VitiCanopy: A Free Computer App to Estimate Canopy Vigor and Porosity for Grapevine
Abstract
:1. Introduction
2. Materials and Methods
2.1. VitiCanopy Development
2.2. Algorithms Used in the VitiCanopy
2.3. Description of VitiCanopy
2.4. Settings Menu
2.5. New Measurement Menu
2.6. Outputs
2.7. Beta Testing of VitiCanopy
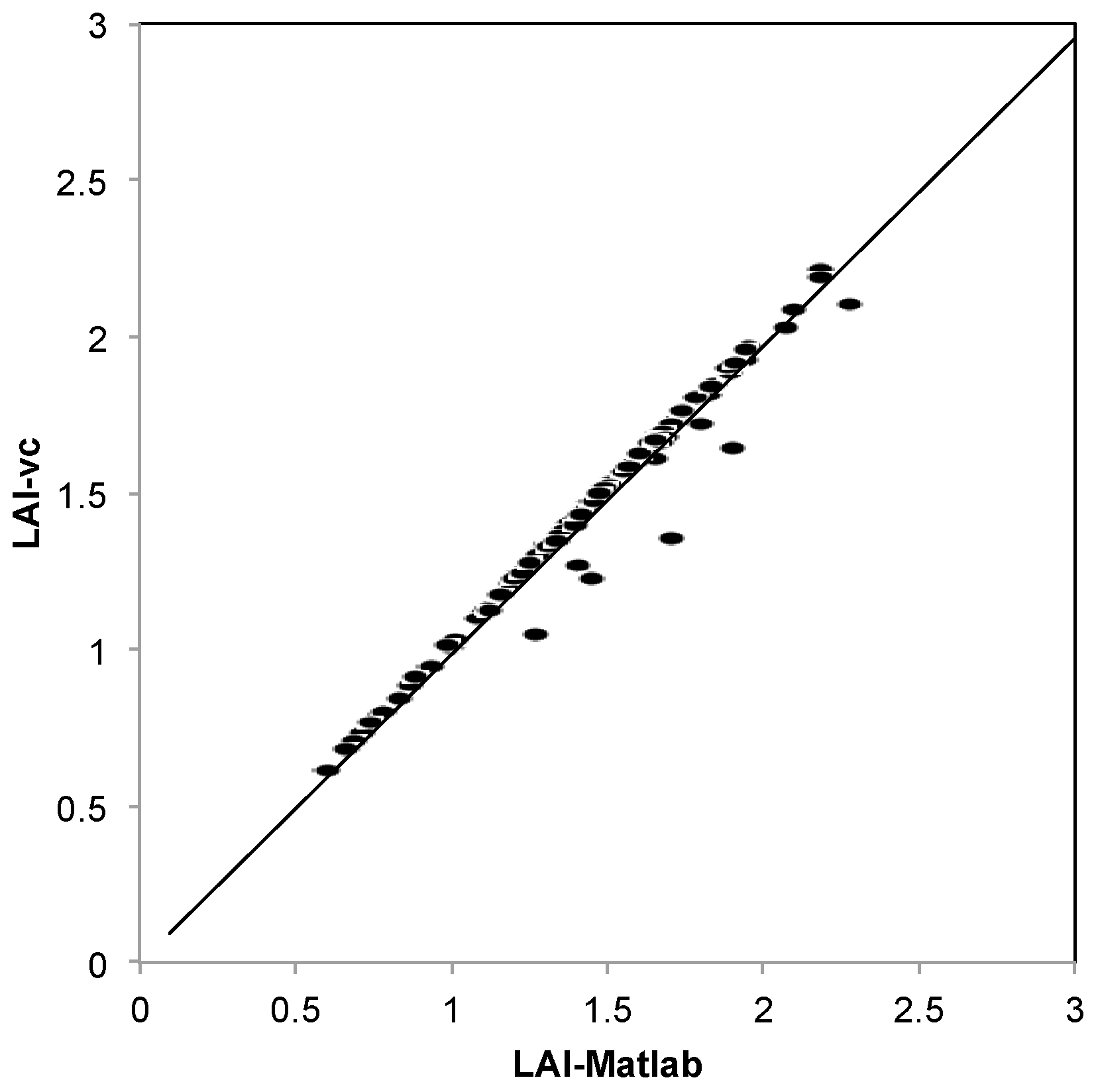
2.7.1. VitiCanopy vs. Matlab
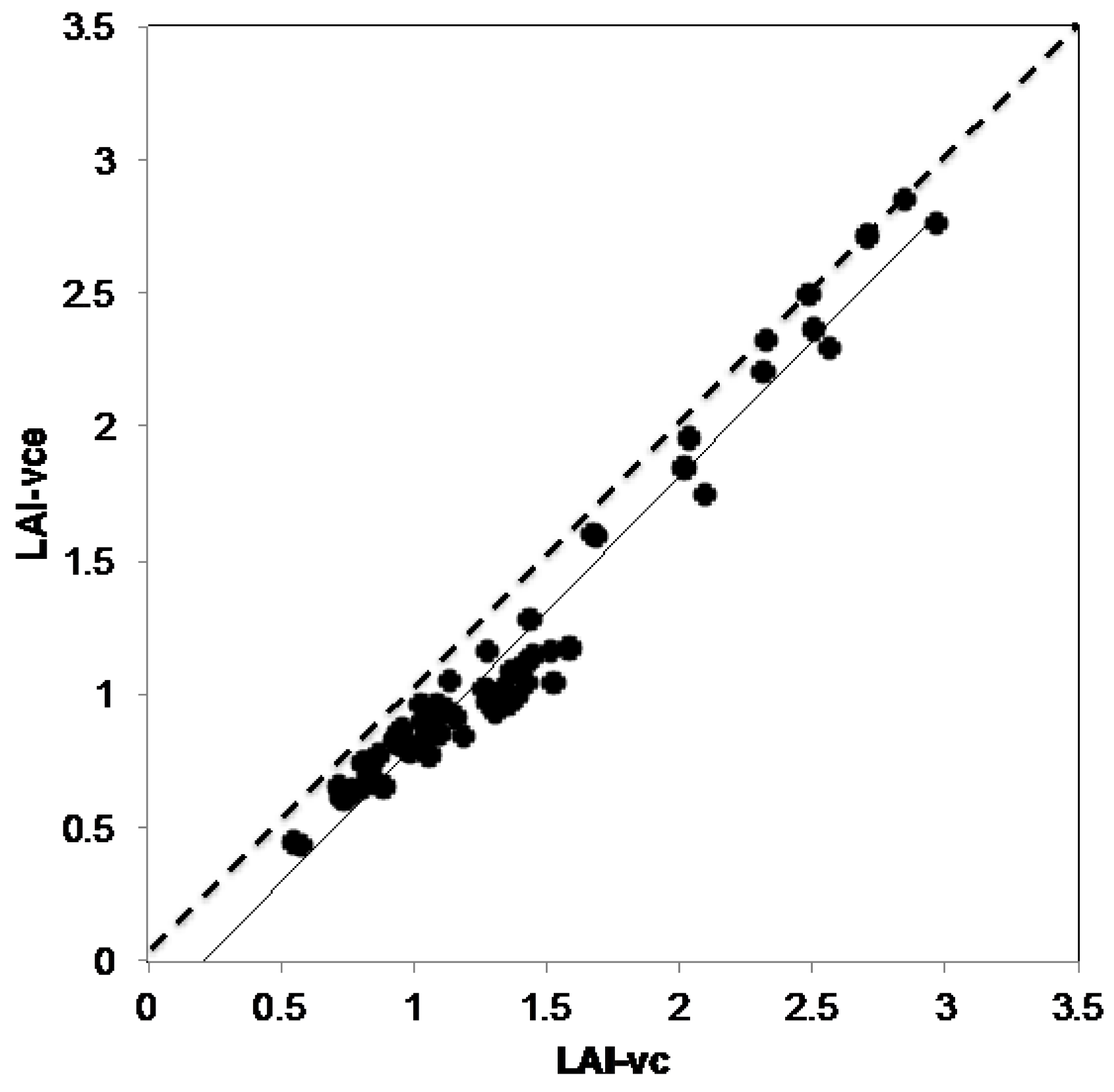
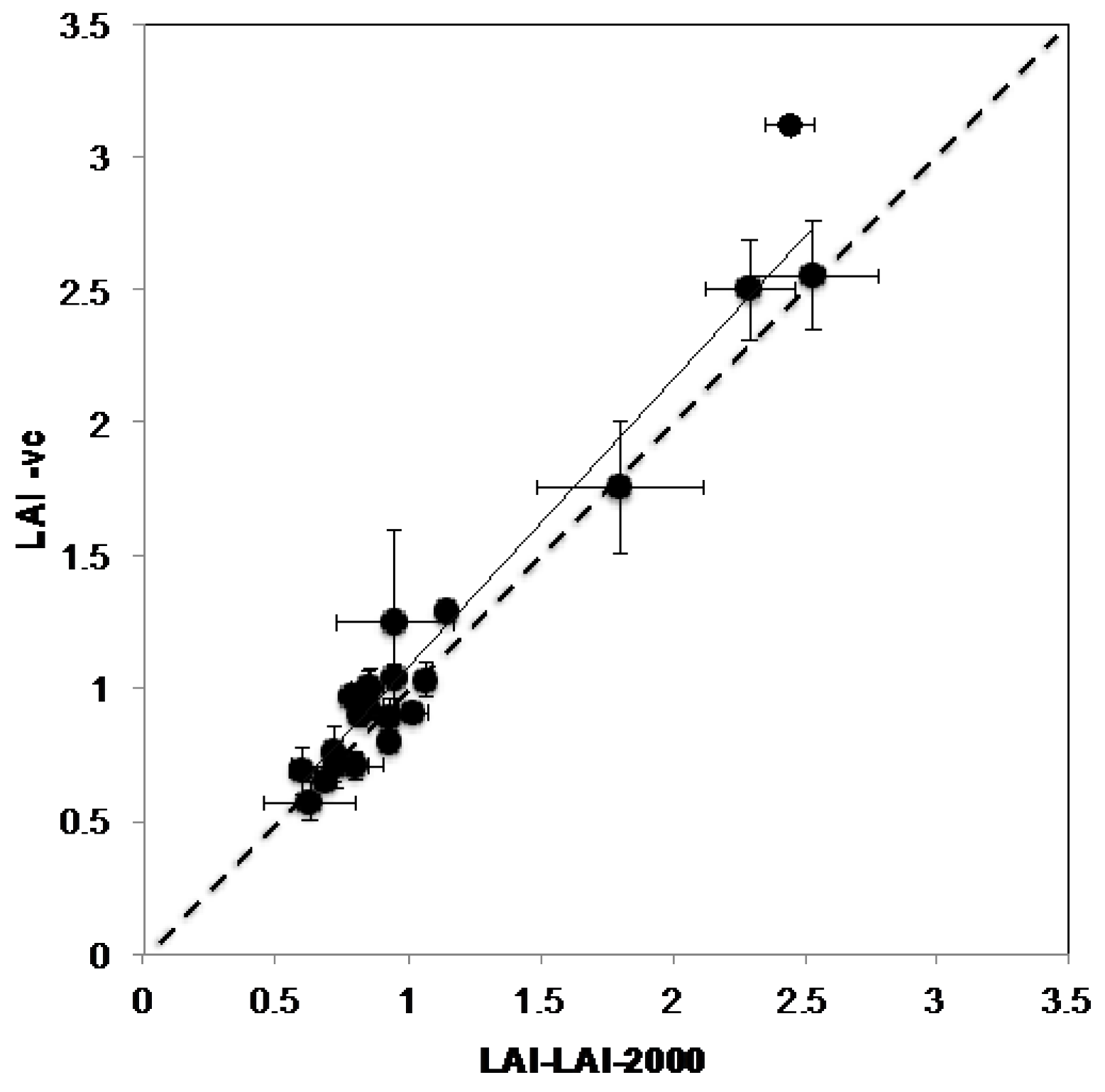
2.7.2. VitiCanopy vs. LAI-2000
2.7.3. VitiCanopy vs. Planimetric and Gravimetric Method
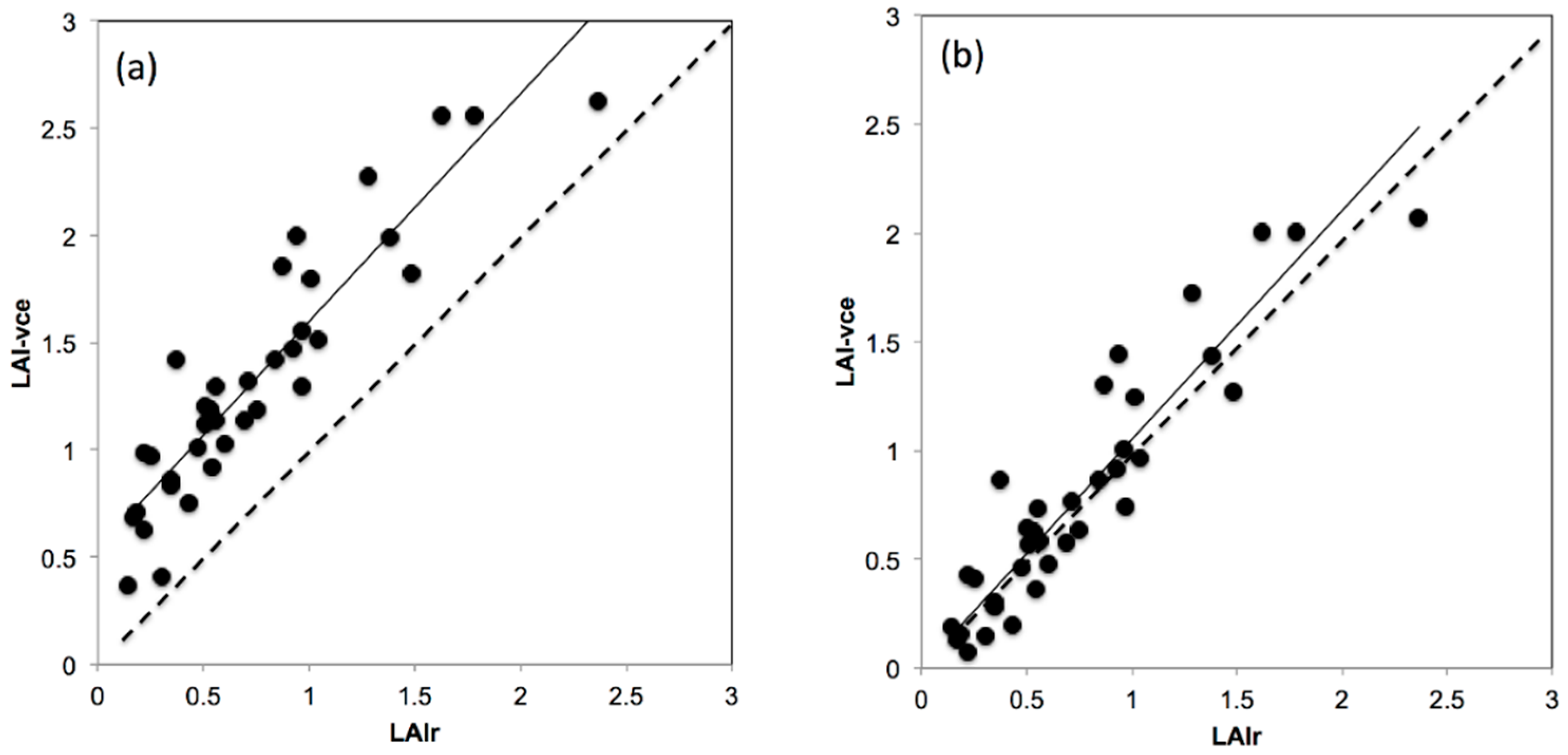
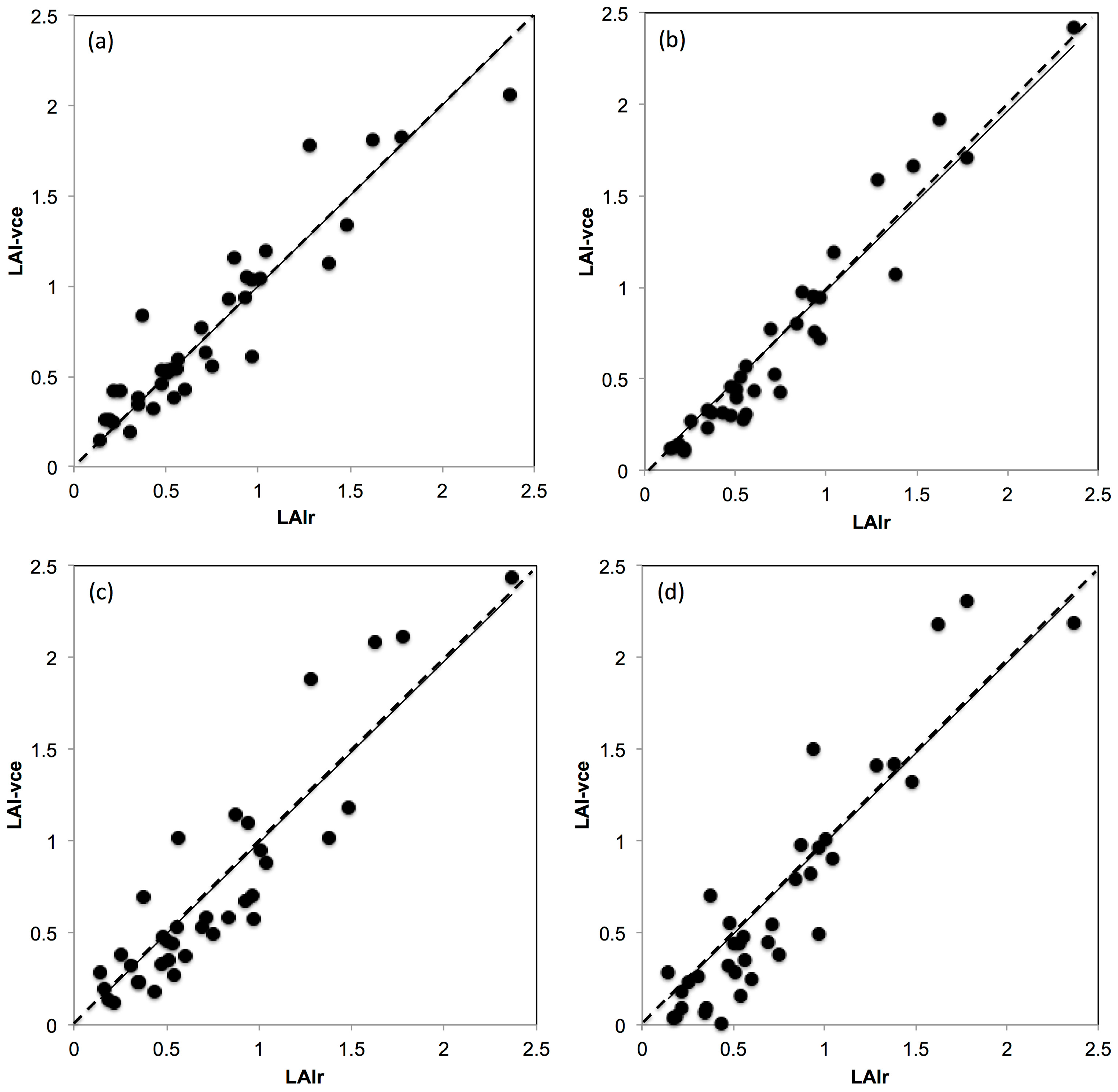
2.7.4. Comparison between Real k vs. Fixed and Modeled k to Estimate LAI
- (i)
- Fixed value (k = 0.7) as reported in literature [16];
- (ii)
- Real k (kr) calculated by using LAIr and inverting Equation (7).
- (iii)
- Proxy of k obtained by using the Lowess method to smooth the data combined with the curve-fitting tool from Matlab. The model was constructed using fc (Equation (2)) and Φ (Equation (3)) as follows: kSmooth = malowess (fc, Φ), which smooths scattered data in fc and Φ. The default window size is 5% of the length of ff.
- (iv)
- Modeled k obtained comparing kr with large gaps (lg), based on the fact that larger gaps will result in higher light transmission to the image acquisition point.
- (v)
- The ratio between luminance, obtained automatically from the metadata of each image (I), using a customized code written in Matlab and the maximum luminance obtained from sky images (Io = 12).
2.8. Statistical Analysis
3. Results
3.1. VitiCanopy vs. MatLab
3.2. VitiCanopy vs. Licor-2000
3.3. VitiCanopy vs. Planimetric and Gravimetric Method
3.4. Comparison between Real k vs. Fixed and Modelled k to Estimate LAI
4. Discussion
4.1. VitiCanopy vs. MatLab
4.2. VitiCanopy vs. Licor-2000
4.3. VitiCanopy and the Effect of Distance of Images from Grapevine Cordons
4.4. VitiCanopy Performance and Cordon Contribution Corrections
4.5. The Effect of Variable k on PAI Using the VitiCanopy App
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dokoozlian, N.K.; Kliewer, W.M. The light environment within grapevine canopies. II. Influence of leaf area density on fruit zone light environment and some canopy assessment parameters. Am. J. Enol. Vitic. 1995, 46, 219–226. [Google Scholar]
- Dokoozlian, N.K.; Kliewer, W.M. The light environment within grapevine canopies. I. Description and seasonal changes during fruit development. Am. J. Enol. Vitic. 1995, 46, 209–218. [Google Scholar]
- Smart, R.E. Principles of grapevine canopy microclimate manipulation with implications for yield and quality. A review. Am. J. Enol. Vitic. 1985, 36, 230–239. [Google Scholar]
- Watson, D.J. Comparative physiological studies on the growth of field crops: I. Variation in net assimilation rate and leaf area between species and varieties, and within and between years. Ann. Bot. 1947, 11, 41–76. [Google Scholar] [CrossRef]
- Bréda, N.J.J. Ground-based measurements of leaf area index: A review of methods, instruments and current controversies. J. Exp. Bot. 2003, 54, 2403–2417. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, S.; Palmer, A.R.; Taylor, D.; Zeppel, M.; Whitley, R.; Eamus, D. An automated procedure for estimating the leaf area index (LAI) of woodland ecosystems using digital imagery, matlab programming and its application to an examination of the relationship between remotely sensed and field measurements of LAI. Funct. Plant Biol. 2008, 35, 1070–1079. [Google Scholar] [CrossRef]
- Jonckheere, I.; Fleck, S.; Nackaerts, K.; Muys, B.; Coppin, P.; Weiss, M.; Baret, F. Review of methods for in situ leaf area index determination: Part I. Theories, sensors and hemispherical photography. Agric. Forest Meteorol. 2004, 121, 19–35. [Google Scholar] [CrossRef]
- Cutini, A.; Matteucci, G.; Mugnozza, G.S. Estimation of leaf area index with the Li-cor LAI 2000 in deciduous forests. Forest Ecol. Manag. 1998, 105, 55–65. [Google Scholar] [CrossRef]
- Weiss, M.; Baret, F.; Smith, G.J.; Jonckheere, I.; Coppin, P. Review of methods for in situ leaf area index (LAI) determination: Part II. Estimation of lai, errors and sampling. Agric. Forest Meteorol. 2004, 121, 37–53. [Google Scholar] [CrossRef]
- Aschonitis, V.G.; Papamichail, D.M.; Lithourgidis, A.; Fano, E.A. Estimation of leaf area index and foliage area index of rice using an indirect gravimetric method. Commun. Soil Sci. Plant Anal. 2014, 45, 1726–1740. [Google Scholar] [CrossRef]
- Montero, F.J.; de Juan, J.A.; Cuesta, A.; Brasa, A. Nondestructive methods to estimate leaf area in vitis vinifera L. HortScience 2000, 35, 696–698. [Google Scholar]
- Poblete-Echeverría, C.A.; Ortega-Farias, S.O. Evaluation of single and dual crop coefficients over a drip-irrigated merlot vineyard (Vitis vinifera L.) using combined measurements of sap flow sensors and an eddy covariance system. Aust. J. Grape Wine Res. 2013, 19, 249–260. [Google Scholar] [CrossRef]
- Pekin, B.; Macfarlane, C. Measurement of crown cover and leaf area index using digital cover photography and its application to remote sensing. Remote Sens. 2009, 1, 1298–1320. [Google Scholar] [CrossRef]
- Macfarlane, C.; Arndt, S.K.; Livesley, S.J.; Edgar, A.C.; White, D.A.; Adams, M.A.; Eamus, D. Estimation of leaf area index in eucalypt forest with vertical foliage, using cover and fullframe fisheye photography. Forest Ecol. Manag. 2007, 242, 756–763. [Google Scholar] [CrossRef]
- Macfarlane, C.; Hoffman, M.; Eamus, D.; Kerp, N.; Higginson, S.; McMurtrie, R.; Adams, M. Estimation of leaf area index in eucalypt forest using digital photography. Agric. Forest Meteorol. 2007, 143, 176–188. [Google Scholar] [CrossRef]
- Fuentes, S.; Poblete-Echeverría, C.; Ortega-Farias, S.; Tyerman, S.; De Bei, R. Automated estimation of leaf area index from grapevine canopies using cover photography, video and computational analysis methods. Aust. J. Grape Wine Res. 2014, 20, 465–473. [Google Scholar] [CrossRef]
- Macfarlane, C.; Grigg, A.; Evangelista, C. Estimating forest leaf area using cover and fullframe fisheye photography: Thinking inside the circle. Agric. Forest Meteorol. 2007, 146, 1–12. [Google Scholar] [CrossRef]
- Poblete-Echeverría, C.; Fuentes, S.; Ortega-Farias, S.; Gonzalez-Talice, J.; Yuri, J.A. Digital cover photography for estimating leaf area index (LAI) in apple trees using a variable light extinction coefficient. Sensors 2015, 15, 2860–2872. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, S.; De Bei, R.; Pozo, C.; Tyerman, S. Development of a smartphone application to characterise temporal and spatial canopy architecture and leaf area index for grapevines. Wine Vitic. J. 2012, 6, 56–60. [Google Scholar]
- Confalonieri, R.; Foi, M.; Casa, R.; Aquaro, S.; Tona, E.; Peterle, M.; Boldini, A.; De Carli, G.; Ferrari, A.; Finotto, G.; et al. Development of an app for estimating leaf area index using a smartphone. Trueness and precision determination and comparison with other indirect methods. Comput. Electron. Agric. 2013, 96, 67–74. [Google Scholar] [CrossRef]
- Confalonieri, R.; Francone, C.; Foi, M. The pocketlai smartphone app: An alternative method for leaf area index estimation. In Proceedings of the 7th International Congress on Environmental Modelling and Software, San Diego, CA, USA, 15–19 June 2014.
- Francone, C.; Pagani, V.; Foi, M.; Cappelli, G.; Confalonieri, R. Comparison of leaf area index estimates by ceptometer and pocketlai smart app in canopies with different structures. Field Crops Res. 2014, 155, 38–41. [Google Scholar] [CrossRef]
- Orlando, F.; Movedi, E.; Paleari, L.; Gilardelli, C.; Foi, M.; Dell’Oro, M.; Confalonieri, R. Estimating leaf area index in tree species using the pocketlai smart app. Appl. Veg. Sci. 2015, 18, 716–723. [Google Scholar] [CrossRef]
- De Bei, R.; Hook, J.; Fuentes, S.; Gilliham, M.; Tyerman, S.; Collins, C. Linking canopy architecture to grape quality using the laicanopy app. In Proceedings of the 19th International Symposium GiESCO, Pech Rouge, France, 31 May–5 June 2015.
- De Bei, R.; Fuentes, S.; Petrie, P.; Kidman, C.; Hook, J.; Gilliham, M.; Tyerman, S.; Collins, C. Optimising vine balance in Australian vineyards: Development of a smartphone application to assess leaf area index. In Proceedings of the ASVO Mildura Seminar, Mildura, Victoria, Australia, 24–25 July 2014.
- Mora, M.; Avila, F.; Carrasco-Benavides, M.; Maldonado, G.; Olguín-Cáceres, J.; Fuentes, S. Automated computation of leaf area index from fruit trees using improved image processing algorithms applied to canopy cover digital photograpies. Comput. Electron. Agric. 2016, 123, 195–202. [Google Scholar] [CrossRef]
- LI-COR. Lai-2000 Plant Canopy anAlyzer Operating Manual; LI-COR: Lincoln, NE, USA, 1992. [Google Scholar]
- Walker, J.; Tunstall, B.R. Field Estimation of Foliage Cover in Australian Woody Vegetation; CSIRO, Institute of Biological Resources, Division of Land Use Research: Canberra, Austrialia, 1981. [Google Scholar]
- Viti Adelaide. VitiCanopy. Available online: https://www.youtube.com/watch?v=Eqzmuruubm8 (accessed on 21 March 2016).
- Neumann, H.H.; Den Hartog, G.; Shaw, R.H. Leaf area measurements based on hemispheric photographs and leaf-litter collection in a deciduous forest during autumn leaf-fall. Agric. Forest Meteorol. 1989, 45, 325–345. [Google Scholar] [CrossRef]


| Distance iPad-Cordon (cm) | Cordon Length in the Image (cm) |
|---|---|
| 90 | 70 |
| 80 | 64 |
| 70 | 54 |
| 60 | 46 |
| 50 | 38 |
| Vineyard | LAI-vc | LAI-vce | Ω(0) | |||
|---|---|---|---|---|---|---|
| Average | Range | Average | Range | Average | Range | |
| Hilltops | 0.99 | 0.72–1.44 | 0.85 | 0.57–1.28 | 0.85 | 0.76–0.90 |
| Langhorne Creek | 2.37 | 1.93–2.71 | 2.26 | 1.72–2.66 | 0.95 | 0.90–0.98 |
| Sunraysia | 1.30 | 1.16–1.39 | 0.97 | 0.90–1.04 | 0.75 | 0.70–0.78 |
| Distance iPad-Cordon (cm) | R2 | b | a | SEE | RMSE |
|---|---|---|---|---|---|
| 50 | 0.76 | 1.08 | 0.35 | 0.15 | 0.19 |
| 60 | 0.79 | 0.86 | 0.47 | 0.05 | 0.11 |
| 70 | 0.89 | 1.01 | 0.31 | 0.65 | 0.30 |
| 80 | 0.89 | 1.06 | 0.28 | 0.30 | 0.21 |
| 90 | 0.70 | 1.15 | 0.19 | 0.38 | 0.28 |
| Proxy of k | R2 | b | SEE | RMSE | p Value |
|---|---|---|---|---|---|
| Method (i) | na; 0.86 | na; 1.05 | na; 1.89 | na; 0.24 | na; 1 × 10−5 |
| Method (ii) | na; 0.94 | na; 0.98 | na; 0.71 | na; 0.15 | na; 1 × 10−5 |
| Method (iii) | 0.38; 0.87 | 1.72; 1.00 | 0.61; 1.11 | 0.13; 0.18 | 1 × 10-4; 1 × 10−5 |
| Method (iv) | 0.05; 0.83 | 1 × 10−5; 0.99 | 2.57; 2.04 | 0.27; 0.24 | ns; 1 × 10−5 |
| Method (v) | 0.25; 0.86 | 0.09; 0.99 | 0.21; 2.04 | 0.08; 0.24 | 1 × 10-3; 1 × 10−5 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Bei, R.; Fuentes, S.; Gilliham, M.; Tyerman, S.; Edwards, E.; Bianchini, N.; Smith, J.; Collins, C. VitiCanopy: A Free Computer App to Estimate Canopy Vigor and Porosity for Grapevine. Sensors 2016, 16, 585. https://doi.org/10.3390/s16040585
De Bei R, Fuentes S, Gilliham M, Tyerman S, Edwards E, Bianchini N, Smith J, Collins C. VitiCanopy: A Free Computer App to Estimate Canopy Vigor and Porosity for Grapevine. Sensors. 2016; 16(4):585. https://doi.org/10.3390/s16040585
Chicago/Turabian StyleDe Bei, Roberta, Sigfredo Fuentes, Matthew Gilliham, Steve Tyerman, Everard Edwards, Nicolò Bianchini, Jason Smith, and Cassandra Collins. 2016. "VitiCanopy: A Free Computer App to Estimate Canopy Vigor and Porosity for Grapevine" Sensors 16, no. 4: 585. https://doi.org/10.3390/s16040585
APA StyleDe Bei, R., Fuentes, S., Gilliham, M., Tyerman, S., Edwards, E., Bianchini, N., Smith, J., & Collins, C. (2016). VitiCanopy: A Free Computer App to Estimate Canopy Vigor and Porosity for Grapevine. Sensors, 16(4), 585. https://doi.org/10.3390/s16040585








