Reading Assessment and Eye Movement Analysis in Bilateral Central Scotoma Due to Age-Related Macular Degeneration
Highlights
- People with late-onset age-related macular degeneration adopt oculomotor strategies.
- Our approach integrates functional tests, morphological imaging and eye tracking.
- Saccade count per paragraph correlates significantly with reading indices.
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection
2.2. Cognitive Impairment Screening
2.3. Reading Indices Assessment
2.4. Microperimetry and Multimodal Macular Imaging
2.5. Eye Tracking Recordings and Eye Movement Analysis
2.6. Statistical Analysis
3. Results
3.1. Reading Indices (Reading VA, Reading Speed, and near Contrast Sensitivity)
3.2. Fixation Stability
3.3. PRL Characteristics (Location, Distance from the Fovea) and AMD Lesion Size
3.4. Eye Movement Analysis
3.5. Correlation Between Eye Movements, Reading Indices, Microperimetry, and Macular Imaging Findings
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of variance |
| AMD | Age-related macular degeneration |
| BE | Better-seeing eye of the AMD Group |
| BCEA | Bivariate contour ellipse area |
| BCVA | Best-corrected visual acuity |
| CE | Control (right) eye |
| DAF | Decreased autofluorescence |
| ETDRS | Early treatment diabetic retinopathy study |
| FAF | Fundus autofluorescence |
| FDR | False discovery rate |
| H | Horizontal |
| HV | Horizontal and vertical |
| I-VT | Velocity threshold |
| KW | Kruskal-Walis |
| logCS | Logarithm of contrast sensitivity |
| logMAR | Logarithm of the minimum angle of resolution |
| MNREAD-SI | Minnesota low vision reading chart in Slovene |
| MMSE | Mini mental state examination |
| MP-3 | Microperimeter—third generation |
| ms | Milliseconds |
| NA | Not available |
| OCT | Optical coherence tomography |
| PRL | Preferential retinal locus |
| SD | Standard deviation |
| V | Vertical |
| VA | Visual acuity |
| WE | Worse-seeing eye of the AMD Group |
| wpm | Words per minute |
| ° | Degrees of visual angle |
| °/s | Degrees per second |
Appendix A

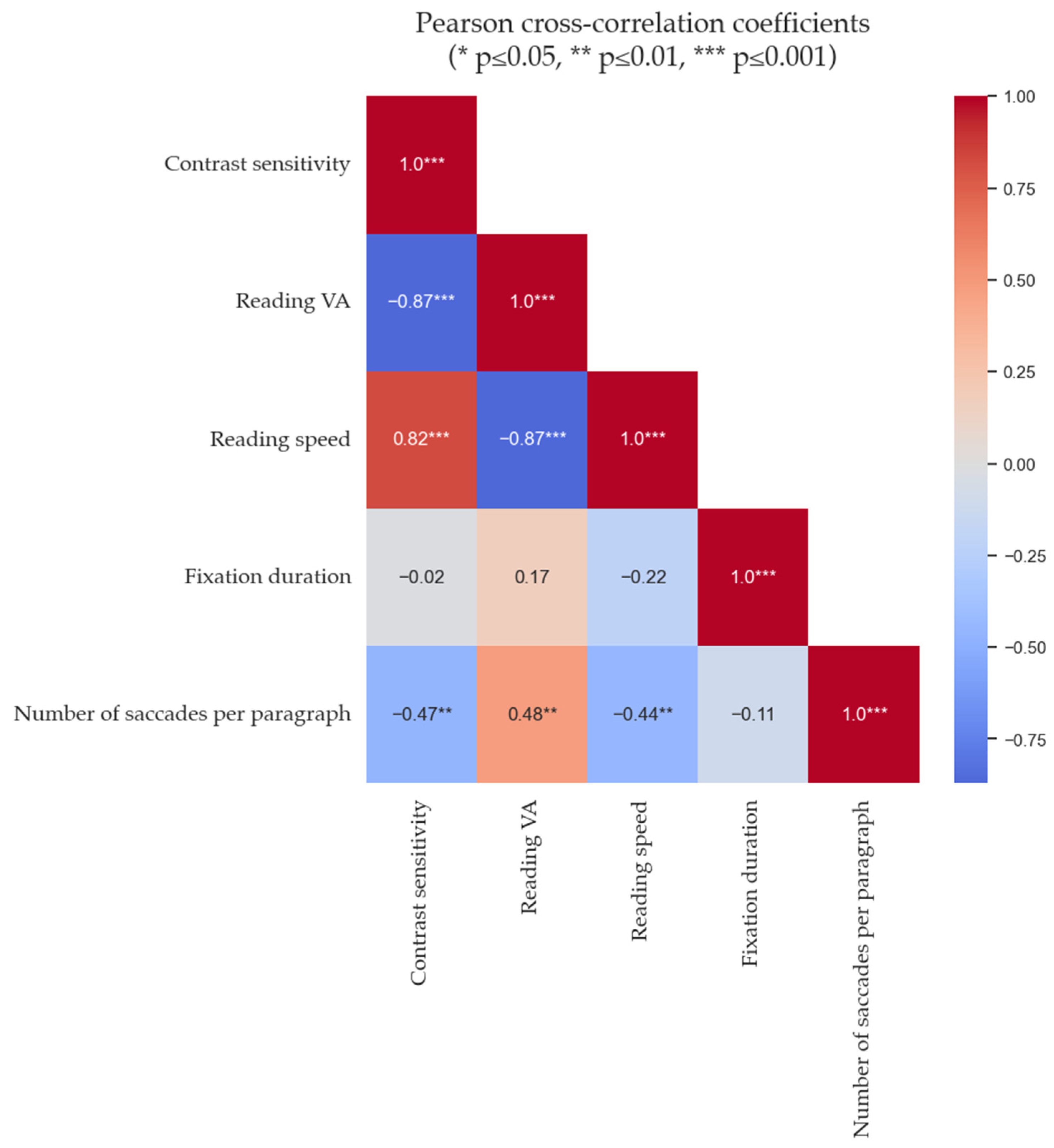


References
- Wong, W.L.; Su, X.; Li, X.; Cheung, C.M.; Klein, R.; Cheng, C.Y.; Wong, T.Y. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: A systematic review and meta-analysis. Lancet Glob. Health 2014, 2, e106–e116. [Google Scholar] [CrossRef]
- Guymer, R.H.; Campbell, T.G. Age-related macular degeneration. Lancet 2023, 401, 1459–1472. [Google Scholar] [CrossRef]
- Mitchell, P.; Liew, G.; Gopinath, B.; Wong, T.Y. Age-related macular degeneration. Lancet 2018, 392, 1147–1159. [Google Scholar] [CrossRef]
- Verghese, P.; Vullings, C.; Shanidze, N. Eye Movements in Macular Degeneration. Annu. Rev. Vis. Sci. 2021, 7, 773–791. [Google Scholar] [CrossRef]
- Crossland, M.D.; Culham, L.E.; Kabanarou, S.A.; Rubin, G.S. Preferred retinal locus development in patients with macular disease. Ophthalmology 2005, 112, 1579–1585. [Google Scholar] [CrossRef] [PubMed]
- Tarita-Nistor, L.; González, E.G.; Markowitz, S.N.; Steinbach, M.J. Plasticity of fixation in patients with central vision loss. Vis. Neurosci. 2009, 26, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Frennesson, C.; Nilsson, S.E. The superior retina performs better than the inferior retina when reading with eccentric viewing: A comparison in normal volunteers. Acta Ophthalmol. Scand. 2007, 85, 868–870. [Google Scholar] [CrossRef] [PubMed]
- Guez, J.E.; Le Gargasson, J.F.; Rigaudiere, F.; O’Regan, J.K. Is there a systematic location for the pseudo-fovea in patients with central scotoma? Vis. Res. 1993, 33, 1271–1279. [Google Scholar] [CrossRef]
- Chung, S.T.L. Reading in the presence of macular disease: A mini-review. Ophthalmic Physiol. Opt. 2020, 40, 171–186. [Google Scholar] [CrossRef]
- Altınbay, D.; İdil, Ş.A. Fixation Stability and Preferred Retinal Locus in Advanced Age-Related Macular Degeneration. Turk. J. Ophthalmol. 2022, 52, 23–29. [Google Scholar] [CrossRef]
- Tarita-Nistor, L.; González, E.G.; Markowitz, S.N.; Steinbach, M.J. Fixation characteristics of patients with macular degeneration recorded with the mp-1 microperimeter. Retina 2008, 28, 125–133. [Google Scholar] [CrossRef]
- Crossland, M.D.; Culham, L.E.; Rubin, G.S. Fixation stability and reading speed in patients with newly developed macular disease. Ophthalmic Physiol. Opt. 2004, 24, 327–333. [Google Scholar] [CrossRef]
- Guadron, L.; Titchener, S.A.; Abbott, C.J.; Ayton, L.N.; van Opstal, J.; Petoe, M.A.; Goossens, J. The Saccade Main Sequence in Patients With Retinitis Pigmentosa and Advanced Age-Related Macular Degeneration. Investig. Ophthalmol. Vis. Sci. 2023, 64, 1. [Google Scholar] [CrossRef]
- Calabrèse, A.; Bernard, J.B.; Faure, G.; Hoffart, L.; Castet, E. Eye movements and reading speed in macular disease: The shrinking perceptual span hypothesis requires and is supported by a mediation analysis. Investig. Ophthalmol. Vis. Sci. 2014, 55, 3638–3645. [Google Scholar] [CrossRef]
- Crossland, M.D.; Rubin, G.S. Eye movements and reading in macular disease: Further support for the shrinking perceptual span hypothesis. Vis. Res. 2006, 46, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Giacomelli, G.; Farini, A.; Baldini, I.; Raffaelli, M.; Bigagli, G.; Fossetti, A.; Virgili, G. Saccadic movements assessment in eccentric fixation: A study in patients with Stargardt disease. Eur. J. Ophthalmol. 2021, 31, 2556–2562. [Google Scholar] [CrossRef] [PubMed]
- Shanidze, N.M.; Lively, Z.; Lee, R.; Verghese, P. Saccadic contributions to smooth pursuit in macular degeneration. Vis. Res. 2022, 200, 108102. [Google Scholar] [CrossRef]
- Van der Stigchel, S.; Bethlehem, R.A.; Klein, B.P.; Berendschot, T.T.; Nijboer, T.C.; Dumoulin, S.O. Macular degeneration affects eye movement behavior during visual search. Front. Psychol. 2013, 4, 579. [Google Scholar] [CrossRef] [PubMed]
- Vullings, C.; Lively, Z.; Verghese, P. Saccades during visual search in macular degeneration. Vis. Res. 2022, 201, 108113. [Google Scholar] [CrossRef]
- Klaib, A.F.; Alsrehin, N.O.; Melhem, W.Y.; Bashtawi, H.O.; Magableh, A.A. Eye tracking algorithms, techniques, tools, and applications with an emphasis on machine learning and Internet of Things technologies. Expert Syst. Appl. 2021, 166, 114037. [Google Scholar] [CrossRef]
- Termsarasab, P.; Thammongkolchai, T.; Rucker, J.C.; Frucht, S.J. The diagnostic value of saccades in movement disorder patients: A practical guide and review. J. Clin. Mov. Disord. 2015, 2, 14. [Google Scholar] [CrossRef]
- Leigh, R.J.; Zee, D.S. The Neurology of Eye Movements; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Granda, G.; Mlakar, J.; Vodušek, D.B. The Slovene version of Mini mental state examination—Standardization on volunteers from 55 to 75 years old. Slov. Med. J. 2003, 72, 575–582. [Google Scholar]
- Mansfield, J.S.; Atilgan, N.; Lewis, A.M.; Legge, G.E. Extending the MNREAD sentence corpus: Computer-generated sentences for measuring visual performance in reading. Vis. Res. 2019, 158, 11–18. [Google Scholar] [CrossRef]
- Longhin, E.; Convento, E.; Pilotto, E.; Bonin, G.; Vujosevic, S.; Kotsafti, O.; Midena, E. Static and dynamic retinal fixation stability in microperimetry. Can. J. Ophthalmol. 2013, 48, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Crossland, M.D.; Engel, S.A.; Legge, G.E. The preferred retinal locus in macular disease: Toward a consensus definition. Retina 2011, 31, 2109–2114. [Google Scholar] [CrossRef]
- Fujii, G.Y.; de Juan, E., Jr.; Sunness, J.; Humayun, M.S.; Pieramici, D.J.; Chang, T.S. Patient selection for macular translocation surgery using the scanning laser ophthalmoscope. Ophthalmology 2002, 109, 1737–1744. [Google Scholar] [CrossRef] [PubMed]
- Strauss, R.W.; Muñoz, B.; Ho, A.; Jha, A.; Michaelides, M.; Mohand-Said, S.; Cideciyan, A.V.; Birch, D.; Hariri, A.H.; Nittala, M.G.; et al. Incidence of Atrophic Lesions in Stargardt Disease in the Progression of Atrophy Secondary to Stargardt Disease (ProgStar) Study: Report No. 5. JAMA Ophthalmol. 2017, 135, 687–695. [Google Scholar] [CrossRef]
- Tobii Eye Tracker. Available online: https://www.tobii.com/ (accessed on 14 February 2025).
- Imaoka, Y.; Flury, A.; de Bruin, E.D. Assessing Saccadic Eye Movements With Head-Mounted Display Virtual Reality Technology. Front. Psychiatry 2020, 11, 572938. [Google Scholar] [CrossRef] [PubMed]
- Leube, A.; Rifai, K.; Rifai, K. Sampling rate influences saccade detection in mobile eye tracking of a reading task. J. Eye Mov. Res. 2017, 10, 1–11. [Google Scholar] [CrossRef]
- Antoniades, C.; Ettinger, U.; Gaymard, B.; Gilchrist, I.; Kristjánsson, A.; Kennard, C.; Leigh, R.J.; Noorani, I.; Pouget, P.; Smyrnis, N. An internationally standardised antisaccade protocol. Vis. Res. 2013, 84, 1–5. [Google Scholar] [CrossRef]
- Tanasković, I.; Miljković, N.; Stojmenova Pečečnik, K.; Sodnik, J. Saccade Identification During Driving Simulation from Eye Tracker Data with Low-Sampling Frequency. In Disruptive Information Technologies for a Smart Society. ICIST 2023; Springer Nature: Cham, Switzerland, 2024; pp. 30–39. [Google Scholar]
- Wierts, R.; Janssen, M.J.; Kingma, H. Measuring saccade peak velocity using a low-frequency sampling rate of 50 Hz. IEEE Trans. Biomed. Eng. 2008, 55, 2840–2842. [Google Scholar] [CrossRef]
- Van Rossum, G.; Drake, F.L. Python 3 Reference Manual; CreateSpace: Scotts Valley, CA, USA, 2009. [Google Scholar]
- Holmqvist, K.; Nyström, M.; Andersson, R.; Dewhurst, R.; Jarodzka, H.; Van de Weijer, J. Eye Tracking: A Comprehensive Guide to Methods and Measures; Oxford University Press: Oxford, UK, 2011. [Google Scholar]
- Nyström, M.; Holmqvist, K. An adaptive algorithm for fixation, saccade, and glissade detection in eyetracking data. Behav. Res. Methods 2010, 42, 188–204. [Google Scholar] [CrossRef]
- Duchowski, A.T.; Duchowski, A.T. Eye Tracking Methodology: Theory and Practice; Springer: Cham, Switzerland, 2017. [Google Scholar]
- Harezlak, K.; Kasprowski, P. Application of eye tracking in medicine: A survey, research issues and challenges. Comput. Med. Imaging Graph. 2018, 65, 176–190. [Google Scholar] [CrossRef]
- Amit, R.; Abeles, D.; Bar-Gad, I.; Yuval-Greenberg, S. Temporal dynamics of saccades explained by a self-paced process. Sci. Rep. 2017, 7, 886. [Google Scholar] [CrossRef]
- Rubin, G.S.; Feely, M. The role of eye movements during reading in patients with age-related macular degeneration (AMD). Neuro-Ophthalmology 2009, 33, 120–126. [Google Scholar] [CrossRef]
- Mahanama, B.; Jayawardana, Y.; Rengarajan, S.; Jayawardena, G.; Chukoskie, L.; Snider, J.; Jayarathna, S. Eye movement and pupil measures: A review. Front. Comput. Sci. 2022, 3, 733531. [Google Scholar] [CrossRef]
- Chen, J.T.; Kuo, Y.C.; Hsu, T.Y.; Wang, C.A. Fatigue and Arousal Modulations Revealed by Saccade and Pupil Dynamics. Int. J. Environ. Res. Public Health 2022, 19, 9234. [Google Scholar] [CrossRef] [PubMed]
- Lal, V.; Truong, D. Eye movement abnormalities in movement disorders. Clin. Park. Relat. Disord. 2019, 1, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, S.S.; Wilk, M.B. An analysis of variance test for normality (complete samples). Biometrika 1965, 52, 591–611. [Google Scholar] [CrossRef]
- Elkin, L.; Kay, M.; Higgins, J.J.; Wobbrock, J.O. An Aligned Rank Transform Procedure for Multifactor Contrast Tests. In Proceedings of the 34th Annual ACM Symposium on User Interface Software and Technology, Virtual, 10–14 October 2021; pp. 754–768. [Google Scholar]
- Wobbrock, J.O.; Findlater, L.; Gergle, D.; Higgins, J.J. The aligned rank transform for nonparametric factorial analyses using only anova procedures. In Proceedings of the SIGCHI conference on Human Factors in Computing Systems, Vancouver, BC, Canada, 7–12 May 2011; pp. 143–146. [Google Scholar]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Maxwell, S.E.; Delaney, H.D.; Kelley, K. Designing Experiments and Analyzing Data: A Model Comparison Perspective; Routledge: New York, NY, USA, 2017. [Google Scholar]
- Richardson, J.T. Eta squared and partial eta squared as measures of effect size in educational research. Educ. Res. Rev. 2011, 6, 135–147. [Google Scholar] [CrossRef]
- Yu, H.; Kwon, M. Altered Eye Movements During Reading With Simulated Central and Peripheral Visual Field Defects. Investig. Ophthalmol. Vis. Sci. 2023, 64, 21. [Google Scholar] [CrossRef]
- Harvey, H.; Walker, R. Reading with peripheral vision: A comparison of reading dynamic scrolling and static text with a simulated central scotoma. Vis. Res. 2014, 98, 54–60. [Google Scholar] [CrossRef]
- Farzaneh, A.; Riazi, A.; Falavarjani, K.G.; Doostdar, A.; Kamali, M.; Sedaghat, A.; Khabazkhoob, M. Evaluating Reading Performance in Different Preferred Retinal Loci in Persian-Speaking Patients with Age-Related Macular Degeneration. J. Curr. Ophthalmol. 2021, 33, 48–55. [Google Scholar] [CrossRef]
- Daibert-Nido, M.; Patino, B.; Markowitz, M.; Markowitz, S.N. Rehabilitation with biofeedback training in age-related macular degeneration for improving distance vision. Can. J. Ophthalmol. 2019, 54, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Hessels, R.S.; Hooge, I.T. Eye tracking in developmental cognitive neuroscience–The good, the bad and the ugly. Dev. Cogn. Neurosci. 2019, 40, 100710. [Google Scholar] [CrossRef] [PubMed]
- Holmqvist, K.; Nyström, M.; Mulvey, F. Eye tracker data quality: What it is and how to measure it. In Proceedings of the Symposium on Eye Tracking Research and Applications, Santa Barbara, CA, USA, 28–30 March 2012; pp. 45–52. [Google Scholar]
- Motnikar, L.; Stojmenova, K.; Štaba, U.Č.; Klun, T.; Robida, K.R.; Sodnik, J. Exploring driving characteristics of fit-and unfit-to-drive neurological patients: A driving simulator study. Traffic Inj. Prev. 2020, 21, 359–364. [Google Scholar] [CrossRef] [PubMed]
- Niehorster, D.C.; Santini, T.; Hessels, R.S.; Hooge, I.T.; Kasneci, E.; Nyström, M. The impact of slippage on the data quality of head-worn eye trackers. Behav. Res. Methods 2020, 52, 1140–1160. [Google Scholar] [CrossRef]
- Bulling, A.; Roggen, D.; Tröster, G. Wearable EOG goggles: Seamless sensing and context-awareness in everyday environments. J. Ambient. Intell. Smart Environ. 2009, 1, 157–171. [Google Scholar] [CrossRef]
- Rudnicka, A.R.; Kapetanakis, V.V.; Jarrar, Z.; Wathern, A.K.; Wormald, R.; Fletcher, A.E.; Cook, D.G.; Owen, C.G. Incidence of Late-Stage Age-Related Macular Degeneration in American Whites: Systematic Review and Meta-analysis. Am. J. Ophthalmol. 2015, 160, 85–93.e3. [Google Scholar] [CrossRef]
- Zetterberg, M. Age-related eye disease and gender. Maturitas 2016, 83, 19–26. [Google Scholar] [CrossRef] [PubMed]




| Parameter | Explanation | Expectation | Reference(s) |
|---|---|---|---|
| Reading duration [s] | Total time a subject spent reading a specific paragraph of text | Reading duration may be prolonged | [41] |
| Saccade amplitude [°] | Angle of eye movement from start to end of a saccade | Smaller amplitude varies, usually higher variability | [19,36,42] |
| Peak saccade velocity [°/s] | Maximum speed during a saccade, around 300°/s for healthy adults | Often reduced or erratic | [36,43,44] |
| Saccade duration [ms] | Duration from onset to offset of a saccade, typically between 30 and 80 ms | Duration may be prolonged | [36,43] |
| Number of detected saccades per paragraph | Count of saccadic eye movements detected during reading paragraphs | Increased number of saccades | [18] |
| Fixation duration [ms] | Duration spent on a single fixation point, approximately 200–300 ms in healthy individuals | Expected to be longer, due impairment in fixation stability | [12] |
| Group | Name of the Parameter |
|---|---|
| Group 1 | age, reading VA, reading speed, near contrast sensitivity, reading duration, saccade amplitude, peak saccade velocity, saccade duration, number of saccades per paragraph, fixation duration |
| Group 2 | ETDRS (LogMAR), static fixation characteristics (circle 2%, circle 4%, static BCEA 1 sd, static BCEA 2 sd, and static BCEA 3 sd) |
| Group 3 | dynamic fixation characteristics (dynamic circle 2%, dynamic circle 4%, dynamic BCEA 1 sd, dynamic BCEA 2 sd, dynamic BCEA 3 sd), PRL-fovea distance, and AMD lesion size |
| Parameter | Descriptive Statistics | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| AMD Group (N = 17) | Control Group (N = 17) | |||||||||
| Mean | SD | 25% | Median | 75% | Mean | SD | 25% | Median | 75% | |
| Reading VA [logMAR] | 0.95 | 0.22 | 0.80 | 0.90 | 1.10 | 0.18 | 0.20 | 0.00 | 0.20 | 0.30 |
| Reading speed [wpm] | 37.12 | 21.95 | 19.00 | 38.00 | 55.00 | 142.65 | 42.79 | 120.00 | 133.00 | 150.00 |
| Contrast sensitivity [logCS] | 1.03 | 0.27 | 0.90 | 1.05 | 1.20 | 1.65 | 0.14 | 1.65 | 1.65 | 1.80 |
| circle 2 [%] | 66.47 | 32.68 | 32.00 | 61.00 | 100.00 | 99.24 | 1.52 | 99.00 | 100.00 | 100.00 |
| circle 4 [%] | 89.71 | 13.92 | 81.00 | 95.00 | 100.00 | 99.59 | 0.87 | 100.00 | 100.00 | 100.00 |
| BCEA 1 sd [area] | 4.45 | 5.14 | 0.30 | 2.50 | 7.80 | 0.35 | 0.39 | 0.10 | 0.02 | 0.50 |
| BCEA 2 sd [area] | 11.98 | 13.84 | 0.90 | 6.80 | 20.90 | 0.91 | 1.03 | 0.30 | 0.40 | 1.30 |
| BCEA 3 sd [area] | 22.92 | 26.49 | 1.70 | 13.00 | 40.00 | 1.76 | 1.95 | 0.60 | 0.80 | 2.50 |
| Name of the Parameter | KW p-Value | Pairwise Comparison | Post Hoc Test p-Value | Effect Size | Effect Size Interpretation |
|---|---|---|---|---|---|
| ETDRS | <0.001 | CE vs. BE | <0.001 | 1.000 | Large |
| CE vs. WE | <0.001 | 1.000 | |||
| BE vs. WE | 0.012 | 0.751 | |||
| LogMAR | CE vs. BE | <0.001 | −1.000 | ||
| CE vs. WE | <0.001 | −1.000 | |||
| BE vs. WE | 0.012 | −0.751 | |||
| circle 2 | CE vs. BE | 0.007 | 0.574 | ||
| CE vs. WE | <0.001 | 0.924 | |||
| BE vs. WE | 0.048 | 0.405 | Medium | ||
| circle 4 | CE vs. BE | 0.022 | 0.481 | Large | |
| CE vs. WE | <0.001 | 0.896 | |||
| BE vs. WE | 0.023 | 0.450 | Medium | ||
| BCEA 1 sd | CE vs. BE | 0.003 | −0.678 | Large | |
| CE vs. WE | <0.001 | −0.945 | |||
| BE vs. WE | 0.056 | −0.439 | Medium | ||
| BCEA 2 sd | CE vs. BE | 0.002 | −0.709 | Large | |
| CE vs. WE | <0.001 | −0.952 | |||
| BE vs. WE | 0.058 | −0.446 | Medium | ||
| BCEA 3 sd | CE vs. BE | 0.002 | −0.709 | Large | |
| CE vs. WE | <0.001 | −0.952 | |||
| BE vs. WE | 0.058 | −0.446 | Medium |
| Parameter | Descriptive Statistics (N = 17) | ||||
|---|---|---|---|---|---|
| Mean | SD | 25% | Median | 75% | |
| Dynamical fixation, circle 2 [%] | 34.76 | 24.86 | 15.00 | 31.00 | 44.00 |
| Dynamical fixation, circle 4 [%] | 65.12 | 25.09 | 53.00 | 75.00 | 83.00 |
| Dynamical fixation, BCEA 1 sd [area] | 17.82 | 16.23 | 7.10 | 10.30 | 22.40 |
| Dynamical fixation, BCEA 2 sd [area] | 47.96 | 43.69 | 19.10 | 27.70 | 60.30 |
| Dynamical fixation, BCEA 3 sd [area] | 91.79 | 83.62 | 36.50 | 53.00 | 115.50 |
| PRL-fovea distance [mm] | 1.69 | 0.99 | 1.05 | 1.70 | 2.58 |
| AMD lesion size [mm2] | 12.34 | 6.07 | 6.87 | 11.39 | 18.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benda, P.Z.; Jakus, G.; Sodnik, J.; Miljković, N.; Tanasković, I.; Stokanović, S.; Meglič, A.; Valentinčič, N.V.; Mekjavić, P.J. Reading Assessment and Eye Movement Analysis in Bilateral Central Scotoma Due to Age-Related Macular Degeneration. J. Eye Mov. Res. 2025, 18, 38. https://doi.org/10.3390/jemr18050038
Benda PZ, Jakus G, Sodnik J, Miljković N, Tanasković I, Stokanović S, Meglič A, Valentinčič NV, Mekjavić PJ. Reading Assessment and Eye Movement Analysis in Bilateral Central Scotoma Due to Age-Related Macular Degeneration. Journal of Eye Movement Research. 2025; 18(5):38. https://doi.org/10.3390/jemr18050038
Chicago/Turabian StyleBenda, Polona Zaletel, Grega Jakus, Jaka Sodnik, Nadica Miljković, Ilija Tanasković, Smilja Stokanović, Andrej Meglič, Nataša Vidovič Valentinčič, and Polona Jaki Mekjavić. 2025. "Reading Assessment and Eye Movement Analysis in Bilateral Central Scotoma Due to Age-Related Macular Degeneration" Journal of Eye Movement Research 18, no. 5: 38. https://doi.org/10.3390/jemr18050038
APA StyleBenda, P. Z., Jakus, G., Sodnik, J., Miljković, N., Tanasković, I., Stokanović, S., Meglič, A., Valentinčič, N. V., & Mekjavić, P. J. (2025). Reading Assessment and Eye Movement Analysis in Bilateral Central Scotoma Due to Age-Related Macular Degeneration. Journal of Eye Movement Research, 18(5), 38. https://doi.org/10.3390/jemr18050038







