1. Introduction
Since 1950, the semiconductor manufacturing industry has continued to grow, raising concerns about the increasing emissions of perfluorinated compounds (PFCs) [
1]. PFCs are characterized by a very high global-warming potential, meaning that they produce a large environmental impact, even at low concentrations. As a result, the Kyoto Protocol (COP3) in 1997 set country-specific targets for reducing PFCs. In 2018, COP24 established guidelines for certain regulations on PFCs in developing countries, highlighting the urgency of the task of eliminating PFCs. Methods for eliminating PFC gas used in the semiconductor manufacturing industry include combustion methods, which burn the PFC gas together with such fuels as oil and natural gas to decompose it into hydrogen fluoride and carbon dioxide [
2,
3], and catalytic methods, which use catalysts to decompose the gas into hydrogen fluoride and carbon dioxide by means of the hydrolysis reaction [
4,
5]. The hydrogen fluoride generated during treatment is dealt with by dissolving it in water and processing it as hydrogen fluoride solution, or by causing it to react with calcium hydroxide to immobilize it as CaF
2 [
6]. Because the catalytic and CaF
2 fixing methods are not suitable for large-scale treatment of PFCs, large-scale treatment facilities have used the combustion method followed by wastewater processing. In the combustion method, NO
x (NO + NO
2) is generated as a byproduct of the decomposition of the PFCs. NO
x, which is generated mainly by the combustion of fuel, is a harmful substance that causes photochemical oxidation processes and other chemical changes in the atmosphere and produces aerosols, such as nitric acid (HNO
3), that cause acid rain. Selective catalytic reduction (SCR) is a common technique for processing NO
x. In the combustion method, however, immediately after the PFCs are burned, a water spray is applied to dissolve the resulting hydrogen fluoride (HF) in water. The problem for SCR is that the water spray brings the target exhaust gas down to a normal temperature, at which the catalyst cannot be activated. Furthermore, the HF can sometimes poison the catalyst for the SCR [
7,
8], which poses a problem for the treatment of the NO
x generated during processing of the PFCs.
Plasma–chemical methods that use nonthermal plasma have been proposed as a method of removing the NO
x at low temperatures [
9,
10,
11,
12,
13,
14]. The plasma–chemical method oxidizes NO to NO
2 by using nonthermal plasma to promote oxidation, and then it applies a scrubber to reduce the NO
2 to N
2. Our research group has achieved highly efficient removal of NO
x through treatment by a combined process of indirect oxidation by nonthermal plasma together with a sodium sulfite (Na
2SO
3) scrubber [
9,
10,
11,
12]. Kim et al. have also achieved NO
x removal efficiency of more than 80% using nonthermal plasma–chemical methods to treat the NO
x discharged from semiconductor plants [
15]. Comparing the NO
x removal efficiency of two different reducing agents, Na
2S and Na
2SO
3, Kim et al. found that Na
2S was highly efficient at removing NO
x at 1/10 the concentration of Na
2SO
3 (0.1 mass%). Although the cost of using Na
2S is approximately three times higher than that of using Na
2SO
3, using Na
2S as the reducing agent suppresses the reaction of the exhaust gas with oxygen, making high-efficiency treatment of NO
x possible at 1/10 concentration (0.1 mass%) and reducing the cost of treating NO
x to one-third the cost of Na
2SO
3 [
15].
With respect to dielectric barrier discharge (DBD) plasma excitation, research has been conducted on the effect of changing the shape of the electrode on its DBD discharge characteristics and ozone-generating characteristics [
16]. This research shows that the discharge energy efficiency and ozone generation efficiency are improved by using a multipoint electrode or trench electrode rather than a plate electrode. In concentric cylindrical plasma reactors, which are commonly used in environmental plasma processing, the high voltage is applied perpendicular to the direction of the gas flow, which means the voltage value imposes limits on the possible cross-sectional area or distance between electrodes. Parallelization is therefore required to increase the processing flow rate. However, in the blade-barrier electrode-based plasma reactor proposed in this study, the discharge direction is parallel to the gas flow, making it suitable for high-flow treatment of exhaust gas with no limit on the flow rate, and promising a highly efficient treatment of exhaust gas.
As mentioned above, previous studies have evaluated individual performance such as NOx reduction by chemical scrubber and NO oxidation by ozone generator, but the performance of the plasma-chemical hybrid technique has not been clarified. In the present study, a method for treating NOx is considered that combines the nonthermal plasma oxidation method at normal temperatures with a wet scrubber. For the plasma process, there are two different plasma oxidation methods: a direct oxidation method, which irradiates plasma directly into the exhaust gas, and an indirect oxidation method, which combines the exhaust with radical gas irradiated with plasma in the air. NO in the simulated exhaust gas is oxidized to NO2 by using a blade-barrier electrode reactor as a plasma reactor, and then NOx reduction is performed by Na2S scrubber.
3. Experimental Apparatus and Method
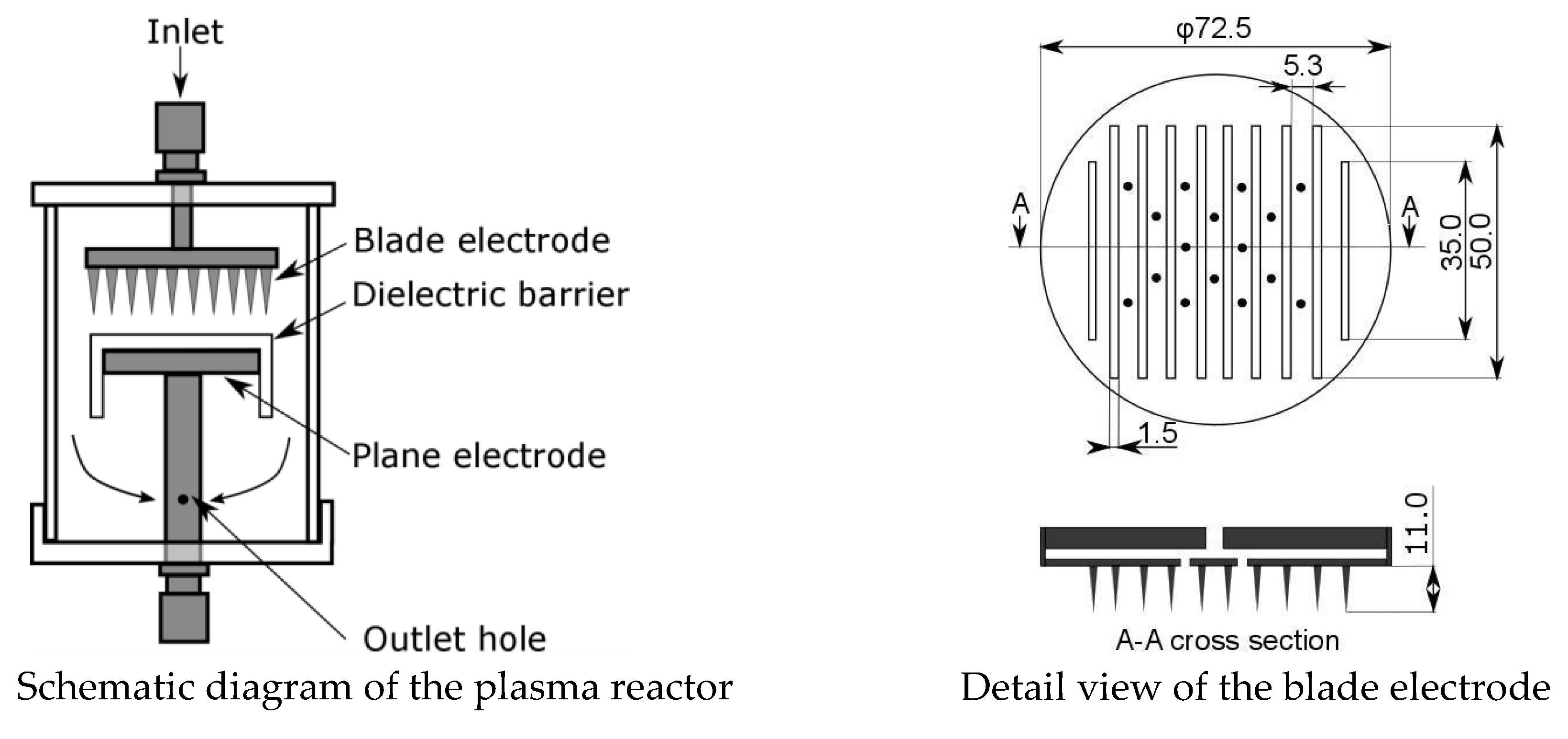
In this study, a blade-barrier electrode plasma reactor is used to generate ozone and radical oxygen for the purpose of oxidizing the NO, as shown in Equations (3) and (4). A schematic diagram of the blade-barrier plasma reactor and a detailed view of the blade electrode are shown in
Figure 1. To increase the strength of the electric field, a multihole (φ2 × 16) blade electrode is used. The gas flows from the top of the reactor, passes through the multihole blade electrode, and enters the discharge part. After the reaction in the discharge part, the gas flows out of the reactor through the holes in the lower electrode (φ2 × 4). To elicit DBD discharge, a dielectric barrier made of acrylic is installed on the lower electrode. The upper electrode is equipped with 10 blades shaped like knife edges. The distance between the blades is 5.3 mm, the width of each blade is 1.5 mm, and the height is 11 mm. The two end blades are 35 mm in length, and the remaining eight blades are 50 mm in length. The upper and lower electrodes are 72.5 mm in diameter with a distance between the electrodes of 7 mm, including the thickness of the dielectric barrier.
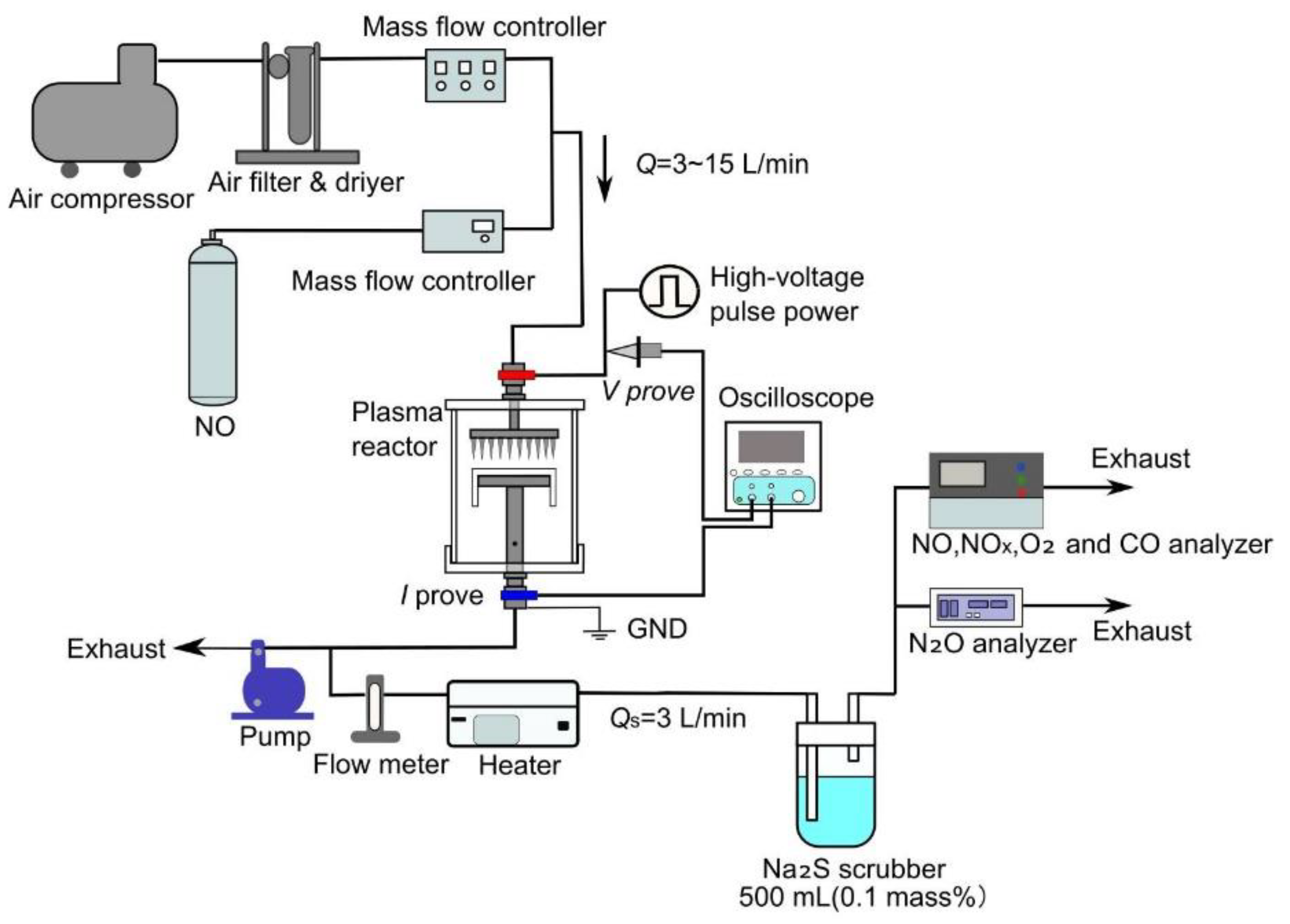
In this study, the practicality of the proposed NO
x removal device is considered by two methods of NO
x removal: direct oxidation and indirect oxidation. The direct oxidation method enables simulated exhaust gas to flow directly into the plasma reactor, where the nonthermal plasma directly irradiates the simulated exhaust gas, oxidizing the NO gas into NO
2. A schematic diagram of the experimental setup for the direct plasma oxidation method is shown in
Figure 2. The external air is compressed with a compressor, the fine particles are removed with a filter, and the moisture is removed with a desiccant to dry the air. The dry air is mixed with NO gas supplied from a gas cylinder (2%, N
2 balance), with the flow rate and concentration regulated by mass flow controllers. After the simulated exhaust gas is passed through the plasma reactor and irradiated with the nonthermal plasma, it is passed through a heater at a flow rate of 3 L/min and heated to 270 °C to remove the O
3 that is generated in the plasma reactor. The remaining gas is then exhausted. After removal of the O
3, the simulated exhaust gas passes through the reduction process by a bubble injection-type Na
2S scrubber and then flows into a gas analyzer. The respective concentrations of NO, NO
x (NO+NO
2), CO, and N
2O were measured with a gas analyzer (Horiba Co. Ltd., PG-240, Kyoto, Japan, chemiluminescense for NO, NO
x, infrared absorption for CO and O
2 for zirconia method,) and N
2O meter (Horiba Co. Ltd., VIA-510, Kyoto, Japan, infrared absorption for N
2O). To ensure that the pressure inside the plasma reactor remained at atmospheric pressure during the experiment, the pressure was monitored upstream of the reactor. In this experiment, the concentration of NO was set at 300 ppm, and the total flow rate flowing into the reactor was changed to 3, 5, 10, and 15 L/min.
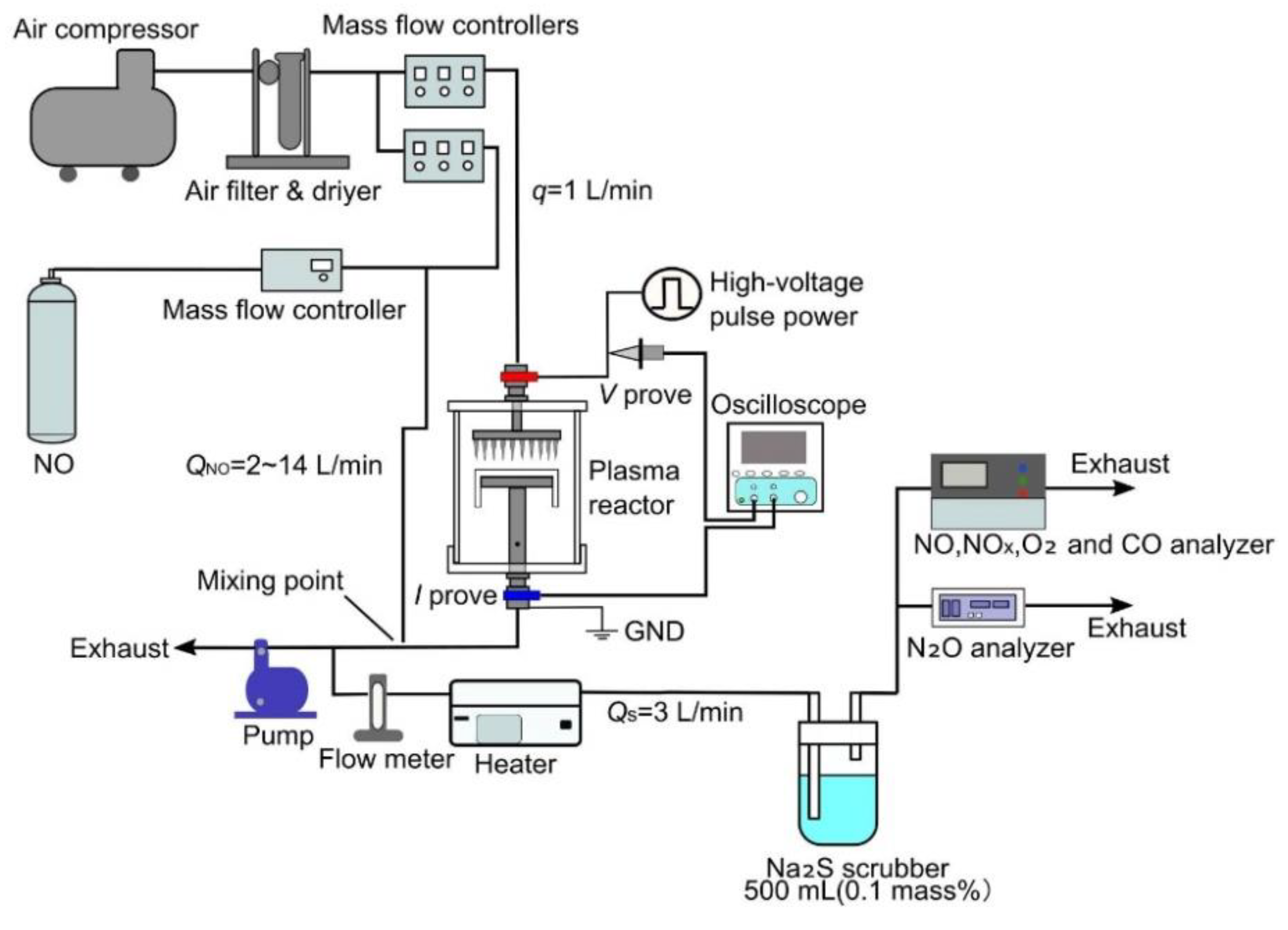
A schematic diagram of the experimental setup for the indirect plasma oxidation method is shown in
Figure 3. Unlike the direct plasma oxidation method, in which the simulated exhaust gas is directly irradiated with nonthermal plasma, in the indirect plasma oxidation method where only dry air flows through the reactor, and ozone is generated by irradiating this air with the nonthermal plasma. This ozone is then combined with the simulated exhaust gas to oxidize the NO in the gas to NO
2. In the direct plasma oxidation method, when the exhaust gas is at a high temperature, the gas volume expands compared to room temperature. This decreases the residence time of the gas and reduces oxidation performance. In contrast, the indirect plasma oxidation method offers the advantage that the air is treated at normal temperatures, so the residence time does not change, and oxidation performance does not decrease. The external air is compressed with a compressor, the fine particles are removed with a filter, and the moisture is removed with a desiccant to dry the air. The dry air is branched, with one part going to the plasma reactor and the other part mixed with NO gas supplied from a gas cylinder (2%, N
2 balance). The dry air irradiated by the nonthermal plasma in the plasma reactor is combined with the simulated exhaust gas and then passed through the heater to remove the O
3. The residence time of the mixture between the mixing point and the heater is 0.2–0.5 s. The residence time is enough time to react and mix NO with ozone since oxidation of NO is immediately occurred by ozone. Especially in this experiment, a tube with diameter of 4mm is used, which is quite small and considers that NO and ozone mix completely in the tube. In this experiment, the concentration of NO was set at 300 ppm and the flow rate of dry air flowing into the reactor at 1.0 L/min. The total flow rate of the air passing through the reactor and mixing with the exhaust gas was changed to 3, 5, 10, and 15 L/min.
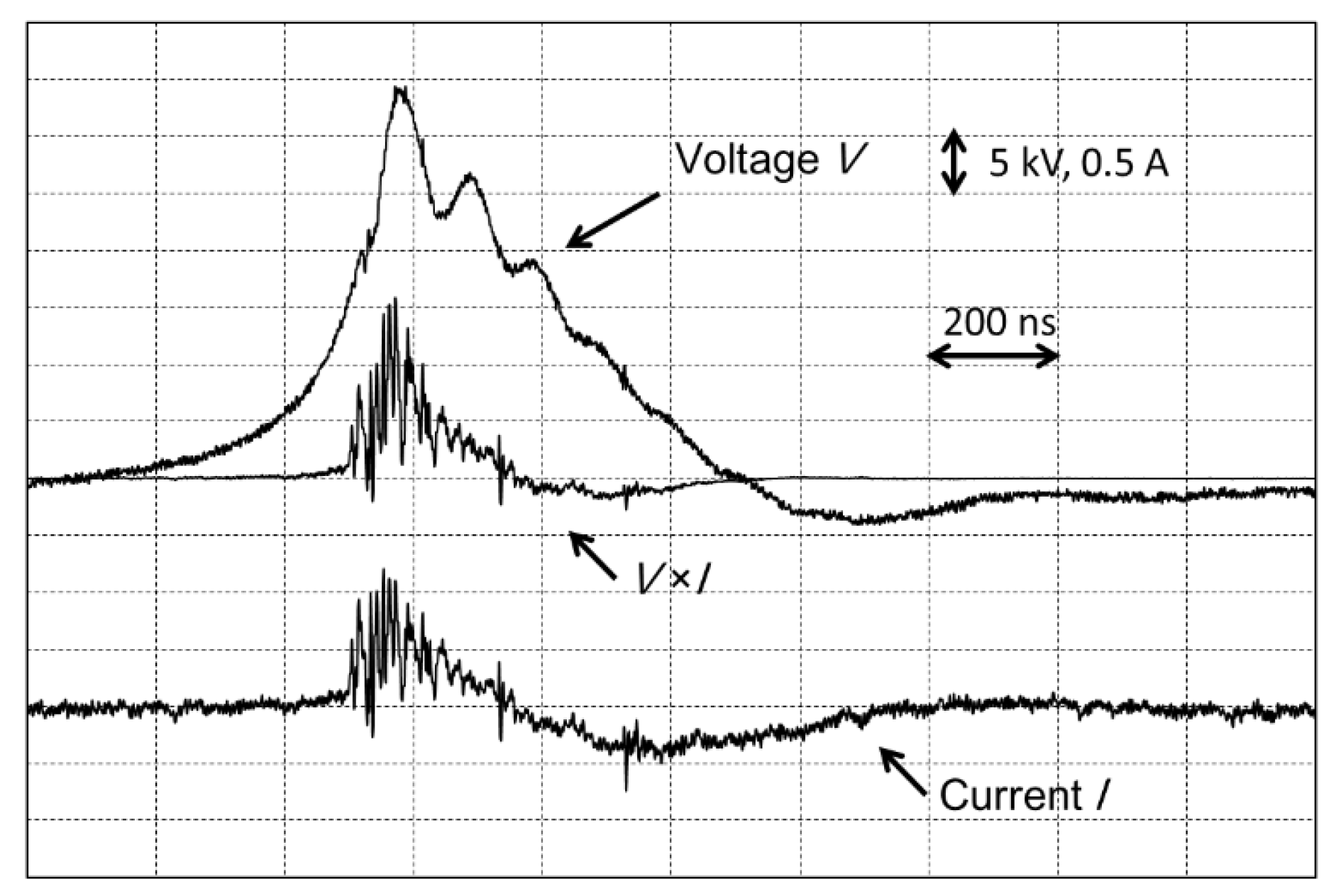
The voltage applied to the plasma reactor was the same for the direct and indirect methods. An insulated gate bipolar transistor power supply was used to apply high-voltage pulses (max. 38 kV) at the following frequencies: 210, 420, 630, 840, and 1020 Hz. The discharge power in the plasma reactor was measured by an oscilloscope (Yokogawa Electric Corporation DL1740, Tokyo, Japan) using a high-voltage probe (Tektronix Co. Ltd., P6015A, Beaverton, USA) and an electric current probe (Tektronix Co. Ltd., P6021, Beaverton, USA).
Figure 4 shows typical waveforms of the voltage, the current, and the product of the voltage and current as measured by the oscilloscope. To find the discharge power, the integral of the product of the voltage and current was calculated over one wavelength and then multiplied by the frequency. For the wet scrubber, sodium sulfide (Na
2S, 9-hydrate, Sigma-Aldrich, Japan Co. LLC, Tokyo, Japan) was used in an aqueous solution of 500 mL with an initial concentration of 0.1 mass%. Before processing, the pH was measured as 11.9 ± 0.2, and the oxidation-reduction potential (ORP) was measured as −300 ± 20 mV using a pH/ORP meter (Horiba Co. Ltd., D-53, Kyoto, Japan). The pH level requires careful attention because if it drops to less than 10, the Na
2S will react with the acid to generate highly toxic H
2S [
18]. An H
2S detector (Honeywell International Inc., ToxiRAE3-H
2S) is therefore installed near the Na
2S scrubber vessel for the duration of the experiment to ensure that the pH would never drop to less than 10.
To evaluate the combined process of the dry plasma process and the wet chemical process using the wet scrubber, in this study NOx concentrations at various discharge power are measured. Furthermore, NO conversion efficiency and NOx removal energy efficiency are calculated and compared.
4. Results and Discussion
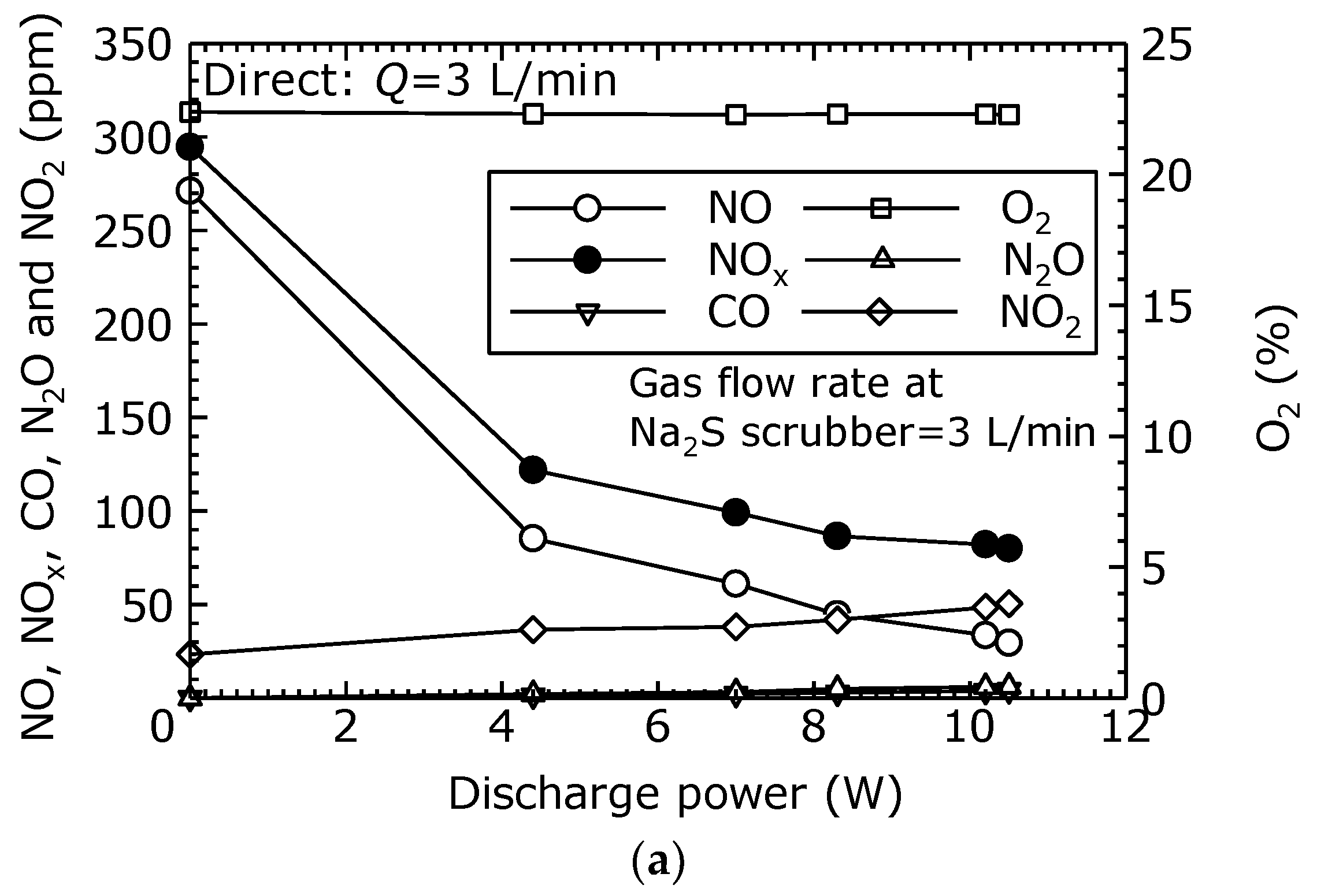
To evaluate the NO oxidation and removal characteristics of the treatment by the combined dry plasma and wet chemical processes using the direct oxidation method, the components of the treated gas relative to the discharge power are measured.
Figure 5a–d show the gas concentrations after the combined process under each flow condition (
Q = 3, 5, 10, and 15 L/min). From
Figure 5a–d, it is seen that the NO decreased as the discharge power increased. Furthermore, as the NO decreased, the NO
2 increased, causing a reduction in NO
x as the NO
2 is removed by the Na
2S scrubber. In
Figure 5a, as the discharge power rose from its initial value to 10.5 W (
Vp-p = 25.6 kV), the NO concentration decreased from 271 to 30 ppm, and the NO
x concentration decreased from 295 to 80 ppm. This represents an 89% reduction of NO and a 73% reduction of NO
x. In
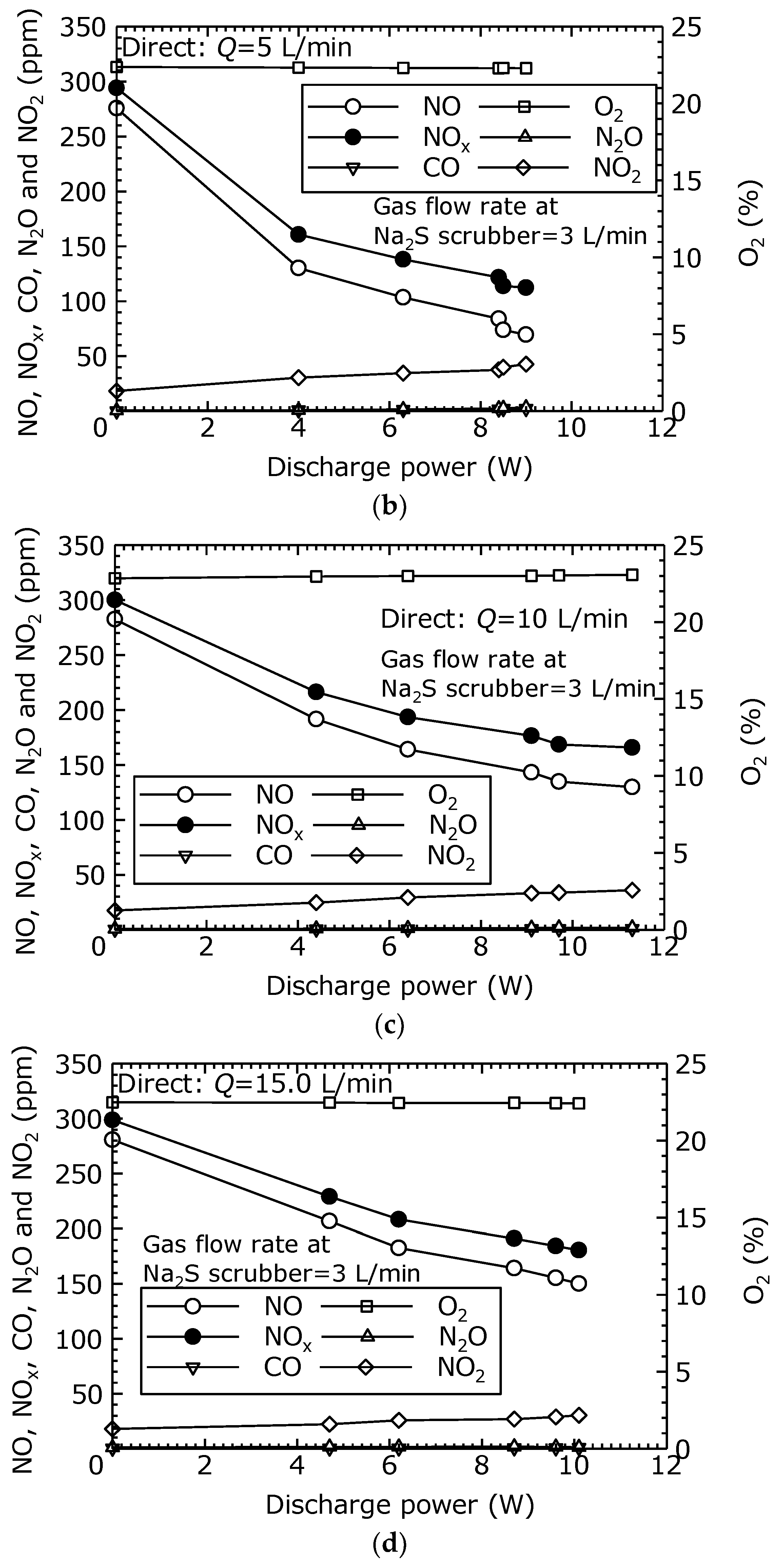
Figure 5b, when the discharge power was 9.0 W (
Vp-p = 24.7 kV), the NO was reduced by 75% and the NO
x by 62%. In
Figure 5c, when the discharge power was 11.3 W (
Vp-p = 26.6 kV), the NO was reduced by 54% and the NO
x by 45%. In
Figure 5d, when the discharge power was 10.1 W (
Vp-p = 24.9 kV), the NO was reduced by 47% and the NO
x by 40%. These results demonstrate that treatment by the combined dry plasma and wet chemical processes effectively reduce NO
x. In
Figure 5a–d, CO and N
2O were each less than 10 ppm.
The NO-to-NO
2 conversion characteristics of the dry plasma process are evaluated by the direct oxidation method. The relationship between the discharge power and the NO conversion efficiency at different flow rates is shown in
Figure 6. In these experiments, the oxygen concentration was 22% to 24% at all flow rates. It is noted that NO
2 is produced at zero discharge power. It is known that a portion of NO converts to NO
2 upon coming into contact with oxygen by following reaction (2NO + O
2 → 2NO
2).
Figure 6 shows that the NO conversion efficiency increases with an increase in discharge power at any flow rate. It also shows that the NO conversion efficiency increased as the flow rate decreased. As the flow rate increased, it reduced the residence time of the simulated exhaust gas in the plasma reactor, thereby decreasing the amount of oxidation promotion reactions shown in Equations (3) and (4).
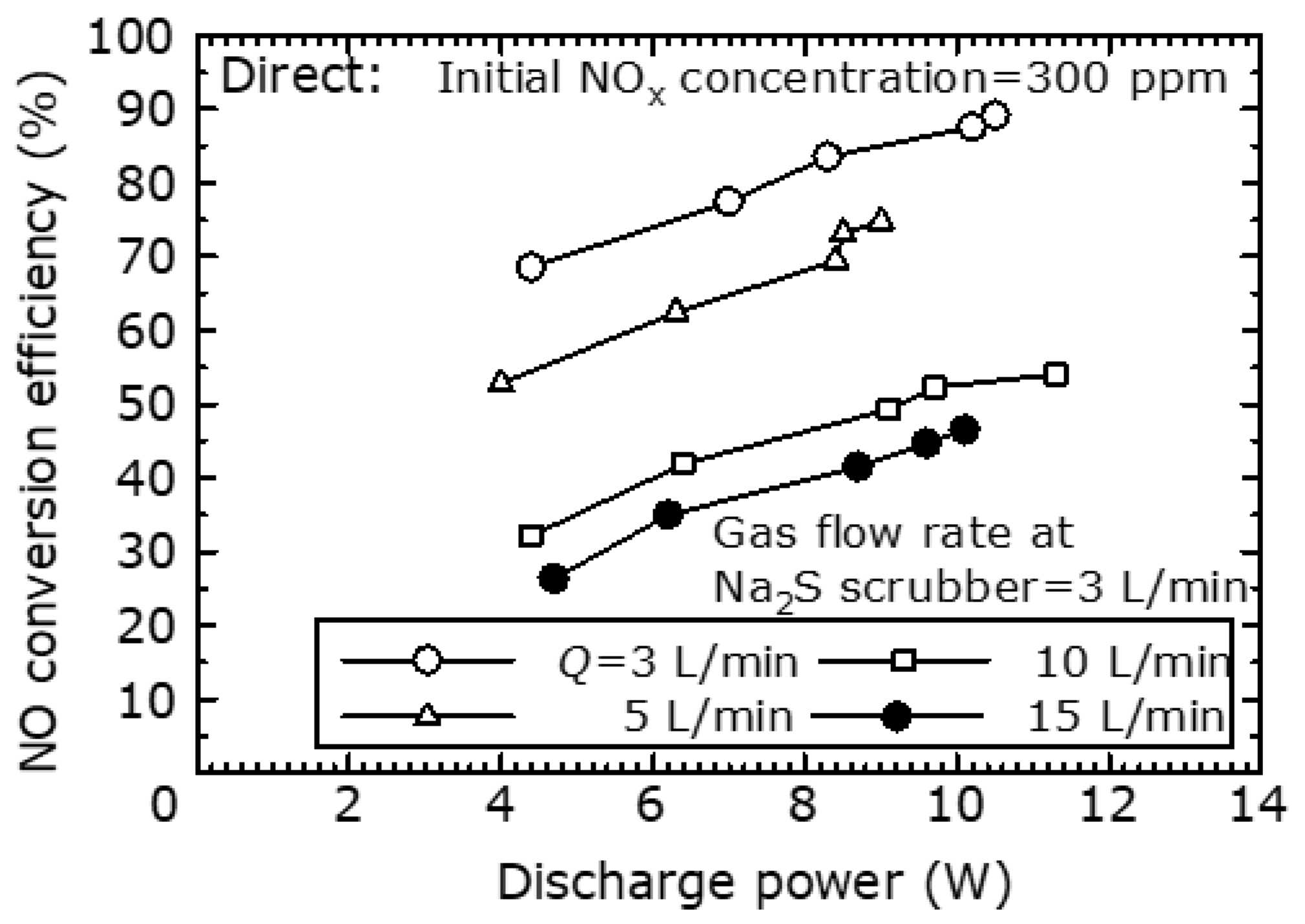
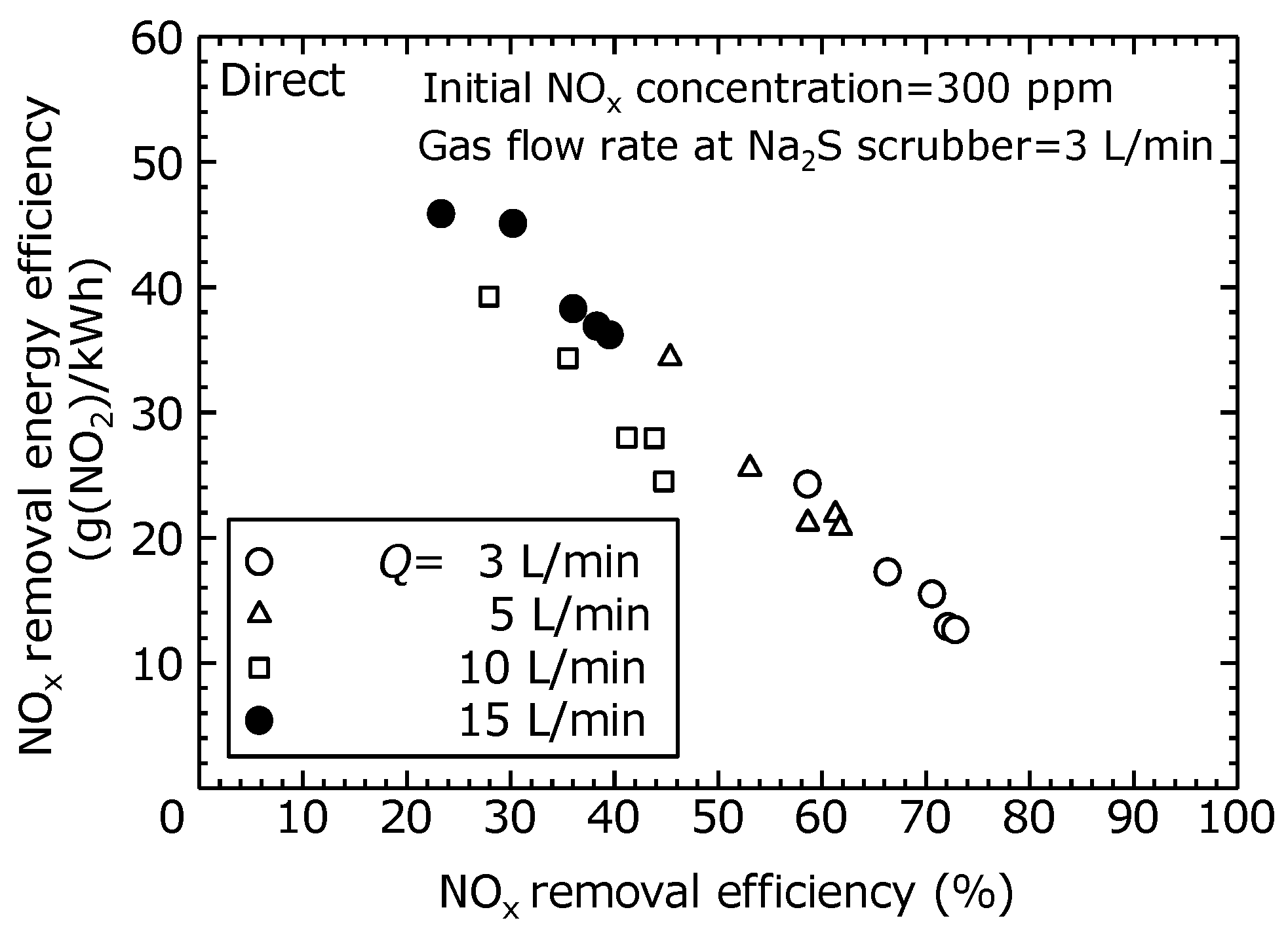
To evaluate the NO
x removal characteristics of the combined dry plasma and wet chemical processes by the direct oxidation method, the NO
x removal efficiency and NO
x removal energy efficiency at different flow rates is shown in
Figure 7. The actual gas flow rate into the scrubber was fixed at 3 L/min. Here, it was assumed that the full flow rate was processed. The NO
x removal energy efficiency was calculated as the total flow rate relative to the NO
x removal amount.
Figure 7 shows that as the total flow rate increased, the NO
x removal efficiency decreased, and the NO
x removal energy efficiency increased. Furthermore, at each flow rate, as the NO
x removal efficiency increased, the NO
x removal energy efficiency decreased. This is because, as shown in
Figure 6, the NO oxidation efficiency rises as the discharge power increases. Within the scope of this experiment, when the NO
x removal efficiency is 60%, the NO
x removal energy efficiency is approximately 20–25 g (NO
2)/kWh.
Examining
Figure 6 and
Figure 7 to evaluate the efficiency of the wet chemical process, the average ratio of the decrease in NO concentration to the decrease in NO
x concentration is 89%, and the Na
2S scrubber can reduce the NO
2 over 80%. At no time was the pH of the solution after treatment greater than 11. In addition, the ORP remained at or more than −263 mV, and no H
2S was detected.
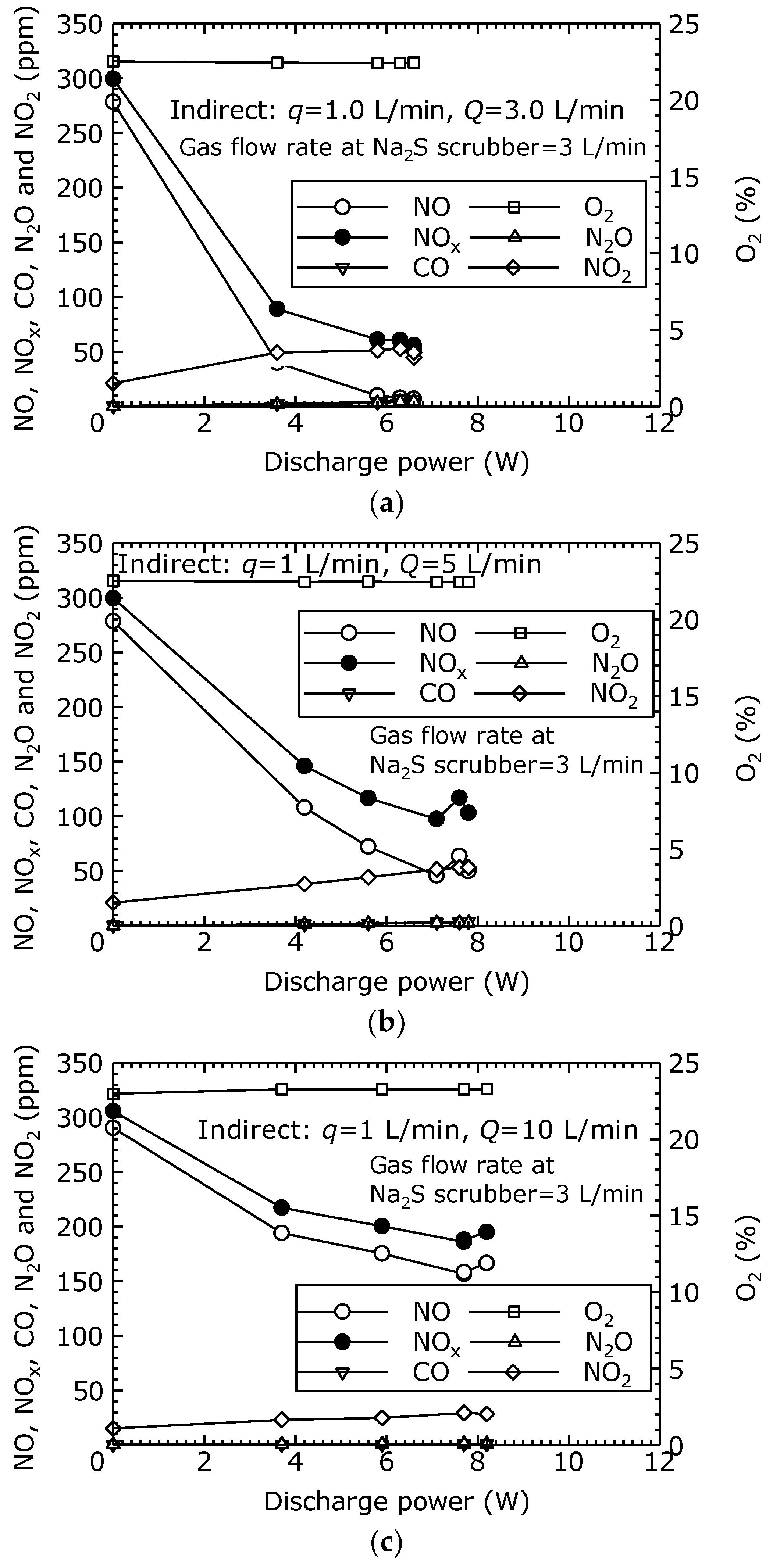
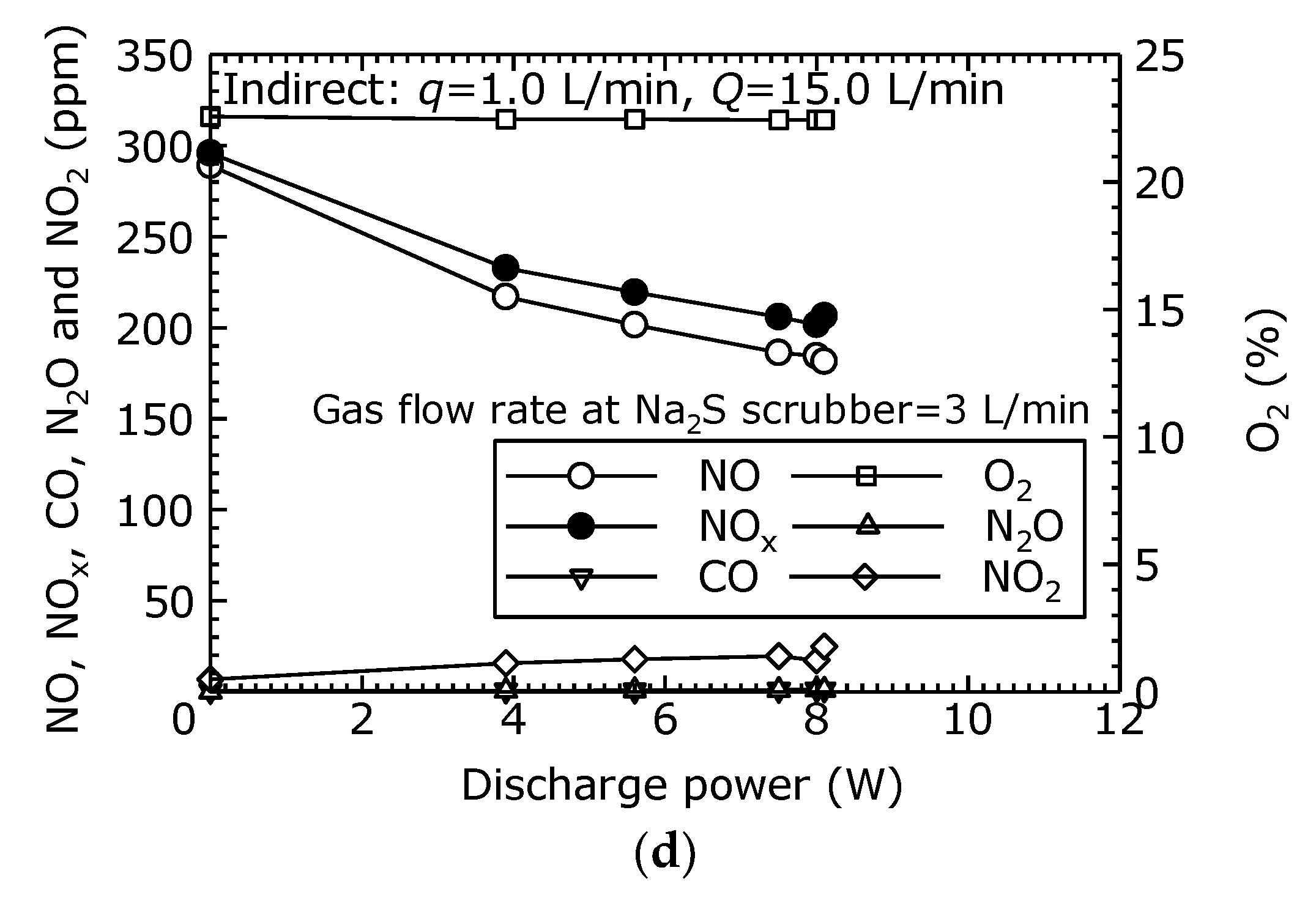
In order to evaluate the NO oxidation and removal characteristics of the combined dry plasma and wet chemical processes using the indirect oxidation method, the components of the treated gas relative to the discharge power was measured.
Figure 8a–d shows the gas concentrations after the combined process under each flow condition (
q = 1 L/min,
Q = 3, 5, 10 and 15 L/min). From
Figure 8a–d, it is seen that the NO decreased as the discharge power increased. Furthermore, as the NO decreased, the NO
2 increased, causing a reduction in NO
x as the NO
2 is removed by the Na
2S scrubber. In
Figure 8a, as the discharge power rises from its initial value to 6.6 W (
Vp-p = 25.8 kV), the NO concentration decreased from 281 to 7 ppm, and the NO
x concentration decreased from 298 to 52 ppm. This represents a 98% reduction of NO and an 83% reduction of NO
x. In
Figure 8b, when the discharge power is 7.8 W (
Vp-p = 31.4 kV), the NO is reduced by 82% and the NO
x by 66%. In
Figure 8c, when the discharge power is 8.3 W (
Vp-p = 26.0 kV), the NO is reduced by 43% and the NO
x by 36%. In
Figure 8d, when the discharge power is 8.1 W (
Vp-p = 27.2 kV), the NO is reduced by 36% and the NO
x by 32%. These results demonstrate that, using the indirect oxidation method as well, treatment by the combined dry plasma and wet chemical processes effectively reduce NO
x. In
Figure 8a–d, CO and N
2O are each less than 10 ppm.
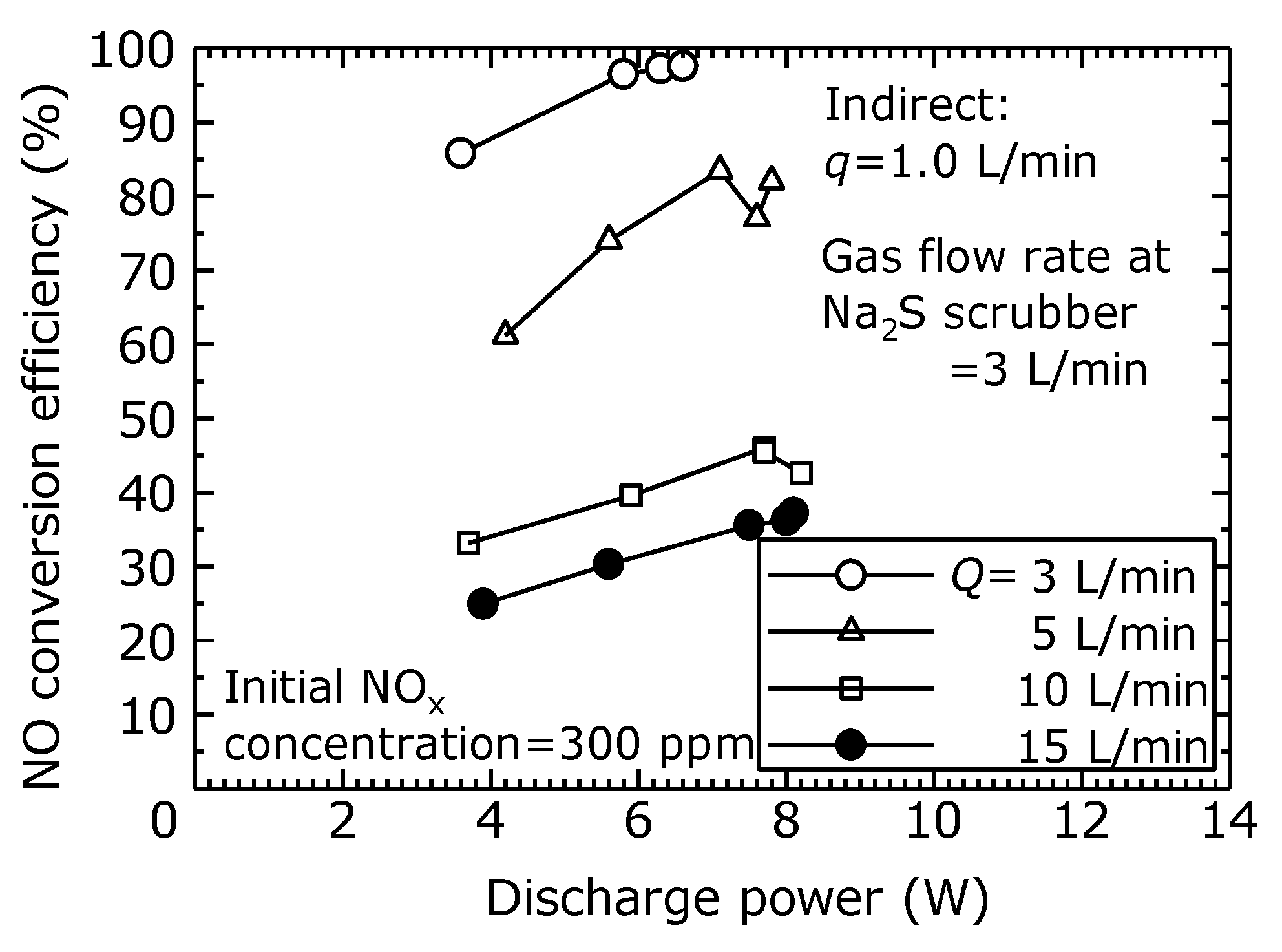
In order to evaluate the NO to NO
2 conversion characteristics of the dry plasma process by the indirect oxidation method, the relationship between the discharge power and the NO conversion efficiency at different flow rates is shown in
Figure 9. In these experiments, the oxygen concentration was 22% to 24% at all flow rates. In the indirect oxidation method, even if the total flow rate is changed, the flow rate into the plasma reactor was constant (
Q = 1 L/min), which means the ozone generated in the plasma reactor stayed constant regardless of the total flow rate. Therefore, as shown in
Figure 9, the NO conversion efficiency varies by roughly the same magnitude as the flow rate (
Q-
q), which is the total flow rate
Q minus the flow rate
q flowing into the plasma reactor. However, when the total flow rate was low, the ozone might be used for further oxidation of NO
2, which suggests that an optimal flow rate may exist. In this experiment, as the flow rate increased, the NO conversion efficiency decreased, but the production of CO and N
2O was suppressed.
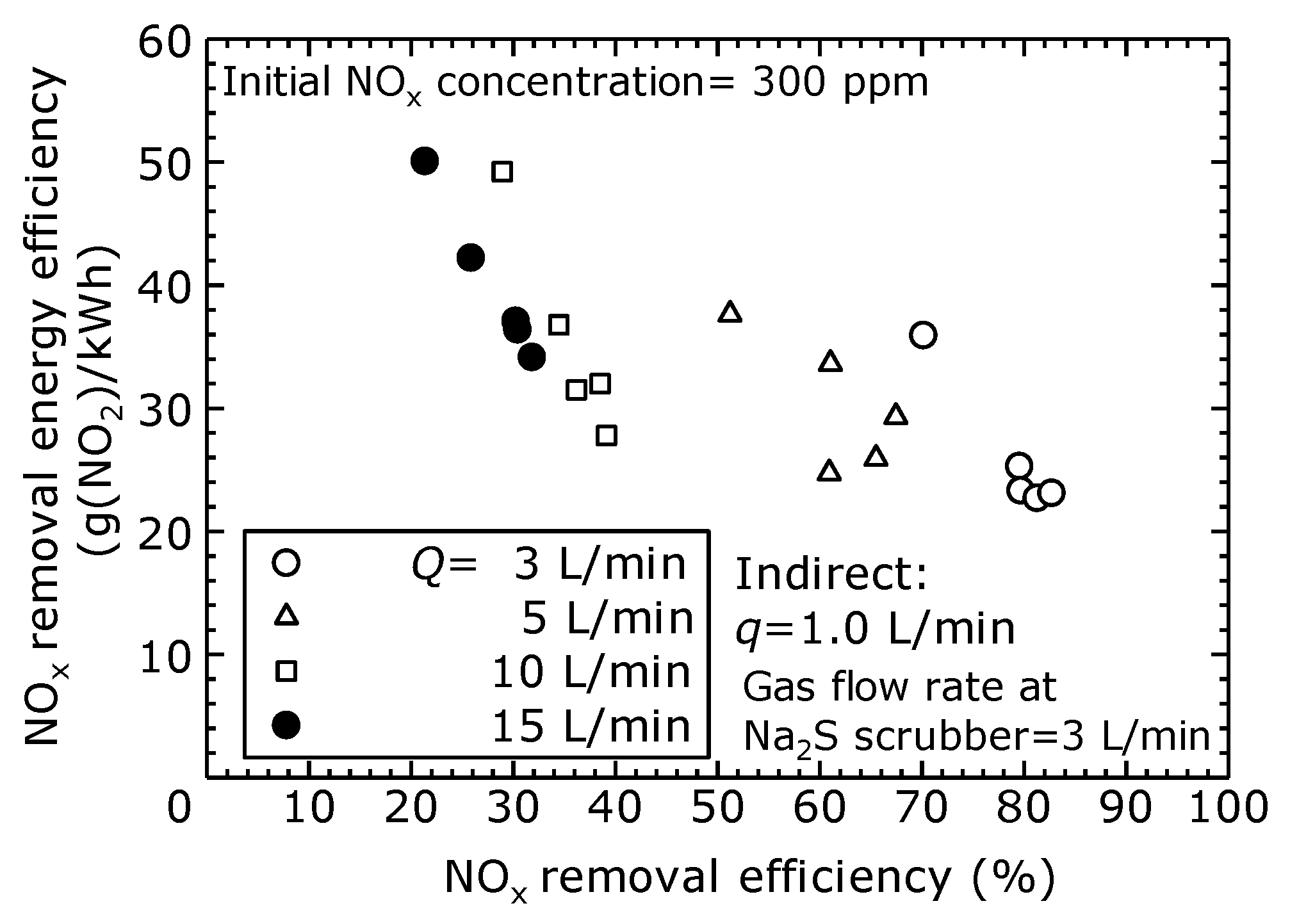
To evaluate the NO
x removal characteristics of the dry plasma and wet chemical processes by the direct oxidation method, the NO
x removal efficiency and NO
x removal energy efficiency at different flow rates is shown in
Figure 10. The NO
x removal energy efficiency was evaluated based on the molecular mass of NO
2 per discharge power with the unit of g(NO
2)/kWh. The actual gas flow rate into the scrubber was fixed at 3 L/min. Here, it is assumed that the full flow rate is processed. The NO
x removal energy efficiency was calculated as the total flow rate relative to the NO
x removal amount.
Figure 10 shows that, as the flow rate increased, the NO
x removal efficiency decreased, and the NO
x removal energy efficiency increased. As shown in
Figure 10, it is clear that the flow rate affects the relationship between the NO
x removal efficiency and the NO
x removal energy efficiency. In the indirect oxidation method, the flow rate into the plasma reactor is constant at
q = 1 L/min, which means the ozone and radical oxygen produced stay constant, regardless of the total flow rate. This explains why the energy efficiency of NO
x removal varies for each flow rate. Within the scope of this experiment, when the NO
x removal efficiency was 80%, the NO
x removal energy efficiency was approximately 22–25 g(NO
2)/kWh.
Examining
Figure 9 and
Figure 10 to evaluate the efficiency of the wet chemical process, it is observed that the average ratio of the decrease in NO concentration to the decrease in NOx concentration is 85%, and the Na
2S scrubber can reduce the NO
2 over 80%. At no time was the pH of the solution after treatment greater than 11. In addition, the ORP remained at or more than −233 mV, and no H
2S was detected.
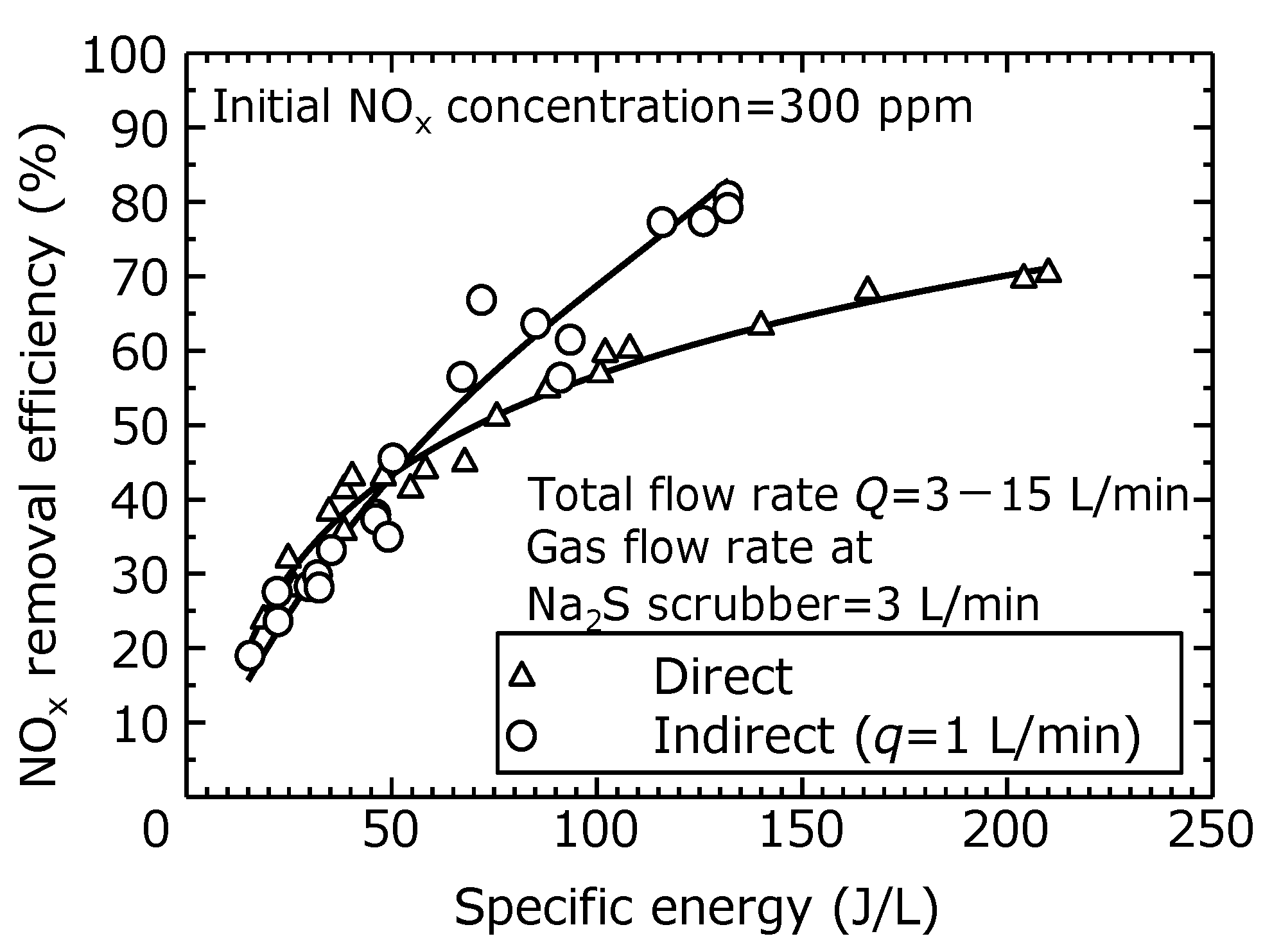
To compare the efficiency of NO
x removal using the direct and indirect oxidation methods in this experiment, the relationship between the NO
x removal efficiency and the specific energy (SE) per unit flow rate is shown in
Figure 11. The SE is calculated based on the total flow rate (3–15 L/min). The solid line is a trendline calculated by the least-squares method.
Figure 11 shows no difference between the direct and indirect oxidation methods at SEs less than 50 J/L, when the NO
x removal efficiency is 20–40%. Although the indirect oxidation method generates a certain amount of ozone in the reactor regardless of the total flow rate, in this experiment, the amount of ozone generated by the indirect oxidation method is not enough to oxidize the NO in the simulated exhaust gas at the high flow rate (of 10 and 15 L/min). As the SE exceeds 50 J/L, the efficiency of NO
x removal by indirect oxidation increases, achieving an 80% removal efficiency at an SE of 132 J/L. It is considered that the amount of ozone generated by indirection oxidation method is larger than that of the direct oxidation method due to the increase of residence time as mentioned above.
These results demonstrate that the combined process of the dry plasma process using the indirect oxidation method and the wet scrubber process can remove NO
x with greater efficiency and energy savings. The plasma dry etching process used in the semiconductor industry requires a high-frequency voltage power supply and consumes several hundred to 1000 W of power. Because the power associated with the NO
x processing performed in this study was less than 10 W, the electricity consumption costs are very low. For example, assuming a 2 m
3/min exhaust class semiconductor plant that discharges NO
x at 25 g(NO
2)/h, the plasma power costs required to achieve 80% or greater NO
x removal using the proposed system at a NO
x removal energy efficiency of 25 g(NO
2)/kWh would be very economical at
$0.20 /h (assuming
$0.20/kWh). There is a risk that H
2S may occur as a byproduct of using Na
2S as the chemical scrubber. However, by maintaining the pH at a sufficiently high value, it is possible to reduce NO
x using Na
2S at a third of the cost of using Na
2SO
3. In the actual semiconductor manufacturing, the concentration of NO
x is emitted 100 ppm at 2000 L/min [
15]. The mass flow rate is much larger than in our experiment (maximum 15 L/min). Since our experiment is fundamental research to remove NO
x at room temperature and atmospherically pressure, practical research should be performed for realizing development. The combined dry plasma and wet chemical processes can therefore be expected to serve as a low-cost and highly efficient method for treating NO
x emissions from the semiconductor manufacturing industry.