Kidney Injury in Children after Hematopoietic Stem Cell Transplant
Abstract
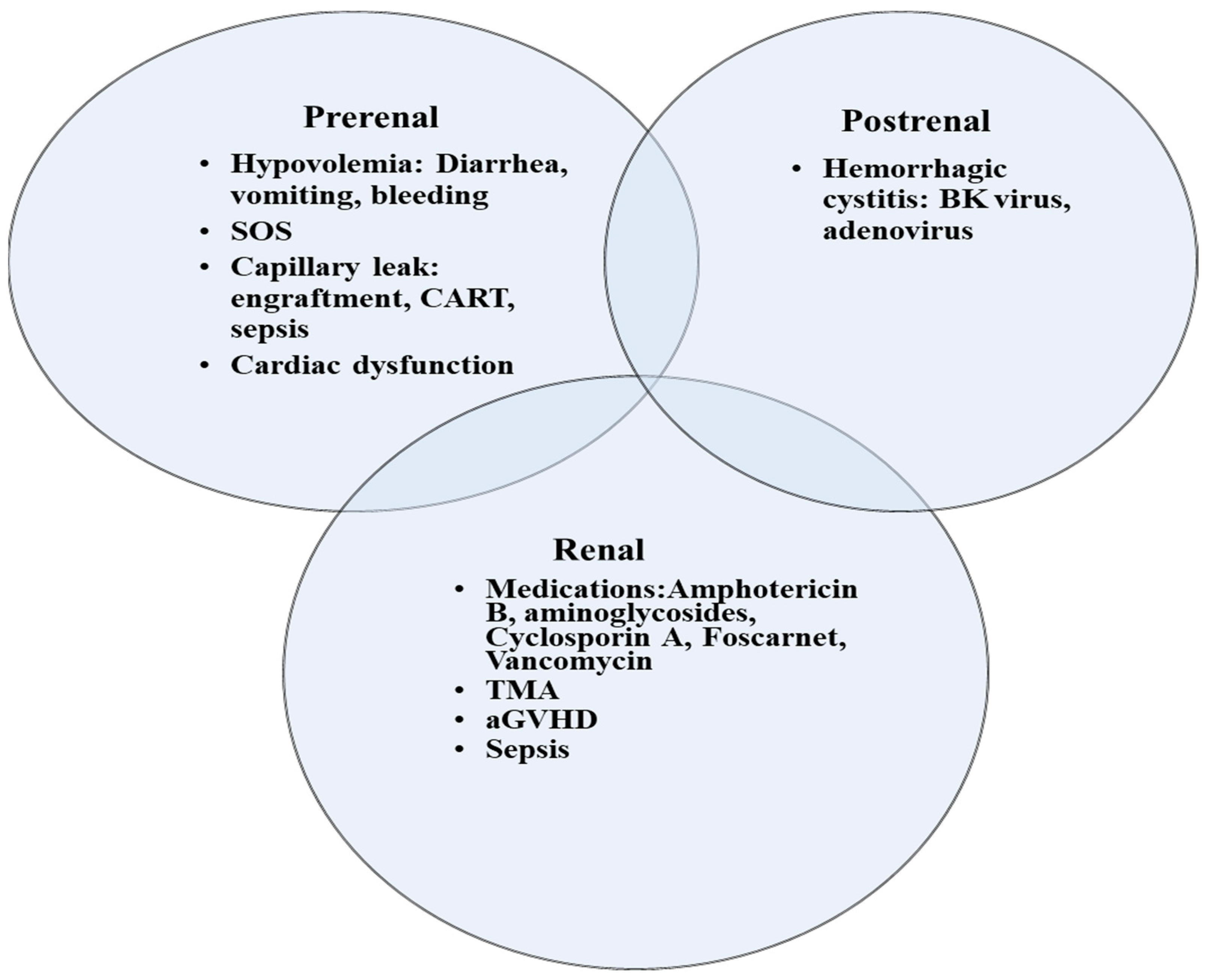
1. Acute Kidney Injury
2. Criteria for AKI Diagnosis and Staging
3. Special Disease Conditions Post HCT That Are Associated with AKI
3.1. Transplant-Associated Thrombotic Microangiopathy
3.2. Sinusoidal Obstruction Syndrome
- Consumptive and transfusion-refractory thrombocytopenia;
- Weight gain on 3 consecutive days despite the use of diuretics, or a weight gain of >5% above baseline weight within 72 h;
- Increase in bilirubin from baseline on 3 consecutive days, or bilirubin ≥ 2 mg/dL within 72 h;
- Hepatomegaly (best if supported by imaging) above baseline value;
- Ascites (best if supported by imaging) above baseline.
3.3. Fluid Overload
3.4. CAR T-Cell Therapy
4. Continuous Kidney Replacement Therapy
5. Transition from CKRT to IHD/Discontinuation of CKRT
6. Outcomes of KRT
7. Biomarkers of AKI in Children with HCT
Cystatin C
8. Tubular Injury Markers
9. Chronic Kidney Disease
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Raina, R.; Abu-Arja, R.; Sethi, S.; Dua, R.; Chakraborty, R.; Dibb, J.T.; Basu, R.K.; Bissler, J.; Felix, M.B.; Brophy, P.; et al. Acute kidney injury in pediatric hematopoietic cell transplantation: Critical appraisal and consensus. Pediatr. Nephrol. 2022, 37, 1179–1203. [Google Scholar] [CrossRef] [PubMed]
- Elbahlawan, L.; Bissler, J.; Morrison, R.R. Continuous Renal Replacement Therapy: A Review of Use and Application in Pediatric Hematopoietic Stem Cell Transplant Recipients. Front. Oncol. 2021, 11, 632263. [Google Scholar] [CrossRef] [PubMed]
- Daraskevicius, J.; Azukaitis, K.; Dziugeviciute-Tupko, J.; Peciulyte, M.; Planciunaite, R.; Vaitkeviciene, G.; Rascon, J.; Jankauskiene, A. Phenotypes and Baseline Risk Factors of Acute Kidney Injury in Children After Allogeneic Hematopoietic Stem Cell Transplantation. Front. Pediatr. 2020, 8, 499. [Google Scholar] [CrossRef] [PubMed]
- Koh, K.N.; Sunkara, A.; Kang, G.; Sooter, A.; Mulrooney, D.A.; Triplett, B.; Onder, A.M.; Bissler, J.; Cunningham, L.C. Acute Kidney Injury in Pediatric Patients Receiving Allogeneic Hematopoietic Cell Transplantation: Incidence, Risk Factors, and Outcomes. Biol. Blood Marrow Transplant. 2018, 24, 758–764. [Google Scholar] [CrossRef]
- Kizilbash, S.J.; Kashtan, C.E.; Chavers, B.M.; Cao, Q.; Smith, A.R. Acute Kidney Injury and the Risk of Mortality in Children Undergoing Hematopoietic Stem Cell Transplantation. Biol. Blood Marrow Transplant. 2016, 22, 1264–1270. [Google Scholar] [CrossRef]
- Satwani, P.; Bavishi, S.; Jin, Z.; Jacobson, J.S.; Baker, C.; Duffy, D.; Lowe, L.; Morris, E.; Cairo, M.S. Risk factors associated with kidney injury and the impact of kidney injury on overall survival in pediatric recipients following allogeneic stem cell transplant. Biol. Blood Marrow Transplant. 2011, 17, 1472–1480. [Google Scholar] [CrossRef]
- Yu, Z.P.; Ding, J.H.; Chen, B.A.; Liu, B.C.; Liu, H.; Li, Y.F.; Ding, B.H.; Qian, J. Risk factors for acute kidney injury in patients undergoing allogeneic hematopoietic stem cell transplantation. Chin. J. Cancer 2010, 29, 946–951. [Google Scholar] [CrossRef]
- Ileri, T.; Ertem, M.; Ozcakar, Z.B.; Ince, E.U.; Biyikli, Z.; Uysal, Z.; Ekim, M.; Yalcinkaya, F. Prospective evaluation of acute and chronic renal function in children following matched related donor hematopoietic stem cell transplantation. Pediatr. Transplant. 2010, 14, 138–144. [Google Scholar] [CrossRef]
- Hazar, V.; Gungor, O.; Guven, A.G.; Aydin, F.; Akbas, H.; Gungor, F.; Tezcan, G.; Akman, S.; Yesilipek, A. Renal function after hematopoietic stem cell transplantation in children. Pediatr. Blood Cancer 2009, 53, 197–202. [Google Scholar] [CrossRef]
- Kist-van Holthe, J.E.; Goedvolk, C.A.; Brand, R.; van Weel, M.H.; Bredius, R.G.; van Oostayen, J.A.; Vossen, J.M.; van der Heijden, B.J. Prospective study of renal insufficiency after bone marrow transplantation. Pediatr. Nephrol. 2002, 17, 1032–1037. [Google Scholar] [CrossRef]
- Didsbury, M.S.; Mackie, F.E.; Kennedy, S.E. A systematic review of acute kidney injury in pediatric allogeneic hematopoietic stem cell recipients. Pediatr. Transplant. 2015, 19, 460–470. [Google Scholar] [CrossRef]
- Madsen, K.; Pelletier, K.; Côté, G.; Kitchlu, A.; Chen, S.; Mattsson, J.; Pasic, I. Acute kidney injury within 100 days post allogeneic hematopoietic cell transplantation is associated with increased risk of post-transplant complications and poor transplant outcomes. Bone Marrow Transplant. 2022, 57, 1411–1420. [Google Scholar] [CrossRef]
- Ando, M.; Ohashi, K.; Akiyama, H.; Sakamaki, H.; Morito, T.; Tsuchiya, K.; Nitta, K. Chronic kidney disease in long-term survivors of myeloablative allogeneic haematopoietic cell transplantation: Prevalence and risk factors. Nephrol. Dial. Transplant. 2010, 25, 278–282. [Google Scholar] [CrossRef]
- Prókai, Á.; Csohány, R.; Sziksz, E.; Pap, D.; Balicza-Himer, L.; Boros, S.; Magda, B.; Vannay, Á.; Kis-Petik, K.; Fekete, A.; et al. Calcineurin-inhibition Results in Upregulation of Local Renin and Subsequent Vascular Endothelial Growth Factor Production in Renal Collecting Ducts. Transplantation 2016, 100, 325–333. [Google Scholar] [CrossRef]
- Wanchoo, R.; Bayer, R.L.; Bassil, C.; Jhaveri, K.D. Emerging Concepts in Hematopoietic Stem Cell Transplantation-Associated Renal Thrombotic Microangiopathy and Prospects for New Treatments. Am. J. Kidney Dis. 2018, 72, 857–865. [Google Scholar] [CrossRef]
- Catherine Joseph, J.R.A.; Benjamin, L. Laskin, sangeeta hingorani. Hematopoietic cell transplant associated kidney injury. In Onco-Nephrology; Elsevier: Amsterdam, The Netherlands, 2020; pp. 89–98. [Google Scholar]
- Redant, S.; De Bels, D.; Barbance, O.; Massaut, J.; Honoré, P.M.; Taccone, F.S.; Biarent, D. Creatinine correction to account for fluid overload in children with acute respiratory distress syndrome treated with extracorporeal membrane oxygenation: An initial exploratory report. Pediatr. Nephrol. 2022, 37, 891–898. [Google Scholar] [CrossRef]
- Van Benschoten, V.; Roy, C.; Gupta, R.; Ouellette, L.; Hingorani, S.; Li, A. Incidence and Risk Factors of Transplantation-Associated Thrombotic Microangiopathy: A Systematic Review and Meta-Analysis. Transplant. Cell Ther. 2022, 28, 266.e261–266.e268. [Google Scholar] [CrossRef]
- Siami, K.; Kojouri, K.; Swisher, K.K.; Selby, G.B.; George, J.N.; Laszik, Z.G. Thrombotic microangiopathy after allogeneic hematopoietic stem cell transplantation: An autopsy study. Transplantation 2008, 85, 22–28. [Google Scholar] [CrossRef]
- Dandoy, C.E.; Rotz, S.; Alonso, P.B.; Klunk, A.; Desmond, C.; Huber, J.; Ingraham, H.; Higham, C.; Dvorak, C.C.; Duncan, C.; et al. A pragmatic multi-institutional approach to understanding transplant-associated thrombotic microangiopathy after stem cell transplant. Blood Adv. 2021, 5, 1–11. [Google Scholar] [CrossRef]
- Jodele, S.; Davies, S.M.; Lane, A.; Khoury, J.; Dandoy, C.; Goebel, J.; Myers, K.; Grimley, M.; Bleesing, J.; El-Bietar, J.; et al. Diagnostic and risk criteria for HSCT-associated thrombotic microangiopathy: A study in children and young adults. Blood 2014, 124, 645–653. [Google Scholar] [CrossRef]
- Jodele, S.; Dandoy, C.E.; Lane, A.; Laskin, B.L.; Teusink-Cross, A.; Myers, K.C.; Wallace, G.; Nelson, A.; Bleesing, J.; Chima, R.S.; et al. Complement blockade for TA-TMA: Lessons learned from a large pediatric cohort treated with eculizumab. Blood 2020, 135, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Coppell, J.A.; Richardson, P.G.; Soiffer, R.; Martin, P.L.; Kernan, N.A.; Chen, A.; Guinan, E.; Vogelsang, G.; Krishnan, A.; Giralt, S.; et al. Hepatic veno-occlusive disease following stem cell transplantation: Incidence, clinical course, and outcome. Biol. Blood Marrow Transplant. 2010, 16, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Bonifazi, F.; Barbato, F.; Ravaioli, F.; Sessa, M.; Defrancesco, I.; Arpinati, M.; Cavo, M.; Colecchia, A. Diagnosis and Treatment of VOD/SOS After Allogeneic Hematopoietic Stem Cell Transplantation. Front. Immunol. 2020, 11, 489. [Google Scholar] [CrossRef] [PubMed]
- Mohty, M.; Malard, F.; Abecassis, M.; Aerts, E.; Alaskar, A.S.; Aljurf, M.; Arat, M.; Bader, P.; Baron, F.; Bazarbachi, A.; et al. Sinusoidal obstruction syndrome/veno-occlusive disease: Current situation and perspectives-a position statement from the European Society for Blood and Marrow Transplantation (EBMT). Bone Marrow Transplant. 2015, 50, 781–789. [Google Scholar] [CrossRef]
- Mahadeo, K.M.; McArthur, J.; Adams, R.H.; Radhi, M.; Angelo, J.; Jeyapalan, A.; Nicol, K.; Su, L.; Rabi, H.; Auletta, J.J.; et al. Consensus Report by the Pediatric Acute Lung Injury and Sepsis Investigators and Pediatric Blood and Marrow Transplant Consortium Joint Working Committees on Supportive Care Guidelines for Management of Veno-Occlusive Disease in Children and Adolescents: Part 2-Focus on Ascites, Fluid and Electrolytes, Renal, and Transfusion Issues. Biol. Blood Marrow Transplant. 2017, 23, 2023–2033. [Google Scholar] [CrossRef]
- Raina, R.; Abusin, G.A.; Vijayaraghavan, P.; Auletta, J.J.; Cabral, L.; Hashem, H.; Vogt, B.A.; Cooke, K.R.; Abu-Arja, R.F. The role of continuous renal replacement therapy in the management of acute kidney injury associated with sinusoidal obstruction syndrome following hematopoietic cell transplantation. Pediatr. Transplant. 2018, 22, e13139. [Google Scholar] [CrossRef]
- Mohty, M.; Battista, M.L.; Blaise, D.; Calore, E.; Cesaro, S.; Maximova, N.; Perruccio, K.; Renard, C.; Wynn, R.; Zecca, M.; et al. A multicentre, multinational, prospective, observational registry study of defibrotide in patients diagnosed with veno-occlusive disease/sinusoidal obstruction syndrome after haematopoietic cell transplantation: An EBMT study. Bone Marrow Transplant. 2021, 56, 2454–2463. [Google Scholar] [CrossRef]
- Alobaidi, R.; Basu, R.K.; DeCaen, A.; Joffe, A.R.; Lequier, L.; Pannu, N.; Bagshaw, S.M. Fluid Accumulation in Critically Ill Children. Crit. Care Med. 2020, 48, 1034–1041. [Google Scholar] [CrossRef]
- Alobaidi, R.; Morgan, C.; Basu, R.K.; Stenson, E.; Featherstone, R.; Majumdar, S.R.; Bagshaw, S.M. Association Between Fluid Balance and Outcomes in Critically Ill Children: A Systematic Review and Meta-analysis. JAMA Pediatr. 2018, 172, 257–268. [Google Scholar] [CrossRef]
- Goldstein, S.L.; Currier, H.; Graf, C.; Cosio, C.C.; Brewer, E.D.; Sachdeva, R. Outcome in children receiving continuous venovenous hemofiltration. Pediatrics 2001, 107, 1309–1312. [Google Scholar] [CrossRef]
- Raymakers-Janssen, P.; Lilien, M.R.; Tibboel, D.; Kneyber, M.C.J.; Dijkstra, S.; van Woensel, J.B.M.; Lemson, J.; Cransberg, K.; van den Heuvel-Eibrink, M.M.; Wosten-van Asperen, R.M. Epidemiology and Outcome of Critically Ill Pediatric Cancer and Hematopoietic Stem Cell Transplant Patients Requiring Continuous Renal Replacement Therapy: A Retrospective Nationwide Cohort Study. Crit. Care Med. 2019, 47, e893–e901. [Google Scholar] [CrossRef]
- Elbahlawan, L.; Morrison, R.; Li, Y.; Huang, S.; Cheng, C.; Avent, Y.; Madden, R. Outcome of Acute Respiratory Failure Secondary to Engraftment in Children After Hematopoietic Stem Cell Transplant. Front. Oncol. 2020, 10, 584269. [Google Scholar] [CrossRef]
- Sallee, C.J.; Smith, L.S.; Rowan, C.M.; Heckbert, S.R.; Angelo, J.R.; Daniel, M.C.; Gertz, S.J.; Hsing, D.D.; Mahadeo, K.M.; McArthur, J.A.; et al. Early Cumulative Fluid Balance and Outcomes in Pediatric Allogeneic Hematopoietic Cell Transplant Recipients With Acute Respiratory Failure: A Multicenter Study. Front. Oncol. 2021, 11, 705602. [Google Scholar] [CrossRef]
- Al-Lawati, Z.H.; Sur, M.; Kennedy, C.E.; Akcan Arikan, A. Profile of Fluid Exposure and Recognition of Fluid Overload in Critically Ill Children. Pediatr. Crit. Care Med. 2020, 21, 760–766. [Google Scholar] [CrossRef]
- Gutgarts, V.; Jain, T.; Zheng, J.; Maloy, M.A.; Ruiz, J.D.; Pennisi, M.; Jaimes, E.A.; Perales, M.A.; Sathick, J. Acute Kidney Injury after CAR-T Cell Therapy: Low Incidence and Rapid Recovery. Biol. Blood Marrow Transplant. 2020, 26, 1071–1076. [Google Scholar] [CrossRef]
- Fitzgerald, J.C.; Weiss, S.L.; Maude, S.L.; Barrett, D.M.; Lacey, S.F.; Melenhorst, J.J.; Shaw, P.; Berg, R.A.; June, C.H.; Porter, D.L.; et al. Cytokine Release Syndrome After Chimeric Antigen Receptor T Cell Therapy for Acute Lymphoblastic Leukemia. Crit. Care Med. 2017, 45, e124–e131. [Google Scholar] [CrossRef]
- Brudno, J.N.; Kochenderfer, J.N. Recent advances in CAR T-cell toxicity: Mechanisms, manifestations and management. Blood Rev. 2019, 34, 45–55. [Google Scholar] [CrossRef]
- Flores, F.X.; Brophy, P.D.; Symons, J.M.; Fortenberry, J.D.; Chua, A.N.; Alexander, S.R.; Mahan, J.D.; Bunchman, T.E.; Blowey, D.; Somers, M.J.; et al. Continuous renal replacement therapy (CRRT) after stem cell transplantation. A report from the prospective pediatric CRRT Registry Group. Pediatr. Nephrol. 2008, 23, 625–630. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, J.O.; Wald, R.; Bagshaw, S.M.; Burns, K.E.; Adhikari, N.K. Hemofiltration compared to hemodialysis for acute kidney injury: Systematic review and meta-analysis. Crit. Care 2012, 16, 1–16. [Google Scholar] [CrossRef]
- Bagshaw, S.M.; Wald, R.; Adhikari, N.K.J.; Bellomo, R.; da Costa, B.R.; Dreyfuss, D.; Du, B.; Gallagher, M.P.; Gaudry, S.; Hoste, E.A.; et al. Timing of Initiation of Renal-Replacement Therapy in Acute Kidney Injury. N. Engl. J. Med. 2020, 383, 240–251. [Google Scholar] [CrossRef]
- Zarbock, A.; Kellum, J.A.; Schmidt, C.; Van Aken, H.; Wempe, C.; Pavenstädt, H.; Boanta, A.; Gerß, J.; Meersch, M. Effect of Early vs Delayed Initiation of Renal Replacement Therapy on Mortality in Critically Ill Patients With Acute Kidney Injury: The ELAIN Randomized Clinical Trial. Jama 2016, 315, 2190–2199. [Google Scholar] [CrossRef] [PubMed]
- Ostermann, M.; Bagshaw, S.M.; Lumlertgul, N.; Wald, R. Indications for and Timing of Initiation of KRT. Clin. J. Am. Soc. Nephrol. 2023, 18, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Khwaja, A. KDIGO clinical practice guidelines for acute kidney injury. Nephron. Clin. Pract. 2012, 120, c179–c184. [Google Scholar] [CrossRef] [PubMed]
- Fayad, A.I.; Buamscha, D.G.; Ciapponi, A. Intensity of continuous renal replacement therapy for acute kidney injury. Cochrane Database Syst. Rev. 2016, 10, Cd010613. [Google Scholar] [CrossRef]
- Zarbock, A.; Küllmar, M.; Kindgen-Milles, D.; Wempe, C.; Gerss, J.; Brandenburger, T.; Dimski, T.; Tyczynski, B.; Jahn, M.; Mülling, N.; et al. Effect of Regional Citrate Anticoagulation vs Systemic Heparin Anticoagulation During Continuous Kidney Replacement Therapy on Dialysis Filter Life Span and Mortality Among Critically Ill Patients With Acute Kidney Injury: A Randomized Clinical Trial. Jama 2020, 324, 1629–1639. [Google Scholar] [CrossRef]
- Mendu, M.L.; Ciociolo, G.R., Jr.; McLaughlin, S.R.; Graham, D.A.; Ghazinouri, R.; Parmar, S.; Grossier, A.; Rosen, R.; Laskowski, K.R.; Riella, L.V.; et al. A Decision-Making Algorithm for Initiation and Discontinuation of RRT in Severe AKI. Clin. J. Am. Soc. Nephrol. 2017, 12, 228–236. [Google Scholar] [CrossRef]
- Liu, C.; Peng, Z.; Dong, Y.; Li, Z.; Andrijasevic, N.M.; Albright, R.C., Jr.; Kashani, K.B. Predicting successful continuous renal replacement therapy liberation in critically ill patients with acute kidney injury. J. Crit. Care 2021, 66, 6–13. [Google Scholar] [CrossRef]
- Elbahlawan, L.; Morrison, R.R. Continuous renal replacement therapy in children post-hematopoietic stem cell transplantation: The present and the future. Curr. Stem. Cell Res. Ther. 2012, 7, 381–387. [Google Scholar] [CrossRef]
- Yang, T.; Sun, S.; Lin, L.; Han, M.; Liu, Q.; Zeng, X.; Zhao, Y.; Li, Y.; Su, B.; Huang, S.; et al. Predictive Factors Upon Discontinuation of Renal Replacement Therapy for Long-Term Chronic Dialysis and Death in Acute Kidney Injury Patients. Artif. Organs 2017, 41, 1127–1134. [Google Scholar] [CrossRef]
- Charlton, J.R.; Portilla, D.; Okusa, M.D. A basic science view of acute kidney injury biomarkers. Nephrol. Dial. Transplant. 2014, 29, 1301–1311. [Google Scholar] [CrossRef]
- Nickolas, T.L.; Schmidt-Ott, K.M.; Canetta, P.; Forster, C.; Singer, E.; Sise, M.; Elger, A.; Maarouf, O.; Sola-Del Valle, D.A.; O’Rourke, M.; et al. Diagnostic and prognostic stratification in the emergency department using urinary biomarkers of nephron damage: A multicenter prospective cohort study. J. Am. Coll. Cardiol. 2012, 59, 246–255. [Google Scholar] [CrossRef]
- Augustynowicz, M.; Bargenda-Lange, A.; Kałwak, K.; Zwolińska, D.; Musiał, K. Markers of acute kidney injury in children undergoing hematopoietic stem cell transplantation. Adv. Clin. Exp. Med. 2019, 28, 1111–1118. [Google Scholar] [CrossRef]
- Shao, X.; Tian, L.; Xu, W.; Zhang, Z.; Wang, C.; Qi, C.; Ni, Z.; Mou, S. Diagnostic value of urinary kidney injury molecule 1 for acute kidney injury: A meta-analysis. PLoS ONE 2014, 9, e84131. [Google Scholar] [CrossRef]
- Susantitaphong, P.; Siribamrungwong, M.; Doi, K.; Noiri, E.; Terrin, N.; Jaber, B.L. Performance of urinary liver-type fatty acid-binding protein in acute kidney injury: A meta-analysis. Am. J. Kidney Dis. 2013, 61, 430–439. [Google Scholar] [CrossRef]
- Suzuki, G.; Ichibayashi, R.; Yamamoto, S.; Nakamichi, Y.; Watanabe, M.; Honda, M. Clinical significance of urinary L-FABP in the emergency department. Int. J. Emerg. Med. 2019, 12, 24. [Google Scholar] [CrossRef]
- Suzuki, G.; Ichibayashi, R.; Yamamoto, S.; Serizawa, H.; Nakamichi, Y.; Watanabe, M.; Honda, M. Urinary liver-type fatty acid-binding protein variation as a predictive value of short-term mortality in intensive care unit patients. Ren. Fail. 2021, 43, 1041–1048. [Google Scholar] [CrossRef]
- Zeng, X.F.; Li, J.M.; Tan, Y.; Wang, Z.F.; He, Y.; Chang, J.; Zhang, H.; Zhao, H.; Bai, X.; Xie, F.; et al. Performance of urinary NGAL and L-FABP in predicting acute kidney injury and subsequent renal recovery: A cohort study based on major surgeries. Clin. Chem. Lab. Med. 2014, 52, 671–678. [Google Scholar] [CrossRef]
- Gauer, S.; Sichler, O.; Obermüller, N.; Holzmann, Y.; Kiss, E.; Sobkowiak, E.; Pfeilschifter, J.; Geiger, H.; Mühl, H.; Hauser, I.A. IL-18 is expressed in the intercalated cell of human kidney. Kidney Int. 2007, 72, 1081–1087. [Google Scholar] [CrossRef]
- Gonzalez, F.; Vincent, F. Biomarkers for acute kidney injury in critically ill patients. Minerva Anestesiol 2012, 78, 1394–1403. [Google Scholar]
- Kashani, K.; Al-Khafaji, A.; Ardiles, T.; Artigas, A.; Bagshaw, S.M.; Bell, M.; Bihorac, A.; Birkhahn, R.; Cely, C.M.; Chawla, L.S.; et al. Discovery and validation of cell cycle arrest biomarkers in human acute kidney injury. Crit. Care 2013, 17, 1–12. [Google Scholar] [CrossRef]
- Xie, Y.; Ankawi, G.; Yang, B.; Garzotto, F.; Passannante, A.; Breglia, A.; Digvijay, K.; Ferrari, F.; Brendolan, A.; Raffaele, B.; et al. Tissue inhibitor metalloproteinase-2 (TIMP-2) • IGF-binding protein-7 (IGFBP7) levels are associated with adverse outcomes in patients in the intensive care unit with acute kidney injury. Kidney Int. 2019, 95, 1486–1493. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Eckardt, K.U.; Dorman, N.M.; Christiansen, S.L.; Cheung, M.; Jadoul, M.; Winkelmayer, W.C. Nomenclature for Kidney Function and Disease: Executive Summary and Glossary from a Kidney Disease: Improving Global Outcomes (KDIGO) Consensus Conference. Kidney Dis. Basel. 2020, 6, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Hingorani, S.; Guthrie, K.A.; Schoch, G.; Weiss, N.S.; McDonald, G.B. Chronic kidney disease in long-term survivors of hematopoietic cell transplant. Bone Marrow Transplant. 2007, 39, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Hingorani, S. Chronic kidney disease after pediatric hematopoietic cell transplant. Biol. Blood Marrow Transplant. 2008, 14, 84–87. [Google Scholar] [CrossRef]
- Prasad, M.; Jain, N.G.; Radhakrishnan, J.; Jin, Z.; Satwani, P. Risk factors for chronic kidney disease following acute kidney injury in pediatric allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2021, 56, 1665–1673. [Google Scholar] [CrossRef]
- Hingorani, S.R.; Seidel, K.; Lindner, A.; Aneja, T.; Schoch, G.; McDonald, G. Albuminuria in hematopoietic cell transplantation patients: Prevalence, clinical associations, and impact on survival. Biol. Blood Marrow Transplant. 2008, 14, 1365–1372. [Google Scholar] [CrossRef]
- Hingorani, S. Renal Complications of Hematopoietic-Cell Transplantation. N. Engl. J. Med. 2016, 374, 2256–2267. [Google Scholar] [CrossRef]
- Flynn, J.T.; Kaelber, D.C.; Baker-Smith, C.M. Subcommittee on screening and management of high blood pressure in children. Clinical Practice Guideline for Screening and Management of High Blood Pressure in Children and Adolescents. Pediatrics. Pediatrics 2017, 140, e20171904. [Google Scholar] [CrossRef]
- Baltatzi, M.; Savopoulos, C.; Hatzitolios, A. Role of angiotensin converting enzyme inhibitors and angiotensin receptor blockers in hypertension of chronic kidney disease and renoprotection. Study results. Hippokratia 2011, 15, 27–32. [Google Scholar]
- Wühl, E.; Trivelli, A.; Picca, S.; Litwin, M.; Peco-Antic, A.; Zurowska, A.; Testa, S.; Jankauskiene, A.; Emre, S.; Caldas-Afonso, A.; et al. Strict blood-pressure control and progression of renal failure in children. N. Engl. J. Med. 2009, 361, 1639–1650. [Google Scholar] [CrossRef]
| Study | No. of Patients | AKI Criteria | Incidence of AKI | Risk Factors | Survival |
|---|---|---|---|---|---|
| Daraskevicius et al. [3] | 51 | pRIFLE | 35.3% | Older age Higher BMI | |
| Koh et al. [4] | 1057 | AKIN | 68.2% Stage 1: 22% Stage 2: 41.4% Stage 3: 36.6% KRT: 30.3% in patients with Stage 3 | Older age Myeloablative conditioning Unrelated donor SOS aGVHD grade II-IV | 1 year OS No AKI: 66.1% Stage 1: 73.4% Stage 2: 63.9% Stage 3: 47.3% Stage 3 requiring KRT: 7.5% |
| Kizilbash et al. [5] | 205 | pRIFLE | 84% R: 71 (35%) I: 67 (33%) F/L/E without dialysis: 21 (10%) F/L/E with dialysis: 14 (7%) | Race: African American SOS | HR mortality F/L/E 8.41 (4.55–15.53) |
| Satwani et al. [6] | 170 | ≥50% decrease in eCrCl from the baseline | 33% at 30 d post HCT 55% at 3 m post HCT CKRT: 14.7% | Myeloablative conditioning | HR mortality: 2 with AKI at 1 m |
| Yu et al. [7] | 96 | 29.2% Stage 1: 14.6% Stage 2: 12.5% Stage 3: 2.1% | aGVHD Myeloablative conditioning | Mortality Grade 1: 57.1% Grades 2–3: 78.6% | |
| Ileri et al. [8] | 57 | AKIN | 42% Stage 1: 21% Stage 2: 16% Stage 3: 5% | Cyclosporin A Amphotericin B SOS | Mortality: 21% within 100 d |
| Hazar et al. [9] | 34 | 26.4% | Mortality: 39% within 6 m | ||
| Kist-van Holthe et al. [10] | 66 | 21% | SOS Cyclosporin A Foscarnet |
| Stage | AKIN Staging | KDIGO Staging | pRIFLE Staging |
|---|---|---|---|
| Stage 1 | Scr: 1.5–2.0x Bl OR ≥ 0.3 mg/dL 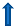 | Scr:1.5–1.9x Bl OR ≥ 0.3 mg/dL 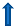 | R = Risk for renal dysfunction eGFR by 25%  |
| UO: <0.5 mL/kg/h for >6 h | <0.5 mL/kg/h for 6–12 h | <0.5 mL/kg/h for 8 h | |
| Stage 2 | Scr: >2–3x Bl | Scr: 2.0–2.9x Bl | I = Injury to the kidney eGFR  by 50% by 50% |
| UO: <0.5 mL/kg/h for >12 h | <0.5 mL/kg/h for 12–24 h | <0.5 mL/kg/h for 16 h | |
| Stage 3 | Scr: >3x Bl OR KRT | Scr: 3.0x Bl OR ≥ 4.0 mg/dL OR KRT OR eGFR < 35 mL/min/1.73 m2 | F = Failure of kidney function eGFR  by 75% by 75% OR eGFR < 35 mL/min/1.73 m2 |
| UO: <0.3 mL/kg/h for 24 h OR Anuria for 12 h | <0.3 mL/kg/h for ≥24 h OR Anuria for ≥12 h | <0.3 mL/kg/h for 24 h OR Anuria for 12 h L = Loss of kidney function Persistent failure > 4 weeks E = End-stage renal disease Persistent failure > 3 months |
| Biomarker | Characteristic | Detection Time | Peak | AUC for AKI Detection | Limitations |
|---|---|---|---|---|---|
| Glomerular injury | |||||
| Cystatin C | 13-kDa protein that is present in all nucleated cells, protease inhibitor | 2–48 h | 6–8 h | Influenced by inflammation, muscle mass, and high-dose steroids | |
| Renal tubular injury | |||||
| NGAL | 25-kDa protein of the family of lipocalins with bacteriostatic function | 2–24 h | 6–12 h | 0.8 (0.72–0.87) | False elevation in sepsis and malignancy |
| NAG | >130-kDa proximal tubular lysosomal enzyme | 2–4 h | 0.6 | Elevated in diabetes and albuminuria | |
| KIM 1 | 38.7-kDa type I transmembrane glycoprotein | 1–24 h | 0.85 | Slow rise and non-specific May be elevated in the settings of chronic proteinuria and inflammatory diseases | |
| Interleukin-18 | 24-kDa cytokine | 4–48 h | 12 h | 0.75 | Low sensitivity/specificity |
| L-FABP | 14-kDa lipid binding protein | 12–72 h | May lose its specificity when liver disease is present | ||
| TIMP 2 | 21-kDa protein, endogenous inhibitor of metalloproteinase activities, involved in G1 cycle arrest | 1–12 h | 0.8 | Proteinuria interferes with the test results Elevated in diabetes | |
| IGFBP7 | 29-kDa protein, IGF-1 receptor antagonist, involved in G1 cycle arrest | 0.76 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
James, V.; Angelo, J.; Elbahlawan, L. Kidney Injury in Children after Hematopoietic Stem Cell Transplant. Curr. Oncol. 2023, 30, 3329-3343. https://doi.org/10.3390/curroncol30030253
James V, Angelo J, Elbahlawan L. Kidney Injury in Children after Hematopoietic Stem Cell Transplant. Current Oncology. 2023; 30(3):3329-3343. https://doi.org/10.3390/curroncol30030253
Chicago/Turabian StyleJames, Vinson, Joseph Angelo, and Lama Elbahlawan. 2023. "Kidney Injury in Children after Hematopoietic Stem Cell Transplant" Current Oncology 30, no. 3: 3329-3343. https://doi.org/10.3390/curroncol30030253
APA StyleJames, V., Angelo, J., & Elbahlawan, L. (2023). Kidney Injury in Children after Hematopoietic Stem Cell Transplant. Current Oncology, 30(3), 3329-3343. https://doi.org/10.3390/curroncol30030253





