Antimicrobial Photodynamic Therapy in Combination with Nystatin in the Treatment of Experimental Oral Candidiasis Induced by Candida albicans Resistant to Fluconazole
Abstract
1. Introduction
2. Results
2.1. Microbiological Evaluation
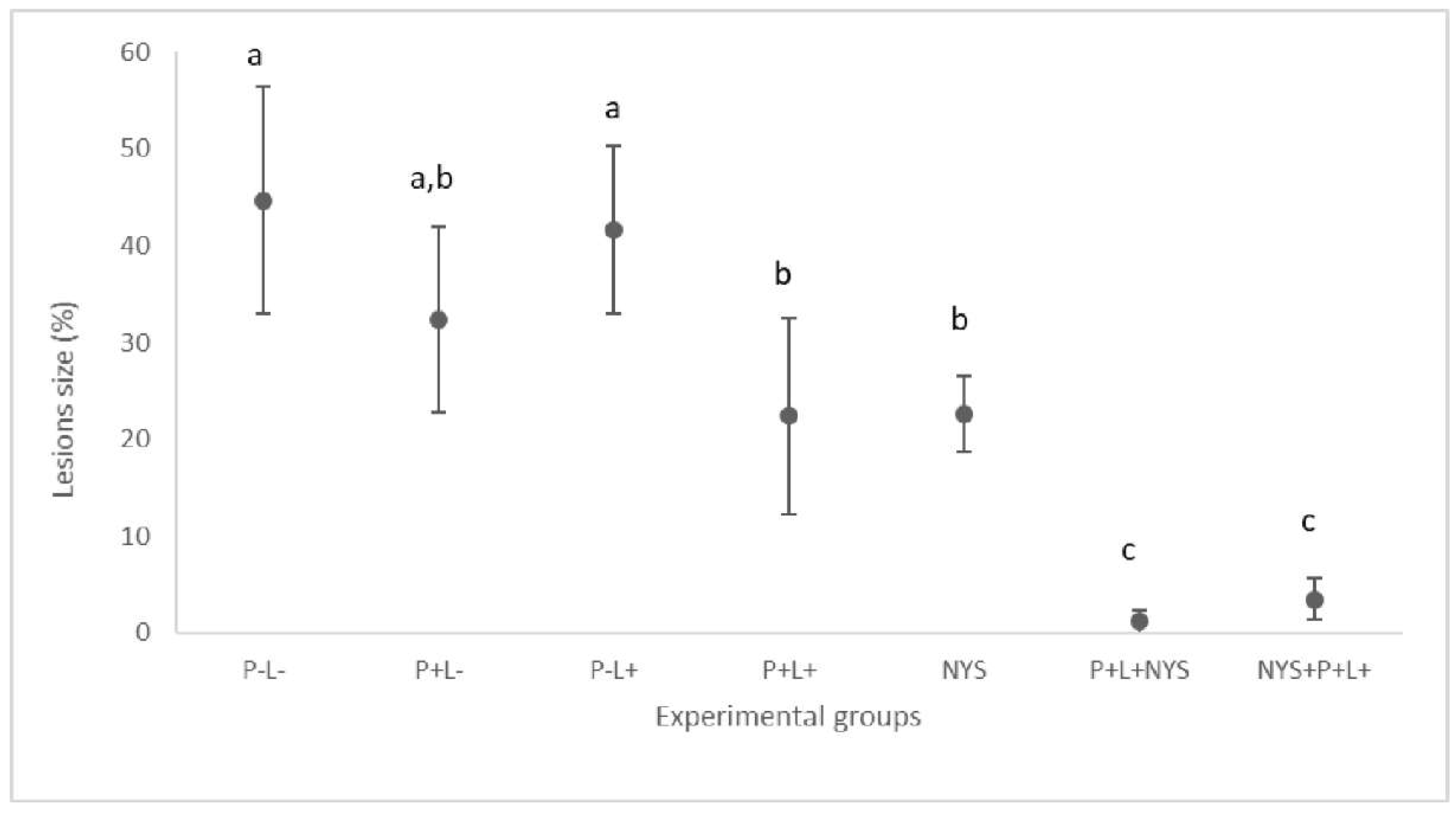
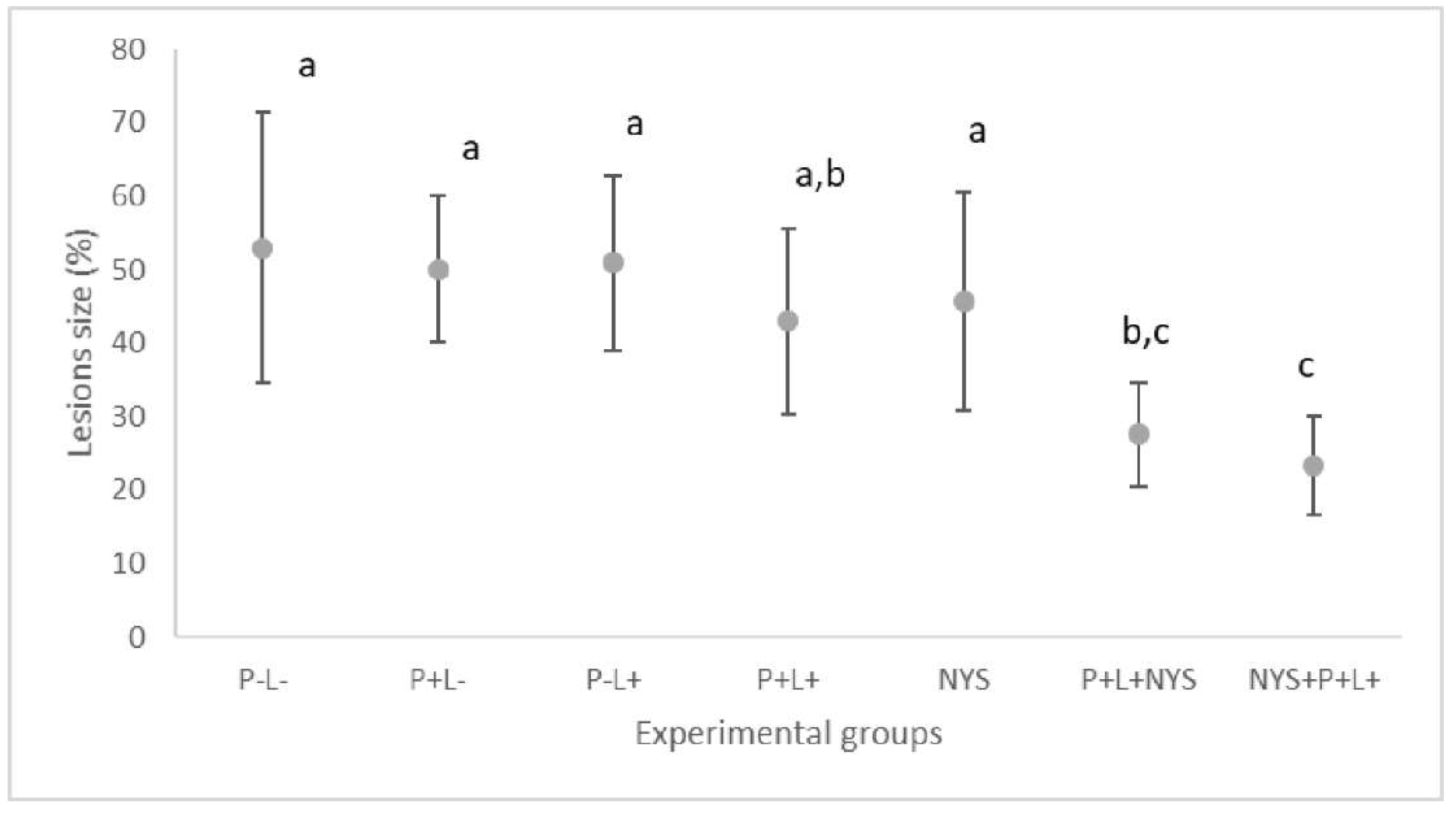
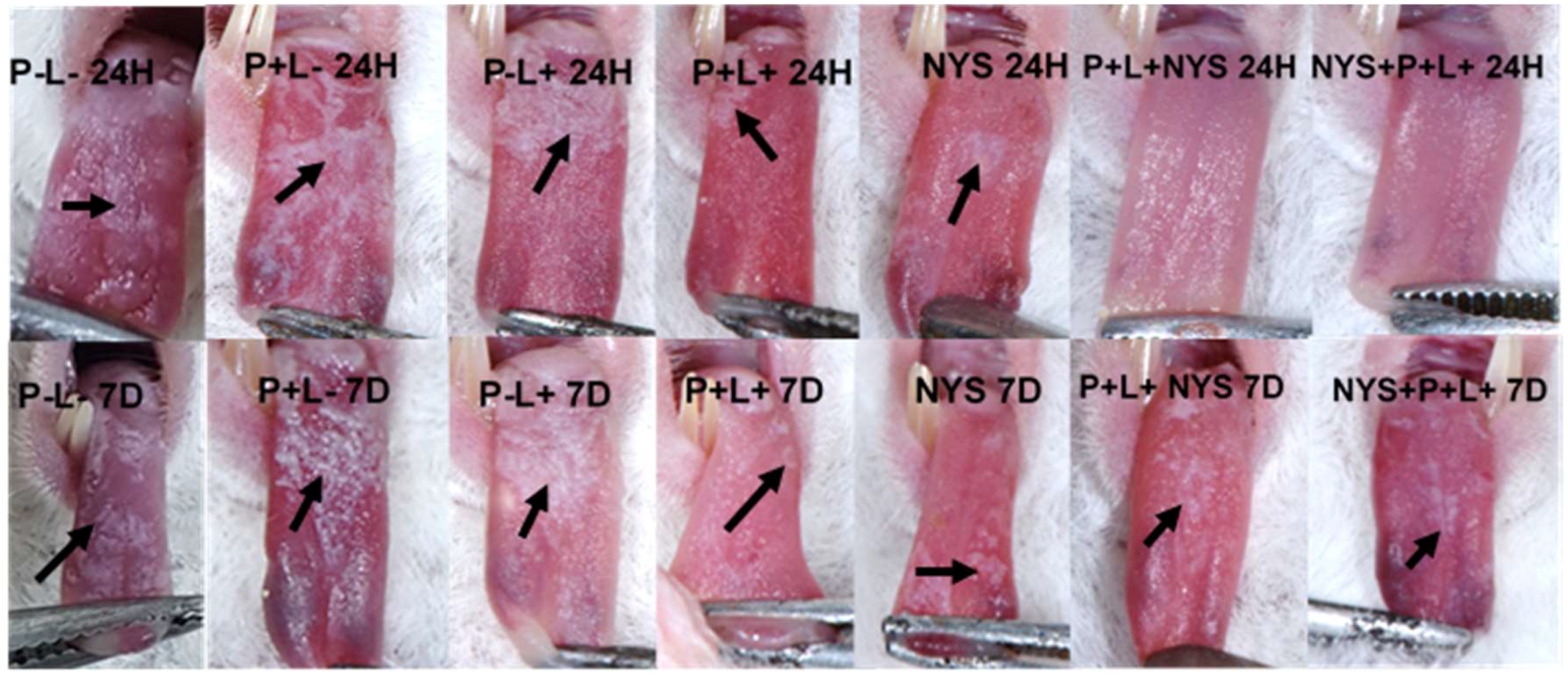
2.2. Macroscopic Evaluation of Lesions
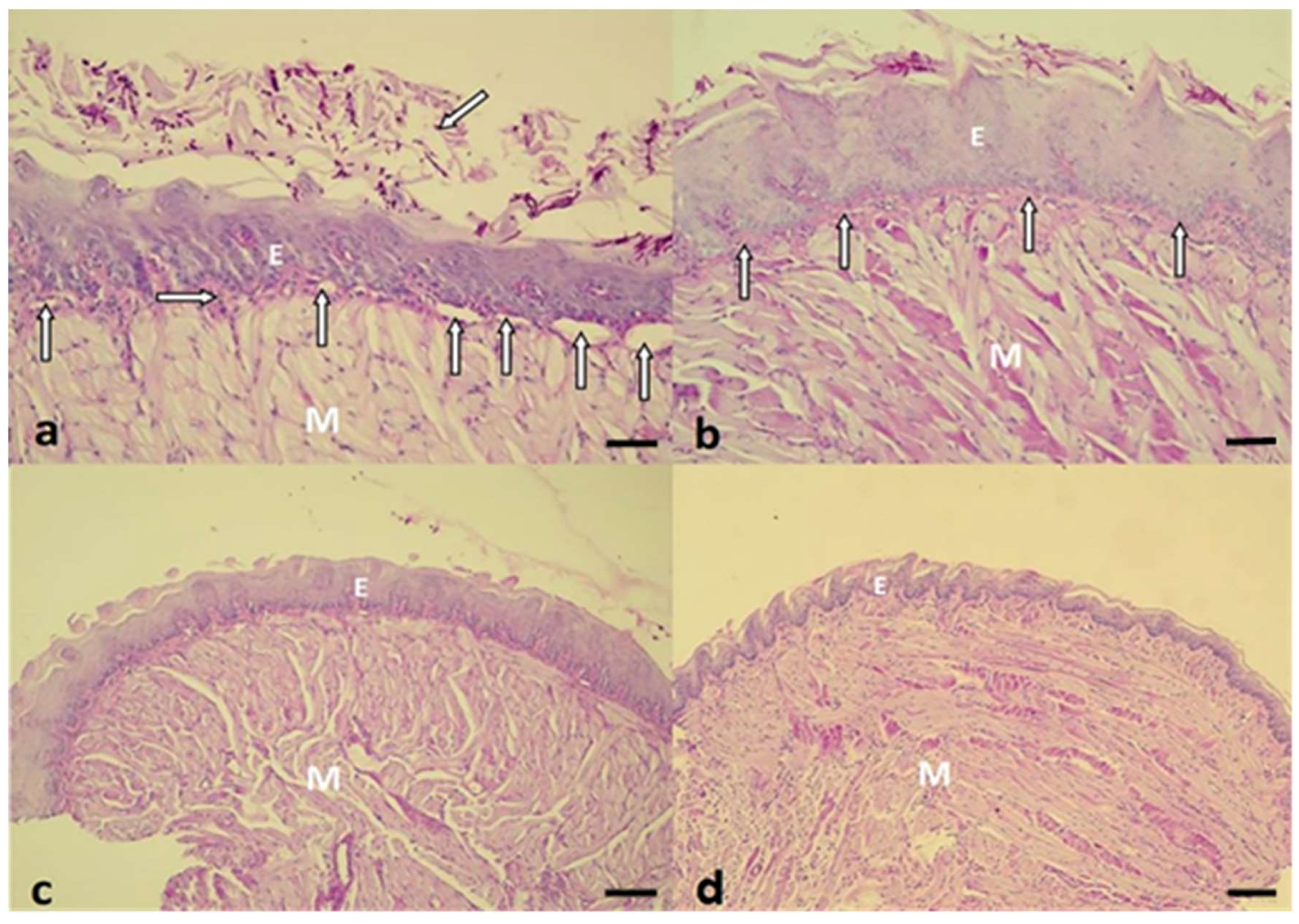
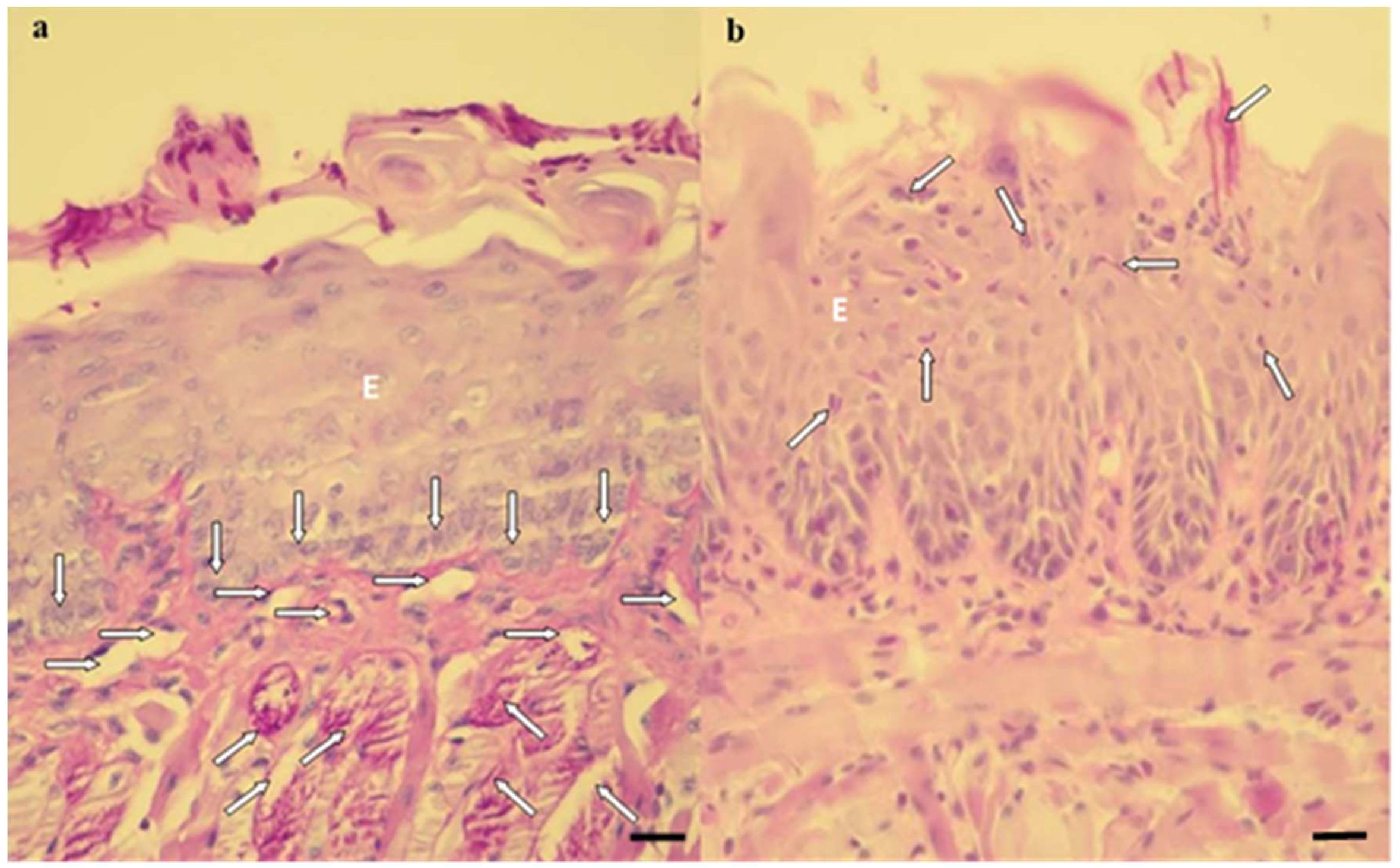
2.3. Histopathological Evaluation
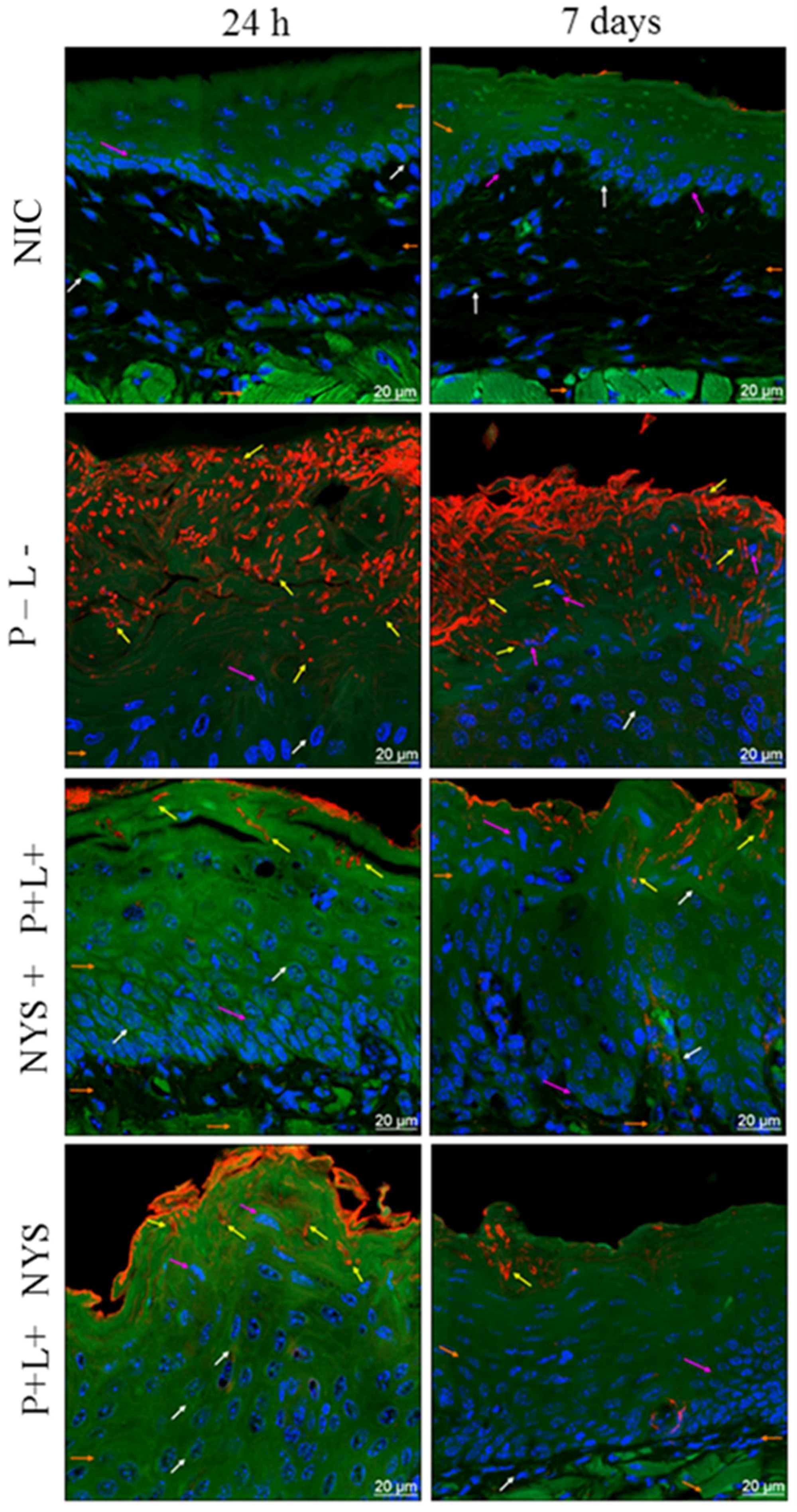
2.4. Confocal Scanning Laser Microscopy (CSLM) Evaluation
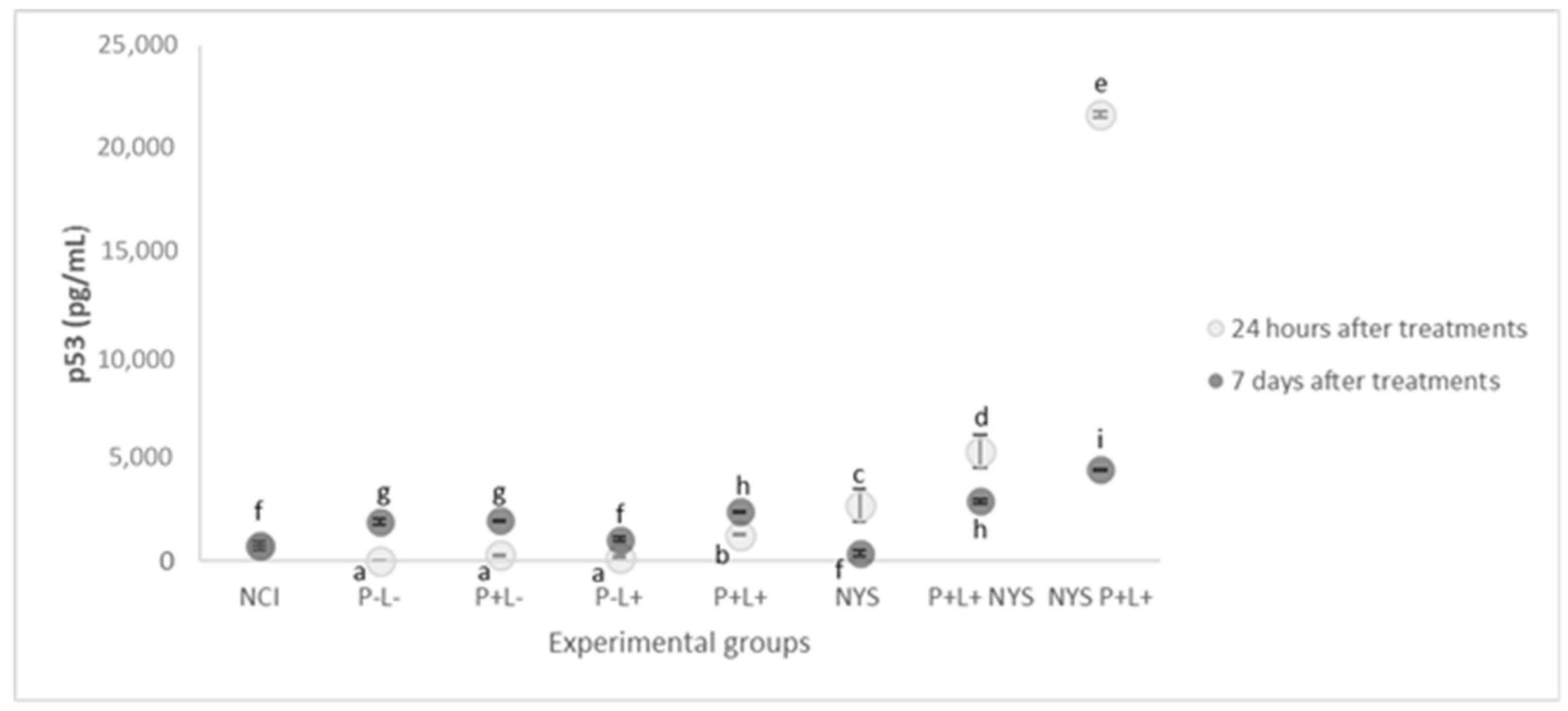
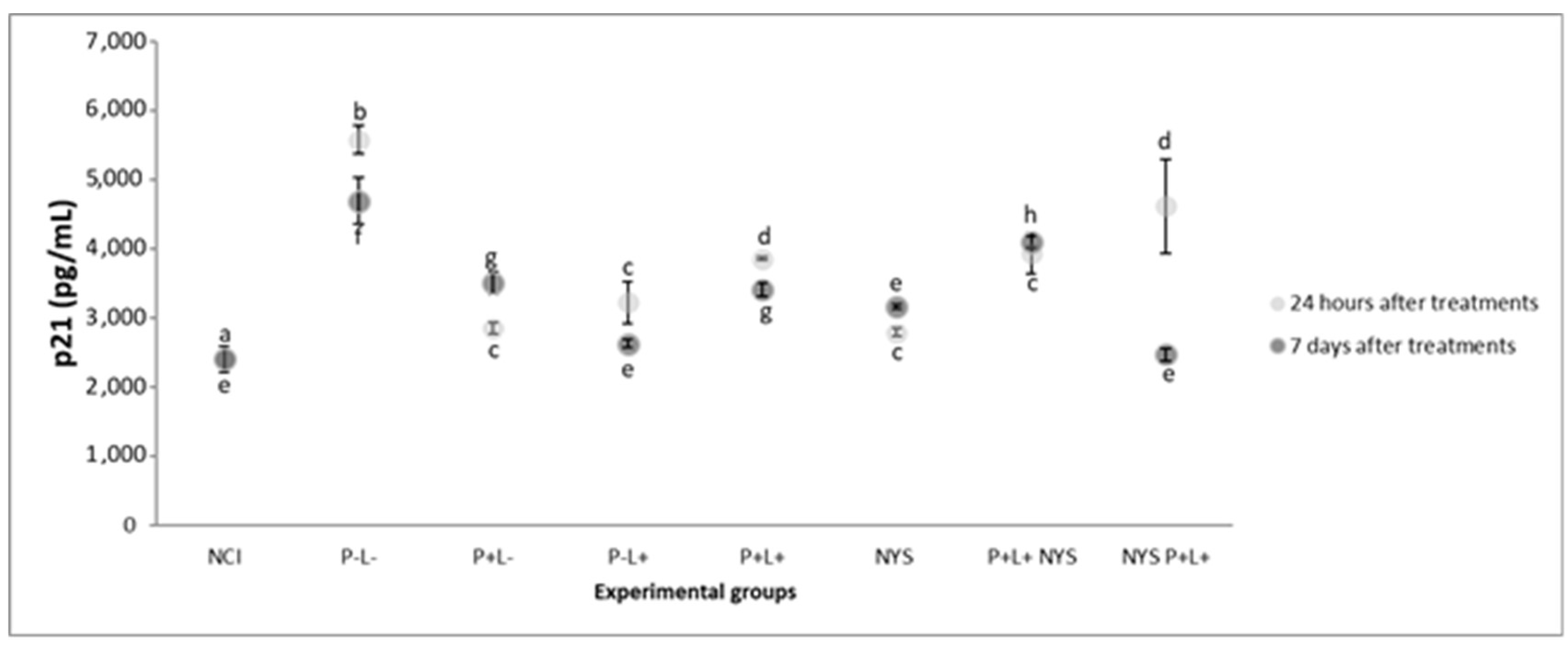
2.5. Expression of p53 and p21
3. Discussion
4. Materials and Methods
4.1. Photossensitizer, LED Device
4.2. Treatments Performed
4.3. Treatment Assessment
4.4. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Martin, R.; Wächtler, B.; Schaller, M.; Wilson, D.; Hube, B. Host-pathogen interactions and virulence-associated genes during Candida albicans oral infections. Int. J. Med. Microbiol. 2011, 301, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Gulati, M.; Nobile, C.J. Candida albicans biofilms: Development, regulation, and molecular mechanisms. Microbes Infect. 2016, 18, 310–321. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.J.P.; Brown, G.D.; Netea, M.G.; Gow, N.A.R. Metabolism impacts upon Candida immunogenicity and pathogenicity at multiple levels. Trends Microbiol. 2014, 22, 614–622. [Google Scholar] [CrossRef] [PubMed]
- Sardi, J.C.O.; Scorzoni, L.; Bernardi, T.; Fusco-Almeida, A.M.; Giannini, M.J.S.M. Candida species: Current epidemiology, pathogenicity, biofilm formation, natural antifungal products and new therapeutic options. J. Med. Microbiol. 2013, 62, 10–24. [Google Scholar] [CrossRef]
- Arendrup, M.C.; Patterson, T.F. Multidrug-resistant Candida: Epidemiology, molecular mechanisms, and treatment. J. Infect. Dis. 2017, 216, S445–S451. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, S.G.; Aalders, M.C.G.; van Marle, J. Mechanistic study of the photodynamic inactivation of Candida albicans by a cationic porphyrin. Antimicrob. Agents Chemother. 2005, 49, 2026–2034. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, R.F.; McCarron, P.A.; Tunney, M.M. Antifungal photodynamic therapy. Microbiol. Res. 2008, 163, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Bonnett, R.; Martínez, G. Photobleaching of sensitizers used in photodynamic therapy. Tetrahedron 2001, 57, 9513–9547. [Google Scholar] [CrossRef]
- De Melo, W.C.M.A.; Avci, P.; de Oliveira, M.N.; Gupta, A.; Vecchio, D.; Sadasivam, M.; Chandran, R.; Huang, Y.Y.; Yin, R.; Perussi, L.R.; et al. Photodynamic inactivation of biofilm: Taking a lightly colored approach to stubborn infection. Expert Rev. Anti Infect. Ther. 2013, 11, 669–693. [Google Scholar] [CrossRef]
- Huang, L.; Xuan, Y.; Koide, Y. Type I and Type II mechanisms of antimicrobial photodynamic therapy: An in vitro study on gram-negative and gram-positive bacteria. Lasers Surg. Med. 2012, 44, 490–499. [Google Scholar] [CrossRef]
- Engelmann, F.M.; Mayer, I.; Gabrielli, D.S.; Toma, H.E.; Kowaltowski, A.J.; Araki, K.; Baptista, M.S. Interaction of cationic meso-porphyrins with liposomes, mitochondria and erythrocytes. J. Bioenerget. Biomembr. 2007, 39, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Quishida, C.C.; Mima, E.G.; Dovigo, L.N.; Jorge, J.H.; Bagnato, V.S.; Pavarina, A.C. Photodynamic inactivation of a multispecies biofilm using Photodithazine(®) and LED light after one and three successive applications. Lasers Med. Sci. 2015, 30, 2303–2312. [Google Scholar] [CrossRef] [PubMed]
- Dovigo, L.N.; Carmello, J.C.; Carvalho, M.T.; Mima, E.G.; Vergani, C.E.; Bagnato, V.S.; Pavarina, A.C. Photodynamic inactivation of clinical isolates of Candida using Photodithazine®. Biofouling 2013, 29, 1057–1067. [Google Scholar] [CrossRef] [PubMed]
- Carmello, J.C.; Alves, F.; Mima, E.G.; Jorge, J.H.; Bagnato, V.S.; Pavarina, A.C. Photoinactivation of single and mixed biofilms of Candida albicans and non-albicans Candida species using Photodithazine®. Photodiagnosis Photodyn. Ther. 2017, 17, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Carmello, J.C.; Dovigo, L.N.; Mima, E.G.; Jorge, J.H.; Costa, C.A.d.; Bagnato, V.S.; Pavarina, A.C. In vivo evaluation of photodynamic inactivation using Photodithazine® against Candida albicans. Photochem. Photobiol. Sci. 2015, 14, 1319–1328. [Google Scholar] [CrossRef] [PubMed]
- Alves, F.; Carmello, J.C.; Mima, E.G.O.; Costa, C.A.S.; Bagnato, V.S.; Pavarina, A.C. Photodithazine-mediated antimicrobial photodynamic therapy against fluconazole-resistant Candida albicans in vivo. Med. Mycol. 2018, 57, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Carmello, J.C.; Alves, F.; Basso, F.G.; de Souza Costa, C.A.; Bagnato, V.S.; de Oliveira Mima, E.G.; Pavarina, A.C. Treatment of oral candidiasis using Photodithazine®-mediated photodynamic therapy in vivo. PLoS ONE 2016, 11, e0156947. [Google Scholar] [CrossRef]
- Antoniadou, A.; Kontoyiannis, D.P. Status of combination therapy for refractory mycoses. Curr. Opin. Infect. Dis. 2003, 16, 539–545. [Google Scholar] [CrossRef]
- Hsieh, Y.H.; Zhang, J.H.; Chuang, W.C.; Yu, K.H.; Huang, X.B.; Lee, Y.C.; Lee, C.I. An in Vitro Study on the effect of combined treatment with photodynamic and chemical therapies on Candida albicans. Int. J. Mol. Sci. 2018, 19, 337. [Google Scholar] [CrossRef]
- Barra, F.; Roscetto, E.; Soriano, A.A.; Vollaro, A.; Postiglione, I.; Pierantoni, M.G.; Palumbo, G.; Catania, M.R. Photodynamic and antibiotic therapy in combination to fight biofilms and resistant surface bacterial infections. Int. J. Mol. Sci. 2015, 16, 20417–20430. [Google Scholar] [CrossRef]
- Agostinis, P.; Berg, K.; Cengel, K.A.; Foster, T.H.; Girotti, A.W.; Gollnick, S.O.; Hahn, S.M.; Hamblin, M.R.; Juzeniene, A.; Kessel, D.; et al. Photodynamic therapy of cancer: An update. CA Cancer J. Clin. 2011, 61, 250–281. [Google Scholar] [CrossRef] [PubMed]
- Kessel, D. Autophagic death probed by photodynamic therapy. Autophagy 2015, 11, 1941–1943. [Google Scholar] [CrossRef] [PubMed]
- Martins, W.K.; Santos, N.F.; Rocha, C.S.; Bacellar, I.O.L.; Tsubone, T.M.; Viotto, A.C.; Matsukuma, A.Y.; Abrantes, A.B.P.; Siani, P.; Dias, L.G.; et al. Parallel damage in mitochondria and lysosomes is an efficient way to photoinduce cell death. Autophagy 2019, 15, 259–279. [Google Scholar] [CrossRef] [PubMed]
- Abrantes, A.B.P.; Dias, G.C.; Souza-Pinto, N.C.; Baptista, M.S. p53-Dependent and p53-Independent responses of cells challenged by photosensitization. Photochem. Photobiol. 2019, 95, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Bacellar, I.O.; Tsubone, T.M.; Pavani, C.; Baptista, M.S. Photodynamic efficiency: From molecular photochemistry to cell death. Int. J. Mol. Sci. 2015, 16, 20523–20559. [Google Scholar] [CrossRef] [PubMed]
- Castano, A.P.; Demidova, T.N.; Hamblin, M.R. Mechanisms in photodynamic therapy: Part one-photosensitizers, photochemistry and cellular localization. Photodiagnosis Photodyn. Ther. 2004, 1, 279–293. [Google Scholar] [CrossRef]
- Li, R.; Hannon, G.J.; Beach, D.; Stillman, B. Subcellular distribution of p21 and PCNA in normal and repair-deficient cells following DNA damage. Curr. Biol. 1996, 6, 189–199. [Google Scholar] [CrossRef]
- Snell, S.B.; Foster, T.H.; Haidaris, C.G. Miconazole induces fungistasis and increases killing of Candida albicans subjected to photodynamic therapy. Photochem. Photobiol. 2012, 88, 596–603. [Google Scholar] [CrossRef]
- Davies, A.; Gebremedhin, S.; Yee, M.; Padilla, R.J.; Duzgunes, N.; Konopka, K.; Dorocka-Bobkowska, B. Cationic porphyrin-mediated photodynamic inactivation of Candida biofilms and the effect of miconazole. J. Physiol. Pharmacol. 2016, 67, 777–783. [Google Scholar]
- Kashef, N.; Hamblin, M.R. Can microbial cells develop resistance to oxidative stress in antimicrobial photodynamic inactivation? Drug Resist. Updat. 2017, 31, 31–42. [Google Scholar] [CrossRef]
- Lopes, M.; Alves, C.T.; Raju, B.R.; Gonçalves, M.S.; Coutinho, P.J.; Henriques, M.; Belo, I. Application of benzo[a]phenoxazinium chlorides in Antimicrobial Photodynamic Therapy of Candida albicans biofilms. J. Photochem. Photobiol. B 2014, 141, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Dovigo, L.N.; Pavarina, A.C.; Mima, E.G.; Giampaolo, E.T.; Vergani, C.E.; Bagnato, V.S. Fungicidal effect of photodynamic therapy against fluconazole-resistant Candida albicans and Candida glabrata. Mycoses 2011, 54, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Shen, M.; Huang, Y.; Lin, H.; Chen, C. Photodynamic inactivation potentiates the susceptibility of antifungal agents against the planktonic and biofilm cells of Candida albicans. Int. J. Mol. Sci. 2018, 19, 434. [Google Scholar] [CrossRef] [PubMed]
- Karanja, C.W.; Hong, W.; Younis, W.; Eldesouky, H.E.; Seleem, M.N.; Cheng, J.X. Stimulated raman imaging reveals aberrant lipogenesis as a metabolic marker for azole-resistant Candida albicans. Anal. Chem. 2017, 89, 9822–9829. [Google Scholar] [CrossRef] [PubMed]
- Maisch, T.; Bosl, C.; Szeimies, R.; Lehn, N.; Abels, C. Photodynamic effects of novel XF porphyrin derivatives on prokaryotic and eukaryotic cells. Antimicrob. Agents Chemother. 2005, 49, 1542–1552. [Google Scholar] [CrossRef] [PubMed]
- Okada, M.; Hisajima, T.; Ishibashi, H.; Miyasaka, T.; Abe, S.; Satoh, T. Pathological analysis of the Candida albicans-infected tongue tissues of a murine oral candidiasis model in the early infection stage. Arch. Oral Biol. 2013, 58, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Sobue, T.; Bertolini, M.; Thompson, A.; Dongari-Bagtzoglou, A. Streptococcus oralis and Candida albicans synergistically activate μ-calpain to degrade e-cadherin from oral epithelial junctions. J. Infect. Dis. 2016, 214, 925–934. [Google Scholar] [CrossRef] [PubMed]
- Abreu-Blanco, M.T.; Watts, J.J.; Verboon, J.M.; Parkhurst, S.M. Cytoskeleton responses in wound repair. Cell. Mol. Life Sci. 2012, 69, 2469–2483. [Google Scholar] [CrossRef]
- Yan, L.; Yang, C.; Tang, J. Disruption of the intestinal mucosal barrier in Candida albicans infections. Microbiol. Res. 2013, 168, 389–395. [Google Scholar] [CrossRef]
- Sakima, V.T.; Barbugli, P.A.; Cerri, P.S.; Chorilli, M.; Carmello, J.C.; Pavarina, A.C.; Mima, E.G.O. Antimicrobial photodynamic therapy mediated by curcumin-loaded polymeric nanoparticles in a murine model of oral candidiasis. Molecules 2018, 23, 2075. [Google Scholar] [CrossRef]
- Galanos, P.; Vougas, K.; Walter, D.; Polyzos, A.; Maya-Mendoza, A.; Haagensen, E.J.; Kokkalis, A.; Roumelioti, F.M.; Gagos, S.; Tzetis, M.; et al. Chronic p53-independent p21 expression causes genomic instability by deregulating replication licensing. Nat. Cell. Biol. 2016, 18, 777–789. [Google Scholar] [CrossRef] [PubMed]
- Nakonieczna, J. Comment on Effectiveness of antimicrobial photodynamic therapy (AmPDT) on Staphylococcus aureus using phenothiazine compound with red laser. Lasers Med. Sci. 2017, 32, 1667–1668. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cieplik, F.; Tabenski, L.; Buchalla, W.; Maisch, T. Antimicrobial photodynamic therapy for inactivation of biofilms formed by oral key pathogens. Front. Microbiol. 2014, 5, 405. [Google Scholar] [CrossRef] [PubMed]
- Taraszkiewicz, A.; Grinholc, M.; Bielawski, K.P.; Kawiak, A.; Nakonieczna, J. Imidazoacridinone derivatives as efficient sensitizers in photoantimicrobial chemotherapy. Appl. Environ. Microbiol. 2013, 79, 3692–3702. [Google Scholar] [CrossRef] [PubMed]
- Kiesslich, T.; Gollmer, A.; Maisch, T.; Berneburg, M.; Plaetzer, K. A comprehensive tutorial on in vitro characterization of new photosensitizers for photodynamic antitumor therapy and photodynamic inactivation of microorganisms. Biomed. Res. Int. 2013, 2013, 840417. [Google Scholar] [CrossRef] [PubMed]
- Alves, F.; Alonso, G.C.; Carmello, J.C.; Mima, E.G.O.; Bagnato, V.S.; Pavarina, A.C. Antimicrobial photodynamic therapy mediated by Photodithazine® in the treatment of denture stomatitis: A case report. Photodiagnosis Photodyn. Ther. 2018, 21, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Takakura, N.; Sato, Y.; Ishibashi, H.; Oshima, H.; Uchida, K.; Yamaguchi, H.; Abe, S. A novel murine model of oral candidiasis with local symptoms characteristic of oral thrush. Microbiol. Immunol. 2003, 47, 321–326. [Google Scholar] [CrossRef] [PubMed]

| Experimental Groups | Treatment Description | Number of Animals, after 24 h of the Treatments | Number of Animals, after 7 Days of the Treatments | Total Number of Animals (n) |
|---|---|---|---|---|
| P+L+ (aPDT) | Application of PDZ at 200 mg/L (for 20 min of pre-irradiation), followed by irradiation with LED (50 J/cm2) for 19 min | 12 | 12 | 24 |
| P+L- | Application of PDZ at 200 mg/L (for 20 min) | 12 | 12 | 24 |
| P-L+ | Irradiation with LED, with a dose of light of 50 J/cm2 | 12 | 12 | 24 |
| P-L- | Animals inoculated with C. albicans, without treatment (positive control of infection) | 12 | 12 | 24 |
| NYS | Animals treated with Nystatin oral suspension (100,000 I.U. for 39 min) | 12 | 12 | 24 |
| P+L+NYS | Application of PDZ (200 mg/L) and LED light (50 J/cm2) + Nystatin oral suspension | 12 | 12 | 24 |
| NYS+P+L+ | Application of Nystatin oral suspension + PDZ (200 mg/L) and LED light (50 J/cm2) | 12 | 12 | 24 |
| NCI | Negative control of infection (healthy animals) | 3 | 3 | 6 |
| TOTAL | 87 | 87 | 174 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janeth Rimachi Hidalgo, K.; Cabrini Carmello, J.; Carolina Jordão, C.; Aboud Barbugli, P.; de Sousa Costa, C.A.; Garcia de Oliveira Mima, E.; Pavarina, A.C. Antimicrobial Photodynamic Therapy in Combination with Nystatin in the Treatment of Experimental Oral Candidiasis Induced by Candida albicans Resistant to Fluconazole. Pharmaceuticals 2019, 12, 140. https://doi.org/10.3390/ph12030140
Janeth Rimachi Hidalgo K, Cabrini Carmello J, Carolina Jordão C, Aboud Barbugli P, de Sousa Costa CA, Garcia de Oliveira Mima E, Pavarina AC. Antimicrobial Photodynamic Therapy in Combination with Nystatin in the Treatment of Experimental Oral Candidiasis Induced by Candida albicans Resistant to Fluconazole. Pharmaceuticals. 2019; 12(3):140. https://doi.org/10.3390/ph12030140
Chicago/Turabian StyleJaneth Rimachi Hidalgo, Karem, Juliana Cabrini Carmello, Cláudia Carolina Jordão, Paula Aboud Barbugli, Carlos Alberto de Sousa Costa, Ewerton Garcia de Oliveira Mima, and Ana Claudia Pavarina. 2019. "Antimicrobial Photodynamic Therapy in Combination with Nystatin in the Treatment of Experimental Oral Candidiasis Induced by Candida albicans Resistant to Fluconazole" Pharmaceuticals 12, no. 3: 140. https://doi.org/10.3390/ph12030140
APA StyleJaneth Rimachi Hidalgo, K., Cabrini Carmello, J., Carolina Jordão, C., Aboud Barbugli, P., de Sousa Costa, C. A., Garcia de Oliveira Mima, E., & Pavarina, A. C. (2019). Antimicrobial Photodynamic Therapy in Combination with Nystatin in the Treatment of Experimental Oral Candidiasis Induced by Candida albicans Resistant to Fluconazole. Pharmaceuticals, 12(3), 140. https://doi.org/10.3390/ph12030140