Influence of Magnetorheological Finishing on Surface Topography and Functional Performance of Shoulder Joint Cap Surface
Abstract
1. Introduction
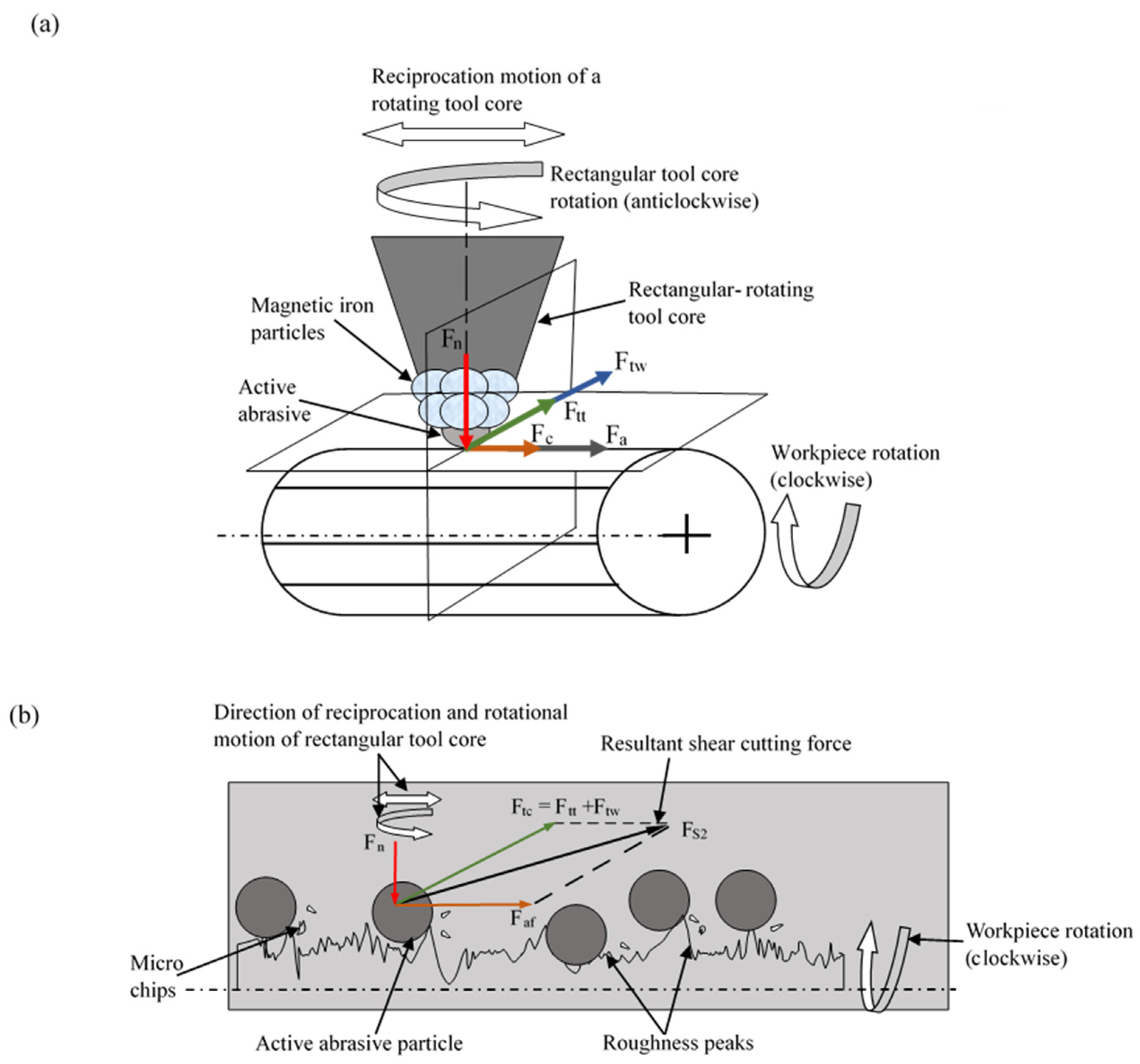
2. Materials and Methods
3. Results and Discussion
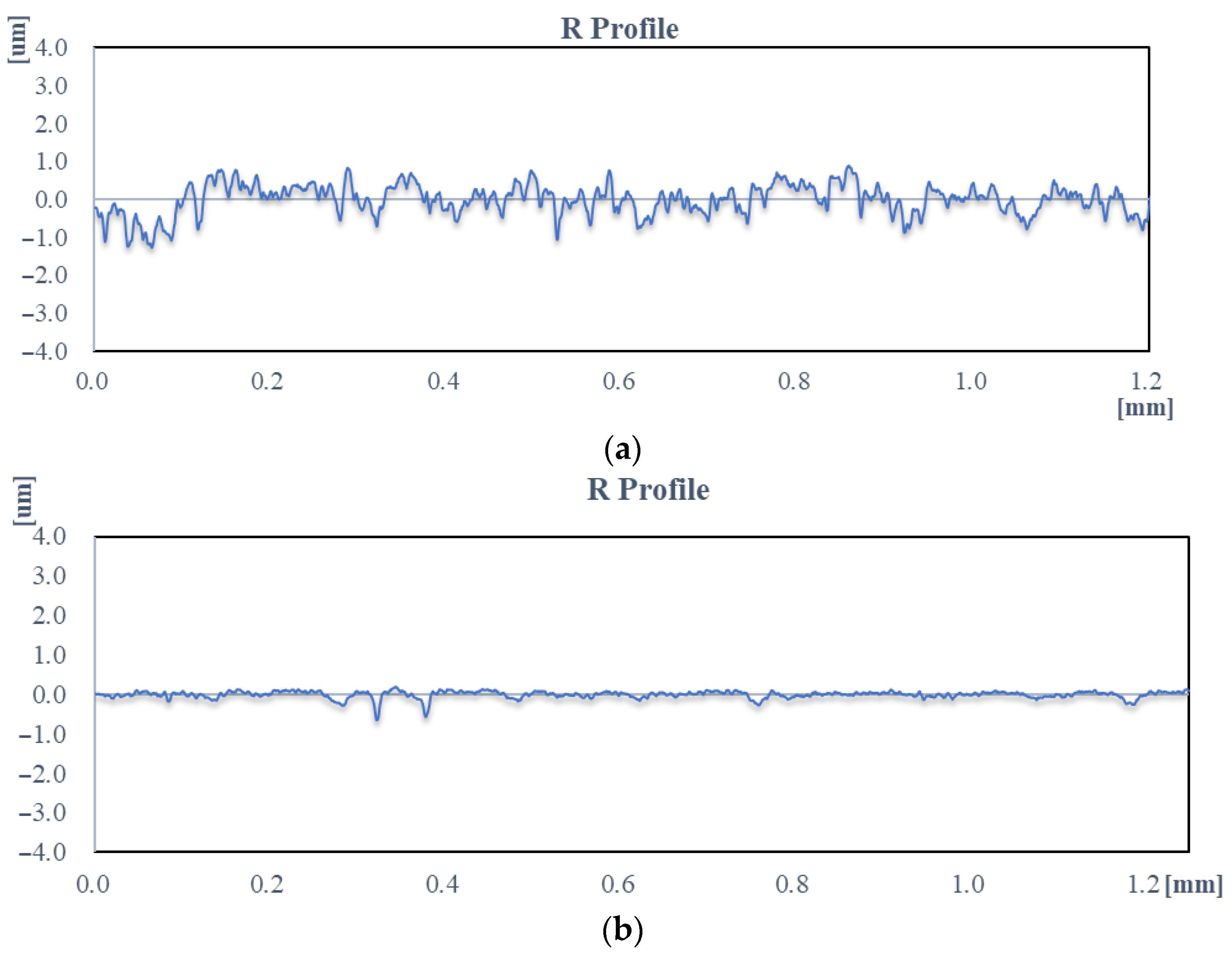
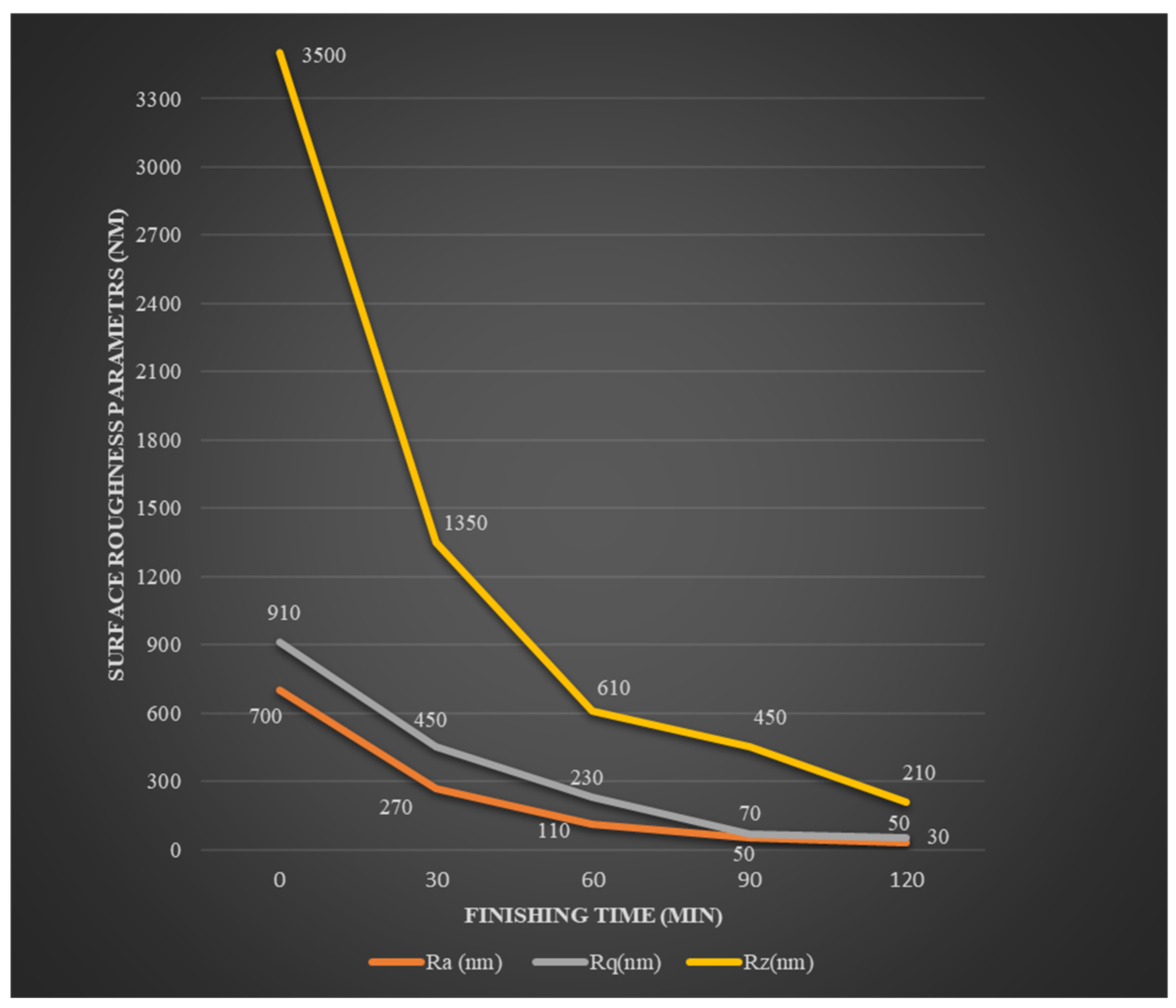
3.1. Surface Roughness Analysis and Nano-Finishing Effectiveness
3.2. Microhardness Enhancement and Its Impact on Wear Resistance
3.3. Tribological Performance: Reduction in Coefficient of Friction and Wear Rate
3.4. Surface Morphology and Topographical Analysis
3.5. Statistical Validation and Process Optimization
- Magnetic field strength: 0.4 T;
- Tool rotational speed: 1800 rpm;
- Workpiece speed: 110 rpm;
- Finishing duration: 120 min.
4. Conclusions
- Magnetorheological finishing (MRF) resulted in significant surface quality improvement, reducing the surface roughness value from 0.35 µm to 0.03 µm, attaining a roughness reduction percentage of 91.4, which is necessary for biomedical applications.
- An SEM study confirmed that the machining marks, surface asperities, and grooves were removed, thus obtaining a defect-free and mirror-like surface, imparting improved biocompatibility to implants.
- The microhardness of the shoulder joint caps was improved by magnetorheological finishing from HV 510 to HV 560, thereby augmenting surface strength and wear resistance, which are critical for the durability of implants.
- Tribological performance was enhanced significantly, with a decrease in coefficient of friction from 0.12 to 0.06, resulting in reduced wear and improved lubrication under physiological conditions.
- MRF implants had a wear rate 38% lower than that of untreated ones, thus endorsing the importance of nano-finishing in enhancing implant longevity and minimizing material degradation.
- According to the statistical analysis using ANOVA, the process parameters showed a strong correlation to the finishing results, with an R2 value of 0.96, which is an indication of good process repeatability and reliability.
- The optimized MRF parameters (magnetic field strength of 0.4 T, tool speed of 1800 rpm, workpiece speed of 110 rpm, and finishing time of 120 min) gave the best results in terms of surface finish and mechanical improvements, thus making the process well suited for biomedical applications.
- MRF offered significantly better results over traditional finishing methods such as grinding and electropolishing in terms of surface smoothness, wear resistance, and hardness enhancement, thereby casting it as a next-generation finishing technique for orthopedic implants.
- This study establishes that MRF is an adoptable and scalable surface finishing technology that contributes to more durable implants, fewer post-surgical complications, and improved patient outcomes, thus ensuring better performance in biomedical applications.
- Future research directions will focus on extending the applicability of MRF in biomedical domains by exploring the use of magnetic nanomaterials for enhanced abrasive control and responsiveness. Further studies will include biological testing, such as osteoblast adhesion, cytocompatibility, and bacterial resistance, to validate the in vivo performance of MRF-treated implants. Additionally, the integration of AI-driven process optimization and toolpath generation will be investigated to adapt MRF techniques for patient-specific implant geometries. These steps will further strengthen the clinical relevance and customization potential of MRF as a precision surface finishing technology for orthopedic applications.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Goldsmith, A.A.J.; Dowson, D.; Issac, G.H.; Lancester, J.G. A comparative joint simulator study of the wear of metal-on-metal and alternative material combinations in hip replacements. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2000, 214, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh, H.R.S.; Eajazi, A.; Shahi, A.S. The bearing surfaces in total hip arthroplasty-options, material characteristics and selection. In Recent Advances in Arthroplasty; Fokter, S.K., Ed.; IntechOpen: London, UK, 2012. [Google Scholar] [CrossRef][Green Version]
- ASTM F2083-11; Standard Specifications For Total Knee Prosthesis. ASTM International: West Conshohocken, PA, USA, 2013; pp. 1–9.
- Dalury, D.F.; Pomeroy, D.L.; Gorab, R.S.; Adams, M.J. Why are total knee arthroplasties being revised? J. Arthroplast. 2013, 28, 120–121. [Google Scholar] [CrossRef] [PubMed]
- Blunt, L.; Charlton, P.; Beaucamp, A.; Jiang, X. The application of optics polishing to free form knee implants. In Proceedings of the 6th Euspen International Conference, Baden, Austria, 28 May–1 June 2006; pp. 1–4. [Google Scholar]
- Brecher, C.; Tuecks, R.; Zunke, R.; Wenzel, C. Development of a force controlled orbital polishing head for free form surface finishing. Prod. Eng. Res. Dev. 2010, 4, 269–277. [Google Scholar] [CrossRef]
- Cheung, C.F.; Ho, L.T.; Charlton, P.; Kong, L.B.; To, S.; Lee, W.B. Analysis of surface generation in the ultra precision polishing of freeform surfaces. Proc. Inst. Mech. Eng. Part B J. Eng. Manuf. 2010, 224, 59–73. [Google Scholar] [CrossRef]
- Loveless, T.R.; Williams, R.E.; Rajurkar, K.P. A study of the effects of abrasive-flow finishing on various machined surfaces. J. Mater. Process. Technol. 1994, 47, 133–151. [Google Scholar] [CrossRef]
- Tzeng, H.J.; Van, B.H.; Hsu, R.T.; Lin, Y.C. Self-modulating abrasive medium and its application to abrasive flow machining for finishing micro channel surfaces. Int. J. Adv. Manuf. Technol. 2007, 32, 1163–1169. [Google Scholar] [CrossRef]
- Rhoades, L.J.; Kohut, T.A.; Nokovich, N.P. Unidirectional Abrasive Flow Machining. U.S. Patent 5,367,833, 29 November 1994. [Google Scholar]
- Li, J.; Liu, W.; Yang, L.; Tian, C.; Zhang, S. A method of motion control about micro-hole abrasive flow machining based on Delphi language. In Proceeding of the International Conference on Mechatronics and Automation, Changchun, China, 18 September 2009; pp. 9–12. [Google Scholar]
- Walia, R.S.; Shan, H.S.; Kumar, P. Parametric optimization of centrifugal force-assisted abrasive flow machining (CFAAFM) by the Taguchi method. Mater. Manuf. Process. 2006, 21, 375–382. [Google Scholar] [CrossRef]
- Sarkar, M.; Jain, V.K. Nanofinishing of freeform surfaces using abrasive flow finishing process. Proc. Inst. Mech. Eng. Part B J. Eng. Manuf. 2015, 231, 1501–1515. [Google Scholar] [CrossRef]
- Subramanian, K.T.; Balashanmugam, N.; Kumar, P.V.S. Nanometric finishing on biomedical implants by abrasive flow finishing. J. Inst. Eng. Ser. C 2016, 97, 55–61. [Google Scholar] [CrossRef]
- Singh, S.; Sankar, M.R.; Jain, V.K. Simulation and experimental investigations into abrasive flow nanofinishing of surgical steel tubes. Mach. Sci. Technol. 2018, 22, 454–475. [Google Scholar] [CrossRef]
- Singh, S.; Sankar, M.R. Development of polymer abrasive medium for nanofinishing of microholes on surgical stainless steel using abrasive flow machining process. Proc. Inst. Mech. Eng. Part B J. Eng. Manuf. 2019, 234, 355–370. [Google Scholar] [CrossRef]
- Singh, M.; Singh, A.; Singh, A.K. A Rotating Core Based Magnetorheological Nano-Finishing Process for External Cylindrical Surfaces. Mater. Manuf. Process. 2018, 33, 1160–1168. [Google Scholar] [CrossRef]
- Singh, M.; Singh, A.K. Improved Magnetorheological Finishing Process with Rectangular Core Tip for External Cylindrical Surfaces. Mater. Manuf. Process. 2019, 34, 1049–1061. [Google Scholar] [CrossRef]
- Singh, M.; Singh, A.K. Performance Investigation of Magnetorheological Finishing of Rolls in Cold Rolling Process. J. Manuf. Process. 2019, 41, 315–329. [Google Scholar] [CrossRef]
- Singh, M.; Singh, A.K. Magnetorheological Finishing of Micro-Punches for Enhanced Performance of Micro-Extrusion Process. Mater. Manuf. Process. 2019, 34, 1646–1657. [Google Scholar] [CrossRef]
- Singh, M.; Singh, A.K. Magnetorheological Finishing of Grooved Drum Surface and Its Performance Analysis in Winding Process. Int. J. Adv. Manuf. Technol. 2020, 106, 2921–2937. [Google Scholar] [CrossRef]
- Singh, M.; Singh, A.K. Theoretical Investigations into Magnetorheological Finishing of External Cylindrical Surfaces for Improved Performance. Proc. Inst. Mech. Eng. Part C J. Process Mech. Eng. 2020, 234, 4872–4892. [Google Scholar] [CrossRef]
- Singh, M.; Singh, A.K. Magnetorheological Finishing of Copper Cylindrical Roller for Its Improved Performance in Printing Machine. Proc. Inst. Mech. Eng. Part E J. Process Mech. Eng. 2020, 235, 103–115. [Google Scholar] [CrossRef]
- Singh, M.; Singh, G.; Jayant, A. Optimization of Turning Parameters of Titanium Chrome-Moly (Ti-Cr-Mo) Alloy Using Taguchi Method. Indian J. Eng. Mater. Sci. (IJEMS) 2021, 27, 776–782. [Google Scholar]
- Park, B.J.; Fang, F.F.; Choi, H.J. Magnetorheology: Materials and application. Soft. Matter 2010, 6, 5246–5253. [Google Scholar] [CrossRef]
- Lokander, M.; Stenberg, B. Performance of isotropic magnetorheological rubber materials. Polym. Test. 2003, 22, 245–251. [Google Scholar] [CrossRef]
- Kubík, M.; Válek, J.; Žáček, J.; Jeniš, F.; Borin, D.; Strecker, Z.; Mazůrek, I. Transient response of magnetorheological fluid on rapid change of magnetic field in shear mode. Sci. Rep. 2022, 12, 10612. [Google Scholar] [CrossRef] [PubMed]
- Ginder, J.M.; Nichols, M.E.; Elie, L.D.; Clark, S.M. Controllable-Stiffness Components Based on Magnetorheological Elastomers. In Proceedings of the SPIE’s 7th Annual International Symposium on Smart Structures and Materials, Newport Beach, CA, USA, 6–9 March 2000. [Google Scholar]
- Kikuchi, T.; Noma, J.; Akaiwa, S.; Ueshima, Y. Response time of magnetorheological fluid–based haptic device. J. Intell. Mater. Syst. Struct. 2016, 27, 859–865. [Google Scholar] [CrossRef]
- Chen, D.W.; Yu, H.H.; Luo, L.J.; Rajesh Kumar, S.; Chen, C.H.; Lin, T.Y.; Jessie Lue, S. Osteoblast biocompatibility and antibacterial effects using 2-methacryloyloxyethyl phosphocholine-grafted stainless-steel composite for implant applications. Nanomaterials 2019, 9, 939. [Google Scholar] [CrossRef] [PubMed]
- Stepanov, G.V.; Abramchuk, S.S.; Grishin, D.A.; Nikitin, L.V.; Kramarenko, E.Y.; Khokhlov, A.R. Effect of a homogeneous magnetic field on the viscoelastic behavior of magnetic elastomers. Polymer 2007, 48, 488–495. [Google Scholar] [CrossRef]
- Kankanala, S.V.; Triantafyllidis, N. On finitely strained magnetorheological elastomers. J. Mech. Phys. Solids 2004, 52, 2869–2908. [Google Scholar] [CrossRef]
- Wei, Y.; Lv, J.; Tang, Z.; Yang, L.; Wu, M.; Zhao, T.; Yin, H. A universal rheological constitutive equation of magnetorheological fluids with a wide shear rate range. J. Magn. Magn. Mater. 2022, 563, 169811. [Google Scholar] [CrossRef]
- Zhang, H.; Hu, Z.; Lei, Y.; Wang, D.; Zhao, H.; Jiang, H. Enhanced performances of magnetorheological fluids based on weakly magnetic organogel. J. Magn. Magn. Mater. 2022, 560, 169572. [Google Scholar] [CrossRef]
- Odenbach, S. Microstructure and rheology of magnetic hybrid materials. Arch. Appl. Mech. 2016, 86, 269–279. [Google Scholar] [CrossRef]
- Yu, M.; Ju, B. Dynamic mechanical properties testing for shear mode of magnetorheological elastomer. Gongneng Cailiao/J. Funct. Mater. 2011, 42, 1939–1942+1946. [Google Scholar]
- Naji, F.A.A.; Murtaza, Q.; Khaled, N.I.; Nasr, M.M. Future perspectives and research trends in advanced chemo-mechanical magneto-rheological finishing for enhanced surface quality. Multiscale Multidiscip. Model. Exp. Des. 2025, 8, 42. [Google Scholar] [CrossRef]
- Kumar, S.; Nasna, P.; Ghosh, G. Recent advancement in biocompatible materials, hybrid bioactive coating, surface modification and post-processing techniques for the fabrication of biomedical implant: Critical review and future prospects. Proc. Inst. Mech. Eng. Part C J. Mech. Eng. Sci. 2025, 239, 3247–3286. [Google Scholar] [CrossRef]
- Tien, D.H.; Thoa, P.T.T.; Van Que, N.; Duy, T.N. Developing material removal rate from polishing force modeling via magnetorheological finishing using an improved Halbach array with a slider crank mechanism for Ti-6Al-4V alloy. Mater. Today Commun. 2025, 45, 112360. [Google Scholar] [CrossRef]
- Wu, Y.; Wan, K.; Lu, J.; Yuan, C.; Cui, Y.; Duan, R.; Yu, J. Research Progress on Surface Modification of Titanium Implants. Coatings 2025, 15, 229. [Google Scholar] [CrossRef]
- Witkowska, J.; Sobiecki, J.; Wierzchoń, T. Advancements in Surface Modification of NiTi Alloys for Orthopedic Implants: Focus on Low-Temperature Glow Discharge Plasma Oxidation Techniques. Int. J. Mol. Sci. 2025, 26, 1132. [Google Scholar] [CrossRef] [PubMed]
- Winkler, R.; Ciria, M.; Ahmad, M.; Plank, H.; Marcuello, C. A Review of the Current State of Magnetic Force Microscopy to Unravel the Magnetic Properties of Nanomaterials Applied in Biological Systems and Future Directions for Quantum Technologies. Nanomaterials 2023, 13, 2585. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Lu, Y.; Xu, J.; Cui, Z.; Yang, H. Magnetic Levitation Actuation and Motion Control System with Active Levitation Mode Based on Force Imbalance. Appl. Sci. 2023, 13, 740. [Google Scholar] [CrossRef]




| Element | Composition (%) |
|---|---|
| Cobalt (Co) | 63 |
| Chromium (Cr) | 25 |
| Molybdenum (Mo) | 5.6 |
| Nickel (Ni) | 3.2 |
| Iron (Fe) | 2.2 |
| Carbon (C) | 0.1 |
| Manganese (Mn) | 0.46 |
| Silicon (Si) | 0.44 |
| S. No | Parameters | Units | Range | ||||
|---|---|---|---|---|---|---|---|
| 1 | Rotational speed of tool core tip (S) | rpm | 800 | 1300 | 1800 | 2300 | 2800 |
| 2 | Rotational speed of workpiece (W) | rpm | 50 | 80 | 110 | 140 | 170 |
| 3 | Feed rate of tool core tip (R) | cm/min | 10 | 20 | 30 | 40 | 50 |
| 4 | Current (I) | A | 1 | 1.5 | 2 | 2.5 | 3 |
| Source | SS | DF | MS | F Value | p Value | %Contribution | |
|---|---|---|---|---|---|---|---|
| Model | 9084 | 11 | 825.81 | 87.91 | <0.0001 | Significant | |
| S | 1457.79 | 1 | 1457.79 | 139.33 | <0.0001 | 16.07 | |
| W | 856.62 | 1 | 856.62 | 91.42 | <0.0001 | 9.43 | |
| R | 361.54 | 1 | 361.54 | 32.16 | 0.0021 | 3.98 | |
| I | 2431.78 | 1 | 2431.78 | 244.19 | <0.0001 | 26.77 | |
| T2 | 396.97 | 1 | 396.97 | 51.39 | 0.0011 | 4.37 | |
| W2 | 428.67 | 1 | 428.67 | 55.52 | 0.0009 | 4.72 | |
| R2 | 1143.67 | 1 | 1143.67 | 123.47 | <0.0001 | 12.59 | |
| I2 | 1697.80 | 1 | 1697.80 | 159.72 | <0.0001 | 18.69 | |
| TW | 457.83 | 1 | 457.83 | 62.81 | 0.0007 | 5.04 | |
| TR | 218.92 | 1 | 218.92 | 28.36 | 0.0049 | 2.41 | |
| RI | 269.79 | 1 | 269.79 | 29.81 | 0.0062 | 2.97 | |
| Residual | 220.08 | 18 | 6.88 | ||||
| Lack of fit | 130.53 | 18 | 5.68 | 0.57 | 0.8666 | Not significant | |
| Pure error | 89.56 | 5 | 9.95 | ||||
| Total | 9524.17 | 29 |
| Finishing Technique | Achievable Surface Roughness (Ra, µm) | Microhardness Improvement (HV) | Coefficient of Friction (CoF) | Process Control and Uniformity | Suitability for Complex Geometries | Biocompatibility Preservation |
|---|---|---|---|---|---|---|
| MRF (This study) | 0.03 | ↑ from 510 to 560 | ↓ from 0.12 to 0.06 | Excellent (magnetically controlled fluid dynamics) | Excellent (customizable for joint cap shape) | High (no phase change, no subsurface damage) |
| Magnetic Abrasive Finishing (MAF) | 0.08–0.12 | ↑ marginal (varies) | ↓ from 0.14 to 0.09 | Moderate (abrasive dispersion, not uniform) | Good for flat/cylindrical parts | Moderate (thermal and mechanical stresses may occur) |
| RMAF (Rotational MAF) | 0.06–0.10 | ↑ slight to moderate | ↓ to ~0.08 | Better than MAF but needs complex setups | Good (for cylindrical/symmetric shapes) | Moderate |
| BEMRF (Ball-End MRF) | 0.05–0.07 | ↑ up to ~10% | ↓ to ~0.07 | High (localized control) | Very good (localized finishing for cavities) | High |
| Conventional Lapping | 0.10–0.20 | No improvement | 0.10–0.15 | Low (non-uniform material removal) | Poor (not suitable for complex shapes) | Low (residual stress risk) |
| Mechanical Polishing | ~0.15 | No significant change | 0.12–0.16 | Low | Poor | Low |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, M.; Singh, G.; Abu-Malouh, R.; Chauhan, S.; Vashishtha, G. Influence of Magnetorheological Finishing on Surface Topography and Functional Performance of Shoulder Joint Cap Surface. Materials 2025, 18, 3397. https://doi.org/10.3390/ma18143397
Singh M, Singh G, Abu-Malouh R, Chauhan S, Vashishtha G. Influence of Magnetorheological Finishing on Surface Topography and Functional Performance of Shoulder Joint Cap Surface. Materials. 2025; 18(14):3397. https://doi.org/10.3390/ma18143397
Chicago/Turabian StyleSingh, Manpreet, Gagandeep Singh, Riyad Abu-Malouh, Sumika Chauhan, and Govind Vashishtha. 2025. "Influence of Magnetorheological Finishing on Surface Topography and Functional Performance of Shoulder Joint Cap Surface" Materials 18, no. 14: 3397. https://doi.org/10.3390/ma18143397
APA StyleSingh, M., Singh, G., Abu-Malouh, R., Chauhan, S., & Vashishtha, G. (2025). Influence of Magnetorheological Finishing on Surface Topography and Functional Performance of Shoulder Joint Cap Surface. Materials, 18(14), 3397. https://doi.org/10.3390/ma18143397








