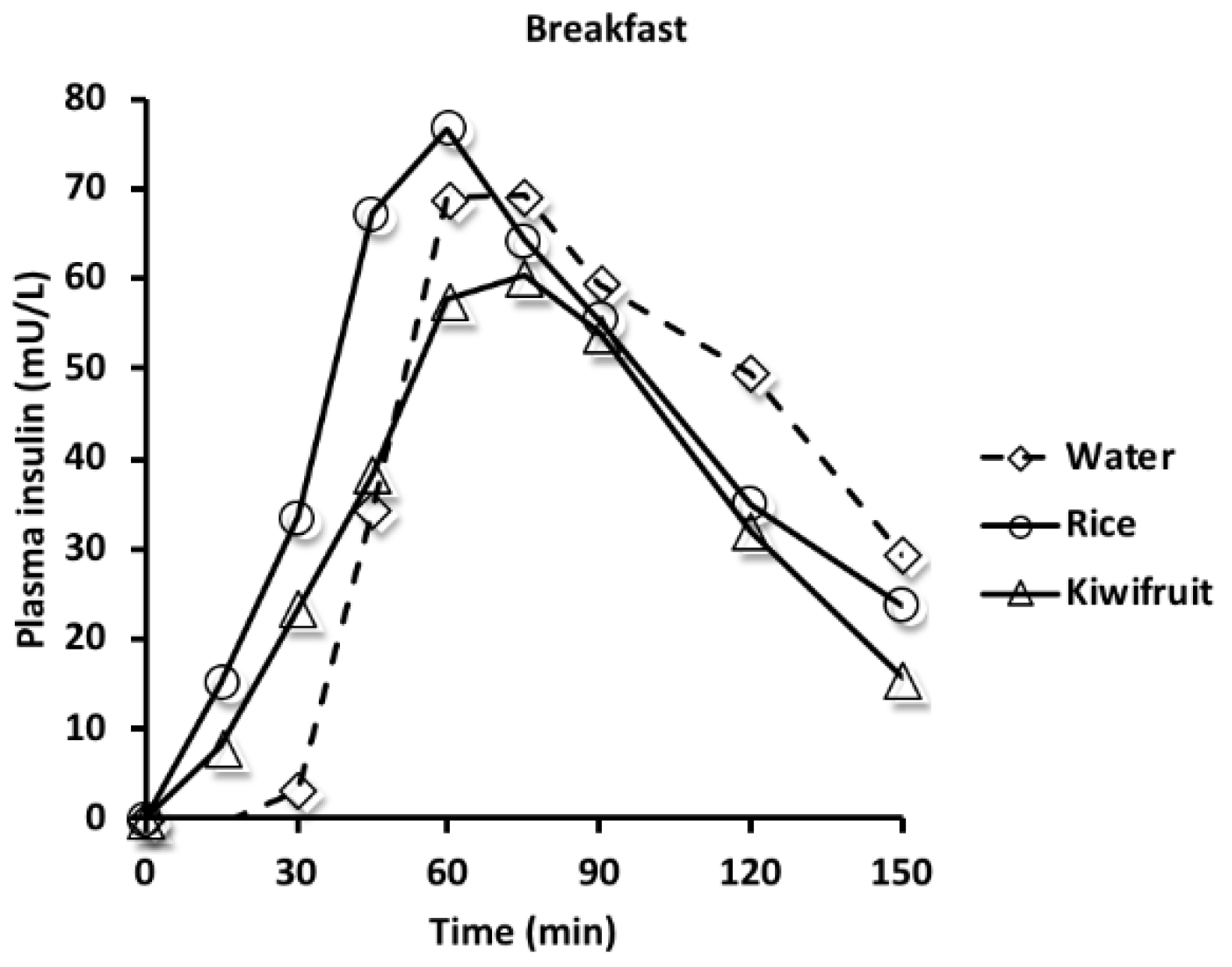
3.2. Hormones
3.3. Satiety and Subsequent Energy Intake
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ceriello, A.; Colagiuri, S. International Diabetes Federation guideline for management of postmeal glucose: A review of recommendations. Diabet. Med. 2008, 25, 1151–1156. [Google Scholar] [CrossRef] [PubMed]
- Trico, D.; Filice, E.; Trifiro, S.; Natali, A. Manipulating the sequence of food ingestion improves glycemic control in type 2 diabetic patients under free-living conditions. Nutr. Diabetes 2016, 6, e226. [Google Scholar] [CrossRef] [PubMed]
- Stacher, G.; Bergmann, H.; Gaupmann, G.; Schneider, C.; Kugi, A.; Hobart, J.; Binder, A.; Mittelbach-Steiner, G. Fat preload delays gastric emptying: Reversal by cisapride. Br. J. Clin. Pharmacol. 1990, 30, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Gentilcore, D.; Chaikomin, R.; Jones, K.L.; Russo, A.; Feinle-Bisset, C.; Wishart, J.M.; Rayner, C.K.; Horowitz, M. Effects of fat on gastric emptying of and the glycemic, insulin, and incretin responses to a carbohydrate meal in type 2 diabetes. J. Clin. Endocrinol. Metab. 2006, 91, 2062–2067. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Little, T.J.; Bound, M.J.; Borg, M.; Zhang, X.; Deacon, C.F.; Horowitz, M.; Jones, K.L.; Rayner, C.K. A protein preload enhances the glucose-lowering efficacy of vildagliptin in type 2 diabetes. Diabetes Care 2016, 39, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Stevens, J.E.; Cukier, K.; Maddox, A.F.; Wishart, J.M.; Jones, K.L.; Clifton, P.M.; Horowitz, M.; Rayner, C.K. Effects of a protein preload on gastric emptying, glycemia, and gut hormones after a carbohydrate meal in diet-controlled type 2 diabetes. Diabetes Care 2009, 32, 1600–1602. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, E.; Schnider, S.; Palmvig, B.; Tauber-Lassen, E.; Pedersen, O. Intake of a diet high in trans monounsaturated fatty acids or saturated fatty acids. Effects on postprandial insulinemia and glycemia in obese patients with NIDDM. Diabetes Care 1997, 20, 881–887. [Google Scholar] [CrossRef] [PubMed]
- Diekman, C.; Malcolm, K. Consumer perception and insights on fats and fatty acids: Knowledge on the quality of diet fat. Ann. Nutr. Metab. 2009, 54, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Nuttall, F.Q.; Gannon, M.C. Metabolic response of people with type 2 diabetes to a high protein diet. Nutr. Metab. (Lond.) 2004, 1, 6. [Google Scholar] [CrossRef] [PubMed]
- Baltali, M.; Korkmaz, M.E.; Kiziltan, H.T.; Muderris, I.H.; Ozin, B.; Anarat, R. Association between postprandial hyperinsulinemia and coronary artery disease among non-diabetic women: A case control study. Int. J. Cardiol. 2003, 88, 215–221. [Google Scholar] [CrossRef]
- Douros, A.; Dell’Aniello, S.; Yu, O.H.Y.; Filion, K.B.; Laurent, A.; Suissa, S. Sulfonylureas as second line drugs in type 2 diabetes and the risk of cardiovascular and hypoglycaemic events: Population based cohort study. Br. Med. J. 2018, 362. [Google Scholar] [CrossRef] [PubMed]
- Robertson, L.; Waugh, N.; Robertson, A. Protein restriction for diabetic renal disease. Cochrane Database Syst. Rev. 2007, 4. [Google Scholar] [CrossRef] [PubMed]
- Holmstrup, M.E.; Owens, C.M.; Fairchild, T.J.; Kanaley, J.A. Effect of meal frequency on glucose and insulin excursions over the course of a day. e-SPEN Eur. e-J. Clin. Nutr. Metab. 2010, 5, e277–e280. [Google Scholar] [CrossRef]
- Papakonstantinou, E.; Kechribari, I.; Mitrou, P.; Trakakis, E.; Vassiliadi, D.; Georgousopoulou, E.; Zampelas, A.; Kontogianni, M.D.; Dimitriadis, G. Effect of meal frequency on glucose and insulin levels in women with polycystic ovary syndrome: A randomised trial. Eur. J. Clin. Nutr. 2016, 70, 646. [Google Scholar] [CrossRef] [PubMed]
- Munsters, M.J.; Saris, W.H. Effects of meal frequency on metabolic profiles and substrate partitioning in lean healthy males. PLoS ONE 2012, 7, e38632. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, O.; Winther, E.; Hermansen, K. Postprandial glucose and insulin responses to rolled oats ingested raw, cooked or as a mixture with raisins in normal subjects and type 2 diabetic patients. Diabet. Med. 1989, 6, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Muraki, I.; Imamura, F.; Manson, J.E.; Hu, F.B.; Willett, W.C.; van Dam, R.M.; Sun, Q. Fruit consumption and risk of type 2 diabetes: Results from three prospective longitudinal cohort studies. BMJ 2013, 347, f5001. [Google Scholar] [CrossRef] [PubMed]
- Montagnese, C.; Santarpia, L.; Buonifacio, M.; Nardelli, A.; Caldara, A.R.; Silvestri, E.; Contaldo, F.; Pasanisi, F. European food-based dietary guidelines: A comparison and update. Nutrition 2015, 31, 908–915. [Google Scholar] [CrossRef] [PubMed]
- Government of Canada. Canada’s Food Guides. Available online: https://www.canada.ca/en/health-canada/services/canada-food-guides.html (accessed on 18 July 2018).
- U.S. Department of Health and Human Services; U.S. Department of Agriculture. 2015–2020 Dietary Guidelines for Americans, 8th ed.; U.S. Department of Health and Human Services: Washington, DC, USA, December 2015. Available online: http://health.gov/dietaryguidelines/2015/guidelines/ (accessed on 18 July 2018).
- Lustig, R.H.; Schmidt, L.A.; Brindis, C.D. Public health: The toxic truth about sugar. Nature 2012, 482, 27–29. [Google Scholar] [CrossRef] [PubMed]
- Tappy, L.; Le, K.A. Metabolic effects of fructose and the worldwide increase in obesity. Physiol. Rev. 2010, 90, 23–46. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, D.S. Examining the health effects of fructose. JAMA 2013, 310, 33–34. [Google Scholar] [CrossRef] [PubMed]
- Teff, K.L.; Grudziak, J.; Townsend, R.R.; Dunn, T.N.; Grant, R.W.; Adams, S.H.; Keim, N.L.; Cummings, B.P.; Stanhope, K.L.; Havel, P.J. Endocrine and metabolic effects of consuming fructose- and glucose-sweetened beverages with meals in obese men and women: Influence of insulin resistance on plasma triglyceride responses. J. Clin. Endocrinol. Metab. 2009, 94, 1562–1569. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Monro, J. Kiwifruit remnants from digestion in vitro have functional attributes of potential importance to health. Food Chem. 2012, 135, 2188–2194. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Edwards, H.; Hedderley, D.; Podd, J.; Monro, J. Kiwifruit non-sugar components reduce glycaemic response to co-ingested cereal in humans. Nutrients 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Livingstone, M.B.E.; Robson, P.J.; Welch, R.W.; Burns, A.A.; Burrows, M.S.; McCormack, C. Methodological issues in the assessment of satiety. Scand. J. Nutr. 2000, 44, 98–103. [Google Scholar] [CrossRef]
- Rolls, B.J.; Kim, S.; McNelis, A.L.; Fischman, M.W.; Foltin, R.W.; Moran, T.H. Time course of effects of preloads high in fat or carbohydrate on food intake and hunger ratings in humans. Am. J. Physiol. 1991, 260, R756–R763. [Google Scholar] [CrossRef] [PubMed]
- Kataoka, M.; Venn, B.J.; Williams, S.M.; Te Morenga, L.A.; Heemels, I.M.; Mann, J.I. Glycaemic responses to glucose and rice in people of Chinese and European ethnicity. Diabet. Med. 2013, 30, e101–e107. [Google Scholar] [CrossRef] [PubMed]
- The New Zealand Institute of Plant and Food Research; The New Zealand Ministry of Health. New Zealand food composition database: New Zealand Food Files 2012 Version 01. Available online: http://www.foodcomposition.co.nz (accessed on 30 April 2013).
- Yang, C.; Chang, C.; Lin, J. A comparison between venous and finger-prick blood sampling on values of blood glucose. Int. Conf. Nutr. Food Sci. 2012, 39, 206–210. [Google Scholar]
- Esposito, K.; Ciotola, M.; Carleo, D.; Schisano, B.; Sardelli, L.; Di Tommaso, D.; Misso, L.; Saccomanno, F.; Ceriello, A.; Giugliano, D. Post-meal glucose peaks at home associate with carotid intima-media thickness in type 2 diabetes. J. Clin. Endocrinol. Metab. 2008, 93, 1345–1350. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Standards of medical care in diabetes-2018 abridged for primary care providers. Clin. Diabetes 2018, 36, 14–37. [Google Scholar]
- Atkinson, F.S.; Foster-Powell, K.; Brand-Miller, J.C. International tables of glycemic index and glycemic load values: 2008. Diabetes Care 2008, 31, 2281–2283. [Google Scholar] [CrossRef] [PubMed]
- Clifton, P.M.; Galbraith, C.; Coles, L. Effect of a low dose whey/guar preload on glycemic control in people with type 2 diabetes—A randomised controlled trial. Nutr. J. 2014, 13, 103. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Xu, Q.; Jiang, R.; Han, T.; Sun, C.; Na, L. Dietary protein consumption and the risk of type 2 diabetes: A systematic review and meta-analysis of cohort studies. Nutrients 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Holman, R.R.; Haffner, S.M.; McMurray, J.J.; Bethel, M.A.; Holzhauer, B.; Hua, T.A.; Belenkov, Y.; Boolell, M.; Buse, J.B.; Buckley, B.M.; et al. Effect of nateglinide on the incidence of diabetes and cardiovascular events. N. Engl. J. Med. 2010, 362, 1463–1476. [Google Scholar] [PubMed]
- Numao, S.; Kawano, H.; Endo, N.; Yamada, Y.; Konishi, M.; Takahashi, M.; Sakamoto, S. Short-term low carbohydrate/high-fat diet intake increases postprandial plasma glucose and glucagon-like peptide-1 levels during an oral glucose tolerance test in healthy men. Eur. J. Clin. Nutr. 2012, 66, 926–931. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, K.; Ihana-Sugiyama, N.; Yamamoto-Honda, R.; Nakamura, T.; Sobe, C.; Kamiya, S.; Kishimoto, M.; Kajio, H.; Kawano, K.; Noda, M. Postprandial glucose surges after extremely low carbohydrate diet in healthy adults. Tohoku J. Exp. Med. 2017, 243, 35–39. [Google Scholar] [CrossRef] [PubMed]
- Tay, J.; Luscombe-Marsh, N.D.; Thompson, C.H.; Noakes, M.; Buckley, J.D.; Wittert, G.A.; Yancy, W.S., Jr.; Brinkworth, G.D. Comparison of low- and high-carbohydrate diets for type 2 diabetes management: A randomized trial. Am. J. Clin. Nutr. 2015, 102, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Saslow, L.R.; Kim, S.; Daubenmier, J.J.; Moskowitz, J.T.; Phinney, S.D.; Goldman, V.; Murphy, E.J.; Cox, R.M.; Moran, P.; Hecht, F.M. A randomized pilot trial of a moderate carbohydrate diet compared to a very low carbohydrate diet in overweight or obese individuals with type 2 diabetes mellitus or prediabetes. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Guldbrand, H.; Dizdar, B.; Bunjaku, B.; Lindstrom, T.; Bachrach-Lindstrom, M.; Fredrikson, M.; Ostgren, C.J.; Nystrom, F.H. In type 2 diabetes, randomisation to advice to follow a low-carbohydrate diet transiently improves glycaemic control compared with advice to follow a low-fat diet producing a similar weight loss. Diabetologia 2012, 55, 2118–2127. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, N.; Vetter, M.L.; Moore, R.H.; Chittams, J.L.; Dalton-Bakes, C.V.; Dowd, M.; Williams-Smith, C.; Cardillo, S.; Wadden, T.A. Effects of a low-intensity intervention that prescribed a low-carbohydrate vs. a low-fat diet in obese, diabetic participants. Obesity (Silver Spring) 2010, 18, 1733–1738. [Google Scholar] [CrossRef] [PubMed]
- Davis, N.J.; Tomuta, N.; Schechter, C.; Isasi, C.R.; Segal-Isaacson, C.J.; Stein, D.; Zonszein, J.; Wylie-Rosett, J. Comparative study of the effects of a 1-year dietary intervention of a low-carbohydrate diet versus a low-fat diet on weight and glycemic control in type 2 diabetes. Diabetes Care 2009, 32, 1147–1152. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.; Wolever, T.M.; Taylor, R.H.; Barker, H.; Fielden, H.; Baldwin, J.M.; Bowling, A.C.; Newman, H.C.; Jenkins, A.L.; Goff, D.V. Glycemic index of foods: A physiological basis for carbohydrate exchange. Am. J. Clin. Nutr. 1981, 34, 362–366. [Google Scholar] [CrossRef] [PubMed]
- Kaur, B.; Quek Yu Chin, R.; Camps, S.; Henry, C.J. The impact of a low glycaemic index (GI) diet on simultaneous measurements of blood glucose and fat oxidation: A whole body calorimetric study. J. Clin. Transl. Endocrinol. 2016, 4, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Shikany, J.M.; Phadke, R.P.; Redden, D.T.; Gower, B.A. Effects of low- and high-glycemic index/glycemic load diets on coronary heart disease risk factors in overweight/obese men. Metabolism 2009, 58, 1793–1801. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Gilhooly, C.H.; Golden, J.K.; Pittas, A.G.; Fuss, P.J.; Cheatham, R.A.; Tyler, S.; Tsay, M.; McCrory, M.A.; Lichtenstein, A.H.; et al. Long-term effects of 2 energy-restricted diets differing in glycemic load on dietary adherence, body composition, and metabolism in CALERIE: A 1-y randomized controlled trial. Am. J. Clin. Nutr. 2007, 85, 1023–1030. [Google Scholar] [CrossRef] [PubMed]
- Wolever, T.M.; Gibbs, A.L.; Mehling, C.; Chiasson, J.L.; Connelly, P.W.; Josse, R.G.; Leiter, L.A.; Maheux, P.; Rabasa-Lhoret, R.; Rodger, N.W.; et al. The Canadian Trial of Carbohydrates in Diabetes (CCD), a 1-y controlled trial of low-glycemic-index dietary carbohydrate in type 2 diabetes: No effect on glycated hemoglobin but reduction in C-reactive protein. Am. J. Clin. Nutr. 2008, 87, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Collier, G.; O’Dea, K. The effect of coingestion of fat on the glucose, insulin, and gastric inhibitory polypeptide responses to carbohydrate and protein. Am. J. Clin. Nutr. 1983, 37, 941–944. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.I.; Yoshino, J.; Stromsdorfer, K.L.; Klein, S.J.; Magkos, F.; Reeds, D.N.; Klein, S.; Mittendorfer, B. Protein ingestion induces muscle insulin resistance independent of leucine-mediated mTOR activation. Diabetes Care 2015, 64, 1555–1563. [Google Scholar] [CrossRef] [PubMed]
- Brand-Miller, J.C.; Stockmann, K.; Atkinson, F.; Petocz, P.; Denyer, G. Glycemic index, postprandial glycemia, and the shape of the curve in healthy subjects: Analysis of a database of more than 1000 foods. Am. J. Clin. Nutr. 2009, 89, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Greenfield, J.R.; Samaras, K.; Chisholm, D.J.; Campbell, L.V. Effect of postprandial insulinemia and insulin resistance on measurement of arterial stiffness (augmentation index). Int. J. Cardiol. 2007, 114, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Silver, H.J.; Dietrich, M.S.; Niswender, K.D. Effects of grapefruit, grapefruit juice and water preloads on energy balance, weight loss, body composition, and cardiometabolic risk in free-living obese adults. Nutr. Metab. (Lond.) 2011, 8. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, S.; Colagiuri, S.; Faramus, E.; Petocz, P.; Brand-Miller, J.C. Postprandial hyperglycemia and insulin sensitivity differ among lean young adults of different ethnicities. J. Nutr. 2002, 132, 2574–2579. [Google Scholar] [CrossRef] [PubMed]


| Component | Kiwifruit | Rice Porridge (Congee) | Steamed Bun |
|---|---|---|---|
| Energy (kJ/100 g) | 238 | 384 | 911 |
| Protein (g/100 g) | 1.02 | 4.1 | 6.6 |
| Fat (g/100 g) | 0.28 | 2.1 | 3.2 |
| Carbohydrate (g/100 g) | 13.1 | 13.9 | 40 |
| Moisture (g/100 g) | 82.4 | 79.7 | 49.1 |
| Ash (g/100 g) | 0.47 | 0.2 | 1.1 |
| Blood Glucose | Mean (SD) | Mean Difference (95% CI) | ||||
|---|---|---|---|---|---|---|
| Water Preload + Rice | Rice Preload + Rice | Kiwifruit Preload + Rice | Rice vs. Water | Kiwifruit vs. Water | Kiwifruit vs. Rice | |
| Breakfast iAUC 1 (mmol/L·min) | 218 (171) | 191 (125) | 153 (93) | −26 (−82, 30) p = 0.357 | −65 (−129, −1) p = 0.047 | −39 (−80, 2) p = 0.063 |
| Lunch iAUC (mmol/L·min) | 365 (185) | 371 (156) | 336 (146) | 6 (−38, 50) p = 0.788 | −29 (−76, 19) p = 0.237 | −35 (−82, 12) p = 0.147 |
| Breakfast peak (mmol/L) | 9.1 (2.0) | 8.4 (1.4) | 8.1 (1.1) | −0.7 (−1.4, 0.1) p = 0.107 | −0.9 (−1.6, −0.2) p = 0.010 | −0.3 (0.2, −0.8) p = 0.264 |
| Lunch peak (mmol/L) | 9.7 (1.6) | 8.7 (1.1) | 8.6 (1.5) | −1.0 (−1.4, −0.7) p < 0.001 | −1.1 (−1.7, −0.5) p < 0.001 | −0.1 (−0.5, 0.4) p = 0.761 |
| Hormone | Mean (SD) | Mean Difference (95% CI) | ||||
|---|---|---|---|---|---|---|
| Water Preload + Rice | Rice Preload + Rice | Kiwifruit Preload + Rice | Rice vs. Water | Kiwifruit vs. Water | Kiwifruit vs. Rice | |
| Insulin iAUC 1 (mU/L·min) | 5962 (2858) | 6552 (3437) | 5167 (2779) | 498 (−688, 1685) p = 0.296 | −887 (−1894, 119) p = 0.152 | −1385 (−2684, −87) p = 0.036 |
| Ghrelin (pg/mL) | 32.9 (25.6) | 35.1 (34.7) | 32.3 (21.2) | 2.2 (−2.5, 6.8) p = 0.359 | −0.6 (−5.3, 4.0) p = 0.787 | −2.8 (−7.4, 1.8) p = 0.235 |
| Glucagon (pg/ml) | 25.3 (10.8) | 24.3 (10.8) | 26.9 (14.9) | −1.0 (−3.0, 1.1) p = 0.353 | 1.6 (−0.5, 3.6) p = 0.134 | 2.5 (0.5, 4.6) p = 0.015 |
| GLP-1 (pg/mL) | 149 (44) | 137 (47) | 142 (41) | −12 (−18, −5) p < 0.001 | −6 (−13, 0.3) p = 0.063 | 5 (−1, 12) p = 0.100 |
| Satiety and Energy Intake | Mean (SD) | Mean Difference (95% CI) | ||||
|---|---|---|---|---|---|---|
| Water Preload + Rice | Rice Preload + Rice | Kiwifruit Preload + Rice | Rice vs. Water | Kiwifruit vs. Water | Kiwifruit vs. Rice | |
| Appetite after breakfast cm·min | 593 (177) | 605 (226) | 687 (218) | 12 (−39, 63) p = 0.650 | 94 (39, 149) p = 0.001 | 82 (26, 138) p = 0.004 |
| Appetite after lunch cm·min | 690 (229) | 631 (242) | 724 (261) | −59 (−103, −15) p = 0.009 | 34 (−19, 86) p = 0.208 | 93 (28, 157) p = 0.005 |
| Energy intake after breakfast (MJ) | 6.08 (2.02) | 5.99 (2.44) | 6.08 (2.34) | −0.09 (−0.94, 0.76) p = 0.836 | 0.00 (−0.88, 0.89) p = 0.995 | 0.09 (−0.72, 0.90) p = 0.823 |
| Energy intake after lunch (MJ) | 4.22 (2.20) | 4.41 (1.87) | 4.32 (1.54) | 0.19 (−0.58, 0.96) p = 0.629 | 0.10 (−0.63, 0.83) p = 0.788 | −0.09 (−0.74, 0.56) p = 0.786 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lubransky, A.; Monro, J.; Mishra, S.; Yu, H.; Haszard, J.J.; Venn, B.J. Postprandial Glycaemic, Hormonal and Satiety Responses to Rice and Kiwifruit Preloads in Chinese Adults: A Randomised Controlled Crossover Trial. Nutrients 2018, 10, 1110. https://doi.org/10.3390/nu10081110
Lubransky A, Monro J, Mishra S, Yu H, Haszard JJ, Venn BJ. Postprandial Glycaemic, Hormonal and Satiety Responses to Rice and Kiwifruit Preloads in Chinese Adults: A Randomised Controlled Crossover Trial. Nutrients. 2018; 10(8):1110. https://doi.org/10.3390/nu10081110
Chicago/Turabian StyleLubransky, Alex, John Monro, Suman Mishra, Hui Yu, Jillian J. Haszard, and Bernard J. Venn. 2018. "Postprandial Glycaemic, Hormonal and Satiety Responses to Rice and Kiwifruit Preloads in Chinese Adults: A Randomised Controlled Crossover Trial" Nutrients 10, no. 8: 1110. https://doi.org/10.3390/nu10081110
APA StyleLubransky, A., Monro, J., Mishra, S., Yu, H., Haszard, J. J., & Venn, B. J. (2018). Postprandial Glycaemic, Hormonal and Satiety Responses to Rice and Kiwifruit Preloads in Chinese Adults: A Randomised Controlled Crossover Trial. Nutrients, 10(8), 1110. https://doi.org/10.3390/nu10081110






