Synthesis, Characterization, and Electrochemical Evaluation of Electrodeposited NiCuZn Powders for Urea Oxidation
Abstract
1. Introduction
2. Materials and Methods
2.1. Material Synthesis
2.2. Morphology and Chemical Analysis
2.2.1. Electrochemical Measurements
2.2.2. Electrochemical Activity Measurements
2.2.3. Electrocatalytic Urea Oxidation
2.2.4. Stability Measurements—Chronoamperometry
2.2.5. Stability Measurements—Multicycles
3. Results
3.1. SEM and EDX
3.2. Electrochemical Tests
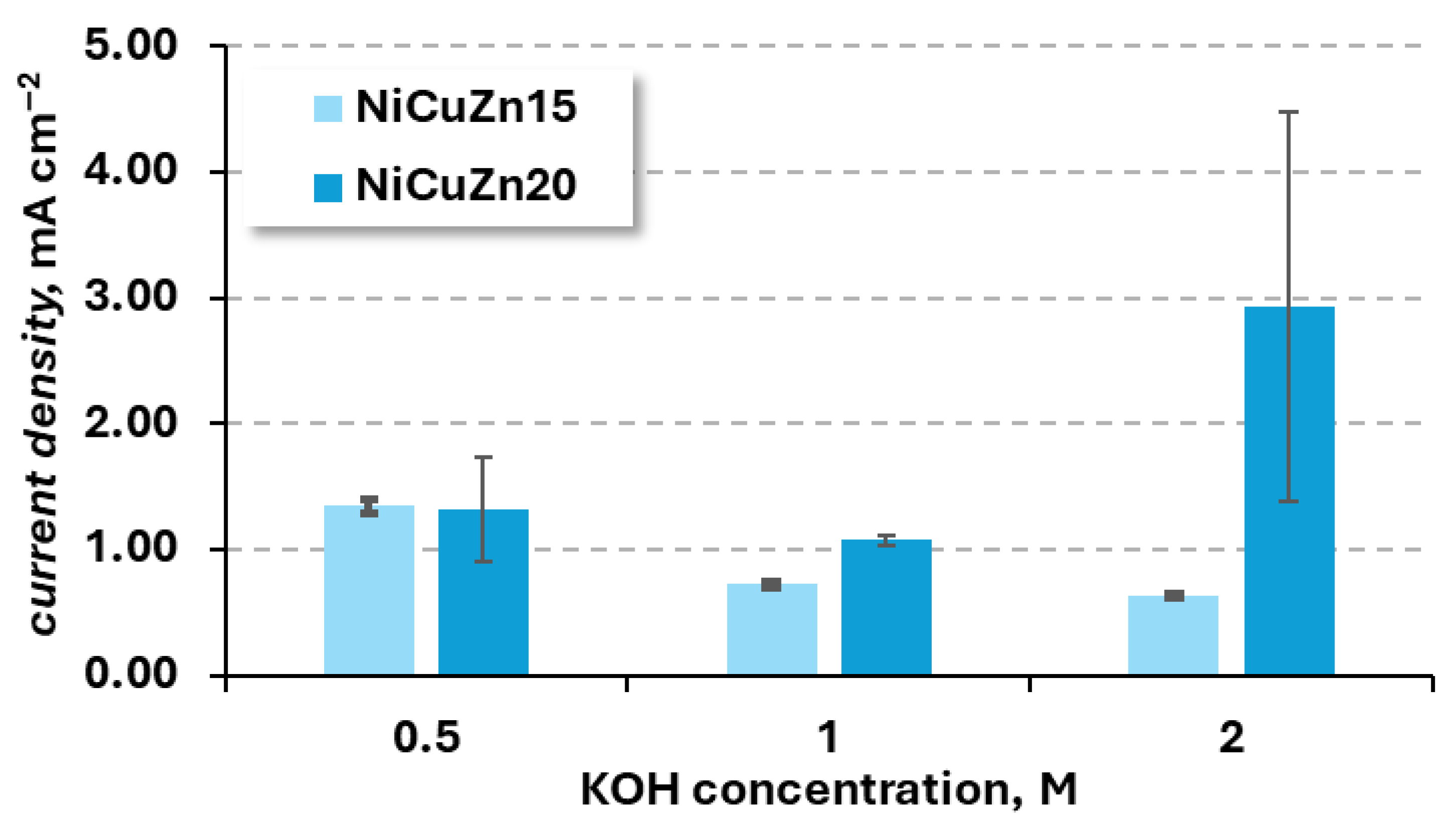
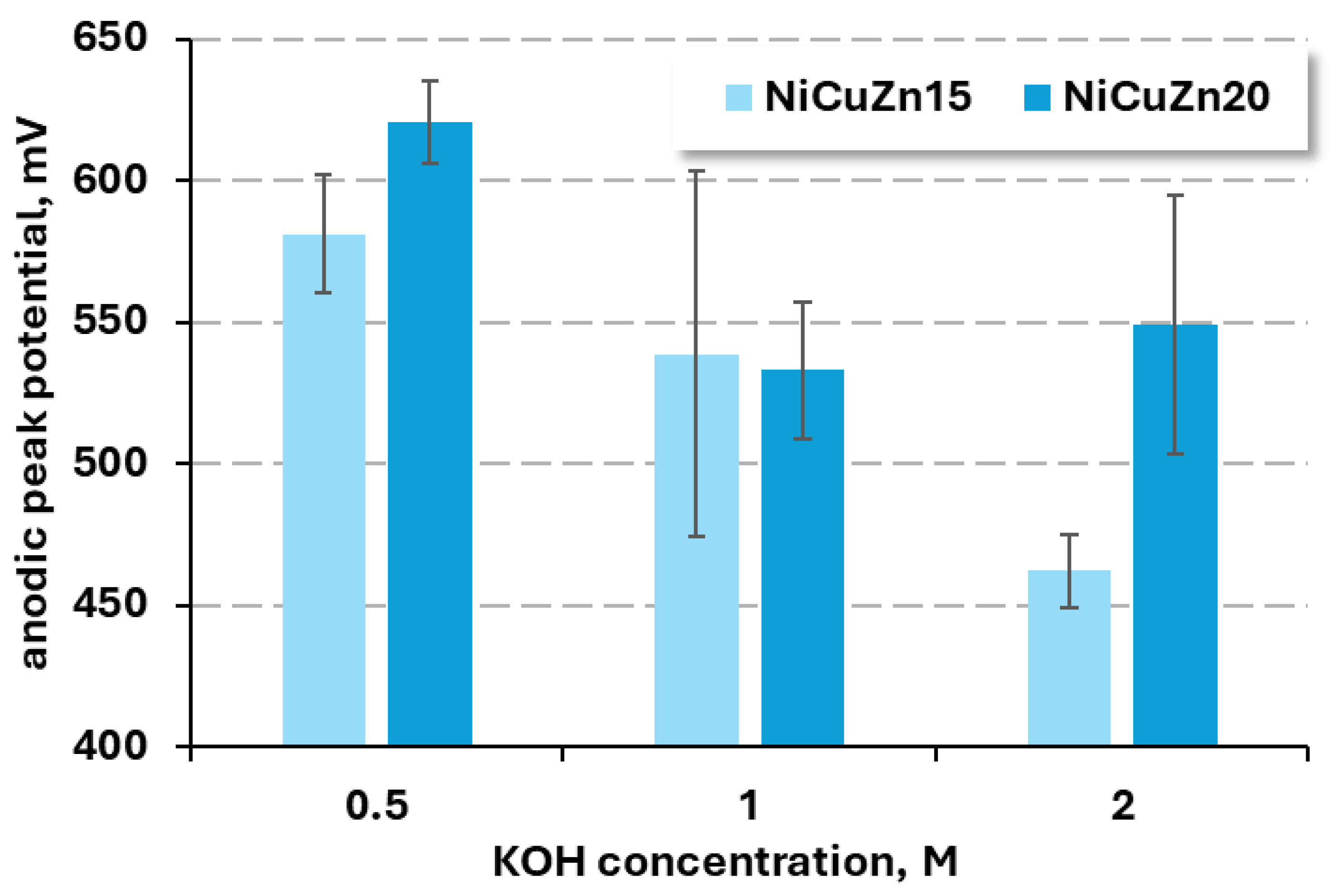
3.2.1. KOH Concentration Influence
3.2.2. Urea Oxidation
3.3. Stability Examination
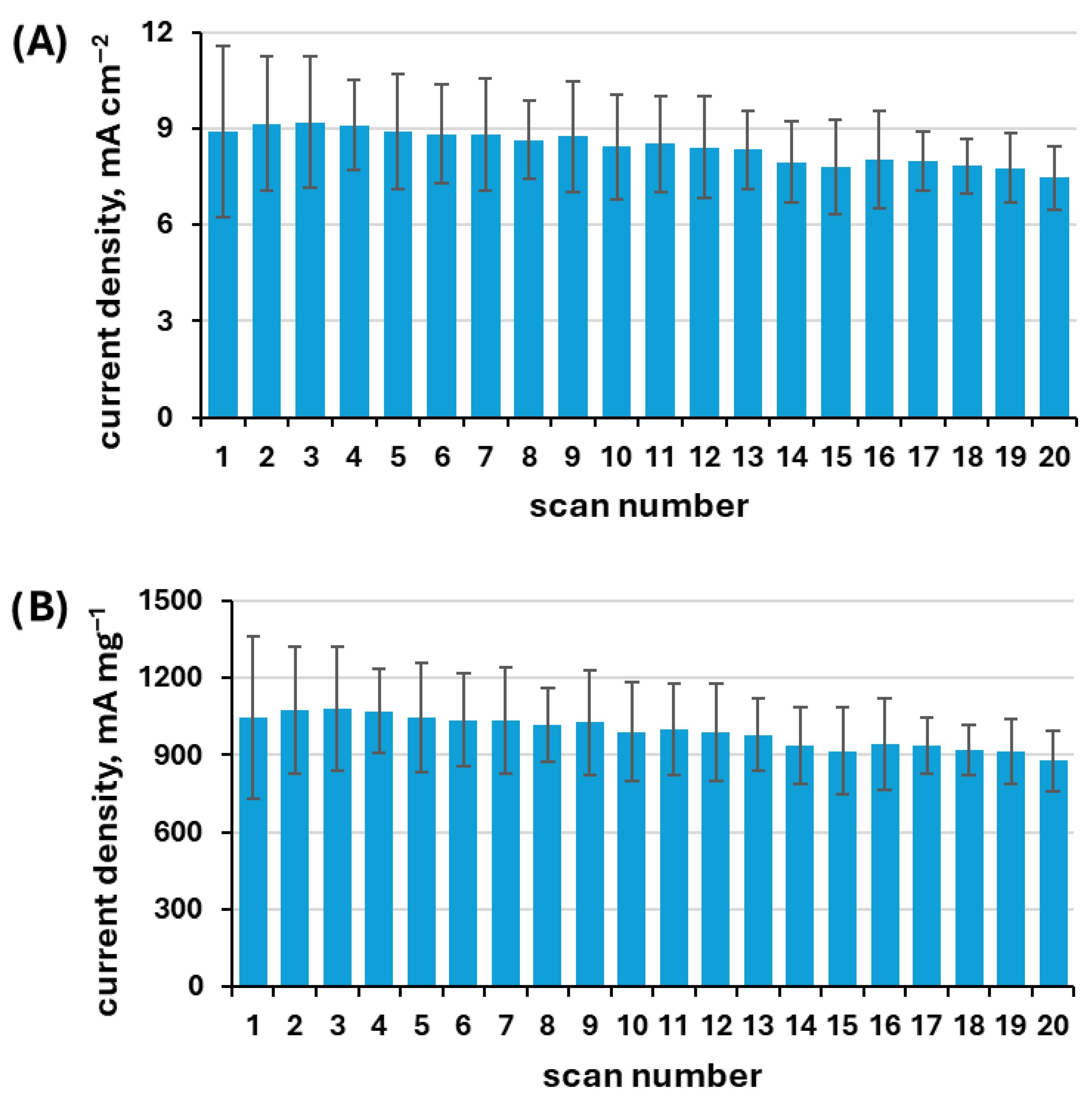
3.3.1. Multiscan Test
3.3.2. Chronoamperometry
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Farghali, M.; Osman, A.I.; Mohamed, I.M.A.; Chen, Z.; Chen, L.; Ihara, I.; Yap, P.S.; Rooney, D.W. Strategies to save energy in the context of the energy crisis: A review. Environ. Chem. Lett. 2023, 21, 2003–2039. [Google Scholar] [CrossRef]
- Guaitolini, S.V.M.; Fardin, J.F. Fuel Cells: History, Principles of Operation, Main Features, and Applications. Adv. Renew. Energ. Power Technol. 2018, 2, 123–150. [Google Scholar] [CrossRef]
- Vakulchuk, R.; Overland, I.; Scholten, D. Renewable energy and geopolitics: A review. Renew. Sustain. Energy Rev. 2020, 122, 109547. [Google Scholar] [CrossRef]
- Moriarty, P.; Honnery, D. What is the global potential for renewable energy? Renew. Sustain. Energy Rev. 2012, 16, 244–252. [Google Scholar] [CrossRef]
- Alex, C.; Shukla, G.; John, N.S. Introduction of surface defects in NiO with effective removal of adsorbed catalyst poisons for improved electrochemical urea oxidation. Electrochim. Acta 2021, 385, 138425. [Google Scholar] [CrossRef]
- Mohamed, I.M.A.; Kanagaraj, P.; Yasin, A.S.; Iqbal, W.; Liu, C. Electrochemical impedance investigation of urea oxidation in alkaline media based on electrospun nanofibers towards the technology of direct-urea fuel cells. J. Alloys Compd. 2020, 816, 152513. [Google Scholar] [CrossRef]
- Gnana Kumar, G.; Farithkhan, A.; Manthiram, A. Direct Urea Fuel Cells: Recent Progress and Critical Challenges of Urea Oxidation Electrocatalysis. Adv. Energy Sustain. Res. 2020, 1, 2000015. [Google Scholar] [CrossRef]
- Yu, X.; Williams, C.T. Recent applications of nickel and nickel-based bimetallic catalysts for hydrodeoxygenation of biomass-derived oxygenates to fuels. Catal. Sci. Technol. 2023, 13, 802–825. [Google Scholar] [CrossRef]
- Yousef, A.; El-Newehy, M.H.; Al-Deyab, S.S.; Barakat, N.A.M. Facile synthesis of Ni-decorated multi-layers graphene sheets as effective anode for direct urea fuel cells. Arab. J. Chem. 2017, 10, 811–822. [Google Scholar] [CrossRef]
- Ahmadi, P.; Torabi, S.H.; Afsaneh, H.; Sadegheih, Y.; Ganjehsarabi, H.; Ashjaee, M. The effects of driving patterns and PEM fuel cell degradation on the lifecycle assessment of hydrogen fuel cell vehicles. Int. J. Hydrogen Energy 2020, 45, 3595–3608. [Google Scholar] [CrossRef]
- King, R.L.; Botte, G.G. Hydrogen production via urea electrolysis using a gel electrolyte. J. Power Sources 2011, 196, 2773–2778. [Google Scholar] [CrossRef]
- Shekhawat, A.; Samanta, R.; Panigrahy, S.; Barman, S. Electrocatalytic Oxidation of Urea and Ethanol on Two-Dimensional Amorphous Nickel Oxide Encapsulated on N-Doped Carbon Nanosheets. ACS Appl. Energy Mater. 2023, 6, 3135–3146. [Google Scholar] [CrossRef]
- Urbańczyk, E.; Wala, M.; Blacha-Grzechnik, A.; Stolarczyk, A.; Maciej, A.; Simka, W. Electrocatalytic methanol oxidation using Ni–Co–graphene composite electrodes. Int. J. Hydrogen Energy 2021, 46, 33272–33286. [Google Scholar] [CrossRef]
- Mansor, M.; Timmiati, S.N.; Lim, K.L.; Wong, W.Y.; Kamarudin, S.K.; Nazirah Kamarudin, N.H. Recent progress of anode catalysts and their support materials for methanol electrooxidation reaction. Int. J. Hydrogen Energy 2019, 44, 14744–14769. [Google Scholar] [CrossRef]
- Zakaria, Z.; Kamarudin, S.K.; Wahid, K.A.A. Polymer electrolyte membrane modification in direct ethanol fuel cells: An update. J. Appl. Polym. Sci. 2023, 140, e53383. [Google Scholar] [CrossRef]
- Baruah, S.; Kumar, A.; Peela, N.R. Advancing paper-based microfluidic ethanol fuel cells with gel-assisted dual electrolytes: A step towards scalable power solutions. Energy Convers. Manag. 2025, 332, 119767. [Google Scholar] [CrossRef]
- Tran, T.Q.N.; Park, B.J.; Yun, W.H.; Duong, T.N.; Yoon, H.H. Metal–organic framework–derived Ni@C and NiO@C as anode catalysts for urea fuel cells. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef]
- Yan, W.; Wang, D.; Botte, G.G. Electrochemical decomposition of urea with Ni-based catalysts. Appl. Catal. B Environ. 2012, 127, 221–226. [Google Scholar] [CrossRef]
- Liu, G.; Xie, C.; Zhang, Y.; Du, Y.; Wang, J.; Lin, J.; Bai, J.; Li, J.; Zhou, C.; Zhou, T.; et al. Synergistic etching of nickel foam by Fe3+ and Cl− ions to synthesize nickel-iron-layered double hydroxide nanolayers with abundant oxygen vacancies for superior urea oxidation. J. Colloid Interface Sci. 2024, 668, 375–384. [Google Scholar] [CrossRef]
- Cai, M.; Zhu, Q.; Wang, X.; Shao, Z.; Yao, L.; Zeng, H.; Wu, X.; Chen, J.; Huang, K.; Feng, S. Formation and Stabilization of NiOOH by Introducing α-FeOOH in LDH: Composite Electrocatalyst for Oxygen Evolution and Urea Oxidation Reactions. Adv. Mater. 2023, 35, e2209338. [Google Scholar] [CrossRef]
- Yang, K.; Hao, L.; Hou, Y.; Zhang, J.; Yang, J.H. Summary and application of Ni-based catalysts for electrocatalytic urea oxidation. Int. J. Hydrogen Energy 2024, 51, 966–981. [Google Scholar] [CrossRef]
- Wala, M.; Blacha–Grzechnik, A.; Stolarczyk, A.; Bajkacz, S.; Dydo, P.; Simka, W. Unexpected electrochemical oxidation of urea on a new NiCuGO composite catalyst. Int. J. Hydrogen Energy 2023, 48, 34229–34243. [Google Scholar] [CrossRef]
- Goel, R.; Jha, R.; Ravikant, C. Investigating the structural, electrochemical, and optical properties of p-type spherical nickel oxide (NiO) nanoparticles. J. Phys. Chem. Solids 2020, 144, 109488. [Google Scholar] [CrossRef]
- Tariq, I.; Iqbal, W.; Haider, A.; Ma, M. Recent advances in carbon nanotube-supported non-noble metal electrocatalysts for urea oxidation reaction. Int. J. Hydrogen Energy 2025, 142, 40–53. [Google Scholar] [CrossRef]
- Ma, Y.; Ma, C.; Wang, Y.; Wang, K. Advanced Nickel-Based Catalysts for Urea Oxidation Reaction: Challenges and Developments. Catalysts 2022, 12, 337. [Google Scholar] [CrossRef]
- Tatarchuk, S.W.; Medvedev, J.J.; Li, F.; Tobolovskaya, Y.; Klinkova, A. Nickel-Catalyzed Urea Electrolysis: From Nitrite and Cyanate as Major Products to Nitrogen Evolution. Angew. Chem. 2022, 134, e202209839. [Google Scholar] [CrossRef]
- Li, S.-M.; Zhang, H.-R.; Liu, J.-H. Corrosion behavior of aluminum alloy 2024-T3 by 8-hydroxy-quinoline and its derivative in 3.5% chloride solution. Trans. Nonferrous Met. Soc. China 2007, 17, 318–325. [Google Scholar] [CrossRef]
- Munde, A.V.; Mulik, B.B.; Chavan, P.P.; Sathe, B.R. Enhanced electrocatalytic activity towards urea oxidation on Ni nanoparticle decorated graphene oxide nanocomposite. Electrochim. Acta 2020, 349, 136386. [Google Scholar] [CrossRef]
- Guo, F.; Ye, K.; Du, M.; Huang, X.; Cheng, K.; Wang, G.; Cao, D. Electrochemical impedance analysis of urea electro-oxidation mechanism on nickel catalyst in alkaline medium. Electrochim. Acta 2016, 210, 474–482. [Google Scholar] [CrossRef]
- Wala-Kapica, M.; Gąsior, A.; Maciej, A.; Smykała, S.; Kazek-Kęsik, A.; Baghayeri, M.; Simka, W. One-Pot Fast Electrochemical Synthesis of Ternary Ni-Cu-Fe Particles for Improved Urea Oxidation. Energies 2024, 17, 5455. [Google Scholar] [CrossRef]
- Li, J.; Wang, S.; Chang, J.; Feng, L. A review of Ni based powder catalyst for urea oxidation in assisting water splitting reaction. Adv. Powder Mater. 2022, 1, 100030. [Google Scholar] [CrossRef]
- Guo, F.; Cao, D.; Du, M.; Ye, K.; Wang, G.; Zhang, W.; Gao, Y.; Cheng, K. Enhancement of direct urea-hydrogen peroxide fuel cell performance by three-dimensional porous nickel-cobalt anode. J. Power Sources 2016, 307, 697–704. [Google Scholar] [CrossRef]
- Basumatary, P.; Konwar, D.; Yoon, Y.S. A novel Ni–Cu/ZnO@MWCNT anode employed in urea fuel cell to attain superior performances. Electrochim. Acta 2018, 261, 78–85. [Google Scholar] [CrossRef]
- Abdel Hameed, R.M.; Medany, S.S. NiO nanoparticles on graphene nanosheets at different calcination temperatures as effective electrocatalysts for urea electro-oxidation in alkaline medium. J. Colloid Interface Sci. 2017, 508, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Yan, W.; Vijapur, S.H.; Botte, G.G. Electrochemically reduced graphene oxide–nickel nanocomposites for urea electrolysis. Electrochim. Acta 2013, 89, 732–736. [Google Scholar] [CrossRef]
- Ye, K.; Zhang, D.; Guo, F.; Cheng, K.; Wang, G.; Cao, D. Highly porous nickel@carbon sponge as a novel type of three-dimensional anode with low cost for high catalytic performance of urea electro-oxidation in alkaline medium. J. Power Sources 2015, 283, 408–415. [Google Scholar] [CrossRef]
- Abd El-Lateef, H.M.; Almulhim, N.F.; Alaulamie, A.A.; Saleh, M.M.; Mohamed, I.M.A. Design of ultrafine nickel oxide nanostructured material for enhanced electrocatalytic oxidation of urea: Physicochemical and electrochemical analyses. Colloids Surf. A Physicochem. Eng. Asp. 2020, 585, 124092. [Google Scholar] [CrossRef]
- Chakrabarty, S.; Offen-Polak, I.; Burshtein, T.Y.; Farber, E.M.; Kornblum, L.; Eisenberg, D. Urea oxidation electrocatalysis on nickel hydroxide: The role of disorder. J. Solid State Electrochem. 2021, 25, 159–171. [Google Scholar] [CrossRef]
- Xiao, M.; Tian, Y.; Yan, Y.; Feng, K.; Miao, Y. Electrodeposition of Ni(OH)2/NiOOH in the Presence of Urea for the Improved Oxygen Evolution. Electrochim. Acta 2015, 164, 196–202. [Google Scholar] [CrossRef]
- Hakami, W.; Danish, E.Y.; Khan, A.N.; Khudaysh, A.; Aslam, M.; Soomro, M.T. An innovative electrochemical approach for selective and sensitive urea sensing using NiNPs modified GCE. Sci. Rep. 2025, 15, 39698. [Google Scholar] [CrossRef]
- Nur Indah Sari, F.; Ke, M.T.; Huang, Y.J.; Zheng, T.M.; Su, Y.H.; Ting, J.M. NiFe sulfide electronic structure modulation via metal doping towards enhanced urea oxidation reaction performance. Appl. Surf. Sci. 2024, 649, 159187. [Google Scholar] [CrossRef]
- Wickramaarachchi, K.; Minakshi, M. Status on electrodeposited manganese dioxide and biowaste carbon for hybrid capacitors: The case of high-quality oxide composites, mechanisms, and prospects. J. Energy Storage 2022, 56, 106099. [Google Scholar] [CrossRef]
- Plowman, B.J.; Jones, L.A.; Bhargava, S.K. Building with bubbles: The formation of high surface area honeycomb-like films via hydrogen bubble templated electrodeposition. Chem. Commun. 2015, 51, 4331–4346. [Google Scholar] [CrossRef]
- Li, Y.; Jia, W.Z.; Song, Y.Y.; Xia, X.H. Superhydrophobicity of 3D Porous Copper Films Prepared Using the Hydrogen Bubble Dynamic Template. Chem. Mater. 2007, 19, 5758–5764. [Google Scholar] [CrossRef]
- Zhang, Z.; Xie, M.; Liu, Z.; Lu, Y.; Zhang, S.; Liu, M.; Liu, K.; Cheng, T.; Gao, C. Ultrathin Pt-Cu-Ni Ternary Alloy Nanowires with Multimetallic Interplay for Boosted Methanol Oxidation Activity. ACS Appl. Energy Mater. 2021, 4, 6824–6832. [Google Scholar] [CrossRef]
- Wang, C.; Du, X.; Zhang, X. Controlled synthesis of Fe doped NiCoM (M=O, P, S and Se) as robust electrocatalyst for urea electrolysis. J. Alloys Compd. 2022, 928, 167094. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, W.; Shen, J.; Zhang, X.; Liu, Z.; Liu, J. Trimetallic nanoplate arrays of Ni-Fe-Mo sulfide on FeNi3 foam: A highly efficient and bifunctional electrocatalyst for overall water splitting. J. Alloys Compd. 2022, 902, 163670. [Google Scholar] [CrossRef]
- Wala, M.; Szewczyk, M.; Leśniak–Ziółkowska, K.; Kazek–Kęsik, A.; Simka, W. Preparation of NiCuGO composite and investigation of its electrocatalytic properties in methanol oxidation. Electrochim. Acta 2022, 425, 140743. [Google Scholar] [CrossRef]
- Wu, M.S.; Chen, F.Y.; Lai, Y.H.; Sie, Y.J. Electrocatalytic oxidation of urea in alkaline solution using nickel/nickel oxide nanoparticles derived from nickel-organic framework. Electrochim. Acta 2017, 258, 167–174. [Google Scholar] [CrossRef]
- Luo, Y.; Zhou, H.; Tong, Y. Advances in Nanostructured Catalysts for Urea-Assisted Water Splitting and Zn-Urea Batteries. ChemSusChem 2026, 19, e202502504. [Google Scholar] [CrossRef] [PubMed]
- Chu, S.; Chen, W.; Chen, G.; Huang, J.; Zhang, R.; Song, C.; Wang, X.; Li, C.; Ostrikov, K. Holey Ni-Cu phosphide nanosheets as a highly efficient and stable electrocatalyst for hydrogen evolution. Appl. Catal. B Environ. 2019, 243, 537–545. [Google Scholar] [CrossRef]
- Zhang, H.M.; Wang, Y.F.; Kwok, Y.H.; Wu, Z.C.; Xia, D.H.; Leung, D.Y.C. A Direct Ammonia Microfluidic Fuel Cell using NiCu Nanoparticles Supported on Carbon Nanotubes as an Electrocatalyst. ChemSusChem 2018, 11, 2889–2897. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.S.; Zheng, Y.X.; Jia, M.L.; Zhang, S.J.; Wang, X.L.; Peng, C. Nanoporous nickel-copper-phosphorus amorphous alloy film for methanol electro-oxidation in alkaline medium. Electrochim. Acta 2015, 154, 54–62. [Google Scholar] [CrossRef]
- Noor, T.; Pervaiz, S.; Iqbal, N.; Nasir, H.; Zaman, N.; Sharif, M.; Pervaiz, E. Nanocomposites of NiO/CuO based MOF with rGO: An efficient and robust electrocatalyst for methanol oxidation reaction in DMFC. Nanomaterials 2020, 10, 1601. [Google Scholar] [CrossRef] [PubMed]
- Wittman, R.M.; Sacci, R.L.; Zawodzinski, T.A. Alkaline Zinc Passivation Mechanism is Controlled by Hydroxide Concentration. J. Electrochem. Soc. 2025, 172, 040514. [Google Scholar] [CrossRef]
- Hampson, N.A.; Shawt, P.E.; Taylor, R. Anodic Behaviour of Zinc in Potassium Hydroxide Solution: II. Horizontal Anodes in Electrolytes Containing Zn(II). Br. Corros. J. 1969, 4, 207–211. [Google Scholar] [CrossRef]
- Vedharathinam, V.; Botte, G.G. Understanding the electro-catalytic oxidation mechanism of urea on nickel electrodes in alkaline medium. Electrochim. Acta 2012, 81, 292–300. [Google Scholar] [CrossRef]
- Hopsort, G.; Carmo, D.P.D.; Latapie, L.; Loubière, K.; Serrano, K.G.; Tzedakis, T. Progress toward a better understanding of the urea oxidation by electromediation of Ni(III)/Ni(II) system in alkaline media. Electrochim. Acta 2023, 442, 141898. [Google Scholar] [CrossRef]
- Schwab, B.; Ruh, A.; Manthey, J.; Drosik, M.Z. Ullmann’s Encycl. Ind. Chem. 2015, 1–25. [Google Scholar] [CrossRef]
- Merten, H. Zinc Alloys. Mold-Mak. Handb. Plast. Eng. 1983, 253–260. [Google Scholar] [CrossRef]
- Ayyanusamy, P.; Swathi Tharani, D.; Alphonse, R.; Minakshi, M.; Sivasubramanian, R. Synthesis of Amorphous Nickel-Cobalt Hydroxides for Ni−Zn Batteries. Chem. A Eur. J. 2024, 30, e202402325. [Google Scholar] [CrossRef]
- Wang, L.; Ren, L.; Wang, X.; Feng, X.; Zhou, J.; Wang, B. Multivariate MOF-Templated Pomegranate-Like Ni/C as Efficient Bifunctional Electrocatalyst for Hydrogen Evolution and Urea Oxidation. ACS Appl. Mater. Interfaces 2018, 10, 4750–4756. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, R.; Chen, W. Graphene-Supported Nanoelectrocatalysts for Fuel Cells: Synthesis, Properties, and Applications. Chem. Rev. 2014, 114, 5117–5160. [Google Scholar] [CrossRef]
- Li, J.; Wang, S.; Sun, S.; Wu, X.; Zhang, B.; Feng, L. A review of hetero-structured Ni-based active catalysts for urea electrolysis. J. Mater. Chem. A 2022, 10, 9308–9326. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, N.; Du, X.; Han, X.; Zhang, X. Transition metal atoms M (M = Mn, Fe, Cu, Zn) doped nickel-cobalt sulfides on the Ni foam for efficient oxygen evolution reaction and urea oxidation reaction. J. Alloys Compd. 2022, 893, 162269. [Google Scholar] [CrossRef]
- Zhang, R.; Wang, Y.; Gao, X.; Duan, D.; Wang, J.; Liu, S. Electrodeposited zinc cobalt bimetallic phosphate as a bifunctional catalyst for hydrogen evolution and urea oxidation. J. Alloys Compd. 2024, 1009, 176818. [Google Scholar] [CrossRef]
- Clark, R.; Moore, A.; MacInnis, M.; Bertin, E. Investigation of urea oxidation as a potential anode reaction during CO2 electrolysis. J. Appl. Electrochem. 2021, 51, 1583–1590. [Google Scholar] [CrossRef]
- Carpenter, K.; Stuve, E.M. Electrooxidation of urea and creatinine on nickel foam-based electrocatalysts. J. Appl. Electrochem. 2021, 51, 945–957. [Google Scholar] [CrossRef]
- Zhang, H.J.; Chen, Z.Q.; Ye, X.T.; Xiao, K.; Liu, Z.Q. Electron delocalized ni active sites in spinel catalysts enable efficient urea oxidation Angew. Chem. Int. Ed. 2025, 64, e202421027. [Google Scholar] [CrossRef] [PubMed]
- Bender, M.T.; Lam, Y.C.; Hammes-Schiffer, S.; Choi, K.S. Unraveling Two Pathways for Electrochemical Alcohol and Aldehyde Oxidation on NiOOH. J. Am. Chem. Soc. 2020, 142, 21538–21547. [Google Scholar] [CrossRef]







| Ingredient | Concentration [M] | pH |
|---|---|---|
| NiSO4 | 0.375 | 9 |
| CuSO4 | 0.125 | |
| Sodium citrate | 0.2 | |
| ZnSO4 | 0.1 |
| [KOH], M | NiCuZn15 | NiCuZn20 | ||
|---|---|---|---|---|
| ipA, mA mg−1 | ipA, mA cm−2 | ipA, mA mg−1 | ipA, mA cm−2 | |
| 0.5 | 158.1 ± 6.4 | 1.34 ± 0.05 | 155.6 ± 49.0 | 1.32 ± 0.42 |
| 1 | 85.3 ± 3.0 | 0.73 ± 0.03 | 126.3 ± 5.3 | 1.07 ± 0.04 |
| 2 | 74.2 ± 1.3 | 0.63 ± 0.01 | 344.8 ± 181.4 | 2.93 ± 1.54 |
| Material | Solution | Onset Potential, E vs RHE, V | Peak Potential, E vs RHE, V | Peak Current Density [mA cm−2] | Reference |
|---|---|---|---|---|---|
| NiCuZn20 | 0.15 M urea/1 M KOH | 1.42 | 1.72 | 10.0 | - |
| Ni-Cu-Fe | 0.15 M urea/1 M KOH | 1.70 | 12.0 | [30] | |
| Ni-Zn-Co | 0.33 M urea/5 M KOH | 1.27 | 1.44 | 24.0 | [18] |
| NiO | 0.4 M urea/6 M KOH | 1.26 | 1.37 | 20.0 | [5] |
| NiO/Gr | 0.3 M urea/0.5 M NaOH | 1.40 | 1.73 | 30.9 | [34] |
| Ni/Pd-C | 0.3 M urea/2 M KOH | 1.37 | 1.47 | 63.0 | [6] |
| NiFe-LDH | 0.33 M urea/1 M KOH | 1.30 | 10.0 | [19] | |
| Ni/Gr | 0.33 M urea/1 M KOH | 1.39 | 1.92 | 81.7 | [9] |
| Ni@GO | 0.33 M urea/1 M KOH | 1.37 | 1.56 | 17.1 | [28] |
| Ultrafine NiO nanoparticles | 0.25 M urea/1 M KOH | 1.42 | 216.1 | [37] | |
| NiCuGO20 | 0.15 M urea/1 M KOH | 1.74 | 5.9 | [22] | |
| NiCu20 | 0.15 M urea/1 M KOH | 1.73 | 3.9 | [22] | |
| Ni@C | 0.1 M urea/0.1 M KOH | 1.41 | 1.62 | 40.0 | [17] |
| NiO@C | 0.1 M urea/0.1 M KOH | 1.35 | 1.67 | 25.0 | [17] |
| Ni Cu/ZnO@ MWCNT | 0.07 M urea/0.4 M KOH | 1.40 | 30.0 | [33] | |
| NiFeTiS | 0.33 M urea/1 M KOH | 2.38 | 100.0 | [41] | |
| Ni/C | 1.33 | [62] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kołkowska, A.; Lisieński, W.; Gardas, Ł.; Shang, W.; Gąsior, A.; Maciej, A.; Wala-Kapica, M.; Simka, W. Synthesis, Characterization, and Electrochemical Evaluation of Electrodeposited NiCuZn Powders for Urea Oxidation. Materials 2026, 19, 1973. https://doi.org/10.3390/ma19101973
Kołkowska A, Lisieński W, Gardas Ł, Shang W, Gąsior A, Maciej A, Wala-Kapica M, Simka W. Synthesis, Characterization, and Electrochemical Evaluation of Electrodeposited NiCuZn Powders for Urea Oxidation. Materials. 2026; 19(10):1973. https://doi.org/10.3390/ma19101973
Chicago/Turabian StyleKołkowska, Agata, Wojciech Lisieński, Łukasz Gardas, Weizhi Shang, Aleksander Gąsior, Artur Maciej, Marta Wala-Kapica, and Wojciech Simka. 2026. "Synthesis, Characterization, and Electrochemical Evaluation of Electrodeposited NiCuZn Powders for Urea Oxidation" Materials 19, no. 10: 1973. https://doi.org/10.3390/ma19101973
APA StyleKołkowska, A., Lisieński, W., Gardas, Ł., Shang, W., Gąsior, A., Maciej, A., Wala-Kapica, M., & Simka, W. (2026). Synthesis, Characterization, and Electrochemical Evaluation of Electrodeposited NiCuZn Powders for Urea Oxidation. Materials, 19(10), 1973. https://doi.org/10.3390/ma19101973







