Characterization of Mutant Aspergillus niger and the Impact on Certain Plants
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Molecular Identification of Aspergillus niger
2.3. Preparation of Mutagen Solutions of Sodium Azide
2.4. Impact of Sodium azide
2.4.1. Direct Evaluation
Fungal Isolates Preparation and Inoculation
Morphological and Microscopic Examination of Mutant Fungal Spores
Physiological Parameters of Mutant Fungi
- (1)
- pH of Broth Media
- (2)
- Total Protein of A. niger
- (3)
- Total Flavonoids of A. niger
- (4)
- DNA Isolation and Molecular Marker (RAPD-PCR)
2.4.2. Indirect Evaluation
Plant Germination and Infection
Morphological Measurements
Physiological Measurements of plants infected with mutant fungi
- (1)
- Total proteins
- (2)
- Total flavonoids
2.5. Statistical Analysis
3. Results
3.1. Molecular Identification of Aspergillus Niger
3.2. Direct Evaluation for Mutant Fungi
3.2.1. Morphology of Fungi and Their Spores
3.2.2. Physiological Parameters of Mutant Fungi
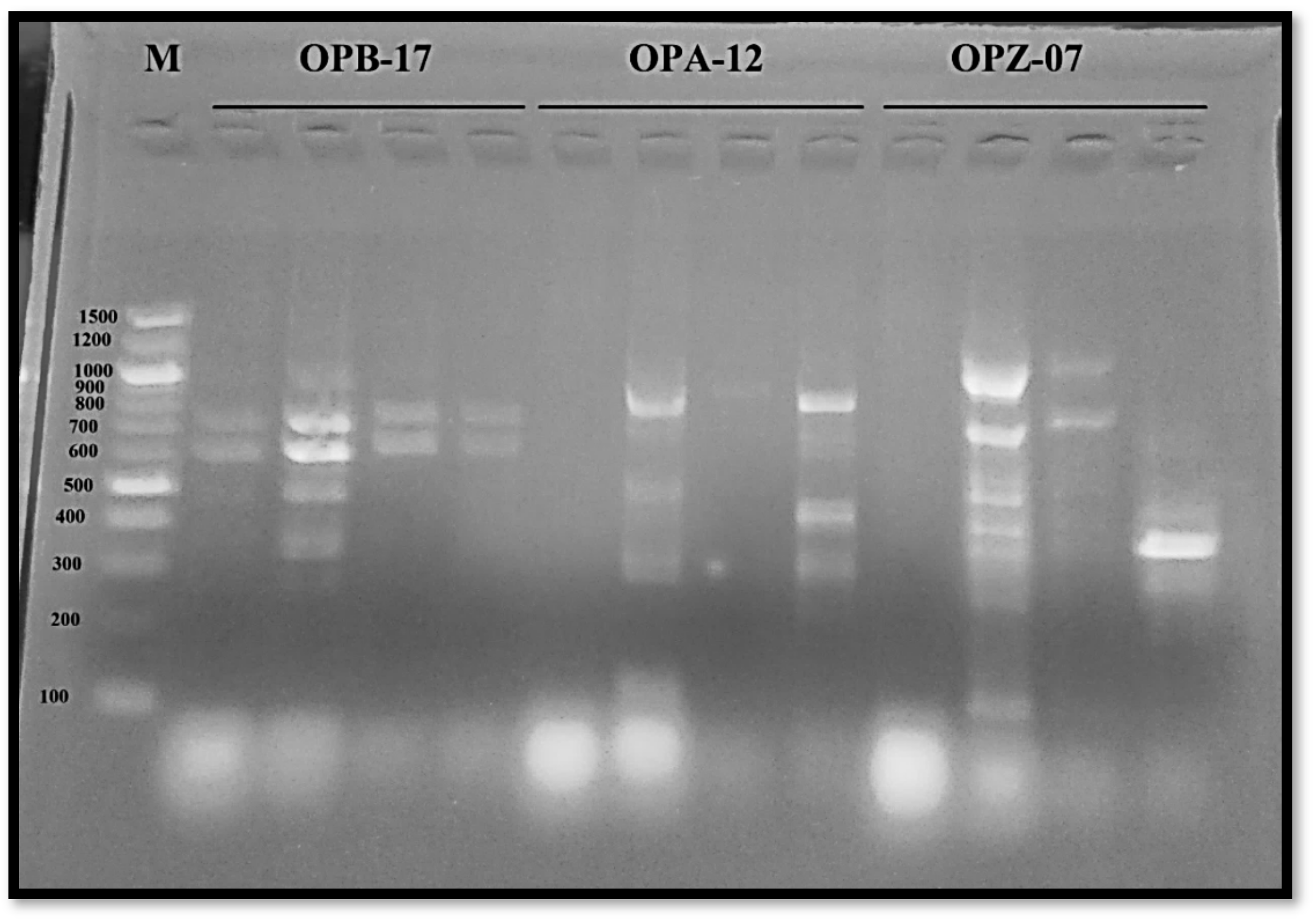
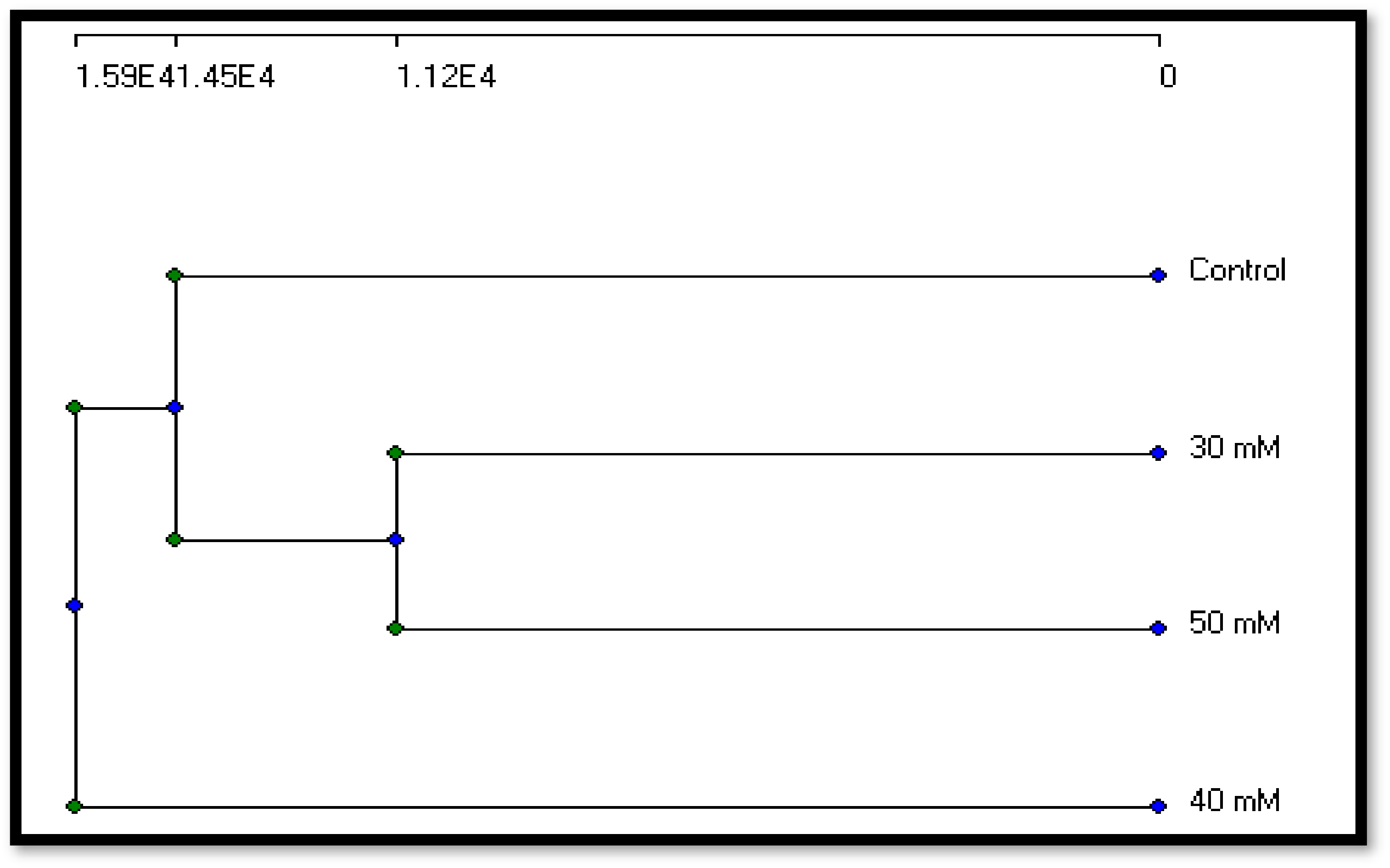
3.2.3. Molecular Variation
3.3. Indirect Evaluation of Plants Infected with Mutant Fungi
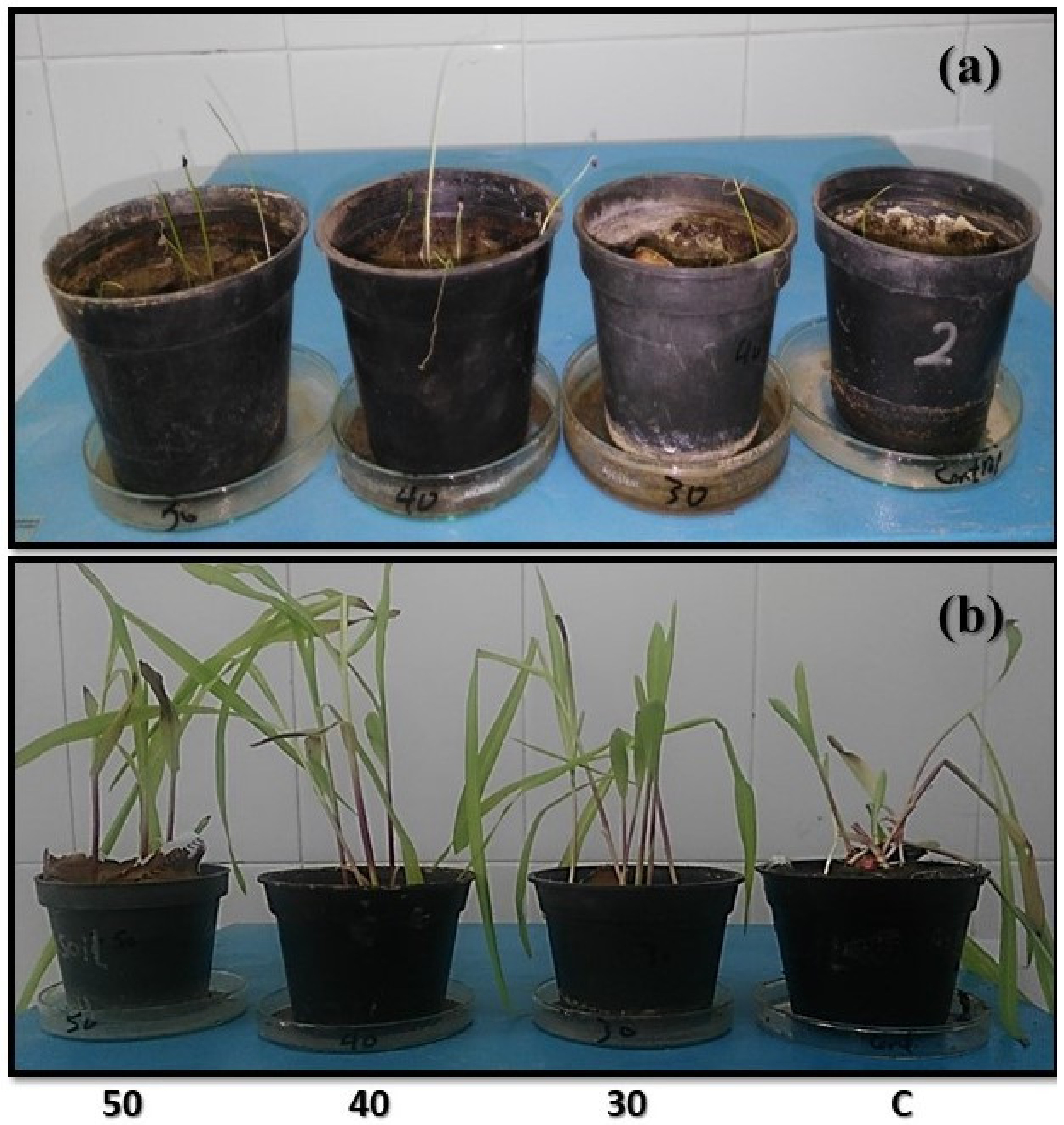
3.3.1. Morphological Parameters and Seed Germination of Infected Plants
3.3.2. Physiological Parameters of Infected Plants
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schuster, E.; Dunn-Coleman, N.; Frisvad, J.C.; van Dijck, P.W.M. On the safety of Aspergillus niger—A review. Appl. Microbiol. Biotechnol. 2002, 59, 426–435. [Google Scholar] [PubMed]
- Blumenthal, C.Z. Production of toxic metabolites in Aspergillus niger, Aspergillus oryzae, and Trichoderma reesei: Justification of mycotoxin testing in food grade enzyme preparations derived from the three fungi. Regul. Toxicol. Pharmacol. 2004, 39, 214–228. [Google Scholar] [CrossRef] [PubMed]
- Shankar, J. An Overview of Toxins in Aspergillus Associated with Pathogenesis. Int. J. Life Sci. Bt. Pharm. Res. 2013, 2, 16–31. [Google Scholar]
- Cortesão, M.; de Haas, A.; Unterbusch, R.; Fujimori, A.; Schütze, T.; Meyer, V.; Moeller, R. Aspergillus niger Spores Are Highly Resistant to Space Radiation. Front. Microbiol. 2020, 11, 560. [Google Scholar] [CrossRef]
- Madika, A.; Ulem, E.A.; Musa, B.; Sulaiman, M.A.; Hussaini, I.M. Screening of Aspergillus Niger Isolated from Soil for Pectinase Production. FUDMA J. Sci. 2020, 4, 244–249. [Google Scholar] [CrossRef]
- El-Nagerabi, S.A.F.; Ahmed, A.H.M.; Elshafie, A.E. In Vitro Evaluation of Selected Plant Extracts as Biocontrol Agents against Black Mold (Aspergillus niger Van Tieghem) of Onion Bulbs (Allium Cepa L.). Int. J. Sci. Technol. Res. 2016, 5, 147–152. [Google Scholar]
- Zohri, A.; Boul-Nasr, M.B.; Adam, M.; Mustafa, M.A.; Amer, E.M. Impact of Enzymes and Toxins Potentiality of Four Aspergillus Species to Cause Aspergillosis. Biol. Med. 2017, 9, 1–7. [Google Scholar]
- El-Mokadem, H.E.; Mostafa, G.G. Induction of mutations in Browallia speciosa using sodium azide and identification of the genetic variation by peroxidase isozyme. Afr. J. Biotechnol. 2014, 13, 106–111. [Google Scholar] [CrossRef]
- Grant, W.F.; Salamone, M.F. Comparative mutagenicity of chemicals selected for test in the International Program on Chemical Safety’s collaborative study on plant systems for the detection of environmental mutagens. Mutagenesis Res. 1994, 310, 187–209. [Google Scholar] [CrossRef]
- Hussain, M.; Gul, M.; Kamal, R.; Iqbal, M.A.; Zulfiqar, S.; Abbas, A.; Röder, M.S.; Muqaddasi, Q.H.; Mehboob-ur-Rahman. Prospects of Developing Novel Genetic Resources by Chemical and Physical Mutagenesis to Enlarge the Genetic Window in Bread Wheat Varieties. Agriculture 2021, 11, 621. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: London, UK, 1990; Volume 18, pp. 315–322. [Google Scholar]
- Suprasanna, P.; Mirajkar, S.J.; Bhagwat, S.G. Induced Mutations and Crop Improvement. Plant. Biol. Biotechnol. 2015, 1, 593–617. [Google Scholar]
- Bradford, M.M. A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Tawfik, E.; Fathy, M. Chemical Mutagens Affecting in vitro Behavior of Gardenia jasminoides. Plant Tissue Cult. Biotechnol. 2020, 30, 209–218. [Google Scholar] [CrossRef]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Doyle, J.J.; Doyle, J.L. Isolation of Plant DNA from Fresh Tissue. Focus 1990, 12, 13–15. [Google Scholar]
- Shuaib, M.; Zeb, A.; Ali, Z.; Ali, W.; Ahmad, T.; Khan, I. Characterization of Wheat Varieties by Seed Storage Protein Electrophoresis. Afr. J. Biotechnol. 2007, 6, 497–500. [Google Scholar]
- Romsdahl, J.; Blachowicz, A.; Chiang, A.J.; Singh, N.; Stajich, J.E.; Kalkum, M.; Venkateswaran, K.; Wanga, C.C.C. Characterization of Aspergillus niger Isolated from the International Space Station. mSystems 2018, 3, e00112-18. [Google Scholar] [CrossRef]
- Soares, C.; Calado, T.; Venâncio, A. Mycotoxin production by Aspergillus niger aggregate strains isolated from harvested maize in three Portuguese regions. Rev. Iberoam. Micol. 2012, 30, 9–13. [Google Scholar] [CrossRef]
- Freire, L.; Guerreiro, T.M.; Pia, A.K.R.; Lima, E.O.; Oliveira, D.N.; Melo, C.F.O.R.; Catharino, R.R.; Sant’ana, A.S. A quantitative study on growth variability and production of ochratoxin A and its derivatives by A. carbonarius and A. niger in grapebased medium. Sci. Rep. 2018, 8, 14573. [Google Scholar] [CrossRef]
- Dijksterhuis, J.; Samson, R.A. Food and crop spoilage on storage. In Mycota X1 Agricultural Applications; Kemken, F., Ed.; Springer: Berlin/Heidelberg, Germany, 2002; pp. 39–52. [Google Scholar]
- Kumar, A.K.; Parikh, B.S. Cellulose-degrading enzymes from Aspergillus terreus D34 and enzymatic saccharification of mild-alkali and dilute-acid pretreated lignocellulosic biomass residues. Bioresour 2015, 2, 7. [Google Scholar] [CrossRef]
- Ramachandran, S. Mise en oeuvre des spores d’Aspergillus niger obtenues par fermentation en milieu solide pour la production d’acide gluconique (Use of spores of Aspergillus niger obtained by Solid-state fermentation for the production of gluconic acid). In Génie Des Proceeds; Université Blaise Pascal-Clermont-Ferrand II: Clermont-Ferrand, France, 2008. [Google Scholar]
- Vernwal, S.K.; Yadav, R.S.; Yadav, K.D. Purification of a peroxidase from Solanum melongena fruit juice. Indian J. Biochem. Biophys. 2006, 43, 239–243. [Google Scholar] [PubMed]
- Lu, L.; Zhao, M.; Zhang, B.B.; Yu, S.Y.; Bian, X.J.; Wang, W.; Wang, Y. Purification and characterization of laccase from Pycnoporus sanguineus and decolorization of an anthraquinone dye by the enzyme. Appl. Microbiol. Biotechnol. 2007, 74, 1232–1239. [Google Scholar] [CrossRef] [PubMed]
- Gromada, A.; Fiedurek, J. Selective isolation of Aspergillus niger mutants with enhanced glucose oxidase production. J. Appl. Microbiol. 1997, 82, 648–652. [Google Scholar] [CrossRef]
- Lucca, A.L.; Jorge, J.A.; Guimaraes, L.H.S. Extracellular β-D-fructofuranosidase from Aspergillus parasiticus: Optimization of the production under submerged fermentation and biochemical characterization. Afr. J. Biotechnol. 2013, 12, 5678–5687. [Google Scholar]
- Salaman, Z.A.; Abdulmalek, H.W.; Al-Judy, N.J.; Dheeb, B.I.; Al Abbasi, S.H.A. Detection the ability of Aspergillus parasiticus local Iraqi isolates for aflatoxin B1 production. Curr. Res. Microbiol. Biotechnol. 2015, 3, 564–568. [Google Scholar]
- Youssef, M.S.; El-Maghraby, O.M.O.; Ibrahim, Y.M. Mycobiota and mycotoxins of Egyptian peanut (Arachis hypogeae L.) seeds. Int. J. Bot. 2008, 4, 349–360. [Google Scholar] [CrossRef]
- Costa, R.; Pereira, G.; Garrido, I.; Tavares-de-Sousa, M.M.; Espinosa, F. Comparison of RAPD, ISSR, and AFLP molecular markers to reveal and classify Orchardgrass (Dactylis glomerata L.) germplasm variations. PLoS ONE 2016, 11, e0152972. [Google Scholar] [CrossRef] [PubMed]
- Longya, A.; Chaipanya, C.; Franceschetti, M.; Maidment, H.R.J.; Banfield, J.M.; Jantasuriyarat, C. Gene duplication and mutation in the emergence of a novel aggressive allele of the AVR-Pik effector in the rice blast fungus. Mol. Plant Microbe Interact. 2019, 32, 740–749. [Google Scholar] [CrossRef]
- Longya, A.; Talumphai, S.; Jantasuriyarat, C. Morphological Characterization and Genetic Diversity of Rice Blast Fungus, Pyricularia oryzae, from Thailand Using ISSR and SRAP Markers. J. Fungi 2020, 6, 38. [Google Scholar] [CrossRef]
- Flaherty, J.E.; Payne, G.A. Overexpression of aflR leads to upregulation of pathway gene transcription and increased aflatoxin production in Aspergillus flavus. Appl. Environ. Microbiol. 1997, 63, 3995–4000. [Google Scholar] [CrossRef]
- McDonald, B.A.; Linde, C. Pathogen population genetics, evolutionary potential, and durable resistance. Annu. Rev. Phytopathol. 2002, 40, 349–379. [Google Scholar] [CrossRef] [PubMed]
- Çelik, O.; Oguz, A.; Karakaya, A. Genetic Diversity of Barley Foliar Fungal Pathogens. Agronomy 2021, 11, 434. [Google Scholar] [CrossRef]
- Sowley, E.; Kankam, F.; Tawiah, E. Comparative Study on the Incidence of Aspergillus flavus in Farmer’s Field and Stored Maize (Zea mays) Seed in Northern Region of Ghana. Asian J. Plant Sci. 2018, 1, 1–7. [Google Scholar] [CrossRef]
- Subramanian, S.; Kiarie, S.; Kiarie, S.; Nyasani, J.; Nyasani, J.; Gahole, L. Impact of Fungal Endophyte Colonization of Maize (Zea mays L.) on Induced Resistance to Thrips- and Aphid-Transmitted Viruses. Plants 2020, 9, 416. [Google Scholar] [CrossRef]

| Control | 30 µM | 40 µM | 50 µM | |
|---|---|---|---|---|
| Growth diameter, cm | 7.76 ± 0.25 | 1.73 ± 0.15 | 1.10 ± 0.10 | 0.78 ± 0.27 |
| Spores count, spore/mL | 93.66 ± 5.68 | 47.00 ± 11.00 | 30.33 ± 3.05 | 22.66 ± 4.04 |
| Control | 30 µM | 40 µM | 50 µM | p-Value | |
|---|---|---|---|---|---|
| pH of media | 5.21 ± 0.34 | 5.65 ± 0.02 | 5.48 ± 0.26 | 5.74 ± 0.01 | 0.002 ** |
| Total protein “mg/g” | 50.52 ± 1.95 | 64.42 ± 0.14 | 64.90 ± 0.08 | 32.57 ± 0.37 | 0.028 * |
| Total flavonoids “mg/g” (fungi) | 20.61 ± 0.47 | 10.45 ± 0.43 | 15.49 ± 0.47 | 12.38 ± 0.58 | 0.008 ** |
| Total flavonoids “mg/g” (media) | 61.04 ± 0.08 | 42.70 ± 0.93 | 47.40 ± 0.98 | 43.48 ± 0.74 | 0.009 ** |
| Primer Code | Primer Sequence | GC% | Tm | Polymorphism % |
|---|---|---|---|---|
| OPB-17 | 5′-AGGGAACGAG-3′ | 60 | 33.1 | 66.66 |
| OPA-12 | 5′-TCGGCGATAG-3′ | 60 | 34 | 85.7 |
| OPZ-07 | 5′-CCAGGAGGAC-3′ | 70 | 34.6 | 83.33 |
| Total polymorphism percentage (%) | 78.56 | |||
| Control | 30 µM | 40 µM | 50 µM | |
|---|---|---|---|---|
| Control | 100 | 24.23 | 25.87 | 36.87 |
| 30 µM | 24.23 | 100 | 16.5 | 31.27 |
| 40 µM | 25.87 | 16.5 | 100 | 17.8 |
| 50 µM | 36.87 | 31.27 | 17.8 | 100 |
| Plant | Mutants | Fresh Weight Loss % | F-Value | Shoot Length Loss % | F-Value | Seed Germination Loss % | F-Value |
|---|---|---|---|---|---|---|---|
| Zea mays | −Ve Control | 0.00 ± 0.00 | 136.32 *** | 0.00 ± 0.00 | 93.56 *** | 0.00 ± 0.00 | 306.67 *** |
| +Ve Control | 80.3 ± 2.34 | 54.2 ± 2.14 | 93.5 ± 9.2 | ||||
| 30 µM | 50.5 ± 2.17 | 37.3 ± 1.24 | 47.5 ± 4.2 | ||||
| 40 µM | 26.4 ± 2.11 | 30.2 ± 1.11 | 33.3 ± 1.4 | ||||
| 50 µM | 12.5 ± 0.89 | 15.7 ± 0.52 | 20.2 ± 1.1 | ||||
| Allium cepa | −Ve Control | 0.00 ± 0.00 | 122.67 *** | 0.00 ± 0.00 | 110.33 *** | 0.00 ± 0.00 | 197.33 *** |
| +Ve Control | 86.3 ± 5.67 | 79.7 ± 5.47 | 91.7 ± 9.25 | ||||
| 30 µM | 59.7 ± 2.45 | 47.5 ± 3.33 | 67.7 ± 8.33 | ||||
| 40 µM | 42.5 ± 2.11 | 36.3 ± 1.27 | 29.3 ± 3.29 | ||||
| 50 µM | 23.7 ± 1.57 | 22.9 ± 1.15 | 11.6 ± 0.77 |
| Plant | Mutants | Proteins | Flavonoids | ||
|---|---|---|---|---|---|
| Mean | F-Value | Mean | F-Value | ||
| Zea mays | −Ve Control | 49.54 ± 0.37 | |||
| +Ve Control | 14.33 ± 0.35 | 298.799 *** | 0.073 ± 0.004 | 246.315 *** | |
| 30 µM | 23.28 ± 2.10 | 0.065 ± 0.005 | |||
| 40 µM | 36.09 ± 0.81 | 0.144 ± 0.006 | |||
| 50 µM | 38.90 ± 0.217 | 0.158 ± 0.003 | |||
| Allium cepa | −Ve Control | 28.26 ± 0.39 | |||
| +Ve Control | 2.38 ± 0.21 | 77.138 *** | 0.057 ± 0.002 | 101.025 *** | |
| 30 µM | 5.19 ± 0.21 | 0.064 ± 0.002 | |||
| 40 µM | 6.85 ± 0.37 | 0.089 ± 0.005 | |||
| 50 µM | 15.85 ± 2.24 | 0.097 ± 0.002 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tawfik, E.; Alqurashi, M.; Aloufi, S.; Alyamani, A.; Baz, L.; Fayad, E. Characterization of Mutant Aspergillus niger and the Impact on Certain Plants. Sustainability 2022, 14, 1936. https://doi.org/10.3390/su14031936
Tawfik E, Alqurashi M, Aloufi S, Alyamani A, Baz L, Fayad E. Characterization of Mutant Aspergillus niger and the Impact on Certain Plants. Sustainability. 2022; 14(3):1936. https://doi.org/10.3390/su14031936
Chicago/Turabian StyleTawfik, Eman, Mohammed Alqurashi, Salman Aloufi, Amal Alyamani, Lina Baz, and Eman Fayad. 2022. "Characterization of Mutant Aspergillus niger and the Impact on Certain Plants" Sustainability 14, no. 3: 1936. https://doi.org/10.3390/su14031936
APA StyleTawfik, E., Alqurashi, M., Aloufi, S., Alyamani, A., Baz, L., & Fayad, E. (2022). Characterization of Mutant Aspergillus niger and the Impact on Certain Plants. Sustainability, 14(3), 1936. https://doi.org/10.3390/su14031936







