Resistance Training Prevents Muscle Loss Induced by Caloric Restriction in Obese Elderly Individuals: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source
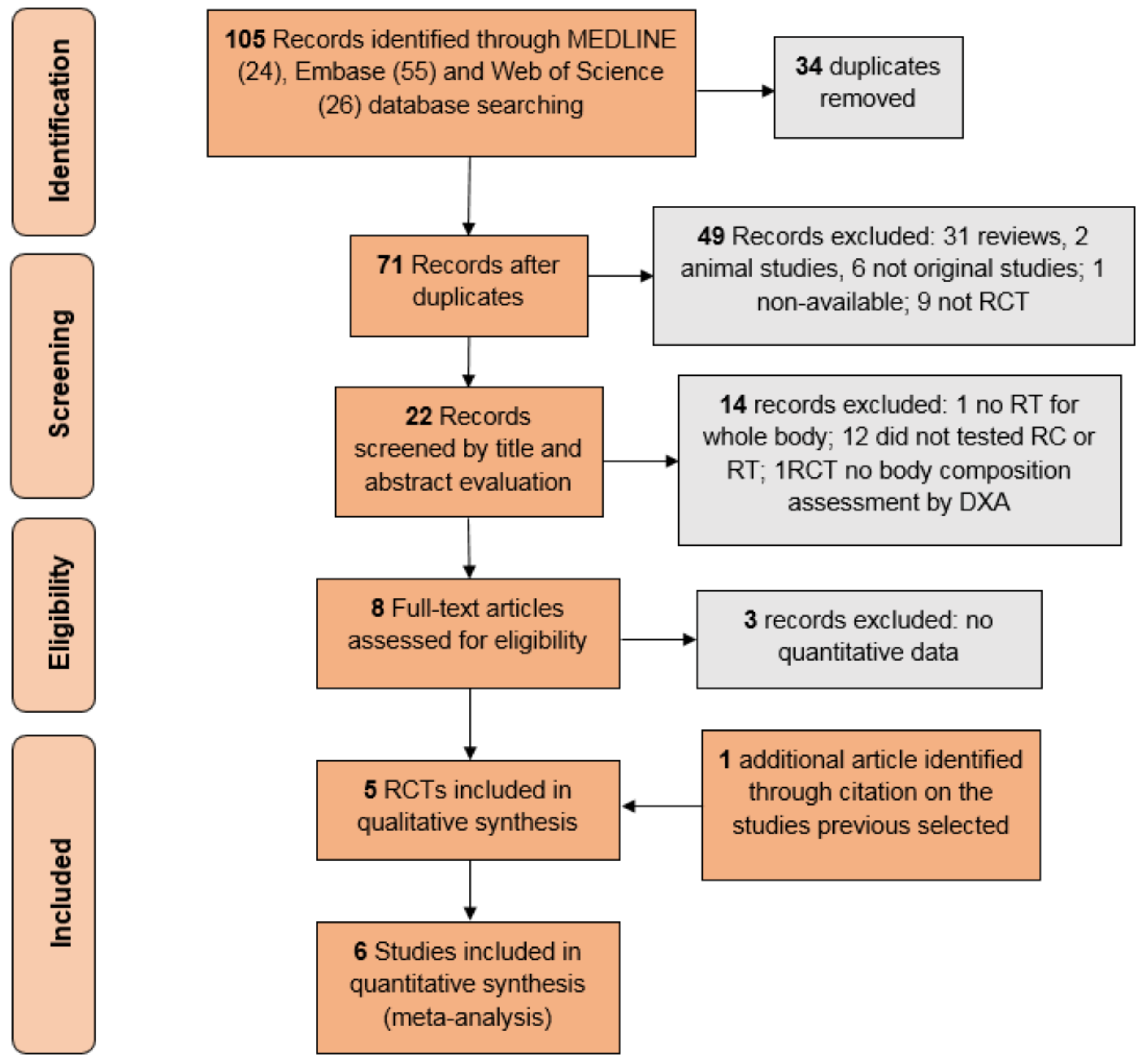
2.2. Study Selection
2.3. Assessment of Risk of Bias
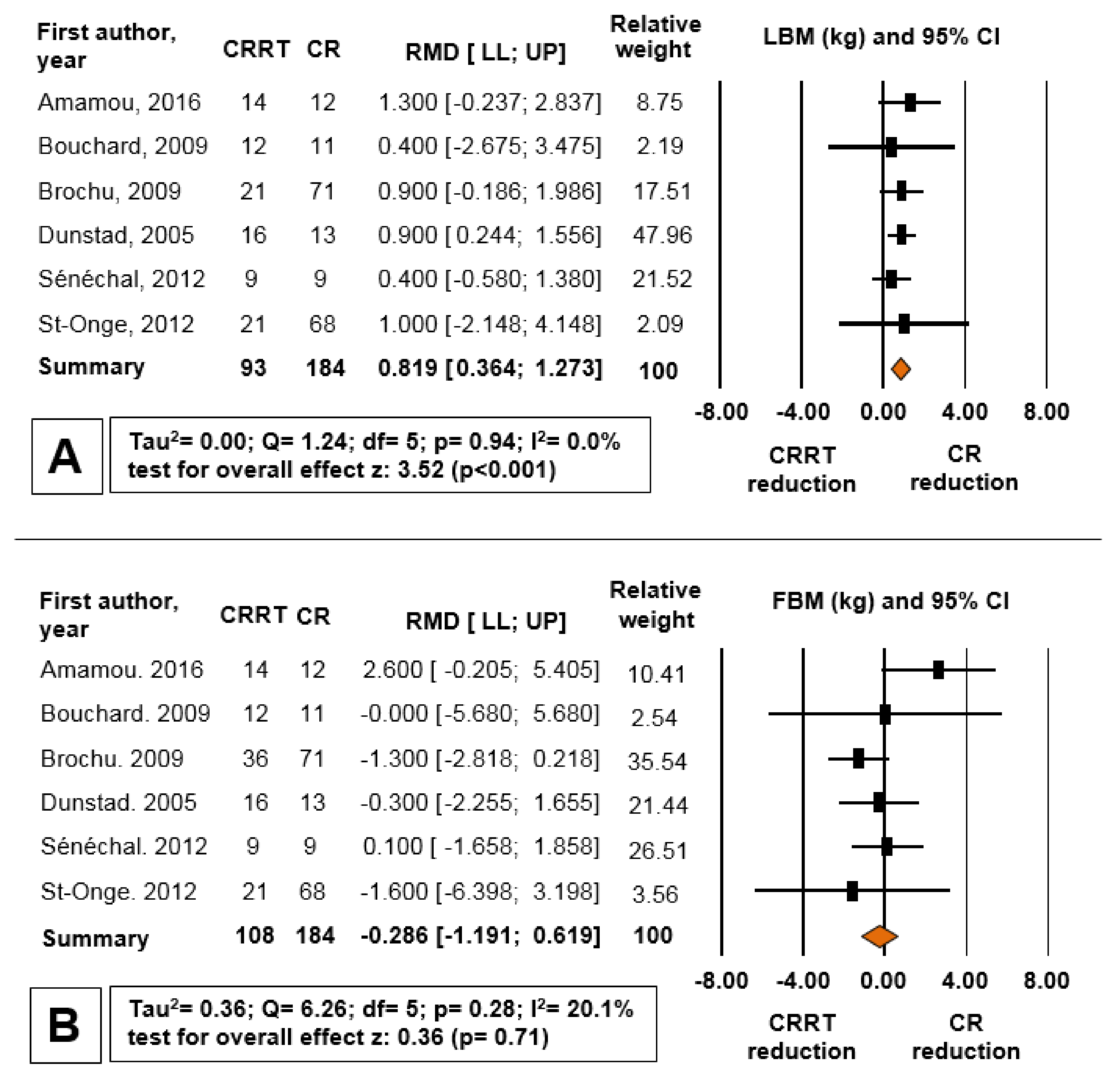
2.4. Statistical Analysis
3. Results
3.1. Studies’ Features
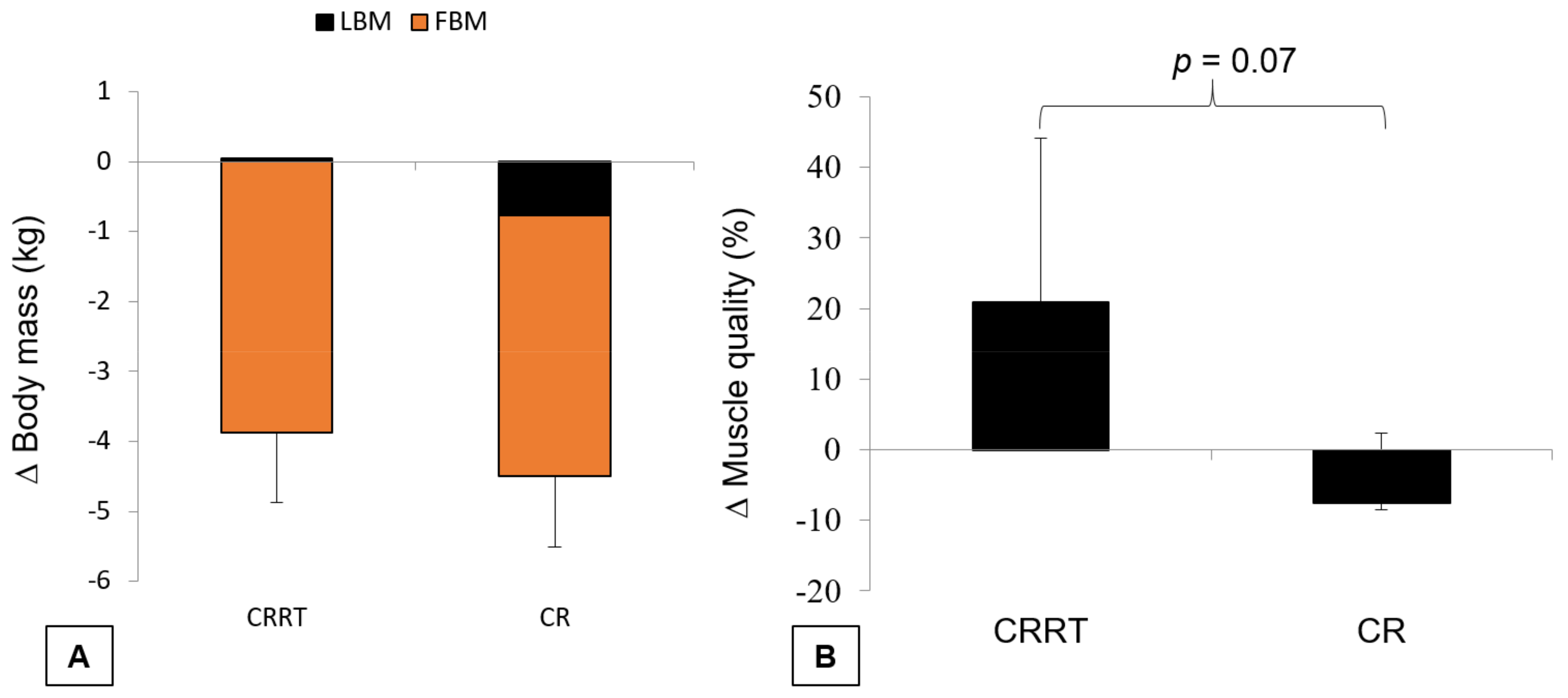
3.2. Evidence Synthesis
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Fontana, L.; Partridge, L. Promoting health and longevity through diet: From model organisms to humans. Cell 2015, 161, 106–118. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, S.J.; Madrigal-Matute, J.; Scheibye-Knudsen, M.; Fang, E.; Aon, M.; Gonzalez-Reyes, J.A.; Cortassa, S.; Kaushik, S.; Gonzalez-Freire, M.; Petel, B.; et al. Effects of Sex, Strain, and Energy Intake on Hallmarks of Aging in Mice. Cell Metab. 2016, 23, 1093–1112. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, P.; Howell, P.R.; Anderson, R.M. Aging and Caloric Restriction Research: A Biological Perspective with Translational Potential. EBioMedicine 2017, 21, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Brestoff, J.R.; Artis, D. Immune regulation of metabolic homeostasis in health and disease. Cell 2015, 161, 146–160. [Google Scholar] [CrossRef] [PubMed]
- Thomou, T.; Mori, M.A.; Dreyfuss, J.M.; Konishi, M.; Sakaguchi, M.; Wolfrum, C.; Rao, T.N.; Winnay, J.N.; Garcia-Martin, R.; Grinspon, S.K.; et al. Adipose-derived circulating miRNAs regulate gene expression in other tissues. Nature 2017, 542, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Reis, F.C.; Branquinho, J.L.; Brandao, B.B.; Guerra, B.A.; Silva, I.D.; Frontini, A.; Thomou, T.; Sartini, L.; Cinti, S.; Kahan, C.R.; et al. Fat-specific Dicer deficiency accelerates aging and mitigates several effects of dietary restriction in mice. Aging (Albany NY) 2016, 8, 1201–1222. [Google Scholar] [CrossRef] [PubMed]
- Amamou, T.; Normandin, E.; Pouliot, J.; Dionne, I.J.; Brochu, M.; Riesco, E. Effect of a High-Protein Energy-Restricted Diet Combined with Resistance Training on Metabolic Profile in Older Individuals with Metabolic Impairments. J. Nutr. Health Aging 2017, 21, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Giannopoulou, I.; Fernhall, B.; Carhart, R.; Weinstock, R.S.; Baynard, T.; Figueroa, A.; Kanaley, J.A. Effects of diet and/or exercise on the adipocytokine and inflammatory cytokine levels of postmenopausal women with type 2 diabetes. Metabolism 2005, 54, 866–875. [Google Scholar] [CrossRef] [PubMed]
- Madsen, E.L.; Rissanen, A.; Bruun, J.M.; Skogstrand, K.; Tonstad, S.; Hougaard, D.M.; Richelsen, B. Weight loss larger than 10% is needed for general improvement of levels of circulating adiponectin and markers of inflammation in obese subjects: A 3-year weight loss study. Eur. J. Endocrinol. 2008, 158, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Weinheimer, E.M.; Sands, L.P.; Campbell, W.W. A systematic review of the separate and combined effects of energy restriction and exercise on fat-free mass in middle-aged and older adults: Implications for sarcopenic obesity. Nutr. Rev. 2010, 68, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Fielding, R.A.; Vellas, B.; Evans, W.J.; Bhasin, S.; Morley, J.E.; Newman, A.B.; Abellan van Kan, G.; Andrieu, S.; Bauer, J.; Breuille, D.; et al. Sarcopenia: An undiagnosed condition in older adults. Current consensus definition: Prevalence, etiology, and consequences. International working group on sarcopenia. J. Am. Med. Dir. Assoc. 2011, 12, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Cermak, N.M.; Res, P.T.; de Groot, L.C.; Saris, W.H.; van Loon, L.J. Protein supplementation augments the adaptive response of skeletal muscle to resistance-type exercise training: A meta-analysis. Am. J. Clin. Nutr. 2012, 96, 1454–1464. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.K.; Quinn, M.A.; Saunders, D.H.; Greig, C.A. Protein Supplementation Does Not Significantly Augment the Effects of Resistance Exercise Training in Older Adults: A Systematic Review. J. Am. Med. Dir. Assoc. 2016, 17, 959.e1–959.e9. [Google Scholar] [CrossRef] [PubMed]
- Nicklas, B.J.; Chmelo, E.; Delbono, O.; Carr, J.J.; Lyles, M.F.; Marsh, A.P. Effects of resistance training with and without caloric restriction on physical function and mobility in overweight and obese older adults: A randomized controlled trial. Am. J. Clin. Nutr. 2015, 101, 991–999. [Google Scholar] [CrossRef] [PubMed]
- Senechal, M.; Bouchard, D.R.; Dionne, I.J.; Brochu, M. The effects of lifestyle interventions in dynapenic-obese postmenopausal women. Menopause 2012, 19, 1015–1021. [Google Scholar] [CrossRef] [PubMed]
- Brochu, M.; Malita, M.F.; Messier, V.; Doucet, E.; Strychar, I.; Lavoie, J.M.; Prud’homme, D.; Rabasa-Lhoret, R. Resistance training does not contribute to improving the metabolic profile after a 6-month weight loss program in overweight and obese postmenopausal women. J. Clin. Endocrinol. Metable 2009, 94, 3226–3233. [Google Scholar] [CrossRef] [PubMed]
- Chmelo, E.A.; Beavers, D.P.; Lyles, M.F.; Marsh, A.P.; Nicklas, B.J.; Beavers, K.M. Legacy effects of short-term intentional weight loss on total body and thigh composition in overweight and obese older adults. Nutr. Diabetes 2016, 6, e203. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Tsujimoto, T.; So, R.; Tanaka, K. Changes in lower extremity muscle mass and muscle strength after weight loss in obese men: A prospective study. Obes. Res. Clin. Pract. 2015, 9, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, D.R.; Soucy, L.; Senechal, M.; Dionne, I.J.; Brochu, M. Impact of resistance training with or without caloric restriction on physical capacity in obese older women. Menopause 2009, 16, 66–72. [Google Scholar] [CrossRef]
- St-Onge, M.; Rabasa-Lhoret, R.; Strychar, I.; Faraj, M.; Doucet, E.; Lavoie, J.M. Impact of energy restriction with or without resistance training on energy metabolism in overweight and obese postmenopausal women: A Montreal Ottawa New Emerging Team group study. Menopause 2013, 20, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Ballor, D.L.; Katch, V.L.; Becque, M.D.; Marks, C.R. Resistance weight training during caloric restriction enhances lean body weight maintenance. Am. J. Clin. Nutr. 1988, 47, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Kordi, R.; Dehghani, S.; Noormohammadpour, P.; Rostami, M.; Mansournia, M.A. Effect of abdominal resistance exercise on abdominal subcutaneous fat of obese women: A randomized controlled trial using ultrasound imaging assessments. J. Manipul. Physiol. Therap. 2015, 38, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, C.; An, P.; Rice, T.; Skinner, J.S.; Wilmore, J.H.; Gagnon, J.; Leon, A.S.; Rao, D.C. Familial aggregation of VO(2max) response to exercise training: Results from the HERITAGE Family Study. J. Appl. Physiol. 1999, 87, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Dunstan, D.W.; Daly, R.M.; Owen, N.; Jolley, D.; Vulikh, E.; Shaw, J.; Zimmet, P. Home-based resistance training is not sufficient to maintain improved glycemic control following supervised training in older individuals with type 2 diabetes. Diabetes Care 2005, 28, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Dunstan, D.W.; Daly, R.M.; Owen, N.; Jolley, D.; De Courten, M.; Shaw, J.; Zimmet, P. High-intensity resistance training improves glycemic control in older patients with type 2 diabetes. Diabetes Care 2002, 25, 1729–1736. [Google Scholar] [CrossRef] [PubMed]
- Maher, C.G.; Sherrington, C.; Herbert, R.D.; Moseley, A.M.; Elkins, M. Reliability of the PEDro scale for rating quality of randomized controlled trials. Phys. Ther. 2003, 83, 713–721. [Google Scholar] [PubMed]
- Boreinstein, M.; Hedges, L.; Higgins, J.; Rothstein, H.R. Introduction to Meta-Analysis; Wiley: Chichester, UK, 2009. [Google Scholar]
- Csapo, R.; Alegre, L.M. Effects of resistance training with moderate vs heavy loads on muscle mass and strength in the elderly: A meta-analysis. Scand. J. Med. Sci. Sports 2016, 26, 995–1006. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.S.; Morey, M.C.; Dutta, C.; Manini, T.M.; Weltman, A.L.; Nelson, M.E.; Morgan, A.L.; Senior, J.G.; Seyffarth, C.; Buchner, D.M. Activity-related energy expenditure in older adults: A call for more research. Med. Sci. Sports Exerc. 2014, 46, 2335–2340. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, J.E.; Blair, S.N.; Jakicic, J.M.; Manore, M.M.; Rankin, J.W.; Smith, B.K. American College of Sports Medicine Position Stand. Appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Med. Sci. Sports Exerc. 2009, 41, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Chodzko-Zajko, W.J.; Proctor, D.N.; Fiatarone Singh, M.A.; Minson, C.T.; Nigg, C.R.; Salem, G.J.; Skinner, J.S. American College of Sports Medicine position stand. Exercise and physical activity for older adults. Med. Sci. Sports Exerc. 2009, 41, 1510–1530. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Bradley, J.S.; McCoski, S.R.; Gonzalez, J.M.; Ealy, A.D.; Johnson, S.E. Reduced skeletal muscle fiber size following caloric restriction is associated with calpain-mediated proteolysis and attenuation of IGF-1 signaling. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2017, 312, R806–R815. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.H.; Churchward-Venne, T.A.; Mitchell, C.J.; Kolar, N.M.; Kassis, A.; Karagounis, L.G.; Burke, L.M.; Hawley, J.A.; Phillips, S.M. Hypoenergetic diet-induced reductions in myofibrillar protein synthesis are restored with resistance training and balanced daily protein ingestion in older men. Am. J. Physiol. Endocrinol. Metable 2015, 308, E734–E743. [Google Scholar] [CrossRef] [PubMed]
- Longland, T.M.; Oikawa, S.Y.; Mitchell, C.J.; Devries, M.C.; Phillips, S.M. Higher compared with lower dietary protein during an energy deficit combined with intense exercise promotes greater lean mass gain and fat mass loss: A randomized trial. Am. J. Clin. Nutr. 2016, 103, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.E.; Suarez, J.A.; Brandhorst, S.; Balasubramanian, P.; Cheng, C.W.; Madia, F.; Fontana, L.; Mirisola, M.G.; Guevara-Aguirre, J.; Wan, J.; et al. Low protein intake is associated with a major reduction in IGF-1, cancer, and overall mortality in the 65 and younger but not older population. Cell Metab. 2014, 19, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Solon-Biet, S.M.; Mitchell, S.J.; Coogan, S.C.; Cogger, V.C.; Gokarn, R.; McMahon, A.C.; Raubenheimer, D.; de Cabo, R.; Simpson, S.J.; Le Couteur, D.G. Dietary Protein to Carbohydrate Ratio and Caloric Restriction: Comparing Metabolic Outcomes in Mice. Cell Rep. 2015, 11, 1529–1534. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.C.; Rabinovitch, P.S.; Kaeberlein, M. mTOR is a key modulator of ageing and age-related disease. Nature 2013, 493, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Barsalani, R.; Brochu, M.; Dionne, I.J. Is there a skeletal muscle mass threshold associated with the deterioration of insulin sensitivity in sedentary lean to obese postmenopausal women? Diabetes Res. Clin. Pract. 2013, 102, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Shiroma, E.J.; Cook, N.R.; Manson, J.E.; Moorthy, M.V.; Buring, J.E.; Rimm, E.B.; Lee, I.M. Strength Training and the Risk of Type 2 Diabetes and Cardiovascular Disease. Med. Sci. Sports Exerc. 2017, 49, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Sylow, L.; Kleinert, M.; Richter, E.A.; Jensen, T.E. Exercise-stimulated glucose uptake-regulation and implications for glycaemic control. Nat. Rev. Endocrinol. 2017, 13, 133–148. [Google Scholar] [CrossRef] [PubMed]
- Pesta, D.H.; Goncalves, R.L.S.; Madiraju, A.K.; Strasser, B.; Sparks, L.M. Resistance training to improve type 2 diabetes: Working toward a prescription for the future. Nutr. Metab. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Delmonico, M.J.; Harris, T.B.; Visser, M.; Park, S.W.; Conroy, M.B.; Velasquez-Mieyer, P.; Boudreau, R.; Manini, T.M.; Nevitt, M.; Newman, A.B.; et al. Longitudinal study of muscle strength, quality, and adipose tissue infiltration. Am. J. Clin. Nutr. 2009, 90, 1579–1585. [Google Scholar] [CrossRef] [PubMed]
- Dam, T.T.; Peters, K.W.; Fragala, M.; Cawthon, P.M.; Harris, T.B.; McLean, R.; Shardell, M.; Alley, D.E.; Kenny, A.; Ferrucci, L.; et al. An evidence-based comparison of operational criteria for the presence of sarcopenia. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69, 584–590. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.; Le, H.D.; Melkani, G.C.; Panda, S. Time-restricted feeding attenuates age-related cardiac decline in Drosophila. Science 2015, 347, 1265–1269. [Google Scholar] [CrossRef] [PubMed]
- Kastman, E.K.; Willette, A.A.; Coe, C.L.; Bendlin, B.B.; Kosmatka, K.J.; McLaren, D.G.; Xu, G.; Canu, E.; Field, A.S.; Alexander, A.L. A calorie-restricted diet decreases brain iron accumulation and preserves motor performance in old rhesus monkeys. J. Neurosci. 2010, 30, 7940–7947. [Google Scholar] [CrossRef] [PubMed]
- Colman, R.J.; Beasley, T.M.; Allison, D.B.; Weindruch, R. Attenuation of sarcopenia by dietary restriction in rhesus monkeys. J. Gerontol. A Biol. Sci. Med. Sci. 2008, 63, 556–559. [Google Scholar] [CrossRef] [PubMed]
- Bodnar, M.; Skalicky, M.; Viidik, A.; Erben, R.G. Interaction between exercise, dietary restriction and age-related bone loss in a rodent model of male senile osteoporosis. Gerontology 2012, 58, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Moreira, L.D.; Oliveira, M.L.; Lirani-Galvao, A.P.; Marin-Mio, R.V.; Santos, R.N.; Lazaretti-Castro, M. Physical exercise and osteoporosis: Effects of different types of exercises on bone and physical function of postmenopausal women. Arq. Bras. Endocrinol. Metabol. 2014, 58, 514–522. [Google Scholar] [CrossRef] [PubMed]

| First Author, Year | Weight Stabilization | Age (Years Mean ± SD) | Sex | Health Status | CR or BM Reduction | Diet (CARBOHYDRATE/Protein/fat%) | RT Load | RT Volume | CRRT Duration (Weeks) |
|---|---|---|---|---|---|---|---|---|---|
| Amamou, 2016 [6] | 4 weeks stabilization | 65.8 ± 3.1 | both | dyslipidemic and diabetics | 472.74 ± 52.5/day | 25–30 g protein supplementation (45–50/25–30/25–30) | 65 to 80% 1RM | 2 × (8 to 15) | 16 |
| Bouchard, 2009 [18] | no | 63 ± 4 | women | health | 0.5 to 1 kg/week | balanced (55/15/30) | 80% 1RM | 3 × 8 | 12 |
| Brochu, 2009 [15] | 2 kg stabilization | 57.2 ± 5 | women | health | 624 ± 133/day (33.4 ± 4.9%) | balanced (55/15/30) | 65 to 75% 1RM | (2 to 3) × (15 to 10) | 24 |
| Dunstan, 2005 [21] | not reported | 67.6 ± 5.2 | both | diabetics | 0.25 kg/week | balanced (70% carbohydrate and protein/30%fat) | 75 to 85% 1RM | 3 × (8 to 10) | 24 |
| Sénéchal, 2012 [14] | no | 62.6 ± 4.1 | women | health | 0.5 to 1 kg/week | balanced (55/15/30) | not reported | 3 × 8 | 12 |
| St-Onge, 2012 [19] | 4 weeks stabilization | 57.6 ± 4 | women | health | 500 to 800 kcal/day | balanced (55/15/30) | 8 to 15RM | (1 to 3) × (8 to 12) | 24 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sardeli, A.V.; Komatsu, T.R.; Mori, M.A.; Gáspari, A.F.; Chacon-Mikahil, M.P.T. Resistance Training Prevents Muscle Loss Induced by Caloric Restriction in Obese Elderly Individuals: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 423. https://doi.org/10.3390/nu10040423
Sardeli AV, Komatsu TR, Mori MA, Gáspari AF, Chacon-Mikahil MPT. Resistance Training Prevents Muscle Loss Induced by Caloric Restriction in Obese Elderly Individuals: A Systematic Review and Meta-Analysis. Nutrients. 2018; 10(4):423. https://doi.org/10.3390/nu10040423
Chicago/Turabian StyleSardeli, Amanda V., Tiemy R. Komatsu, Marcelo A. Mori, Arthur F. Gáspari, and Mara Patrícia T. Chacon-Mikahil. 2018. "Resistance Training Prevents Muscle Loss Induced by Caloric Restriction in Obese Elderly Individuals: A Systematic Review and Meta-Analysis" Nutrients 10, no. 4: 423. https://doi.org/10.3390/nu10040423
APA StyleSardeli, A. V., Komatsu, T. R., Mori, M. A., Gáspari, A. F., & Chacon-Mikahil, M. P. T. (2018). Resistance Training Prevents Muscle Loss Induced by Caloric Restriction in Obese Elderly Individuals: A Systematic Review and Meta-Analysis. Nutrients, 10(4), 423. https://doi.org/10.3390/nu10040423






