Wearable Hardware Design for the Internet of Medical Things (IoMT)
Abstract
:1. Introduction
2. Background and Factors to Consider
2.1. Sample Biomedical Signals
2.1.1. Photoplethysmogram (PPG)
2.1.2. Electromyography (EMG)
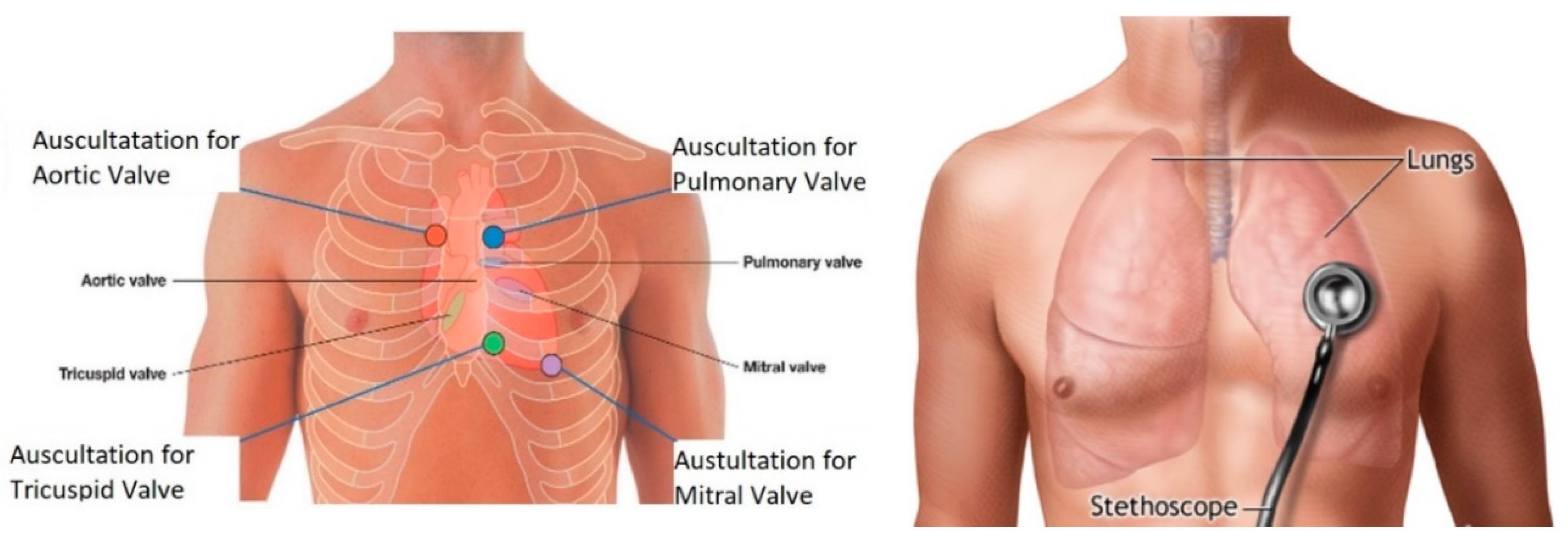
2.1.3. Auscultation of Body Sounds
2.1.4. Gait Analysis
2.2. Important Distinction: Medical versus Non-Medical Wearables
2.3. Factors to Consider
2.3.1. Four Design Factors
2.3.2. On-Chip and Edge Computing
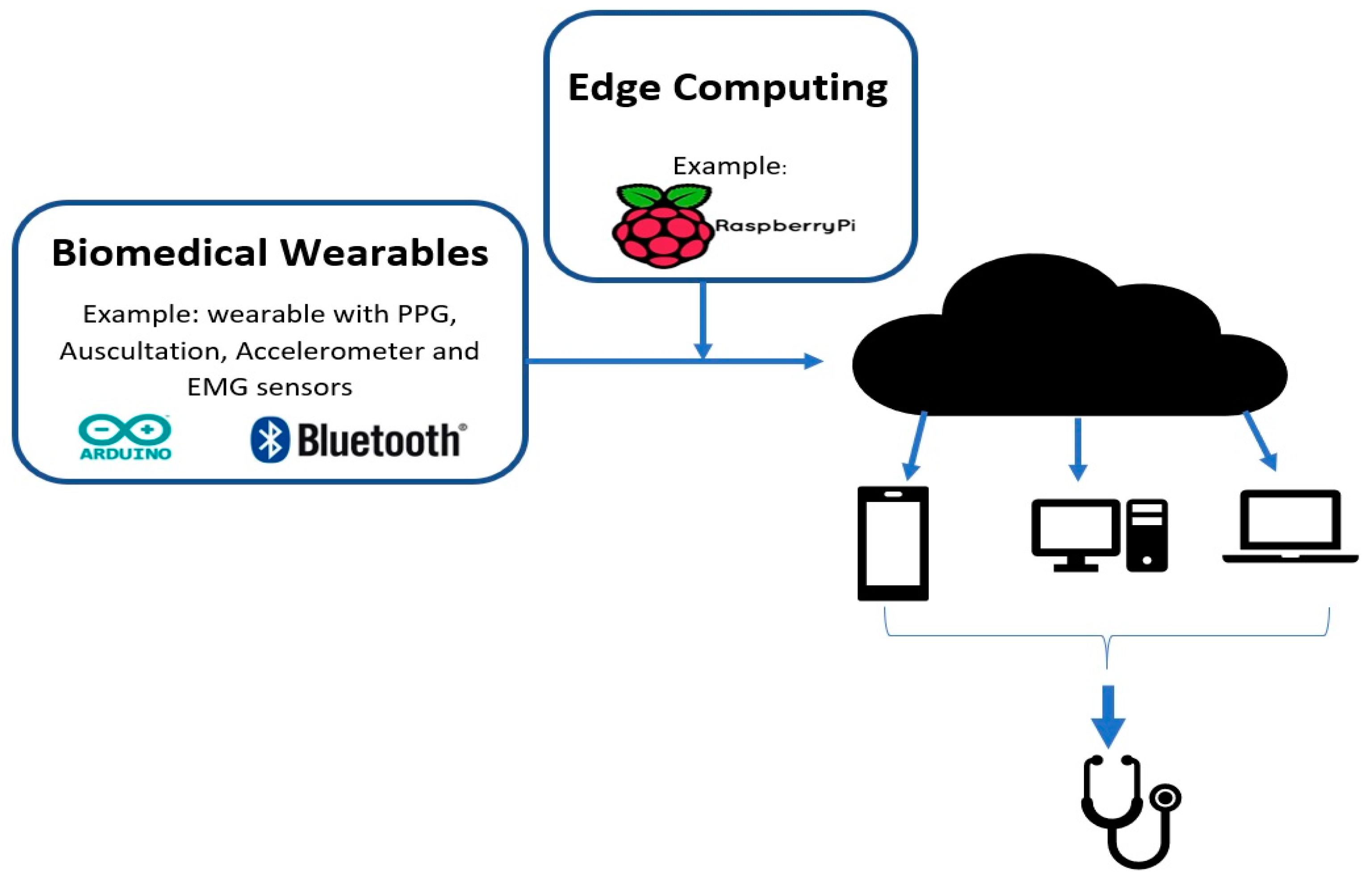
3. Biomedical Wearables and Their Components
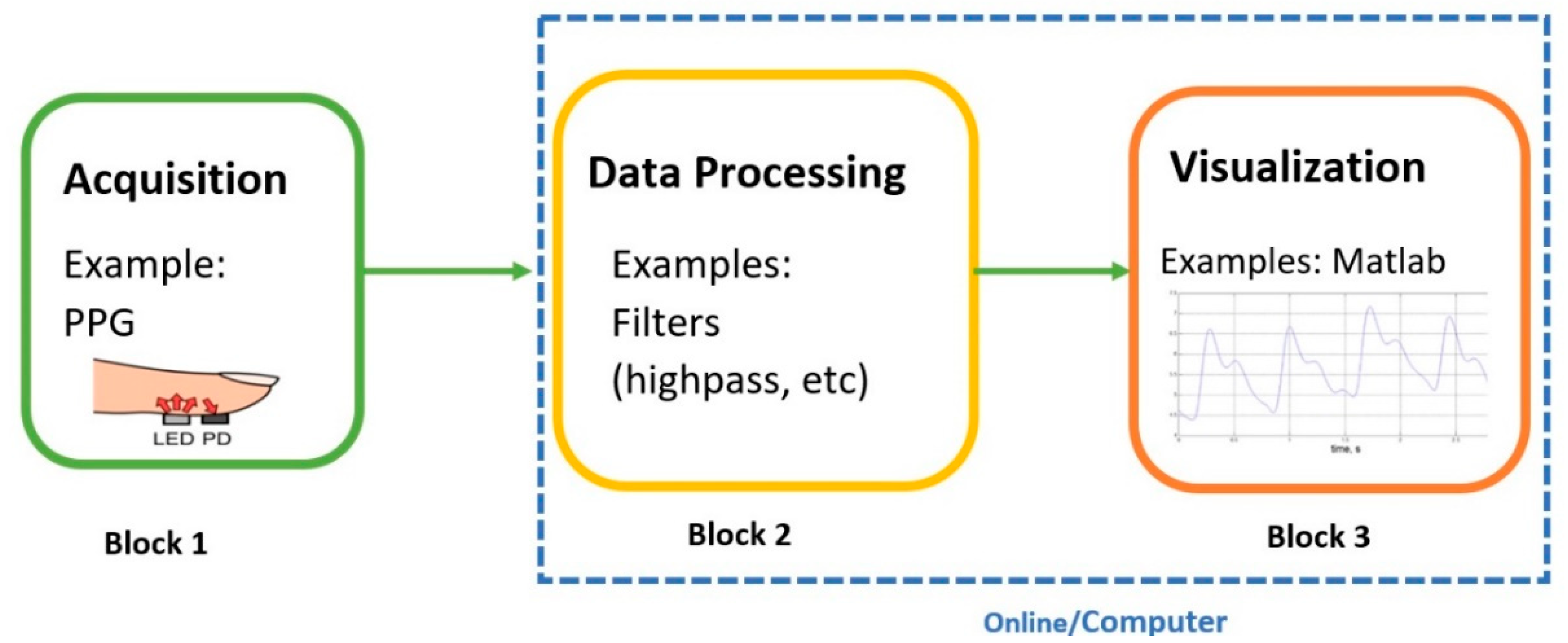
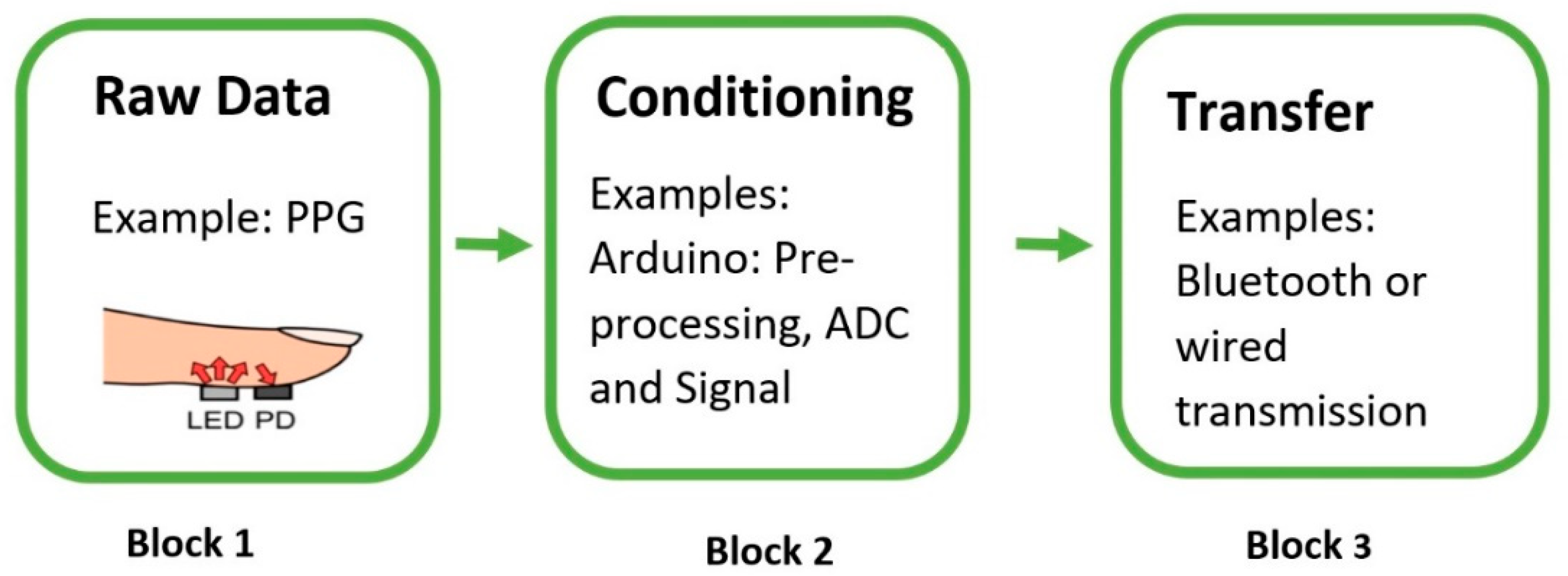
3.1. Allocation of Hardware Design
3.2. Hardware Requirements and Methods
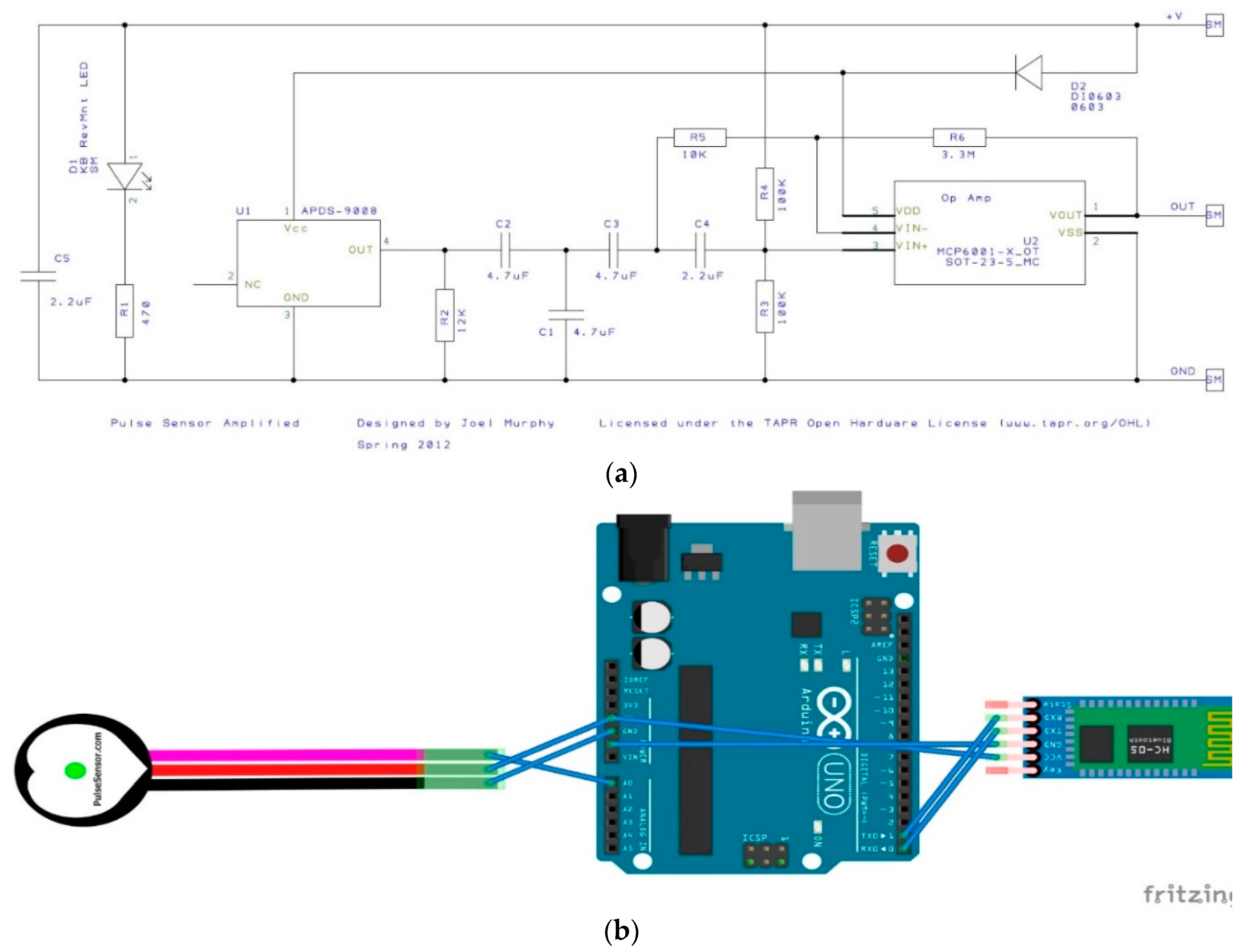
3.2.1. PPG Sensor Description and Bioinstrumentation
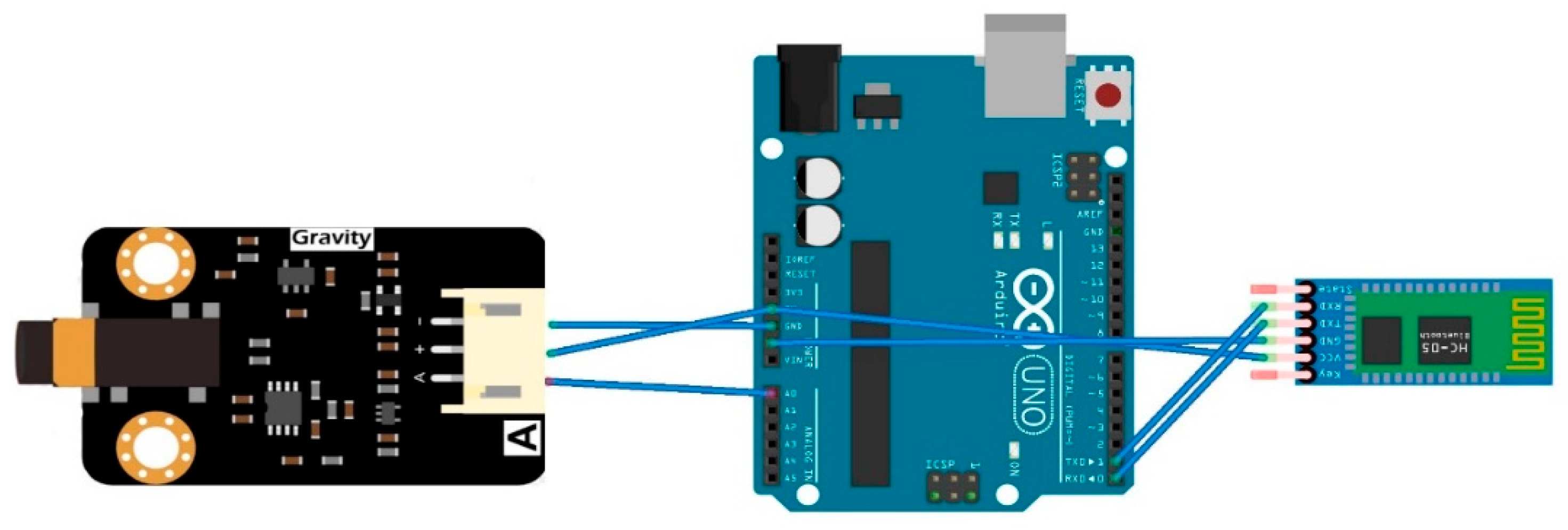
3.2.2. EMG Sensor Requirements and Description
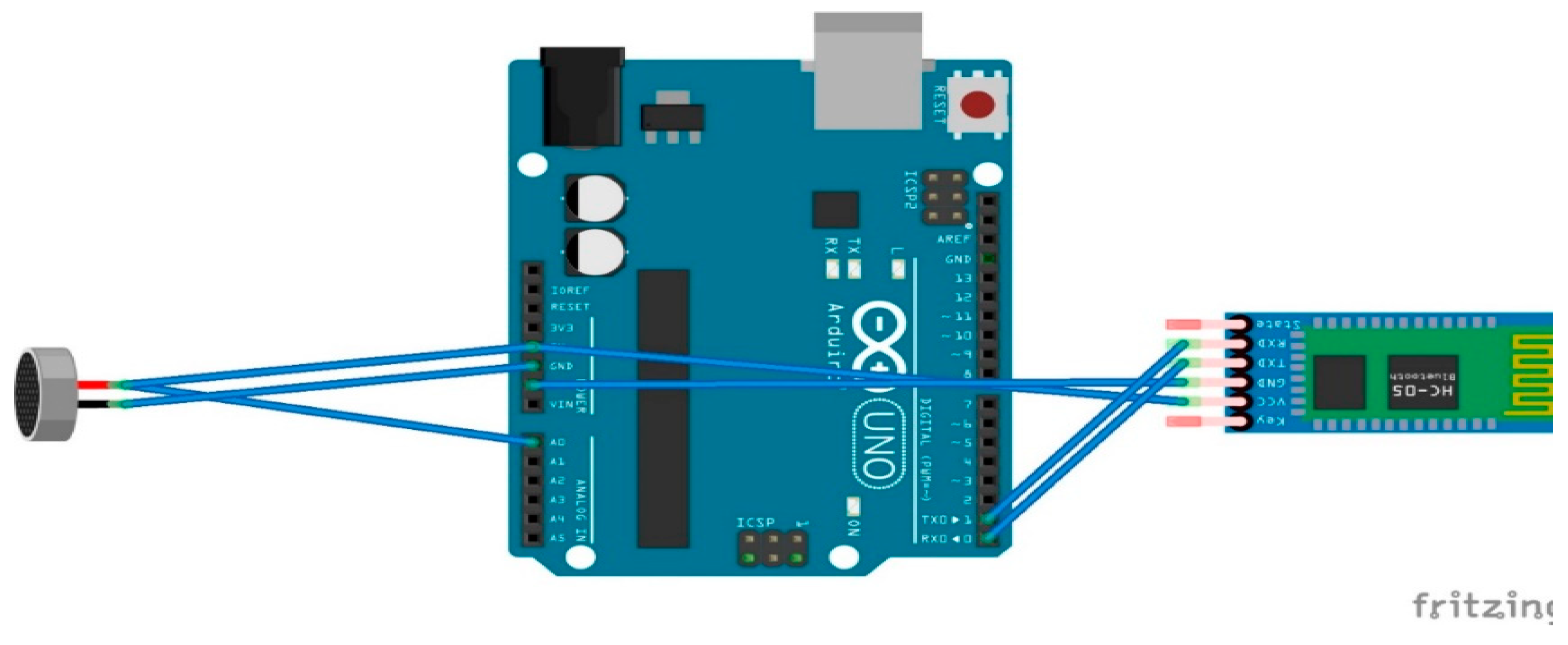
3.2.3. Microphone Requirement and Description
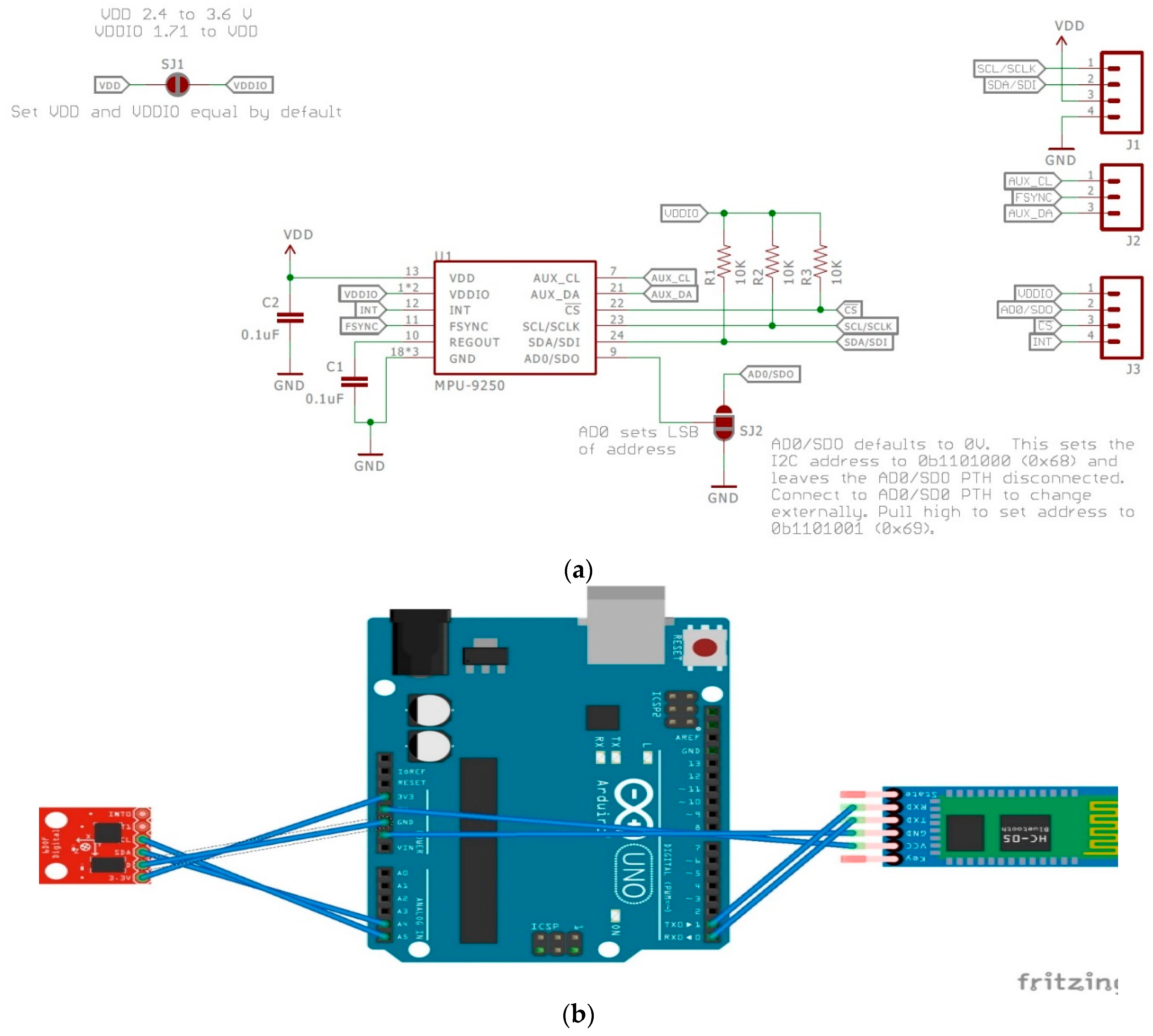
3.2.4. IMU Requirements and Description
3.2.5. Arduino and Raspberry Pi Description
4. Analysis/Discussion
4.1. Hardware Analysis
4.2. Discussion
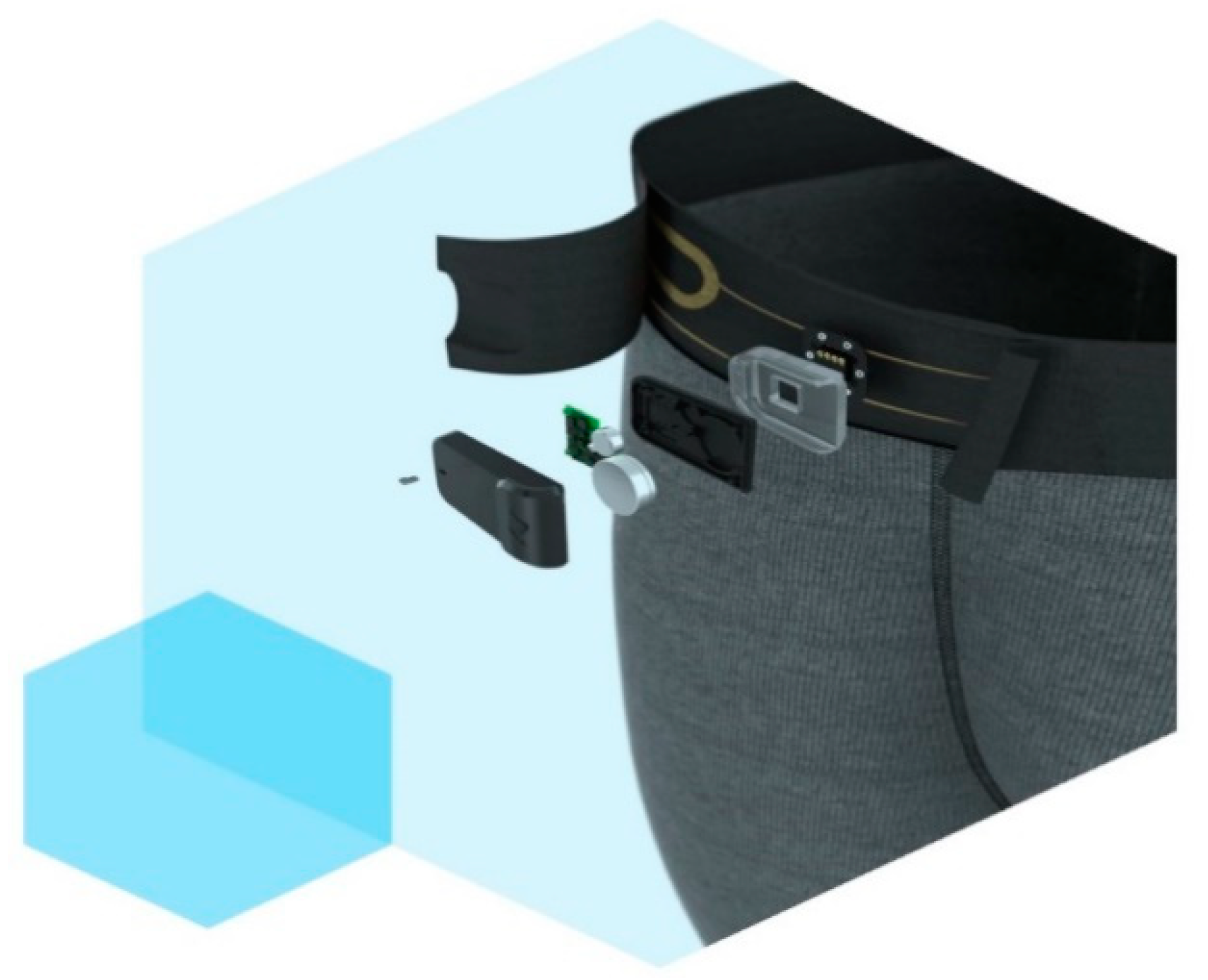
5. Textile Wearables
5.1. Electronic Textiles
5.2. Current Electronic Textiles in the Market
Funding
Acknowledgments
Conflicts of Interest
References
- Ray, S.; Park, J.; Bhunia, S. Guest Editors’ Introduction: Wearables, Implants and Internet of Things. IEEE Trans. Multi-Scale Comput. Syst. 2015, 1, 60–61. [Google Scholar] [CrossRef]
- Ometov, A.; Sergey, B.; Joona, K.; Jarmo, H.; Sergery, A.; Koucheryavy, Y. Facilitating the Delegation of Use for Private Devices in the Era of the Internet of Wearable Things. IEEE Internet Things J. 2017, 4. [Google Scholar] [CrossRef]
- Haghi, M.; Thurow, K.; Stoll, R. Wearable Devices in Medical Internet of Things: Scientific Research and Commercially Available Devices. Healthc. Inform. Res. 2017, 23, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Wei, J. How Wearables Intersect with the Cloud and the Internet of Things: Considerations for the developers of wearables. IEEE Consum. Electron. Mag. 2014, 3, 53–56. [Google Scholar] [CrossRef]
- Athavale, Y.; Krishnan, S. Biosignal Monitoring Using Wearables: Observations and Opportunities. Biomed. Signal Process. Control 2017, 38, 22–33. [Google Scholar] [CrossRef]
- Arduino. Available online: https://www.arduino.cc/ (accessed on 5 March 2018).
- Raspberry PI Zero W. Available online: https://www.raspberrypi.org/products/raspberry-pi-zero-w/ (accessed on 10 April 2015).
- Mayat, U.; Qureshi, F.; Ahmed, S.; Athavale, Y.; Krishnan, S. Towards a low-cost point-of-care screening platform for electronic auscultation of vital body sounds. IEEE Can. Int. Humanit. Technol. Converence 2017, 1–5. [Google Scholar] [CrossRef]
- Government of Canada. “Medical Devices”. Available online: https://www.canada.ca/en/health-canada/services/drugs-health-products/reports-publications/medical-devices.html (accessed on 24 April 2018).
- Telemedicine Market Worth USD 78.82 Billion by 2020. Cision PR Newswire. Available online: https://www.prnewswire.com/news-releases/telemedicine-market-worth-usd-7882-billion-by-2020-674057123.html (accessed on 29 March 2018).
- Zhang, X.; Li, M. Prototype on Interoperability of Internet of Things Gateway Oreiented to Telehealth and Telemedicine. Energy Procedia 2011, 13, 8276–8284. [Google Scholar] [CrossRef]
- Bagha, S.; Shaw, L. A Real Time Analysis of PPG Signal for Measurement of SpO2 and Pulse Rate. Int. J. Comput. Appl. 2011, 26. [Google Scholar] [CrossRef]
- Antonsson, E.K.; Mann, R.W. The Frequency Content of Gait. J. Biomech. 1985, 18, 39–41. [Google Scholar] [CrossRef]
- Ghamari, M.; Soltapur, C.; Cabrera, S.; Romero, R.; Martinek, R.; Nazeran, H. Design and Prototyping of a wristband-type Wireless Photoplethysmographic device for Heart Rate Variability Signal Analysis. IEEE Eng. Med. Biol. Soc. 2016, 467–4970. [Google Scholar] [CrossRef]
- Gwo-Jia, J.; Aripriharta, G.-J. The PPG Physiological Signal for Heart Rate Variability Analysis. Wirel. Pers. Commun. 2017, 97, 5229–5276. [Google Scholar]
- Yang, D.; Zhu, J.; Zhy, P. SpO2 and heart rate measurment with wearable watch based PPG. IET Int. Conf. Biomed. Image Signal Process. 2015. [Google Scholar] [CrossRef]
- Dubey, H.; Kumaresan, R.; Mankodiya, K. Harmonic Sum-based method for heart rate estimation using PPG signals affected with motion artifacts. J. Ambient Intell. Humaniz. Comput. 2018, 9, 137–150. [Google Scholar] [CrossRef]
- iHeart. Available online: https://goiheart.com/ (accessed on 21 October 2018).
- Palladino, V. How Wearable Heart Rate Sensors Work—Which One Is Best for You. Available online: https://arstechnica.com/gadgets/2017/04/how-wearable-heart-rate-monitors-work-and-which-is-best-for-you/ (accessed on 21 October 2018).
- Milosevic, B.; Benatti, S.; Farella, E. Design Challenges for Wearable EMG Applications. In Proceedings of the Design, Automation & Test in Europe Conference & Exhibition (DATE), Lausanne, Switzerland, 27–31 March 2017. [Google Scholar]
- Tavakoli, M.; Benussi, C.; Lopes, P.; Osorio, A. Robust hand gesture recognition with a double channel surface EMG wearable armband and SVM classifier. Biomed. Signal Process. Control 2018, 46, 121–130. [Google Scholar] [CrossRef]
- Myo Armband. Available online: https://www.myo.com/ (accessed on 21 October 2018).
- Abdollapur, M.; Ghaffari, A.; Chiasi, S.; Mollakazme, J. Detection of pathological heart sounds. Physiol. Meas. 2017, 38, 1616–1630. [Google Scholar] [CrossRef] [PubMed]
- Voin, V.; Oskouian, R.; Loukas, M.; Tubbs, S. Auscultation of the Heart: The Basics with Anotomical Correlation. Clin. Anat. 2017, 30, 58–60. [Google Scholar] [CrossRef] [PubMed]
- Bohadana, A.; Izbicki, G.; Kraman, S. Fundamentals of Lung Auscultation. N. Engl. J. Med. 2014, 370, 744–751. [Google Scholar] [CrossRef] [PubMed]
- Surface Anatomy of the Heart. Duke Medicine. Available online: https://web.duke.edu/anatomy/Lab03/Lab4_preLab.html (accessed on 18 October 2018).
- Breath Sounds. MedlinePluse. Available online: https://medlineplus.gov/ency/article/007535.htm (accessed on 17 October 2018).
- Fortune, E.; Lugade, V.; Morrow, M.; Kaufman, K. Validity of Using Tri-Axial Accelerometers to Measure Human Movement—Part II: Step counts at a Wide Range of Gait Velocities. Med. Eng. Phys. 2014, 36, 659–669. [Google Scholar] [CrossRef] [PubMed]
- Moov Now. Available online: https://welcome.moov.cc/ (accessed on 20 October 2018).
- May, M.; McFadden, A. What is a Wearable Device? Lifestyle Device vs. Medical Device. In Regulatory Focus; Regularity Affairs Professionals Society: Rockville, MD, USA, 2017; Available online: https://ctsi.wakehealth.edu/Portals/0/Human%20Research%20Unsecure/IDE_What-is-a-Wearable-Device.pdf?ver=2017-01-24-121015-290 (accessed on 6 July 2018).
- Shi, W.; Cao, J.; Zhang, Q.; Li, Y.; Xu, L. Edge Computing: Vision and Challenges. IEEE Internet Things J. 2016, 3, 637–646. [Google Scholar] [CrossRef]
- Toshiyo, T.; Yuka, M.; Masaki, S.; Masaki, Y. Wearable Photoplethysmographic Sensors—Past and Present. Electronics 2014, 3, 282–302. [Google Scholar] [CrossRef]
- Electret Microphone. Available online: https://challengeelectronics.com/downloads/microphones/omni_directional/CEM-C9745JAD462P2.54R.pdf (accessed on 23 March 2018).
- MPU-9250 Product Specification. Available online: https://www.invensense.com/wp-content/uploads/2015/02/PS-MPU-9250A-01-v1.1.pdf (accessed on 28 March 2018).
- Analog EMG Sensor. Available online: https://www.dfrobot.com/wiki/index.php/Analog_EMG_Sensor_by_OYMotion_SKU:SEN0240 (accessed on 24 March 2018).
- HC-05 Bluethooth Module. Available online: http://www.electronicaestudio.com/docs/istd016A.pdf (accessed on 2 March 2018).
- Pulse Sensor. Available online: https://pulsesensor.com/ (accessed on 24 March 2018).
- MPU-9250 Hookup Guide. Available online: https://learn.sparkfun.com/tutorials/mpu-9250-hookup-guide (accessed on 10 July 2018).
- Donald, U.; Fontelo, P. Prototype telepathology solutions that use the Raspberry Pi and mobile devices. In Proceedings of the 2017 IEEE Global Humanitarian Technology Conference (GHTC), San Jose, CA, USA, 19–22 October 2017. [Google Scholar] [CrossRef]
- Solutions for Personalized Healthcare. Available online: http://www.finlandhealth.fi/-/solution-for-personalized-healthcare (accessed on 5 July 2018).
- Digital Health. Available online: http://www.finlandhealth.fi/-/digital-health (accessed on 6 July 2018).
- Pretz, K. Before Wearables Can Be Used for Health Care Monitoring, These Issues Must Be Addressed. The Institute: The IEEE News Source. Available online: http://theinstitute.ieee.org/technology-topics/life-sciences/before-wearables-can-be-used-for-health-care-monito (accessed on 25 August 2018).
- Feng, X.; Onafeso, B.; Liu, E. Investigating Big Data Healthcare Security Issues with Raspberry Pi. In Proceedings of the IEEE International conference on Computer and Information Technology, Liverpool, UK, 26–28 October 2015. [Google Scholar]
- Syrotuck, N. The 5 Sensors Your Wearable Needs—Are You Missing Any? Available online: https://www.mddionline.com/5-sensors-your-wearable-needs%E2%80%94are-you-missing-any (accessed on 6 July 2018).
- Krishnan, S.; Athavale, Y. Trends in biomedical signal feature extraction. Biomed. Signal Process. Control 2018, 43, 41–63. [Google Scholar] [CrossRef]
- Weng, W.; Chen, P.; He, S.; Sun, X.; Peng, H. Smart Electronic Textiles. Angew. Chem. Int. Ed. 2016, 55, 6140–6169. [Google Scholar] [CrossRef] [PubMed]
- Khalilabad, M.; Yazdanshenas, M. Fabricating Electroconductive cotton textiles using graphene. Carbohydr. Polym. 2013, 96, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Skiin: A Myant Company. Available online: https://skiin.com/products/ (accessed on 27 June 2018).
- Dion, G.; Anday, E.; Dandekar, K. On the Use of Knitted Antennas and Inductively Coupled RFID-Tags for Wearable Applications. IEEE Trans. Biomed. Circuits Syst. 2016, 10, 1047–1057. [Google Scholar] [CrossRef]





| Signal | Frequency Range (Hz) |
|---|---|
| PPG | 0.5–5 1 |
| EMG | 50–150 1 |
| Cardiac Auscultation | 20–420 1 |
| Gait Analysis | 0–15 1 |
| Device | Operating Voltage | Inbuilt ADC | Supply Current | Output Voltage Range | Cost 2 (CAD) |
|---|---|---|---|---|---|
| Pulse Sensor (SEN-11574) | 3–5.5 V | N/A 1 | 3–4 mA | 0.3–5 V | 24.95 |
| EMG Sensor (SEN-0240 | 3.3–5.5 V | N/A | 20 mA | 0–3 V | 50.03 |
| Electret microphone (CEM-C9745JAD462P2.54R) | 1–10 V | N/A | 0.5 mA | ≤10 V | 0.95 |
| MPU9250 | 2.4–3.6 V | 16 bit | 450 μA–3.2 mA | 2.4–3.6 V | 14.95 |
| Arduino Uno | 6–20 V | 10 bit | 20–50 mA | N/A | 35.95 |
| HC-05 Bluetooth Module | 3.3–5 V | 8 bit transfer | ≈35 mA | N/A | 11.99 |
| Raspberry PI Zero W | 5 V | 11–17 bit | 1.2 A | N/A | 28.95 |
| Batteries | N/A | N/A | 400–600 mAH | 9 V | 1.47 |
| (a) | |||||||
| Pulse Sensor | EMG Sensor | ||||||
| Sensor | Black (ground) | Red (power) | Purple (Signal) | −(ground) | +(power) | A (Signal) | |
| Arduino | GND | 5 V | A0 (Analog in) | GND | 5 V | A0 (Analog in) | |
| (b) | |||||||
| Electret Microphone | IMU | ||||||
| Sensor | Black (ground) | Red (power) | Red (power) | VDD | GND | SCL | SDA |
| Arduino | GND | 5 V | A0 (Analog in) | 5 V | GND | A5 (Analog in) | A4 (Analog in) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qureshi, F.; Krishnan, S. Wearable Hardware Design for the Internet of Medical Things (IoMT). Sensors 2018, 18, 3812. https://doi.org/10.3390/s18113812
Qureshi F, Krishnan S. Wearable Hardware Design for the Internet of Medical Things (IoMT). Sensors. 2018; 18(11):3812. https://doi.org/10.3390/s18113812
Chicago/Turabian StyleQureshi, Fayez, and Sridhar Krishnan. 2018. "Wearable Hardware Design for the Internet of Medical Things (IoMT)" Sensors 18, no. 11: 3812. https://doi.org/10.3390/s18113812
APA StyleQureshi, F., & Krishnan, S. (2018). Wearable Hardware Design for the Internet of Medical Things (IoMT). Sensors, 18(11), 3812. https://doi.org/10.3390/s18113812




