Abstract
The documentation of local food resources among linguistic/cultural minorities is essential for fostering measures aimed at sustaining food biocultural heritage. Moreover, interdisciplinary studies on food cultural heritage represent a vital aspect of promoting environmental and social sustainability. The current study aimed to record the traditional foraging of wild food plants (WFPs) among three minority groups (Kalasha, Muslim Ismaili Yidgha, and Muslim Sunni Kamkata-vari speakers) as well as the dominant (Sunni Muslim) Kho/Chitrali people in the Kalasha and Lotkoh valleys, Chitral, NW Pakistan. A field survey recorded fifty-five locally gathered wild food plants and three mycological taxa. Most of the WFPs were used raw as snacks or as cooked vegetables, and Yidgha speakers reported the highest number of WFPs. Although the wild food plant uses of the four considered groups were quite similar, Yidgha speakers exclusively reported the use of Heracleum candicans, Matricaria chamomilla, Seriphidium brevifolium, and Sisymbrium irio. Similarly, Kalasha speakers reported the highest number of use reports, and along with Yidgha speakers they quoted a few WFPs that were frequently used only by them. The results of the study showed a remarkable degree of cultural adaptation of the minority groups to the dominant Kho/Chitrali culture, but also some signs of cultural resilience among those linguistic and religious minorities that were historically more marginalized (Kalasha and Yidgha speakers). The recorded food biocultural heritage should be seriously considered in future development programs aimed at fostering social cohesion and sustainability.
1. Introduction
In today’s complex socio-ecological systems, biocultural approaches shaping human culture relationships have gained extraordinary interest across different scientific disciplines [1]. Although, historically, biocultural-centered reflections mainly emerged from the field of biological anthropology, where they addressed the impacts of social environment on human biology and health, their original dimensions are much wider and more complex [2]. Since the concept of biocultural diversity has been defined by Maffi [3] as “the diversity of life in all its manifestations—biological, cultural and linguistic—which are interrelated within a complex socio-ecological adaptive system”, this idea found an important arena for discussion within the Convention on Biological Diversity (CBD); The United Nations Educational, Scientific, and Cultural Organization (UNESCO) [4]; and later the Florence Declaration of 2014 [5]. Researchers have claimed that biocultural approaches have the ability to bridge various knowledge systems and policy frameworks which could play a useful role in pursuing sustainability goals [1,6].
Sustainability science recognizes the inextricable link between nature and culture, which in many ways spans values, norms, beliefs, practices, knowledge, livelihoods, and also languages. Living in complex socio-ecological systems, the interconnections between the two pillars of the living planet can only be understood through a holistic approach [7]. Sustainable approaches to solve problems primarily fall into two main categories: (a) the conservation of biocultural entities; and (b) fostering their resilience—i.e., their balanced coevolution and transformation [8].
Biocultural heritage underpins the various interrelations that biological diversity has with the language, cultural memory, ecological knowledge, and social values of local and indigenous communities [9,10,11,12,13]. Therefore, the biocultural heritage held by local communities could play an important role in both social and environmental sustainability [10,11,12,13,14,15]. As endorsed by the Convention on Biological Diversity [16], it is highly advisable to respect and protect the innovations, knowledge, and practices that local communities have shaped in connection to the sustainable use of biological resources. The key role that local ecological knowledge (LEK) and lifestyle play in maintaining sustainable management practices must be acknowledged because, without such recognition, stewardship as a praxis will remain overlooked as a fundamental driver of sustainability [17]. Threats to biocultural heritage can be understood as those indirect drivers that erode the knowledge and capacity of human communities to live within ecological limits and can be found throughout contemporary economic and development practices [18]. Practices related to biocultural heritage are also closely linked to the construction of identities and social cohesion [19,20,21,22,23]. Global mobility and modernization processes have brought about several challenges to the LEK system, in terms of the standardization and suppression of local diversities and bio-cultural heritage. Moreover, in a multi-ethnic society, these processes create the situation in which majority or dominant cultures tend to impose, in one way or another, their agenda and value system on minority groups. Therefore, multicultural environments are always confronted with unequal power relationships in which an imposed mono-culturalism often emerges at the expense of minority heritage systems. UNESCO has protected minorities and their “intangible cultural heritage (ICH)” since 1989, pursuant to the UNESCO Recommendation on the Safeguarding of Traditional Culture and Folklore [24]. For every culture, but especially for minority and indigenous communities, ICH is an indispensable pillar of cultural identity. Among the different expressions of ICH, an important role is played by perceptions, uses, and practices linked to the natural environment, i.e., by ethnobotanical, ethnozoological, and ethnoecological knowledge systems. The ethnobotany of wild food plants (WFPs) is becoming an increasingly important topic in ethnoscience, as ethnobotanical knowledge, mainly retained by women or attached to domestic practices managed by women, has often been neglected, despite its importance in fostering the food sovereignty and security of local communities.
This ethnobotanical study aimed to document the biocultural heritage linked to wild food plants (WFPs) among three linguistic minorities residing in the Chitral region of north-west Pakistan, which could play a central role in shaping local social sustainability and sovereignty. Among the various minority groups in the region, Kalasha, Yidgha, and Kamkatka-vari speakers represent the most widely recognized minorities of the area. The Kalasha, who speak a Dardic language, represent a religious, ethnic, and linguistic minority group and are considered the last pagan tribes of the entire Hindu Kush mountain range; the community is highly marginalized, vulnerable, and endangered and is increasingly facing pressures from globalization and social change, which may be influencing youth and community development [25]. Yidgha speakers represent an important linguistic minority in NW Pakistan, since they speak a moribund Iranic language, while occupying the upper regions of the Lotkoh Valley in Western Chitral. Kamkata-vari speakers represent the main group of the Nuristani group languages, which are spread along the Afghan–Pakistani border in Chitral. Finally, the study also included the majority Kho people of Chitral (also known as Chitralis), who speak Khowar, a Dardic language.
In this study, we therefore investigated the effect that linguistic and religious affiliations have on the consumption of WFPs in the remote mountainous valleys of Chitral, northern Pakistan. The objective of this study was to analyze the differences and commonalities in traditional WFP uses among different linguistic and religious communities living in the Kalasha and Lotkoh valleys, which encompassed the following three sub-objectives: (a) to assess the wild food ethnobotany of the considered area; (b) to compare the collected data with the food ethnobotanical literature of Pakistan in order to identify possible novel wild plant food uses; and (c) to compare the gathered data among the four considered linguistic communities in order to possibly understand the cultural adaption processes the minority groups may have undergone.
2. Materials and Methods
2.1. Study Area and Communities
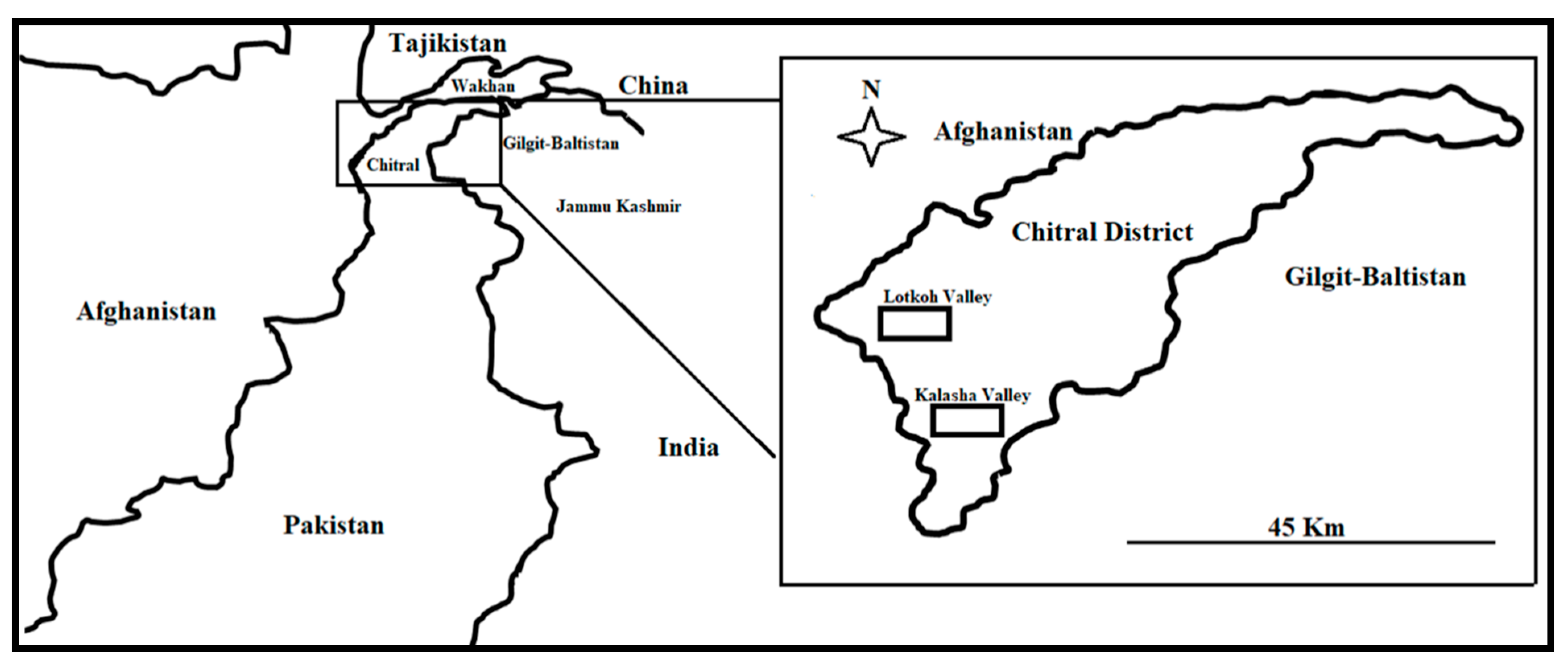
The study was conducted in the Kalasha and Lotkoh valleys, Chitral District, north-west Pakistan (Figure 1). The two valleys are part of the Hindu Kush mountain range, whose landscape is depicted in Figure 2.

Figure 1.
Map of the study area and visited valleys.

Figure 2.
Landscape of the study area: (a) Kalasha Valley, (b) Lotkoh Valley.
The demographic characteristics of the selected studied groups are presented in Table 1. In the Kalasha Valley, the Kalasha were the earliest settlers, while the Kho migrated there in the 14th century. Israr-ud-Din [26] claimed that the Kalasha ruled Chitral for centuries, but currently they have a restricted geographical distribution and are concentrated only in the Rumbur, Bumburet, and Birir valleys of District Chitral. In 1320 A.D., the Kalasha were invaded by Rais Mehtars (Khowar speakers), at which time they retreated into the few southern valleys they presently occupy [27]. In the Lotkoh Valley, the Yidgha occupy all the villages located in the upper reaches of the valley except for Gobar village, where both the Yidgha and Kamkata-vari/Nuristani people live together. The Yidgha came to the valley in the 11th century, while Kamkata-vari speakers settled later in the 17th and 18th centuries.

Table 1.
Characteristics of the study participants.
2.2. Field Study
A field ethnobotanical study was carried out from July to August 2019 in 8 mountain villages in the Kalasha and Lotkoh valleys of Chitral region, north Pakistan. The main purpose of the survey was to identify and record the traditional ethnobotanical uses of WFPs and mushrooms among the four selected linguistic and religious groups that live in different villages across the two valleys. It is important to underline that—as should always happen in modern participatory-centered ethnobiology—we considered all the “wild plants” as emically perceived by the locals, without any “etic” consideration regarding their plant biology (i.e., non-native plants, semi-domesticated species, and plants that have reverted to a wild state were included if locals considered them “wild”). Study participants were selected among middle-aged and elderly inhabitants (range: 52 to 69 years old) using snowball techniques, favoring those locals who are active farmers and shepherds and are therefore more exposed to wild natural resources and can be considered traditional knowledge holders in the study area (Figure 3). For the semi-structured interviews, twenty participants were chosen from each of the studied group, including both male and female community members. It is important to note that we were not allowed to interview female community members of the Sunni cultural group in order to respect their practice of Pardah (veil).

Figure 3.
Some of the informants interviewed during the field study.
During the course of the study, the code of ethics recommended by the International Society of Ethnobiology (ISE) [28] was strictly followed. From each of the participants, verbal consent was obtained before conducting the interview. Interviews were carried out in the Urdu language as well as in local languages with the help of a local translator. The interviews mainly focused on the WFPs that were gathered and consumed by each of the studied groups. Interviews aimed to record WFPs used as vegetables, as salads, as snacks, as seasoning, in fermentation, and in recreational teas. In the current study, the criteria adopted for recording the gathered WFPs were established by Termote et al. [29]. Specific questions were asked about the WFPs used in lacto-fermented foods and dairy products. Moreover, questions were asked about the consumption of edible mushrooms. For each of the reported species, the local name, used part(s), food uses, and gathering area were documented. In addition, qualitative ethnographic information of the studied communities was obtained through open-ended questions as well as participant/self-observation. The reported WFPs were then collected from the study area and were identified by the third author using the national taxonomic databases, especially the Flora of Pakistan [30,31,32,33]. After correct identification, each taxon was given a voucher specimen number and deposited at the Herbarium of the Department of Botany, University of Swat, Khyber Pakhtunkhwa, Pakistan. The identification of the few wild plants for which it was not possible to collect vouchers was made on the basis of the folk name and detailed plant description only. Each taxon was given an updated botanical name using The Plant List database [34], and for the mushroom taxa the Index Fungorum [35] was followed. Plant family assignments were verified through the Angiosperm Phylogeny Website [36].
2.3. Data Analysis
The recorded ethnobotanical taxa were processed using MS Excel. Later, the data were grouped into two datasets: one containing the overall used plants and the other comprising the most frequently reported plants (plant uses quoted by more than 50% of the participants). These two datasets, which were generated for each of the four studied communities, were compared through proportional Venn diagrams which were drafted using free software (http://bioinformatics.psb.ugent.be/webtools/Venn/). Venn diagrams are primary diagrams showing all possible relationships between finite collections of different sets and depict elements as points in the plane and set as regions inside closed curves.
Additionally, a comparison of the ethnobotanical data among the considered linguistic groups was carried out by calculating the Jaccard Similarity Index (for each pair of the considered communities), used for gauging the similarity and diversity of sample sets, following the application designed by González-Tejero et al. [37] for the ethnobotanical domain.
The Jaccard similarity index was calculated as follows:
- J(X, Y) = |X∩Y|/|X∪Y|;
- X = Individual set of plant uses recorded among group X;
- Y = individual set of plant uses recorded among group Y.
Furthermore, the collected data were qualitatively compared with the existing Pakistani food ethnobotanical literature [38,39,40,41,42,43,44,45,46] in order to identify possible novel plant uses.
3. Results and Discussion
3.1. Wild Food Plants and Their Uses
The wild plant/mushroom-based gastronomic cultural heritage of the studied linguistic and religious minorities comprises fifty-eight taxa, which are used in the considered valleys. All the recorded wild taxa are native except Medicago sativa, Matricaria chamomilla, Chenopodium album, Amaranthus hybridus, and Portulaca quadrifida, which are naturalized aliens that grow wild as weeds in anthropogenic environments. This study represents the first effort to record the local names of reported WFPs among the investigated groups. The WFPs locally gathered and consumed are presented in Table A1 (see Appendix A).
Some of the WFPs locally gathered by the local communities are illustrated in Figure 4. With reference to plant parts, fruit was the dominant organ used and the majority of WFPs were utilized as raw snacks, followed by wild taxa, which were used as cooked vegetables. Raw snacks are indeed an interesting phenomenon in food anthropology, and they have been reported in several ethnobotanical studies [47,48,49], where it has been argued that snacks may have emerged during the development of mobile pastoralism. The findings here underline the idea that pastoralism might play a crucial role in shaping food habits and an element of cultural identity. Moreover, pastoralism also shapes human–environment relationships, as has been well described by UNESCO [50,51,52,53]. Pastoralism is often related to specific categories of plants; for instance, in Iraq, Kurdish pastoralists were found to consume more snacks than the neighboring and more horticulturalist-driven Kakai [48] and much more than Assyrians [54]. As most of the raw snacks were fruits, sweetness can be recognized as an important cultural preference in the pastoralist culture of the studied communities. The most commonly reported snack plants mentioned by all the studied groups were Berberis lycium, Cotoneaster nummularius, Crataegus songarica, Echinops echinatus, Elaeagnus angustifolia, Prunus dulcis, Rheum ribes, Solanum americanum, and Tulipa clusiana (Sõukand and Kalle) [55]. Moreover, Solanum americanum is known to contain toxic alkaloids [56], which are mainly found in its fruit [57], but nevertheless locals in the current study area snack on a limited amount of berries and they did not report any toxic effects.

Figure 4.
A few wild specimens collected for the herbarium: (a) Sorbus aria; (b) Rubus fruticosus; (c) Berberis lycium; (d) Portulaca quadrifida; (e) Allium sp.; (f) Nasturtium officinale; (g) Rheum ribes; (h) Rumex dentatus; (i) unidentified fungal taxon.
In the study area, we observed in a local field that wild Sorbus aria was grafted onto Crataegus songarica by a Kalasha man, which indicates that desire for certain plants drives people to alter the environment to suit their own needs (Figure 5). Some of the most frequently reported plant species that were cooked as vegetables included Allium spp., Amaranthus hybridus, Arum maculatum, Capparis spinosa, Chenopodium album, Medicago sativa, Morchella vulgaris, Portulaca quadrifida, Rumex spp., and Urtica dioica, and most of these reported wild vegetables were largely gathered in anthropogenic environments. Considering the importance of gathering environments, anthropogenic environments are important habitats providing space for WFPs [58]. Moreover, the average and total values of the cultural importance of plants gathered in different environments highlights the significance of the gathering environment, compared to looking solely at the number of plant species gathered [59]. Among the reported species, ten taxa were utilized in making tea, while very few were used in salads or as seasoning.

Figure 5.
(a) Sorbus aria that was previously grafted onto Crataegus songarica by a Kalasha man at the margin of a corn field; (b) collecting wild food plants for the herbarium in a summer pasture in the Lotkoh Valley.
Almost half of the recorded WFPs (22 taxa) were reported by more than fifty percent of the participants, even though in many parts of the world traditional knowledge has drastically decreased [38,45,60,61,62,63,64,65]. In a recent other study that the authors conducted in north Pakistan [38], three quarters of the quoted wild food plants were reported by more than fifty percent of informants. On the other hand, in a nearby region Abbas et al. [39] reported that the traditional ecological knowledge (TEK) of WFPs is partially eroded and that the majority of the reported species were quoted by approximately only one third of the informants. The TEK of wild food plant ingredients is not only linked to local biodiversity and the availability of plants, but is also deeply embedded in daily food practices, which are in turn highly variable and influenced by a complex combination of socio-cultural factors, such as the pervasiveness of industrialized food, food security status/socio-economic conditions, the importance of cultural identities, and so on. Where this heritage is still alive, locals have greater knowledge of wild ingredients; this was the case, for example, among the communities around the Thakht-e-Sulaiman Hills, NW Pakistan [42]. The studied groups frequently reported Allium spp., Berberis lycium, Cotoneaster nummularius, Carum carvi, Chenopodium album, Crataegus songarica, Eremurus stenophyllus, Medicago sativa, Morchella esculenta, Portulaca quadrifida, Rheum ribes, and Taraxacum campylodes. The WFPs reported in the study were mostly gathered during daily routines and were nearly identical among the groups. All the wild vegetables were mainly consumed in young stages of growth, and the green leaves or aerial parts of respective plant taxa were generally used. It is essential to note that local communities did not mention any wild plants used in fermentation. After conducting a comprehensive comparison with the Pakistani food ethnobotanical literature, the following wild plants emerged as possible novel or rare food ingredients for the country: Angelica glauca, Arum maculatum, Descurainia Sophia, Ferula narthex, Heracleum candicans, Juniperus excels, Sisymbrium irio, Viola odorata, and Ziziphora clinopodioides.
We also found some wild vegetables which have completely disappeared from the traditional food system, such as Arum maculatum, Medicago sativa, Rumex spp., and Urtica dioica. It is also alarming that, in recent decades, social and environmental change has greatly impacted the dependence of local communities on their local ecosystem. Market access and other economic factors involved in the process of the rapid social evolution of these communities could be a serious threat to sustainability for both the environment and natural resources. TEK regarding WFPs is mainly held by community elders, and intergenerational transmission is quite limited because of the low number of young people living in these villages, as they spend most of their time in cities in search of jobs, business, and education. TEK is often recognized by young villagers as something useless and outdated, which often “needs to much time” as also reported by Kalle and Sõukand [66], who coined the term “unlearning debt” to define the phenomenon in which explicit knowledge of local practices is still alive in the memory of the aged people, but does not transfer anymore to younger generations and thus is destined to be forgotten.
3.2. Cross-Cultural Comparison
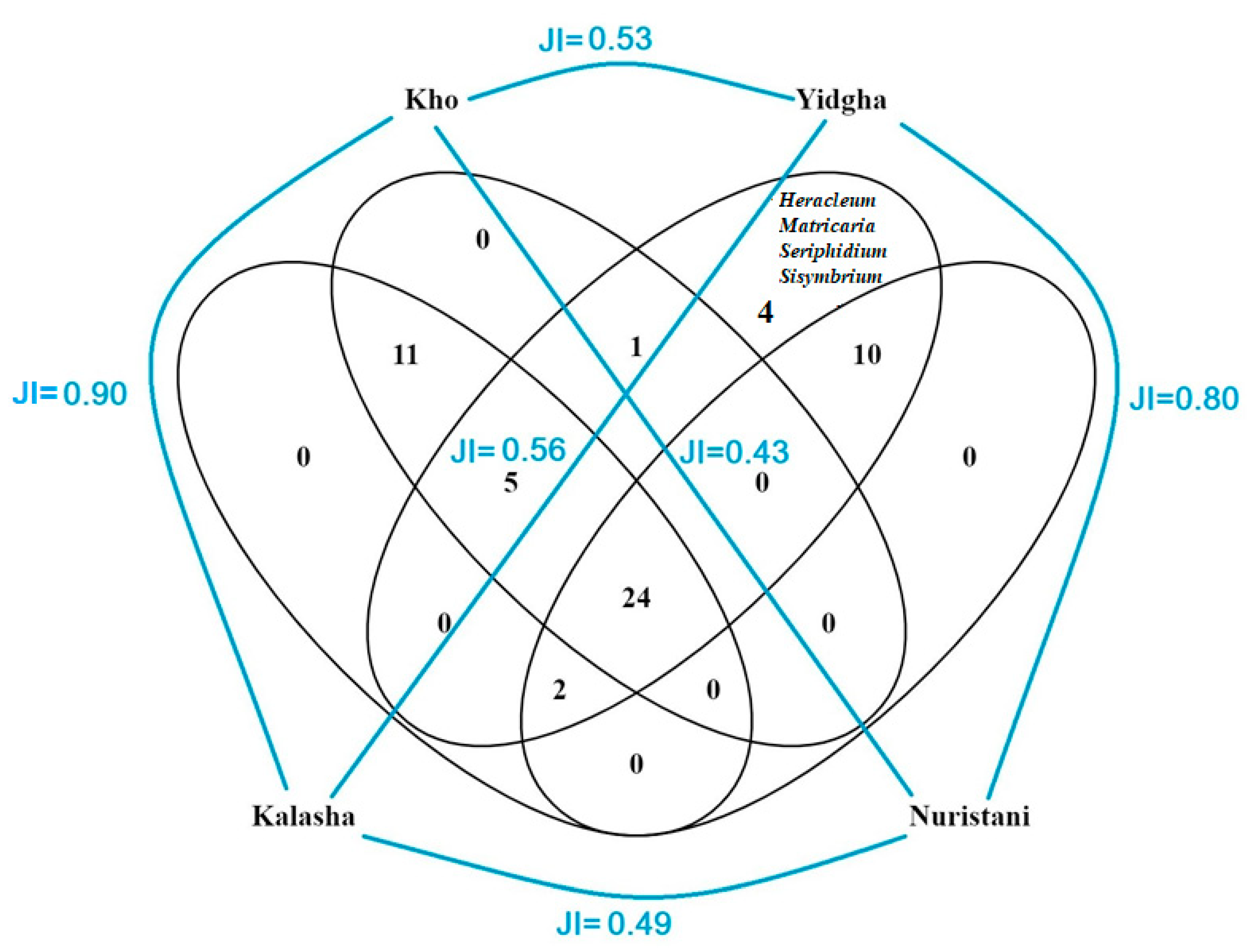
Cross-cultural comparison shows that the WFP uses of the four studied groups are quite similar. The WFPs used among the four studied communities and their related Jaccard similarity indexes are presented through a Venn diagram (Figure 6). The remarkable overlap of wild food plant uses between the Kalasha and the Kho and also Yidgha and Kamkata-vari speakers may suggest the strong cultural adaptation of the Kalasha to the majority Kho people, and remarkable social exchange between Yidgha and Kamkata-vari speakers, given the fact that they live together in the Lotkoh Valley.

Figure 6.
Venn diagram showing Jaccard similarity indexes and overlap of all reported wild food plants and mushrooms among the studied groups.
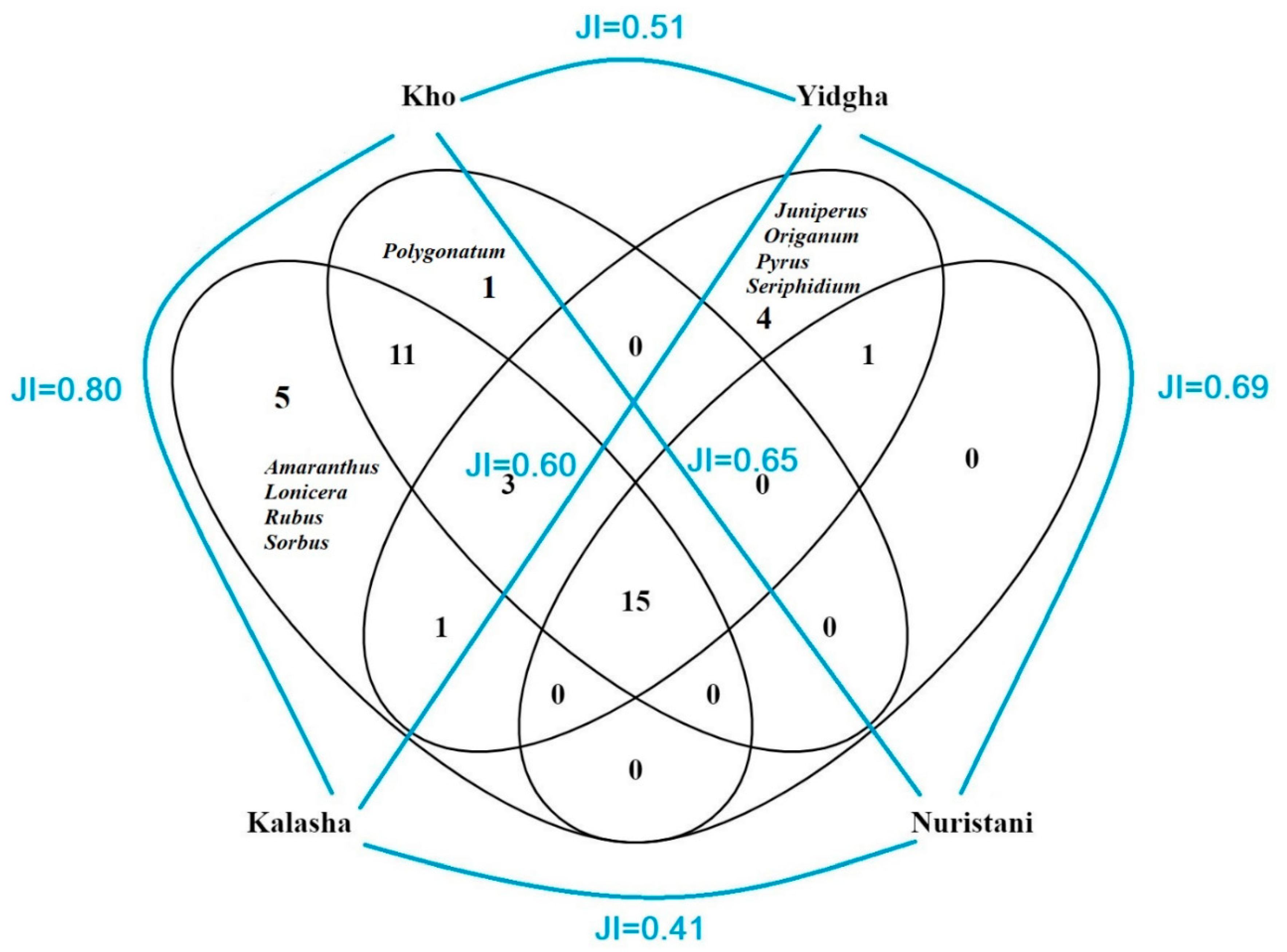
A comparative analysis of frequently used WFPs indicated that the Kalasha and the Yidgha used certain plants which were rarely reported by other cultural groups (Figure 7). Descriptive statistics indicate that Kalasha speakers show the greatest number of use reports (562 use reports; 28 use reports per participant), followed by the Kho (507; 25), the Yidgha (464; 23), and Kamkata-vari speakers (306; 15). More importantly, the Yidgha mentioned certain WFPs, such as Heracleum candicans, Matricaria chamomilla, Seriphidium brevifolium, and Sisymbrium irio, which were not reported among any other group. High similarity indexes among the different groups demonstrate that traditional ecological knowledge (TEK) is following the pathway of homogenization, and this phenomenon may occur because plant gathering and possibly food customs are generally not perceived as being identitarian in the study area. Moreover, the sharing of plant knowledge may be considered a phenomenon of cultural assimilation and standardization to the dominant culture of the Kho/Chitralis. Cultural assimilation might have been triggered by historical, political, and religious pressures that the minor groups underwent. For instance, the Kalasha represent an ethnic group in southern Chitral that has been politically and religiously oppressed for almost three hundred years. In 1320 A.D., the Chitral area was invaded by a few Rais Mehtars (Khowar speakers) and came under their political and religious influence [26,27]. Consequently, many original Kalasha people were Islamized, which led to intermarriages with other cultural groups such as the Kho and the Nuristani. These intermarriages may have resulted in a homogenization of kinship relations and, attached to them, the oral transmission of TEK in the study area, as hypothesized in other studies [38,66,67,68,69].

Figure 7.
Venn diagram showing Jaccard similarity indexes and overlap of the most frequently reported wild food plants and mushrooms (quoted by more than 50% of informants) among the studied groups.
In the same way, Yidgha speakers represent a linguist minority in the upper Lotkoh Valley [70] which underwent significant socio-cultural and linguistic exchanges with the Kho majority during the past few decades and may possibly act as a bridge for the flow of traditional knowledge. Socio-linguistic adaptability could clearly be observed while recording similar local names for certain plant taxa. For instance, Celtis australis was called Binjo, Portulaca quadrifida was referred to as Pichili, Rubus fruticosus was called Atchu, and Silene conoidea was referred to as Hapupar. This linguistic adaptation was possibly linked to a broader cultural adaptation that minority groups in a given area underwent toward majority groups or groups speaking the Kho lingua franca. Since identity is mainly expressed through language and sometimes religion as well, the commonalities in local names of certain taxa may indicate an erosion of the linguistic and bio-cultural heritage of each individual minority group. We also observed on occasion that some of the participants among the Yidgha and Kamkata-viri speakers did not remember the local names of certain WFPs in their local languages and thus reported local names in Kho. The loss and homogenization of knowledge among minority groups may be facilitated by linguistic erosion—i.e., the erosion of vernacular mnemonics [71]. As found also in the Caucasus and North Pakistan [38,69], minority groups sometimes use terms expressed in the majority language for naming wild plants. It is quite alarming that the linguistic adaptation of minority groups in our study area (especially Yidgha and Kamkata-vari speakers) could also come at the expense of language survival and that minority languages are moribund and subject to what Skutnabb-Kangas [72] defined in 2000 as “linguicide”.
Linguicide is not only relevant to linguistic issues, as, even more dramatically, it often entails more subtle cultural assimilation processes, in which communities no longer perceive their own local customs as valuable and instead tend to mimic the dominant code/culture. In our study area, we can hypothesize, especially when looking at the still partially idiosyncratic wild food ethnobotany of Kalasha and Yidgha speakers, that before WFPs were “homogenized”, unique plant ingredients and food utilizations may have existed and, later in the 20th century, disappeared or were “forcibly” forgotten. The linguistic literature also indicates that in the past these exchange processes may have been more mutually beneficial, as, for example, numerous Khowar words actually have a Yidgha origin [73]. Today, it seems that the Yidgha have been culturally assimilated into the Kho population, and their position on the ancient trade route between Chitral town and the Dorah Pass may have contributed to their subjugation by the dominant Chitralis.
3.3. The Importance of the Cultural Heritage of Marginalized Minorities
In order to implement social sustainability, socio-ecological systems urgently need to respect and celebrate cultural diversity, especially that of marginalized groups. Marginalized groups are often vulnerable and feel, or are made to feel, less privileged, less important, and less respected than those who hold more power, status, privilege, and opportunities in society. In general, they may be considered outside of the “mainstream” way of thinking and behaving [74]. One of the most difficult feelings to rid oneself of is the emotional turmoil connected with being marginalized by a person or group in a position of power [75]. Being seen and treated as less than others or excluded in some way has a long-lasting impact on the mental health of an individual, and marginalized individuals are also more susceptible to the effects of stress [76,77,78]. On an emotional and psychological level, individuals can feel isolated from the social fabric of their larger communities and can feel a sense of paranoia, which is a function of how they might be perceived and treated by others. They can feel invisible, as though their concerns are not significant enough to be heard, and self-doubt and frustration are common psychological responses. Therefore, some marginalized individuals or groups are also at higher risk of self-harm and suicide [79].
One way to eliminate the feeling of deprivation among ethnic minorities is to pay special attention to the protection of their biocultural heritage. Due to their less privileged status in society, ethnic minorities are more vulnerable to losing their heritage; although they do not want to lose their cultural entity, they always try to integrate into the mainstream, where the “dominant way of thinking and behaving” is frequently followed. Therefore, it is essential to honor and celebrate their biocultural heritage in order to better make the majority groups aware of the cultural richness and values they possess; this will possibly counter the negative impact of intolerance and discrimination.
In a pluralistic society, it has always been difficult to maintain social cohesion among the different members of the society without considering three dimensions: social relationships, a sense of belonging, and orientation toward the common good [80]. To maintain social cohesion, one of the most important things which needs to be seriously reconsidered is the celebration of biocultural elements in future development programs. This will not only foster a sense of belonging and security for minorities, but also help them to fight exclusion and marginalization, and to reformulate their social networks to share cultural values and aspirations. As rightly described by the United Nations World Commission on Environment and Development (WCED), the success of sustainable development requires a favorable social context that can only be achieved by means of social cohesion [81]. Social cohesion demands the integration and coordination of all members and groups of a given society, and in the ongoing context it is dire to bring ethnic minorities into mainstream society to achieve sustainable goals. As described earlier, the field of biocultural diversity has given rise to transdisciplinary research, and the impetus for the emergence of this field came from the observation that linguistic, cultural, and biological diversity are under threat which may bring about serious consequences for humanity and the earth. Therefore, sustainability science recognizes the tight coupling between humans and their environment within a complex, adaptive system which requires a holistic approach to studying it [7].
3.4. Role of Wild Food Plants and Future Food Sovereignty
Wild food plants have remained an important ingredient of the local food basket in the study area. During the field survey, we visited pastures located in the upper elevations of valleys which are locally known as “Soon” in Kalasha, “Ghari” in Khowar, “Passo” in Kamkata-vari, and “Kharovoo” in Yidgha. These summer pastures are considered reservoirs of several important food and medicinal plant species, as described in another study conducted in the Yasin Valley of Gilgit-Baltistan [38]. During the study we also found some plants being sold in markets, including Eremurus stenophyllus and Rheum ribes, which are very popular wild vegetables in the Chitral region. Additionally, we noticed certain WFPs, such as Allium spp., which the local people brought to their houses from the mountains. Locals emphasized that the foraging of WFPs has been decreasing in recent times, although certain wild vegetables like Allium spp., Chenopodium spp., Eremurus stenophyllus, and Portulaca quadrifida are still foraged and part of the local everyday food system. Informants quoted that the growth of WFPs has drastically decreased due to advancing anthropogenic activities such as unhealthy agricultural practices and environmental degradation. People living in summer pastures cause extensive overgrazing which disturbs the natural health of the ecosystem. In the valleys, rapidly increasing agriculture practices have led to the conversion of a considerable portion of the land to agricultural fields (Figure 8). Research has indicated that various socioeconomic and biophysical factors that have led to a depletion of natural resources across the Hindu Kush Himalayan region, which has resulted in a significant loss of ecosystem services [82]. It is well established that wild food constitutes a substantial part of household food consumption around the world, but rapid land use changes influence the availability of wild foods, which has implications for smallholders’ food and nutrient intake. Therefore, it is imperative to pay attention to natural resources and protect them from further exploitation in order to counter future food insecurity. Most importantly, stakeholders in the area should adopt a more nutrition-sensitive approach, in which local small-scale agricultural practices and biodiversity conservation policies are reviewed, in order to facilitate wild food provisioning and food security. In this context, the Food and Agriculture Organization (FAO) also recognizes that “nutrition and biodiversity converge to a common path leading to food security and sustainable development” and that “wild species and intra-species biodiversity have key roles in global nutrition security” [83]. Just as the environment is facing losses through prevailing unhealthy anthropogenic practices, deteriorating nature, communities are becoming more significantly disconnected from the natural world and its ecological processes. Keeping sustainable growth initiatives as priorities, we need to value the TEK of natural resources and, at the same time, integrate traditional food practices within the existing farming practices of the local area. It is certain that wild food resources cannot satisfy demand but, at the same time, without them the gap between food supply and demand will be much wider in the future than predicted. It is vital to formulate policies on food security and the conservation of biodiversity and to recognize a transition zone in order to perfectly integrate these areas so as to elevate the significance of wild species.

Figure 8.
Local people working in agricultural fields: (a) Kalasha Valley, (b) Lotkoh Valley.
4. Conclusions
This study recorded considerable TEK of WFP uses and represents the first scientific documentation reporting wild plant food ingredients along with their vernacular names among the linguistic minorities of the Chitral area. The study reported fifty-five plant taxa and three mushrooms which were used in traditional food systems among the different researched groups. A remarkable number of WFPs were used as snacks, as well as wild vegetables, which may represent the dominance of the pastoral lifestyle that has been practiced by these communities for generations. Aerial parts and fruits were the most widely consumed plant parts in the study area. A comparative analysis indicated that the WFP uses of the studied groups were quite similar, although the Yidgha reported the highest number of WFPs. The Kalasha reported the greatest number of use reports for the quoted taxa and, along with the Yidgha, also retained a deeper knowledge of specific, frequently reported WFPs. A comparative analysis with the ethnobotanical literature of Pakistan revealed a few taxa as novel or rare wild plant ingredients: Angelica glauca, Arum maculatum, Descurainia Sophia, Ferula narthex, Heracleum candicans, Juniperus excelsa, Sisymbrium irio, Viola odorata, and Ziziphora clinopodioides. Most importantly, the biocultural heritage of WFPs held by the three linguistic minorities—i.e., the Kalasha, Kamkata-vari, and Yidgha—seems to face certain threats and challenges, since these groups have presumably undergone linguistic and cultural adaptation processes to the dominant Kho/Chitrali culture.
The main limitations of the current research are that the field study was conducted during one single season, was not repeated for several years, and the selected sample was unequal in terms of gender among the Khowar and Nuristani speakers. Nevertheless, the current research could provide a baseline for future community-centered rural development programs aimed at implementing food security and food sovereignty. The local ethnobotanical knowledge urgently needs to be not only documented but also promoted and revitalized so as to possibly inspire, for example, initiatives in the field of ecological and sustainable tourism and gastronomy. Furthermore, in order to promote social sustainability, future research directions should more thoroughly address the ethnobotanical heritage of minorities in Pakistan and Asia in general, since these studies may be extremely important for increasing awareness of the value of cultural diversity among stakeholders and the general public. We also sincerely hope that the current study can encourage social cohesion in Chitral, as well as an effective recognition of the immense value of the linguistic and religious diversity in Pakistan.
Author Contributions
A.P. and M.A.A. planned and designed the theoretical and methodological framework of the research study; M.A.A. carried out the field study; Z.U. identified the ethnobotanical taxa and helped in addressing reviewer comments; M.A.A. analyzed the data, provided the cultural interpretation of the research findings, and drafted the first version of the manuscript, which was later revised by A.P. A.P. supervised all stages of the publication process. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the University of Gastronomic Sciences, Pollenzo, Italy.
Acknowledgments
Special thanks are due to all the study participants of the different groups who generously shared their knowledge.
Conflicts of Interest
The authors have no conflict of interest.
Appendix A

Table A1.
Gathered WFPs and mushrooms recorded in the study area.
Table A1.
Gathered WFPs and mushrooms recorded in the study area.
| Botanical Taxon/Taxa; Family; Voucher Specimen Code | Recorded Local Names | Parts Used | Gathering Areas | Recorded Gastronomic Uses | Number of Quotations | Previously Reported in Pakistan | |||
|---|---|---|---|---|---|---|---|---|---|
| Ka | Kh | N | Y | ||||||
| Allium carolinianum DC.; Amaryllidaceae; SWAT005962 | Latirk Y | Aerial parts | Mountain pastures | Cooked | 12 | 11 | 10 | 11 | Yes |
| Latruk Ka,Kh,N | |||||||||
| Allium spp.; Amaryllidaceae | Balakh-sha Y | Whole plant | Mountain pastures | Cooked | 17 | 13 | 11 | 15 | Yes |
| Kachiandook Ka | |||||||||
| Keelkasoo rKh | |||||||||
| Kolgho Y | |||||||||
| Koo N | |||||||||
| Lomanrr-u-shook N | |||||||||
| Treshto Kh | |||||||||
| Zoomshakho Kh | |||||||||
| Amaranthus hybridus L.; Amaranthaceae; SWAT005470 | Gandari Ka | Leaves | Fields, home gardens | Cooked | 16 | 16 | 14 | 17 | Yes |
| Kholdoor Y | |||||||||
| Khordool Kh | |||||||||
| Kondakh Kh | |||||||||
| Karpatik N | |||||||||
| Angelica glauca Edgew.; Apiaceae; SWAT005963 | Soghmano Y | Shoot | Mountain pastures | Raw snacks | - | - | 3 | 5 | No |
| Razo N | |||||||||
| Arum maculatum L.; Araceae; SWAT005964 | Barishtavon Ka | Leaves | Forests | Cooked | 15 | 12 | - | - | No |
| Pramolo Kh | |||||||||
| Berberis lycium Royle; Berberidaceae; SWAT004744 | Azito Y | Fruits | Foothills | Raw snacks | 10 | 11 | 10 | 15 | Yes |
| Chinitai N | |||||||||
| Choveng Kh | |||||||||
| Chrokee Ka | |||||||||
| Capparis spinosa L.; Capparaceae; SWAT005965 | Kaveer Ka,Kh | Flowers | Foothills, fields, plain areas | Cooked | 13 | 12 | - | - | Yes |
| Carum carvi L.; Apiaceae; SWAT005966 | Ayo N | Fruits | Mountain pastures, meadows, home gardens | Seasoning Tea | 15 | 15 | 14 | 16 | Yes |
| Hajyash Ka | |||||||||
| Hojooj Kh | |||||||||
| Zeero Y | |||||||||
| Celtis australis subsp. caucasica (Willd.) C.C.Towns.; Cannabaceae; SWAT005474 | Binjo Kh,Y | Fruits | River banks, cliffs | Raw snacks | 16 | 12 | - | 6 | Yes |
| Bizo Ka | |||||||||
| Chenopodium album L.; Amaranthaceae; SWAT005499 | Gandari Ka | Leaves | Fields and gardens | Cooked | 16 | 5 | 4 | 7 | Yes |
| Kondakh Kh | |||||||||
| Sheghiko Y | |||||||||
| Tretheray N | |||||||||
| Chenopodium foliosum Asch.; Amaranthaceae; SWAT005510 | Atchu Kh,Y | Fruits | Fields and meadows | Raw snacks | 10 | 14 | - | 6 | Yes |
| Ongacha Ka | |||||||||
| Clinopodium umbrosum (M.Bieb.) Kuntze; Lamiaceae; SWAT005506 | Ghalaghali Ka | Aerial parts | Fields and gardens | Cooked | 14 | 12 | - | - | Yes |
| Jalajali Kh | |||||||||
| Cotoneaster nummularius Fisch. & C.A.Mey.; Rosaceae; SWAT005485 | Amakhni Y | Fruits | Foothills | Raw snacks | 17 | 15 | 15 | 18 | Yes |
| Mikay N | |||||||||
| Mikeen Ka,Kh | |||||||||
| Crataegus songarica K. Koch; Rosaceae; SWAT005473 | Chachinkh Y | Fruits | Fields, foothills | Raw snacks | 13 | 13 | 11 | 12 | Yes |
| Gooni Kh,Y | |||||||||
| Jinjo Ka | |||||||||
| Seeu N | |||||||||
| Daucus carota L.; Apiaceae; SWAT005484 | Khaizgoom Kh | Roots | Fields | Raw snacks | 14 | 15 | - | - | Yes |
| Toruk Ka | |||||||||
| Descurainia sophia (L.) Webb ex Prantl; Brassicaceae; SWAT005793, SWAT005513 | Chatkosh N | Fruits | Mountain pastures | Tea | 5 | - | 4 | 4 | No |
| Chatkash Ka | |||||||||
| Kheelnakheel Y | |||||||||
| Echinops echinatus Roxb.; Asteraceae; SWAT005490 | Chamcheer Kh | Roots | Fields, gardens, meadows | Raw snacks | 17 | 13 | 3 | 6 | Yes |
| Changhan Ka | |||||||||
| Khakho Y | |||||||||
| Tanlou N | |||||||||
| Elaeagnus angustifolia L.; Elaeagnaceae; SWAT005806, SWAT005808 | Sichen Ka | Fruits Bark | River banks, stream courses, fields | Fruit; Raw snacks Bark; Tea | 12 | 17 | 3 | 7 | Yes |
| Sinjoor Kh | |||||||||
| Sonji N | |||||||||
| Soziu Y | |||||||||
| Eremurus stenophyllus (Boiss. & Buhse) Baker; Xanthorrhoeaceae; SWAT005967 | Shay Y | Leaves | Mountain pastures | Cooked | 14 | 14 | 13 | 16 | Yes |
| Shingish Ka | |||||||||
| Tekh-shakh Kh | |||||||||
| Vish N | |||||||||
| Ferula narthex Boiss.; Apiaceae; SWAT005968 | Aroos N | Leaves | Foothills | Salad | - | - | 8 | 14 | No |
| Rew Y | |||||||||
| Heracleum candicans Wall. ex DC.; Apiaceae; SWAT005500 | Krushak/Krushoo Y | Leaves | Mountain pastures | Tea | - | - | - | 5 | No |
| Juniperus excelsa M.Bieb.; Cupressaceae; SWAT005497, SWAT005498 | Siritchderray N | Fruits Leaves | Mountains | Tea | - | - | 7 | 14 | No |
| Tarparatong Y | |||||||||
| Lonicera griffithii Hook.f. & Thomson; Caprifoliaceae; SWAT005488 | Kagadrach Ka | Fruits | Mountain forests | Raw snacks | 15 | 10 | - | - | Yes |
| Moeedrach Kh | |||||||||
| Matricaria chamomilla L.; Asteraceae; SWAT005773, SWAT005772 | Sherwisht Y | Flowers | Foothills | Tea | - | - | - | 5 | No |
| Medicago monantha (C.A.Mey.) Trautv.; Leguminosae; SWAT004745 | Bargheekpola Y | Aerial parts | Fields | Raw snacks | - | - | 3 | 9 | No |
| Kovoropoo N | |||||||||
| Medicago sativa L.; Leguminosae; SWAT005797, SWAT005795 | Irghogho Y | Aerial parts | Fields | Cooked | 11 | 13 | 11 | 12 | Yes |
| Meekeush N | |||||||||
| Moshich Ka,Kh | |||||||||
| Mentha longifolia (L.) L.; Lamiaceae; SWAT005792, SWAT005790 | Ben Kh | Aerial parts | Water courses, home gardens | Salad Seasoning Tea | 20 | 20 | 20 | 20 | Yes |
| Boinj Ka | |||||||||
| Gilinwainrr N | |||||||||
| Walna Y | |||||||||
| Morchella esculenta (L.) Pers.; Morchellaceae; SWAT004746 | Shoti Kh | Aerial parts | Mountain forests, damp places | Cooked | 15 | 16 | 9 | 11 | Yes |
| Kotchi Kh,Ka | |||||||||
| Ghoshghosha Y | |||||||||
| Goktalook N | |||||||||
| Nasturtium officinale R.Br.; Brassicaceae; SWAT005482 | Chilawoor Ka,Kh | Leaves | Water courses | Salad | 14 | 14 | - | 10 | Yes |
| Terghay Y | |||||||||
| Origanum vulgare L.; Lamiaceae; SWAT005777, SWAT005504 | Gondostak Y | Leaves | Meadows | Tea | - | - | 8 | 12 | No |
| Chinai-wanrr N | |||||||||
| Pinus gerardiana Wall. ex D.Don; Pinaceae; SWAT004752 | Jalghoza Kh | Kernels | Mountain forests | Raw snacks Roasted | 20 | 20 | - | - | Yes |
| Kovaireekjak Ka | |||||||||
| * Polygonatum verticillatum (L.) All.; Polygonaceae: SWAT005969 | Manghoor Kh | Aerial parts | Mountains | Cooked | - | 15 | - | 4 | Yes |
| Margho Y | |||||||||
| Portulaca quadrifida L.; Portulacaceae; SWAT005970 | Dorol-yus N | Aerial parts | Fields, home gardens | Cooked Salad | 18 | 17 | 15 | 16 | Yes |
| Pichili Kh,Y | |||||||||
| Rohairak Ka | |||||||||
| Prunus dulcis (Mill.) D.A.Webb; Rosaceae; SWAT005480 | Kaanda Ka | Fruits Gum | Mountain forests | Fruits: Raw snacks Gum: Raw snacks | 6 | 8 | 5 | 6 | Yes |
| Kando Kh | |||||||||
| Ketay N | |||||||||
| Kitagh Y | |||||||||
| Pyrus pashia Buch.-Ham. ex D.Don; Rosaceae: SWAT005971 | Itchtong N | Fruits | Mountains | Raw snacks | 4 | - | 5 | 6 | Yes |
| Kachatong Ka | |||||||||
| Yarshkugoo Y | |||||||||
| Quercus baloot Griff.; Fagaceae; SWAT004748 | Banj Kh | Kernels | Mountain forests | Raw snacks | 20 | 20 | - | - | Yes |
| Bonjmoot Ka | |||||||||
| Rheum ribes L.; Polygonaceae; SWAT004749 | Chewo N | Aerial parts | Foothills | Raw snacks | 11 | 15 | 13 | 18 | Yes |
| Chokri Y | |||||||||
| Chokriyo Y | |||||||||
| Khoongara Y | |||||||||
| Ishpar Kh | |||||||||
| Srakohon Ka | |||||||||
| Ribes alpestre Wall. ex Decne.; Grossulariaceae; SWAT005802 | Chinai-mikay N | Fruits | Meadows, mountains | Raw snacks | - | - | 6 | 9 | Yes |
| Tawdogho Y | |||||||||
| Ribes orientale Desf.; Grossulariaceae; SWAT005781 | Ashisht Y | Fruits | Mountains | Raw snacks | - | - | 6 | 9 | Yes |
| Zeepodik N | |||||||||
| Rubus vestitus Weihe; Rosaceae; SWAT005471 | Atchu Kh,Y | Fruits | Fields, soggy places | Raw snacks | 10 | 7 | - | 6 | Yes |
| Ongacha Ka | |||||||||
| Rumex dentatus L.; Polygonaceae; SWAT005468 | Chiko N | Leaves | Home gardens, fields | Cooked | 11 | 11 | 7 | 9 | Yes |
| Chrikon Ka | |||||||||
| Salkhikoo Y | |||||||||
| Sirkonzoor Kh | |||||||||
| Rumex hastatus D. Don; Polygonaceae; SWAT005801 | Cheeryak Y | Leaves | Home gardens, fields | Raw snacks Cooked | 10 | 6 | - | 7 | Yes |
| Chroki Ka | |||||||||
| Shotako Kh | |||||||||
| Seriphidium brevifolium (Wall. ex DC.) Ling & Y.R.Ling; Asteraceae; SWAT004751 | Borborrook/Bospook | Fruits | Foothills | Raw snacks | - | - | - | 14 | Yes |
| Silene conoidea L.; Caryophyllaceae; SWAT005481, SWAT005514 | Alodoichak Ka | Aerial parts | Fields | Cooked | 15 | 10 | 6 | 10 | Yes |
| Hapupar Kh,Y | |||||||||
| Sookchalyoos N | |||||||||
| Sisymbrium irio L.; Brassicaceae; SWAT005462 | Shinai Y | Fruits | Mountain pastures, fields | Seasoning | - | - | - | 7 | No |
| Solanum americanum Mill.; Solanaceae; SWAT005503, SWAT005803 | Anechek N | Fruits | Field banks | Raw snacks | 7 | 8 | 6 | 6 | Yes |
| Keemalook Ka | |||||||||
| Parmalik Y | |||||||||
| Pilmileek Kh | |||||||||
| Pirmileek Kh | |||||||||
| Sorbus aria (L.) Crantz; Rosaceae; SWAT004747 | Tweshvalavo Ka | Fruits | Mountain forests | Raw snacks | 18 | 7 | - | - | Yes |
| Shoth-Palaokh Kh | |||||||||
| Sorbus sp.; Rosaceae | Matravalavo Ka | Fruits | Mountain forests | Raw snacks | 12 | 5 | - | - | Yes |
| Shutpaloakh Kh | |||||||||
| Taraxacum campylodes G.E.Haglund; Asteraceae; SWAT005972 | Chinaiparr N | Leaves | Fields, home gardens | Salad | 11 | 10 | 10 | 10 | Yes |
| Choghoz-gulla Y | |||||||||
| Ishkanacho Kh | |||||||||
| Kashpiyak Ka | |||||||||
| Pavoo Kh | |||||||||
| Tulipa clusiana DC. var. chrysantha (A.D.HALL) Sealy; Liliaceae: SWAT005973 | Ishkoon Kh | Bulbs | Foothills | Raw snacks | 9 | 7 | 4 | 6 | Yes |
| Kashisho Ka | |||||||||
| Mirjooneek Kh,Y | |||||||||
| Meejang N | |||||||||
| Plakhdini Kh | |||||||||
| Urtica dioica L.; Urticaceae; SWAT005501 | Chomanyak Ka | Leaves | Fields, home gardens | Cooked | 14 | 13 | 9 | 14 | Yes |
| Drozono Kh | |||||||||
| Kholkhadi Y | |||||||||
| Dotchi N | |||||||||
| Viola odorata L.; Violaceae; SWAT005974 | Benavoosh N,Y | Leaves | Meadows | Tea | - | - | 6 | 8 | No |
| Ziziphora clinopodioides Lam.; Lamiaceae; SWAT005975 | Zoghoor N,Y | Aerial parts | Mountain pastures | Tea | - | - | 12 | 10 | No |
| Unidentified plant taxon | Semay-gai N | Aerial parts | Mountain pastures | Cooked | - | - | 5 | 6 | Yes |
| Somani Y | |||||||||
| Unidentified plant taxon | Kalamadook Ka | Bulbs | Foothills | Raw snacks | 13 | 9 | - | - | - |
| Paiponik Kh | |||||||||
| Unidentified fungal taxon | Aleekpalook N | Aerial parts | Forests | Cooked | 17 | 14 | 10 | 10 | Yes |
| Kamio Ka | |||||||||
| Brangalo Kh | |||||||||
| Kharposht Y | |||||||||
| Unidentified fungal taxon | Daidap Ka,Kh | Aerial parts | Orchards (grows on walnut tree trunks) | Cooked | 15 | 12 | - | - | - |
Ka: Kalasha people; Kh: Kho people; N: Kamkata-vari/Nuristani people; Y: Yidgha people; *: identifications based on plant description and local names only.
References
- Merçon, J.; Vetter, S.; Tengö, M.; Cocks, M.; Balvanera, P.; Rosell, J.A.; Ayala-Orozco, B. From local landscapes to international policy: Contributions of the biocultural paradigm to global sustainability. Glob. Sustain. 2019, 2, 1–11. [Google Scholar] [CrossRef]
- Wiley, A.S.; Cullin, J.M. What do anthropologists mean when they use the term biocultural? Am. Anthropol. 2016, 118, 554–569. [Google Scholar] [CrossRef]
- Maffi, L. Linguistic, Cultural, and Biological Diversity. Annu. Rev. Anthropol. 2005, 34, 599–617. [Google Scholar] [CrossRef]
- UNESCO-CBD. Joint Program between Biological and Cultural Diversity; UNESCO: Paris, France, 2010; Available online: http://www.unesco.org/new/en/natural-sciences/special-themes/biodiversity-initiative/biodiversity-culture/unesco-cbd-joint-programme/ (accessed on 2 November 2020).
- UNESCO-CBD. Florence Declaration on the Links between Biological and Cultural Diversity; UNESCO: Florence, Italy, 2014; Available online: https://www.landscapeunifi.it/en/florence-declaration-on-the-links-between-biological-and-cultural-diversity/ (accessed on 2 November 2020).
- Sterling, E.J.; Filardi, C.; Toomey, A.; Sigouin, A.; Betley, E.; Gazit, N.; Newell, J.; Albert, S.; Alvira, D.; Bergamini, N.; et al. Biocultural approaches to well-being and sustainability indicators across scales. Nat. Ecol. Evol. 2017, 1, 1798–1806. [Google Scholar] [CrossRef] [PubMed]
- Folke, C. Resilience: The emergence of a perspective for social-ecological systems analyses. Glob. Environ. Chang. 2006, 16, 253–267. [Google Scholar] [CrossRef]
- Hanspach, J.; Haide, L.J.; Oteros-Rozas, E.; Olafsson, A.S.; Gulsrud, N.M.; Raymond, C.M.; Albert, C.; Torralba, M.; Martín-López, B.; Bieling, C.; et al. Biocultural approaches to sustainability: A systematic review of the scientific literature. People Nat. 2020, 2, 643–659. [Google Scholar] [CrossRef]
- Argumedo, A.; Pimbert, M. ‘Protecting farmers’ rights with indigenous biocultural heritage territories: The experience of the potato park’, report. Int. Inst. Environ. Dev. 2008. Available online: http://pubs.iied.org/pdfs/G03072.pdf (accessed on 10 September 2020).
- Davidson-Hunt, I.J.; Turner, K.L.; Mead, A.T.P.; Cabrera-Lopez, J.; Bolton, R.; Idrobo, C.J.; Miretski, I.; Morrison, A.; Robson, J.P. Biocultural design: A new conceptual framework for sustainable development in rural indigenous and local communities. S.A.P.I.EN.S. 2012, 5. Available online: https://sapi-ens.revues.org/1382 (accessed on 2 November 2020).
- Gavin, M.C.; McCarter, J.; Mead, A.; Berkes, F.; Stepp, J.R.; Peterson, D.; Tang, R. Defining biocultural approaches to conservation. Trends Ecol. Evol. 2015, 30, 140–145. [Google Scholar] [CrossRef]
- Hunn, E. The value of subsistence for the future of the world. In Ethnoecology: Situated Knowledge/Located Lives; Nazarea, V., Ed.; University of Arizona Press: Tucson, Arizona, 1999; pp. 22–36. [Google Scholar]
- Maffi, L.; Woodley, E. Biocultural Diversity Conservation: A Global Sourcebook; Routledge: London, UK, 2012. [Google Scholar]
- Harmon, D. Losing species, losing languages: Connections between biological and linguistic diversity. Southwest J. Ling. 1996, 15, 89–108. [Google Scholar]
- Muhlhausler, P. The interdependence of linguistic and biological diversity. In The Politics of Multiculturalism in the Asia/Pacific; Myers, D., Ed.; Northern Territory University Press: Darwin, Australia, 1995; pp. 154–161. [Google Scholar]
- CBD. Convention on Biological Diversity; United Nations: Rio de Janeiro, Brazil; New York, NY, USA, 1992. [Google Scholar]
- Poole, A. Where is goal 18? The need for biocultural heritage in the sustainable development goals. Environ. Values 2018, 27, 55–80. [Google Scholar] [CrossRef]
- Pretty, J.; Adams, B.; Berkes, F.; de Athayde, S.; Dudley, N.; Hunn, E.; Maffi, L.; Milton, K.; Rapport, D.; Robbins, P.; et al. The Intersections of Biological Diversity and Cultural Diversity: Towards Integration. Conserv. Soc. 2009, 7, 100–112. [Google Scholar]
- Berkes, F.; Colding, J.; Folke, C. Rediscovery of traditional ecological knowledge as adaptive management. Ecol. Appl. 2000, 10, 1251–1262. [Google Scholar] [CrossRef]
- Berkes, F. Rethinking community-based conservation. Conserv. Biol. 2004, 18, 621–630. [Google Scholar] [CrossRef]
- Olsson, P.; Folke, C.; Berkes, F. Adaptive co-management for building resilience in social-ecological systems. Environ. Manag. 2004, 34, 75–90. [Google Scholar] [CrossRef]
- Ruiz-Mallén, I.; Corbera, E. Community-based conservation and traditional ecological knowledge: Implications for social-ecological resilience. Ecol. Soc. 2013, 18, 12. [Google Scholar] [CrossRef]
- Gómez-Baggethun, E.; Corbera, E.; Reyes-García, V. Traditional ecological knowledge and Global Environmental Change: Research findings and policy implications. Ecol. Soc. 2013, 184, 72. [Google Scholar] [CrossRef] [PubMed]
- UNESCO. Recommendation on the Safeguarding of Traditional Culture and Folklore: Adopted by the General Conference at Its Twenty-Fifth Session, Paris, 1989; UNESCO: Paris, France, 1989. [Google Scholar]
- Choudhry, F.R.; Khan, T.M.; Park, M.S.A.; Golden, K.J. Mental health conceptualization and resilience factors in the Kalasha youth: An indigenous ethnic and religious minority community in Pakistan. Front. Public Health 2018, 6, 187. [Google Scholar] [CrossRef]
- Israr-ud-Din. The people of Chitral: A survey of their ethnic diversity. Pak. Geo. Rev. 1969, 24, 45–57. [Google Scholar]
- Israr-ud-Din. Chitral—A historical sketch. J. Area Study Cent. Asia 1979, 3, 1–13. [Google Scholar]
- International Society of Ethnobiology (ISE). Code of Ethics. 2008. Available online: www.ethnobiology.net/whatwe-do/core-programs/ise-ethics-program/code-of-ethics/ (accessed on 1 September 2020).
- Termote, C.; Van Damme, P.; Djailo, B.D.A. Eating from the wild: Turumbu, Mbole and Bali traditional knowledge on non-cultivated edible plants, District Tshopo, DRCongo. Genet. Resour. Crop Evol. 2011, 58, 585–618. [Google Scholar] [CrossRef]
- Ali, S.I.; Qaiser, M. (Eds.) Flora of Pakistan; University of Karachi: Karachi, Pakistan, 1993–2009. [Google Scholar]
- Nasir, E.; Ali, S.I. (Eds.) Flora of Pakistan; No. 132–190; University of Karachi: Karachi, Pakistan, 1980–1989. [Google Scholar]
- Nasir, E.; Ali, S.I. (Eds.) Flora of Pakistan; No. 191–193; University of Karachi: Karachi, Pakistan, 1989–1992. [Google Scholar]
- Nasir, E.; Ali, S.I. (Eds.) Flora of West Pakistan; No. 1–131; University of Karachi: Karachi, Pakistan, 1970–1979. [Google Scholar]
- The Plant List. Version 1.1. 2013. Available online: http://www.theplantlist.org/ (accessed on 10 June 2020).
- Index Fungorum. 2020. Available online: http://www.indexfungorum.org/Names/Names.asp (accessed on 1 September 2020).
- Stevens, P.F. Angiosperm Phylogeny Website, Version 14. 2017. Available online: http://www.mobot.org/MOBOT/research/APweb (accessed on 10 June 2020).
- González-Tejero, M.R.; Casares-Porcel, M.; Sánchez-Rojas, C.P.; Ramiro-Gutiérrez, J.M.; Molero-Mesa, J.; Pieroni, A.; Giusti, M.E.; Censorii, E.; De Pasquale, C.; Della, A.; et al. Medicinal plants in the Mediterranean area: Synthesis of the results of the project Rubia. J. Ethnopharmacol. 2008, 116, 341–357. [Google Scholar] [CrossRef] [PubMed]
- Aziz, M.A.; Abbasi, A.M.; Ullah, Z.; Pieroni, A. Shared but Threatened: The Heritage of Wild Food Plant Gathering among Different Linguistic and Religious Groups in the Ishkoman and Yasin Valleys, North Pakistan. Foods 2020, 9, 601. [Google Scholar] [CrossRef]
- Abbas, W.; Hussain, W.; Hussain, W.; Badshah, L.; Hussain, K.; Pieroni, A. Traditional wild vegetables gathered by four religious groups in Kurram District, Khyber Pakhtunkhwa, North-West Pakistan. Genet. Resour. Crop Evol. 2020, 67, 1521–1536. [Google Scholar] [CrossRef]
- Shad, A.A.; Shah, H.U.; Bakht, J. Ethnobotanical assessment and nutritive potential of wild food plants. J. Anim. Plant Sci. 2013, 23, 92–99. [Google Scholar]
- Shah, A.H.; Khan, S.M.; Shah, A.H.; Mehmood, A.; Rahman, I.; Ahmad, H. Cultural uses of plants among Basikhel tribe of district Tor Ghar, Khyber Pakhtunkhwa, Pakistan. Pak. J. Bot. 2015, 47, 23–41. [Google Scholar]
- Ahmad, K.; Pieroni, A. Folk knowledge of wild food plants among the tribal communities of Thakht-e-Sulaiman Hills, North-West Pakistan. J. Ethnobiol. Ethnomed. 2016, 12, 7. [Google Scholar] [CrossRef] [PubMed]
- Tareen, N.M.; Rehman, M.A.; Shinwari, Z.K.; Bibi, T. Ethnomedicinal utilization of wild edible vegetables in district Harnai of Balochistan Province-Pakistan. Pak. J. Bot. 2016, 3, 1159–1171. [Google Scholar]
- Ahmad, K.; Weckerle, C.S.; Nazir, A. Ethnobotanical investigation of wild vegetables used among local communities in northwest Pakistan. Acta Soc. Bot. Pol. 2019, 88, 3616. [Google Scholar] [CrossRef]
- Abbasi, A.M.; Khan, M.A.; Shah, M.H.; Shah, M.M.; Pervez, A.; Ahmad, M. Ethnobotanical appraisal and cultural values of medicinally important wild edible vegetables of Lesser Himalayas-Pakistan. J. Ethnobiol. Ethnomed. 2013, 9, 66. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.P.Z.; Ahmad, M.; Zafar, M.; Sultana, S.; Ali, M.I.; Sun, H. Ethnomedicinal uses of edible wild fruits (EWFs) in Swat Valley, Northern Pakistan. J. Ethnopharmacol. 2015, 173, 191–203. [Google Scholar] [CrossRef]
- Luczaj, L.; Kujawska, M. Botanists and their childhood memories: An underutilized expert source in ethnobotanical research. Bot. J. Linn. Soc. 2012, 168, 334–343. [Google Scholar] [CrossRef]
- Pieroni, A.; Zahir, H.; Amin, H.I.M.; Sõukand, R. Where tulips and crocuses are popular food snacks: Kurdish traditional foraging reveals traces of mobile pastoralism in Southern Iraqi Kurdistan. J. Ethnobiol. Ethnomed. 2019, 15, 59. [Google Scholar] [CrossRef]
- Mattalia, G.; Sõukand, R.; Corvo, P.; Pieroni, A. Wild food thistle gathering and pastoralism: An inextricable link in the biocultural landscape of Barbagia, Central Sardinia (Italy). Sustainability 2020, 12, 5105. [Google Scholar] [CrossRef]
- Fernández-Giménez, M.E.; Estaque, F.F. Pyrenean pastoralists’ ecological knowledge: Documentation and application to natural resource management and adaptation. Hum. Ecol. 2012, 40, 287–300. [Google Scholar] [CrossRef]
- Ghimire, S.K.; Aumeeruddy-Thomas, Y. Ethnobotanical classification and plant nomenclature system of high altitude agro-pastoralists in Dolpo, Nepal. Bot. Orient. J. Plant Sci. 2009, 6, 56–68. [Google Scholar] [CrossRef]
- Oteros-Rozas, E.; Ontillera-Sánchez, R.; Sanosa, P.; Gómez-Baggethun, E.; Reyes-García, V.; González, J.A. Traditional ecological knowledge among transhumant pastoralists in Mediterranean Spain. Ecol. Sociol. 2013, 18, 33. [Google Scholar] [CrossRef]
- Tamou, C. Understanding Relations between Pastoralism and Its Changing Natural Environment. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2017. [Google Scholar]
- Pieroni, A.; Sõukand, R.; Amin, H.I.M.; Zahir, H.; Kukk, T. Celebrating multi-religious co-existence in Central Kurdistan: The bio-culturally diverse traditional gathering of wild vegetables among Yazidis, Assyrians, and Muslim Kurds. Hum. Ecol. 2018, 46, 217–227. [Google Scholar] [CrossRef]
- Sõukand, R.; Kalle, R. Perceiving the biodiversity of food at chest-height: Use of the fleshy fruits of wild trees and shrubs in Saaremaa, Estonia. Hum. Ecol. 2016, 44, 265–272. [Google Scholar] [CrossRef]
- Milner, S.E.; Brunton, N.P.; Jones, P.W.; O’Brien, N.M.; Collins, S.G.; Maguire, A.R. Bioactivities of glycoalkaloids and their aglycones from Solanum species. J. Agric. Food. Chem. 2011, 59, 3454–3484. [Google Scholar] [CrossRef]
- Carle, R. Investigations on the content of steroidal alkaloids and sapogenins within Solanum sect. Solanum (¼ sect. Morella) (Solanaceae). Plant Syst. Evol. 1981, 138, 61–71. [Google Scholar] [CrossRef]
- Menendez-Baceta, G.; Aceituno-Mata, L.; Tardío, J.; Reyes-García, V.; Pardo-de-Santayana, M. Wild edible plants traditionally gathered in Gorbeialdea (Biscay, Basque Country). Genet. Resour. Crop Evol. 2012, 59, 1329–1347. [Google Scholar] [CrossRef]
- Pawera, L.; Verner, V.; Termote, C.; Sodombekov, I.; Kandakov, A.; Karabaev, N.; Skalicky, M.; Polesny, Z. Medical ethno-botany of herbal practitioners in the Turkestan range, south-western Kyrgyzstan. Acta Soc. Bot. Pol. 2016, 85, 3483. [Google Scholar] [CrossRef]
- Nordeide, M.B.; Hatloy, A.; Folling, M.; Lied, E.; Oshaug, A. Nutrient composition and nutritional importance of green leaves and wild food resources in an agricultural district, Koutiala, in southern Mali. Int. J. Food Sci. Nutr. 1996, 47, 455–468. [Google Scholar] [CrossRef]
- Pimentel, D.; McNair, M.; Buck, L.; Pimentel, M.; Kamil, J. The value of forests to world food security. Hum. Ecol. 1997, 25, 91–120. [Google Scholar] [CrossRef]
- Sundriyal, M.; Sundriyal, D.C. Wild edible plants of the Sikkim Himalaya: Nutritive values of selected species. J. Econ. Bot. 2001, 55, 377–390. [Google Scholar] [CrossRef]
- Abbasi, A.M.; Khan, M.A.; Zafar, M. Ethno-medicinal assessment of some selected wild edible fruits and vegetables of Lesser-Himalayas, Pakistan. Pak. J. Bot. 2013, 45, 215–222. [Google Scholar]
- Luczaj, Ł.; Končić, M.Z.; Miličević, T.; Dolina, K.; Pandža, M. Wild vegetable mixes sold in the markets of Dalmatia (southern Croatia). J. Ethnobiol. Ethnomed. 2013, 9, 2. [Google Scholar] [CrossRef] [PubMed]
- Konsam, S.; Thongam, B.; Handique, A.K. Assessment of wild leafy vegetables traditionally consumed by the ethnic communities of Manipur, northeast India. J. Ethnobiol. Ethnomed. 2016, 12, 9. [Google Scholar] [CrossRef] [PubMed]
- Kalle, R.; Sõukand, R. Current and remembered past uses of wild food plants in Saaremaa, Estonia: Changes in the context of unlearning debt. Econ. Bot. 2016, 70, 235–253. [Google Scholar] [CrossRef]
- Pieroni, A.; Hovsepyan, R.; Manduzai, A.K.; Sõukand, R. Wild food plants traditionally gathered in central Armenia: Archaic ingredients or future sustainable foods? Environ. Dev. Sustain. 2020. [Google Scholar] [CrossRef]
- Pieroni, A.; Sõukand, R. Ethnic and religious affiliations affect traditional wild plant foraging in Central Azerbaijan. Genet. Resour. Crop. Evol. 2019, 66, 1495–1513. [Google Scholar] [CrossRef]
- Sõukand, R.; Pieroni, A. Resilience in the mountains: Biocultural refugia of wild food in the Greater Caucasus Range, Azerbaijan. Biodivers. Conserv. 2019, 28, 3529–3545. [Google Scholar] [CrossRef]
- Biddulph, J. Tribes of the Hindoo Koosh; Government Printing Office: Calcutta, India, 1880; Reprinted in Ali Kamran Publishers: Lahore, Pakistan, 1986.
- McCarter, J.; Gavin, M.C. Perceptions of the value of traditional ecological knowledge to formal school curricula: Opportunities and challenges from Malekula Island, Vanuatu. J. Ethnobiol. Ethnomed. 2011, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Skutnabb-Kangas, T. Linguistic Genocide in Education—Or Worldwide Diversity and Human Rights? Routledge: New York, NY, USA, 2000. [Google Scholar]
- Morgenstierne, G. Indo-Iranian Frontier Languages, Volume II, Iranian Pamir Languages; Instituttet for Sammenlignende Kultur Forskning: Oslo, Norway, 1938. [Google Scholar]
- Marshall, G. A Dictionary of Sociology; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- Daniel, M.; Linder, G.F. “Marginal People.” Encyclopedia of Public Health; Breslow, L., Ed.; Gale Group, Inc.: Farmington Hills, MI, USA, 2002. [Google Scholar]
- Alexander, G.L.; Kinman, E.L.; Miller, L.C.; Patrick, T.B. Marginalization and health geomatics. J. Biomed. Inf. 2003, 36, 400–407. [Google Scholar] [CrossRef][Green Version]
- Cleary, M.; Horsfall, J.; Escott, P. Marginalization and associated concepts and processes in relation to mental health/illness. Issues Ment. Health Nurs. 2014, 35, 224–226. [Google Scholar] [CrossRef]
- Lynam, M.J.; Cowley, S. Understanding marginalization as a social determinant of health. Crit. Public Health 2007, 17, 137–149. [Google Scholar] [CrossRef]
- Kannan, D. The Psychological Costs of Marginalization: Being Seen as Less Than Has a Long-Standing Impact on the Health of An Individual. Available online: https://www.whiteswanfoundation.org/mental-health-matters/society-and-mental-health/the-psychological-costs-of-marginalization (accessed on 1 September 2020).
- Schiefer, D.; van der Noll, J. The Essentials of Social Cohesion: A Literature Review. Soc. Indic. Res. 2017, 132, 579–603. [Google Scholar] [CrossRef]
- WCED. Our Common Future. World Commission on Environment and Development; Oxford University Press: New York, NY, USA, 1987. [Google Scholar]
- Tiwari, P.C.; Joshi, B. Natural and socio-economic factors affecting food security in the Himalayas. Food Secur. 2012, 4, 195–207. [Google Scholar] [CrossRef]
- FAO. The State of Food Insecurity in the World; FAO: Rome, Italy, 2009. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).