Radioimmunotherapy in Non-Hodgkin’s Lymphoma: Retrospective Adverse Event Profiling of Zevalin and Bexxar
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Integration
2.2. Statistical Analysis
- FAERS Bexxar cohort: 290 AEs (FAERS cases annotated with the ‘Tositumomab’ drug record).
- FAERS Zevalin cohort: 1196 AEs (FAERS cases annotated with the ‘Ibritumomab’ drug record).
- VigiBase Bexxar cohort: 307 AEs (VigiBase cases with product (drug) names mentioning ‘Bexxar’ or ‘Tositumomab’).
- VigiBase Zevalin cohort: 1125 AEs (VigiBase cases with product (drug) names mentioning ‘Zevalin’ or ‘Ibritumomab’).
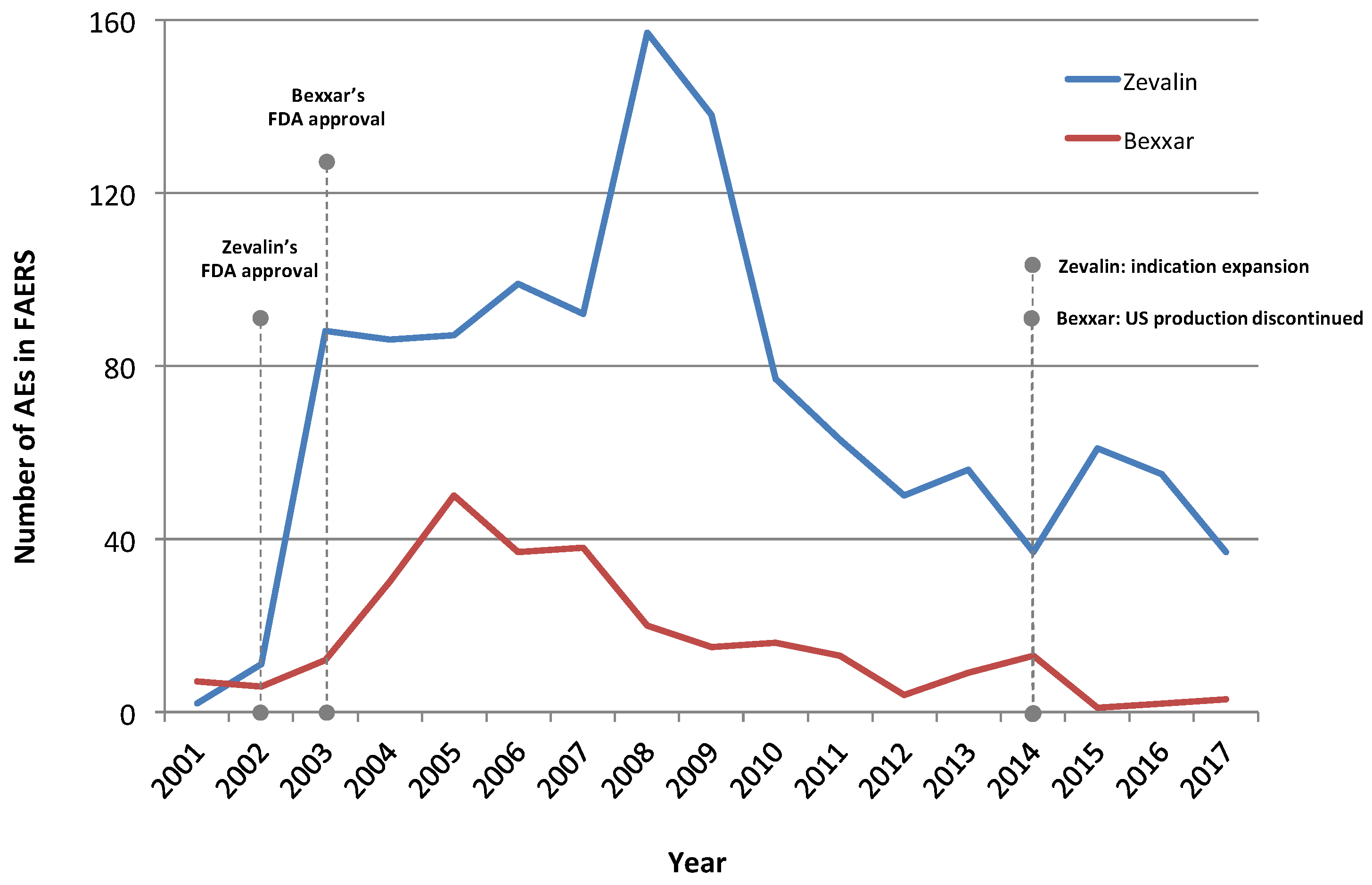
3. Results
3.1. Adverse Event Profiling
3.1.1. Zevalin Reactions
3.1.2. Bexxar Reactions
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cragg, M.S.; Walshe, C.A.; Ivanov, A.O.; Glennie, M.J. The biology of CD20 and its potential as a target for mAb therapy. Curr. Dir. Autoimmun. 2005, 8, 140–174. [Google Scholar] [PubMed]
- Cheson, B.D.; Leonard, J.P. Monoclonal antibody therapy for B-cell non-Hodgkin’s lymphoma. N. Engl. J. Med. 2008, 359, 613–626. [Google Scholar] [CrossRef] [PubMed]
- Cheson, B.D. Radioimmunotherapy of non-Hodgkin lymphomas. Blood 2003, 101, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Green, D.J.; Press, O.W. Whither Radioimmunotherapy: To Be or Not To Be? Cancer Res. 2017, 77, 2191–2196. [Google Scholar] [CrossRef] [PubMed]
- Shimoni, A.; Zwas, S.T. Radioimmunotherapy and Autologous Stem-Cell Transplantation in the Treatment of B-Cell Non-Hodgkin Lymphoma. Semin. Nucl. Med. 2016, 46, 119–125. [Google Scholar] [CrossRef]
- Radiosensitivity of tumors. JAMA 1934, 102, 619–620. [CrossRef][Green Version]
- Murthy, V.; Thomas, K.; Foo, K.; Cunningham, D.; Johnson, B.; Norman, A.; Horwich, A. Efficacy of palliative low-dose involved-field radiation therapy in advanced lymphoma: A phase II study. Clin. Lymphoma Myeloma 2008, 8, 241–245. [Google Scholar] [CrossRef]
- Haas, R.L.M.; Poortmans, P.; de Jong, D.; Aleman, B.M.P.; Dewit, L.G.H.; Verheij, M.; Hart, A.M.; van Oers, M.H.J.; van der Hulst, M.; Baars, J.W.; et al. High response rates and lasting remissions after low-dose involved field radiotherapy in indolent lymphomas. J. Clin. Oncol. 2003, 21, 2474–2480. [Google Scholar] [CrossRef]
- Illidge, T.; Specht, L.; Yahalom, J.; Aleman, B.; Berthelsen, A.K.; Constine, L.; Dabaja, B.; Dharmarajan, K.; Ng, A.; Ricardi, U.; et al. Modern radiation therapy for nodal non-Hodgkin lymphoma-target definition and dose guidelines from the International Lymphoma Radiation Oncology Group. Int. J. Radiat. Oncol. Biol. Phys. 2014, 89, 49–58. [Google Scholar] [CrossRef]
- Kaminski, M.S.; Zasadny, K.R.; Francis, I.R.; Milik, A.W.; Ross, C.W.; Moon, S.D.; Crawford, S.M.; Burgess, J.M.; Petry, N.A.; Butchko, G.M. Radioimmunotherapy of B-cell lymphoma with [131I] anti-B1 (anti-CD20) antibody. N. Engl. J. Med. 1993, 329, 459–465. [Google Scholar] [CrossRef]
- Vose, J.M.; Wahl, R.L.; Saleh, M.; Rohatiner, A.Z.; Knox, S.J.; Radford, J.A.; Zelenetz, A.D.; Tidmarsh, G.F.; Stagg, R.J.; Kaminski, M.S. Multicenter phase II study of iodine-131 tositumomab for chemotherapy-relapsed/refractory low-grade and transformed low-grade B-cell non-Hodgkin’s lymphomas. J. Clin. Oncol. 2000, 18, 1316–1323. [Google Scholar] [CrossRef] [PubMed]
- Witzig, T.E.; Flinn, I.W.; Gordon, L.I.; Emmanouilides, C.; Czuczman, M.S.; Saleh, M.N.; Cripe, L.; Wiseman, G.; Olejnik, T.; Multani, P.S.; et al. Treatment with ibritumomab tiuxetan radioimmunotherapy in patients with rituximab-refractory follicular non-Hodgkin’s lymphoma. J. Clin. Oncol. 2002, 20, 3262–3269. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, G.A.; Gordon, L.I.; Multani, P.S.; Witzig, T.E.; Spies, S.; Bartlett, N.L.; Schilder, R.J.; Murray, J.L.; Saleh, M.; Allen, R.S.; et al. Ibritumomab tiuxetan radioimmunotherapy for patients with relapsed or refractory non-Hodgkin lymphoma and mild thrombocytopenia: A phase II multicenter trial. Blood 2002, 99, 4336–4342. [Google Scholar] [CrossRef] [PubMed]
- Morschhauser, F.; Radford, J.; Van Hoof, A.; Vitolo, U.; Soubeyran, P.; Tilly, H.; Huijgens, P.C.; Kolstad, A.; d’Amore, F.; Gonzalez Diaz, M.; et al. Phase III trial of consolidation therapy with yttrium-90-ibritumomab tiuxetan compared with no additional therapy after first remission in advanced follicular lymphoma. J. Clin. Oncol. 2008, 26, 5156–5164. [Google Scholar] [CrossRef] [PubMed]
- Kaminski, M.S.; Tuck, M.; Estes, J.; Kolstad, A.; Ross, C.W.; Zasadny, K.; Regan, D.; Kison, P.; Fisher, S.; Kroll, S.; et al. 131I-tositumomab therapy as initial treatment for follicular lymphoma. N. Engl. J. Med. 2005, 352, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Press, O.W.; Unger, J.M.; Braziel, R.M.; Maloney, D.G.; Miller, T.P.; Leblanc, M.; Fisher, R.I.; Southwest Oncology Group. Phase II trial of CHOP chemotherapy followed by tositumomab/iodine I-131 tositumomab for previously untreated follicular non-Hodgkin’s lymphoma: Five-year follow-up of Southwest Oncology Group Protocol. SJ Clin. Oncol. 2006, 24, 4143–4149. [Google Scholar] [CrossRef] [PubMed]
- Zinzani, P.L.; Tani, M.; Pulsoni, A.; Gobbi, M.; Perotti, A.; De Luca, S.; Fabbri, A.; Zaccaria, A.; Voso, M.T.; Fattori, P.; et al. Fludarabine and mitoxantrone followed by yttrium-90 ibritumomab tiuxetan in previously untreated patients with follicular non-Hodgkin lymphoma trial: A phase II non-randomised trial (FLUMIZ). Lancet Oncol. 2008, 9, 352–358. [Google Scholar] [CrossRef]
- Zinzani, P.L.; Rossi, G.; Franceschetti, S.; Botto, B.; Di Rocco, A.; Cabras, M.G.; Petti, M.C.; Stefoni, V.; Broccoli, A.; Fanti, S.; et al. Phase II trial of short-course R-CHOP followed by 90Y-ibritumomab tiuxetan in previously untreated high-risk elderly diffuse large B-cell lymphoma patients. Clin. Cancer Res. 2010, 16, 3998–4004. [Google Scholar] [CrossRef]
- Illidge, T.M. Radioimmunotherapy of lymphoma: A treatment approach ahead of its time or past its sell-by date? J. Clin. Oncol. 2010, 28, 2944–2946. [Google Scholar] [CrossRef]
- Soldatos, T.G.; Perdigão, N.; Brown, N.P.; Sabir, K.S.; O’Donoghue, S.I. How to learn about gene function: Text-mining or ontologies? Methods 2015, 74, 3–15. [Google Scholar] [CrossRef]
- Soldatos, T.G.; Taglang, G.; Jackson, D.B. In Silico Profiling of Clinical Phenotypes for Human Targets Using Adverse Event Data. High Throughput 2018, 7, 37. [Google Scholar] [CrossRef] [PubMed]
- Evans, S.J.; Waller, P.C.; Davis, S. Use of proportional reporting ratios (PRRs) for signal generation from spontaneous adverse drug reaction reports. Pharmacoepidemiol. Drug Saf. 2001, 10, 483–486. [Google Scholar] [CrossRef]
- Van Puijenbroek, E.P.; Bate, A.; Leufkens, H.G.M.; Lindquist, M.; Orre, R.; Egberts, A.C.G. A comparison of measures of disproportionality for signal detection in spontaneous reporting systems for adverse drug reactions. Pharmacoepidemiol. Drug Saf. 2002, 11, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, N.G.; Huang, P.; Buchanan, J.W.; Wahl, R.L. Radioimmunotherapy in non-Hodgkin lymphoma: Opinions of nuclear medicine physicians and radiation oncologists. J. Nucl. Med. 2011, 52, 830–838. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, N.G.; Ma, J.; Huang, P.; Buchanan, J.; Wahl, R.L. Radioimmunotherapy in non-Hodgkin lymphoma: Opinions of U.S. medical oncologists and hematologists. J. Nucl. Med. 2010, 51, 987–994. [Google Scholar] [CrossRef] [PubMed]
- ZEVALIN® (Ibritumomab Tiuxeta)—Package Insert from www.zevalin.com. Available online: http://www.zevalin.com/downloads/Zevalin_Package_Insert.pdf (accessed on 15 August 2019).
- DailyMed—ZEVALIN- Ibritumomab Tiuxetan. Available online: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=ccf29429-bf8e-4cce-bd12-1de56853fc5b (accessed on 15 August 2019).
- Zevalin. European Medicines Agency. 2018. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/zevalin (accessed on 15 August 2019).
- DailyMed—RITUXAN- Rituximab Injection, Solution. Available online: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=b172773b-3905-4a1c-ad95-bab4b6126563 (accessed on 15 August 2019).
- DailyMed—TRUXIMA- Rituximab-Abbs Injection, Solution [Internet]. [Cited 2019 Aug 15]. Available online: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=9af3ddc7-4217-417a-ac89-8704edc5bc44 (accessed on 15 August 2019).
- Rixathon. European Medicines Agency. 2018. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/rixathon (accessed on 15 August 2019).
- Truxima. European Medicines Agency. 2018. Available online: https://www.ema.europa.eu/en/medicines/human/EPAR/truxima (accessed on 15 August 2019).
- Delaloye, A.B.; Antonescu, C.; Louton, T.; Kuhlmann, J.; Hagenbeek, A. Dosimetry of 90Y-ibritumomab tiuxetan as consolidation of first remission in advanced-stage follicular lymphoma: Results from the international phase 3 first-line indolent trial. J. Nucl. Med. 2009, 50, 1837–1843. [Google Scholar] [CrossRef] [PubMed]
- Wiseman, G.A.; Kornmehl, E.; Leigh, B.; Erwin, W.D.; Podoloff, D.A.; Spies, S.; Sparks, R.B.; Stabin, M.G.; Witzig, T.; White, C.A. Radiation dosimetry results and safety correlations from 90Y-ibritumomab tiuxetan radioimmunotherapy for relapsed or refractory non-Hodgkin’s lymphoma: Combined data from 4 clinical trials. J. Nucl. Med. 2003, 44, 465–474. [Google Scholar] [PubMed]
- Wiseman, G.A.; Leigh, B.R.; Dunn, W.L.; Stabin, M.G.; White, C.A. Additional radiation absorbed dose estimates for Zevalin radioimmunotherapy. Cancer Biother. Radiopharm. 2003, 18, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Wahl, R.L.; Zasadny, K.R.; MacFarlane, D.; Francis, I.R.; Ross, C.W.; Estes, J.; Fisher, S.; Regan, D.; Kroll, S.; Kaminski, M.S. Iodine-131 anti-B1 antibody for B-cell lymphoma: An update on the Michigan Phase I experience. J. Nucl. Med. 1998, 39 (Suppl. 8), 21S–27S. [Google Scholar]
- BEXXAR (Tositumomab and Iodine I 131 Tositumomab). Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/125011s0126lbl.pdf (accessed on 15 August 2019).
- Soldatos, T.G.; Dimitrakopoulou-Strauss, A.; Larribere, L.; Hassel, J.C.; Sachpekidis, C. Retrospective Side Effect Profiling of the Metastatic Melanoma Combination Therapy Ipilimumab-Nivolumab Using Adverse Event Data. Diagnostics 2018, 8, 76. [Google Scholar] [CrossRef]
- Soldatos, T.G.; Iakovou, I.; Sachpekidis, C. Retrospective Toxicological Profiling of Radium-223 Dichloride for the Treatment of Bone Metastases in Prostate Cancer Using Adverse Event Data. Medicina 2019, 55, 149. [Google Scholar] [CrossRef]
- Soldatou, V.; Soldatos, A.; Soldatos, T. Examining Socioeconomic and Computational Aspects of Vaccine Pharmacovigilance. BioMed Res. Int. 2019. Available online: https://www.hindawi.com/journals/bmri/2019/6576483/ (accessed on 21 February 2019).
- Racz, R.; Soldatos, T.G.; Jackson, D.; Burkhart, K. Association Between Serotonin Syndrome and Second-Generation Antipsychotics via Pharmacological Target-Adverse Event Analysis. Clin. Transl. Sci. 2018, 11, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Soldatos, T.G.; Jackson, D.B. Adverse Event Circumstances and the Case of Drug Interactions. Healthcare 2019, 7, 45. [Google Scholar] [CrossRef] [PubMed]
- Barr, P.M.; Li, H.; Burack, W.R.; LeBlanc, M.; Smith, S.M.; Gopal, A.K.; Floyd, J.D.; Persky, D.O.; Press, O.W.; Fisher, R.I.; et al. R-CHOP, radioimmunotherapy, and maintenance rituximab in untreated follicular lymphoma (SWOG S0801): A single-arm, phase 2, multicentre study. Lancet Haematol. 2018, 5, e102–e108. [Google Scholar] [CrossRef]
- Lansigan, F.; Costa, C.A.; Zaki, B.; Yen, S.; Winer, E.P.; Ryan, H.; Findley, D.; Metzler, S.; Shaw, L.; Toaso, B.; et al. Multicenter, Open Label, Phase II Study of Bendamustine and Rituximab followed by 90-yttrium (Y) Ibritumomab Tiuxetan for Untreated Follicular Lymphoma (Fol-BRITe). Clin. Cancer Res. 2019, 26, 3755. [Google Scholar] [CrossRef] [PubMed]
- Lugtenburg, P.J.; Zijlstra, J.M.; Doorduijn, J.K.; Böhmer, L.H.; Hoogendoorn, M.; Berenschot, H.W.; Beeker, A.; van der Burg-de Graauw, N.C.; Schouten, H.C.; Bilgin, Y.M.; et al. Rituximab-PECC induction followed by 90 Y-ibritumomab tiuxetan consolidation in relapsed or refractory DLBCL patients who are ineligible for or have failed ASCT: Results from a phase II HOVON study. Blood 2005, 126, 2707. [Google Scholar] [CrossRef]
| Outcome | % Zevalin AEs | % Bexxar AEs | ||
|---|---|---|---|---|
| FAERS | VigiBase | FAERS | VigiBase | |
| Death | 33.4 | 22.8 | 22.1 | 24.1 |
| Hospitalization | 41.5 | 22.1 | 31.0 | 20.5 |
| Life threatening | 7.4 | 3.9 | 7.9 | 5.2 |
| Other | 46.3 | 31.8 | 36.6 | 23.1 |
| Reaction (MedDRA PT) Name | Number of AEs_in Cohort | PRR | Cohort % |
|---|---|---|---|
| Thrombocytopenia 1 | 151 | 21.4 | 12.6 |
| Neutropenia | 124 | 19.0 | 10.4 |
| Sepsis | 98 | 13.0 | 8.2 |
| White blood cell count decreased 2 | 84 | 13.2 | 7.0 |
| Myelodysplastic syndrome (MDS) | 83 | 74.1 | 6.9 |
| Platelet count decreased 1 | 80 | 12.0 | 6.7 |
| Pyrexia | 79 | 3.4 | 6.6 |
| Pancytopenia | 78 | 20.0 | 6.5 |
| Febrile neutropenia | 76 | 20.9 | 6.4 |
| Anaemia 3 | 65 | 4.7 | 5.4 |
| Bone marrow failure | 63 | 37.3 | 5.3 |
| Diarrhoea | 60 | 1.7 | 5.0 |
| Pneumonia | 57 | 2.9 | 4.8 |
| Haemoglobin decreased 3 | 51 | 6.6 | 4.3 |
| Meningitis tuberculous | 49 | 1489.1 | 4.1 |
| Multi-organ failure | 44 | 15.9 | 3.7 |
| Acute myeloid leukaemia (AML) | 43 | 42.5 | 3.6 |
| Leukopenia 2 | 43 | 13.6 | 3.6 |
| Neutrophil count decreased | 42 | 19.4 | 3.5 |
| Progressive multifocal leukoencephalopathy | 38 | 64.5 | 3.2 |
| Nausea | 36 | 0.7 | 3.0 |
| Cytomegalovirus infection | 33 | 37.4 | 2.8 |
| Septic shock | 33 | 12.7 | 2.8 |
| Mucosal inflammation | 32 | 19.7 | 2.7 |
| Infection | 32 | 4.0 | 2.7 |
| Renal failure | 31 | 3.5 | 2.6 |
| Reaction (MedDRA PT) Name | Number of AEs_in Cohort | PRR | Cohort % |
|---|---|---|---|
| Thrombocytopenia 1 | 165 | 20.1 | 14.7 |
| Neutropenia | 157 | 24.2 | 14.0 |
| Platelet count decreased 1 | 117 | 35.2 | 10.4 |
| Pancytopenia | 90 | 37.2 | 8.0 |
| Sepsis | 71 | 20.0 | 6.3 |
| White blood cell count decreased 2 | 71 | 18.4 | 6.3 |
| Myelodysplastic syndrome (MDS) | 69 | 170.1 | 6.1 |
| Anaemia 3 | 59 | 6.1 | 5.2 |
| Pneumonia | 53 | 5.2 | 4.7 |
| Pyrexia | 51 | 1.4 | 4.5 |
| Febrile neutropenia | 50 | 26.2 | 4.4 |
| Leukopenia 2 | 48 | 9.1 | 4.3 |
| Neutrophil count decreased | 42 | 24.1 | 3.7 |
| Meningitis tuberculous | 37 | 3877.3 | 3.3 |
| Nausea | 37 | 0.6 | 3.3 |
| Fatigue | 34 | 1.2 | 3.0 |
| Diarrhoea | 34 | 1.0 | 3.0 |
| Acute myeloid leukaemia (AML) | 33 | 93.7 | 2.9 |
| Bone marrow failure | 32 | 15.1 | 2.8 |
| Pain | 29 | 1.2 | 2.6 |
| Progressive multifocal leukoencephalopathy | 27 | 131.5 | 2.4 |
| Haemoglobin decreased 3 | 25 | 7.0 | 2.2 |
| Dyspnoea | 24 | 0.7 | 2.1 |
| Dehydration | 23 | 4.6 | 2.0 |
| Vomiting | 23 | 0.6 | 2.0 |
| Asthenia | 23 | 1.2 | 2.0 |
| Reaction (MedDRA PT) Name | Number of AEs_in Cohort | PRR | Cohort % |
|---|---|---|---|
| Myelodysplastic syndrome (MDS) | 46 | 168.7 | 15.9 |
| Pyrexia | 30 | 5.3 | 10.3 |
| Acute myeloid leukaemia (AML) | 22 | 89.4 | 7.6 |
| Fatigue | 22 | 2.0 | 7.6 |
| Dyspnoea | 21 | 2.4 | 7.2 |
| Nausea | 18 | 1.4 | 6.2 |
| Anaemia | 17 | 5.1 | 5.9 |
| Thrombocytopenia + | 16 | 9.3 | 5.5 |
| Pancytopenia | 14 | 14.8 | 4.8 |
| Hypotension | 14 | 4.4 | 4.8 |
| Febrile neutropenia | 13 | 14.7 | 4.5 |
| Neutropenia | 13 | 8.2 | 4.5 |
| Chills | 13 | 6.7 | 4.5 |
| Lymphadenopathy | 12 | 20.4 | 4.1 |
| Pneumonia | 12 | 2.6 | 4.1 |
| Vomiting | 12 | 1.6 | 4.1 |
| Dizziness | 12 | 1.5 | 4.1 |
| Infusion related reaction | 11 | 14.9 | 3.8 |
| Rash | 11 | 1.9 | 3.8 |
| Platelet count decreased + | 10 | 6.2 | 3.4 |
| Back pain | 10 | 2.7 | 3.4 |
| Arthralgia | 10 | 1.8 | 3.4 |
| Asthenia | 10 | 1.7 | 3.4 |
| Pleural effusion | 9 | 8.1 | 3.1 |
| Cardiac failure congestive | 9 | 4.2 | 3.1 |
| Decreased appetite | 9 | 2.5 | 3.1 |
| Weight decreased | 9 | 2.1 | 3.1 |
| Reaction (MedDRA PT) Name | Number of AEs_in Cohort | PRR | Cohort % |
|---|---|---|---|
| Myelodysplastic syndrome (MDS) | 43 | 386.9 | 14.0 |
| Acute myeloid leukaemia (AML) | 27 | 280.5 | 8.8 |
| Pyrexia | 27 | 2.7 | 8.8 |
| Fatigue | 22 | 2.8 | 7.2 |
| Dyspnoea | 20 | 2.3 | 6.5 |
| Thrombocytopenia + | 18 | 8.0 | 5.9 |
| Nausea | 17 | 1.0 | 5.5 |
| Febrile neutropenia | 15 | 28.7 | 4.9 |
| Pancytopenia | 15 | 22.7 | 4.9 |
| Anaemia | 15 | 5.7 | 4.9 |
| Neutropenia | 13 | 7.3 | 4.2 |
| Pneumonia | 13 | 4.7 | 4.2 |
| Hypotension | 13 | 4.0 | 4.2 |
| Chills | 13 | 3.7 | 4.2 |
| Lymphadenopathy | 12 | 16.6 | 3.9 |
| Infusion related reaction | 11 | 24.8 | 3.6 |
| Infection | 11 | 8.3 | 3.6 |
| Back pain | 11 | 4.1 | 3.6 |
| Rash | 11 | 0.8 | 3.6 |
| Platelet count decreased + | 10 | 11.0 | 3.3 |
| Pain in extremity | 10 | 2.8 | 3.3 |
| Arthralgia | 10 | 2.2 | 3.3 |
| Asthenia | 10 | 1.9 | 3.3 |
| Vomiting | 10 | 0.9 | 3.3 |
| White blood cell count decreased | 9 | 8.6 | 2.9 |
| Cardiac failure congestive | 9 | 7.9 | 2.9 |
| Weight decreased | 9 | 3.7 | 2.9 |
| Zevalin | Bexxar |
|---|---|
| Blood/Cytopenias | |
| Thrombocytopenia Neutropenia Leukopenia Pancytopenia Anemia Bone marrow failure | Anemia Thrombocytopenia Pancytopenia Neutropenia |
| Infections/Inflammations | |
| Sepsis Pyrexia + Febrile neutropenia Pneumonia Meningitis tuberculous Cytomegalovirus infection Septic schock Mucosal inflammation | Pyrexia + Febrile neutropenia Chills Pneumonia |
| Secondary malignancies | |
| Myelodysplastic syndrome (MDS) | Myelodysplastic syndrome (MDS) |
| Acute myeloid leukaemia (AML) | Acute myeloid leukaemia (AML) |
| Gastrointestinal | |
| Diarrhoea Nausea | Nausea |
| Other | |
| Multi-organ failure Progressive multifocal leukoencephalopathy Renal failure Fatigue Pain Asthenia Dehydration | Fatigue Dyspnoea Hypotension Chills Lymphadenopathy Dizziness Infusion related reaction Rash Back pain Arthralgia Asthenia Pleural effusion Cardiac failure congestive Decreased appetite Weight decreased |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sachpekidis, C.; Jackson, D.B.; Soldatos, T.G. Radioimmunotherapy in Non-Hodgkin’s Lymphoma: Retrospective Adverse Event Profiling of Zevalin and Bexxar. Pharmaceuticals 2019, 12, 141. https://doi.org/10.3390/ph12040141
Sachpekidis C, Jackson DB, Soldatos TG. Radioimmunotherapy in Non-Hodgkin’s Lymphoma: Retrospective Adverse Event Profiling of Zevalin and Bexxar. Pharmaceuticals. 2019; 12(4):141. https://doi.org/10.3390/ph12040141
Chicago/Turabian StyleSachpekidis, Christos, David B. Jackson, and Theodoros G. Soldatos. 2019. "Radioimmunotherapy in Non-Hodgkin’s Lymphoma: Retrospective Adverse Event Profiling of Zevalin and Bexxar" Pharmaceuticals 12, no. 4: 141. https://doi.org/10.3390/ph12040141
APA StyleSachpekidis, C., Jackson, D. B., & Soldatos, T. G. (2019). Radioimmunotherapy in Non-Hodgkin’s Lymphoma: Retrospective Adverse Event Profiling of Zevalin and Bexxar. Pharmaceuticals, 12(4), 141. https://doi.org/10.3390/ph12040141






