Design and Characterization of 3D Printed Auxetic PLA-HA Composite Scaffolds for Biomedical Application
Highlights
- Three-Dimensional printed auxetic structures of a biocompatible PLA-40% HA composite.
- Presence of HA in the thermoplastic improves the wettability, Young modulus, and wear resistance.
- Soaking in PBS degrades the polymer, causing the composite to present worse mechanical resistance.
- Re-entrant auxetic structure demonstrated notable mechanical retention after biodegradation.
Abstract
1. Introduction
2. Materials and Methods
2.1. Starting Materials
2.2. Samples Design and Printing
2.3. XRD Analysis
2.4. Density Analysis
2.5. Raman Spectroscopy
2.6. Wettability Test
2.7. Nanoindentation
2.8. Tribological Test
2.9. In Vitro Biodegradation Test
2.10. Compression Test
2.11. Statistical Analysis
3. Results and Discussion
3.1. Structural Characterization of the Dense Printed PLA and PLA-HA
3.1.1. Density Analysis
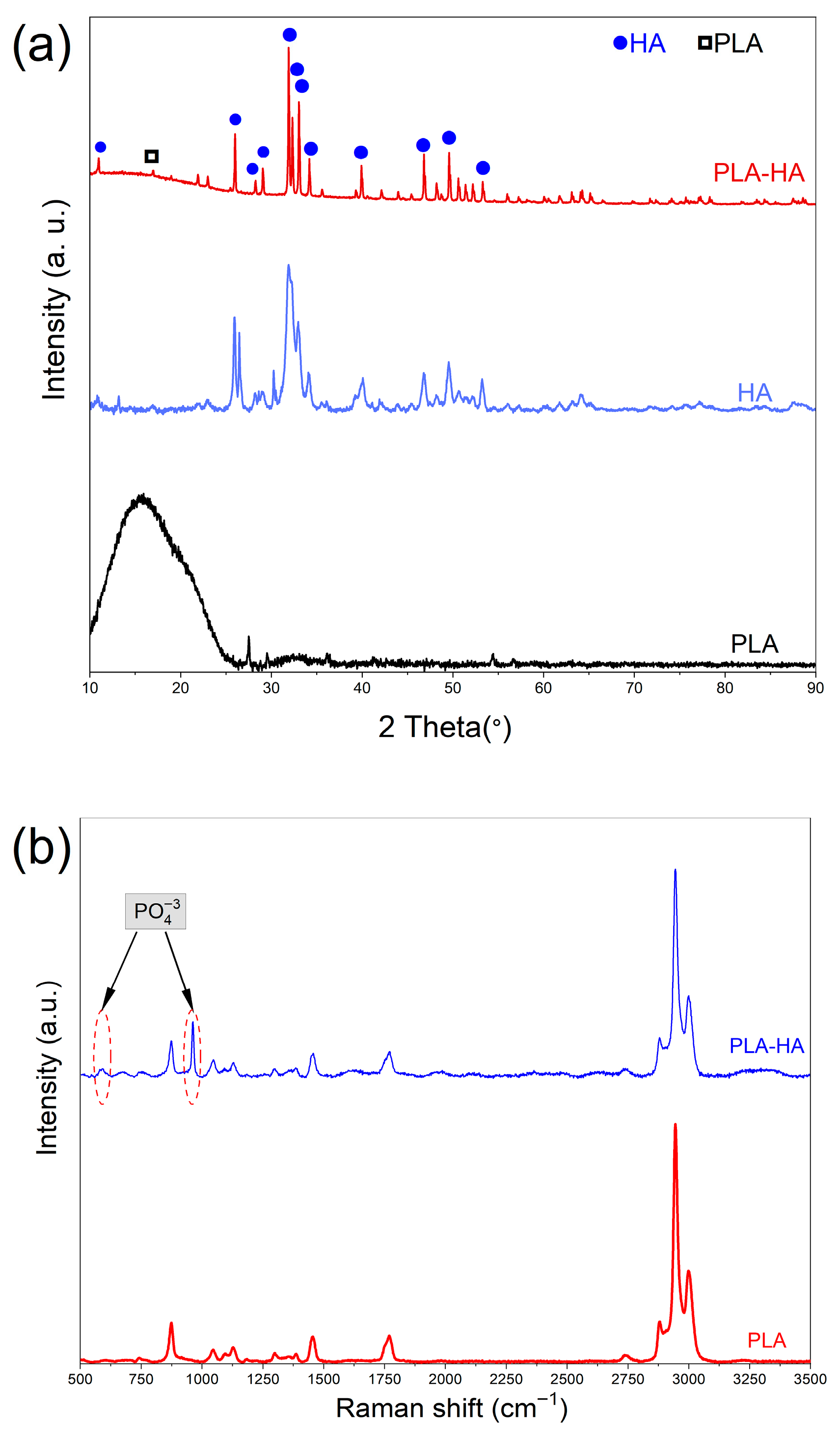
3.1.2. XRD Analysis
3.1.3. RAMAN Spectroscopy
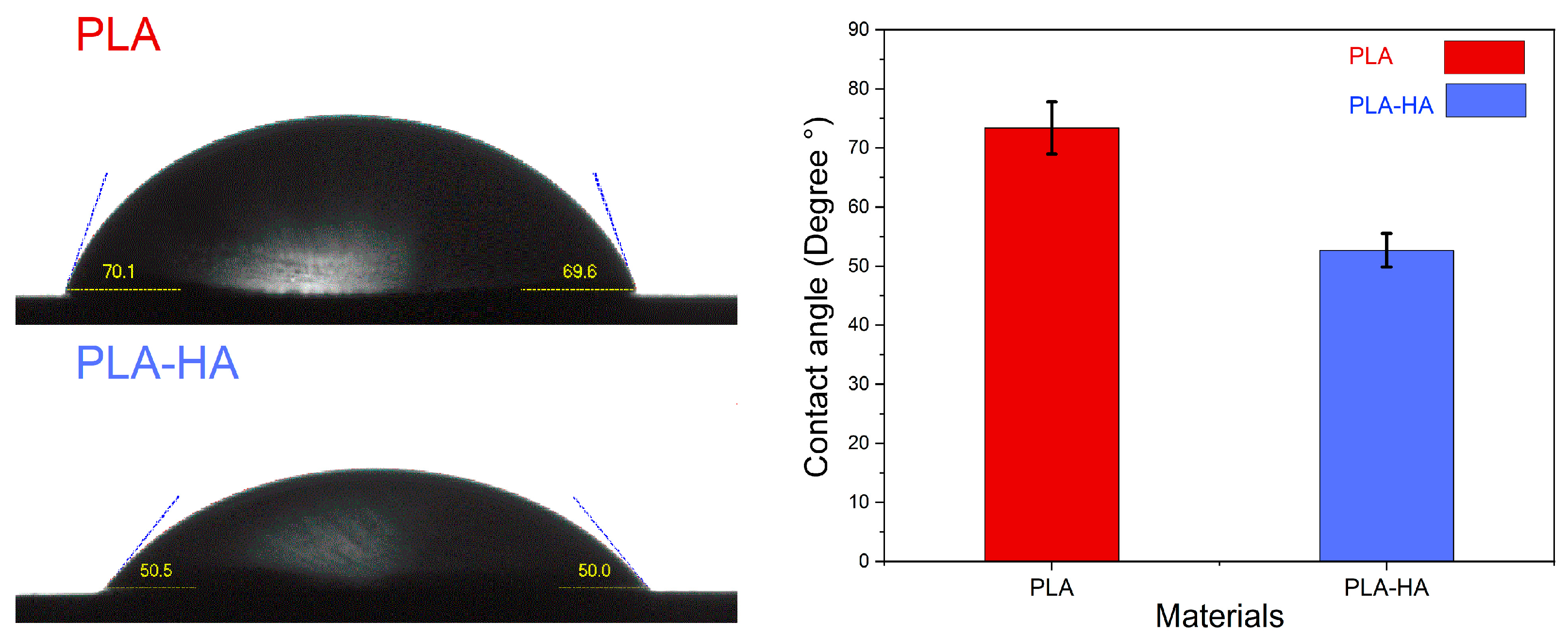
3.1.4. Wettability Measurement
3.1.5. Nanoindentation Test
3.1.6. Tribological Test
3.2. Biodegradation of the Auxetic PLA-HA Scaffold
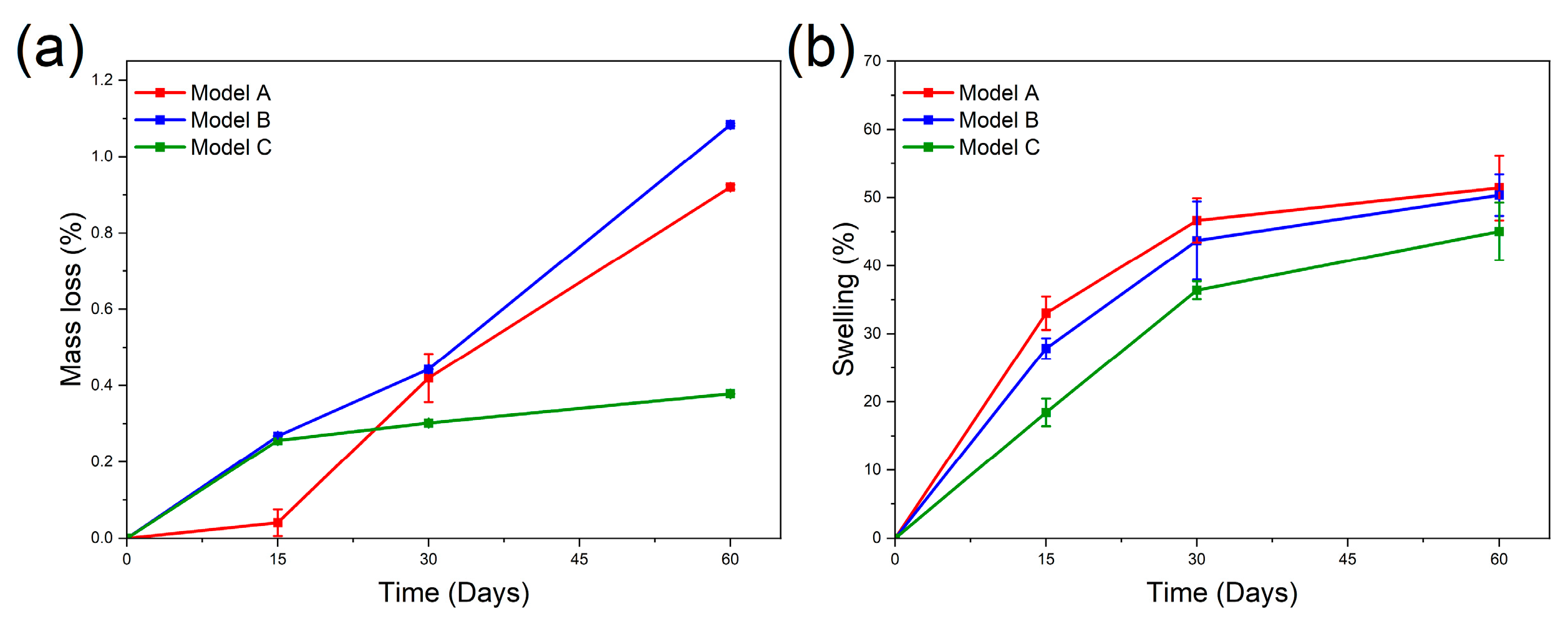
3.2.1. Mass Loss and Swelling
3.2.2. Scanning Electron Microscopy (SEM)
3.2.3. XRD Study Following In Vitro Tests
3.2.4. Hardness Following In Vitro Test
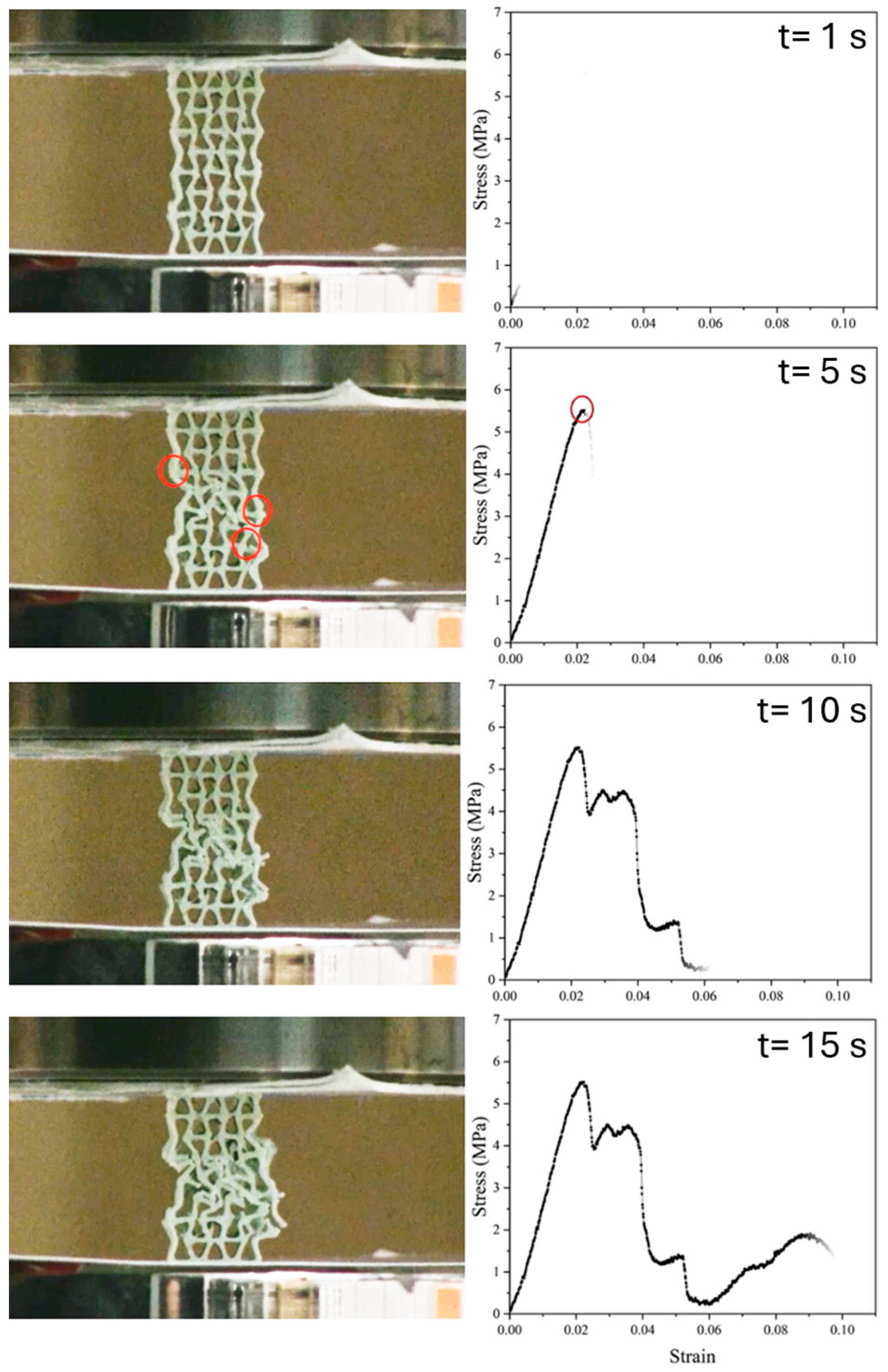
3.2.5. Compression Test After In Vitro
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Guo, Z.; Poot, A.A.; Grijpma, D.W. Advanced polymer-based composites and structures for biomedical applications. Eur. Polym. J. 2021, 149, 110388. [Google Scholar] [CrossRef]
- Festas, A.; Ramos, A.; Davim, J. Medical devices biomaterials—A review. Proc. Inst. Mech. Eng. Part L J. Mater. Des. Appl. 2020, 234, 218–228. [Google Scholar] [CrossRef]
- Sahu, M.K.; Yadav, R.; Tiwari, S.P. Recent advances in nanotechnology. Int. J. Nanomater. Nanotechnol. Nanomed. 2023, 9, 015–023. [Google Scholar] [CrossRef]
- Hu, X.; Zhao, W.; Zhang, Z.; Xie, J.; He, J.; Cao, J.; Li, Q.; Yan, Y.; Xiong, C.; Li, K. Novel 3D printed shape-memory PLLA-TMC/GA-TMC scaffolds for bone tissue engineering with the improved mechanical properties and degradability. Chin. Chem. Lett. 2023, 34, 107451. [Google Scholar] [CrossRef]
- Custodio, C.L.; Broñola, P.J.M.; Cayabyab, S.R.; Lagura, V.U.; Celorico, J.R.; Basilia, B.A. Powder Loading Effects on the Physicochemical and Mechanical Properties of 3D Printed Poly Lactic Acid/Hydroxyapatite Biocomposites. Int. J. Bioprint. 2021, 7, 326. [Google Scholar] [CrossRef]
- Subramaniyan, M.; Karuppan, S.; Helaili, S.; Ahmad, I. Structural, mechanical, and in-vitro characterization of hydroxyapatite loaded PLA composites. J. Mol. Struct. 2024, 1306, 137862. [Google Scholar] [CrossRef]
- Zhao, W.; Yue, C.; Liu, L.; Leng, J.; Liu, Y. Mechanical behavior analyses of 4D printed metamaterials structures with excellent energy absorption ability. Compos. Struct. 2023, 304, 116360. [Google Scholar] [CrossRef]
- Tajvar, S.; Hadjizadeh, A.; Samandari, S.S. Scaffold degradation in bone tissue engineering: An overview. Int. Biodeterior. Biodegrad. 2023, 180, 105599. [Google Scholar] [CrossRef]
- Bertsch, C.; Maréchal, H.; Gribova, V.; Lévy, B.; Debry, C.; Lavalle, P.; Fath, L. Biomimetic bilayered scaffolds for tissue engineering: From current design strategies to medical applications. Adv. Healthc. Mater. 2023, 12, 2203115. [Google Scholar] [CrossRef]
- Paltanea, G.; Manescu, V.; Antoniac, I.; Antoniac, A.; Nemoianu, I.V.; Robu, A.; Dura, H. A review of biomimetic and biodegradable magnetic scaffolds for bone tissue engineering and oncology. Int. J. Mol. Sci. 2023, 24, 4312. [Google Scholar] [CrossRef]
- Kalsi, S.; Singh, J.; Sehgal, S.; Sharma, N. Biomaterials for tissue engineered bone Scaffolds: A review. Mater. Today Proc. 2023, 81, 888–893. [Google Scholar] [CrossRef]
- Jeyachandran, D.; Cerruti, M. Glass, Ceramic, Polymeric, and Composite Scaffolds with Multiscale Porosity for Bone Tissue Engineering. Adv. Eng. Mater. 2023, 25, 2201743. [Google Scholar] [CrossRef]
- Ershad-Langroudi, A.; Babazadeh, N.; Alizadegan, F.; Mousaei, S.M.; Moradi, G. Polymers for implantable devices. J. Ind. Eng. Chem. 2024, 137, 61–86. [Google Scholar] [CrossRef]
- Cao, S.; Wei, Y.; Bo, R.; Yun, X.; Xu, S.; Guan, Y.; Zhao, J.; Lan, Y.; Zhang, B.; Xiong, Y. Inversely engineered biomimetic flexible network scaffolds for soft tissue regeneration. Sci. Adv. 2023, 9, eadi8606. [Google Scholar] [CrossRef]
- Biglari, N.; Zare, E.N. Conjugated polymer-based composite scaffolds for tissue engineering and regenerative medicine. Alex. Eng. J. 2024, 87, 277–299. [Google Scholar] [CrossRef]
- Wang, X.; Peng, Y.; Wu, Y.; Cao, S.; Deng, H.; Cao, Z. Chitosan/silk fibroin composite bilayer PCL nanofibrous mats for bone regeneration with enhanced antibacterial properties and improved osteogenic potential. Int. J. Biol. Macromol. 2023, 230, 123265. [Google Scholar] [CrossRef]
- Parupelli, S.K.; Saudi, S.; Bhattarai, N.; Desai, S. 3D printing of PCL-ceramic composite scaffolds for bone tissue engineering applications. Int. J. Bioprinting 2023, 9, 0196. [Google Scholar] [CrossRef]
- Shukla, S.; Behera, B. Auxetic fibrous materials and structures in medical engineering—A review. J. Text. Inst. 2023, 114, 1078–1089. [Google Scholar] [CrossRef]
- Dong, K.; Wang, Y.; Wang, Z.; Qiu, W.; Zheng, P.; Xiong, Y. Reusability and energy absorption behavior of 4D printed continuous fiber-reinforced auxetic composite structures. Compos. Part A Appl. Sci. Manuf. 2023, 169, 107529. [Google Scholar] [CrossRef]
- Wanniarachchi, C.T.; Arjunan, A.; Baroutaji, A.; Singh, M. 3D printing customised stiffness-matched meta-biomaterial with near-zero auxeticity for load-bearing tissue repair. Bioprinting 2023, 33, e00292. [Google Scholar] [CrossRef]
- Ghavidelnia, N.; Bodaghi, M.; Hedayati, R. Femur auxetic meta-implants with tuned micromotion distribution. Materials 2020, 14, 114. [Google Scholar] [CrossRef]
- Ongaro, F.; Pugno, N. Mechanical modelling of viscoelastic hierarchical suture joints and their optimisation and auxeticity. Mech. Mater. 2023, 186, 104785. [Google Scholar] [CrossRef]
- Vellaparambil, R.; Han, W.-S.; Di Giovanni, P.; Avril, S. Computational Comparison of the Mechanical Behavior of Aortic Stent-Grafts Derived from Auxetic Unit Cells. Cardiovasc. Eng. Technol. 2023, 15, 199–210. [Google Scholar] [CrossRef]
- Dudek, K.K.; Martínez, J.A.I.; Ulliac, G.; Kadic, M. Micro-scale auxetic hierarchical mechanical metamaterials for shape morphing. Adv. Mater. 2022, 34, 2110115. [Google Scholar] [CrossRef]
- Shirzad, M.; Zolfagharian, A.; Bodaghi, M.; Nam, S.Y. Auxetic metamaterials for bone-implanted medical devices: Recent advances and new perspectives. Eur. J. Mech.-A/Solids 2023, 98, 104905. [Google Scholar] [CrossRef]
- Urquhart, L.; Tamburrino, F.; Neri, P.; Wodehouse, A.; Fingland, C.; Razionale, A.V. An examination of auxetic componentry for applications in human-centred biomedical product design settings. Int. J. Interact. Des. Manuf. 2023, 18, 5457–5467. [Google Scholar] [CrossRef]
- Ikram, H.; Al Rashid, A.; Koç, M. Additive manufacturing of smart polymeric composites: Literature review and future perspectives. Polym. Compos. 2022, 43, 6355–6380. [Google Scholar] [CrossRef]
- Zhong, R.; Ren, X.; Zhang, X.Y.; Luo, C.; Zhang, Y.; Xie, Y.M.J.C.; Materials, B. Mechanical properties of concrete composites with auxetic single and layered honeycomb structures. Constr. Build. Mater. 2022, 322, 126453. [Google Scholar] [CrossRef]
- Chen, Z.; Li, J.; Wu, B.; Chen, X.; Xie, Y.M.J.C.S. Enhanced mechanical properties of re-entrant auxetic honeycomb with self-similar inclusion. Compos. Struct. 2024, 331, 117921. [Google Scholar] [CrossRef]
- Li, K.; Zhang, Y.; Hou, Y.; Su, L.; Zeng, G.; Xu, X.J.T.-W.S. Mechanical properties of re-entrant anti-chiral auxetic metamaterial under the in-plane compression. Thin-Walled Struct. 2023, 184, 110465. [Google Scholar] [CrossRef]
- He, P.; Wang, S.; Zhang, M.; Sang, L.; Tong, L.; Hou, W. Compression performance of 3D-printed thermoplastic auxetic structures. Thin-Walled Struct. 2024, 197, 111558. [Google Scholar] [CrossRef]
- Chikkanna, N.; Krishnapillai, S.; Ramachandran, V. In-plane and out-of-plane quasi-static compression performance enhancement of 3D printed re-entrant diamond auxetic metamaterial with geometrical tuning and fiber reinforcement. Def. Technol. 2023, 25, 1–17. [Google Scholar] [CrossRef]
- Sanchez, O.; Besharatloo, H.; Yus, J.; Sanchez-Herencia, A.; Ferrari, B. Material thermal extrusion of conductive 3D electrodes using highly loaded graphene and graphite colloidal feedstock. Addit. Manuf. 2023, 72, 103643. [Google Scholar] [CrossRef]
- Ortega-Columbrans, P.; Ferrandez-Montero, A.; Yus, J.; Sanchez-Herencia, A.; Ferrari, B. Processing of membranes and 3D scaffolds based on n-TiO2 colloidally dispersed on a thermoplastic matrix for photocatalytic pollutant removal. Catalist Today 2024, 426, 114371. [Google Scholar] [CrossRef]
- Alonzo, M.; Primo, F.A.; Kumar, S.A.; Mudloff, J.A.; Dominguez, E.; Fregoso, G.; Ortiz, N.; Weiss, W.M.; Joddar, B. Bone tissue engineering techniques, advances, and scaffolds for treatment of bone defects. Curr. Opin. Biomed. Eng. 2021, 17, 100248. [Google Scholar] [CrossRef]
- Singhvi, M.S.; Zinjarde, S.S.; Gokhale, D.V. Polylactic acid: Synthesis and biomedical applications. J. Appl. Microbiol. 2019, 127, 1612–1626. [Google Scholar] [CrossRef]
- Liu, S.; Qin, S.; He, M.; Zhou, D.; Qin, Q.; Wang, H. Current applications of poly (lactic acid) composites in tissue engineering and drug delivery. Compos. Part B Eng. 2020, 199, 108238. [Google Scholar] [CrossRef]
- Premphet, P.; Leksakul, K.; Boonyawan, D.; Vichiansan, N. Process parameters optimization and mechanical properties of 3D PLA/HA printing scaffold. Mater. Today Proc. 2023. [Google Scholar] [CrossRef]
- Negrescu, A.-M.; Mocanu, A.-C.; Miculescu, F.; Mitran, V.; Constantinescu, A.-E.; Cimpean, A. In Vitro Studies on 3D-Printed PLA/HA/GNP Structures for Bone Tissue Regeneration. Biomimetics 2024, 9, 55. [Google Scholar] [CrossRef]
- de Andrade, J.C.; Cabral, F.; Clemens, F.J.; Vieira, J.L.; Soares, M.B.; Hotza, D.; Fredel, M.C. Effect of stearic acid on the mechanical and rheological properties of PLA/HA biocomposites. Mater. Today Commun. 2023, 35, 106357. [Google Scholar] [CrossRef]
- Namasivayam Sukumaar, V.; Arjunan, S.; Pandiaraj, L.N.; Narayanan, A.J.J.o.T.C.M. Experimental investigation of 3D printed polylactic acid and polylactic acid–hydroxyapatite composite through material extrusion technique for biomedical application. J. Thermoplast. Compos. Mater. 2024, 38, 08927057241255883. [Google Scholar] [CrossRef]
- Alarifi, I.M. Evaluation of a design for a three-dimensional-printed artificial bone structure. Polym. Compos. 2023, 44, 8240–8252. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, B.; Li, M.; Li, J.; Zhang, C.; Han, Y.; Wang, L.; Wang, K.; Zhou, C.; Liu, L. 3D printing of PLA/n-HA composite scaffolds with customized mechanical properties and biological functions for bone tissue engineering. Compos. Part B Eng. 2021, 224, 109192. [Google Scholar] [CrossRef]
- Ferrández-Montero, A.; Ortega-Columbrans, P.; Eguiluz, A.; Sanchez-Herencia, A.J.; Detsch, R.; Boccaccini, A.R.; Ferrari, B. Biocompatible colloidal feedstock for material extrusion processing of bioceramic-based scaffolds. Polym. Compos. 2024, 45, 7237–7255. [Google Scholar] [CrossRef]
- Shan, E.; Chamorro, C.; Ferrández-Montero, A.; Martin-Rodriguez, R.M.; Ferrari, B.; Sanchez-Herencia, A.J.; Virto, L.; Marín, M.J.; Figuero, E.; Sanz, M. In Vitro Biological Properties Assessment of 3D-Printed Hydroxyapatite–Polylactic Acid Scaffolds Intended for Bone Regeneration. J. Funct. Biomater. 2025, 16, 218. [Google Scholar] [CrossRef] [PubMed]
- Madji, S.; Belmedani, M.; Mekatel, E.; Trari, M. Co-precipitation synthesis and characterization of the sillenite Bi12CoO20: Application to photoactivity for water treatment on the heterojunction with ZnO. J. Mater. Sci. Mater. Electron. 2023, 34, 2093. [Google Scholar] [CrossRef]
- Yang, R.; Dong, X.; Chen, G.; Lin, F.; Huang, Z.; Manzo, M.; Mao, H. Novel Terahertz Spectroscopy Technology for Crystallinity and Crystal Structure Analysis of Cellulose. Polymers 2021, 13, 6. [Google Scholar] [CrossRef]
- Alam, M.K.; Hossain, M.S.; Bahadur, N.M.; Ahmed, S. A comparative study in estimating of crystallite sizes of synthesized and natural hydroxyapatites using Scherrer Method, Williamson-Hall model, Size-Strain Plot and Halder-Wagner Method. J. Mol. Struct. 2024, 1306, 137820. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Scalera, F.; Gervaso, F.; Montagna, F.; Sannino, A.; Maffezzoli, A. One-step solvent-free process for the fabrication of high loaded PLA/HA composite filament for 3D printing. J. Therm. Anal. 2018, 134, 575–582. [Google Scholar] [CrossRef]
- ASTM G99; Standard Test Method for Wear Testing with a Pin-on-Disk Apparatus. ASTM International: West Conshohocken, PA, USA, 2017.
- ASTM F1635; Standard Test Method for In Vitro Degradation Testing of Hydrolytically Degradable Polymer Resins and Fabricated Forms for Surgical Implants. ASTM International: West Conshohocken, PA, USA, 2016.
- ASTM D695; Standard Test Method for Compressive Properties of Rigid Plastics. ASTM International: West Conshohocken, PA, USA, 2015.
- ASTM E384-22; Standard Test Method for Microindentation Hardness of Materials. ASTM International: West Conshohocken, PA, USA, 2022.
- Marzuki, A.P.; Nasir, M.A.M.; Salleh, F.M.; Tharazi, I.; Ismail, M.H.; Murat, B.I.S. Rheological properties of hydroxyapatite (HAp)/poly-lactic acid (PLA) composite at different weight percentages in producing 3D printing filament. Proc. Mech. Eng. Res. Day 2022, 2022, 43–45. [Google Scholar]
- Tanase-Opedal, M.; Espinosa, E.; Rodríguez, A.; Chinga-Carrasco, G. Lignin: A Biopolymer from Forestry Biomass for Biocomposites and 3D Printing. Materials 2019, 12, 3006. [Google Scholar] [CrossRef]
- Chieng, B.W.; Ibrahim, N.; Yunus, W.; Hussein, M.; Then, Y.Y.; Loo, Y. Effects of Graphene Nanoplatelets and Reduced Graphene Oxide on Poly(lactic acid) and Plasticized Poly(lactic acid): A Comparative Study. Polymers 2014, 6, 2232–2246. [Google Scholar] [CrossRef]
- Pérez-Davila, S.; Garrido-Gulías, N.; González-Rodríguez, L.; López-Álvarez, M.; Serra, J.; López-Periago, J.E.; González, P. Physicochemical Properties of 3D-Printed Polylactic Acid/Hydroxyapatite Scaffolds. Polymers 2023, 15, 2849. [Google Scholar] [CrossRef]
- Yazdani, A.; Höhne, G.W.H.; Misture, S.T.; Graeve, O.A. A method to quantify crystallinity in amorphous metal alloys: A differential scanning calorimetry study. PLoS ONE 2020, 15, e0234774. [Google Scholar] [CrossRef] [PubMed]
- Pandele, A.M.; Constantinescu, A.; Radu, I.C.; Miculescu, F.; Ioan Voicu, S.; Ciocan, L.T. Synthesis and characterization of pla-micro-structured hydroxyapatite composite films. Materials 2020, 13, 274. [Google Scholar] [CrossRef]
- Fontana-Escartín, A.; Simon, N.; Bertran, O.; Contini, A.; Torras, J.; Alemán, C. Plasma-Treated Poly(Lactic Acid): Deciphering the Structure of a Versatile Engineering Material. ACS Omega 2025, 10, 61915–61926. [Google Scholar] [CrossRef]
- Nguyen, N.M.; Kakarla, A.B.; Nukala, S.G.; Kong, C.; Baji, A.; Kong, I. Evaluation of physicochemical properties of a hydroxyapatite polymer nanocomposite for use in fused filament fabrication. Polymers 2023, 15, 3980. [Google Scholar] [CrossRef] [PubMed]
- Rodovalho, A.J.R.L.; Barbosa, W.T.; Vieira, J.L.; Oliva, C.A.d.; Gonçalves, A.P.B.; Cardoso, P.d.S.M.; Modolon, H.B.; Montedo, O.R.K.; Arcaro, S.; Hodel, K.V.S.; et al. Influence of size and crystallinity of nanohydroxyapatite (nHA) particles on the properties of Polylactic Acid/nHA nanocomposite scaffolds produced by 3D printing. J. Mater. Res. Technol. 2024, 30, 3101–3111. [Google Scholar] [CrossRef]
- Bernardo, M.P.; da Silva, B.C.R.; Hamouda, A.E.I.; de Toledo, M.A.S.; Schalla, C.; Rütten, S.; Goetzke, R.; Mattoso, L.H.C.; Zenke, M.; Sechi, A. PLA/Hydroxyapatite scaffolds exhibit in vitro immunological inertness and promote robust osteogenic differentiation of human mesenchymal stem cells without osteogenic stimuli. Sci. Rep. 2022, 12, 2333. [Google Scholar] [CrossRef]
- Wijerathne, H.; Yan, D.; Zeng, B.; Xie, Y.; Hu, H.; Wickramaratne, M.; Han, Y. Effect of nano-hydroxyapatite on protein adsorption and cell adhesion of poly(lactic acid)/nano-hydroxyapatite composite microspheres. SN Appl. Sci. 2020, 2, 722. [Google Scholar] [CrossRef]
- Jaganathan, S.; Kandasamy, R.; Venkatachalam, R.; Gunalan, M.; Dhairiyasamy, R. Advances in Optimizing Mechanical Performance of 3D-Printed Polymer Composites: A Microstructural and Processing Enhancements Review. Adv. Polym. Technol. 2024, 2024, 3168252. [Google Scholar] [CrossRef]
- Grigora, M.-E.; Terzopoulou, Z.; Baciu, D.; Steriotis, T.; Charalambopoulou, G.; Gounari, E.; Bikiaris, D.N.; Tzetzis, D. 3D printed poly(lactic acid)-based nanocomposite scaffolds with bioactive coatings for tissue engineering applications. J. Mater. Sci. 2023, 58, 2740–2763. [Google Scholar] [CrossRef]
- Majzoobi, G.H.; Rahmani, K.; Mohammadi, M.; Bakhtiari, H.; Das, R. Tribological behaviour of Ti/HA and Ti/SiO2 functionally graded materials fabricated at different strain rates. Biotribology 2023, 35–36, 100233. [Google Scholar] [CrossRef]
- Rachaiah, B.; Tamalapura Puttaswamy, J.; Nagaraju, S.B.; Vedavathi, D. Investigation on the wear characteristics of 3D printed graphene-reinforced PLA composites. Discov. Mater. 2024, 4, 75. [Google Scholar] [CrossRef]
- Hu, Y.; Xia, D.; Shen, H.; Nan, J.; Ma, N.; Guo, Z.; Wang, X.; Jin, Q. Cold sintering constructed in situ drug-loaded high strength HA-PLA composites: Potential bone substitution material. Ceram. Int. 2023, 49, 11655–11663. [Google Scholar] [CrossRef]
- Jiang, F.; Yang, S.; Qi, C.; Liu, H.-T. Two plateau characteristics of re-entrant auxetic honeycomb along concave direction. Thin-Walled Struct. 2022, 179, 109665. [Google Scholar] [CrossRef]
- Wolf, M.H.; Gil-Castell, O.; Cea, J.; Carrasco, J.C.; Ribes-Greus, A. Degradation of Plasticised Poly(lactide) Composites with Nanofibrillated Cellulose in Different Hydrothermal Environments. J. Polym. Environ. 2023, 31, 2055–2072. [Google Scholar] [CrossRef]
- Limsukon, W.; Rubino, M.; Rabnawaz, M.; Lim, L.-T.; Auras, R. Hydrolytic degradation of poly(lactic acid): Unraveling correlations between temperature and the three phase structures. Polym. Degrad. Stab. 2023, 217, 110537. [Google Scholar] [CrossRef]
- Vaid, R.; Yildirim, E.; Pasquinelli, M.A.; King, M.W. Hydrolytic Degradation of Polylactic Acid Fibers as a Function of pH and Exposure Time. Molecules 2021, 26, 7554. [Google Scholar] [CrossRef] [PubMed]
- Kasprzak, M.; Szabłowska, A.; Kurzyk, A.; Tymowicz-Grzyb, P.; Najmrodzki, A.; Woźniak, A.; Antosik, A.; Pagacz, J.; Szterner, P.; Plichta, A.; et al. Effects of Sterilization and Hydrolytic Degradation on the Structure, Morphology and Compressive Strength of Polylactide-Hydroxyapatite Composites. Int. J. Mol. Sci. 2022, 23, 10454. [Google Scholar] [CrossRef]
- Nazir, F.; Iqbal, M. Comparative Study of Crystallization, Mechanical Properties, and In Vitro Cytotoxicity of Nanocomposites at Low Filler Loadings of Hydroxyapatite for Bone-Tissue Engineering Based on Poly(l-lactic acid)/Cyclo Olefin Copolymer. Polymers 2021, 13, 3865. [Google Scholar] [CrossRef] [PubMed]
- Bauer, L.; Rogina, A.; Ivanković, M.; Ivanković, H. Medical-Grade Poly(Lactic Acid)/Hydroxyapatite Composite Films: Thermal and In Vitro Degradation Properties. Polymers 2023, 15, 1512. [Google Scholar] [CrossRef] [PubMed]
- Sardar, M.Z.; Gide, K.M.; Stuart, A.; Rostobaya, V.; Zarrabinia, K.; Beheshti, A.; Bagheri, S. Evaluating mixing methods for FFF-printed PLA-HA composites: Towards high-performance composites and advancements in additive manufacturing. Int. J. Adv. Manuf. Technol. 2024, 136, 1267–1280. [Google Scholar] [CrossRef]
- Available online: https://zenodo.org/records/19800236 (accessed on 26 April 2026).











| Designation | Length, L (mm) | Width, W (mm) | Angle θ (°) | Wall Thickness, T (mm) |
|---|---|---|---|---|
| A | 3.00 | 1.78 | 70 | 0.4 |
| B | 4.00 | 2.31 | 70 | 0.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Benziada, M.A.; Sanchez-Herencia, A.J.; Daoud, I.; Besharatloo, H.; Ferrari, B.; Miroud, D.; Ferrandez-Montero, A. Design and Characterization of 3D Printed Auxetic PLA-HA Composite Scaffolds for Biomedical Application. Materials 2026, 19, 1972. https://doi.org/10.3390/ma19101972
Benziada MA, Sanchez-Herencia AJ, Daoud I, Besharatloo H, Ferrari B, Miroud D, Ferrandez-Montero A. Design and Characterization of 3D Printed Auxetic PLA-HA Composite Scaffolds for Biomedical Application. Materials. 2026; 19(10):1972. https://doi.org/10.3390/ma19101972
Chicago/Turabian StyleBenziada, Mohammed Amine, Antonio Javier Sanchez-Herencia, Isamil Daoud, Hossein Besharatloo, Begoña Ferrari, Djamel Miroud, and Ana Ferrandez-Montero. 2026. "Design and Characterization of 3D Printed Auxetic PLA-HA Composite Scaffolds for Biomedical Application" Materials 19, no. 10: 1972. https://doi.org/10.3390/ma19101972
APA StyleBenziada, M. A., Sanchez-Herencia, A. J., Daoud, I., Besharatloo, H., Ferrari, B., Miroud, D., & Ferrandez-Montero, A. (2026). Design and Characterization of 3D Printed Auxetic PLA-HA Composite Scaffolds for Biomedical Application. Materials, 19(10), 1972. https://doi.org/10.3390/ma19101972











