Predicting Mushroom Productivity from Long-Term Field-Data Series in Mediterranean Pinus pinaster Ait. Forests in the Context of Climate Change
Abstract
1. Introduction
2. Materials and Methods
2.1. Data
2.2. Statistical Analysis
2.3. Simulation Scenarios
3. Results
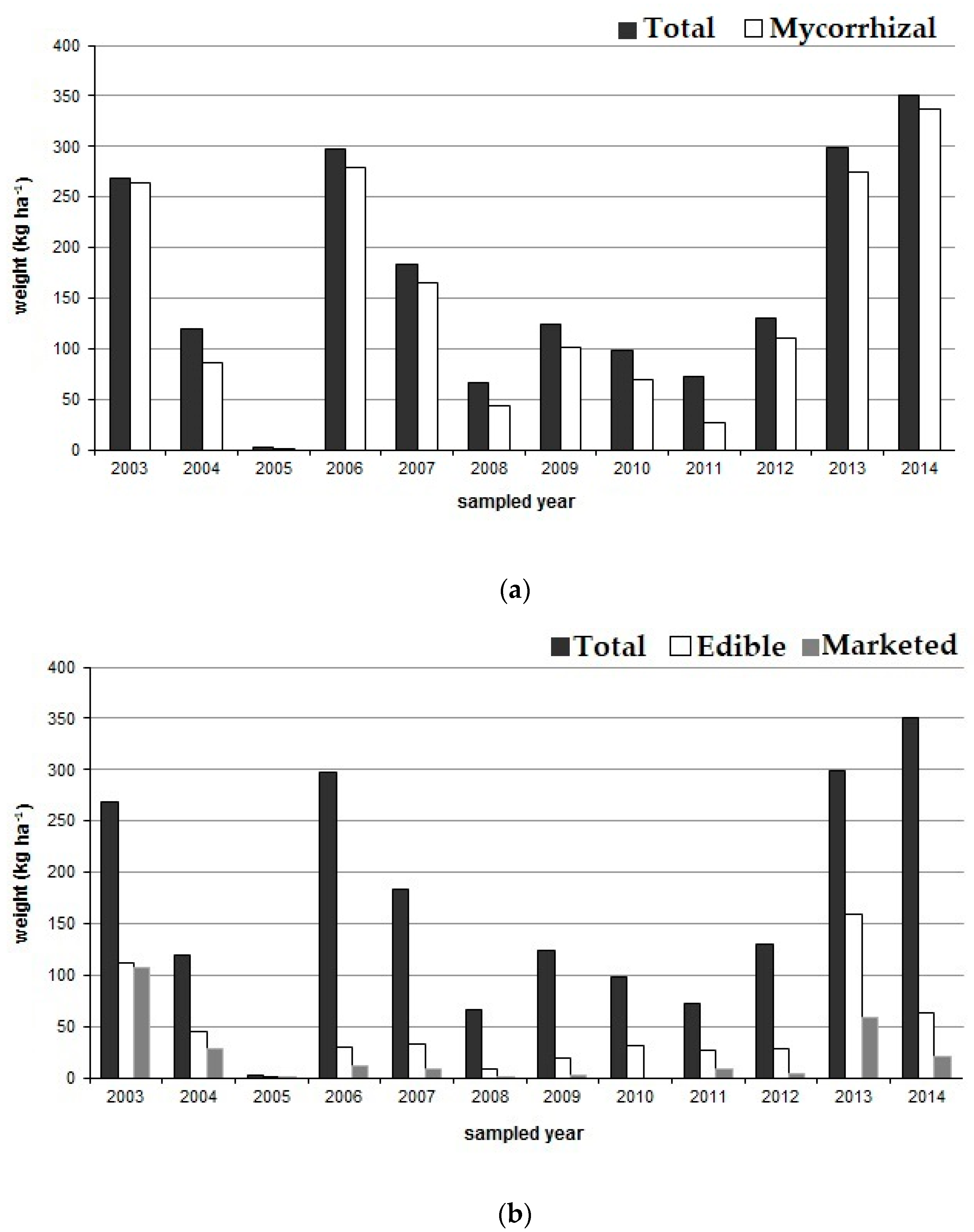
3.1. Mushroom Productivity
3.2. Mushroom Productivity Model
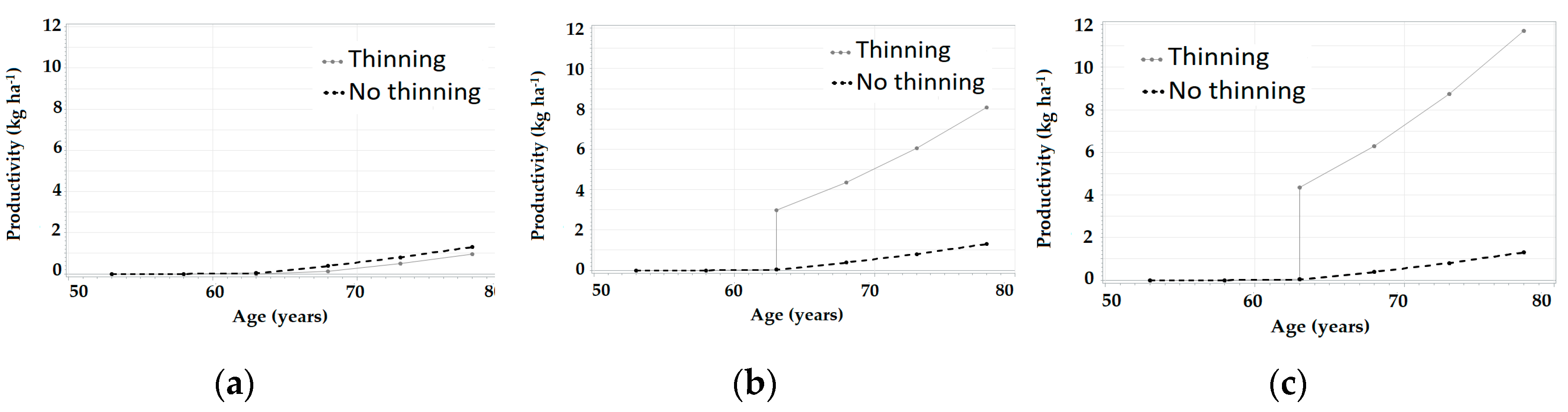
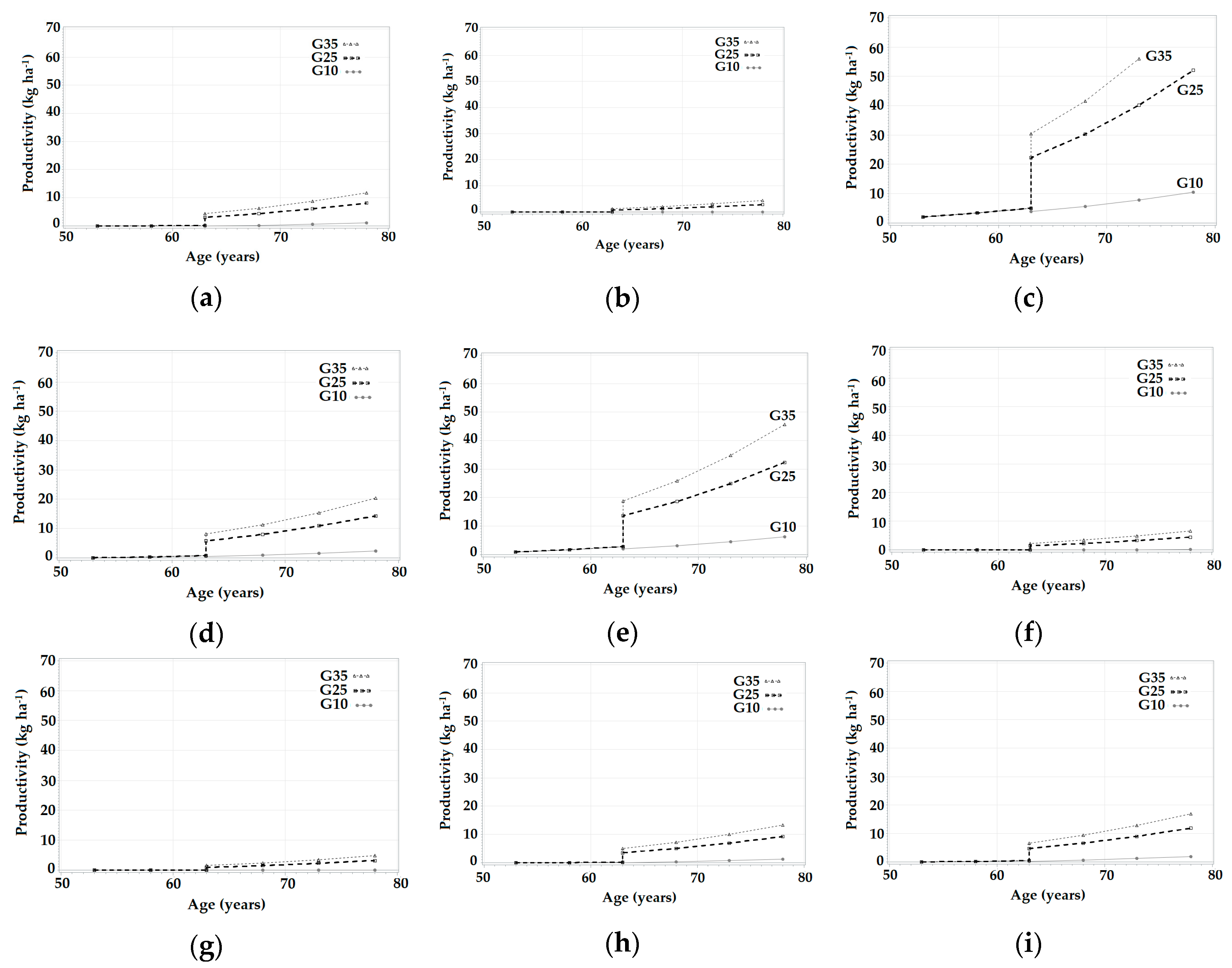
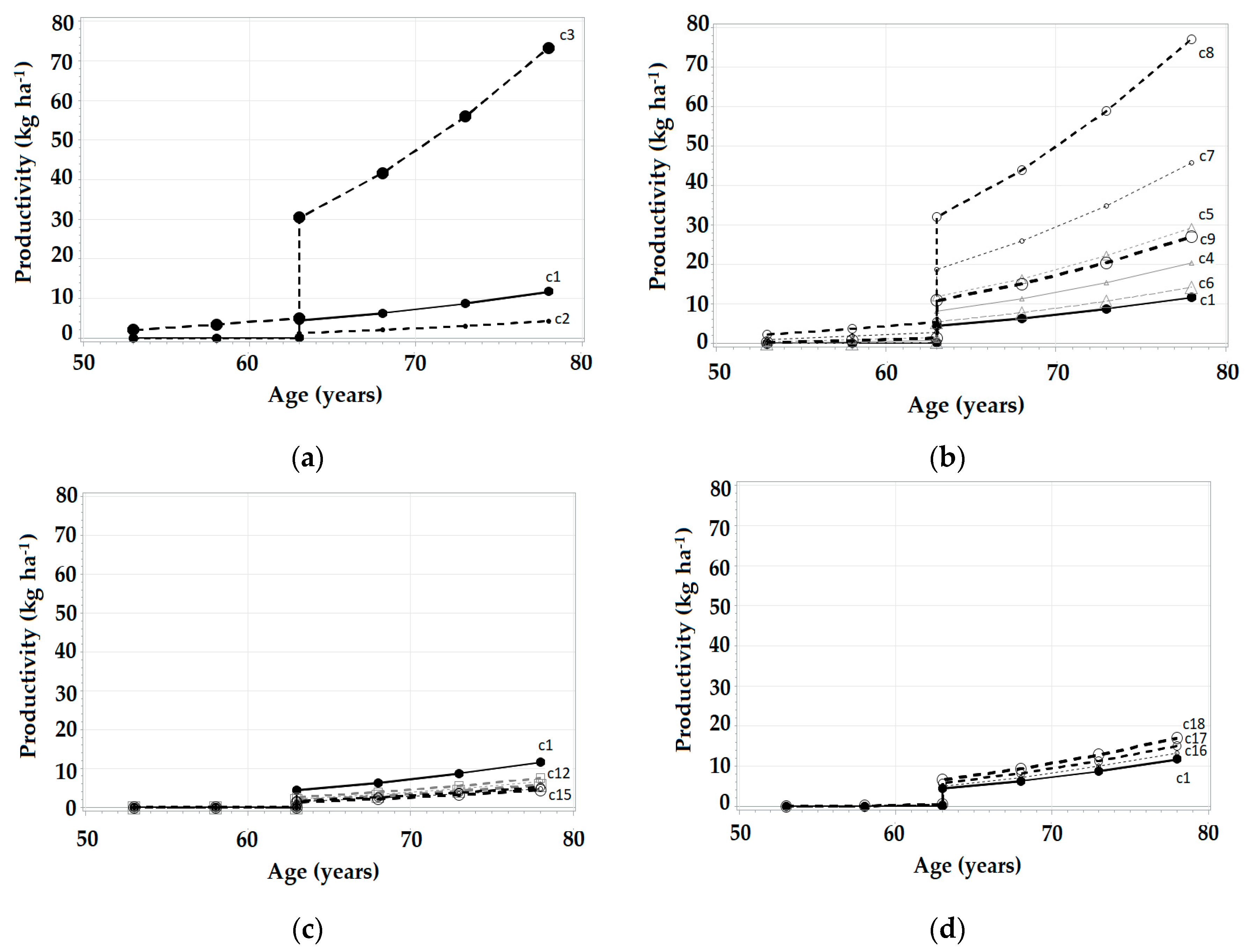
3.3. Simulations in SiManFor Platform
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Luoma, D.L.; Eberhart, J.L.; Molina, R.; Amaranthus, M.P. Response of ectomycorrhizal fungus sporocarp production to varying levels and patterns of green-tree retention. For. Ecol. Manag. 2004, 202, 337–354. [Google Scholar] [CrossRef]
- Brundrett, M.C. Mycorrhizal associations and other means of nutrition of vascular plants: Understanding the global diversity of host plants by resolving conflicting information and developing reliable means of diagnosis. Plant Soil 2009, 320, 37–77. [Google Scholar] [CrossRef]
- Sardans, J.; Peñuelas, J. Plant–soil interactions in Mediterranean forest and shrublands: Impacts of climatic change. Plant Soil 2013, 365, 1–33. [Google Scholar] [CrossRef]
- Clemmensen, K.E.; Bahr, A.; Ovaskainen, O.; Dahlberg, A.; Ekblad, A.; Wallander, H.; Stenlid, J.; Finlay, R.D.; Wardle, D.A.; Lindahl, B.D. Roots and associated fungi drive long-term carbon sequestration in boreal forest. Science 2013, 339, 1615–1618. [Google Scholar] [CrossRef] [PubMed]
- Querejeta, J.I. Chapter 17—Soil water retention and availability as influenced by mycorrhizal symbiosis: Consequences for individual plants, communities, and ecosystems. In Mycorrhizal Mediation of Soil; Johnson, N.C., Gehring, C., Jansa, J., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 299–317. [Google Scholar]
- Duchesne, L.C.; Ellis, B.E.; Peterson, R.L. Disease suppression and antibiosis by the ectomycorrhizal fungus Paxilus involutus. New Phytol. 1989, 111, 693–698. [Google Scholar] [CrossRef]
- Eastwood, D.C.; Floudas, D.; Binder, M.; Majcherczyk, A.; Schneider, P.; Aerts, A.; Asiegbu, F.O.; Baker, S.E.; Barry, K.; Bendiksby, M.; et al. The plant cell walledecomposing machinery underlies the functional diversity of forest fungi. Science 2011, 333, 762–765. [Google Scholar] [CrossRef] [PubMed]
- Alexander, S.J.; Pilz, D.; Weber, N.S.; Brown, E.; Rockwell, V.A. Mushrooms, trees and money. Value estimates of commercial mushrooms and timber in the Pacific Northwest. Environ. Manag. 2002, 30, 129–141. [Google Scholar] [CrossRef] [PubMed]
- Palahí, M.; Pukkala, T.; Bonet, J.A.; Colinas, C.; Fischer, C.R.; de Aragón, J.M. Effect of the inclusion of mushroom values on the optimal management of even-aged pine stands of Catalonia. For. Sci. 2009, 55, 503–511. [Google Scholar]
- Castaño, C.; Alday, J.G.; Lindahl, B.D.; de Aragón, J.M.; de Miguel, S.; Colinas, C.; Parladé, J.; Pera, J.; Bonet, J.A. Lack of thinning effects over inter-annual changes in soil fungal community and diversity in a Mediterranean pine forests. For. Ecol. Manag. 2018, 424, 420–427. [Google Scholar] [CrossRef]
- Alday, J.G.; de Aragón, J.M.; de Miguel, S.; Bonet, J.A. Mushroom biomass and diversity are driven by different spatio-temporal scales along Mediterranean: Elevation gradients. Sci. Rep. 2017, 7, 45824. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.E.; Molina, R.; Huso, M.M.P.; Luoma, D.L.; Mckay, D.; Castellano, M.A.; Lebel, T.; Valachovic, I. Species richness, abundance and composition of hypogeous and epigeous ectomucorrhizal fungal sporocarps in Young, rotation age and old-growth stand of Douglas fir (Ptseudotsuga menziensii) in the Cascade Range of Oregon, USA. Can. J. Bot. 2002, 80, 186–204. [Google Scholar] [CrossRef]
- Bonet, J.A.; Pukkala, T.; Fischer, C.R.; Palahí, M.; de Aragón, J.M.; Colinas, C. Empirical models for predicting the production of wild mushrooms in Scots pine (Pinus sylvestris L.) forests in the Central Pyrenees. Ann. For. Sci 2008, 65, 206–215. [Google Scholar] [CrossRef]
- Collado, E.; Camarero, J.J.; de Aragón, J.M.; Pemán, J.; Bonet, J.A.; de Miguel, S. Linking fungal dynamics, tree growth and forest management in a Mediterranean pine ecosystem. For. Ecol. Manag. 2018, 422, 223–232. [Google Scholar] [CrossRef]
- Primicia, I.; Camarero, J.J.; de Aragón, J.M.; de Miguel, S.; Bonet, J.A. Linkages between climate, seasonal wood formation and mycorrhizalmushroom yields. Agric. For. Meteorol. 2016, 228–229, 339–348. [Google Scholar] [CrossRef]
- Castaño, C.; Lindahl, B.D.; Alday, J.G.; Hagenbo, A.; de Aragón, J.M.; Parladé, J.; Pera, J.; Bonet, J.A. Soil microclimate changes affect soil fungal communities in a Mediterranean pine forest. New Phytol. 2018, 220, 1211–1221. [Google Scholar] [CrossRef] [PubMed]
- Karavani, A.; De Cáceres, M.; de Aragón, J.M.; Bonet, J.A.; de Miguel, S. Effect of climatic and soil moisture conditions on mushroom productivity and related ecosystem services in Mediterranean pine stands facing climate change. Agric. For. Meteorol. 2018, 248, 432–440. [Google Scholar] [CrossRef]
- Hernández-Rodríguez, M.; de Miguel, S.; Pukkala, T.; de Rueda, J.A.O.; Martín-Pinto, P. Climate-sensitive models for mushroom yields and diversity in Cistus ladanifer scrublands. Agric. For. Meteorol. 2015, 213, 173–182. [Google Scholar] [CrossRef]
- Alday, J.G.; Bonet, J.A.; de Rueda, J.A.O.; de Aragón, J.M.; Martín-Pinto, P.; de Miguel, S.; Hernández-Rodríguez, M.; Martínez-Peña, F. Record breaking mushroom yields in Spain. Fungal Ecol. 2017, 26, 144–146. [Google Scholar] [CrossRef]
- Vogt, K.A.; Bloomfield, J.; Ammirati, J.F.; Ammirati, S.R. Sporocarp production by basidiomycetes, with emphasis on forest ecosystems. In The Fungal Community. Its Organization and Role in the Ecosystem, 2nd ed.; Dekker, M., Ed.; Marcel Dekker: New York, NY, USA, 1992; pp. 563–581. [Google Scholar]
- De Aragón, J.M.; Bonet, J.A.; Fischer, C.R.; Colinas, C. Productivity of ectomycorrhizal and selected edible saprotrophic fungi in pine forests of the pre-Pyrenees mountains, Spain: Predictive equations for forest management of mycolocial resources. For. Ecol. Manag. 2007, 252, 239–256. [Google Scholar] [CrossRef]
- Vasquez Gassibe, P.; Fraile, R.; Hernández-Rodríguez, M.; de Rueda, J.A.O.; Bravo, F.; Martín-Pinto, P. Post-fire production of mushrooms in Pinus pinaster forests using classificatory models. J. For. Res. 2014, 19, 348–356. [Google Scholar] [CrossRef]
- Dahl, F.A.; Galteland, T.; Gjelsvik, R. Statistical modelling of wildwood mushroom abundance. Scand. J. For. Res. 2008, 23, 224–249. [Google Scholar] [CrossRef]
- Martínez-Peña, F.; de Miguel, S.; Pukkala, T.; Bonet, J.A.; Ortega-Martínez, P.; Aldea, J.; de Aragón, J.M. Yield models for ectomycorrhizal mushrooms in Pinus sylvestris forests with special focus on Boletus edulis and Lactarius group deliciosus. For. Ecol. Manag. 2012, 282, 63–69. [Google Scholar] [CrossRef]
- Bonet, J.A.; Palahí, M.; Colinas, C.; Pukkala, T.; Fischer, C.R.; Miina, J.; de Aragón, J.M. Modelling the production and species richness of wild mushrooms in pine forests of Central Pyrenees in north-eastern Spain. Can. J. For. Res. 2010, 40, 347–356. [Google Scholar] [CrossRef]
- Tahvanainen, V.; Miina, J.; Kurttila, M.; Salo, K. Modelling the yields of marketed mushrooms in Picea abies stands in eastern Finland. For. Ecol. Manag. 2016, 362, 79–88. [Google Scholar] [CrossRef]
- De Miguel, S.; Bonet, J.A.; Pukkala, T.; de Aragón, J.M. Impact of forest management intensity on landscape-level mushroom productivity: A regional model-based scenario analysis. For. Ecol. Manag. 2014, 330, 218–227. [Google Scholar] [CrossRef]
- Taye, Z.M.; Martínez-Peña, F.; Bonet, J.A.; de Aragón, J.M.; de Miguel, S. Meteorological conditions and site characteristics driving edible mushroom production in Pinus pinaster forests of Central Spain. Fungal Ecol. 2016, 23, 30–41. [Google Scholar] [CrossRef]
- Fox, J.C.; Ades, P.K.; Bi, H. Stochastic structure and individual-tree growth models. For. Ecol. Manag. 2001, 154, 261–276. [Google Scholar] [CrossRef]
- Bravo, F.; Rodríguez, F.; Ordóñez, A.C. A web-based application to simulate alternatives for sustainable forest management: SIMANFOR. For. Syst. 2012, 21, 4–8. [Google Scholar] [CrossRef]
- Agreda, T.; Águeda, B.; Olano, J.M.; Vicente-Serrano, S.M.; Fernández-Toirán, M. Increased evapotranspiration demand in a Mediterranean climate might cause a decline in fungal yields under global warming. Glob. Chang. Biol. 2015, 21, 3499–3510. [Google Scholar] [CrossRef] [PubMed]
- Büntgen, U.; Egli, S.; Galván, J.D.; Diez, J.M.; Aldea, J.; Latorre, J.; Martínez-Peña, F. Drought-induced changes in the phenology, productivity and diversity of Spanish fungi. Fungal Ecol. 2015, 16, 6–18. [Google Scholar] [CrossRef]
- Vásquez, P.; de Rueda, J.O.; Martín-Pinto, P.P. pinaster under extreme ecological conditions provides high fungal production and diversity. For. Ecol. Manag. 2015, 337, 161–173. [Google Scholar] [CrossRef]
- Ohenoja, E.; Koistinen, R. Fruit body production of larger fungi in Finland. 2. Edible fungi in northern Finland 1976–1978. Ann. Bot. Fenn. 1984, 21, 357–366. [Google Scholar]
- Baptista, P.; Martins, A.; Tavares, R.M.; Lino-Neto, T. Diversity and fruiting pattern of macrofungi associated with chestnut (Castanea sativa) in the Trás-os-Montes region (Northeast Portugal). Fungal Ecol. 2010, 3, 9–19. [Google Scholar] [CrossRef]
- Moser, M.; Plant, S.; Kibby, G. Keys to Agarics and Boleti: Polyporales, Boletales, Agaricales, Russulales; Roger Phillips: London, UK, 1983; pp. 1–535. [Google Scholar]
- Breitenbach, J. Champignons de Suisse Tome 1. Les Ascomycètes; Mykologia: Lucerne, Switzerland, 1984; pp. 1–310. [Google Scholar]
- Breitenbach, J. Champignons de Suisse Tome 2. Champignons sans Lames; Mykologia: Lucerne, Switzerland, 1986; pp. 1–412. [Google Scholar]
- Breitenbach, J. Champignons de Suisse Tome 3. Bolets et Champignons à Lames: Première Partie; Mykologia: Lucerne, Switzerland, 1991; pp. 1–364. [Google Scholar]
- Breitenbach, J. Champignons de Suisse Tome 4. Champignons à lames. Deuxième Partie; Mykologia: Lucerne, Switzerland, 1995; pp. 1–371. [Google Scholar]
- Breitenbach, J. Champignons de Suisse Tome 5. Champignons à lames. Troisième Partie; Mykologia: Lucerne, Switzerland, 2000; pp. 1–340. [Google Scholar]
- Breitenbach, J. Champignons de Suisse Tome 6. Russulaceae. Actaires. Russules; Mykologia: Lucerne, Switzerland, 2005; pp. 1–263. [Google Scholar]
- Bon, M. Guía de Campo de los Hongos de Europa; Omega: Barcelona, Spain, 1987; pp. 1–352. [Google Scholar]
- Knudsen, H.; Vesterholt, J. Funga Nordica: Agaricoid, Boletoid and Cyphelloid Genera; Nordsvamp: Copenhagen, Denmark, 2008; pp. 1–965. [Google Scholar]
- Bonet, J.; Fischer, C.; Colinas, C. The relationship between forest age and aspect on the production of sporocarps of ectomycorrhizal fungi in Pinus sylvestris forest of the central Pyrenees. For. Ecol. Manag. 2004, 203, 157–175. [Google Scholar] [CrossRef]
- Martín-Pinto, P.; Pajares, J.; Diez, J. In vitro effects of four ectomycorrhizal fungi, Boletus edulis, Rhizopogon roseolus, Laccaria laccata and Lactarius deliciosus on Fusarium damping off in Pinus nigra seedlings. New For. 2006, 32, 323–334. [Google Scholar] [CrossRef]
- Meyer, H.A. A mathematical expression for height curves. J. For. 1940, 38, 415–420. [Google Scholar]
- Regent Instruments Inc. WinDENDROtm; Regent Instruments Inc.: Québec, QC, Canada, 2003. [Google Scholar]
- Ruiz-Peinado, R.; Río, M.; Montero, G. New models for estimating the carbon sink capacity of Spanish softwood species. For. Syst. 2011, 20, 176–188. [Google Scholar] [CrossRef]
- Rodríguez, F.; Broto, M.; Lizarralde, I. CubiFor: Herramienta para cubicar, clasificar productos y calcular biomasa y CO2 en masas forestales de Castilla y León. Rev. Montes 2008, 95, 33–39. [Google Scholar]
- De la Varga, H.; Agueda, B.; Martínez-Pena, F.; Parlade, J.; Pera, J. Quantification of extraradical soil mycelium and ectomycorrhizas of Boletus. For. Ecol. Manag. 2015, 337, 161–173. [Google Scholar] [CrossRef]
- ISSS-ISRIC-FAO. World Reference Base for Soil Resources. Draft; Wageningen: Rome, Italy, 1994; pp. 1–133. [Google Scholar]
- MAPA. Métodos Oficiales de Análisis. Tomo III; Ministerio de Agricultura, Pesca y Alimentación: Madrid, Spain, 1994; pp. 1–662. [Google Scholar]
- Nicholson, G. Methods of Soil, Plant and Water Analysis; New Zealand Forest Research Institute: Rotorua, New Zealand, 1984; pp. 1–24. [Google Scholar]
- Benton, J.J.; Wolf, J.B.; Mills, H.A. Plant Analysis Handbook, a Practical Sampling, Preparation, Analysis and Interpretation Guide; Micro-Macro Publishing: Athens, GA, USA, 1991; pp. 1–213. [Google Scholar]
- Sas Institute Inc. SAS/Stattm User’s Guide, Relase 9.4; Sas Institute Inc.: Cary, NC, USA, 2016; pp. 1–550. [Google Scholar]
- Baskerville, G.L. Use of logarithmic regression in the estimation of plant biomass. Can. J. For. Res. 1972, 2, 49–53. [Google Scholar] [CrossRef]
- Huang, S.; Yang, Y.; Wang, Y. A critical look at procedures for validating growth and yield models. In Modelling Forest Systems; Amaro, A., Reed, D., Soares, P., Eds.; CAB International: Wallingford, UK, 2003; pp. 271–293. [Google Scholar]
- Rio, M.; López, E.; Montero, G. Manual de Gestión para Masas Procedentes de Repoblación de Pinus pinaster Ait, Pinus sylvestris L. y Pinus nigra Arn. en Castilla y León; Consejería de Medio Ambiente, Junta de Castilla y León: Valladolid, Spain, 2006; pp. 1–102. [Google Scholar]
- Bravo, F.; Herrero, C.; Ordóñez, A.C.; De La Parra, B.; Cuesta, J.; Santos, M.; Ruano, I.; Manso, R. Instalación de Ensayos de Gestión Forestal Adaptativa en Distintos Tipos de Pinares en el Bosque Modelo de Palencia; Consejería de Medio Ambiente, Junta de Castilla y León: Valladolid, Spain, 2015; pp. 1–15. [Google Scholar]
- O’Dell, T.E.; Ammirati, J.F.; Schreiner, E.G. Species richness and abundance of ectomycorrhizal basidiomycete sporocarps on a moisture gradient in the Tsuga heterophylla zone. Can. J. Bot. 1999, 77, 1699–1711. [Google Scholar] [CrossRef]
- Bonet, J.A.; de-Miguel, S.; de Aragón, J.M.; Pukkala, T.; Palahí, M. Immediate effect of thinning on the yield of Lactarius group deliciosus in Pinus pinaster forests in North-Eastern Spain. For. Ecol. Manag. 2012, 265, 211–217. [Google Scholar] [CrossRef]
- Straatsma, G.; Ayer, F.; Egli, S. Species richness, abundance and phenology of fungal fruit bodies over 21 years in a Swiss forest plot. Mycol. Res. 2001, 105, 515–523. [Google Scholar] [CrossRef]
- Straatsma, G.; Krisai-Greilhuber, I. Assemblage structure, species richness, abundance, and distribution of fungal fruit bodies in a seven year plot-based survey near Vienna. Mycol. Res. 2003, 107, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Krebs, C.J.; Carrier, P.; Boutin, S.; Boonstra, R.; Hofer, E. Mushroom crops in relation to weather in southeastern Yukon. Botany 2008, 86, 1497–1502. [Google Scholar] [CrossRef]
- Berraondo, I.; Herrero, C.; de la Parra, B.; Olaizola, J.; Pando, V.; Oria de Rueda, J.A. Modelización de la producción de Tricholoma portentosum (Fr.) Quél. en masas de Pinus sylvestris L. In Proceedings of the V Congreso Forestal Español, Ávila, Spain, 21–25 September 2009. [Google Scholar]
- Martínez-Peña, F. Producción y Aprovechamiento de Boletus edulis Bull.: Fr. en un Bosque de Pinus sylvestris L.; Consejería de Medio Ambiente. Junta de Castila y León: Valladolid, Spain, 2003; pp. 1–134. [Google Scholar]
- Kauserud, C.L.; Stige, J.O.; Vik, J.O.; Okland, R.H.; Hoiland, K.; Stenseth, N.C. Mushroom fruiting and climate change. Proc. Natl. Acad. Sci. USA 2008, 105, 3811–3814. [Google Scholar] [CrossRef] [PubMed]
- Shaw, P.J.A.; Kibby, C.; Mayes, J. Effects of thinning treatment on an ectomycorrhizal succession under Scots pine. Mycol. Res. 2003, 107, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Amaranthus, M.P.; Page-Dumroese, D.; Harvey, A.; Cazares, E.; Bednar, L.F. Soil Compaction and Organic Matter Affect Conifer Seedling Nonmycorrhizal and Ectomycorrhizal Root Tip Abundance and Diversity; Research Paper PNW-RP-494; USDA Forest Service: Portland, OR, USA, 1996; pp. 1–12. [Google Scholar]
- Kropp, B.R.; Albee, S. The effects of silvicultural treatments on occurrence of mycorrhizal sporocarps in a Pinus contorta forest: A preliminary survey. Biol. Conserv. 1996, 78, 313–318. [Google Scholar] [CrossRef]
- Pilz, D.; Molina, R.; Mayo, J. Effects of thinning young forests on Chanterelle mushroom production. J. For. 2006, 104, 9–14. [Google Scholar] [CrossRef]
- Tomao, A.; Bonet, J.A.; de Aragón, J.M.; de Miguel, S. Is silviculture able to enhance wild forest mushroom resources? Current knowledge and future perspectives. For. Ecol. Manag. 2017, 402, 102–114. [Google Scholar] [CrossRef]
- Salerni, E.; Perini, C. Experimental study for increasing productivity of Boletus edulis s.l. in Italy. For. Ecol. Manag. 2004, 201, 161–170. [Google Scholar] [CrossRef]
- Nogués Bravo, D.; Araújo, M.B.; Lasanta, T.; López Moreno, J.L. Climate change in Mediterranean mountains during the 21st century. Ambio 2008, 37, 280–285. [Google Scholar] [CrossRef]
- IPCC. Summary for Policymakers. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. In Climate Change 2013: The Physical Science Basis; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK, 2013; pp. 1–1535. [Google Scholar]
| Plot | Stand | Site | Soil | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BA (m2 ha−1) | N (trees ha−1) | QMD (cm) | Hdom (m) | V (m3 ha−1) | Broot (mg ha−1) | Alt. | S (%) | Exp. | pH | SOM (%) | C/N | AP | MT | |
| 1 | 52.9 | 408 | 40.6 | 19.1 | 1.082 | 0.1438 | 985 | 0.1 | SE | 5.2 | 5.28 | 25.09 | 573 | 10.1 |
| 2 | 40.3 | 333 | 39.2 | 18.5 | 0.987 | 0.1321 | 985 | 0.1 | SE | 5.4 | 4.86 | 28.88 | 573 | 10.1 |
| 3 | 45.3 | 379 | 39.0 | 18.2 | 0.971 | 0.1302 | 985 | 0.1 | SE | 5.1 | 4.39 | 22.02 | 573 | 10.1 |
| 4 | 12.1 | 133 | 33.9 | 19.8 | 0.788 | 0.1037 | 700 | 0.011 | N | 6.6 | 0.53 | 11.34 | 455 | 11.8 |
| 5 | 14.8 | 183 | 32.0 | 20.0 | 0.682 | 0.0908 | 700 | 0.011 | N | 6.5 | 0.68 | 19.57 | 455 | 11.8 |
| 6 | 10.5 | 146 | 30.3 | 18.1 | 0.564 | 0.0772 | 700 | 0.011 | N | 6.0 | 0.80 | 14.83 | 455 | 11.8 |
| 7 | 18.6 | 129 | 42.8 | 18.5 | 1.234 | 0.1624 | 890 | 0.01 | N | 6.6 | 0.82 | 18.60 | 429 | 12.1 |
| 8 | 18.1 | 146 | 39.8 | 18.5 | 1.029 | 0.1372 | 890 | 0.01 | N | 6.6 | 1.67 | 23.55 | 429 | 12.1 |
| 9 | 21.4 | 183 | 38.5 | 18.4 | 0.954 | 0.1278 | 890 | 0.01 | N | 6.6 | 1.24 | 19.30 | 429 | 12.1 |
| Plot | fw (kg ha−1) | dw (kg ha−1) | ns ha−1 | fw (kg ha−1) | dw (kg ha−1) | ns ha−1 | fw (kg ha−1) | dw (kg ha−1) | ns ha−1 |
|---|---|---|---|---|---|---|---|---|---|
| 2003 | 2004 | 2005 | |||||||
| 1 | 241.5 | 17.6 | 15,000 | 73.4 | 10.4 | 12,000 | 2.9 | 0.3 | 4000 |
| 2 | 392.7 | 27.3 | 15,200 | 101.7 | 24.8 | 12,600 | 0.8 | 0.0 | 1600 |
| 3 | 169.7 | 11.1 | 11,200 | 181.5 | 21.1 | 10,000 | 3.1 | 0.2 | 2900 |
| 2006 | 2007 | 2008 | |||||||
| 1 | 444.8 | 30.3 | 30,400 | - | - | - | 13.4 | 1.5 | 80,800 |
| 2 | 436.9 | 27.5 | 24,800 | - | - | - | 22.6 | 2.4 | 63,000 |
| 3 | 729.0 | 59.9 | 32,900 | - | - | - | 26.9 | 2.8 | 75,500 |
| 4 | 17.0 | 1.9 | 22,800 | 62.1 | 9.5 | 30,800 | 44.1 | 4.5 | 66,500 |
| 5 | 6.8 | 0.9 | 10,600 | 4.8 | 0.7 | 7800 | 5.0 | 0.6 | 17,200 |
| 6 | 166.4 | 17.8 | 20,700 | 118.7 | 18.3 | 27,100 | 120.4 | 10.2 | 40,700 |
| 7 | 391.9 | 38.8 | 50,900 | 454.7 | 79.4 | 112,900 | 201.1 | 17.7 | 88,100 |
| 8 | 198.5 | 19.5 | 224,500 | 216.2 | 39.7 | 110,400 | 64.0 | 6.0 | 49,500 |
| 9 | 290.0 | 29.2 | 26,900 | 245.7 | 42.7 | 62,700 | 92.5 | 9.7 | 33,600 |
| 2009 | 2010 | 2011 | |||||||
| 1 | 115.3 | 10.9 | 128,900 | 133.6 | 15.2 | 75,100 | 117.8 | 11.0 | 81,300 |
| 2 | 95.7 | 7.4 | 128,100 | 97.0 | 8.0 | 57,300 | 104.7 | 8.1 | 101,100 |
| 3 | 26.2 | 2.3 | 76,500 | 65.4 | 10.5 | 57,800 | 102.8 | 8.1 | 102,700 |
| 4 | 159.2 | 15.3 | 28,600 | - | - | - | 45.5 | 4.3 | 54,900 |
| 5 | 2.8 | 0.3 | 3400 | - | - | - | 13.2 | 1.4 | 24,900 |
| 6 | 410.0 | 42.5 | 25,400 | - | - | - | 22.4 | 2.2 | 33,700 |
| 7 | 122.0 | 13.1 | 28,600 | - | - | - | 137.0 | 10.9 | 50,600 |
| 8 | 100.8 | 9.2 | 33,800 | - | - | - | 27.3 | 2.5 | 24,100 |
| 9 | 84.5 | 8.6 | 24,100 | - | - | - | 78.5 | 8.3 | 22,600 |
| 2012 | 2013 | 2014 | |||||||
| 1 | 70.3 | 5.1 | 18,800.0 | 497.7 | 51.9 | 113,500 | 684.5 | 61.5 | 94,300 |
| 2 | 22.0 | 2.0 | 16,200.0 | 193.9 | 20.9 | 138,400 | 396.8 | 43.5 | 66,400 |
| 3 | 37.7 | 2.3 | 20,400.0 | 67.4 | 7.0 | 65,800 | 341.2 | 48.5 | 54,300 |
| 4 | 57.3 | 5.6 | 45,500.0 | 150.8 | 22.3 | 14,800 | 124.8 | 17.8 | 36,400 |
| 5 | 170.4 | 19.1 | 50,600.0 | 224.3 | 28.6 | 18,300 | 102.2 | 14.9 | 12,500 |
| 6 | 65.8 | 6.9 | 53,000 | 354.6 | 53.7 | 17,500 | 162.9 | 24.2 | 46,800 |
| 7 | 467.0 | 47.7 | 101,100 | 478.3 | 65.3 | 34,300 | 648.5 | 56.8 | 52,400 |
| 8 | 179.0 | 17.6 | 52,500 | 298.3 | 38.0 | 35,900 | 460.6 | 38.8 | 34,700 |
| 9 | 98.5 | 10.1 | 48,500 | 421.4 | 61.7 | 26,800 | 233.2 | 19.1 | 19,400 |
| Variable | Correlation | Correlation Level | |
|---|---|---|---|
| Stand | N (trees ha−1) | −0.21536 | 0.0713 |
| QMD (cm) | 0.43380 | 0.0002 | |
| Hdom (m) | −0.27097 | 0.0223 | |
| V (m3 ha−1) | 0.46478 | <0.0001 | |
| Vs (m3 ha−1) | 0.46474 | <0.0001 | |
| Bbranches > 7 (Mg ha−1) | 0.46548 | <0.0001 | |
| Bbranches < 2 (Mg ha−1) | 0.45570 | <0.0001 | |
| Broot (Mg ha−1) | 0.45907 | <0.0001 | |
| SMI parameter | 0.47147 | <0.0001 | |
| Soil | pH | 0.20622 | 0.0845 |
| NMg | −0.20905 | 0.0802 | |
| NK | −0.25277 | 0.0334 | |
| Climatic | SP (mm) | 0.49268 | <0.0001 |
| NP (mm) | 0.26051 | 0.0282 | |
| MT (°C) | 0.22344 | 0.0611 | |
| ST (°C) | 0.31817 | 0.0069 | |
| OT (°C) | 0.22684 | 0.0571 | |
| NT (°C) | 0.20052 | 0.0936 |
| Effect | Estimate | Standard Error | DenDF | F-Value | Pr > F |
|---|---|---|---|---|---|
| Incercept | 10.5544 | 1.2807 | 8 | 8.24 | <0.0001 |
| Ln SMI | 3.1786 | 0.5619 | 69 | 5.66 | <0.0001 |
| TsPs | 0.000906 | 0.000173 | 69 | 5.24 | <0.0001 |
| TnPn | 0.001926 | 0.000129 | 69 | 14.96 | <0.0001 |
| Estimated Variance Parameter | |
|---|---|
| Plot | 0.1360 |
| 2003 | 0.3898 |
| 2004 | 0.7622 |
| 2005 | 15.0219 |
| 2006 | 0.7538 |
| 2007 | 0.5748 |
| 2008 | 0.9821 |
| 2009 | 1.7094 |
| 2010 | 0.02406 |
| 2011 | 0.4127 |
| 2012 | 1.3938 |
| 2013 | 3.6923 |
| 2014 | 0.01082 |
| 0.6119 |
| Age (year) | Stand Variables | Climatic Scenarios | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hdom (m) | N (trees ha−1) | G (m2 ha−1) | c1 | c2 | c3 | c4 | c5 | c6 | c7 | c8 | c9 | c10 | c11 | c12 | c13 | c14 | c15 | c16 | c17 | c18 | |
| 53 | 14.85 | 1793.13 | 38.6 | 0.00 | 0.00 | 2.02 | 0.00 | 0.23 | 0.00 | 0.90 | 2.17 | 0.13 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| 58 | 15.56 | 1778.37 | 43.01 | 0.00 | 0.00 | 3.37 | 0.26 | 0.77 | 0.00 | 1.75 | 3.59 | 0.64 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.06 |
| 63 | 15.79 | 1763.43 | 47,22 | 0.03 | 0.00 | 5.04 | 0.74 | 1.45 | 0.23 | 2.80 | 5.35 | 1.27 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.16 | 0.30 | 0.46 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herrero, C.; Berraondo, I.; Bravo, F.; Pando, V.; Ordóñez, C.; Olaizola, J.; Martín-Pinto, P.; Oria de Rueda, J.A. Predicting Mushroom Productivity from Long-Term Field-Data Series in Mediterranean Pinus pinaster Ait. Forests in the Context of Climate Change. Forests 2019, 10, 206. https://doi.org/10.3390/f10030206
Herrero C, Berraondo I, Bravo F, Pando V, Ordóñez C, Olaizola J, Martín-Pinto P, Oria de Rueda JA. Predicting Mushroom Productivity from Long-Term Field-Data Series in Mediterranean Pinus pinaster Ait. Forests in the Context of Climate Change. Forests. 2019; 10(3):206. https://doi.org/10.3390/f10030206
Chicago/Turabian StyleHerrero, Celia, Iosu Berraondo, Felipe Bravo, Valentín Pando, Cristóbal Ordóñez, Jaime Olaizola, Pablo Martín-Pinto, and Juan Andrés Oria de Rueda. 2019. "Predicting Mushroom Productivity from Long-Term Field-Data Series in Mediterranean Pinus pinaster Ait. Forests in the Context of Climate Change" Forests 10, no. 3: 206. https://doi.org/10.3390/f10030206
APA StyleHerrero, C., Berraondo, I., Bravo, F., Pando, V., Ordóñez, C., Olaizola, J., Martín-Pinto, P., & Oria de Rueda, J. A. (2019). Predicting Mushroom Productivity from Long-Term Field-Data Series in Mediterranean Pinus pinaster Ait. Forests in the Context of Climate Change. Forests, 10(3), 206. https://doi.org/10.3390/f10030206






