Immune-Enhancement and Anti-Inflammatory Activities of Fatty Acids Extracted from Halocynthia aurantium Tunic in RAW264.7 Cells
Abstract
1. Introduction
2. Results
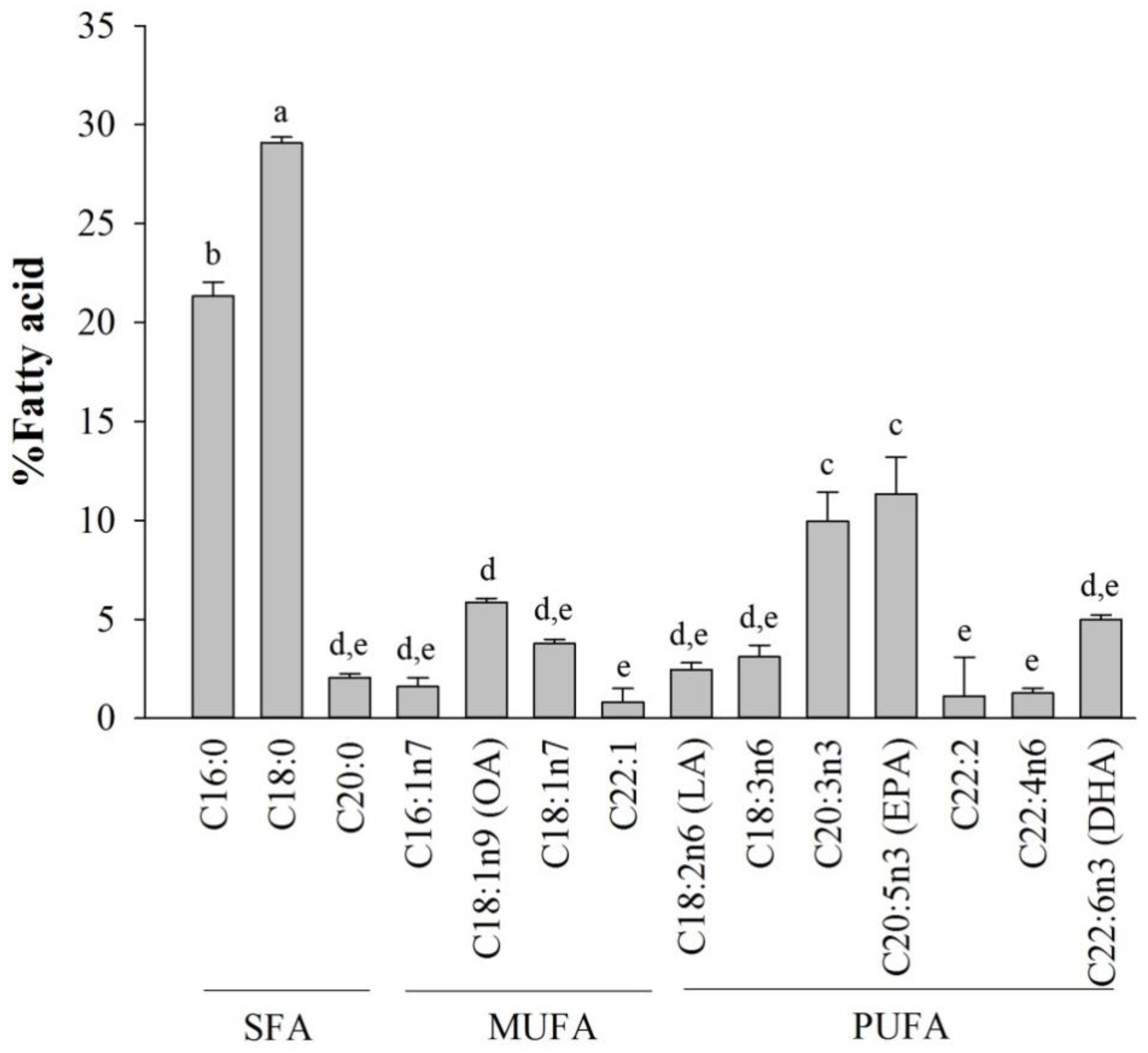
2.1. Fatty Acid Analysis of Halocynthia aurantium Tunic
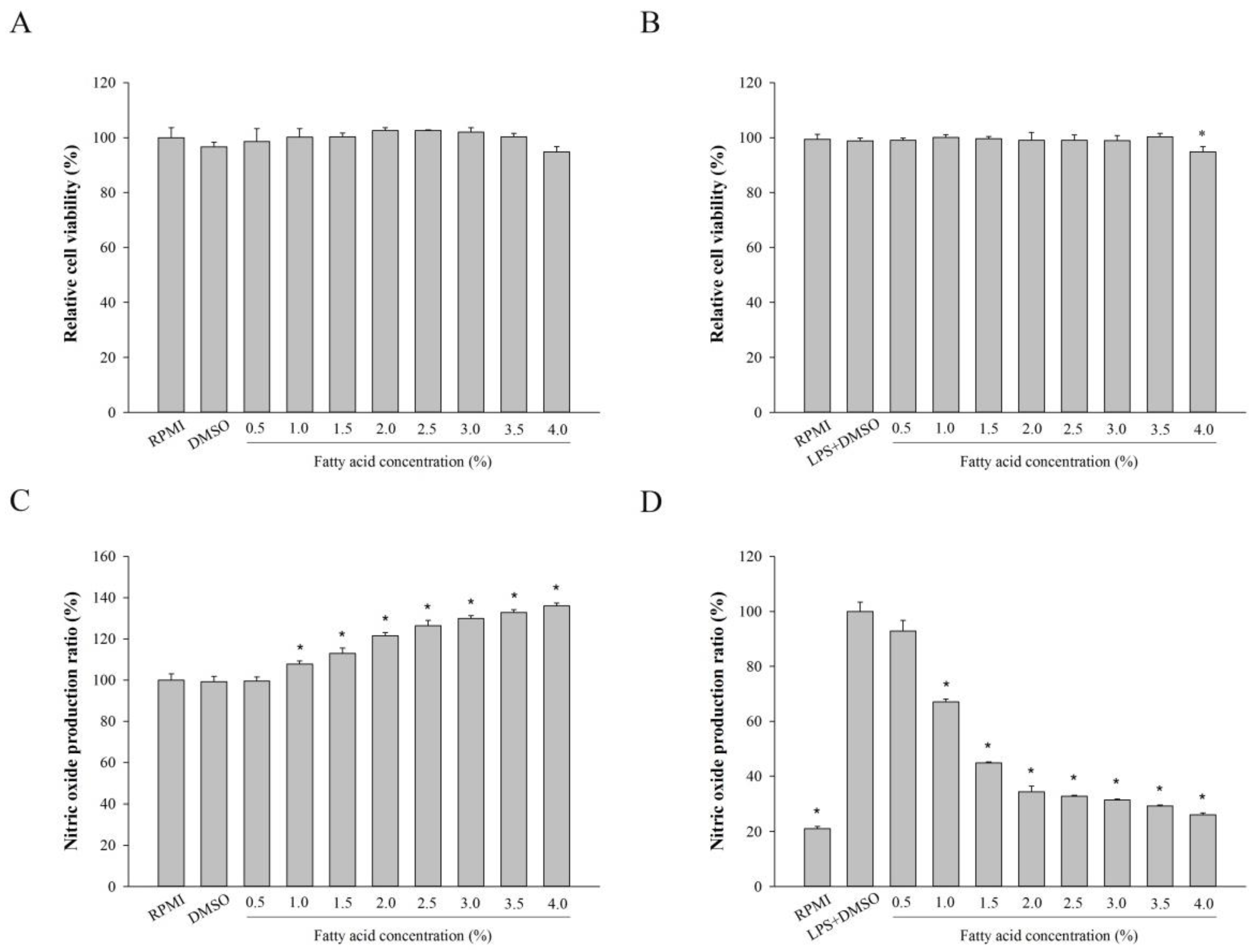
2.2. Effect of Fatty Acids from H. aurantium, Tunic Cytotoxicity and NO Production on Macrophages
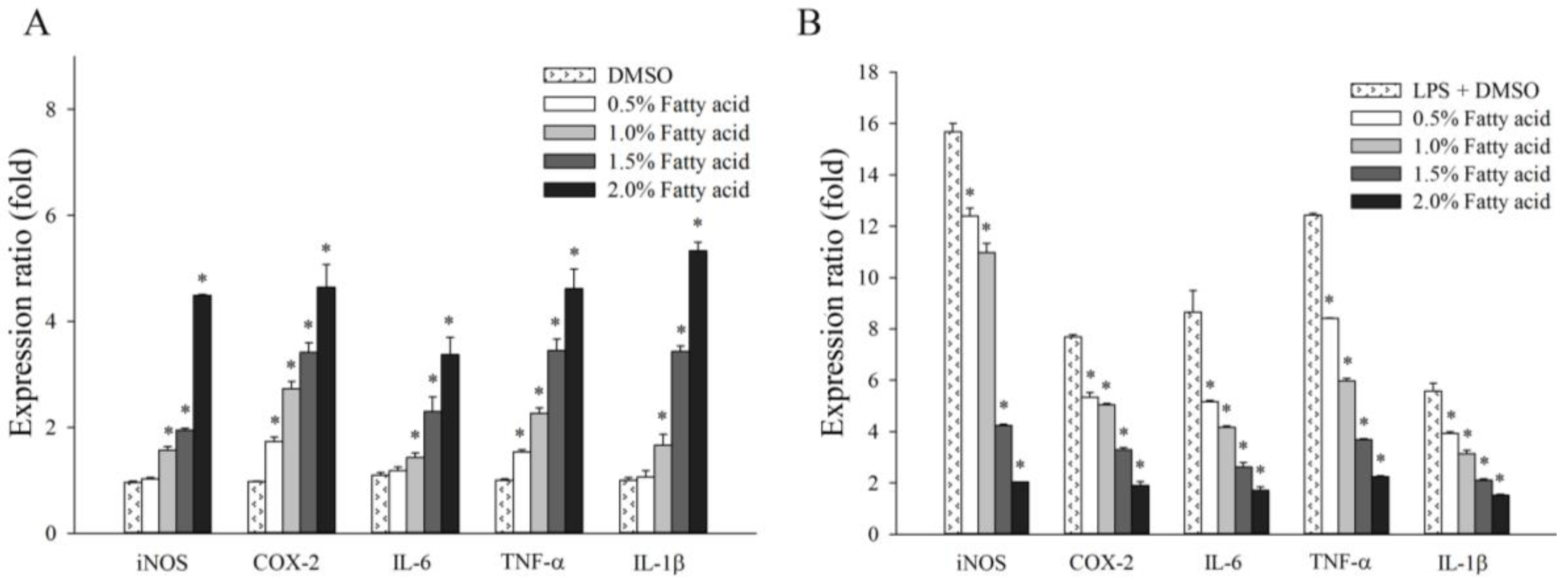
2.3. Effect of Fatty Acids from H. aurantium Tunic on Immune Gene Expressions
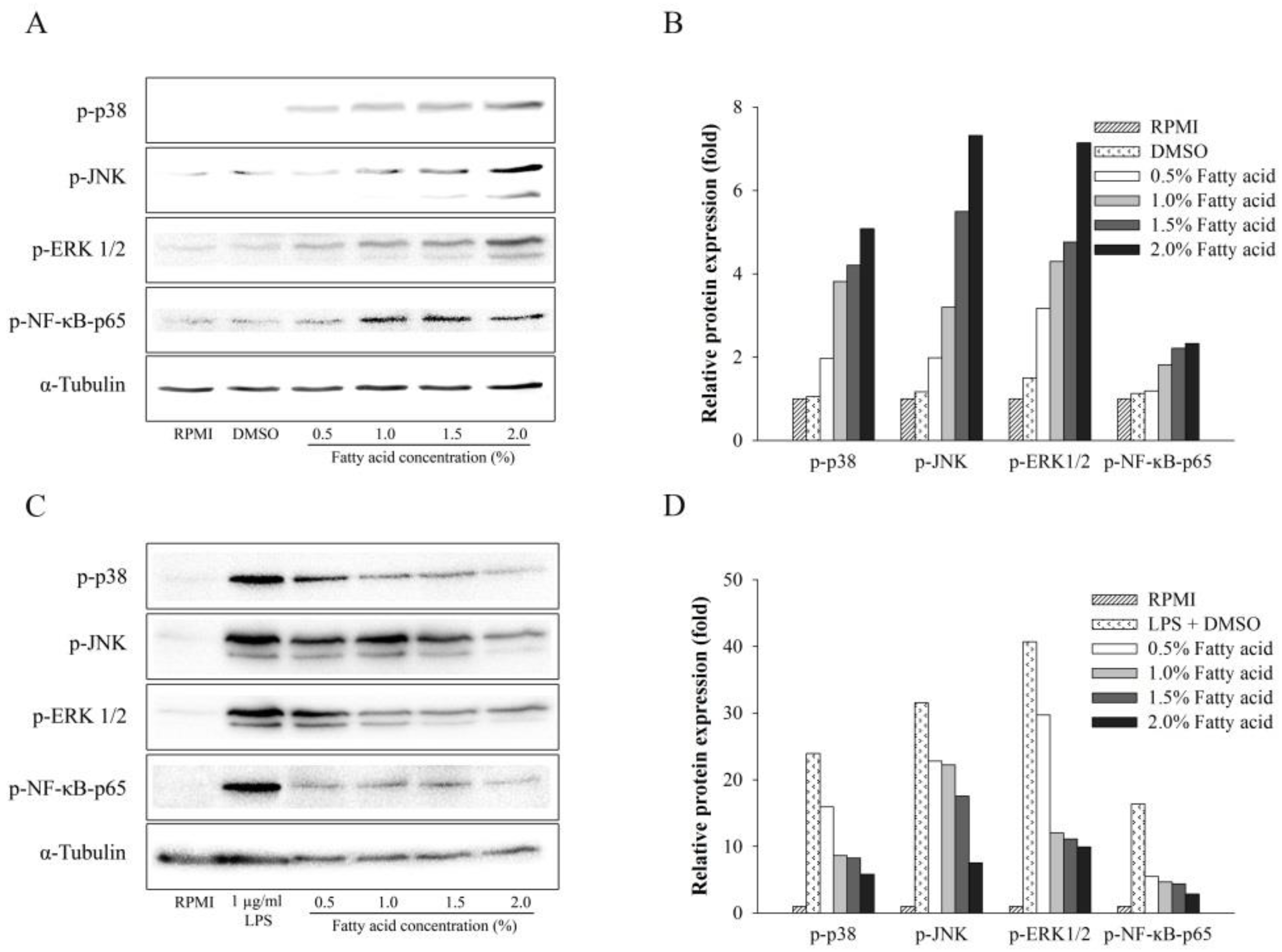
2.4. Effect of Fatty Acids from H. aurantium Tunic on MAPK and NF-κB Signaling Pathways
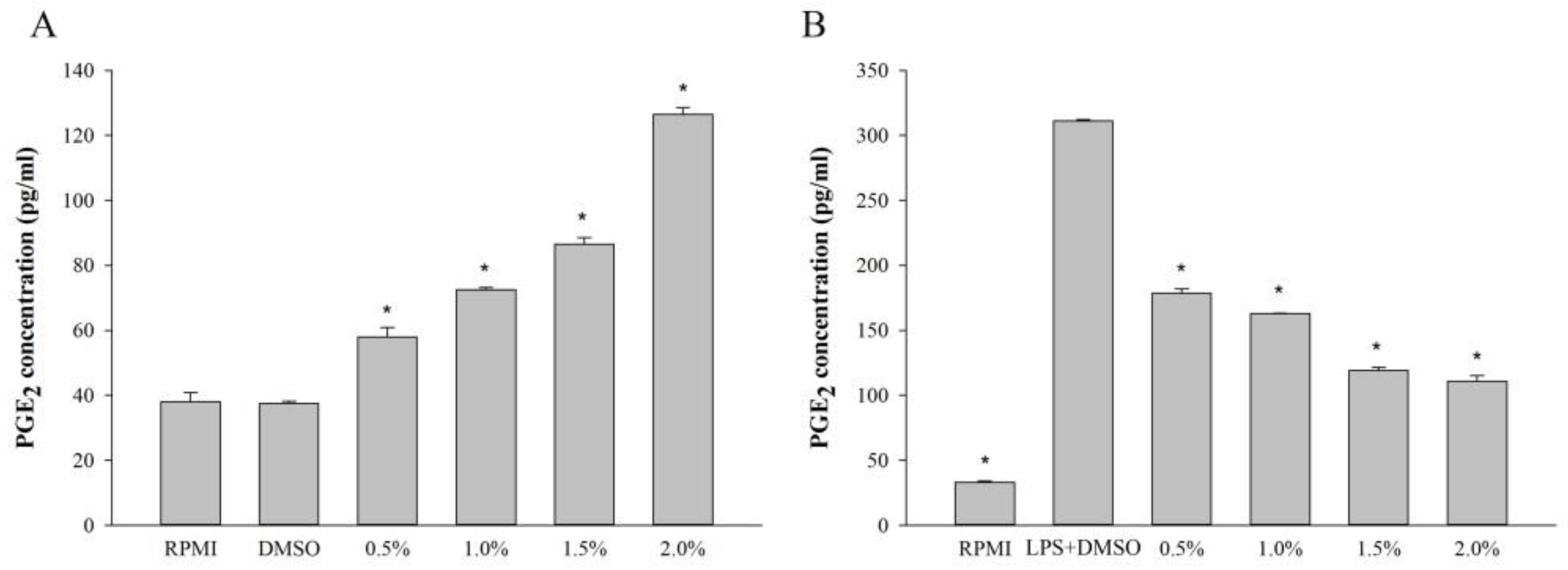
2.5. Effect of Fatty Acids from H. aurantium Tunic on PGE2 Levels
3. Discussion
4. Materials and Methods
4.1. Halocynthia aurantium Sample
4.2. Fatty Acid Extraction and Analysis
4.3. Macrophage Proliferation and Nitric Oxide Production
4.4. RNA Isolation and cDNA Synthesis
4.5. Expression Analysis of Immune Gene by Real-Time PCR
4.6. Western Blot Assay
4.7. Quantification of PGE2
4.8. Statistics
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Roche, H.M. Unsaturated fatty acids. Proc. Nutr. Soc. 1999, 58, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Walls, J.; Sinclair, L.; Finlay, D. Nutrient sensing, signal transduction and immune responses. Semin. Immunol. 2016, 28, 396–407. [Google Scholar] [CrossRef] [PubMed]
- Superko, H.R.; Superko, A.R.; Lundberg, G.P.; Margolis, B.; Garrett, B.C.; Nasir, K.; Agatston, A.S. Omega-3 fatty acid blood levels clinical significance update. Curr. Cardiovasc. Risk Rep. 2014, 8, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Ford, J.H. Saturated fatty acid metabolism is key link between cell division, cancer, and senescence in cellular and whole organism aging. Age (Dordr) 2010, 32, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.S.; Huang, T.; Yang, J.; Fu, Y.Q.; Li, D. Marine N-3 polyunsaturated fatty acids are inversely associated with risk of type 2 diabetes in Asians: A systematic review and meta-analysis. PLoS ONE 2012, 7, e44525. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Omega-3 fatty acids in inflammation and autoimmune diseases. J. Am. Coll. Nutr. 2002, 21, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 polyunsaturated fatty acids and inflammatory processes: Nutrition or pharmacology? Br. J. Clin. Pharmacol. 2013, 75, 645–662. [Google Scholar] [CrossRef] [PubMed]
- Davidson, J.; Rotondo, D.; Rizzo, M.T.; Leaver, H.A. Therapeutic implications of disorders of cell death signalling: Membranes, micro-environment, and eicosanoid and docosanoid metabolism. Br. J. Pharmacol. 2012, 166, 1193–1210. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, L.S. Eicosanoids: Prostaglandins, thromboxanes, leukotrienes, and other derivatives of carbon-20 unsaturated fatty acids. J. Neurochem. 1982, 38, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Adam, O.; Beringer, C.; Kless, T.; Lemmen, C.; Adam, A.; Wiseman, M.; Adam, P.; Klimmek, R.; Forth, W. Anti-inflammatory effects of a low arachidonic acid diet and fish oil in patients with rheumatoid arthritis. Rheumatol. Int. 2003, 23, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 fatty acids and inflammatory processes: From molecules to man. Biochem. Soc. Trans. 2017, 45, 1105–1115. [Google Scholar] [CrossRef] [PubMed]
- Dagorn, F.; Dumay, J.; Wielgosz-Collin, G.; Rabesaotra, V.; Viau, M.; Monniot, C.; Biard, J.F.; Barnathan, G. Phospholipid distribution and phospholipid fatty acids of the tropical tunicates Eudistoma sp. and Leptoclinides uniorbis. Lipids 2010, 45, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Mikami, N.; Hosokawa, M.; Miyashita, K. Effects of sea squirt (Halocynthia roretzi) lipids on white adipose tissue weight and blood glucose in diabetic/obese KK-Ay mice. Mol. Med. Rep. 2010, 3, 449–453. [Google Scholar] [CrossRef] [PubMed]
- Rohmah, Z.; Jeong, U.C.; Ticar, B.F.; Kim, J.-S.; Lee, J.-J.; Kang, S.J.; Choi, B.D. Effect of ascidian (Halocynthia roretzi, Drasche 1884) tunics carotenoids on enhancing growth and muscle coloring of sea-reared rainbow trout (Oncorhynchus mykiss, Walbaum 1792). Aquacult. Rep. 2016, 4, 22–29. [Google Scholar] [CrossRef]
- Konishi, I.; Hosokawa, M.; Sashima, T.; Maoka, T.; Miyashita, K. Suppressive effects of alloxanthin and diatoxanthin from Halocynthia roretzi on LPS-induced expression of pro-inflammatory genes in RAW264.7 cells. J. Oleo Sci. 2008, 57, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Nishino, H.; Tsushima, M.; Matsuno, T.; Tanaka, Y.; Okuzumi, J.; Murakoshi, M.; Satomi, Y.; Takayasu, J.; Tokuda, H.; Nishino, A.; et al. Anti-neoplastic effect of halocynthiaxanthin, a metabolite of fucoxanthin. Anticancer Drugs 1992, 3, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Park, M.W.; Lee, C.S.; Kim, S.K.; Kim, W.K. Effects of temperature and salinity on development of sea peach Halocynthia aurantium. J. Environ. Sci. 2009, 18, 1171–1179. [Google Scholar] [CrossRef]
- Lechner, M.; Lirk, P.; Rieder, J. Inducible nitric oxide synthase (iNOS) in tumor biology: The two sides of the same coin. Semin. Cancer Biol. 2005, 15, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Baugh, J.A.; Bucala, R. Mechanisms for modulating TNF alpha in immune and inflammatory disease. Curr. Opin. Drug Discov. Devel. 2001, 4, 635–650. [Google Scholar] [PubMed]
- Popa, C.; Netea, M.G.; van Riel, P.L.; van der Meer, J.W.; Stalenhoef, A.F. The role of TNF-alpha in chronic inflammatory conditions, intermediary metabolism, and cardiovascular risk. J. Lipid Res. 2007, 48, 751–762. [Google Scholar] [CrossRef] [PubMed]
- Meirow, Y.; Baniyash, M. Immune biomarkers for chronic inflammation related complications in non-cancerous and cancerous diseases. Cancer Immunol. Immunother. 2017, 66, 1089–1101. [Google Scholar] [CrossRef] [PubMed]
- Fomenko, S.E.; Kushnerova, N.F.; Lesnikova, L.N. Experimental assessment of the efficiency of erythrocyte membrane repair by an extract of the tunic of the ascidian purple sea squirt in carbon tetrachloride poisoning. Pharm. Chem. J. 2013, 46, 606–611. [Google Scholar] [CrossRef]
- Yang, X.; Haghiac, M.; Glazebrook, P.; Minium, J.; Catalano, P.M.; Hauguel-de Mouzon, S. Saturated fatty acids enhance TLR4 immune pathways in human trophoblasts. Hum. Reprod. 2015, 30, 2152–2159. [Google Scholar] [CrossRef] [PubMed]
- De Pablo Manuel, A.; De Cienfuegos Gerardo, Á. Modulatory effects of dietary lipids on immune system functions. Immunol. Cell Biol. 2000, 78, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Guertin, M.H.; Robitaille, K.; Pelletier, J.F.; Duchesne, T.; Julien, P.; Savard, J.; Bairati, I.; Fradet, V. Effects of concentrated long-chain omega-3 polyunsaturated fatty acid supplementation before radical prostatectomy on prostate cancer proliferation, inflammation, and quality of life: Study protocol for a phase IIb, randomized, double-blind, placebo-controlled trial. BMC Cancer 2018, 18, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Lee, H.J.; Kang, S.B.; Park, W.J. Fatty acid desaturases, polyunsaturated fatty acid regulation, and biotechnological advances. Nutrients 2016, 8, 23. [Google Scholar] [CrossRef] [PubMed]
- Olefsky, J.M.; Glass, C.K. Macrophages, inflammation, and insulin resistance. Annu. Rev. Physiol. 2010, 72, 219–246. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.S.; Bradshaw, H.B.; Chen, J.S.; Tan, B.; Walker, J.M. Prostaglandin E2 glycerol ester, an endogenous COX-2 metabolite of 2-arachidonoylglycerol, induces hyperalgesia and modulates NFκB activity. Br. J. Pharmacol. 2008, 153, 1538–1549. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Liu, J.; Liu, C.; Zeng, X.; Li, X.; Zhao, J. Anti-inflammatory effects of p-coumaric acid in LPS-stimulated RAW264.7 cells: Involvement of NF-κB and MAPKs pathways. J. Med. Chem. 2016, 6, 327–330. [Google Scholar] [CrossRef]
- Tak, P.P.; Firestein, G.S. NF-κB: A key role in inflammatory diseases. J. Clin. Investig. 2001, 107, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Baeuerle, P.A.; Baltimore, D. NF-κB: Ten years after. Cell 1996, 87, 13–20. [Google Scholar] [CrossRef]
- Kim, J.B.; Han, A.R.; Park, E.Y.; Kim, J.Y.; Cho, W.; Lee, J.; Seo, E.K.; Lee, K.T. Inhibition of LPS-induced iNOS, COX-2 and cytokines expression by poncirin through the NF-kappaB inactivation in RAW 264.7 macrophage cells. Biol. Pharm. Bull. 2007, 30, 2345–2351. [Google Scholar] [CrossRef] [PubMed]
- Cargnello, M.; Roux, P.P. Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol. Mol. Biol. Rev. 2011, 75, 50–83. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.Y.; Hsieh, L.L.; Tang, R.P.; Liao, S.K.; Yeh, K.Y. Interleukin-6 (IL-6) released by macrophages induces IL-6 secretion in the human colon cancer HT-29 cell line. Hum. Immunol. 2009, 70, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Han, L.; Song, S.; Niu, Y.; Meng, M.; Wang, C. Eicosapentaenoic acid (EPA) induced macrophages activation through GPR120-mediated Raf-ERK1/2-IKKβ-NF-κB p65 signaling pathways. Nutrients 2017, 9, 937. [Google Scholar] [CrossRef] [PubMed]
- Novak, T.E.; Babcock, T.A.; Jho, D.H.; Helton, W.S.; Espat, N.J. NF-κB inhibition by omega-3 fatty acids modulates LPS-stimulated macrophage TNF-alpha transcription. Am. J. Physiol. 2003, 284, L84–L89. [Google Scholar] [CrossRef]
- Zhao, Y.; Joshi-Barve, S.; Barve, S.; Chen, L.H. Eicosapentaenoic acid prevents LPS-induced TNF-α expression by preventing NF-κB activation. J. Am. Coll. Nutr. 2004, 23, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Garces, R.; Mancha, M. One-step lipid extraction and fatty acid methyl esters preparation from fresh plant tissues. Anal. Biochem. 1993, 211, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Park, W.J.; Kothapalli, K.S.; Lawrence, P.; Tyburczy, C.; Brenna, J.T. An alternate pathway to long-chain polyunsaturates: The FADS2 gene product Δ8-desaturates 20:2n-6 and 20:3n-3. J. Lipid Res. 2009, 50, 1195–1202. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Cho, M.L.; Karnjanapratum, S.; Shin, I.S.; You, S.G. In vitro and in vivo immunomodulatory activity of sulfated polysaccharides from Enteromorpha prolifera. Int. J. Biol. Macromol. 2011, 49, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Cao, R.A.; Lee, Y.; You, S.G. Water soluble sulfated-fucans with immune-enhancing properties from Ecklonia cava. Int. J. Biol. Macromol. 2014, 67, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, B.A.; Narayanan, N.K.; Simi, B.; Reddy, B.S. Modulation of inducible nitric oxide synthase and related proinflammatory genes by the omega-3 fatty acid docosahexaenoic acid in human colon cancer cells. Cancer Res. 2003, 63, 972–979. [Google Scholar] [PubMed]
| Gene | Accession No. | Sequence | Product Size (bp) |
|---|---|---|---|
| iNOS | BC062378.1 | F: TTCCAGAATCCCTGGACAAG R: TGGTCAAACTCTTGGGGTTC | 180 |
| IL-1β | NM_008361.4 | F: GGGCCTCAAAGGAAAGAATC R: TACCAGTTGGGGAACTCTGC | 183 |
| IL-6 | NM_031168.2 | F: AGTTGCCTTCTTGGGACTGA R: CAGAATTGCCATTGCACAAC | 191 |
| COX-2 | NM_011198.4 | F: AGAAGGAAATGGCTGCAGAA R: GCTCGGCTTCCAGTATTGAG | 194 |
| TNF-α | D84199.2 | F: ATGAGCACAGAAAGCATGATC R: TACAGGCTTGTCACTCGAATT | 276 |
| β-Actin | NM_007393.5 | F: CCACAGCTGAGAGGAAATC R: AAGGAAGGCTGGAAAAGAGC | 193 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Monmai, C.; Go, S.H.; Shin, I.-S.; You, S.G.; Lee, H.; Kang, S.B.; Park, W.J. Immune-Enhancement and Anti-Inflammatory Activities of Fatty Acids Extracted from Halocynthia aurantium Tunic in RAW264.7 Cells. Mar. Drugs 2018, 16, 309. https://doi.org/10.3390/md16090309
Monmai C, Go SH, Shin I-S, You SG, Lee H, Kang SB, Park WJ. Immune-Enhancement and Anti-Inflammatory Activities of Fatty Acids Extracted from Halocynthia aurantium Tunic in RAW264.7 Cells. Marine Drugs. 2018; 16(9):309. https://doi.org/10.3390/md16090309
Chicago/Turabian StyleMonmai, Chaiwat, Seok Hyeon Go, II-Shik Shin, Sang Guan You, Hyungjae Lee, Seok Beom Kang, and Woo Jung Park. 2018. "Immune-Enhancement and Anti-Inflammatory Activities of Fatty Acids Extracted from Halocynthia aurantium Tunic in RAW264.7 Cells" Marine Drugs 16, no. 9: 309. https://doi.org/10.3390/md16090309
APA StyleMonmai, C., Go, S. H., Shin, I.-S., You, S. G., Lee, H., Kang, S. B., & Park, W. J. (2018). Immune-Enhancement and Anti-Inflammatory Activities of Fatty Acids Extracted from Halocynthia aurantium Tunic in RAW264.7 Cells. Marine Drugs, 16(9), 309. https://doi.org/10.3390/md16090309






