Evaluation of A Concentrated Preterm Formula as a Liquid Human Milk Fortifier in Preterm Babies at Increased Risk of Feed Intolerance
Abstract
1. Introduction
2. Materials and Methods
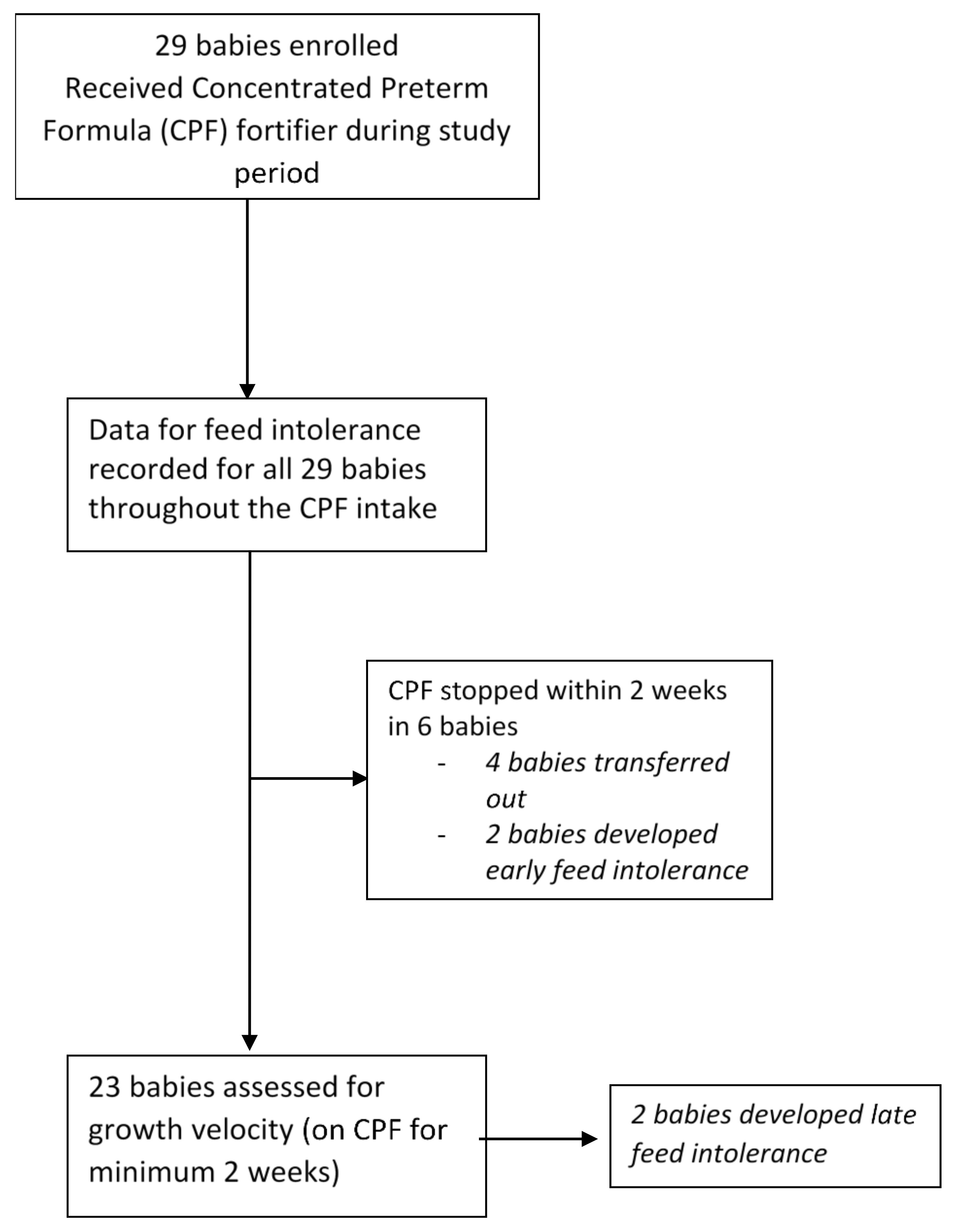
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Agostoni, C.; Buonocore, G.; Carnielli, V.P.; De Curtis, M.; Darmaun, D.; Decsi, T.; Domellöf, M.; Embleton, N.D.; Fusch, C.; et al. Enteral nutrient supply for preterm infants: Commentary from the European Society of Paediatric Gastroenterology, Hepatology and Nutrition Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2010, 50, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Eidelman, A.I. Breastfeeding and the use of human milk: An analysis of the American Academy of Pediatrics 2012 Breastfeeding Policy Statement. Breastfeed Med. 2012, 7, 323–324. [Google Scholar] [CrossRef] [PubMed]
- ESPGHAN Committee on Nutrition; Agostoni, C.; Braegger, C.; Decsi, T.; Kolacek, S.; Koletzko, B.; Michaelsen, K.F.; Mihatsch, W.; Moreno, L.A.; Puntis, J.; et al. Breast-feeding: A commentary by the ESPGHAN Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2009, 49, 112–125. [Google Scholar] [CrossRef] [PubMed]
- Lucas, A.; Cole, T.J. Breast milk and neonatal necrotising enterocolitis. Lancet 1990, 336, 1519–1523. [Google Scholar] [CrossRef]
- Lucas, A.; Morley, R.; Cole, T.J.; Lister, G.; Leeson-Payne, C. Breast milk and subsequent intelligence quotient in children born preterm. Lancet 1992, 339, 261–264. [Google Scholar] [CrossRef]
- McGuire, W.; Anthony, M.Y. Donor human milk versus formula for preventing necrotising enterocolitis in preterm infants: Systematic review. Arch. Dis. Child. Fetal Neonatal Ed. 2003, 88, 11–14. [Google Scholar] [CrossRef]
- Sisk, P.M.; Lovelady, C.A.; Dillard, R.G.; Gruber, K.J.; O’Shea, T.M. Early human milk feeding is associated with a lower risk of necrotizing enterocolitis in very low birth weight infants. J Perinatol. 2007, 27, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.; Gerss, J. Longitudinal analysis of macronutrients and minerals in human milk produced by mothers of preterm infants. Clin. Nutr. 2011, 30, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Arslanoglu, S.; Moro, G.E.; Ziegler, E.E.; The Wapm Working Group on Nutrition, null. Optimization of human milk fortification for preterm infants: New concepts and recommendations. J. Perinat. Med. 2010, 38, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Hawthorne, K.M.; Abrams, S.A. Safety and efficacy of human milk fortification for very-low-birthweight infants. Nutr. Rev. 2004, 62, 482–485. [Google Scholar] [CrossRef] [PubMed]
- Kuschel, C.A.; Harding, J.E. Multicomponent fortified human milk for promoting growth in preterm infants. Cochrane. Database Syst. Rev. 2004. [Google Scholar] [CrossRef]
- Maggio, L.; Costa, S.; Gallini, F. Human milk fortifiers in very low birth weight infants. Early Hum. Dev. 2009, 85, S59–S61. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.V.E.; Embleton, N.D.; Harding, J.E.; McGuire, W. Multi-nutrient fortification of human milk for preterm infants. Cochrane. Database Syst. Rev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Schanler, R.J. Suitability of human milk for the low-birthweight infant. Clin. Perinatol. 1995, 22, 207–222. [Google Scholar] [CrossRef]
- Schanler, R.J.; Abrams, S.A. Postnatal attainment of intrauterine macromineral accretion rates in low birth weight infants fed fortified human milk. J. Pediatr. 1995, 126, 441–447. [Google Scholar] [CrossRef]
- Arslanoglu, S.; Bertino, E.; Coscia, A.; Tonetto, P.; Giuliani, F.; Moro, G.E. Update of adjustable fortification regimen for preterm infants: A new protocol. J. Biol. Regul. Homeost. Agents 2012, 26, 65–67. [Google Scholar] [PubMed]
- Arslanoglu, S.; Moro, G.E.; Ziegler, E.E. Adjustable fortification of human milk fed to preterm infants: Does it make a difference? J. Perinatol. 2006, 26, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, S.; Schanler, R.J.; Kim, J.H.; Patel, A.L.; Trawöger, R.; Kiechl-Kohlendorfer, U.; Chan, G.M.; Blanco, C.L.; Abrams, S.; Cotten, C.M.; et al. An exclusively human milk-based diet is associated with a lower rate of necrotizing enterocolitis than a diet of human milk and bovine milk-based products. J. Pediatr. 2010, 156, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Pearson, F.; Johnson, M.J.; Leaf, A.A. Milk osmolality: Does it matter? Arch. Dis. Child. Fetal Neonatal Ed. 2013, 98, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Goldblum, O.M.; Holzman, I.R.; Fisher, S.E. Intragastric feeding in the neonatal dog. Its effect on intestinal osmolality. Am. J. Dis. Child. 1981, 135, 631–633. [Google Scholar] [CrossRef] [PubMed]
- Schmid, H.R.; Ehrlein, H.J. Effects of enteral infusion of hypertonic saline and nutrients on canine jejunal motor patterns. Dig. Dis. Sci. 1993, 38, 1062–1072. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, L.; Bokiniec, R.; King, C.; Weaver, G.; Edwards, A.D. Increased osmolality of breast milk with therapeutic additives. Arch. Dis. Child. Fetal Neonatal Ed. 2004, 89, F514–F517. [Google Scholar] [CrossRef] [PubMed]
- Palcich, G.; de Moraes Gillio, C.; Aragon-Alegro, L.C.; Pagotto, F.J.; Farber, J.M.; Landgraf, M.; Destro, M.T. Enterobacter sakazakii in dried infant formulas and milk kitchens of maternity wards in São Paulo, Brazil. J. Food Prot. 2009, 72, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Stanger, J.; Zwicker, K.; Albersheim, S.; Murphy, J.J. Human milk fortifier: An occult cause of bowel obstruction in extremely premature neonates. J. Pediatr. Surg. 2014, 49, 724–726. [Google Scholar] [CrossRef] [PubMed]
- Flikweert, E.R.; La Hei, E.R.; De Rijke, Y.B.; Van de Ven, K. Return of the milk curd syndrome. Pediatr. Surg. Int. 2003, 19, 628–631. [Google Scholar] [CrossRef] [PubMed]
- Murase, M.; Miyazawa, T.; Taki, M.; Sakurai, M.; Miura, F.; Mizuno, K.; Itabashi, K.; Toki, A. Development of fatty acid calcium stone ileus after initiation of human milk fortifier. Pediatr. Int. 2013, 55, 114–116. [Google Scholar] [CrossRef] [PubMed]
- Willeitner, A.; Anderson, M.; Lewis, J. Highly Concentrated Preterm Formula as an Alternative to Powdered Human Milk Fortifier: A Randomized Controlled Trial. J. Pediatr. Gastroenterol. Nutr. 2017, 65, 574–578. [Google Scholar] [CrossRef] [PubMed]
- Tsang, R.C.; Uauy, R.; Koletzko, B.; Zlotkin, S. Nutrition of the Preterm Infant: Scientific Basis and Practical Guidelines; Digital Educational Publishing, Inc.: Cincinnati, OH, USA, 2005. [Google Scholar]
- Boyce, C.; Watson, M.; Lazidis, G.; Reeve, S.; Dods, K.; Simmer, K.; McLeod, G. Preterm human milk composition: A systematic literature review. Br. J. Nutr. 2016, 116, 1033–1045. [Google Scholar] [CrossRef] [PubMed]
- Gidrewicz, D.A.; Fenton, T.R. A systematic review and meta-analysis of the nutrient content of preterm and term breast milk. BMC Pediatr. 2014, 14, 216. [Google Scholar] [CrossRef] [PubMed]
- Fenton, T.R. A new growth chart for preterm babies: Babson and Benda’s chart updated with recent data and a new format. BMC Pediatr 2003, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Fenton, T.R.; Kim, J.H. A systematic review and meta-analysis to revise the Fenton growth chart for preterm infants. BMC Pediatr. 2013, 13, 59. [Google Scholar] [CrossRef] [PubMed]
- Fenton, T.R.; Sauve, R.S. Using the LMS method to calculate z-scores for the Fenton preterm infant growth chart. Eur. J. Clin. Nutr. 2007, 61, 1380–1385. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.L.; Engstrom, J.L.; Meier, P.P.; Kimura, R.E. Accuracy of methods for calculating postnatal growth velocity for extremely low birth weight infants. Pediatrics 2005, 116, 1466–1473. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.L.; Engstrom, J.L.; Meier, P.P.; Jegier, B.J.; Kimura, R.E. Calculating postnatal growth velocity in very low birth weight (VLBW) premature infants. J. Perinatol. 2009, 29, 618–622. [Google Scholar] [CrossRef] [PubMed]
- Bell, M.J.; Ternberg, J.L.; Feigin, R.D.; Keating, J.P.; Marshall, R.; Barton, L.; Brotherton, T. Neonatal necrotizing enterocolitis. Therapeutic decisions based upon clinical staging. Ann. Surg. 1978, 187, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ehrenkranz, R.A.; Walsh, M.C.; Vohr, B.R.; Jobe, A.H.; Wright, L.L.; Fanaroff, A.A.; Wrage, L.A.; Poole, K. National Institutes of Child Health and Human Development Neonatal Research Network Validation of the National Institutes of Health consensus definition of bronchopulmonary dysplasia. Pediatrics 2005, 116, 1353–1360. [Google Scholar] [CrossRef] [PubMed]
- International Committee for the Classification of Retinopathy of Prematurity The International Classification of Retinopathy of Prematurity revisited. Arch. Ophthalmol. 2005, 123, 991–999. [CrossRef] [PubMed]
- Abdallah, E.A.A.; Said, R.N.; Mosallam, D.S.; Moawad, E.M.I.; Kamal, N.M.; Fathallah, M.G.E.-D. Serial serum alkaline phosphatase as an early biomarker for osteopenia of prematurity. Medicine (Baltimore) 2016, 95, e4837. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Chan, G.; Schanler, R.; Groh-Wargo, S.; Bloom, B.; Dimmit, R.; Williams, L.; Baggs, G.; Barrett-Reis, B. Growth and tolerance of preterm infants fed a new extensively hydrolyzed liquid human milk fortifier. J. Pediatr. Gastroenterol. Nutr. 2015, 61, 665–671. [Google Scholar] [CrossRef] [PubMed]
- Fanaro, S. Feeding intolerance in the preterm infant. Early Hum. Dev. 2013, 89 (Suppl. 2), S13–S20. [Google Scholar] [CrossRef]
- Thoene, M.; Lyden, E.; Weishaar, K.; Elliott, E.; Wu, R.; White, K.; Timm, H.; Anderson-Berry, A. Comparison of a powdered, acidified liquid, and non-acidified liquid human milk fortifier on clinical outcomes in premature infants. Nutrients 2016, 8, 451. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, K.; Narnag, A.; Mahajan, R. Effect of human milk fortification in appropriate for gestation and small for gestation preterm babies: A randomized controlled trial. Indian Pediatr. 2007, 44, 286–290. [Google Scholar] [PubMed]
- Villar, J.; Giuliani, F.; Barros, F.; Roggero, P.; Coronado Zarco, I.A.; Rego, M.A.S.; Ochieng, R.; Gianni, M.L.; Rao, S.; Lambert, A.; et al. Monitoring the postnatal growth of preterm infants: A paradigm change. Pediatrics 2018, 141. [Google Scholar] [CrossRef] [PubMed]
- Moya, F.; Sisk, P.M.; Walsh, K.R.; Berseth, C.L. A new liquid human milk fortifier and linear growth in preterm infants. Pediatrics 2012, 130, e928–e935. [Google Scholar] [CrossRef] [PubMed]
- Hintz, S.R.; Kendrick, D.E.; Stoll, B.J.; Vohr, B.R.; Fanaroff, A.A.; Donovan, E.F.; Poole, W.K.; Blakely, M.L.; Wright, L.; Higgins, R.; et al. Neurodevelopmental and growth outcomes of extremely low birth weight infants after necrotizing enterocolitis. Pediatrics 2005, 115, 696–703. [Google Scholar] [CrossRef] [PubMed]
- Nicholl, R.M.; Gamsu, H.R. Changes in growth and metabolism in very low birthweight infants fed with fortified breast milk. Acta. Paediatr. 1999, 88, 1056–1061. [Google Scholar] [CrossRef] [PubMed]
- Ehrenkranz, R.A.; Dusick, A.M.; Vohr, B.R.; Wright, L.L.; Wrage, L.A.; Poole, W.K. Growth in the neonatal intensive care unit influences neurodevelopmental and growth outcomes of extremely low birth weight infants. Pediatrics 2006, 117, 1253–1261. [Google Scholar] [CrossRef] [PubMed]
- Erickson, T.; Gill, G.; Chan, G.M. The effects of acidification on human milk’s cellular and nutritional content. J. Perinatol. 2013, 33, 371–373. [Google Scholar] [CrossRef] [PubMed]
- Thoene, M.; Hanson, C.; Lyden, E.; Dugick, L.; Ruybal, L.; Anderson-Berry, A. Comparison of the effect of two human milk fortifiers on clinical outcomes in premature infants. Nutrients 2014, 6, 261–275. [Google Scholar] [CrossRef] [PubMed]
- Manz, F.; Kalhoff, H.; Remer, T. Renal acid excretion in early infancy. Pediatr. Nephrol. 1997, 11, 231–243. [Google Scholar] [CrossRef] [PubMed]
| 24 kcal/oz Fortified Preterm Human Milk * | ||
|---|---|---|
| Per 100 mL | Powder HMF | CPF |
| Dilution/Mixing | 4 packets to 100 mL HM | 40 mL CPF with 60 mL HM |
| Calories | 81 | 81 |
| Protein | 2.6 | 2.1 |
| Iron | 1.48 | 0.74 |
| Ca | 119 | 90.6 |
| P | 59.3 | 46 |
| Vitamin D | 154 | 63 |
| Osmolality | 325 | 304 |
| n = 29 | Value |
|---|---|
| Gestation age, week + day; median (IQR) | 26 + 3 (24 + 6–28 + 2) |
| Birth weight, g; median (IQR) | 833 (635–1050) |
| Male sex; n (%) | 20 (68.9%) |
| C section; n (%) | 20 (68.9%) |
| Enteral feeds start day; median (IQR) | 2 (2–5) |
| Day of life when full feeds achieved; median (IQR) | 30 (16–53) |
| TPN days; median (IQR) | 29 (17–61) |
| CPF start day; median (IQR) | 47 (31–60) |
| Number of days CPF received; median (IQR) | 28 (13–39) |
| Retinopathy of Prematurity (ROP) needing treatment *; n (%) | 6 (20.6%) |
| Chronic Lung Disease (CLD); n (%) | 18 (62.1%) |
| Late onset sepsis (blood) $ | 10 (34.5%) |
| Metabolic bone disease | 13 (44.8%) |
| Discharge/Transfer gestation week; median (IQR) | 39 (36–44) |
| Discharge/Transfer weight g; median (IQR) | 2795 (2300–3907) |
| Baby | GA * | Birth Weight (g) | Day of Start of CPF | Days on CPF When Intolerance Noted | Abdominal Distension | Emesis | Change in Stool/Stoma Output | Clinical/Culture Positive Sepsis | If Restarted on CPF |
|---|---|---|---|---|---|---|---|---|---|
| A | 27 + 1 | 1000 | 58 | 2 | No | Yes | No | Clinical | No |
| B | 24 + 0 | 840 | 47 | 10 | No | No | Yes | No | No |
| C | 28 + 0 | 574 | 40 | 21 | Yes | No | No | Clinical | Yes |
| D | 29 + 6 | 1135 | 29 | 28 | Yes | No | No | Culture | No |
| Characteristic | Value |
|---|---|
| Total observation days on CPF median (IQR) | 34 (24–49) |
| Days on CPF 24 kcal/oz median (IQR) | 27 (12.5–35.5) |
| Growth velocity prior to CPF (g/kg/day) median (IQR) | 12.53 (11.0–15.4) |
| Growth velocity on CPF (g/kg/day) median (IQR) | 15.87 (11.7–19.0) |
| Weight at start of CPF (grams) median (IQR) | 1500 (1254–1746) |
| Weight at end of CPF (grams) median (IQR) | 2128 (2500–2778) |
| Weight gain on CPF (g/day) median (IQR) | 31.4 (22.9–36.2) |
| Head growth prior to CPF (n = 13) cm/week median (IQR) | 0.75 (0.53–0.76) |
| Head growth on CPF (n = 13) cm/week median (IQR) | 0.79 (0.69–0.86) |
| Length growth prior to CPF (n = 12) cm/week median (IQR) | 0.88 (0.84–0.93) |
| Length growth on CPF (n = 12) cm/week median (IQR) | 0.77 (0.67–1.08) |
| Maximum BUN (n = 27) mmol/L median (IQR) | 2.4 (1.25–4) |
| Maximum pre-albumin (n = 27) mg/L median (IQR) | 103 (73.5–119.5) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pillai, A.; Albersheim, S.; Matheson, J.; Lalari, V.; Wei, S.; Innis, S.M.; Elango, R. Evaluation of A Concentrated Preterm Formula as a Liquid Human Milk Fortifier in Preterm Babies at Increased Risk of Feed Intolerance. Nutrients 2018, 10, 1433. https://doi.org/10.3390/nu10101433
Pillai A, Albersheim S, Matheson J, Lalari V, Wei S, Innis SM, Elango R. Evaluation of A Concentrated Preterm Formula as a Liquid Human Milk Fortifier in Preterm Babies at Increased Risk of Feed Intolerance. Nutrients. 2018; 10(10):1433. https://doi.org/10.3390/nu10101433
Chicago/Turabian StylePillai, Anish, Susan Albersheim, Julie Matheson, Vikki Lalari, Sylvia Wei, Sheila M Innis, and Rajavel Elango. 2018. "Evaluation of A Concentrated Preterm Formula as a Liquid Human Milk Fortifier in Preterm Babies at Increased Risk of Feed Intolerance" Nutrients 10, no. 10: 1433. https://doi.org/10.3390/nu10101433
APA StylePillai, A., Albersheim, S., Matheson, J., Lalari, V., Wei, S., Innis, S. M., & Elango, R. (2018). Evaluation of A Concentrated Preterm Formula as a Liquid Human Milk Fortifier in Preterm Babies at Increased Risk of Feed Intolerance. Nutrients, 10(10), 1433. https://doi.org/10.3390/nu10101433






