1. Introduction
Currently antiresorptive agents (ARs), such as bisphosphonate (BP) and denosumab (DMB), are widely used to reduce bone resorption due to osteoporosis and skeletal-related events from metastatic bone tumors and multiple myeloma [
1,
2,
3,
4,
5,
6]. However, the presence of medication-related osteonecrosis of the jaw (MRONJ) is one of the severe side effects of ARs [
7,
8,
9,
10,
11]. The American Association of Oral and Maxillofacial Surgeons (AAOMS) position paper in 2014 [
7] recommends a 2-month AR drug holiday before tooth extraction for patients with osteoporosis taking ARs for more than 4 years or with risk factors, such as diabetes or steroid administration; although there is not sufficient evidence to determine whether ARs should be withdrawn. The AAOMS position paper was revised in 2022 [
12]; the working group was unable to reach a consensus for drug holidays recommendations, which will be addressed on a case-by-case basis. However, there are several negative reports on AR drug holidays before oral surgery, and the necessity and duration of AR drug holidays before oral surgery are controversial [
13].
BP is deposited on the bone matrix and taken up by osteoclasts during the bone resorption process, causing apoptosis of osteoclasts by destroying their cytoskeleton and inhibiting bone resorption [
14]. BP deposition in the bone matrix is considered to last from several months to several years. In contrast, DMB selectively inhibits the receptor activator of nuclear factor kappa B ligand (RANKL) expressed on osteoblasts, and suppresses osteoclast differentiation, function, and survival, thereby reducing bone resorption [
15]. Considering these pharmacokinetic distinctions, there may be a difference in the drug holiday duration required to recover osteoclast function between BP and DMB, although few reports have examined drug holidays and the clinical significance of BP or DMB.
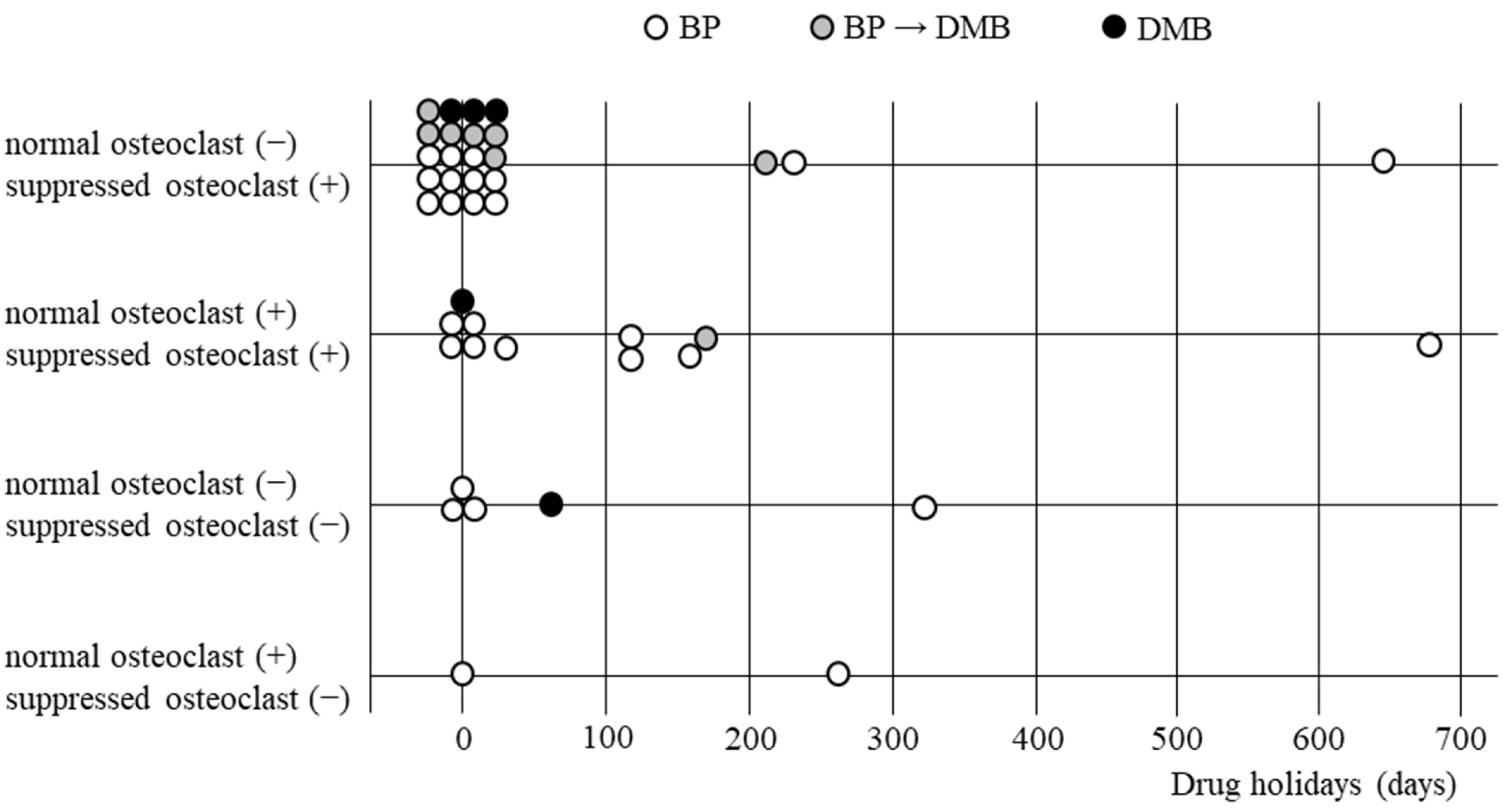
Morishita et al. [
16] recently histologically examined the relationship between drug holidays duration and osteoclast suppression in surgical specimens from patients treated with oral BPs and the reported osteoclast suppression was not recovered after an approximate 6-month drug holiday. However, there are no reports examining the relationship between the DMB drug holiday duration and osteoclast suppression. The purpose of this study is to examine whether the withdrawal of ARs recovers osteoclast suppression and whether there are clinical benefits for withdrawal in osteoporosis patients with MRONJ receiving BP and DMB.
2. Materials and Methods
2.1. Impact of AR Drug Holidays on Treatment Outcomes
A total of 166 osteoporosis patients with MRONJ were treated at the Oral and Maxillofacial Surgery of Nagasaki University Hospital between January 2012 to December 2020, of which 139 were treated with surgical therapy and 27 with conservative therapy. Some of the cases used in this study overlap with some of the cases used in our group’s previous reports on treatment methods for MRONJ, such as reports on treatment methods for maxillary MRONJ [
17], reports on the relationship between drug holiday and sequestrum separation [
18], and reports on segmental mandibulectomy [
19]. Diagnostic criteria for MRONJ are based on the AAOMS Position Paper [
7]. Sex, age, site (upper jaw/lower jaw), stage according to the criteria of AAOMS Position Paper [
7], AR type (BP, DMB), administration period (<4 years/≥4 years), corticosteroid, diabetes, serum albumin, serum creatinine, treatment methods, AR drug holiday (<3 months/≥3 months), and treatment outcomes were investigated from the medical records. We have made surgical therapy the first choice for MRONJ regardless of the MRONJ stage, but conservative therapy was selected when surgery was not possible due to the patient’s wishes or poor general condition. Conservative therapy included the use of antimicrobial mouthwash, rinsing of fistulas, periodontal pockets, or gaps between exposed necrotic bone and surrounding soft tissue, and administration of oral antimicrobial agents. Surgical therapy included marginal resection such as partial maxillectomy and marginal mandibulectomy, and segmental mandibulectomy (
Figure 1).
We have resected not only the sequestrum but some healthy bone around it. The extent of bone resection was planned based on preoperative CT images but was ultimately determined based on the intraoperative findings such as the presence or absence of bleeding from the bone and changes in bone color. Healing was defined as the disappearance of all symptoms, including bone exposure, and the time to healing was also recorded. Non-healing included improved symptoms, unchanged, and worsened symptoms, and the follow-up period was also recorded. Patients whose symptoms disappeared once and were judged to be “healing” but later relapsed were classified as “non-healing”.
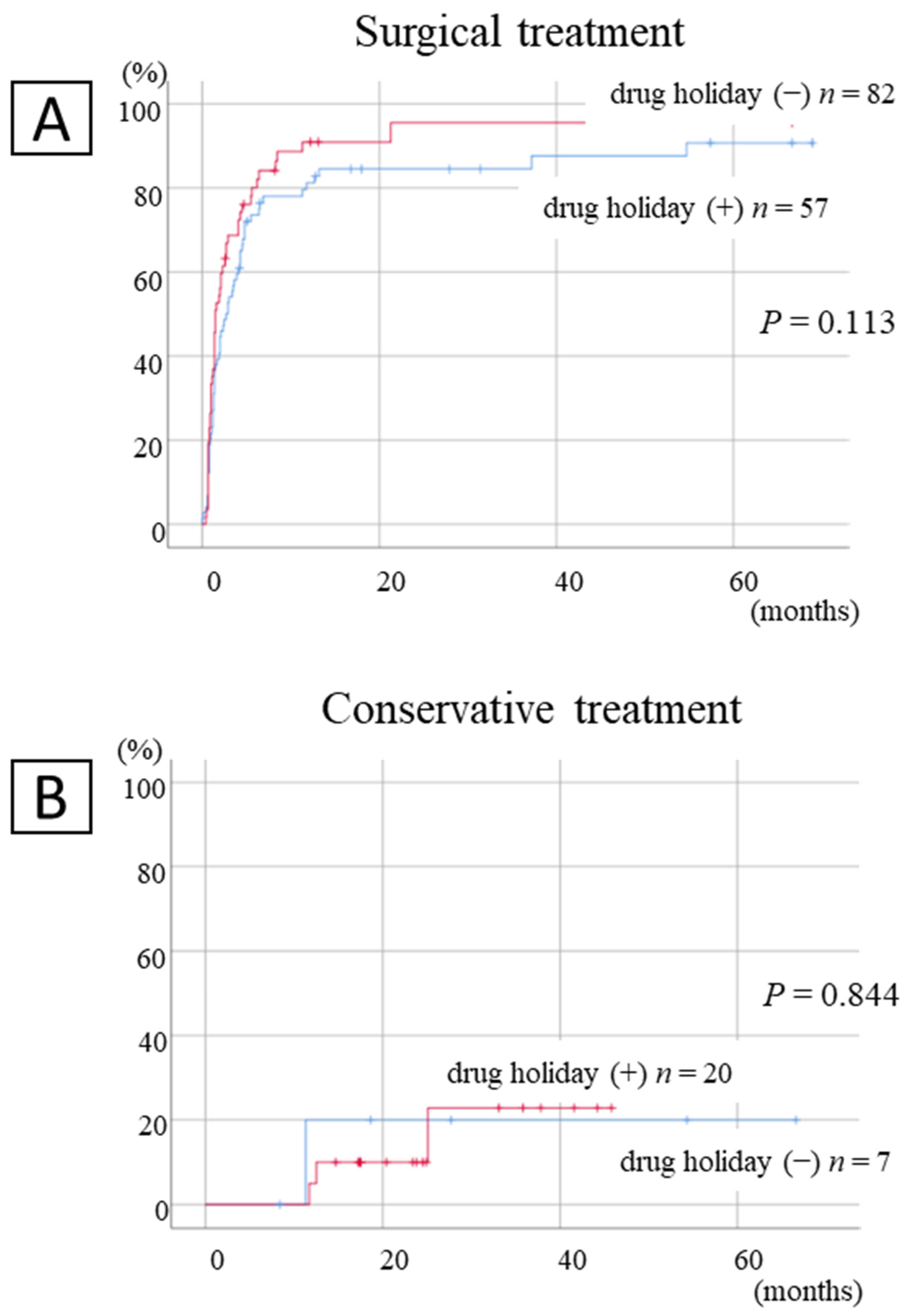
The cumulative cure rate with and without drug suspension was obtained using the Kaplan-Meier method and tested by the log-rank test, divided into surgical cases and preserved cases. Furthermore, each variable examined was analyzed for association with healing rates using a univariate Cox regression analysis, followed by a multiple Cox proportional hazard model including variables with p-values smaller than 0.1 in the univariate analysis and the drug holidays longer than 3 months as covariates. The relationship between AR drug holidays and the healing rate was determined using the Kaplan-Meier method, separately for conservative and surgical therapy cases. All statistical analyses were performed using SPSS software (version 26.0; Japan IBM Co., Ltd., Tokyo, Japan), and a 2-tailed p-value of <0.05 was considered significant.
2.2. Histological Investigation of Recovery from Osteoclast Suppression and Drug Holidays
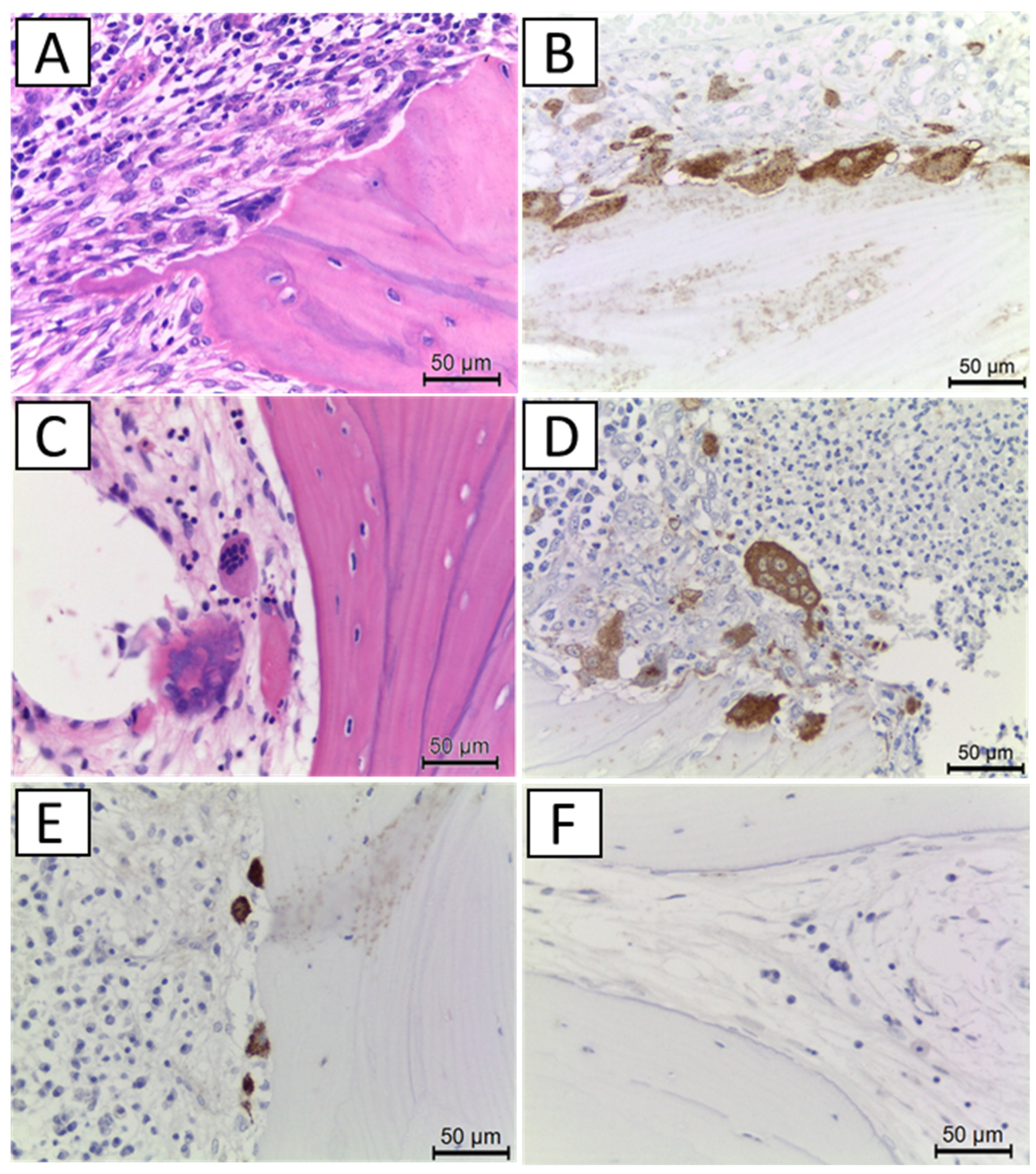
Histopathology sections were decalcified in 0.5 M ethylenediaminetetraacetic acid solution with gentle agitation at 4 °C, postfixed in a fixing solution, dehydrated, and embedded in paraffin to prepare 4-μm-thick sections. Antigen activation in immunostaining was performed in Dako Target Retrieval Solution (DAKO, Carpinteria, CA, USA) using the warm bath method at 80 °C for 15 min. The primary antibody was rabbit anti-cathepsin K polyclonal antibody (1:2000, Abcum: ab19027) and incubated at 4 °C overnight. Secondary antibodies were EnVision+ System- HRP Labelled Polymer Anti-Rabbit (DAKO). For immunohistochemical reactions, Liquid DAB+ Substrate Chromogen System (DAKO) was used according to the manufacturer’s protocol. Mayer’s hematoxylin was used for nuclear staining.
The number and morphology of osteoclasts in the tissue sections were evaluated by hematoxylin and eosin staining and cathepsin K immunostaining (
Figure 2). Cathepsin K-positive, multinucleated giant cells around the bone surface were identified as normal osteoclasts. On the other hand, cathepsin K-positive, giant multinucleated cells released from the bone surface, cathepsin K-positive mononuclear or small multinucleated cells on the bone surface, and no cathepsin-K positive multinucleated cells on the bone surface were regarded as suppressed osteoclasts.
4. Discussion
In this study, we investigated the relationship between the AR drug holidays and treatment outcomes in osteoporosis patients receiving ARs who underwent surgical treatment, and histopathological examinations of osteoclast suppression were also conducted. The results indicated that osteoclast suppression was not recovered by 3-month drug holidays in patients administered BP or DMB, and 3-month drug holidays did not contribute to the improvement of the treatment outcomes.
According to Damm et al. [
20], the skeletal system has a mixture of resting surfaces (osteocytes: 85%), resorbing surfaces (osteoclasts: 2%), and forming surfaces (osteoblasts: 10–12%). The lifespan of bone cells is several to 10 years, the lifespan of osteoclasts is 2 weeks, and the lifespan of osteoblasts is approximately 1 to 3 months. BP and DMB suppress bone resorption by different mechanisms, but both exert a bone resorption inhibitory effect by suppressing osteoclasts. Therefore, impaired bone remodeling is considered one of the causes of MRONJ onset. Approximately 50% of the BP taken into the serum is rapidly excreted from the kidney within a few hours, thus that the blood concentration decreases rapidly. However, BP specifically binds to the bone matrix, and the BP incorporated into the bone matrix is considered to remain in the bone until remodeling; its half-life ranges from several to 10 years. BP is released from the bone matrix in the acidic environment of the resorption fossa during the bone resorption process of osteoclasts, and the osteoclasts take up this to destroy the osteoclast skeleton, thereby inducing apoptosis and suppressing bone resorption. Osteoclasts under the influence of BP are observed as polynuclear, large osteoclasts detached from the bone surface [
21].
In contrast, DMB taken up in serum circulates in the blood including blood vessels in the bone marrow. The half-life in blood is 25.4 days. Unlike the BP preparation, it does not show specific binding to the bone matrix and does not accumulate. Therefore, the action of DMB is considered to be more reversible than the BP preparation [
22]. By specifically binding to RANKL, DMB inhibits the binding of RANKL to the receptor activator of nuclear factor kappa B (RANK) expressed on the surface of osteoclast precursor cells, suppresses differentiation into mature osteoclasts, and activates the existing mature osteoclasts. It also suppresses osteoclasts and survival. As a result, the total number of osteoclasts under the influence of DMB decreases, and small immature osteoclasts can be observed [
23]. Since we wanted to investigate the difference between the period of drug holiday osteoclast inhibition in both drugs, we registered both BP and DMB cases as target cases this time. Although the number of DMB cases was small and a clear conclusion could not be drawn, we were able to suggest that osteoclasts do not recover even in DMB cases after several months of withdrawal.
Several conflicting opinions have been reported regarding the effects of AR drug holidays before tooth extraction. Otto et al. [
24] demonstrated that zoledronic acid withdrawal before and after tooth extraction reduced the incidence of MRONJ in an animal experimental model. In contrast, Salgueiro et al. [
25] stated that a 5-month drug holiday of the injectable BP drug does not affect the onset of MRONJ. Hasegawa et al. [
13,
26,
27] also found in a multicenter study with a large number of cases that drug suspension did not affect the incidence of MRONJ after tooth extraction in osteoporosis patients receiving BP and high-dose ARs. In clinical practice, there is no report with a high level of evidence indicating the incidence of MRONJ decreased due to drug suspension before tooth extraction.
Regarding AR drug holidays during MRONJ treatment, the AAOMS and JSOMS position papers stated that it is desirable to withdraw ARs from the time of MRONJ diagnosis until the end of treatment [
7,
8]. Kim et al. [
28] reported that in the MRONJ surgical treatment group, the treatment results of the 4-months or more drug holiday group were better than those of the non-drug group. Ramaglia et al. [
29] also demonstrated that a drug holiday protocol promotes post-surgery healing, but the application must be determined for each patient’s condition. However, their papers are retrospective studies with a small number of cases and the level of evidence is low. In contrast, Hayashida et al. [
30] collected a large number of MRONJ cases in a multicenter study and reported that AR drug holidays for approximately 3 months before surgery did not affect the treatment results of MRONJ surgery. Morishita et al. [
18] reported that 2- to 6-month AR drug holidays before surgery did not affect treatment outcomes in osteoporosis patients with MRONJ. Otsuru et al. [
31] also found that 2 to 4 months of preoperative high-dose AR drug holidays did not affect treatment outcomes in patients with cancer. As described above, the effectiveness of AR drug holidays during MRONJ treatment is also controversial, but there is no report with a high level of evidence that drug holidays improve MRONJ’s surgical results. In the present study, it was clarified that drug holidays of BP and DMB for several months did not recover the osteoclast suppression and did not affect the treatment outcome of MRONJ. Therefore, we believe that withdrawal of ARs should not be performed before tooth extraction or during MRONJ treatment.
This study has several limitations. Since the study was retrospective with a small number of cases, the criteria and duration of AR drug holidays differed depending on the case, and the patient background varied greatly, thus it is difficult to generalize the results obtained. The cases used in the histological examination were limited to those who underwent marginal resection or segmental resection, and selection bias may have occurred. For future research, we are considering increasing the number of cases, conducting more detailed examinations, and searching for local bone metabolism markers in molecular biology as well as a histological search.