Impact of Different Epoxidation Approaches of Tall Oil Fatty Acids on Rigid Polyurethane Foam Thermal Insulation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Epoxidation of TOFA with Ion Exchange Resin Amberlite IR-120 H
2.3. Epoxidation of TOFA with Novozym® 435
2.4. Synthesis of Polyols Using Two Different ETOFA
2.5. Characterisation of Products and Precursors
2.6. Rigid PU foam Preparation and Characterisation
3. Results and Discussion
3.1. Characteristics of Synthesised High Functionality Polyols
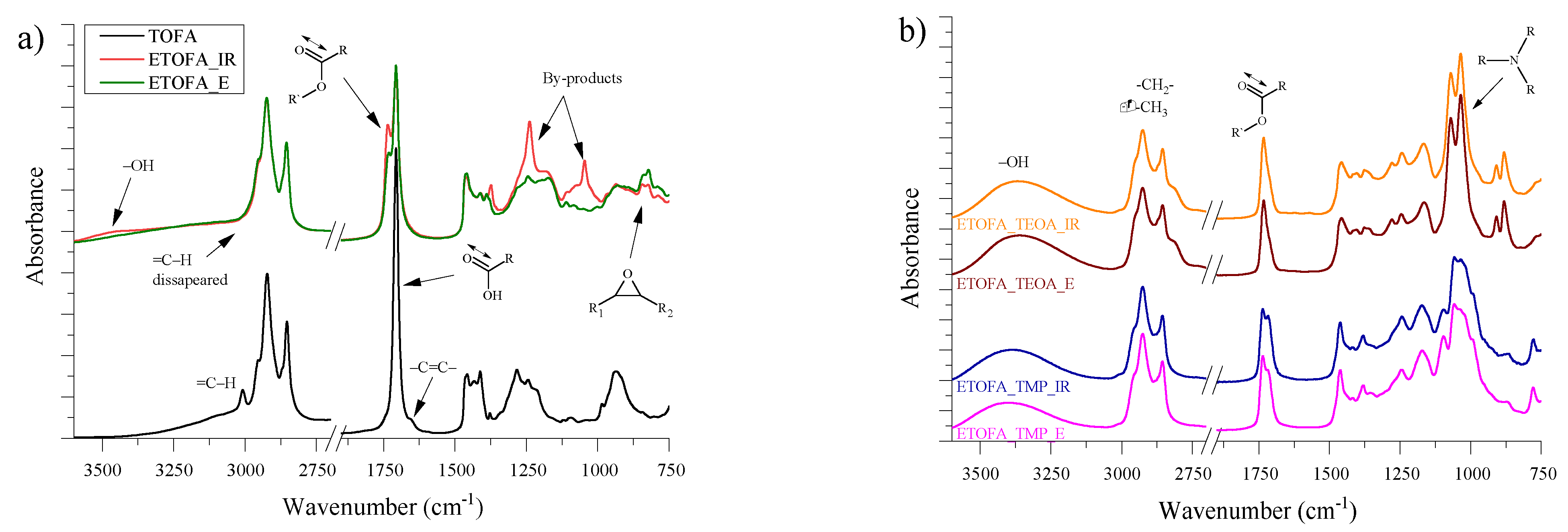
3.2. FTIR Analysis of Synthesised Bio-Polyols from ETOFA
3.3. NMR Analysis of Epoxidated TOFA Using Different Catalysts
3.4. SEC Analysis of Synthesised High Functionality Bio-Polyols
3.5. Characteristics of Rigid PU Foams Developed from Synthesised High Functional Bio-Polyols
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ETOFA | Epoxidised tall oil fatty acids |
| ETOFA_E | Epoxidised tall oil fatty acids obtained from tall oil fatty acids epoxidation with lipase catalyst Novozym® 435 |
| ETOFA_IR | Epoxidised tall oil fatty acids obtained from tall oil fatty acids epoxidation with ion exchange resin Amberlite IR-120 H |
| ETOFA_TEOA_E | Polyol that is obtained from epoxidised tall oil, enzyme is used as a catalyst, oxirane ring opened with triethanolamine |
| ETOFA_TEOA_IR | Polyol that is obtained from epoxidised tall oil, ion exchange resin is used as a catalyst, oxirane ring opened with triethanolamine |
| ETOFA_TMP_E | Polyol that is obtained from epoxidised tall oil if enzyme is used as a catalyst, oxirane ring opened with trimethylolpropane |
| ETOFA_TMP_IR | Polyol that is obtained from epoxidised tall oil if ion exchange resin is used as a catalyst, oxirane ring opened with trimethylolpropane |
| –NCO | Isocyanate group |
| OH value | Hydroxyl value |
| pMDI | 4,4′-diphenylmethane diisocyanate |
| PU | Polyurethane |
| PU_TEOA_E | Polyurethane formulated using polyol that is obtained from epoxidised tall oil, enzyme is used as a catalyst, oxirane ring opened with triethanolamine |
| PU_TEOA_IR | Polyurethane formulated using polyol that is obtained from epoxidised tall oil, ion exchange resin is used as a catalyst, oxirane ring opened with triethanolamine |
| PU_TMP_E | Polyurethane formulated using polyol that is obtained from epoxidised tall oil, enzyme is used as a catalyst, oxirane ring opened with trimethylolpropane |
| PU_TMP_IR | Polyurethane formulated using polyol that is obtained from epoxidised tall oil, ion exchange resin is used as a catalyst, oxirane ring opened with trimethylolpropane |
| TEOA | Triethanolamine |
| TMP | Trimethylolpropane |
| TO_TEOA | Polyol that is obtained by tall oil tall oil esterification with trietanolamine |
| TOFA | Tall oil fatty acids |
References
- Zhang, C.; Garrison, T.F.; Madbouly, S.A.; Kessler, M.R. Recent advances in vegetable oil-based polymers and their composites. Prog. Polym. Sci. 2017, 71, 91–143. [Google Scholar] [CrossRef]
- Biermann, U.; Bornscheuer, U.; Meier, M.A.R.; Metzger, J.O.; Schäfer, H.J. Oils and Fats as Renewable Raw Materials in Chemistry. Angew. Chem. Int. Ed. 2011, 50, 3854–3871. [Google Scholar] [CrossRef]
- Desroches, M.; Escouvois, M.; Auvergne, R.; Caillol, S.; Boutevin, B. From vegetable oils to polyurethanes: Synthetic routes to polyols and main industrial products. Polym. Rev. 2012, 52, 38–79. [Google Scholar] [CrossRef]
- Li, Y.; Luo, X.; Hu, S. Bio-Based Polyols and Polyurethanes, 1st ed.; Springer International Publishing: Berlin/Heidelberg, Germany, 2015; ISBN 9783319215389. [Google Scholar]
- Montero De Espinosa, L.; Meier, M.A.R.R. Plant oils: The perfect renewable resource for polymer science?! Eur. Polym. J. 2011, 47, 837–852. [Google Scholar] [CrossRef]
- Lomège, J.; Negrell, C.; Robin, J.J.; Lapinte, V.; Caillol, S. Oleic acid-based poly(alkyl methacrylate) as bio-based viscosity control additive for mineral and vegetable oils. Polym. Eng. Sci. 2019, 59, E164–E170. [Google Scholar] [CrossRef]
- Noreen, A.; Zia, K.M.; Zuber, M.; Tabasum, S.; Zahoor, A.F. Bio-based polyurethane: An efficient and environment friendly coating systems: A review. Prog. Org. Coat. 2016, 91, 25–32. [Google Scholar] [CrossRef]
- Tan, S.G.; Chow, W.S. Biobased epoxidized vegetable oils and its greener epoxy blends: A review. Polym. Plast. Technol. Eng. 2010, 49, 1581–1590. [Google Scholar] [CrossRef]
- Mungroo, R.; Pradhan, N.C.; Goud, V.V.; Dalai, A.K. Epoxidation of canola oil with hydrogen peroxide catalyzed by acidic ion exchange resin. J. Am. Oil Chem. Soc. 2008, 85, 887–896. [Google Scholar] [CrossRef]
- Kirpluks, M.; Kalnbunde, D.; Walterova, Z.; Cabulis, U. Rapeseed Oil as Feedstock for High Functionality Polyol Synthesis. J. Renew. Mater. 2017, 5, 1–23. [Google Scholar] [CrossRef]
- Lee, P.L.; Wan Yunus, W.M.Z.; Yeong, S.K.; Abdullah, D.K.; Lim, W.H. Optimization of the epoxidation of methyl ester of palm fatty acid distillate. J. Oil Palm Res. 2009, 21, 675–682. [Google Scholar]
- Dinda, S.; Patwardhan, A.V.; Goud, V.V.; Pradhan, N.C. Epoxidation of cottonseed oil by aqueous hydrogen peroxide catalysed by liquid inorganic acids. Bioresour. Technol. 2008, 99, 3737–3744. [Google Scholar] [CrossRef]
- Patel, M.M.; Patel, B.P.; Patel, N.K. Utilization of soya-based polyol for High solid PU-coating application. Int. J. Plast. Technol. 2012, 16, 67–79. [Google Scholar] [CrossRef]
- Sinadinović-Fišer, S.; Janković, M.; Borota, O. Epoxidation of castor oil with peracetic acid formed in situ in the presence of an ion exchange resin. Chem. Eng. Process. Process Intensif. 2012, 62, 106–113. [Google Scholar] [CrossRef]
- Abolins, A.; Yakushin, V.; Vilsone, D. Properties of polyurethane coatings based on linseed oil phosphate ester polyol. J. Renew. Mater. 2018, 6, 737–745. [Google Scholar] [CrossRef]
- Goud, V.V.; Patwardhan, A.V.; Pradhan, N.C. Studies on the epoxidation of mahua oil (Madhumica indica) by hydrogen peroxide. Bioresour. Technol. 2006, 97, 1365–1371. [Google Scholar] [CrossRef]
- de Haro, J.C.; Izarra, I.; Rodríguez, J.F.; Pérez, Á.; Carmona, M. Modelling the epoxidation reaction of grape seed oil by peracetic acid. J. Clean. Prod. 2016, 138, 70–76. [Google Scholar] [CrossRef]
- Abolins, A.; Kirpluks, M.; Vanags, E.; Fridrihsone, A.; Cabulis, U. Tall Oil Fatty Acid Epoxidation Using Homogenous and Heterogeneous Phase Catalysts. J. Polym. Environ. 2020, 28, 1822–1831. [Google Scholar] [CrossRef]
- Kirpluks, M.; Vanags, E.; Abolins, A.; Fridrihsone, A.; Cabulis, U. Chemo-enzymatic oxidation of tall oil fatty acids as a precursor for further polyol production. J. Clean. Prod. 2019, 215, 390–398. [Google Scholar] [CrossRef]
- Vanags, E.; Kirpluks, M.; Cabulis, U.; Walterova, Z. Highly functional polyol synthesis from epoxidized tall oil fatty acids. J. Renew. Mater. 2018, 6, 764–771. [Google Scholar] [CrossRef]
- Goud, V.V.; Patwardhan, A.V.; Dinda, S.; Pradhan, N.C. Kinetics of epoxidation of jatropha oil with peroxyacetic and peroxyformic acid catalysed by acidic ion exchange resin. Chem. Eng. Sci. 2007, 62, 4065–4076. [Google Scholar] [CrossRef]
- Hazmi, A.S.A.; Aung, M.M.; Abdullah, L.C.; Salleh, M.Z.; Mahmood, M.H. Producing Jatropha oil-based polyol via epoxidation and ring opening. Ind. Crops Prod. 2013, 50, 563–567. [Google Scholar] [CrossRef]
- Maisonneuve, L.; Chollet, G.; Grau, E.; Cramail, H. Vegetable oils: A source of polyols for polyurethane materials. OCL 2016, 23, D508. [Google Scholar] [CrossRef]
- Hachemi, I.; Kumar, N.; Mäki-Arvela, P.; Roine, J.; Peurla, M.; Hemming, J.; Salonen, J.; Murzin, D.Y. Sulfur-free Ni catalyst for production of green diesel by hydrodeoxygenation. J. Catal. 2017, 347, 205–221. [Google Scholar] [CrossRef]
- Chemistry, C. Preperation of biodiesel and separation of hemicellulose. Cellul. Chem. Technol. 2016, 50, 247–255. [Google Scholar]
- Aro, T.; Fatehi, P. Tall oil production from black liquor: Challenges and opportunities. Sep. Purif. Technol. 2017, 175, 469–480. [Google Scholar] [CrossRef]
- Panda, H. Handbook on Tall Oil Rosin Production, Processing and Utilization; Asia Pacific Business Press Inc.: Delhi, India, 2013. [Google Scholar]
- Demirbas, A. Methylation of wood fatty and resin acids for production of biodiesel. Fuel 2011, 90, 2273–2279. [Google Scholar] [CrossRef]
- Lubguban, A.A.; Ruda, R.J.G.; Aquiatan, R.H.; Paclijan, S. Soy-Based Polyols and Polyurethanes. KIMIKA 2017, 1, 1–19. [Google Scholar] [CrossRef]
- Kurańska, M.; Leszczyńska, M.; Kubacka, J.; Prociak, A.; Ryszkowska, J. Effects of Modified Used Cooking Oil on Structure and Properties of Closed-Cell Polyurethane foams. J. Polym. Environ. 2020, 28, 2780–2788. [Google Scholar] [CrossRef]
- Omonov, T.S.; Kharraz, E.; Curtis, J.M. The epoxidation of canola oil and its derivatives. RSC Adv. 2016, 6, 92874–92886. [Google Scholar] [CrossRef]
- Ranganathan, S.; Sieber, V. Development of semi-continuous chemo-enzymatic terpene epoxidation: Combination of anthraquinone autooxidation and the lipase-mediated epoxidation process. React. Chem. Eng. 2017, 2, 885–895. [Google Scholar] [CrossRef]
- Re, R.N.; Proessdorf, J.C.; La Clair, J.J.; Subileau, M.; Burkart, M.D. Tailoring chemoenzymatic oxidation: Via in situ peracids. Org. Biomol. Chem. 2019, 17, 9418–9424. [Google Scholar] [CrossRef]
- Cai, X.; Zheng, J.L.; Aguilera, A.F.; Vernières-Hassimi, L.; Tolvanen, P.; Salmi, T.; Leveneur, S. Influence of ring-opening reactions on the kinetics of cottonseed oil epoxidation. Int. J. Chem. Kinet. 2018, 50, 726–741. [Google Scholar] [CrossRef]
- Zhang, X.; Wan, X.; Cao, H.; Dewil, R.; Deng, L.; Wang, F.; Tan, T.; Nie, K. Chemo-enzymatic epoxidation of Sapindus mukurossi fatty acids catalyzed with Candida sp. 99–125 lipase in a solvent-free system. Ind. Crops Prod. 2017, 98, 10–18. [Google Scholar] [CrossRef]
- Mashhadi, F.; Habibi, A.; Varmira, K. Determination of Activation Energy and Ping-Pong Kinetic Model Constants of Enzyme-Catalyzed Self-Epoxidation of Free Fatty Acids using Micro-reactor. Catal. Lett. 2018, 148, 3236–3247. [Google Scholar] [CrossRef]
- Danov, S.M.; Kazantsev, O.A.; Esipovich, A.L.; Belousov, A.S.; Rogozhin, A.E.; Kanakov, E.A. Recent advances in the field of selective epoxidation of vegetable oils and their derivatives: A review and perspective. Catal. Sci. Technol. 2017, 7, 3659–3675. [Google Scholar] [CrossRef]
- Kumar, A.; Dhar, K.; Kanwar, S.S.; Arora, P.K. Lipase catalysis in organic solvents: Advantages and applications. Biol. Proced. Online 2016, 18, 1–11. [Google Scholar] [CrossRef]
- Milchert, E.; Malarczyk-Matusiak, K.; Musik, M. Technological aspects of vegetable oils epoxidation in the presence of ion exchange resins: A review. Polish J. Chem. Technol. 2016, 18, 128–133. [Google Scholar] [CrossRef]
- Mateo, C.; Palomo, J.M.; Fernandez-Lorente, G.; Guisan, J.M.; Fernandez-Lafuente, R. Improvement of enzyme activity, stability and selectivity via immobilization techniques. Enzyme Microb. Technol. 2007, 40, 1451–1463. [Google Scholar] [CrossRef]
- Ivdre, A.; Soto, G.D.; Cabulis, U. Polyols Based on Poly(ethylene terephthalate) and Tall Oil: Perspectives for Synthesis and Production of Rigid Polyurethane Foams. J. Renew. Mater. 2016, 4, 285–293. [Google Scholar] [CrossRef]
- Zhang, C.; Ding, R.; Kessler, M.R. Reduction of epoxidized vegetable oils: A novel method to prepare bio-based polyols for polyurethanes. Macromol. Rapid Commun. 2014, 35, 1068–1074. [Google Scholar] [CrossRef]
- Zeltins, V.; Yakushin, V.; Cabulis, U.; Kirpluks, M. Crude Tall Oil as Raw Material for Rigid Polyurethane Foams with Low Water Absorption. Solid State Phenom. 2017, 267, 17–22. [Google Scholar] [CrossRef]
- Pfister, D.P.; Xia, Y.; Larock, R.C. Recent advances in vegetable oil-based polyurethanes. ChemSusChem 2011, 4, 703–717. [Google Scholar] [CrossRef] [PubMed]
- Barczewski, M.; Kurańska, M.; Sałasińska, K.; Michałowski, S.; Prociak, A.; Uram, K.; Lewandowski, K. Rigid polyurethane foams modified with thermoset polyester-glass fiber composite waste. Polym. Test. 2020, 81. [Google Scholar] [CrossRef]
- Członka, S.; Strakowska, A.; Strzelec, K.; Kairyte, A.; Kremensas, A. Bio-based polyurethane composite foams with improved mechanical, thermal, and antibacterial properties. Materials 2020, 13, 1108. [Google Scholar] [CrossRef]
- Ekkaphan, P.; Sooksai, S.; Chantarasiri, N.; Petsom, A. Bio-Based Polyols from Seed Oils for Water-Blown Rigid Polyurethane Foam Preparation. Int. J. Polym. Sci. 2016, 2016. [Google Scholar] [CrossRef]
- Alagi, P.; Ghorpade, R.; Jang, J.H.; Patil, C.; Jirimali, H.; Gite, V.; Hong, S.C. Functional soybean oil-based polyols as sustainable feedstocks for polyurethane coatings. Ind. Crops Prod. 2018, 113, 249–258. [Google Scholar] [CrossRef]
- Omrani, I.; Farhadian, A.; Babanejad, N.; Shendi, H.K.; Ahmadi, A.; Nabid, M.R. Synthesis of novel high primary hydroxyl functionality polyol from sunflower oil using thiol-yne reaction and their application in polyurethane coating. Eur. Polym. J. 2016, 82, 220–231. [Google Scholar] [CrossRef]
- Fridrihsone-Girone, A.; Stirna, U.; Misane, M.; Lazdiņa, B.; Deme, L. Spray-applied 100% volatile organic compounds free two component polyurethane coatings based on rapeseed oil polyols. Prog. Org. Coat. 2016, 94, 90–97. [Google Scholar] [CrossRef]
- Considine, D.M.; Considine, G.D. Van Nostrand’s Scientific Encyclopedia; Springer US: Boston, MA, USA, 1995; ISBN 978-1-4757-6920-3. [Google Scholar]
- Kurańska, M.; Prociak, A. The influence of rapeseed oil-based polyols on the foaming process of rigid polyurethane foams. Ind. Crops Prod. 2016, 89, 182–187. [Google Scholar] [CrossRef]
- Zieleniewska, M.; Leszczyński, M.K.; Kurańska, M.; Prociak, A.; Szczepkowski, L.; Krzyżowska, M.; Ryszkowska, J. Preparation and characterisation of rigid polyurethane foams using a rapeseed oil-based polyol. Ind. Crops Prod. 2015, 74, 887–897. [Google Scholar] [CrossRef]
- Kirpluks, M.; Vanags, E.; Abolins, A.; Michalowski, S.; Fridrihsone, A.; Cabulis, U. High Functionality Bio-Polyols from Tall Oil and Rigid Polyurethane Foams Formulated Solely Using Bio-Polyols. Materials 2020, 13, 1985. [Google Scholar] [CrossRef] [PubMed]
- Kirpluks, M.; Pomilovskis, R.; Vanags, E.; Abolins, A.; Mierina, I.; Fridrihsone, A. Optimisation of the chemo-enzymatic epoxidation of tall oil fatty acids using response surface methodology. J. Clean. Prod. 2020. Manuscript submitted for publication. [Google Scholar]
- Hawkins, M.C.; O’Toole, B.; Jackovich, D. Cell Morphology and Mechanical Properties of Rigid Polyurethane Foam. J. Cell. Plast. 2005, 41, 267–285. [Google Scholar] [CrossRef]
- Szycher, M. Szycher’s Handbook of Polyurethanes, 1st ed.; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar]
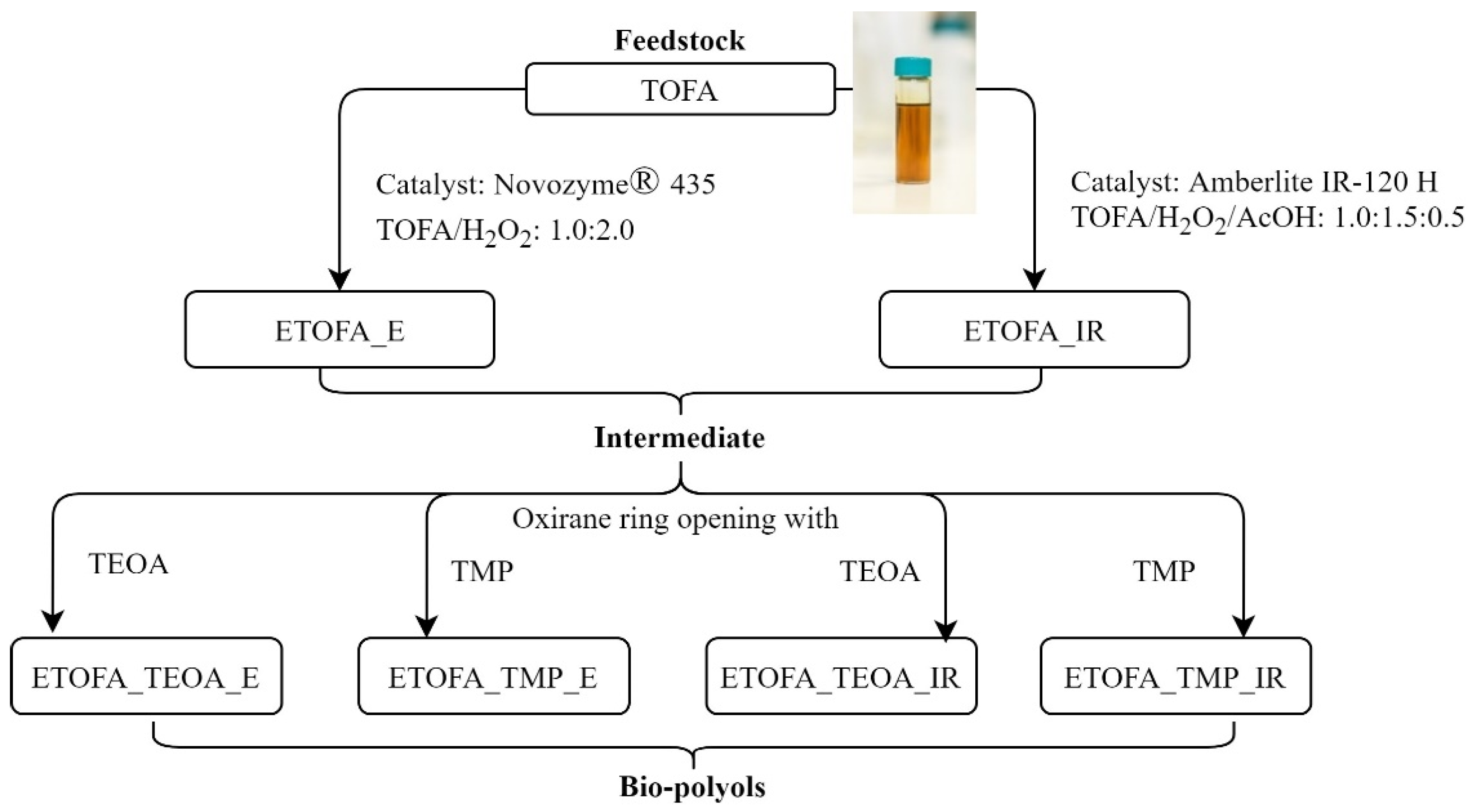




| Multifunctional Alcohol for Polyol Synthesis | Mass (g) Added if Polyol is Synthesised from ETOFA_IR | Mass (g) Added if Polyol is Synthesised from ETOFA_E |
|---|---|---|
| TEOA | 145.0 | 175.4 |
| TMP | 130.4 | 157.8 |
| Components | Reagents, Parts by Weight | ||||
|---|---|---|---|---|---|
| PU_TEOA_IR | PU_TEOA_E | PU_TMP_IR | PU_TMP_E | ||
| Bio-polyols | ETOFA_TEOA_IR | 85.0 | – | – | – |
| ETOFA_TEOA_E | – | 85.0 | – | – | |
| ETOFA_TMP_IR | – | – | 85.0 | – | |
| ETOFA_TMP_E | – | – | – | 85.0 | |
| TO_TEOA | 10.0 | 10.0 | 10.0 | 10.0 | |
| Glycerol | 5.0 | 5.0 | 5.0 | 5.0 | |
| Blowing agents | c-pentane | 12.0 | 12.0 | 12.0 | 10.0 |
| Water | 2.0 | 2.0 | 2.0 | 2.0 | |
| Catalysts | Polycat® 5 | – | – | 0.5 | 0.5 |
| Polycat® NP10 | 3.0 | 3.0 | 3.0 | 3.0 | |
| PC CAT TKA 30 | 1.5 | 1.5 | 1.5 | 1.5 | |
| Surfactant | L-6915 | 2.5 | 2.5 | 2.5 | 2.5 |
| Flame retardant | TCPP | 31 (8 wt.%) | 30 (8 wt.%) | 32 (8 wt.%) | 27 (8 wt.%) |
| Isocyanate | pMDI | 243.4 | 218.3 | 246.2 | 190.5 |
| Isocyanate index | – | 150 | 150 | 150 | 150 |
| Renewable materials in PU foam, % | 16.3 | 18.1 | 16.2 | 19.8 | |
| Polyol | OH Value, mg KOH/g | Acid Value, mg KOH/g | Moisture, % | Viscosity (20 °C), mPa·s | Density (20 °C), g/cm3 | fn | Mn, g/mol | pd |
|---|---|---|---|---|---|---|---|---|
| ETOFA_TEOA_IR | 510 | <5 | 0.14 | 7400 | 1.047 | 8.1 | 893 | 1.78 |
| ETOFA_TEOA_E | 519 | <5 | 0.05 | 10,800 | 1.048 | 8.3 | 899 | 1.88 |
| ETOFA_TMP_IR | 427 | <5 | 0.24 | 77,000 | 1.056 | 9.7 | 1279 | 1.58 |
| ETOFA_TMP_E | 335 | <5 | 0.06 | 278,300 | 1.058 | 12.6 | 2112 | 1.57 |
| PU Material | PU_TEOA_IR | PU_TEOA_E | PU_TMP_IR | PU_TMP_E |
|---|---|---|---|---|
| Technological parameters | – | |||
| Foaming start time, s | 28 | 29 | 32 | 23 |
| Foaming rise time, s | 61 | 61 | 78 | 67 |
| PU foam apparent density, kg/m3 | 36.4 | 35.4 | 35.8 | 38.5 |
| Closed cell content, % | 90 | 91 | 91 | 95 |
| Thermal conductivity, mW/(m·K) | 23.0 | 23.3 | 21.3 | 21.2 |
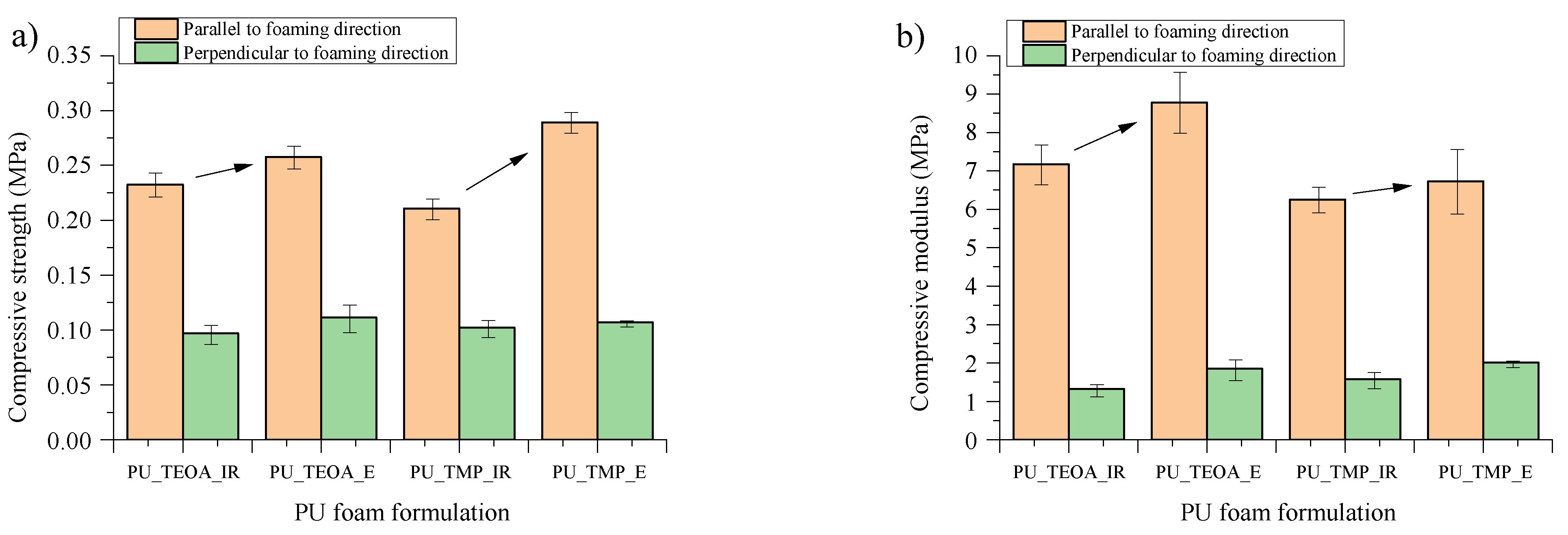
| PU Material | Compressive Strength, MPa | Compressive Modulus, MPa, | PU Foam Apparent Density, kg/m3 | Closed Cell Content, % | Thermal Conductivity, mW/(m·K) |
|---|---|---|---|---|---|
| PU_TEOA_IR | 0.191 | 5.6 | 36.4 | 90.0 | 23.0 |
| PU_TEOA_E | 0.195 | 6.7 | 35.4 | 91.0 | 23.3 |
| PU_TMP_IR | 0.198 | 5.9 | 35.8 | 91.0 | 21.3 |
| PU_TMP_E | 0.309 | 7.2 | 38.5 | 95.0 | 21.2 |
| PU_60_PWCO 1 | 0.275 | n.a | 41.3 | 85.3 | 26.4 |
| PU-BMG/0 2 | 0.248 | n.a | 42.1 | 87.5 | 22.5 |
| PU-0 3 | 0.250 | 6.4 | 37.0 | 88.0 | n.a |
| PU-PSO 4 | 0.238 | n.a | 40.8 | n.a | 33.9 |
| Raypol® 4218 4 | 0.303 | n.a | 44.1 | n.a | 36.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abolins, A.; Pomilovskis, R.; Vanags, E.; Mierina, I.; Michalowski, S.; Fridrihsone, A.; Kirpluks, M. Impact of Different Epoxidation Approaches of Tall Oil Fatty Acids on Rigid Polyurethane Foam Thermal Insulation. Materials 2021, 14, 894. https://doi.org/10.3390/ma14040894
Abolins A, Pomilovskis R, Vanags E, Mierina I, Michalowski S, Fridrihsone A, Kirpluks M. Impact of Different Epoxidation Approaches of Tall Oil Fatty Acids on Rigid Polyurethane Foam Thermal Insulation. Materials. 2021; 14(4):894. https://doi.org/10.3390/ma14040894
Chicago/Turabian StyleAbolins, Arnis, Ralfs Pomilovskis, Edgars Vanags, Inese Mierina, Slawomir Michalowski, Anda Fridrihsone, and Mikelis Kirpluks. 2021. "Impact of Different Epoxidation Approaches of Tall Oil Fatty Acids on Rigid Polyurethane Foam Thermal Insulation" Materials 14, no. 4: 894. https://doi.org/10.3390/ma14040894
APA StyleAbolins, A., Pomilovskis, R., Vanags, E., Mierina, I., Michalowski, S., Fridrihsone, A., & Kirpluks, M. (2021). Impact of Different Epoxidation Approaches of Tall Oil Fatty Acids on Rigid Polyurethane Foam Thermal Insulation. Materials, 14(4), 894. https://doi.org/10.3390/ma14040894






