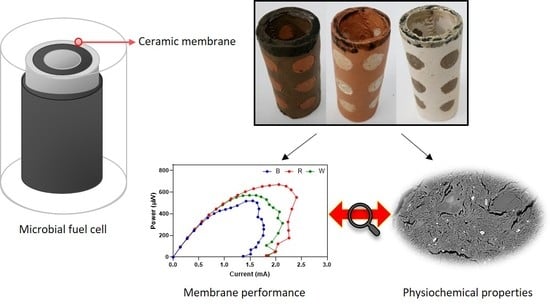
A Comprehensive Study of Custom-Made Ceramic Separators for Microbial Fuel Cells: Towards “Living” Bricks
Abstract
1. Introduction
2. Materials and Methods
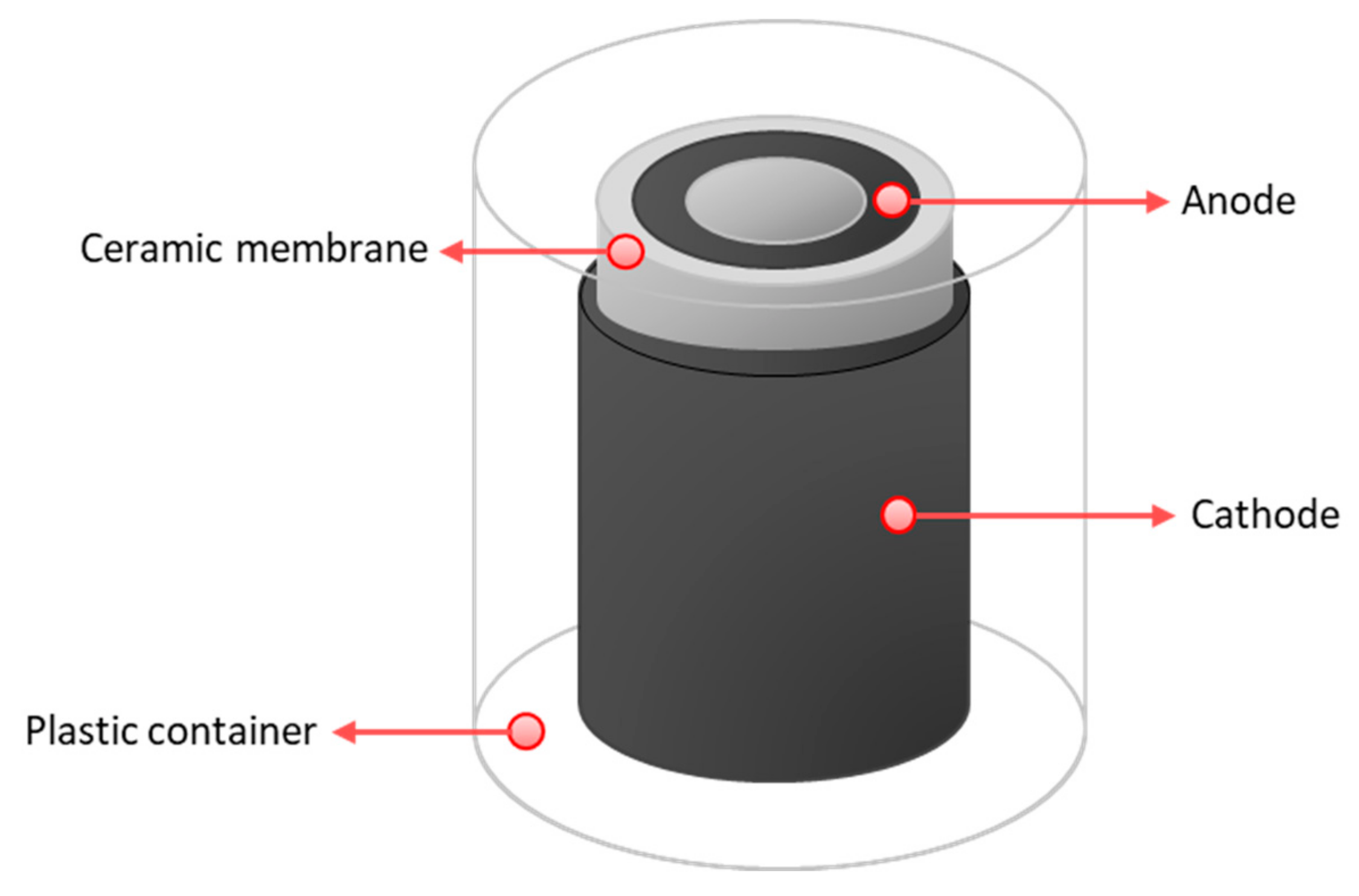
2.1. Custom-Made Ceramics
2.2. Microbial Fuel Cell Designs, Inoculation and Operation
2.3. Polarisation Test and Data Logging
2.4. Physiochemical Property Analysis of Ceramic Separators
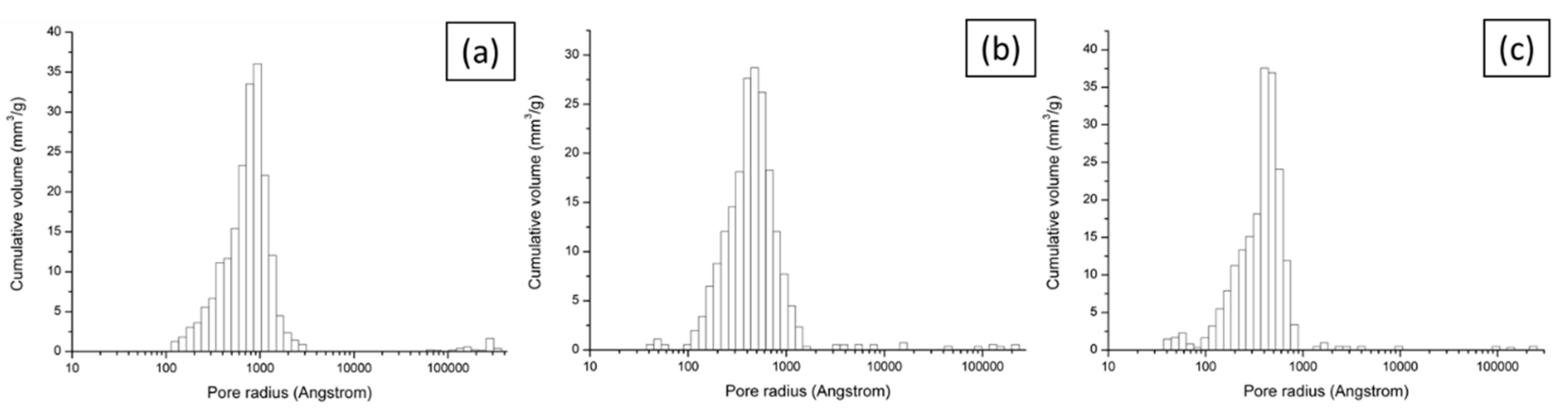
2.4.1. Porosity Investigation
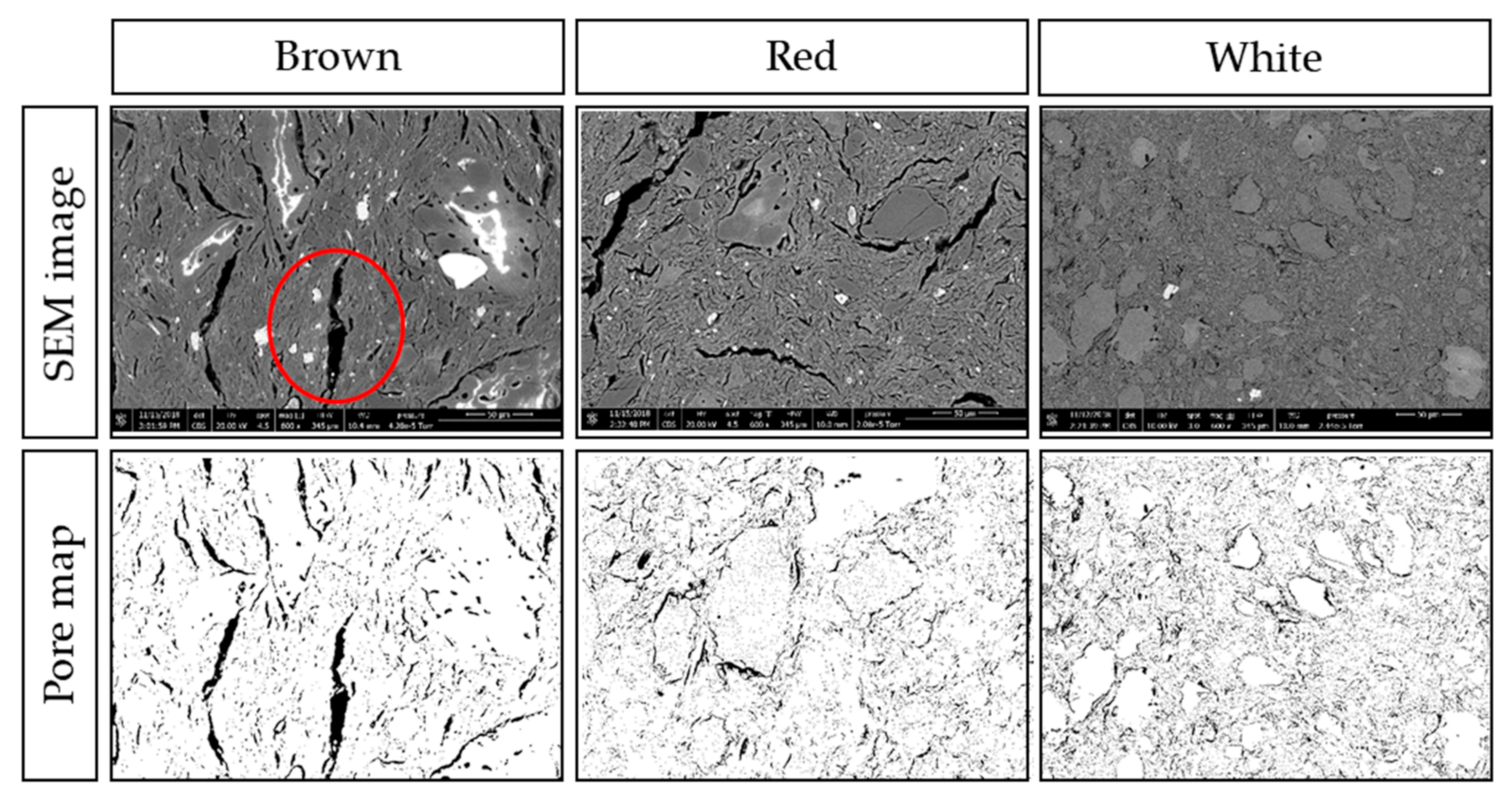
2.4.2. Composition Analysis
2.5. Statistical Analysis
3. Results and Discussion
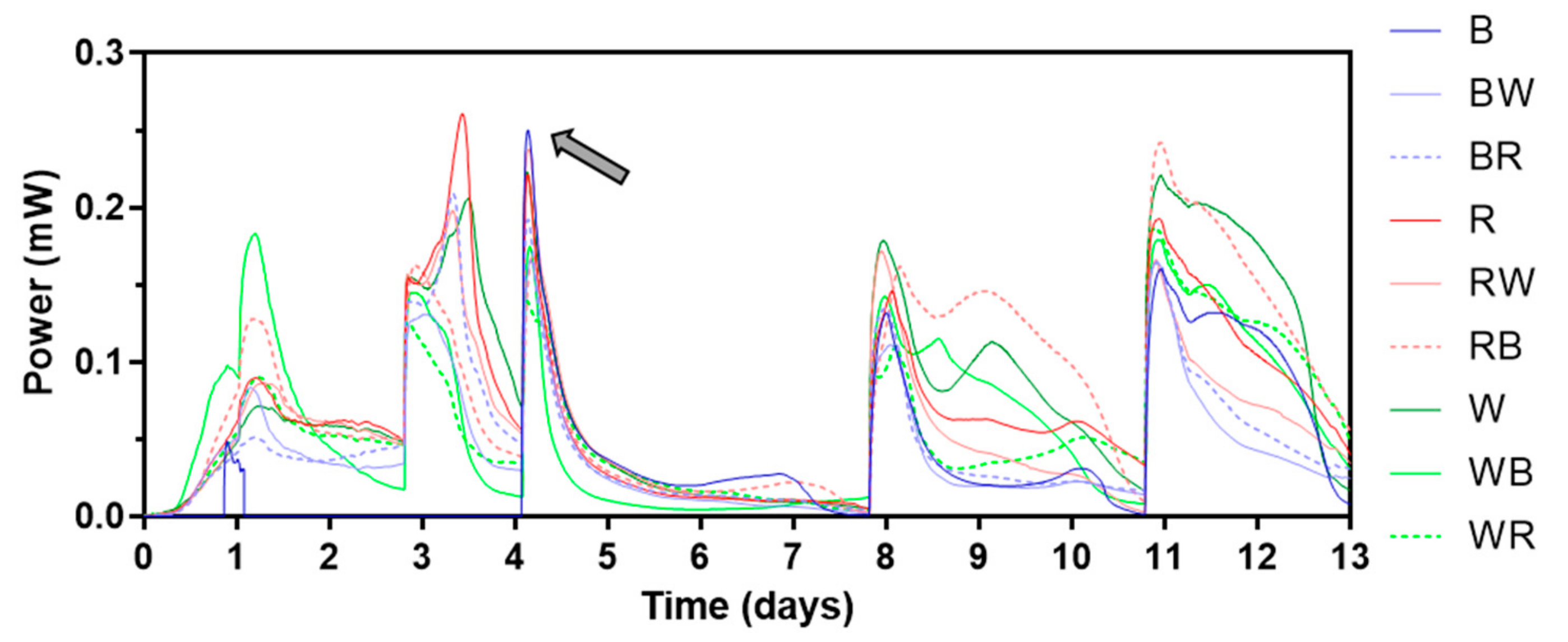
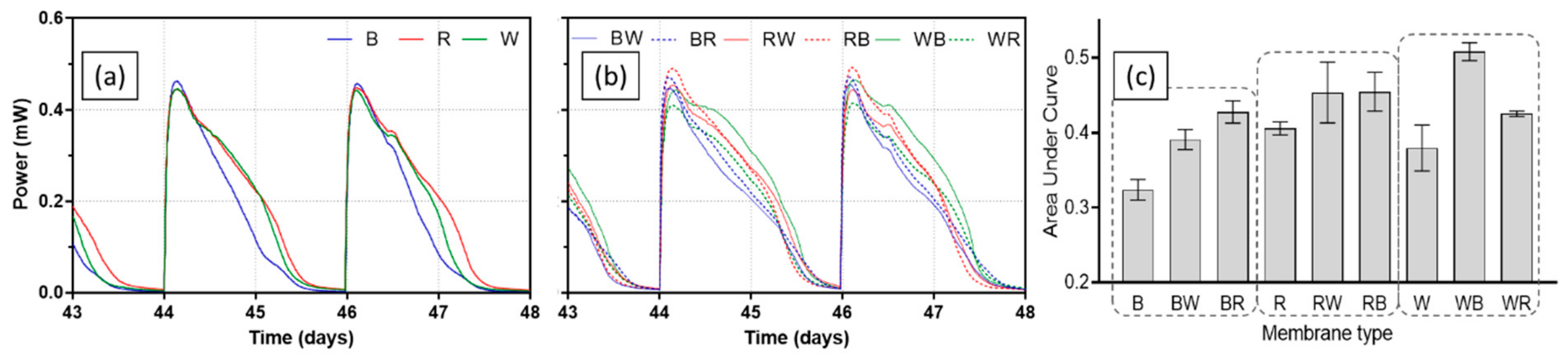
3.1. Effect of Ceramic Type on MFC Performance
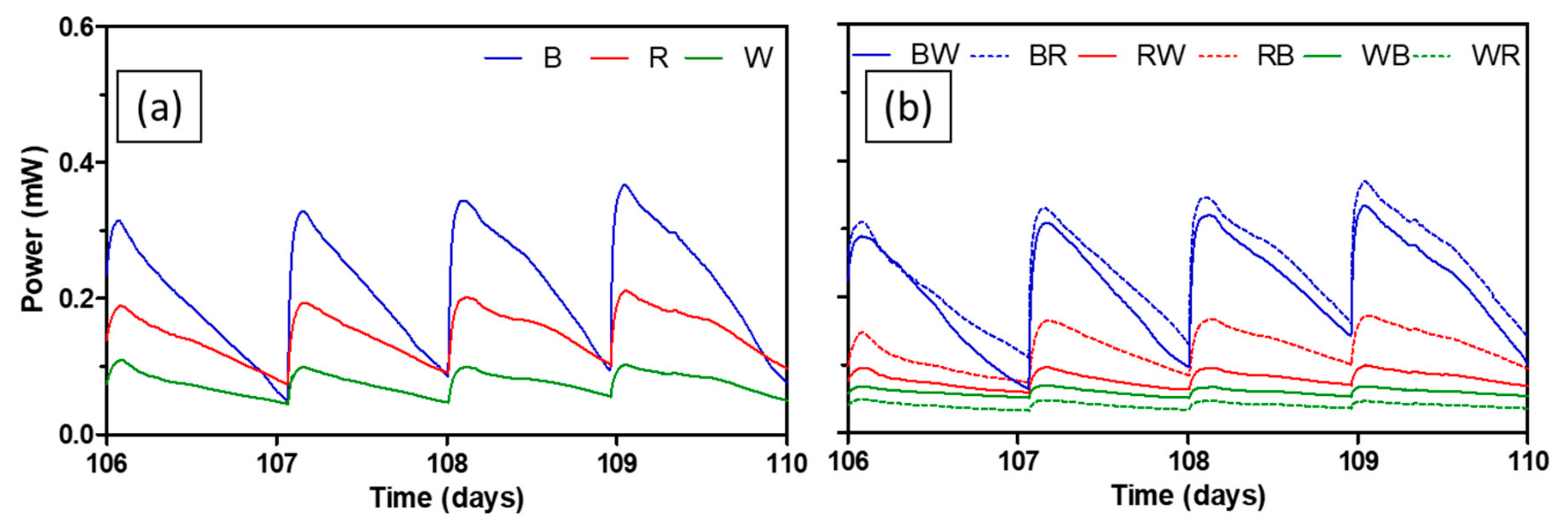
3.2. Long-Term Operation
3.3. Physiochemical Properties of Test Ceramics
3.4. Composition and Structure of Ceramic Separators
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dong, Y.; Qu, Y.; He, W.; Du, Y.; Liu, J.; Han, X.; Feng, Y. A 90-liter stackable baffled microbial fuel cell for brewery wastewater treatment based on energy self-sufficient mode. Bioresour. Technol. 2015, 195, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Goto, Y.; Yoshida, N. Scaling up Microbial Fuel Cells for Treating Swine Wastewater. Water 2019, 11, 1803. [Google Scholar] [CrossRef]
- Ge, Z.; He, Z. Long-term performance of a 200 liter modularized microbial fuel cell system treating municipal wastewater: Treatment, energy, and cost. Environ. Sci. Water Res. Technol. 2016, 2, 274–281. [Google Scholar] [CrossRef]
- Liang, P.; Duan, R.; Jiang, Y.; Zhang, X.; Qiu, Y.; Huang, X. One-year operation of 1000-L modularized microbial fuel cell for municipal wastewater treatment. Water Res. 2018, 141, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Online TOX/BOD System—KORBI. Available online: http://www.korbi.com/eng/products_type/habt-3000/?pageds=1&k=BOD (accessed on 6 September 2019).
- Trapero, J.R.; Horcajada, L.; Linares, J.J.; Lobato, J. Is microbial fuel cell technology ready? An economic answer towards industrial commercialization. Appl. Energy 2017, 185, 698–707. [Google Scholar] [CrossRef]
- Santoro, C.; Arbizzani, C.; Erable, B.; Ieropoulos, I. Microbial fuel cells: From fundamentals to applications. A review. J. Power Sources 2017, 356, 225–244. [Google Scholar] [CrossRef]
- Kondaveeti, S.; Kakarla, R.; Kim, H.S.; Kim, B.G.; Min, B. The performance and long-term stability of low-cost separators in single-chamber bottle-type microbial fuel cells. Environ. Technol. 2018, 39, 288–297. [Google Scholar] [CrossRef]
- Liu, H.; Logan, B.E. Electricity generation using an air-cathode single chamber microbial fuel cell in the presence and absence of a proton exchange membrane. Environ. Sci. Technol. 2004, 38, 4040–4046. [Google Scholar] [CrossRef]
- Palanisamy, G.; Jung, H.Y.; Sadhasivam, T.; Kurkuri, M.D.; Kim, S.C.; Roh, S.H. A comprehensive review on microbial fuel cell technologies: Processes, utilization, and advanced developments in electrodes and membranes. J. Clean. Prod. 2019, 221, 598–621. [Google Scholar] [CrossRef]
- Koók, L.; Bakonyi, P.; Harnisch, F.; Kretzschmar, J.; Chae, K.J.; Zhen, G.; Kumar, G.; Rózsenberszki, T.; Tóth, G.; Nemestóthy, N.; et al. Biofouling of membranes in microbial electrochemical technologies: Causes, characterization methods and mitigation strategies. Bioresour. Technol. 2019, 279, 327–338. [Google Scholar] [CrossRef]
- Bajracharya, S.; Sharma, M.; Mohanakrishna, G.; Dominguez Benneton, X.; Strik, D.P.; Sarma, P.M.; Pant, D. An overview on emerging bioelectrochemical systems (BESs): Technology for sustainable electricity, waste remediation, resource recovery, chemical production and beyond. Renew. Energy 2016, 98, 153–170. [Google Scholar] [CrossRef]
- Oliot, M.; Galier, S.; Roux de Balmann, H.; Bergel, A. Ion transport in microbial fuel cells: Key roles, theory and critical review. Appl. Energy 2016, 183, 1682–1704. [Google Scholar] [CrossRef]
- Dhar, B.R.; Lee, H.S. Membranes for bioelectrochemical systems: Challenges and research advances. Environ. Technol. 2013, 34, 1751–1764. [Google Scholar] [CrossRef] [PubMed]
- Jana, P.S.; Behera, M.; Ghangrekar, M.M. Performance comparison of up-flow microbial fuel cells fabricated using proton exchange membrane and earthen cylinder. Int. J. Hydrog. Energy 2010, 35, 5681–5686. [Google Scholar] [CrossRef]
- Gajda, I.; Greenman, J.; Santoro, C.; Serov, A.; Atanassov, P.; Melhuish, C.; Ieropoulos, I.A. Multi-functional microbial fuel cells for power, treatment and electro-osmotic purification of urine. J. Chem. Technol. Biotechnol. 2019, 94, 2098–2106. [Google Scholar] [CrossRef]
- Winfield, J.; Gajda, I.; Greenman, J.; Ieropoulos, I. A review into the use of ceramics in microbial fuel cells. Bioresour. Technol. 2016, 215, 296–303. [Google Scholar] [CrossRef]
- Santoro, C.; Flores-Cadengo, C.; Soavi, F.; Kodali, M.; Merino-Jimenez, I.; Gajda, I.; Greenman, J.; Ieropoulos, I.; Atanassov, P. Ceramic microbial fuel cells stack: Power generation in standard and supercapacitive mode. Sci. Rep. 2018, 8, 3281. [Google Scholar] [CrossRef]
- Baker, R.W. Membrane Separation. In Encyclopedia of Separation Science; Elsevier: Cambridge, MA, USA, 2000; pp. 189–210. [Google Scholar]
- Winfield, J.; Greenman, J.; Huson, D.; Ieropoulos, I. Comparing terracotta and earthenware for multiple functionalities in microbial fuel cells. Bioprocess Biosyst. Eng. 2013, 36, 1913–1921. [Google Scholar] [CrossRef]
- Pasternak, G.; Greenman, J.; Ieropoulos, I. Comprehensive study on ceramic membranes for low-cost microbial fuel cells. ChemSusChem 2016, 9, 88–96. [Google Scholar] [CrossRef]
- Merino Jimenez, I.; Greenman, J.; Ieropoulos, I. Electricity and catholyte production from ceramic MFCs treating urine. Int. J. Hydrog. Energy 2017, 42, 1791–1799. [Google Scholar] [CrossRef]
- Khalili, H.B.; Mohebbi-Kalhori, D.; Afarani, M.S. Microbial fuel cell (MFC) using commercially available unglazed ceramic wares: Low-cost ceramic separators suitable for scale-up. Int. J. Hydrog. Energy 2017, 42, 8233–8241. [Google Scholar] [CrossRef]
- Winfield, J.; Chambers, L.D.; Rossiter, J.; Greenman, J.; Ieropoulos, I. Urine-activated origami microbial fuel cells to signal proof of life. J. Mater. Chem. A 2015, 3, 7058–7065. [Google Scholar] [CrossRef]
- Living Architecture—Transform Our Habitats from Inert Spaces into Programmable Sites. Available online: https://livingarchitecture-h2020.eu/ (accessed on 10 September 2019).
- Djangang, C.N.; Elimbi, A.; Melo, U.C.; Lecomte, G.L.; Nkoumbou, C.; Soro, J.; Bonnet, J.P.; Blanchart, P.; Njopwouo, D. Sintering of clay-chamotte ceramic composites for refractory bricks. Ceram. Int. 2008, 34, 1207–1213. [Google Scholar] [CrossRef]
- You, J.; Walter, X.A.; Greenman, J.; Melhuish, C.; Ieropoulos, I. Stability and reliability of anodic biofilms under different feedstock conditions: Towards microbial fuel cell sensors. Sens. Bio-Sens. Res. 2015, 6, 43–50. [Google Scholar] [CrossRef]
- ASTM C373-18, Standard Test Methods for Determination of Water Absorption and Associated Properties by Vacuum Method for Pressed Ceramic Tiles and Glass Tiles and Boil Method for Extruded Ceramic Tiles and Non-Tile Fired Ceramic Whiteware Products; ASTM International: West Conshohocken, PA, USA, 2018.
- Logan, B.E.; Hamelers, B.; Rozendal, R.; Schröder, U.; Keller, J.; Freguia, S.; Aelterman, P.; Verstraete, W.; Rabaey, K. Microbial fuel cells: Methodology and technology. Environ. Sci. Technol. 2006, 40, 5181–5192. [Google Scholar] [CrossRef] [PubMed]
- Gajda, I.; Greenman, J.; Melhuish, C.; Ieropoulos, I. Simultaneous electricity generation and microbially-assisted electrosynthesis in ceramic MFCs. Bioelectrochemistry 2015, 104, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Malvankar, N.S.; Tuominen, M.T.; Lovley, D.R. Biofilm conductivity is a decisive variable for high-current-density Geobacter sulfurreducens microbial fuel cells. Energy Environ. Sci. 2012, 5, 5790. [Google Scholar] [CrossRef]
- Kundu, P.P.; Dutta, K.; Kumar, P.; Bharti, R.P.; Kumar, V.; Kundu, P.P. Polymer Electrolyte Membranes for Microbial Fuel Cells: Part A. Nafion-Based Membranes. In Progress and Recent Trends in Microbial Fuel Cells; Elsevier: Amsterdam, The Netherlands, 2018; pp. 47–72. ISBN 978-0-444-64017-8. [Google Scholar]
- Natarajan, K.A. Biofouling and microbially influenced corrosion. In Biotechnology of Metals, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 355–393. ISBN 978-0-12-804022-5. [Google Scholar]
- Gajda, I.; Greenman, J.; Melhuish, C.; Santoro, C.; Li, B.; Cristiani, P.; Ieropoulos, I. Electro-osmotic-based catholyte production by Microbial Fuel Cells for carbon capture. Water Res. 2015, 86, 108–115. [Google Scholar] [CrossRef]
- Stoerzinger, K.A.; Risch, M.; Han, B.; Shao-Horn, Y. Recent Insights into Manganese Oxides in Catalyzing Oxygen Reduction Kinetics. ACS Catal. 2015, 5, 6021–6031. [Google Scholar] [CrossRef]
- Zhang, Y.; Hu, Y.; Li, S.; Sun, J.; Hou, B. Manganese dioxide-coated carbon nanotubes as an improved cathodic catalyst for oxygen reduction in a microbial fuel cell. J. Power Sources 2011, 196, 9284–9289. [Google Scholar] [CrossRef]
- Rout, S.; Nayak, A.K.; Varanasi, J.L.; Pradhan, D.; Das, D. Enhanced energy recovery by manganese oxide/reduced graphene oxide nanocomposite as an air-cathode electrode in the single-chambered microbial fuel cell. J. Electroanal. Chem. 2018, 815, 1–7. [Google Scholar] [CrossRef]
- Shahbazi Farahani, F.; Mecheri, B.; Reza Majidi, M.; Costa de Oliveira, M.A.; D’Epifanio, A.; Zurlo, F.; Placidi, E.; Arciprete, F.; Licoccia, S. MnOx-based electrocatalysts for enhanced oxygen reduction in microbial fuel cell air cathodes. J. Power Sources 2018, 390, 45–53. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, C.; Zhuang, L.; Li, W.; Zhou, S.; Zhang, J. Manganese dioxide as an alternative cathodic catalyst to platinum in microbial fuel cells. Biosens. Bioelectron. 2009, 24, 2825–2829. [Google Scholar] [CrossRef] [PubMed]

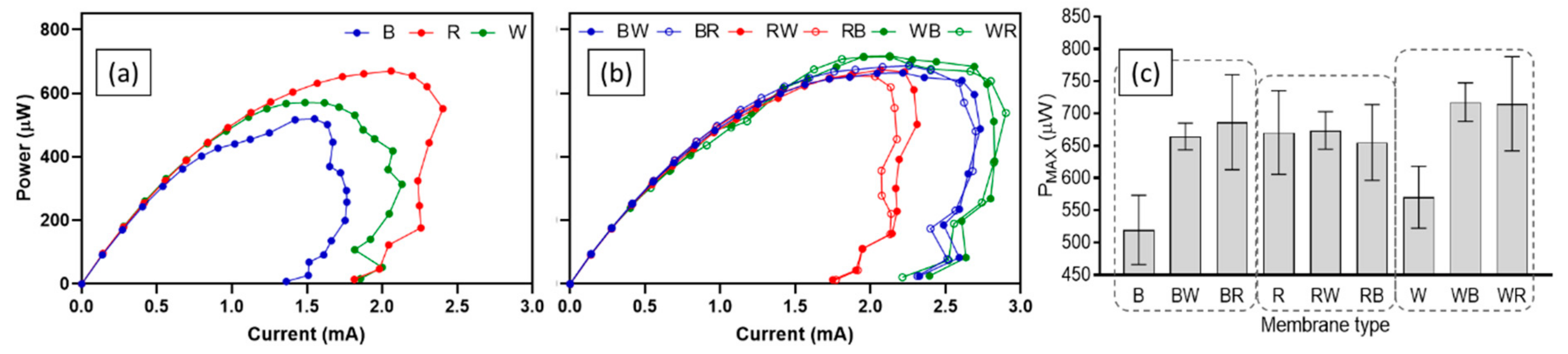

| Density 1) (g·cm 3) | Apparent Porosity 1) (%) | Total Porosity 2) (%) | Average Pore Diameter 2) (nm) | Maximum Power (µW) | Maximum Power Density 3) (W·m−3) | |
|---|---|---|---|---|---|---|
| Brown | 2.7 | 23.3 | 22.0 | 188 | 519.9 | 52.0 |
| Red | 2.7 | 25.6 | 23.7 | 95 | 670.5 | 67.1 |
| White | 2.6 | 26.8 | 23.9 | 81 | 570.5 | 57.1 |
| Chemical Composition 1) (wt.%) | CO2 | SiO2 | Al2O3 | TiO2 | Fe2O3 | CaO | MgO | K2O | Na2O | MnO | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Brown | MSDS | - | 64.5 | 20.0 | 1.3 | 6.5 | 0.3 | 0.4 | 2.2 | 0.1 | 4.8 |
| EDXS | 13.9 | 57.0 | 17.7 | 1.4 | 4.2 | 0.3 | 0.4 | 2.7 | 0.3 | 1.9 | |
| Red | MSDS | 68.9 | 20.5 | 1.3 | 6.0 | 0.3 | 0.4 | 2.5 | 0.1 | 0.0 | |
| EDXS | 10.7 | 57.0 | 21.5 | 1.6 | 10.3 | 0.4 | 0.6 | 3.3 | 0.3 | - | |
| White | MSDS | 72.0 | 22.0 | 1.8 | 1.0 | 0.3 | 0.2 | 2.3 | 0.3 | 0.0 | |
| EDXS | 18.1 | 51.3 | 21.5 | 2.2 | 2.7 | - | 0.5 | 3.2 | 0.3 | - | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
You, J.; Wallis, L.; Radisavljevic, N.; Pasternak, G.; Sglavo, V.M.; Hanczyc, M.M.; Greenman, J.; Ieropoulos, I. A Comprehensive Study of Custom-Made Ceramic Separators for Microbial Fuel Cells: Towards “Living” Bricks. Energies 2019, 12, 4071. https://doi.org/10.3390/en12214071
You J, Wallis L, Radisavljevic N, Pasternak G, Sglavo VM, Hanczyc MM, Greenman J, Ieropoulos I. A Comprehensive Study of Custom-Made Ceramic Separators for Microbial Fuel Cells: Towards “Living” Bricks. Energies. 2019; 12(21):4071. https://doi.org/10.3390/en12214071
Chicago/Turabian StyleYou, Jiseon, Lauren Wallis, Nevena Radisavljevic, Grzegorz Pasternak, Vincenzo M. Sglavo, Martin M Hanczyc, John Greenman, and Ioannis Ieropoulos. 2019. "A Comprehensive Study of Custom-Made Ceramic Separators for Microbial Fuel Cells: Towards “Living” Bricks" Energies 12, no. 21: 4071. https://doi.org/10.3390/en12214071
APA StyleYou, J., Wallis, L., Radisavljevic, N., Pasternak, G., Sglavo, V. M., Hanczyc, M. M., Greenman, J., & Ieropoulos, I. (2019). A Comprehensive Study of Custom-Made Ceramic Separators for Microbial Fuel Cells: Towards “Living” Bricks. Energies, 12(21), 4071. https://doi.org/10.3390/en12214071