Biomass Thermochemical Conversion via Pyrolysis with Integrated CO2 Capture
Abstract
1. Introduction
2. Materials and Methods
2.1. Material Characteristic
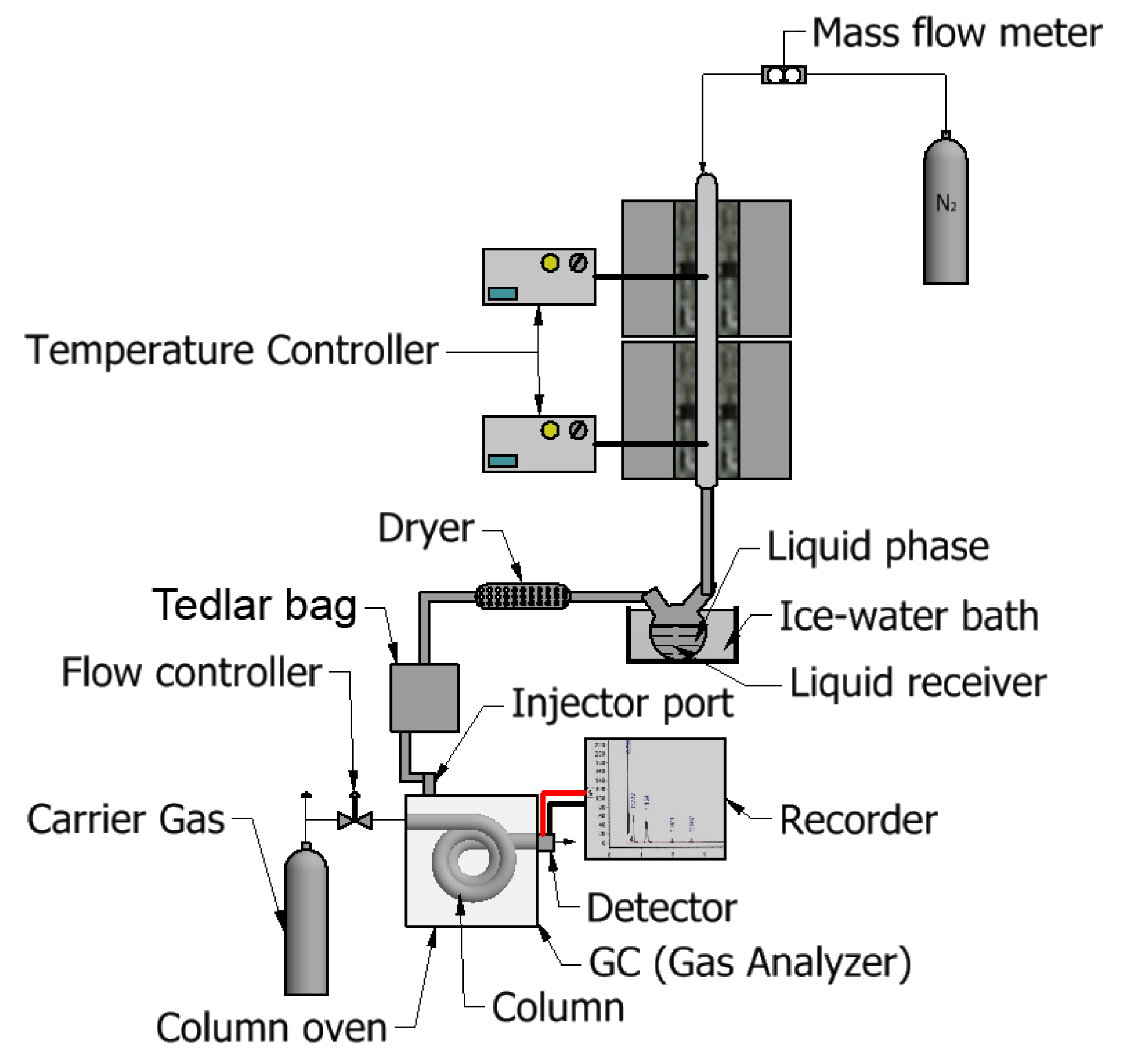
2.2. Experimental Procedure and Methods
3. Results and Discussion
3.1. Analysis of Solid Pyrolysis Products
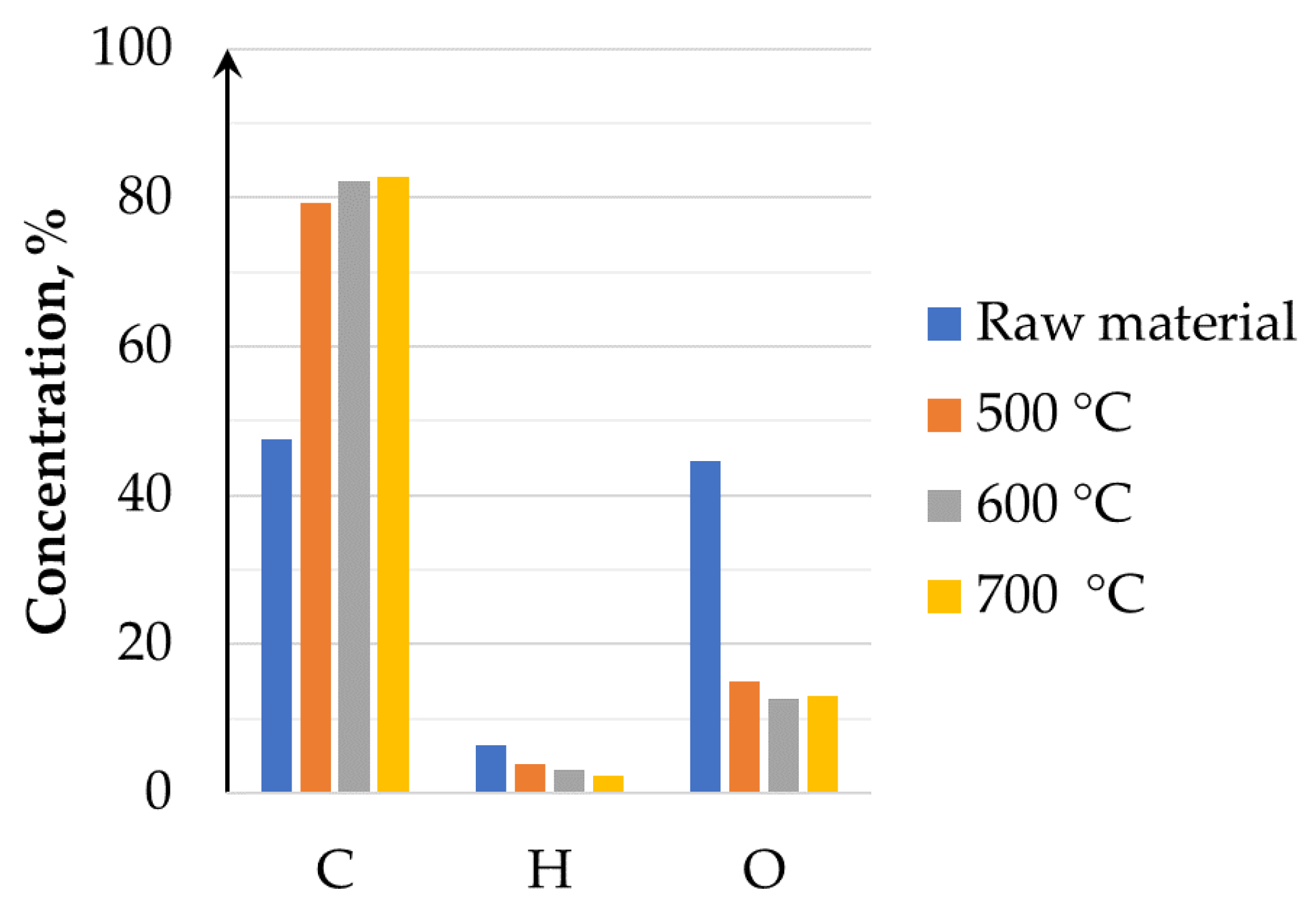
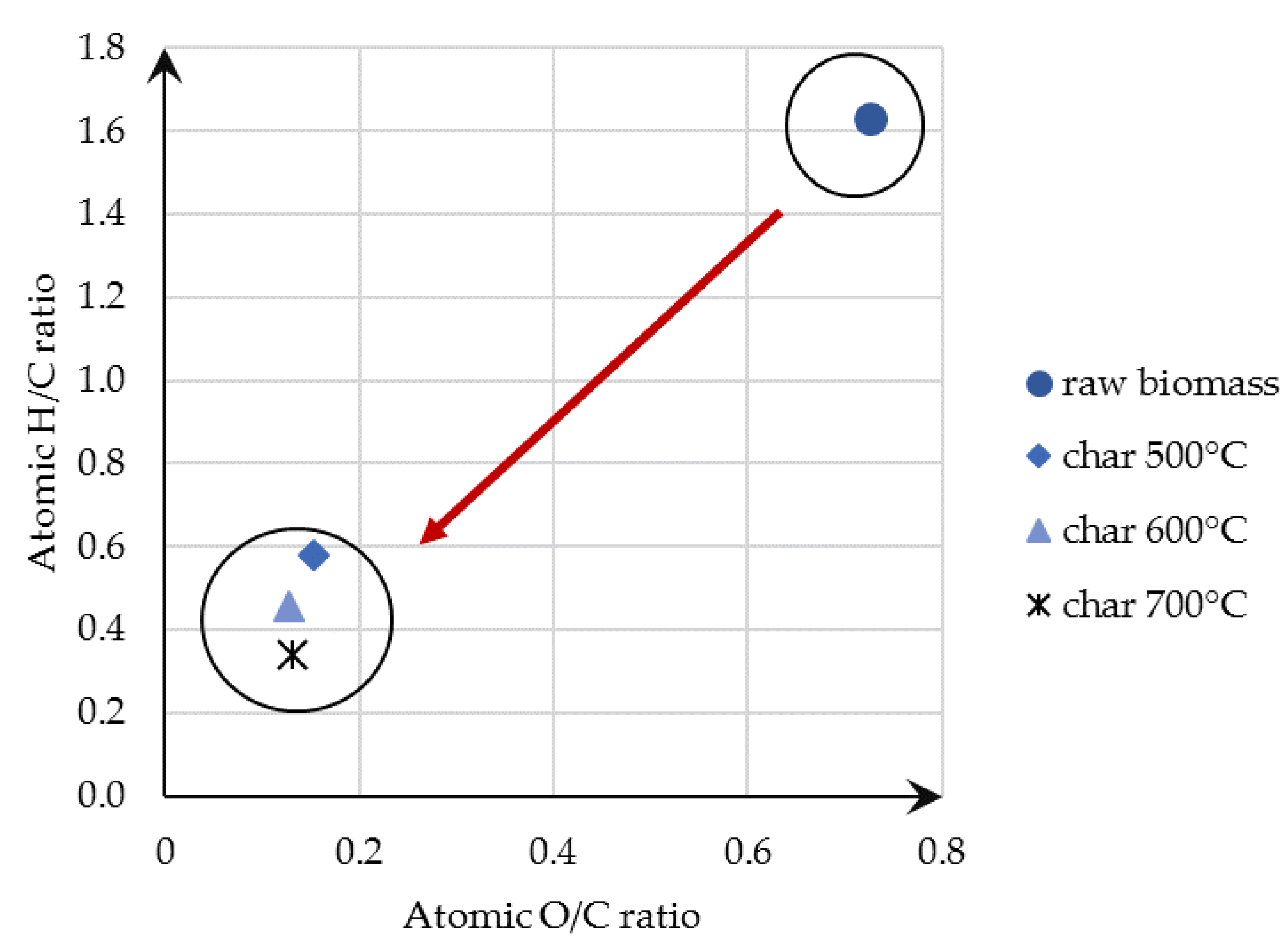
3.1.1. Pyrolysis Char Analysis
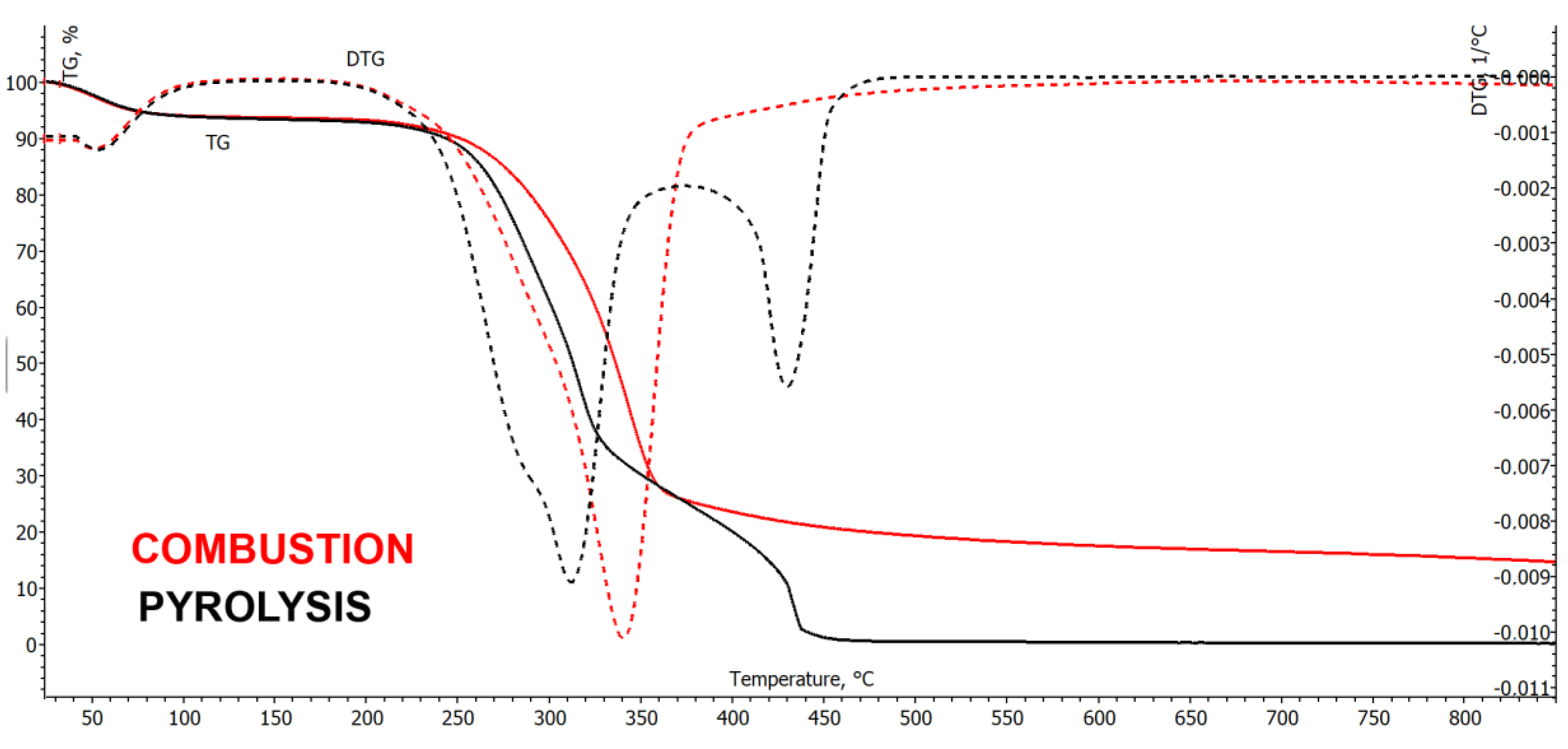
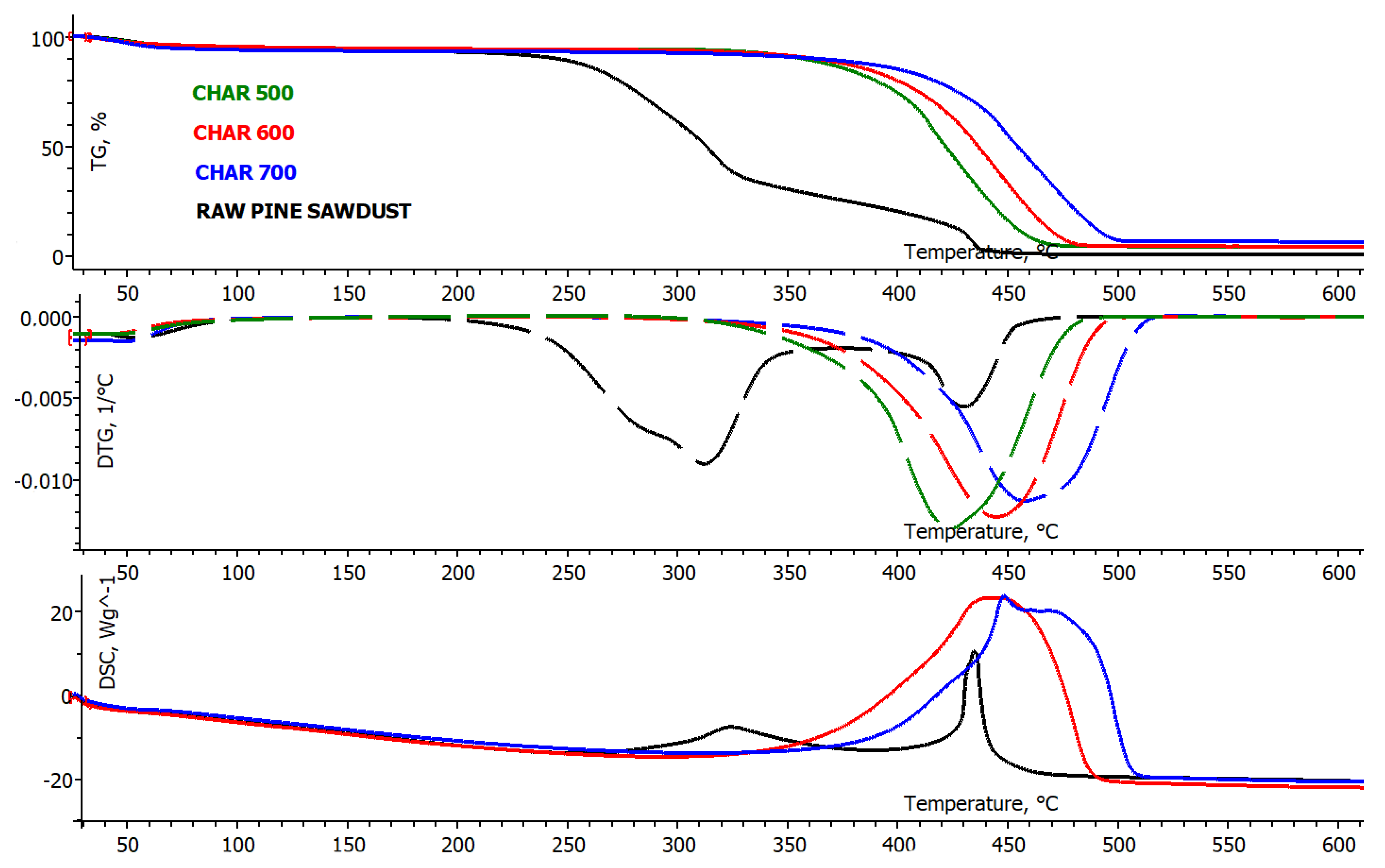
3.1.2. Thermogravimetric Analysis of Combustion Properties of Pine Sawdust and Chars
3.1.3. Scanning Electron Microscope Analysis of Raw Material and Obtained Chars
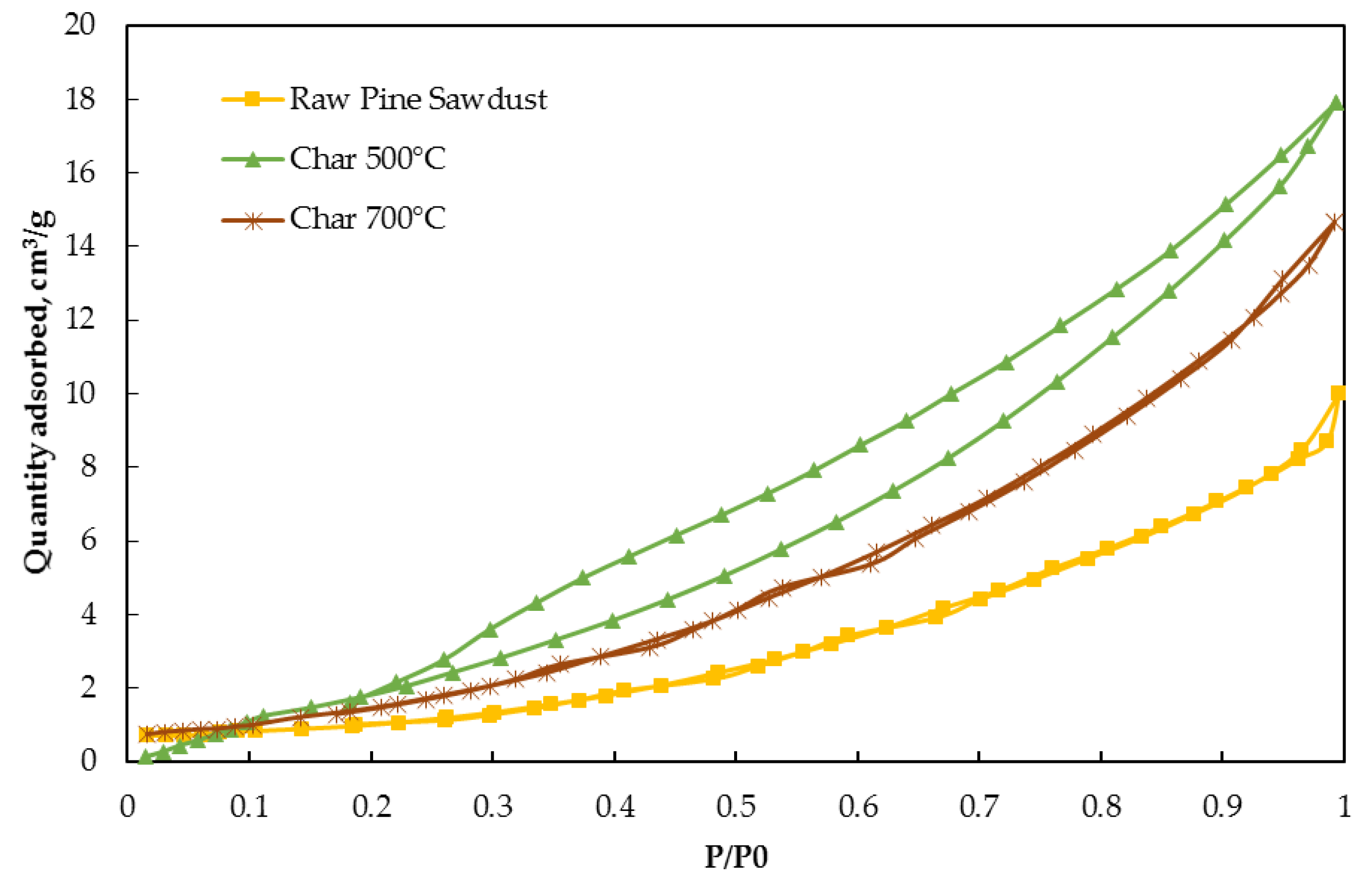
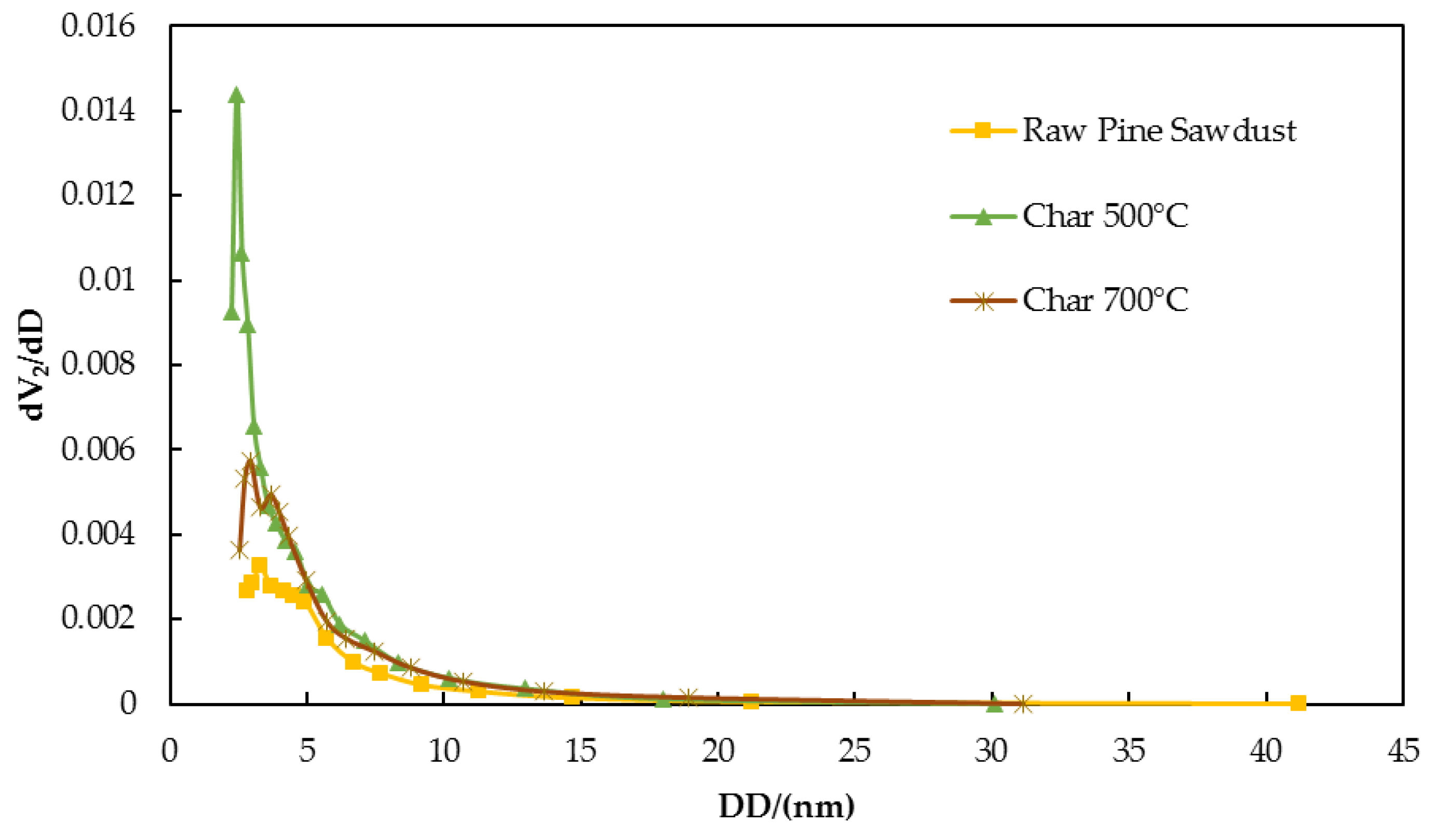
3.1.4. Adsorption Properties of Chars
3.2. Analysis of Sorbent (CaO)
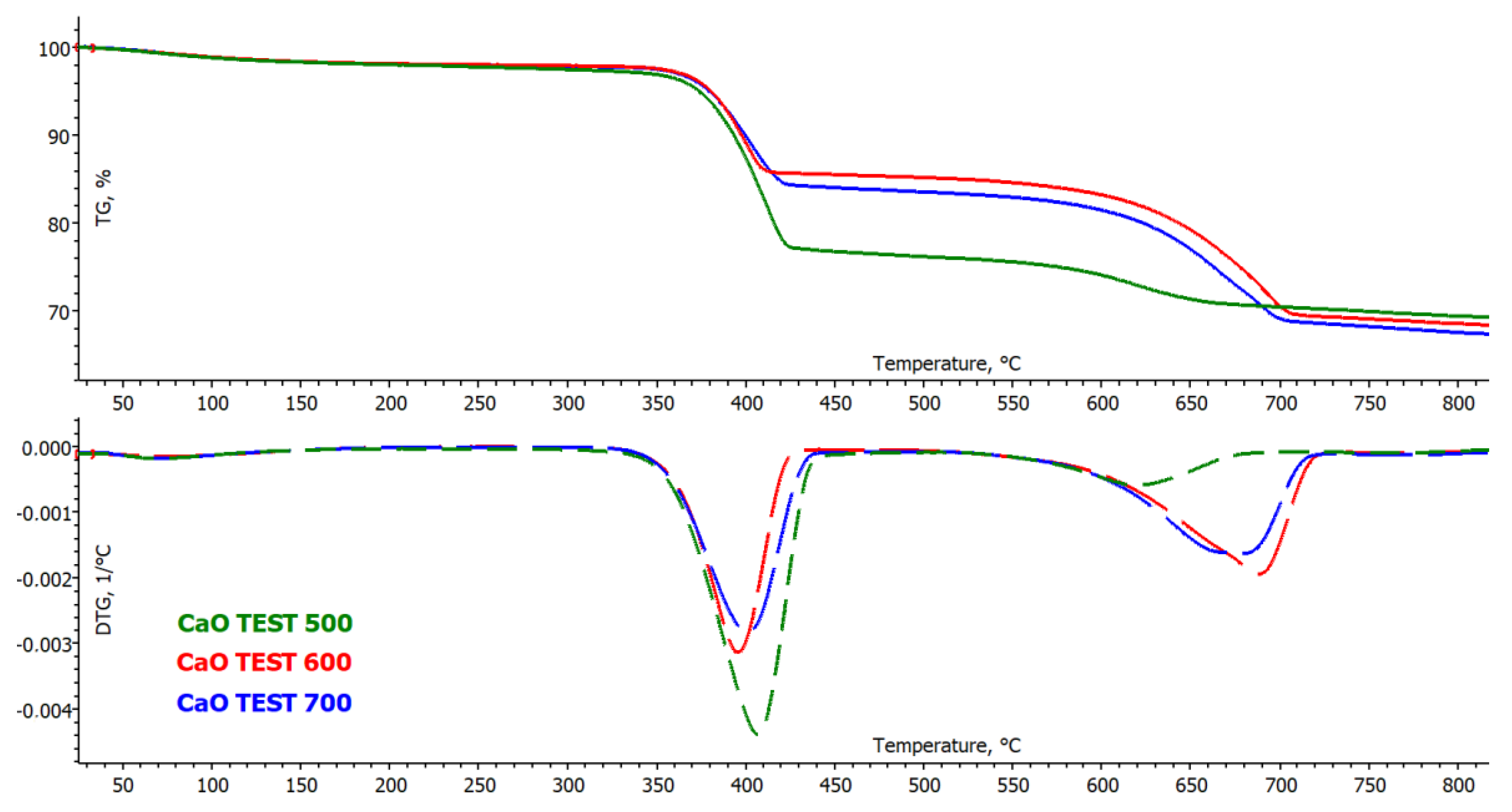
3.2.1. Thermogravimetric Analysis of Sorbent (CaO)
3.2.2. Scanning Electron Microscope Analysis of Sorbent (CaO)
3.2.3. X-ray Diffraction Analysis of Sorbent (CaO)
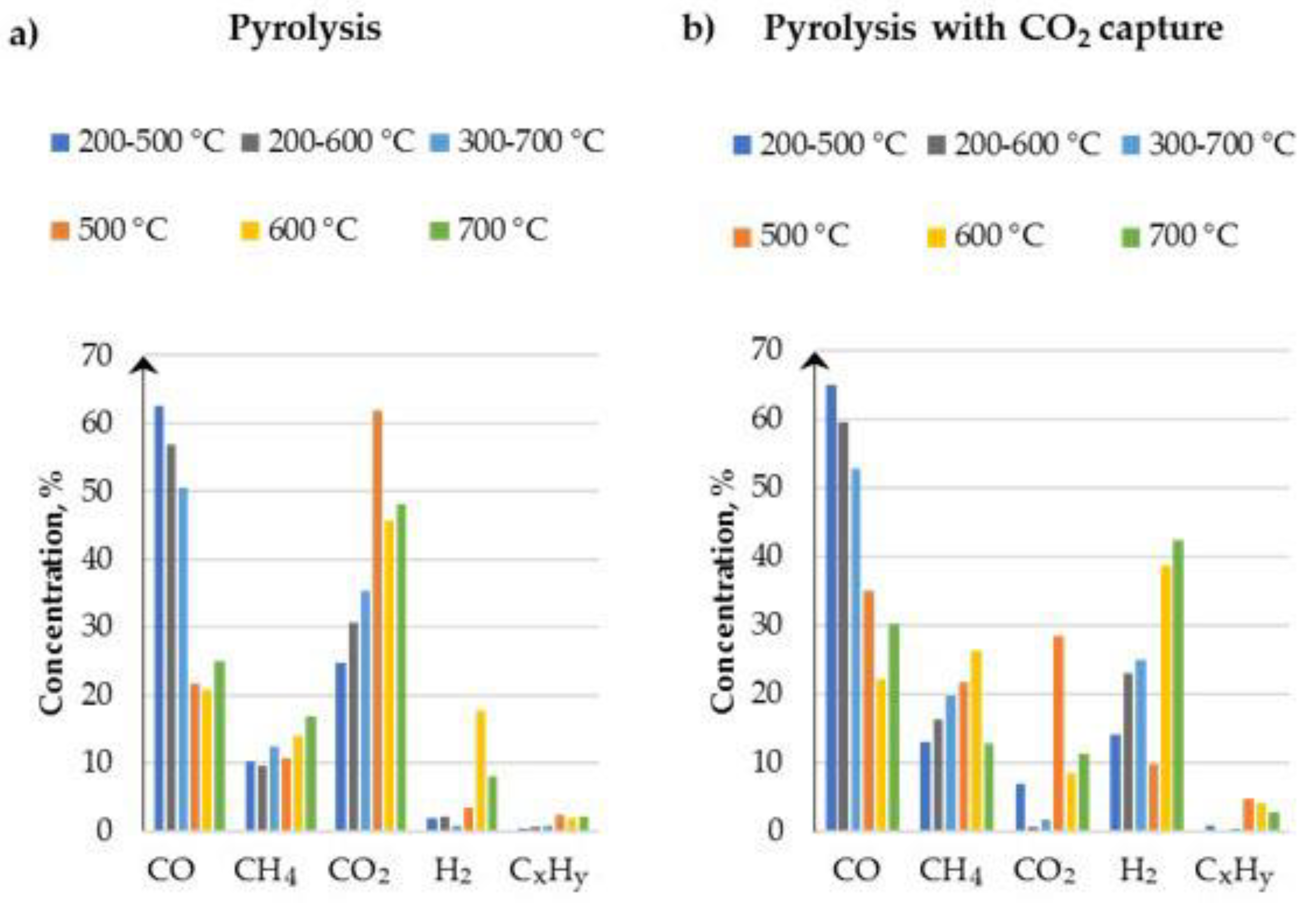
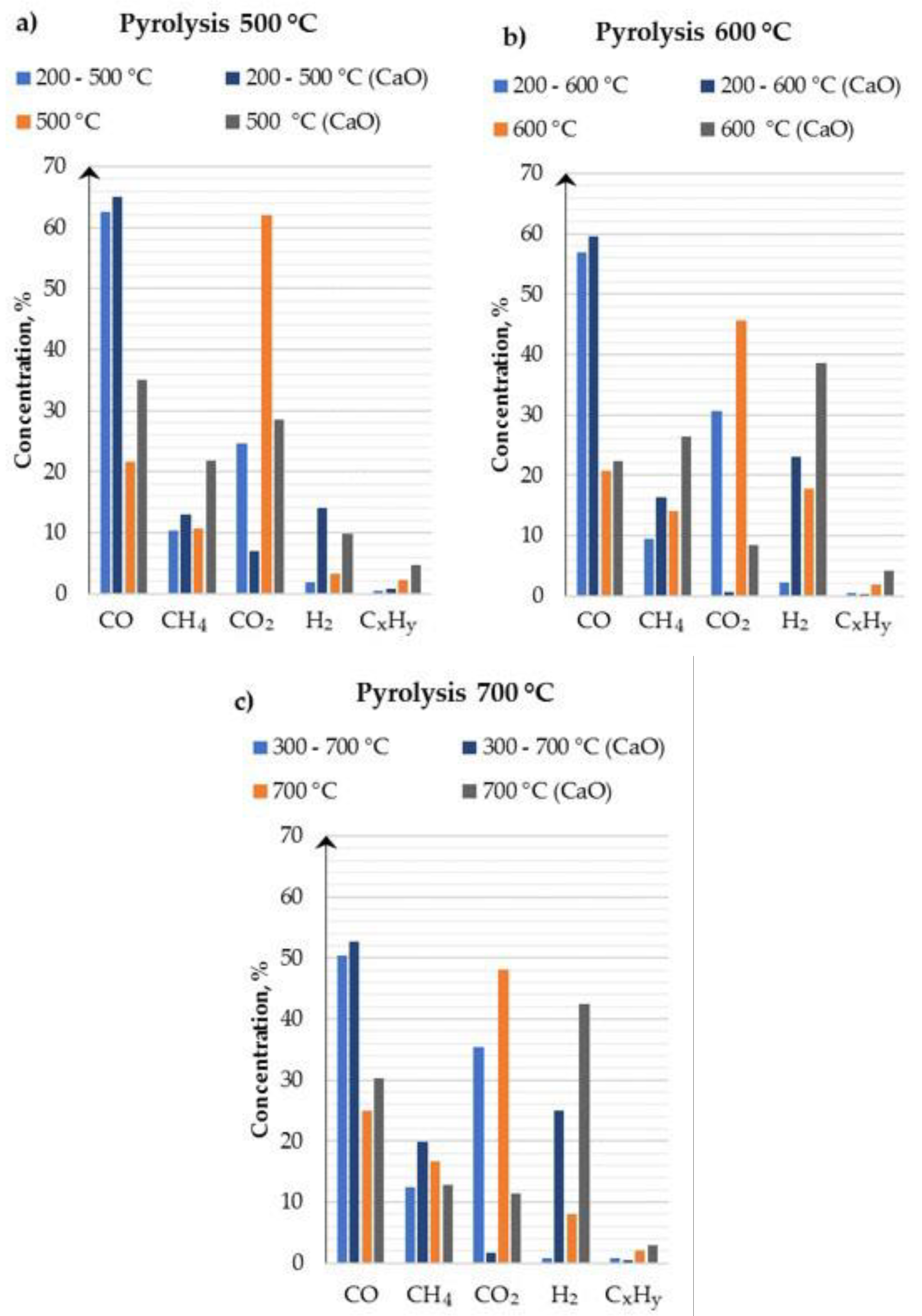
3.3. Gaseous Pyrolysis Products
4. Conclusions
- The solid phase (biochar) obtained during the pyrolysis process is a valuable energy carrier with good combustion properties of a high carbon content and lower VM in comparison to the raw biomass. The analysis of studied temperature range allowed us to conclude that a temperature increase leads to the production of biochar with a higher carbon content and lower VM, as the gaseous phase of biomass was processed and converted into syngas. Biochar mimics the properties of coal. Its high carbon content enables an effective combustion process, which is also reflected in the calculated main combustion parameters.
- The pyrolysis process temperature directly influences the syngas as well as biochar composition. With the increase of process temperature, higher hydrogen and methane concentrations are obtained. However, concentrations of higher hydrocarbons, such as C2H4, C2H6, C3H6 and C3H8, decrease. These concentration changes are caused by the promotion of carbon and hydrogen contained in the biomass which are processed into pure hydrogen and methane forms.
- The addition of a sorbent such as CaO for CO2 capture improves the final gas composition. The presence of sorbent allows the capture of carbon dioxide from produced syngas. As the process temperature increases, the concentration of CO2 in syngas decreases. The conducted tests and analysis prove the ability of capturing the CO2 released during the pyrolysis process and transforming it into a carbonate phase by CaO. The processes of tar cracking are promoted by the presence of CaO with parallel removal of CO2 and lead to an increase in H2 production.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zheng, X.; Streimikiene, D.; Balezentis, T.; Mardani, A.; Cavallaro, F.; Liao, H. A review of greenhouse gas emission profiles, dynamics, and climate change mitigation efforts across the key climate change players. J. Clean. Prod. 2019, 234, 1113–1133. [Google Scholar] [CrossRef]
- European Commission, Communication from the commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions Energy Roadmap 2050. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A52011DC0885 (accessed on 26 February 2020).
- Climate Transparency. Brown to Green: The G20 Transition towards a Net-zero Emissions Economy 2019. Available online: https://www.odi.org/publications/11501-brown-green-g20-transition-towards-net-zero-emissions-economy-2019 (accessed on 26 February 2020).
- Bel, G.; Joseph, S. Climate change mitigation and the role of technological change: Impact on selected headline targets of Europe’s 2020 climate and energy package. Renew. Sustain. Energy Rev. 2018, 82, 3798–3807. [Google Scholar] [CrossRef]
- Connolly, D.; Lund, H.; Mathiesen, B.V. Smart Energy Europe: The technical and economic impact of one potential 100% renewable energy scenario for the European Union. Renew. Sustain. Energy Rev. 2016, 60, 1634–1653. [Google Scholar] [CrossRef]
- Patel, M.; Zhang, X.; Kumar, A. Techno-economic and life cycle assessment on lignocellulosic biomass thermochemical conversion technologies: A review. Renew. Sustain. Energy Rev. 2016, 53, 1486–1499. [Google Scholar] [CrossRef]
- Magdziarz, A.; Wilk, M.; Wądrzyk, M. Pyrolysis of hydrochar derived from biomass—Experimental investigation. Fuel 2020, 267, 117246. [Google Scholar] [CrossRef]
- Dayton, D.C.; Foust, T.D. Chapter Two—Biomass Characterization. Analytical Methods for Biomass Characterization and Conversion. Emerging Issues in Analytical Chemistry 2020. pp. 19–35. Available online: https://www.sciencedirect.com/book/9780128156056/analytical-methods-for-biomass-characterization-and-conversion (accessed on 26 February 2020).
- Kumar, A.; Jones, D.D.; Hanna, M.A. Thermochemical biomass gasification: A review of the current status of the technology. Energies 2009, 2, 556. [Google Scholar] [CrossRef]
- World Biomass Association, Global Biomass Potential Towards 2035. Available online: https://worldbioenergy.org/uploads/Factsheet_Biomass potential.pdf (accessed on 26 February 2020).
- Osman, A.I.; Abdelkader, A.; Farrell, C.; Rooney, D.; Morgan, K. Reusing, recycling and up-cycling of biomass: A review of practical and kinetic modelling approaches. Fuel Process. Technol. 2019, 192, 179–202. [Google Scholar] [CrossRef]
- Ren, J.; Liu, Y.L.; Zhao, X.Y.; Cao, J.P. Biomass thermochemical conversion: A review on tar elimination from biomass catalytic gasification. J. Energy Inst. 2019. [Google Scholar] [CrossRef]
- Mlonka-Mędrala, A.; Magdziarz, A.; Dziok, T.; Sieradzka, M.; Nowak, W. Laboratory studies on the influence of biomass particle size on pyrolysis and combustion using TG GC/MS. Fuel 2019, 252, 635–645. [Google Scholar] [CrossRef]
- Da Silva, C.M.S.; Carneiro, A.D.C.O.; Vital, B.R.; Figueiró, C.G.; de Fialho, L.F.; de Magalhães, M.A.; Carvalho, A.G.; Cândido, W.L. Biomass torrefaction for energy purposes—Definitions and an overview of challenges and opportunities in Brazil. Renew. Sustain. Energy Rev. 2018, 82, 2426–2432. [Google Scholar] [CrossRef]
- Shah, S.A.A.; Zhou, P.; Walasai, G.D.; Mohsin, M. Energy security and environmental sustainability index of South Asian countries: A composite index approach. Ecol. Indic. 2019, 106, 105507. [Google Scholar] [CrossRef]
- Wang, Z. Does biomass energy consumption help to control environmental pollution? Evidence from BRICS countries. Sci. Total Environ. 2019, 670, 1075–1083. [Google Scholar]
- Basu, P. Biomass Gasification, Pyrolysis and Torrefaction. 2018. Available online: https://www.sciencedirect.com/book/9780128129920/biomass-gasification-pyrolysis-and-torrefaction (accessed on 26 February 2020).
- JCR for the European Commission’s Knowledge Centre for Bioeconomy. Brief on Biomass for Energy in the European Union. 2019. Available online: https://ec.europa.eu/jrc/en/publication/brief-biomass-energy-european-union (accessed on 26 February 2020).
- Zhang, P.; Zhang, X.; Li, Y.; Han, L. Influence of pyrolysis temperature on chemical speciation, leaching ability, and environmental risk of heavy metals in biochar derived from cow manure. Bioresour. Technol. 2020, 302, 122850. [Google Scholar] [CrossRef] [PubMed]
- Quan, C.; Gao, N.; Song, Q. Pyrolysis of biomass components in a TGA and a fixed-bed reactor: Thermochemical behaviors, kinetics, and product characterization. J. Anal. Appl. Pyrolysis 2016, 121, 84–92. [Google Scholar] [CrossRef]
- Sobek, S.; Werle, S. Kinetic modelling of waste wood devolatilization during pyrolysis based on thermogravimetric data and solar pyrolysis reactor performance. Fuel 2020, 261, 116459. [Google Scholar] [CrossRef]
- Joardder, M.U.H.; Halder, P.K.; Rahim, M.A.; Masud, M.H. Solar Pyrolysis: Converting Waste into Asset Using Solar Energy. Clean Energy for Sustainable Development: Comparisons and Contrasts of New Approaches. 2017, pp. 213–235. Available online: https://eprints.qut.edu.au/174157/ (accessed on 26 February 2020).
- Rony, A.H.; Kong, L.; Lu, W.; Dejam, M.; Adidharma, H.; Gasem, K.A.M.; Zheng, Y.; Norton, U.; Fan, M. Kinetics, thermodynamics, and physical characterization of corn stover (Zea mays) for solar biomass pyrolysis potential analysis. Bioresour. Technol. 2019, 284, 466–473. [Google Scholar] [CrossRef]
- Sobek, S.; Werle, S. Solar pyrolysis of waste biomass: Part 1 reactor design. Renew. Energy 2019, 143, 1939–1948. [Google Scholar] [CrossRef]
- Tran, K.-Q.; Werle, S.; Trinh, T.T.; Magdziarz, A.; Sobek, S.; Pogrzeba, M. Fuel characterization and thermal degradation kinetics of biomass from phytoremediation plants. Biomass Bioenergy 2020, 134, 105469. [Google Scholar] [CrossRef]
- Uzun, H.; Yıldız, Z.; Goldfarb, J.L.; Ceylan, S. Improved prediction of higher heating value of biomass using an artificial neural network model based on proximate analysis. Bioresour. Technol. 2017, 234, 122–130. [Google Scholar] [CrossRef]
- García, R.; Pizarro, C.; Lavín, A.G.; Bueno, J.L. Biomass proximate analysis using thermogravimetry. Bioresour. Technol. 2013, 139, 1–4. [Google Scholar] [CrossRef]
- Erol, M.; Haykiri-Acma, H.; Küçükbayrak, S. Calorific value estimation of biomass from their proximate analyses data. Renew. Energy 2010, 35, 170–173. [Google Scholar] [CrossRef]
- Parikh, J.; Channiwala, S.A.; Ghosal, G.K. A correlation for calculating HHV from proximate analysis of solid fuels. Fuel 2005, 84, 487–494. [Google Scholar] [CrossRef]
- Toscan, A.; Morais, A.R.C.; Paixão, S.M.; Alves, L.; Andreaus, J.; Camassola, M.; Dillon, A.J.P.; Lukasik, R.M. High-pressure carbon dioxide/water pre-treatment of sugarcane bagasse and elephant grass: Assessment of the effect of biomass composition on process efficiency. Bioresour. Technol. 2017, 224, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Ronsse, F.; Nachenius, R.W.; Wolter, P. Carbonization of Biomass. Recent Advances in Thermo-Chemical Conversion of Biomass. 2015, pp. 293–324. Available online: https://www.sciencedirect.com/book/9780444632890/recent-advances-in-thermochemical-conversion-of-biomass (accessed on 26 February 2020).
- Chen, W.H.; Peng, J.; Bi, X.T. A state-of-the-art review of biomass torrefaction, densification and applications. Renew. Sustain. Energy Rev. 2015, 44, 847–866. [Google Scholar] [CrossRef]
- Mureddu, M.; Dessì, F.; Orsini, A.; Ferrara, F.; Pettinau, A. Air- and oxygen-blown characterization of coal and biomass by thermogravimetric analysis. Fuel 2018, 212, 626–637. [Google Scholar] [CrossRef]
- Parshetti, G.K.; Kent Hoekman, S.; Balasubramanian, R. Chemical, structural and combustion characteristics of carbonaceous products obtained by hydrothermal carbonization of palm empty fruit bunches. Bioresour. Technol. 2013, 135, 683–689. [Google Scholar] [CrossRef]
- Lin, Y.; Ma, X.; Ning, X.; Yu, Z. TGA-FTIR analysis of co-combustion characteristics of paper sludge and oil-palm solid wastes. Energy Convers. Manag. 2015, 89, 727–734. [Google Scholar] [CrossRef]
- Lin, Y.; Ma, X.; Peng, X.; Hu, S.; Yu, Z.; Fang, S. Effect of hydrothermal carbonization temperature on combustion behavior of hydrochar fuel from paper sludge. Appl. Therm. Eng. 2015, 91, 574–582. [Google Scholar] [CrossRef]
- He, C.; Giannis, A.; Wang, J.Y. Conversion of sewage sludge to clean solid fuel using hydrothermal carbonization: Hydrochar fuel characteristics and combustion behavior. Appl. Energy 2013, 111, 257–266. [Google Scholar] [CrossRef]
- Naderi, M. Surface Area: Brunauer-Emmett-Teller (BET). Progress in Filtration and Separation. 2015, pp. 585–608. Available online: https://www.sciencedirect.com/science/article/pii/B9780123847461000148 (accessed on 26 February 2020).
- Zhang, S.; Su, Y.; Xiong, Y.; Zhang, H. Physicochemical structure and reactivity of char from torrefied rice husk: Effects of inorganic species and torrefaction temperature. Fuel 2020, 262, 116667. [Google Scholar] [CrossRef]
- Zbiorowa, P. Poradnik Fizykochemiczny (Physicochemical Guide, in Polish); Wydawnictwa Naukowo-Techniczne: Warszawa, Poland, 1974. [Google Scholar]
- Lee, S.W.; Kim, Y.J.; Lee, Y.H.; Guim, H.; Han, S.M. Behavior and characteristics of amorphous calcium carbonate and calcite using CaCO3 film synthesis. Mater. Des. 2016, 112, 367–373. [Google Scholar] [CrossRef]
- Jerzak, W.; Murzyn, P.; Kuźnia, M.; Magiera, A. Trace elements retention in bottom ashes during coal combustion with hydrated lime additions. Energy Sources Part A Recover. Util. Environ. Eff. 2019. [Google Scholar] [CrossRef]
- RRUFF. Available online: http://rruff.info/ (accessed on 26 February 2020).
- Zheng, Y.; Tao, L.; Huang, Y.; Liu, C.; Wang, Z.; Zheng, Z. Improving aromatic hydrocarbon content from catalytic pyrolysis upgrading of biomass on a CaO/HZSM-5 dual-catalyst. J. Anal. Appl. Pyrolysis 2019, 140, 355–366. [Google Scholar] [CrossRef]
- Mlonka-Mędrala, A.; Magdziarz, A.; Gajek, M.; Nowińska, K.; Nowak, W. Alkali metals association in biomass and their impact on ash melting behaviour. Fuel 2020, 261, 116421. [Google Scholar] [CrossRef]
- Lin, J.; Ma, R.; Luo, J.; Sun, S.; Cui, C.; Fang, L.; Huang, H. Microwave pyrolysis of food waste for high-quality syngas production: Positive effects of a CO2 reaction atmosphere and insights into the intrinsic reaction mechanisms. Energy Convers. Manag. 2020, 206, 112490. [Google Scholar] [CrossRef]




| Proximate analysis ad (wt%) | |||
| Volatile matter | Fixed carbon | Ash | Moisture |
| 82.03 | 13.91 | 1.29 | 2.77 |
| Ultimate analysis (wt%) | |||
| C | H | O a | N |
| 45.84 | 6.44 | 45.84 | 0.17 |
| Sample | PSD | Char 500 °C | Char 600 °C | Char 700 °C |
|---|---|---|---|---|
| Tig, °C | 239.8 | 346.6 | 362.4 | 381.8 |
| Tb, °C | 456.5 | 481.3 | 493.9 | 512.6 |
| TDTG, °C | 318.8 | 437.8 | 459.1 | 471.6 |
| (dW/dt)max, wt. %/min | 9.98 | 15.68 | 12.85 | 13.58 |
| (dW/dt)av, wt. %/min | 1.23 | 1.19 | 1.19 | 1.16 |
| tp, min | 28.7 | 39.9 | 42.0 | 43.3 |
| ti, min | 21.1 | 31.4 | 33.0 | 34.9 |
| tf, min | 42.3 | 44.7 | 45.9 | 47.8 |
| Δt1/2, min | 24.1 | 37.3 | 38.4 | 40.2 |
| Di, wt. %/min3 | 0.0165 | 0.0125 | 0.0093 | 0.0090 |
| Df, 10−4 wt. %/min4 | 3.42 | 2.36 | 1.74 | 1.63 |
| S, 10−7 ∙min−2 ∙ °C−3 | 4.67 | 3.24 | 2.35 | 2.11 |
| Hf, °C | 949 | 1506 | 1596 | 1670 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sieradzka, M.; Gao, N.; Quan, C.; Mlonka-Mędrala, A.; Magdziarz, A. Biomass Thermochemical Conversion via Pyrolysis with Integrated CO2 Capture. Energies 2020, 13, 1050. https://doi.org/10.3390/en13051050
Sieradzka M, Gao N, Quan C, Mlonka-Mędrala A, Magdziarz A. Biomass Thermochemical Conversion via Pyrolysis with Integrated CO2 Capture. Energies. 2020; 13(5):1050. https://doi.org/10.3390/en13051050
Chicago/Turabian StyleSieradzka, Małgorzata, Ningbo Gao, Cui Quan, Agata Mlonka-Mędrala, and Aneta Magdziarz. 2020. "Biomass Thermochemical Conversion via Pyrolysis with Integrated CO2 Capture" Energies 13, no. 5: 1050. https://doi.org/10.3390/en13051050
APA StyleSieradzka, M., Gao, N., Quan, C., Mlonka-Mędrala, A., & Magdziarz, A. (2020). Biomass Thermochemical Conversion via Pyrolysis with Integrated CO2 Capture. Energies, 13(5), 1050. https://doi.org/10.3390/en13051050









