Evaluation of Drug-Loading Ability of Poly(Lactic Acid)/Hydroxyapatite Core–Shell Particles
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
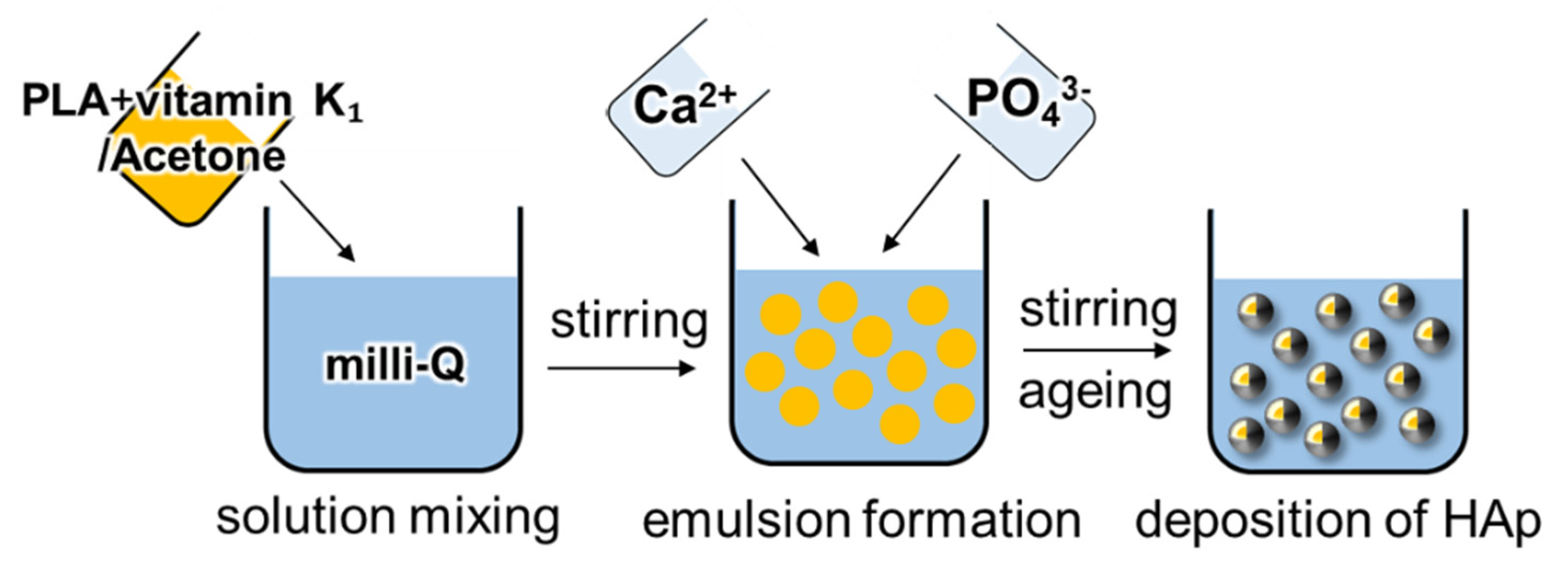
2.2. Preparation of Vitamin K1-Loaded PLA/HAp Core–Shell Particles
2.3. Characterization of the Vitamin K1-Loaded PLA/HAp Core–Shell Particles
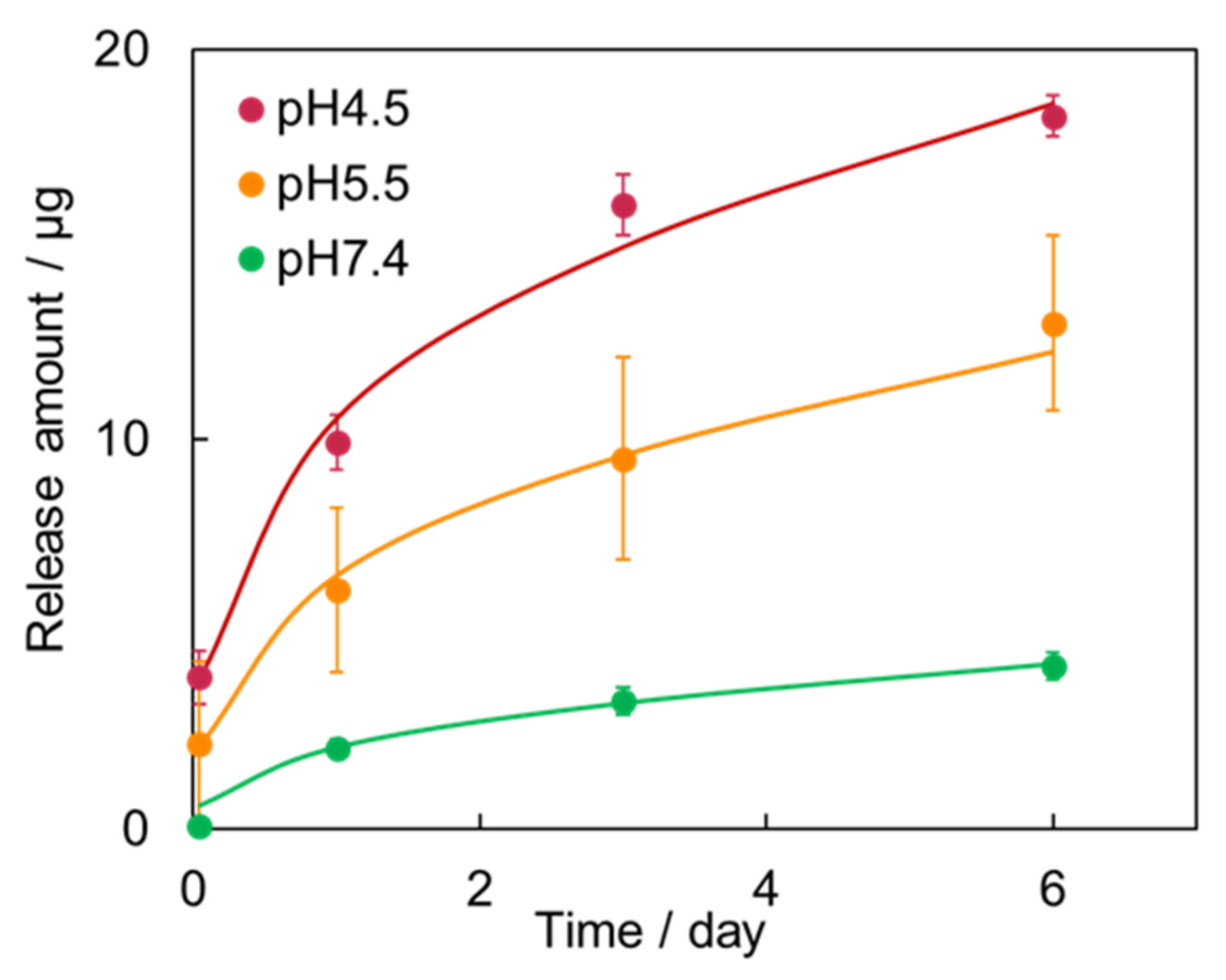
2.4. Drug-Release Test of the Vitamin K1-Loaded PLA/HAp Core–Shell Particles
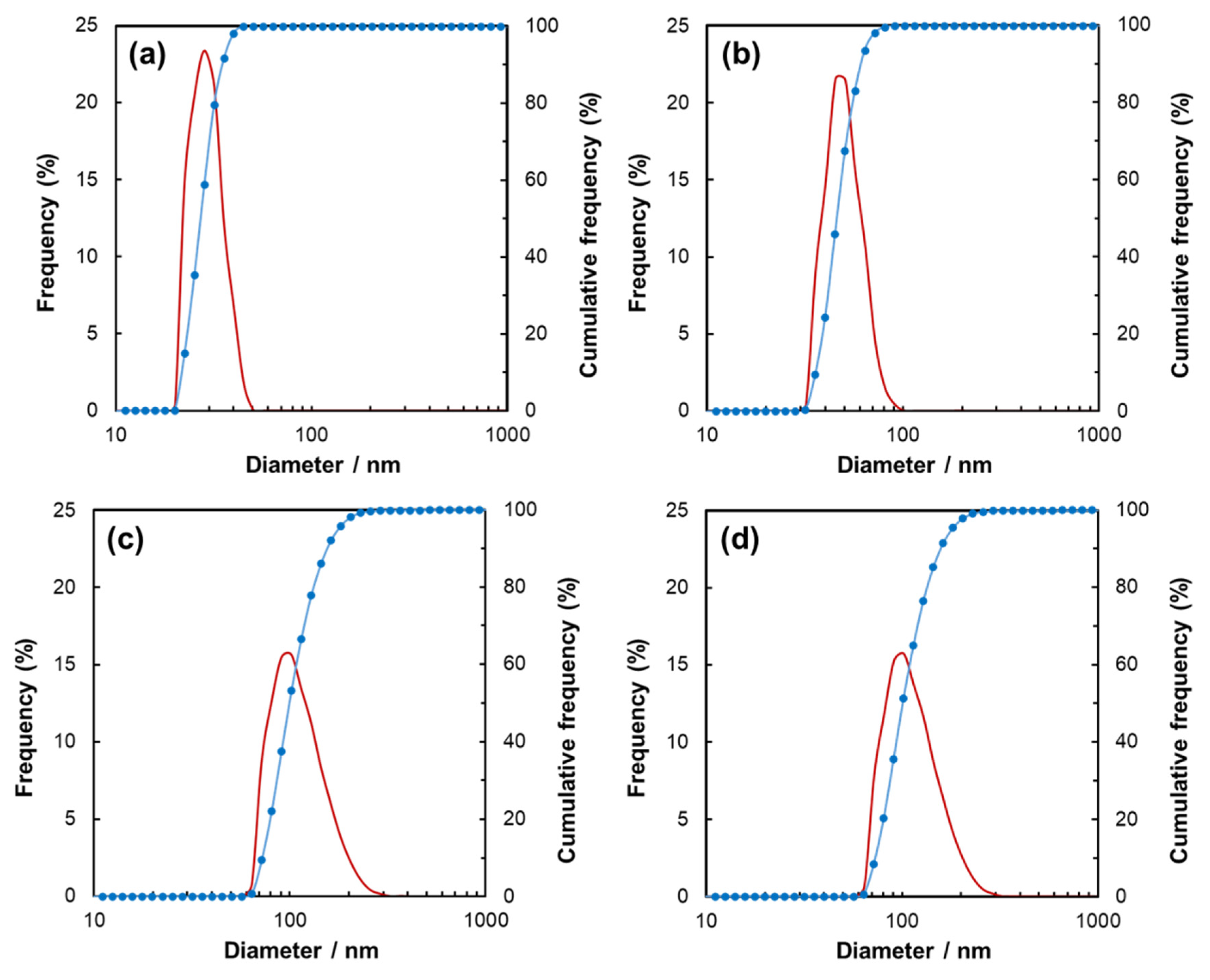
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ju, Y.; Guo, H.; Edman, M.; Hamm-Alvarez, S.F. Application of advances in endocytosis and membrane trafficking to drug delivery. Adv. Drug Del. Rev. 2020, 157, 118–141. [Google Scholar] [CrossRef]
- Blume, G.; Cevc, G. Liposomes for the sustained drug release in vivo. Biochim. Biophys. Acta 1990, 1029, 91–97. [Google Scholar] [CrossRef]
- Nagasaki, Y.; Okada, T.; Scholz, C.; Iijima, M.; Kato, M.; Kataoka, K. The Reactive Polymeric Micelle Based on An Aldehyde-Ended Poly(ethylene glycol)/Poly(lactide) Block Copolymer. Macromolecules 1998, 31, 1473–1479. [Google Scholar] [CrossRef]
- Peltonen, L.; Aitta, J.; Hyvönen, S.; Karjalainen, M.; Hirvonen, J. Improved entrapment efficiency of hydrophilic drug substance during nanoprecipitation of poly (I) lactide nanoparticles. AAPS PharmSciTech 2009, 5, 115. [Google Scholar] [CrossRef]
- Ogawa, Y.; Okada, H.; Yamamoto, Y.; Shimamoto, T. In Vivo Release Profiles of Leuprolide Acetate from Microcapsules Prepared with Polylactic Acids or Copoly (Lactic/Glycolic) Acids and In Vivo Degradation of These Polymers. Chem. Pharm. Bull. 1988, 36, 2576–2581. [Google Scholar] [CrossRef] [PubMed]
- Nagai, Y.; Lee, J.J.; Yamane, H. Hydrolytic degradation of low molecular weigh poly(lactic acid)s and their drug eluting behavior. J. Soc. Mater. Sci. Jpn. 2011, 60, 2–7. [Google Scholar] [CrossRef]
- Fredenberg, S.; Wahlgren, M.; Reslow, M.; Axelsson, A. The mechanisms of drug release in poly(lactic-co-glycolic acid)-based drug delivery systems—A review. Int. J. Pharm. 2011, 415, 34–52. [Google Scholar] [CrossRef]
- Butoescu, N.; Seemayer, C.A.; Foti, M.; Jordan, O.; Doelker, E. Dexamethasone-containing PLGA superparamagnetic microparticles as carriers for the local treatment of arthritis. Biomaterials 2009, 30, 1772–1780. [Google Scholar] [CrossRef]
- Patil, S.D.; Papadmitrakopoulos, F.; Burgess, D.J. Concurrent delivery of dexamethasone and VEGF for localized inflammation control and angiogenesis. J. Control. Release 2007, 117, 68–79. [Google Scholar] [CrossRef]
- Torchilin, V. Tumor delivery of macromolecular drugs based on the EPR effect. Adv. Drug Del. Rev. 2011, 63, 131–135. [Google Scholar] [CrossRef]
- Matsumura, Y.; Maeda, H. A new concept for macromolecular therapeutics in cancer chemotherapy: Mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986, 46, 6387. [Google Scholar] [PubMed]
- Maeda, H. SMANCS and polymer-conjugated macromolecular drugs: Advantages in cancer chemotherapy. Adv. Drug Del. Rev. 1991, 6, 181–202. [Google Scholar] [CrossRef]
- Hollis, C.P.; Weiss, H.L.; Leggas, M.; Evers, B.M.; Gemeinhart, R.A.; Li, T. Biodistribution and bioimaging studies of hybrid paclitaxel nanocrystals: Lessons learned of the EPR effect and image-guided drug delivery. J. Control. Release 2013, 172, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Nakamura, H.; Maeda, H. The EPR effect: Unique features of tumor blood vessels for drug delivery, factors involved, and limitations and augmentation of the effect. Adv. Drug Del. Rev. 2011, 63, 136–151. [Google Scholar] [CrossRef] [PubMed]
- Oyane, A.; Tsurushima, H.; Sogo, Y.; Ito, A.; Mutsuzaki, H. Development of apatite based biomaterials utilizing, biologically functional molecules. New Glass 2009, 24, 35–42. [Google Scholar]
- Kim, H.-W.; Knowles, J.C.; Kim, H.-E. Hydroxyapatite/poly(ε-caprolactone) composite coatings on hydroxyapatite porous bone scaffold for drug delivery. Biomaterials 2004, 25, 1279–1287. [Google Scholar] [CrossRef]
- dos Apostolos, R.C.R.; Andrade, G.F.; da Silva, W.M.; de Assis Gomes, D.; de Miranda, M.C.; de Sousa, E.M.B. Hybrid polymeric systems of mesoporous silica/hydroxyapatite nanoparticles applied as antitumor drug delivery platform. Int. J. Appl. Ceram. Technol. 2019, 16, 1836–1849. [Google Scholar] [CrossRef]
- Kundu, B.; Ghosh, D.; Sinha, M.K.; Sen, P.S.; Balla, V.K.; Das, N.; Basu, D. Doxorubicin-intercalated nano-hydroxyapatite drug-delivery system for liver cancer: An animal model. Ceram. Int. 2013, 39, 9557–9566. [Google Scholar] [CrossRef]
- Sun, W.; Fan, J.L.; Wang, S.Z.; Kang, Y.; Du, J.J.; Peng, X.J. Biodegradable Drug-Loaded Hydroxyapatite Nanotherapeutic Agent for Targeted Drug Release in Tumors. ACS Appl. Mater. Interfaces 2018, 10, 7832–7840. [Google Scholar] [CrossRef]
- Verma, G.; Shetake, N.G.; Pandrekar, S.; Pandey, B.N.; Hassan, P.A.; Priyadarsini, K.I. Development of surface functionalized hydroxyapatite nanoparticles for enhanced specificity towards tumor cells. Eur. J. Pharm. Sci. 2020, 144, 105206. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, Y.; Wada, K.; Nakatani, M.; Yamada, S.; Onoue, S. Formulation design for poorly water-soluble drugs based on biopharmaceutics classification system: Basic approaches and practical applications. Int. J. Pharm. 2011, 420, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Yan, J.; Mei, H.; Cai, S.; Li, S.; Cheng, F.; Cao, J.; He, B. High-drug-loading capacity of redox-activated biodegradable nanoplatform for active targeted delivery of chemotherapeutic drugs. Regen. Biomater. 2020, 7, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Su, T.; Li, S.; Lai, Y.; He, B.; Gu, Z. Polymeric micelles with π–π conjugated moiety on glycerol dendrimer as lipophilic segments for anticancer drug delivery. Biomater. Sci. 2014, 2, 775–783. [Google Scholar] [CrossRef]
- Cheng, F.; Guan, X.; Cao, H.; Su, T.; Cao, J.; Chen, Y.; Cai, M.; He, B.; Gu, Z.; Luo, X. Characteristic of core materials in polymeric micelles effect on their micellar properties studied by experimental and dpd simulation methods. Int. J. Pharm. 2015, 492, 152–160. [Google Scholar] [CrossRef]
- Peng, Z.; Li, S.; Han, X.; Al-Youbi, A.O.; Bashammakh, A.S.; El-Shahawi, M.S.; Leblanc, R.M. Determination of the composition, encapsulation efficiency and loading capacity in protein drug delivery systems using circular dichroism spectroscopy. Anal. Chim. Acta 2016, 937, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Batrakova, E.V.; Dorodnych, T.Y.; Klinskii, E.Y.; Kliushnenkova, E.N.; Shemchukova, O.B.; Goncharova, O.N.; Arjakov, S.A.; Alakhov, V.Y.; Kabanov, A.V. Anthracycline antibiotics non-covalently incorporated into the block copolymer micelles: In vivo evaluation of anti-cancer activity. Br. J. Cancer 1996, 74, 1545–1552. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Chen, H.; Zeng, D.; Tian, Y.; Chen, F.; Feng, J.; Shi, J. Core/Shell Structured Hollow Mesoporous Nanocapsules: A Potential Platform for Simultaneous Cell Imaging and Anticancer Drug Delivery. ACS Nano 2010, 4, 6001–6013. [Google Scholar] [CrossRef]
- Danhier, F.; Ansorena, E.; Silva, J.M.; Coco, R.; Le Breton, A.; Préat, V. PLGA-based nanoparticles: An overview of biomedical applications. J. Control. Release 2012, 161, 505–522. [Google Scholar] [CrossRef]
- Rao, J.P.; Geckeler, K.E. Polymer nanoparticles: Preparation techniques and size-control parameters. Prog. Polym. Sci. 2011, 36, 887–913. [Google Scholar] [CrossRef]
- Nagata, F.; Miyajima, T.; Kato, K. Preparation of phylloquinone-loaded poly(lactic acid)/hydroxyapatite core–shell particles and their drug release behavior. Adv. Powder Technol. 2016, 27, 903–907. [Google Scholar] [CrossRef]
- Hanasaki, M.; Nagata, F.; Miyajima, T.; Kato, K. Controlling particle size of poly(lactic acid)/hydroxyapatite nanoparticles. Trans. Mat. Res. Soc. Jpn. 2018, 43, 135–138. [Google Scholar] [CrossRef]
- Lee, S.; Miyajima, T.; Sugawara-Narutaki, A.; Kato, K.; Nagata, F. Development of paclitaxel-loaded poly(lactic acid)/hydroxyapatite core–shell nanoparticles as a stimuli-responsive drug delivery system. R. Soc. Open Sci. 2021, 8. [Google Scholar] [CrossRef]
- Sato, K.; Kogure, T.; Kumagai, Y.; Tanaka, J. Crystal Orientation of Hydroxyapatite Induced by Ordered Carboxyl Groups. J. Colloid Interface Sci. 2001, 240, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Tanizawa, Y.; Sawamura, K.; Suzuki, T. Inhibition of hydroxyapatite formation and growth by condensed phosphate. Chem. Soc. Jpn. 1989, 1989, 1706–1711. [Google Scholar] [CrossRef]
- Rehman, I.; Bonfield, W. Characterization of hydroxyapatite and carbonated apatite by photo acoustic FTIR spectroscopy. J. Mater. Sci. Mater. Med. 1997, 8, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Maçon, A.L.B.; Lee, S.; Poologasundarampillai, G.; Kasuga, T.; Jones, J.R. Synthesis and dissolution behaviour of CaO/SrO-containing sol–gel-derived 58S glasses. J. Mater. Sci. 2017, 52, 8858–8870. [Google Scholar] [CrossRef]
- Lee, S.; Nakano, T.; Kasuga, T. Structure, dissolution behavior, cytocompatibility, and antibacterial activity of silver-containing calcium phosphate invert glasses. J. Biomed. Mater. Res. A 2017, 105, 3127–3135. [Google Scholar] [CrossRef] [PubMed]
- Kister, G.; Cassanas, G.; Vert, M. Effects of morphology, conformation and configuration on the IR and Raman spectra of various poly(lactic acid)s. Polymer 1998, 39, 267–273. [Google Scholar] [CrossRef]
- Breton, J.; Burie, J.R.; Berthomieu, C.; Berger, G.; Nabedryk, E. The binding sites of quinones in photosynthetic bacterial reaction centers investigated by light-induced FTIR difference spectroscopy: Assignment of the QA vibrations in Rhodobacter sphaeroides using 18O- or 13C-labeled ubiquinone and vitamin K1. Biochemistry 1994, 33, 4953–4965. [Google Scholar] [CrossRef] [PubMed]
- Kokubo, T.; Kushitani, H.; Sakka, S.; Kitsugi, T.; Yamamuro, T. Solutions able to reproduce in vivo surface-structure changes in bioactive glass-ceramic A-W. J. Biomed. Mater. Res. 1990, 24, 721–734. [Google Scholar] [CrossRef] [PubMed]
- Kuśnieruk, S.; Wojnarowicz, J.; Chodara, A.; Chudoba, T.; Gierlotka, S.; Lojkowski, W. Influence of hydrothermal synthesis parameters on the properties of hydroxyapatite nanoparticles. Beilstein J. Nanotechnol. 2016, 7, 1586–1601. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-B.; Chen, A.-Z.; Weng, L.-J.; Chen, M.-Y.; Xie, X.-L. Effect of Drug-loading Methods on Drug Load, Encapsulation Efficiency and Release Properties of Alginate/Poly-L-Arginine/Chitosan Ternary Complex Microcapsules. Macromol. Biosci. 2004, 4, 27–30. [Google Scholar] [CrossRef]
- El-Say, K.M. Maximizing the encapsulation efficiency and the bioavailability of controlled-release cetirizine microspheres using Draper-Lin small composite design. Drug Des. Dev. Ther. 2016, 10. [Google Scholar] [CrossRef] [PubMed]
- Al-Amin, M.D.; Bellato, F.; Mastrotto, F.; Garofalo, M.; Malfanti, A.; Salmaso, S.; Caliceti, P. Dexamethasone Loaded Liposomes by Thin-Film Hydration and Microfluidic Procedures: Formulation Challenges. Int. J. Mol. Sci. 2020, 21, 1611. [Google Scholar] [CrossRef] [PubMed]
- Brown, W.E.; Patel, P.R.; Chow, L.C. Formation of CaHPO4 2H2O from Enamel Mineral and Its Relationship to Caries Mechanism. J. Dent. Res. 1975, 54, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Juhász, Á.; Ungor, D.; Berta, K.; Seres, L.; Csapó, E. Spreadsheet-based nonlinear analysis of in vitro release properties of a model drug from colloidal carriers. J. Mol. Liq. 2021, 328, 115405. [Google Scholar] [CrossRef]



| Sample Code | Addition of Phosphate Ion | After Aging (72 h) | |
|---|---|---|---|
| Before | 1 h after | ||
| PLHA-V0 | 7.61 | 6.81 | 6.19 |
| PLHA-V50 | 7.67 | 6.86 | 6.23 |
| PLHA-V100 | 7.61 | 6.80 | 6.13 |
| PLHA-V200 | 7.70 | 6.88 | 6.35 |
| Sample | HAp (%) | Organic Component (%) |
|---|---|---|
| PLHA-V0 | 50.3 | 49.7 |
| PLHA-V50 | 27.8 | 72.2 |
| PLHA-V100 | 24.9 | 75.1 |
| PLHA-V200 | 15.5 | 84.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suzuki, S.; Lee, S.; Miyajima, T.; Kato, K.; Sugawara-Narutaki, A.; Sakurai, M.; Nagata, F. Evaluation of Drug-Loading Ability of Poly(Lactic Acid)/Hydroxyapatite Core–Shell Particles. Materials 2021, 14, 1959. https://doi.org/10.3390/ma14081959
Suzuki S, Lee S, Miyajima T, Kato K, Sugawara-Narutaki A, Sakurai M, Nagata F. Evaluation of Drug-Loading Ability of Poly(Lactic Acid)/Hydroxyapatite Core–Shell Particles. Materials. 2021; 14(8):1959. https://doi.org/10.3390/ma14081959
Chicago/Turabian StyleSuzuki, Seiya, Sungho Lee, Tatsuya Miyajima, Katsuya Kato, Ayae Sugawara-Narutaki, Makoto Sakurai, and Fukue Nagata. 2021. "Evaluation of Drug-Loading Ability of Poly(Lactic Acid)/Hydroxyapatite Core–Shell Particles" Materials 14, no. 8: 1959. https://doi.org/10.3390/ma14081959
APA StyleSuzuki, S., Lee, S., Miyajima, T., Kato, K., Sugawara-Narutaki, A., Sakurai, M., & Nagata, F. (2021). Evaluation of Drug-Loading Ability of Poly(Lactic Acid)/Hydroxyapatite Core–Shell Particles. Materials, 14(8), 1959. https://doi.org/10.3390/ma14081959







