Linking Heart Function to Prognosis: The Role of a Novel Echocardiographic Index and NT-proBNP in Acute Heart Failure
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Patient Population
2.2. Data Collection
2.3. Echocardiographic Assessment
2.4. Definition of the Echocardiographic Index
2.5. Statistical Analysis
3. Results
3.1. Baseline Characteristics
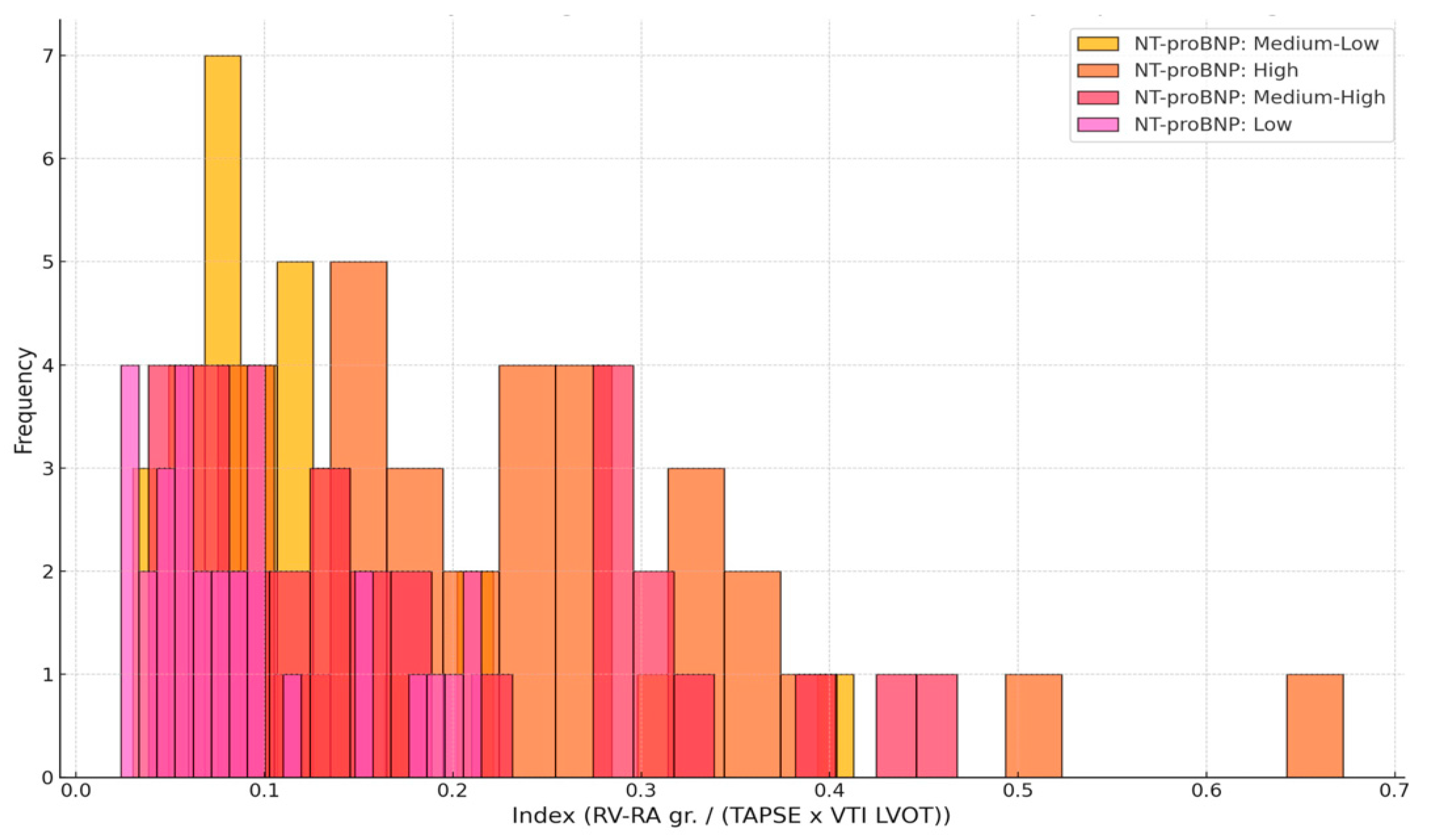
3.2. Relationship Between the Echocardiographic Index and NT-proBNP
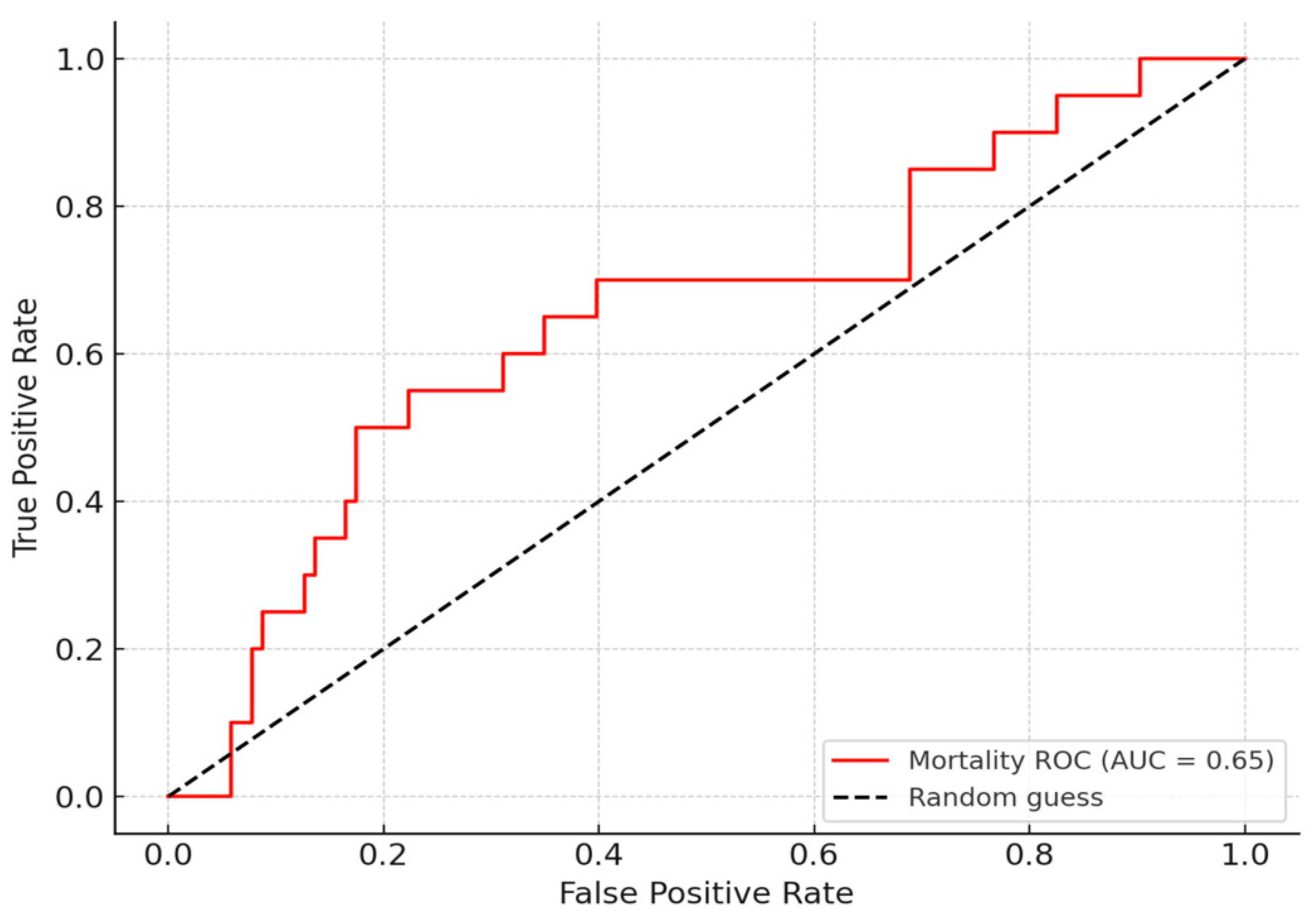
3.3. Predictive Accuracy of the Index for Elevated NT-proBNP
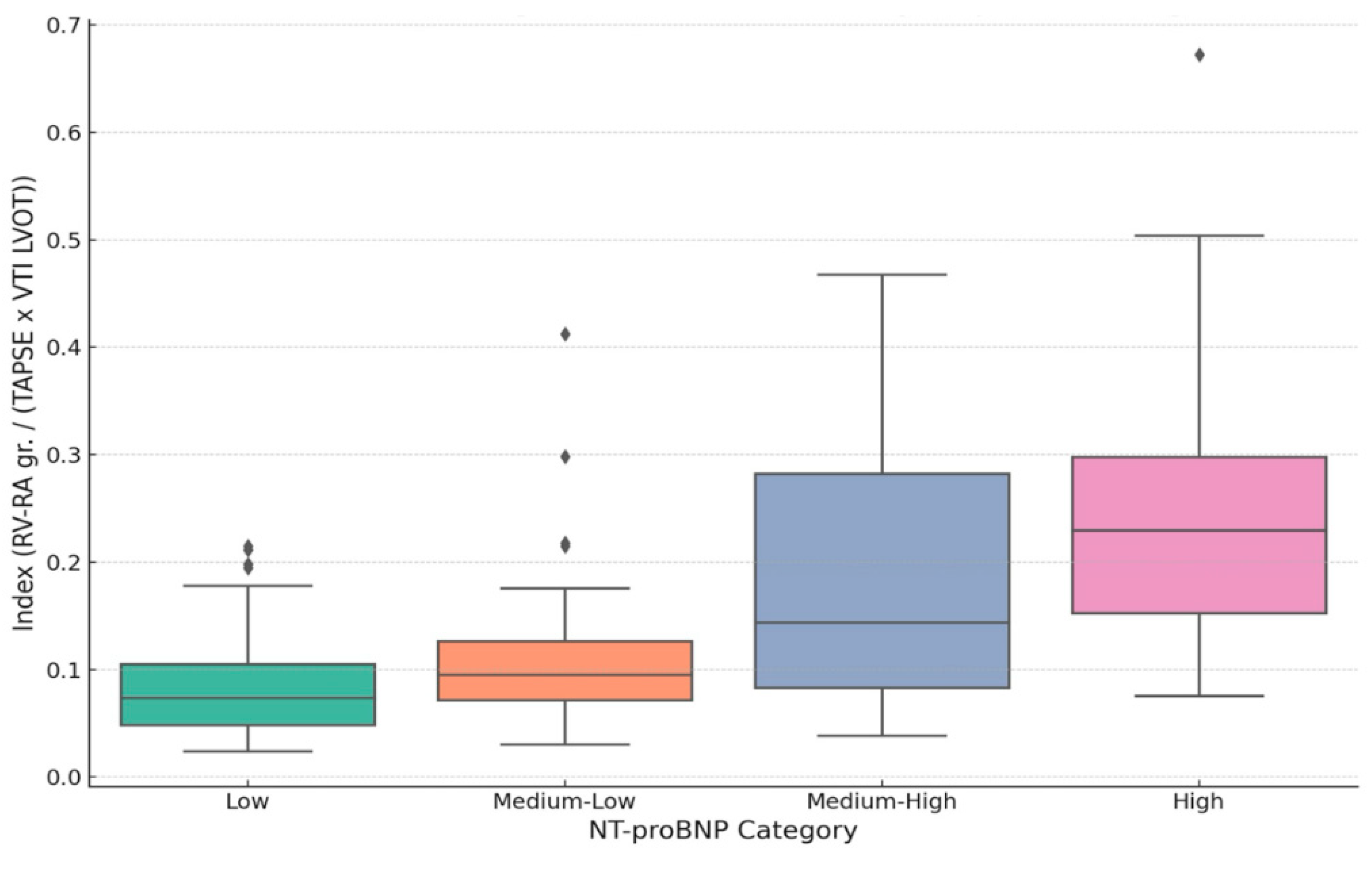
3.4. Index Distribution by NT-proBNP Category
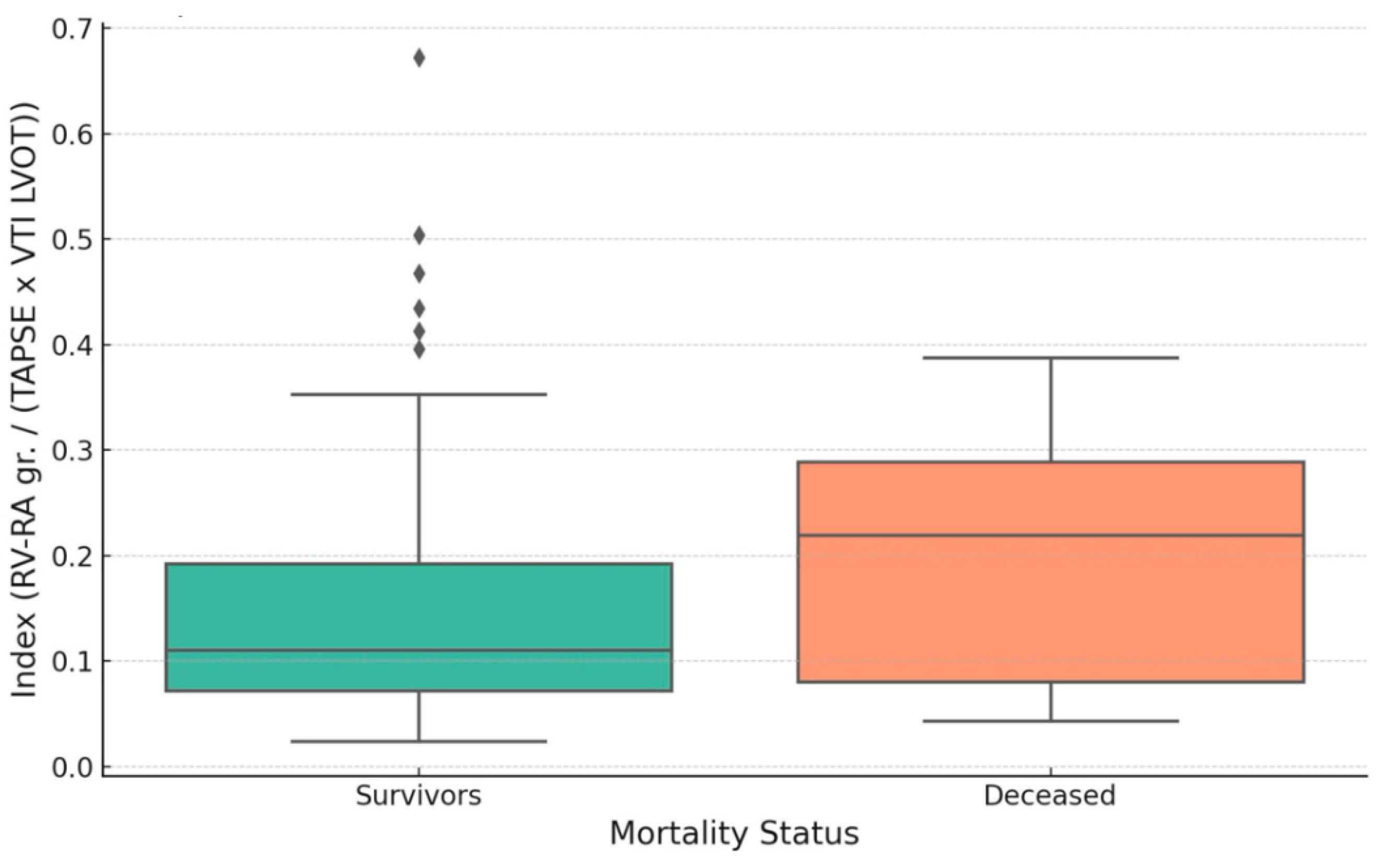
3.5. Correlation with In-Hospital Mortality
3.6. Multivariable Analysis of Mortality Predictors
4. Discussion
4.1. Pathophysiological Interpretation
4.2. Clinical Relevance
4.3. Comparison with the Existing Literature
4.4. Strengths and Limitations
4.5. Future Perspectives
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AHF | Acute Heart Failure |
| ALT | Alanine Aminotransferase |
| AST | Aspartate Aminotransferase |
| AUC | Area Under the Curve |
| BP | Blood Pressure |
| EF | Ejection Fraction |
| FAC | Fractional Area Change |
| GLS | Global Longitudinal Strain |
| HF | Heart Failure |
| INR | International Normalized Ratio |
| IQR | Interquartile Range |
| IVT | Time-Velocity Integral |
| JASP | Jeffrey’s Amazing Statistics Program |
| LV | Left Ventricle |
| LVEF | Left Ventricular Ejection Fraction |
| LVOT | Left Ventricular Outflow Tract |
| NT-proBNP | N-terminal pro–B-type Natriuretic Peptide |
| PASP | Pulmonary Artery Systolic Pressure |
| ROC | Receiver Operating Characteristic |
| RV | Right Ventricle |
| RV-GLS | Right Ventricular Global Longitudinal Strain |
| RV-RA | Right Ventricular–Right Atrial |
| RVGLS/PASP | Ratio of RVGLS to PASP |
| SD | Standard Deviation |
| SPSS | Statistical Package for the Social Sciences |
| TAPSE | Tricuspid Annular Plane Systolic Excursion |
| TAPSE/PASP | Ratio of TAPSE to PASP |
| VTI | Velocity–Time Integral |
References
- Stewart, S.; Jenkins, A.; Buchan, S.; McGuire, A.; Capewell, S.; McMurray, J.J. The current cost of heart failure to the National Health Service in the UK. Eur. J. Heart Fail. 2002, 4, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; de Ferranit, S.; Després, J.-P.; Fullerton, H.J.; Howard, V.J.; et al. Heart disease and stroke statistics–2015 update: A report from the American Heart Association. Circulation 2015, 131, e29–e322. [Google Scholar] [CrossRef]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.F.; Coats, A.J.S.; Falk, V.; González-Juanatey, G.R.; Harjola, V.-P.; Jankowska, E.A.; et al. 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force of the European Society of Cardiology. Eur. Heart J. 2016, 37, 2129–2200. [Google Scholar] [CrossRef] [PubMed]
- Yancy, C.W.; Jessup, M.; Bozkurt, B.; Butler, J.; Casey, D.E., Jr.; Drazner, M.H.; Fonarow, G.C.; Geraci, S.A.; Horwich, T.; Januzzi, J.L.; et al. ACCF/AHA guideline for the management of heart failure: A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 2013, 62, e147–e239. [Google Scholar] [CrossRef] [PubMed]
- Yancy, C.W.; Jessup, M.; Bozkurt, B.; Butler, J.; Casey, D.E., Jr.; Colvin, M.M.; Drazner, M.H.; Filippatos, G.S.; Fonarow, G.C.; Givertz, M.M.; et al. 2017 ACC/AHA/HFSA focused update of the 2013 ACCF/AHA guideline for the management of heart failure: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. Circulation 2017, 136, e137–e161. [Google Scholar] [CrossRef] [PubMed]
- Papadimitriou, L.; Georgiopoulou, V.V.; Kort, S.; Butler, J.; Kalogeropoulos, A.P. Echocardiography in acute heart failure: Current perspectives. J. Card. Fail. 2016, 22, 82–94. [Google Scholar] [CrossRef]
- Dini, F.L.; Cameli, M.; Stefanini, A.; Aboumarie, H.S.; Lisi, M.; Lindqvist, P.; Henein, M.Y. Echocardiography in the assessment of heart failure patients. Diagnostics 2024, 14, 2730. [Google Scholar] [CrossRef]
- Park, J.J.; Park, J.B.; Park, J.H.; Cho, G.Y. Global longitudinal strain to predict mortality in patients with acute heart failure. J. Am. Coll. Cardiol. 2018, 71, 1947–1957. [Google Scholar] [CrossRef]
- Yingchoncharoen, T.; Agarwal, S.; Popović, Z.B.; Marwick, T.H. Normal ranges of left ventricular strain: A meta-analysis. J. Am. Soc. Echocardiogr. 2013, 26, 185–191. [Google Scholar] [CrossRef]
- Tan, C.; Rubenson, D.; Srivastava, A.; Mohan, R.; Smith, M.R.; Billick, K.; Bardarian, S.; Heywood, J.G. Left ventricular outflow tract velocity time integral outperforms ejection fraction and Doppler-derived cardiac output for predicting outcomes in a select advanced heart failure cohort. Cardiovasc. Ultrasound 2017, 15, 18. [Google Scholar] [CrossRef]
- Omote, K.; Nagai, T.; Iwano, H.; Tsujinaga, S.; Kamiya, K.; Aikawa, T.; Konishi, T.; Sato, T.; Kato, Y.; Komoriyama, H.; et al. Left ventricular outflow tract velocity time integral in hospitalized heart failure with preserved ejection fraction. ESC Heart Fail. 2020, 7, 167–175. [Google Scholar] [CrossRef]
- Gentile, F.; Buoncristiani, F.; Sciarrone, P.; Bazan, L.; Panichella, G.; Gasparini, S.; Chubuchny, V.; Taddei, C.; Poggianti, E.; Fabiani, I.; et al. Left ventricular outflow tract velocity-time integral improves outcome prediction in patients with secondary mitral regurgitation. Int. J. Cardiol. 2023, 392, 131272. [Google Scholar] [CrossRef] [PubMed]
- Harjola, V.P.; Mebazaa, A.; Čelutkienė, J.; Bettex, D.; Bueno, H.; Chioncel, O.; Mara, G.; Leiro, C.; Falk, V.; Filippatos, G.; et al. Contemporary management of acute right ventricular failure: A statement from the Heart Failure Association and the Working Group on Pulmonary Circulation and Right Ventricular Function of the European Society of Cardiology. Eur. J. Heart Fail. 2016, 18, 226–241. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, C.; Rahko, P.S.; Blauwet, L.A.; Canaday, B.; Finstuen, J.A.; Foster, M.C.; Horton, K.; Ogunyankin, K.O.; Palma, R.A.; Velazquez, E.J. Guidelines for performing a comprehensive transthoracic echocardiographic examination in adults: Recommendations from the American Society of Echocardiography. J. Am. Soc. Echocardiogr. 2019, 32, 1–64. [Google Scholar] [CrossRef] [PubMed]
- Ghio, S.; Gavazzi, A.; Campana, C.; Inserra, C.; Klersy, C.; Sebastiani, R.; Arbustini, E.; Recusani, F.; Tavazzi, L. Independent and additive prognostic value of right ventricular systolic function and pulmonary artery pressure in patients with chronic heart failure. J. Am. Coll. Cardiol. 2001, 37, 183–188. [Google Scholar] [CrossRef]
- Guazzi, M.; Bandera, F.; Pelissero, G.; Castelvecchio, S.; Menicanti, L.; Ghio, S.; Temporelli, P.L.; Arena, R. Tricuspid annular plane systolic excursion and pulmonary arterial systolic pressure relationship in heart failure: An index of right ventricular contractile function and prognosis. Am. J. Physiol. Heart Circ. Physiol. 2013, 305, H1373–H1381. [Google Scholar] [CrossRef]
- Anastasiou, V.; Papazoglou, A.S.; Moysidis, D.V.; Daios, S.; Barmpagiannos, K.; Gossios, T.; Efthimiadis, G.K.; Karamitsos, T.; Ziakas, A.; Kamperidis, V. The prognostic impact of right ventricular–pulmonary arterial coupling in heart failure: A systematic review and meta-analysis. Heart Fail. Rev. 2024, 29, 13–26. [Google Scholar] [CrossRef]
- Naseem, M.; Alkassas, A.; Alaarag, A. Tricuspid annular plane systolic excursion/pulmonary arterial systolic pressure ratio as a predictor of in-hospital mortality for acute heart failure. BMC Cardiovasc. Disord. 2022, 22, 414. [Google Scholar] [CrossRef]
- Chen, J.S.; Pei, Y.; Li, C.E.; Li, N.Y.; Guo, T.; Yu, J. Prognostic value of heart failure echocardiography index in HF patients with preserved, mid-ranged and reduced ejection fraction. BMC Cardiovasc. Disord. 2020, 20, 351. [Google Scholar] [CrossRef]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef]
- Kang, S.H.; Park, J.J.; Choi, D.J.; Yoon, C.H.; Oh, I.Y.; Kang, S.M.; Yoo, B.S.; Jeon, E.S.; Kim, G.G.; Cho, M.C.; et al. Prognostic value of NT-proBNP in heart failure with preserved versus reduced EF. Heart 2015, 101, 1881–1888. [Google Scholar] [CrossRef]
- Januzzi, J.L.; van Kimmenade, R.; Lainchbury, J.; Bayes-Genis, A.; Ordoñez-Llanos, J.; Santalo-Bel, M.; Pinto, Y.M.; Richards, M. NT-proBNP testing for diagnosis and short-term prognosis in acute destabilized heart failure: An international pooled analysis of 1256 patients: The International Collaborative of NT-proBNP Study. Eur. Heart. J. 2006, 27, 330–337. [Google Scholar] [CrossRef]
- Richards, A.M. Biomarkers in acute heart failure—cardiac and kidney. Card. Fail. Rev. 2015, 1, 107–111. [Google Scholar] [CrossRef]
- Park, J.H.; Kim, M.; Park, J.J.; Park, J.B.; Cho, G.Y. Prognostic role of RVGLS/PASP ratio, a new echocardiographic parameter of the right ventricle–pulmonary artery coupling, in patients with acute heart failure. Int. J. Heart Fail. 2024, 6, 165–173. [Google Scholar] [CrossRef]
- Bok, Y.; Kim, J.Y.; Park, J.H. Prognostic role of right ventricular–pulmonary artery coupling assessed by TAPSE/PASP ratio in patients with acute heart failure. J. Cardiovasc. Imaging 2023, 31, 200–206. [Google Scholar] [CrossRef]
- Chen, A.A.; Wood, M.J.; Krauser, D.G.; Baggish, A.L.; Tung, R.; Anwaruddin, S.; Picard, M.H.; Januzzi, J.L. NT-proBNP levels, echocardiographic findings, and outcomes in breathless patients: Results from the ProBNP Investigation of Dyspnoea in the Emergency Department (PRIDE) echocardiographic substudy. Eur. Heart J. 2006, 27, 839–845. [Google Scholar] [CrossRef] [PubMed]


| Variable | Total (N = 123) | Survivors (N = 103) | Non-Survivors (N = 20) | p-Value |
|---|---|---|---|---|
| Age (years) | 71.34 ± 13.98 | 70.3 ± 14.34 | 76.4 ± 10.85 | 0.039 |
| Male sex, % | 50.4 | 50.4 | 50.0 | 0.968 |
| Smoking, % | 22.76 | 25.24 | 10.0 | 0.137 |
| Hypertension, % | 86.99 | 88.34 | 80.0 | 0.310 |
| Dyslipidemia, % | 91.86 | 95.14 | 75.0 | 0.003 |
| Obesity, % | 39.83 | 35.92 | 60.0 | 0.044 |
| Diabetes mellitus, % | 39.83 | 38.83 | 45.0 | 0.606 |
| Systolic BP (mmHg) | 142.59 ± 29.54 | 144.33 ± 27.43 | 133.6 ± 38.23 | 0.244 |
| Diastolic BP (mmHg) | 85.83 ± 16.68 | 87.84 ± 15.75 | 75.45 ± 17.91 | 0.002 |
| Heart rate (bpm) | 100.98 ± 28.12 | 101.5 ± 29.18 | 98.3 ± 22.32 | 0.644 |
| NT-proBNP (pg/mL) | 9228.3 ± 9736.4 | 7569.6 ± 8565.9 | 17,770.8 ± 11,093.4 | 0.001 |
| TAPSE (mm) | 18.32 ± 5.16 | 18.53 ± 5.09 | 17.25 ± 5.49 | 0.309 |
| VTI LVOT (cm) | 15.5 ± 5.22 | 15.97 ± 5.30 | 13.07 ± 4.06 | 0.022 |
| RV-RA gradient (mmHg) | 33.64 ± 12.14 | 33.09 ± 12.05 | 37.0 ± 12.44 | 0.190 |
| LVEF (%) | 41.21 ± 16.78 | 41.15 ± 16.58 | 41.5 ± 18.23 | 0.934 |
| Valvular heart disease, % | 73.17 | 71.84 | 80.0 | 0.529 |
| Variable | Odds Ratio | p-Value | CI 95% |
|---|---|---|---|
| VTI LVOT | 0.86 | 0.002 | 0.775 to 0.943 |
| RV-RA gradient | 1.04 | 0.037 | 1.002 to 1.080 |
| TAPSE | 0.90 | 0.034 | 0.812 to 0.991 |
| OR | OR 95% CI Lower | OR 95% CI Upper | p-Value | |
|---|---|---|---|---|
| Index (GRVDAD/TAPSE x IVTLVOT) | 1.121555 | 0.010058 | 125.0652 | 0.961959 |
| Age | 1.019246 | 0.968348 | 1.072819 | 0.465779 |
| eGFR at Admission | 1.010532 | 0.981655 | 1.040259 | 0.478765 |
| LVEF | 25.99523 | 0.557571 | 1211.957 | 0.09652 |
| log(NT-proBNP) | 4.719458 | 1.989161 | 11.19732 | 0.000431 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Published by MDPI on behalf of the Lithuanian University of Health Sciences. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Popescu, D.-C.; Ciobanu, M.; Țînț, D.; Nechita, A.-C. Linking Heart Function to Prognosis: The Role of a Novel Echocardiographic Index and NT-proBNP in Acute Heart Failure. Medicina 2025, 61, 1412. https://doi.org/10.3390/medicina61081412
Popescu D-C, Ciobanu M, Țînț D, Nechita A-C. Linking Heart Function to Prognosis: The Role of a Novel Echocardiographic Index and NT-proBNP in Acute Heart Failure. Medicina. 2025; 61(8):1412. https://doi.org/10.3390/medicina61081412
Chicago/Turabian StylePopescu, Dan-Cristian, Mara Ciobanu, Diana Țînț, and Alexandru-Cristian Nechita. 2025. "Linking Heart Function to Prognosis: The Role of a Novel Echocardiographic Index and NT-proBNP in Acute Heart Failure" Medicina 61, no. 8: 1412. https://doi.org/10.3390/medicina61081412
APA StylePopescu, D.-C., Ciobanu, M., Țînț, D., & Nechita, A.-C. (2025). Linking Heart Function to Prognosis: The Role of a Novel Echocardiographic Index and NT-proBNP in Acute Heart Failure. Medicina, 61(8), 1412. https://doi.org/10.3390/medicina61081412







