What Do the First 597 Global Fungal Red List Assessments Tell Us about the Threat Status of Fungi?
Abstract
:1. Introduction
2. Materials and Methods
3. Results
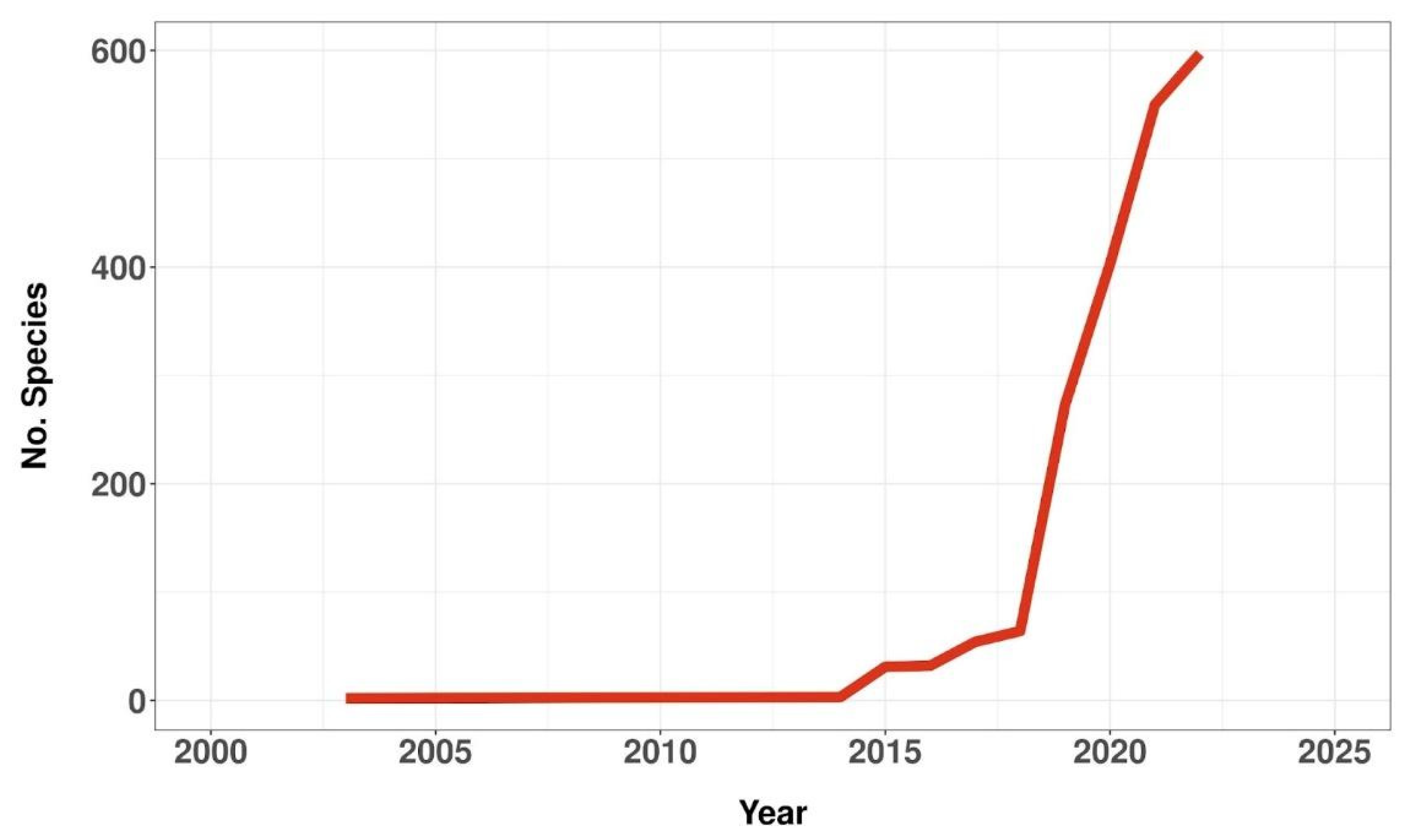
3.1. Assessed Species over Time
Hypocreopsis amplectens—Tea-tree Fingers Critically Endangered
Lepraria lanata—Appalachian Dust Bunnies Endangered
Fomitiporia nubicola Vulnerable
Suillus luteus—Slippery Jack Least Concern
Cortinarius crypticus Data Deficient
3.2. Red List Categories and Declines
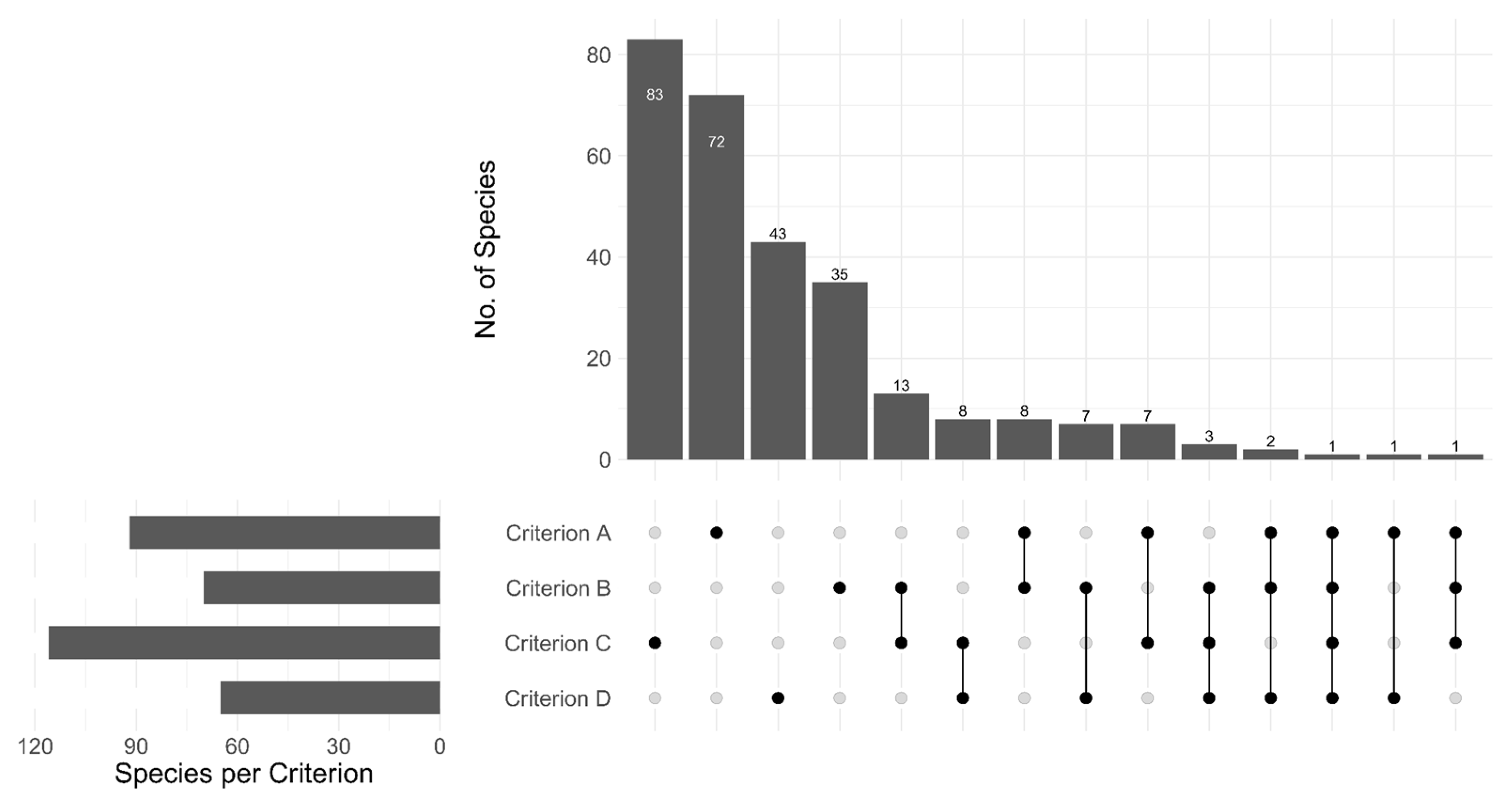
3.3. Application of the Red List Criteria in Assessments of Fungi
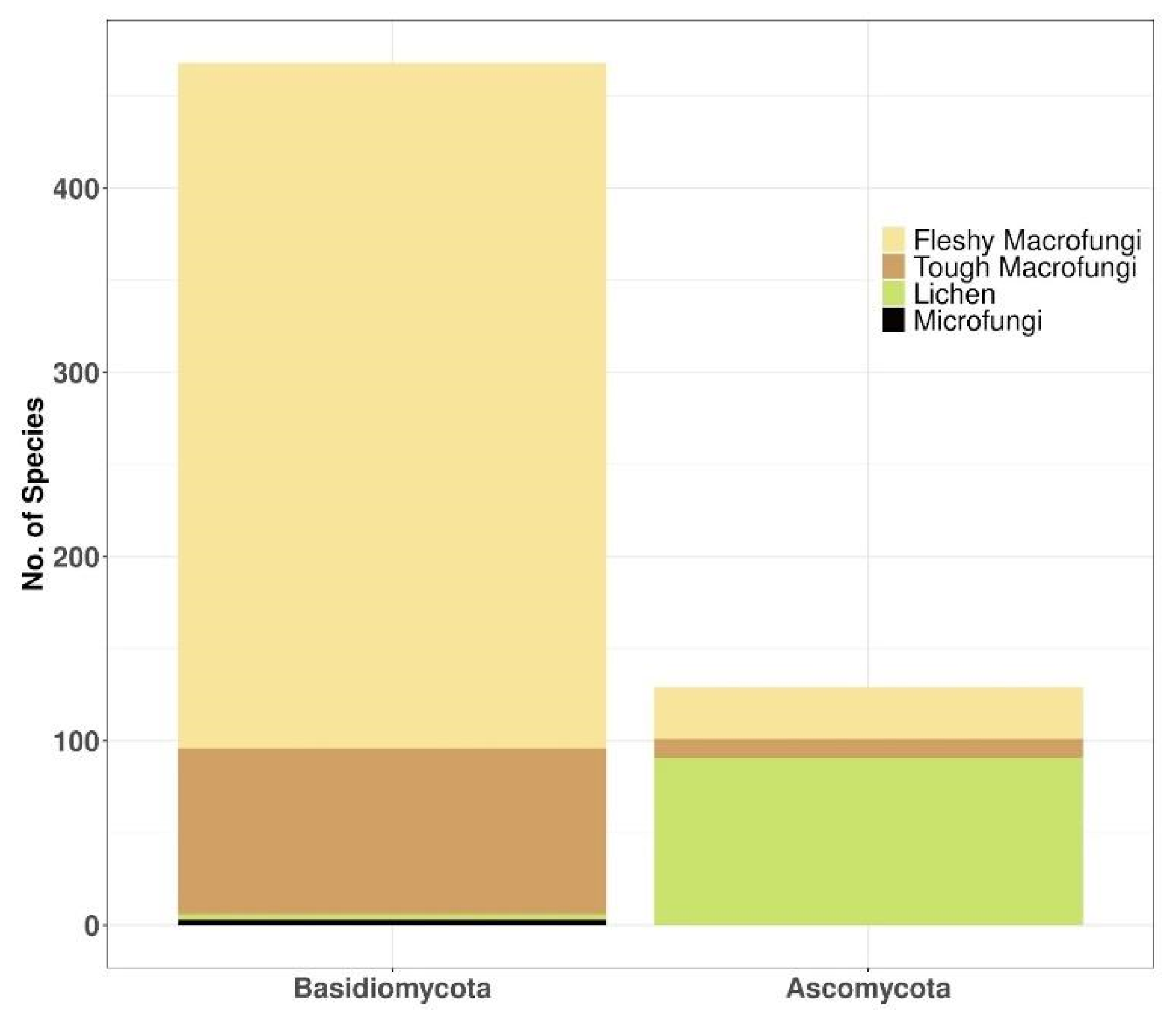
3.4. Taxonomy and Growth Form
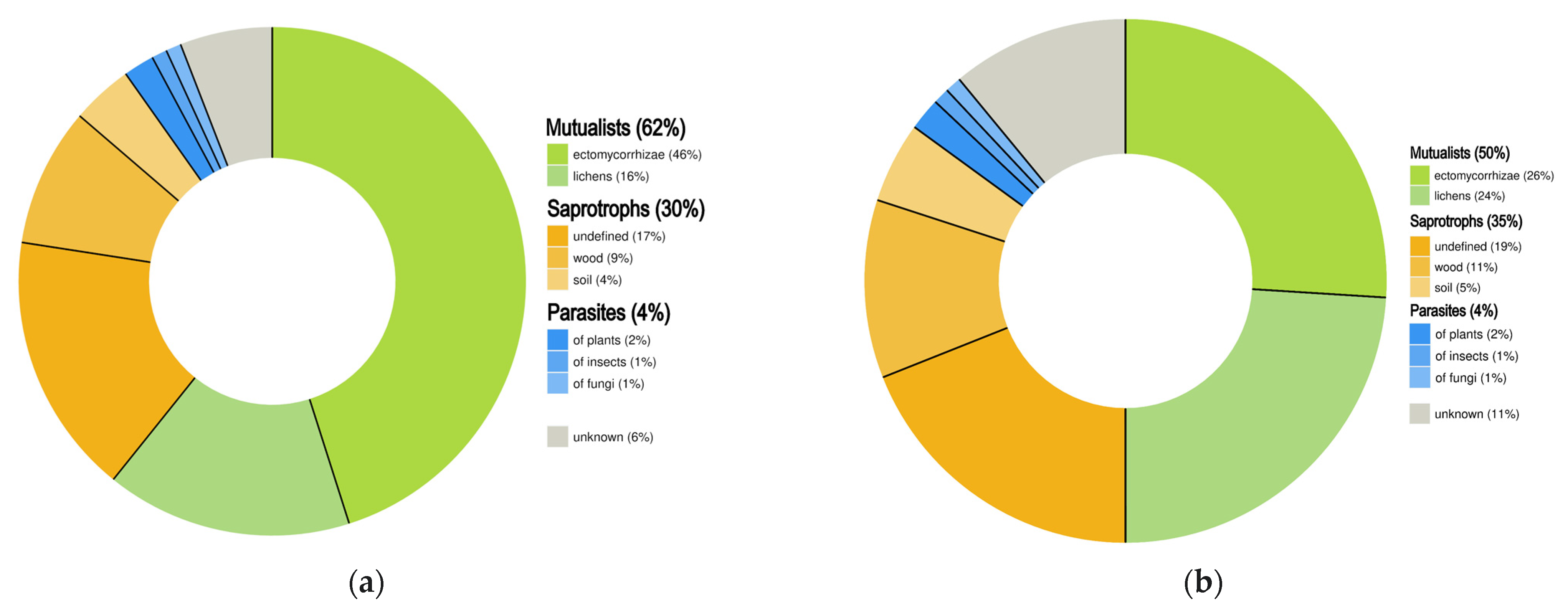
3.5. Trophic Guild
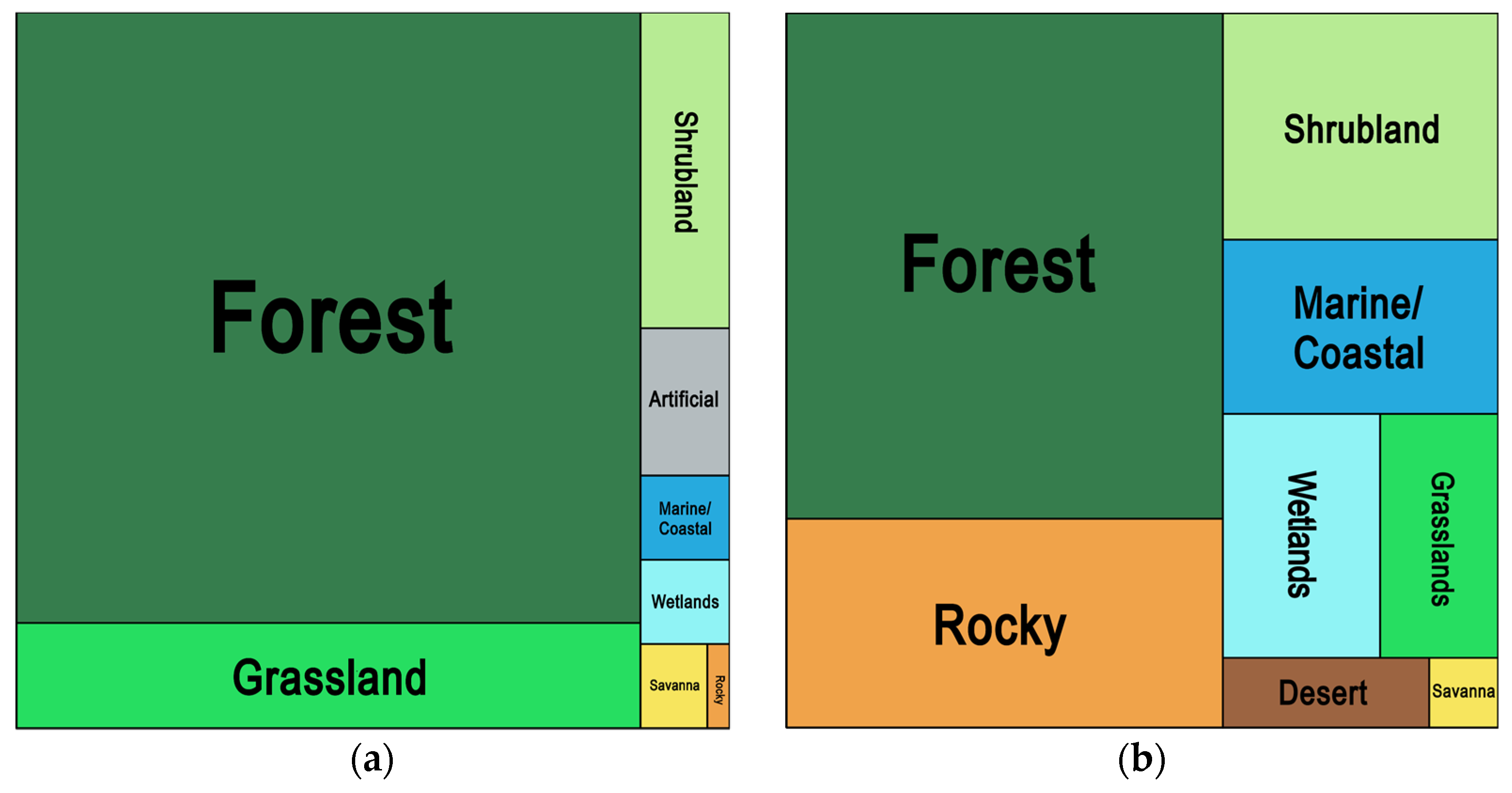
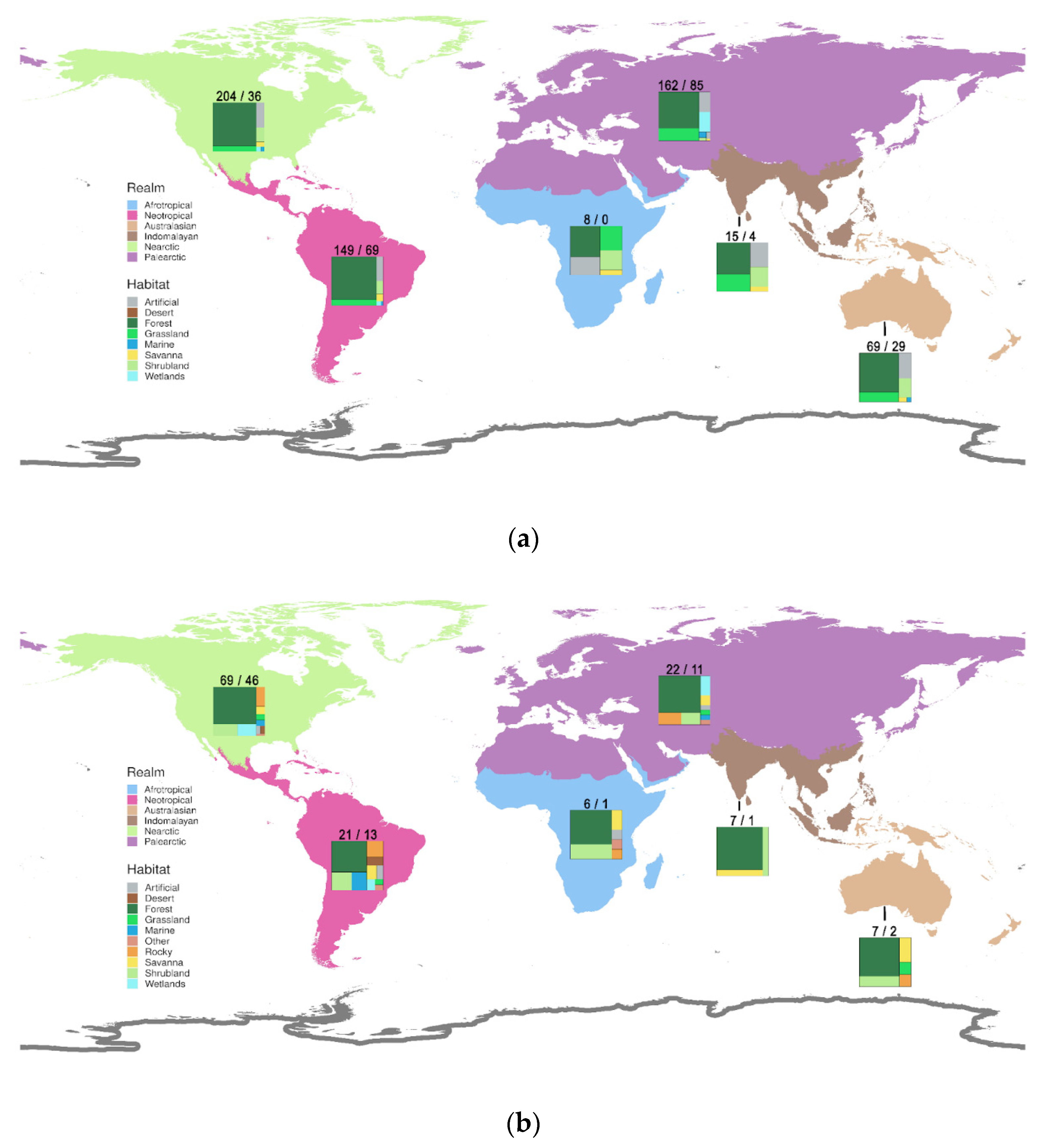
3.6. Geography and Habitats
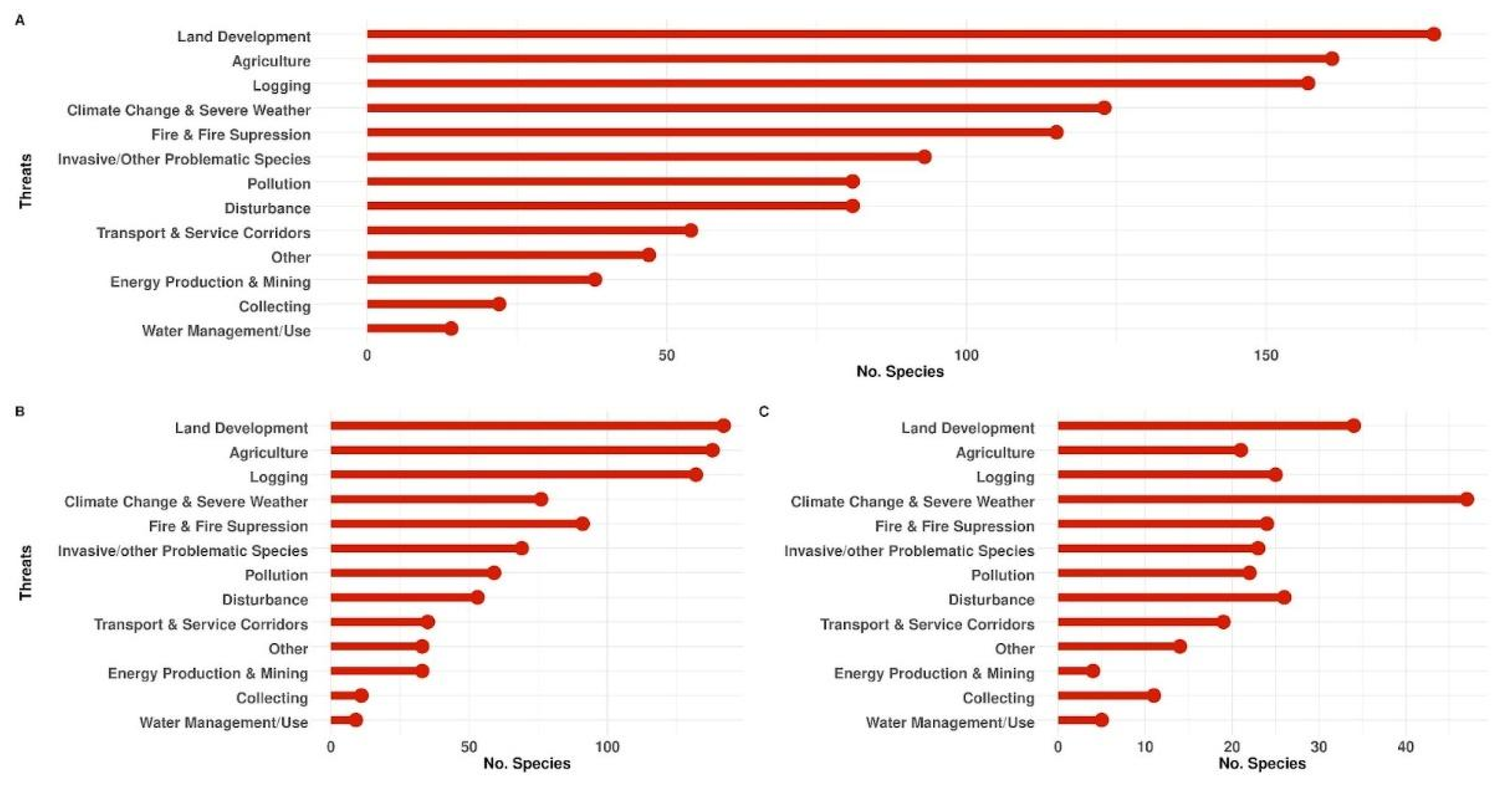
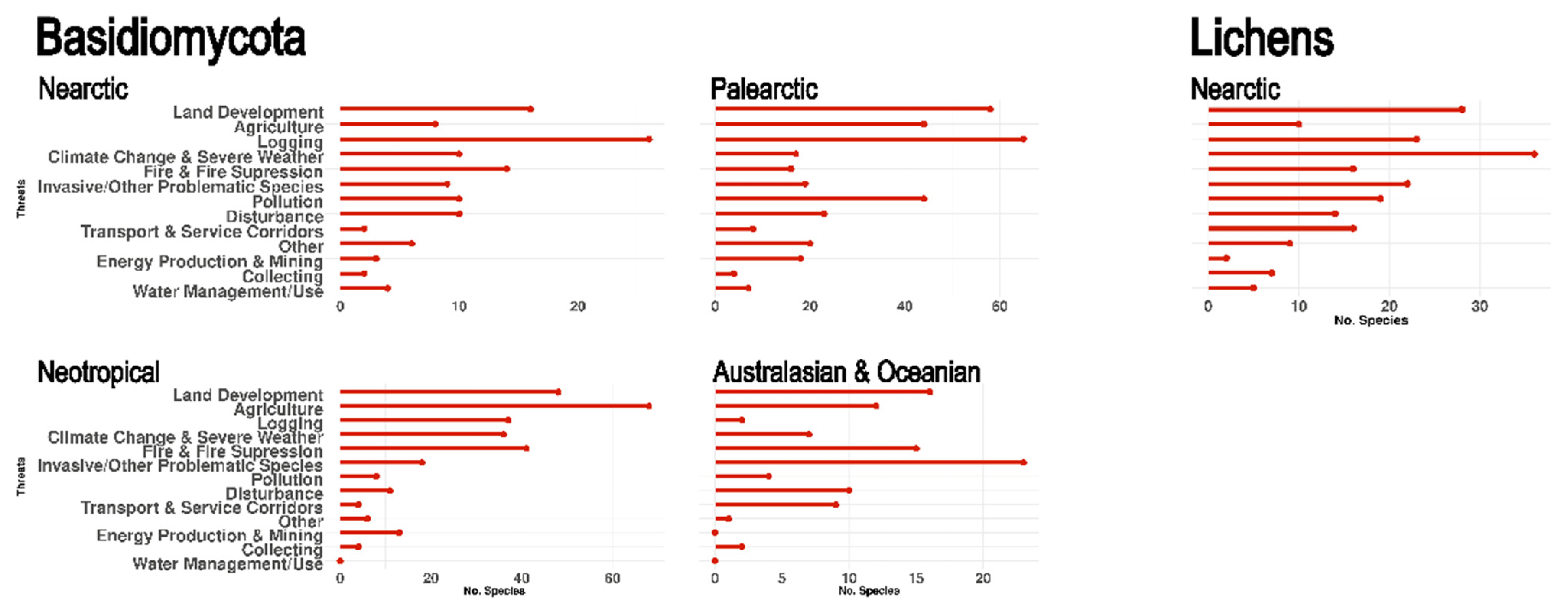
3.7. Threats
3.8. Uses
4. Discussion
4.1. Risk of Extinction and Criteria Used
4.2. Threats
4.3. Gaps
4.4. Future Efforts
4.5. From Red Listing to on-the-Ground Actions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Webster, J.; Weber, R. Introduction to Fungi; Cambridge University Press: Cambridge, UK, 2007; ISBN 1-139-46150-8. [Google Scholar]
- Blackwell, M. The Fungi: 1, 2, 3… 5.1 Million Species? Am. J. Bot. 2011, 98, 426–438. [Google Scholar] [CrossRef] [PubMed]
- Hawksworth, D.L.; Lücking, R. Fungal Diversity Revisited: 2.2 to 3.8 Million Species. Microbiol. Spectr. 2017, 5-4, 10. [Google Scholar]
- Pérez-Izquierdo, L.; Rincón, A.; Lindahl, B.D.; Buée, M. Fungal Community of Forest Soil: Diversity, Functions, and Services. In Forest Microbiology; Elsevier: Amsterdam, The Netherlands, 2021; pp. 231–255. [Google Scholar]
- Prescott, T.; Wong, J.; Panaretou, B.; Boa, E.; Bond, A.; Chowdhury, S.; Davis, L.; Østergaard, L. Useful Fungi. In State of the World’s Fungi 2018; Royal Botanic Gardens, Kew: Richmond, UK, 2018; ISBN 978-1-84246-678-0. [Google Scholar]
- Meyer, V.; Basenko, E.Y.; Benz, J.P.; Braus, G.H.; Caddick, M.X.; Csukai, M.; De Vries, R.P.; Endy, D.; Frisvad, J.C.; Gunde-Cimerman, N. Growing a Circular Economy with Fungal Biotechnology: A White Paper. Fungal Biol. Biotechnol. 2020, 7, 1–23. [Google Scholar] [CrossRef]
- Cheek, M.; Nic Lughadha, E.; Kirk, P.; Lindon, H.; Carretero, J.; Looney, B.; Douglas, B.; Haelewaters, D.; Gaya, E.; Llewellyn, T. New Scientific Discoveries: Plants and Fungi. Plants People Planet 2020, 2, 371–388. [Google Scholar] [CrossRef]
- Baldrian, P.; Větrovský, T.; Lepinay, C.; Kohout, P. High-Throughput Sequencing View on the Magnitude of Global Fungal Diversity. Fungal Divers. 2022, 114, 539–547. [Google Scholar] [CrossRef]
- Ruddiman, W.F. The Anthropocene. Annu. Rev. Earth Planet. Sci. 2013, 41, 45–68. [Google Scholar] [CrossRef]
- Mace, G.M. Drivers of Biodiversity Change. In Trade-Offs in Conservation: Deciding What to Save; John Wiley & Sons: Hoboken, NJ, USA, 2010; pp. 349–364. [Google Scholar]
- Habibullah, M.S.; Din, B.H.; Tan, S.-H.; Zahid, H. Impact of Climate Change on Biodiversity Loss: Global Evidence. Environ. Sci. Pollut. Res. 2022, 29, 1073–1086. [Google Scholar] [CrossRef]
- Chapin Iii, F.S.; Zavaleta, E.S.; Eviner, V.T.; Naylor, R.L.; Vitousek, P.M.; Reynolds, H.L.; Hooper, D.U.; Lavorel, S.; Sala, O.E.; Hobbie, S.E. Consequences of Changing Biodiversity. Nature 2000, 405, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Cardinale, B.J.; Duffy, J.E.; Gonzalez, A.; Hooper, D.U.; Perrings, C.; Venail, P.; Narwani, A.; Mace, G.M.; Tilman, D.; Wardle, D.A. Biodiversity Loss and Its Impact on Humanity. Nature 2012, 486, 59–67. [Google Scholar] [CrossRef]
- Platto, S.; Zhou, J.; Wang, Y.; Wang, H.; Carafoli, E. Biodiversity Loss and COVID-19 Pandemic: The Role of Bats in the Origin and the Spreading of the Disease. Biochem. Biophys. Res. Commun. 2021, 538, 2–13. [Google Scholar] [CrossRef] [PubMed]
- Barnosky, A.D.; Matzke, N.; Tomiya, S.; Wogan, G.O.; Swartz, B.; Quental, T.B.; Marshall, C.; McGuire, J.L.; Lindsey, E.L.; Maguire, K.C. Has the Earth’s Sixth Mass Extinction Already Arrived? Nature 2011, 471, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Dahlberg, A.; Mueller, G.M. Applying IUCN Red-Listing Criteria for Assessing and Reporting on the Conservation Status of Fungal Species. Fungal Ecol. 2011, 4, 147–162. [Google Scholar] [CrossRef]
- Tedersoo, L.; Bahram, M.; Põlme, S.; Kõljalg, U.; Yorou, N.S.; Wijesundera, R.; Ruiz, L.V.; Vasco-Palacios, A.M.; Thu, P.Q.; Suija, A. Global Diversity and Geography of Soil Fungi. Science 2014, 346, 1256688. [Google Scholar] [CrossRef] [PubMed]
- Geml, J.; Arnold, A.E.; Semenova-Nelsen, T.A.; Nouhra, E.R.; Drechsler-Santos, E.R.; Góes-Neto, A.; Morgado, L.N.; Ódor, P.; Hegyi, B.; Oriol, G.; et al. Community Dynamics of Soil-Borne Fungal Communities along Elevation Gradients in Neotropical and Palaeotropical Forests. Mol. Ecol. 2022, 31, 2044–2060. [Google Scholar] [CrossRef] [PubMed]
- IUCN Standards and Petitions Committee. Guidelines for Using the IUCN Red List Categories and Criteria. Version 15.1; IUCN Standards and Petitions Committee: Gland, Switzerland, 2022. [Google Scholar]
- Lughadha, E.N.; Bachman, S.P.; Leão, T.C.; Forest, F.; Halley, J.M.; Moat, J.; Acedo, C.; Bacon, K.L.; Brewer, R.F.; Gâteblé, G.; et al. Extinction Risk and Threats to Plants and Fungi. Plants People Planet 2020, 2, 389–408. [Google Scholar] [CrossRef]
- Williams, B.A.; Watson, J.E.M.; Butchart, S.H.M.; Ward, M.; Brooks, T.M.; Butt, N.; Bolam, F.C.; Stuart, S.N.; Mair, L.; McGowan, P.J.K.; et al. A Robust Goal Is Needed for Species in the Post-2020 Global Biodiversity Framework. Conserv. Lett. 2021, 14, e12778. [Google Scholar] [CrossRef]
- Betts, J.; Young, R.P.; Hilton-Taylor, C.; Hoffmann, M.; Rodríguez, J.P.; Stuart, S.N.; Milner-Gulland, E.J. A Framework for Evaluating the Impact of the IUCN Red List of Threatened Species. Conserv. Biol. 2020, 34, 632–643. [Google Scholar] [CrossRef] [PubMed]
- United Nations. Transforming Our World: The 2030 Agenda for Sustainable Development; United Nations: New York, NY, USA, 2015. [Google Scholar]
- IUCN. The IUCN Red List of Threatened Species. Version 2022-1. Available online: https://www.iucnredlist.org (accessed on 21 July 2022).
- Dahlberg, A.; Genney, D.R.; Heilmann-Clausen, J. Developing a Comprehensive Strategy for Fungal Conservation in Europe: Current Status and Future Needs. Fungal Ecol. 2010, 3, 50–64. [Google Scholar] [CrossRef]
- Ainsworth, M.; Canteiro, C.; Dahlberg, A.; Douglas, B.; Furci, G.; Minter, D.; Mueller, G.M.; Scheidegger, C.; Senn-Irlet, B.; Wilkins, T.; et al. Conservation of Fungi. In State of the World’s Fungi 2018; Royal Botanic Gardens, Kew: Richmond, UK, 2018; ISBN 978-1-84246-678-0. [Google Scholar]
- Mueller, G.M.; Dahlberg, A.; Krikorev, M. Bringing Fungi into the Conservation Conversation: The Global Fungal Red List Initiative. Fungal Conserv. 2014, 4, 12–16. [Google Scholar]
- Nguyen, N.H.; Song, Z.; Bates, S.T.; Branco, S.; Tedersoo, L.; Menke, J.; Schilling, J.S.; Kennedy, P.G. FUNGuild: An Open Annotation Tool for Parsing Fungal Community Datasets by Ecological Guild. Fungal Ecol. 2016, 20, 241–248. [Google Scholar] [CrossRef]
- Van Rossum, G.; Drake, F.L. Python/C Api Manual-Python 3; CreateSpace: Scotts Valley, CA, USA, 2009; ISBN 1-4414-1273-5. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing, Version 4.2.0; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Wickham, H. Programming with Ggplot2. In ggplot2; Springer: Berling/Heidelberg, Germany, 2016; pp. 241–253. [Google Scholar]
- Krassowski, M. Krassowski/Complex-Upset. Available online: http://doi.org/10.5281/zenodo.3700590 (accessed on 21 July 2022).
- The GIMP Development Team. GIMP—GNU Image Manipulation Program; The Gimp Development Team: Charlotte, NC, USA, 2019. [Google Scholar]
- Hochkirch, A.; Samways, M.J.; Gerlach, J.; Böhm, M.; Williams, P.; Cardoso, P.; Cumberlidge, N.; Stephenson, P.J.; Seddon, M.B.; Clausnitzer, V.; et al. A Strategy for the next Decade to Address Data Deficiency in Neglected Biodiversity. Conserv. Biol. 2021, 35, 502–509. [Google Scholar] [CrossRef]
- Fischer, M.W.; Stolze-Rybczynski, J.L.; Cui, Y.; Money, N.P. How Far and How Fast Can Mushroom Spores Fly? Physical Limits on Ballistospore Size and Discharge Distance in the Basidiomycota. Fungal Biol. 2010, 114, 669–675. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Winkler, K.; Fuchs, R.; Rounsevell, M.; Herold, M. Global Land Use Changes Are Four Times Greater than Previously Estimated. Nat. Commun. 2021, 12, 2501. [Google Scholar] [CrossRef] [PubMed]
- Antrop, M. Changing Patterns in the Urbanized Countryside of Western Europe. Landsc. Ecol. 2000, 15, 257–270. [Google Scholar] [CrossRef]
- Ying, Q.; Hansen, M.C.; Potapov, P.V.; Tyukavina, A.; Wang, L.; Stehman, S.V.; Moore, R.; Hancher, M. Global Bare Ground Gain from 2000 to 2012 Using Landsat Imagery. Remote Sens. Environ. 2017, 194, 161–176. [Google Scholar] [CrossRef]
- Galindo-Leal, C.; Câmara, I.G. Atlantic Forest Hotspot Status: An Overview. In The Atlantic Forest of South America: Biodiversity Status, Threats, and Outlook; Conservation International: Crystal City, VA, USA, 2003; pp. 3–11. [Google Scholar]
- IPCC. Climate Change 2022: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Pörtner, H.-O., Roberts, D.C., Tignor, M., Poloczanska, E.S., Mintenbeck, K., Alegría, A., Craig, M., Langsdorf, S., Löschke, S., Möller, V., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2022; p. 3056. [Google Scholar]
- Miller, J.E.; Root, H.T.; Safford, H.D. Altered Fire Regimes Cause Long-term Lichen Diversity Losses. Glob. Change Biol. 2018, 24, 4909–4918. [Google Scholar] [CrossRef]
- Miller, J.E.; Weill, A.M.; Villella, J. Epiphytic Macrolichen Communities Take Decades to Recover after High-severity Wildfire in Chaparral Shrublands. Divers. Distrib. 2022, 28, 454–462. [Google Scholar] [CrossRef]
- Stone, D.; Villella, J.; Root, H. Hypotrachyna riparia. IUCN Red List Threat. Species 2022, e.T195432681A195432683; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2022. [Google Scholar] [CrossRef]
- Leonard, P.L. Bondarzewia retipora. IUCN Red List Threat. Species 2019, e.T154430456A154430616; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2019. [Google Scholar] [CrossRef]
- Vargas, N.; Gonçalves, S.C.; Franco-Molano, A.E.; Restrepo, S.; Pringle, A. In Colombia the Eurasian Fungus Amanita Muscaria Is Expanding Its Range into Native, Tropical Quercus Humboldtii Forests. Mycologia 2019, 111, 758–771. [Google Scholar] [CrossRef]
- Allen, J.L.; McMullin, R.T.; Tripp, E.A.; Lendemer, J.C. Lichen Conservation in North America: A Review of Current Practices and Research in Canada and the United States. Biodivers. Conserv. 2019, 28, 3103–3138. [Google Scholar] [CrossRef]
- Nash, T.H., III. Lichen Biology, 1st ed.; Cambridge University Press: Cambridge, UK, 1996. [Google Scholar]
- Conti, M.E.; Cecchetti, G. Biological Monitoring: Lichens as Bioindicators of Air Pollution Assessment—A Review. Environ. Pollut. 2001, 114, 471–492. [Google Scholar] [CrossRef]
- Nash, T.H., III (Ed.) Lichen Sensitivity to Air Pollution. In Lichen Biology; Cambridge University Press: Cambridge, UK, 2008; pp. 299–314. ISBN 978-0-521-87162-4. [Google Scholar]
- Arnolds, E.E.F. Decline of Ectomycorrhizal Fungi in Europe. Agric. Ecosyst. Environ. 1991, 35, 209–244. [Google Scholar] [CrossRef]
- Wallenda, T.; Kottke, I. Nitrogen Deposition and Ectomycorrhizas. New Phytol. 1998, 139, 169–187. [Google Scholar] [CrossRef]
- Avis, P.G.; Mueller, G.M.; Lussenhop, J. Ectomycorrhizal Fungal Communities in Two North American Oak Forests Respond to Nitrogen Addition. New Phytol. 2008, 179, 472–483. [Google Scholar] [CrossRef]
- Griffith, G.W.; Gamarra, J.P.; Holden, E.M.; Mitchel, D.; Graham, A.; Evans, D.A.; Evans, S.E.; Aron, C.; Noordeloos, M.E.; Kirk, P.M. The International Conservation Importance of Welsh’waxcap’grasslands. Mycosphere 2013, 4, 969–984. [Google Scholar] [CrossRef]
- Feng, L.; Smith, S.J.; Braun, C.; Crippa, M.; Gidden, M.J.; Hoesly, R.; Klimont, Z.; Van Marle, M.; Van Den Berg, M.; Van Der Werf, G.R. The Generation of Gridded Emissions Data for CMIP6. Geosci. Model Dev. 2020, 13, 461–482. [Google Scholar] [CrossRef]
- Ellis, C.J. A Risk-Based Model of Climate Change Threat: Hazard, Exposure, and Vulnerability in the Ecology of Lichen Epiphytes. Botany 2013, 91, 1–11. [Google Scholar] [CrossRef]
- Allen, J.; Lendemer, J.; McMullin, T. Lepraria lanata. IUCN Red List Threat. Species 2020, e.T80702927A80702930; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2020. [Google Scholar] [CrossRef]
- Allen, J.L.; Lendemer, J.C. Quantifying the Impacts of Sea-Level Rise on Coastal Biodiversity: A Case Study on Lichens in the Mid-Atlantic Coast of Eastern North America. Biol. Conserv. 2016, 202, 119–126. [Google Scholar] [CrossRef]
- Sokoloff, P.; McMullin, T. Seirophora aurantiaca. IUCN Red List Threat. Species 2020, e.T175710010A175710692; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2020. [Google Scholar] [CrossRef]
- Gange, A.C.; Gange, E.G.; Sparks, T.H.; Boddy, L. Rapid and Recent Changes in Fungal Fruiting Patterns. Science 2007, 316, 71. [Google Scholar] [CrossRef]
- Kauserud, H.; Stige, L.C.; Vik, J.O.; Økland, R.H.; Høiland, K.; Stenseth, N.C. Mushroom Fruiting and Climate Change. Proc. Natl. Acad. Sci. USA 2008, 105, 3811–3814. [Google Scholar] [CrossRef] [PubMed]
- Vogt-Schilb, H.; Richard, F.; Malaval, J.-C.; Rapior, S.; Fons, F.; Bourgade, V.; Schatz, B.; Buentgen, U.; Moreau, P.-A. Climate-Induced Long-Term Changes in the Phenology of Mediterranean Fungi. Fungal Ecol. 2022, 60, 101166. [Google Scholar] [CrossRef]
- Bidartondo, M.I.; Ellis, C.; Kauserud, H.; Kennedy, P.G.; Lilleskov, E.; Suz, L.; Andrew, C. Climate Change: Fungal Responses and Effects. In State of the World’s Fungi 2018; Willis, K.J., Ed.; Royal Botanical Gardens, Kew: Richmond, UK, 2018; pp. 62–69. [Google Scholar]
- Dymytrova, L.; Brändli, U.-B.; Ginzler, C.; Scheidegger, C. Forest History and Epiphytic Lichens: Testing Indicators for Assessing Forest Autochthony in Switzerland. Ecol. Indic. 2018, 84, 847–857. [Google Scholar] [CrossRef]
- McMullin, R.T.; Wiersma, Y.F. Out with OLD Growth, in with Ecological Contin NEW Ity: New Perspectives on Forest Conservation. Front. Ecol. Environ. 2019, 17, 176–181. [Google Scholar] [CrossRef]
- Molina, R.; Pilz, D.; Smith, J. Conservation and Management of Forest Fungi in the Pacific Northwestern. Fungal Conserv. Issues Solut. 2001, 22, 19. [Google Scholar]
- Reese Næsborg, R. Calicium sequoiae. IUCN Red List Threat. Species 2021, e.T180412795A184974492; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2021. [Google Scholar] [CrossRef]
- Reese Næsborg, R. Xylopsora canopeorum. IUCN Red List Threat. Species 2022, e.T194662559A213315050; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2022. [Google Scholar] [CrossRef]
- Vasco-Palacios, A.M.; Zuluaga, M.; Calle, A.; Drechsler-Santos, E.R.; Kossmann, T.; da Cunha, K.M.; Sandoval-Leiva, P. Phylloporus fibulatus (Amended Version of 2020 Assessment). IUCN Red List Threat. Species 2020, e.T172831186A179542468; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2020. [Google Scholar] [CrossRef]
- Castellano, M. Gastrolactarius camphoratus. IUCN Red List Threat. Species 2015, e.T75111171A75111471; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2015. [Google Scholar] [CrossRef]
- Hyde, K.D. The Numbers of Fungi. Fungal Divers. 2022, 114, 1. [Google Scholar] [CrossRef]
- Kirk, P.M.; Cannon, P.F.; Minter, D.W.; Stalpers, J.A. Dictionary of the Fungi, 10th ed.; CABI Publishing: Wallingford, UK, 2008; ISBN 1-84593-933-6. [Google Scholar]
- Wijayawardene, N.N.; Hyde, K.D.; Al-Ani, L.K.T.; Tedersoo, L.; Haelewaters, D.; Becerra, A.G.; Schnittler, M.; Shchepin, O.N.; Novozhilov, Y.K.; Silva-Filho, A.G.S. Outline of Fungi and Fungus-like Taxa; CONICET: San Juan, Argentina, 2020. [Google Scholar]
- James, T.Y.; Stajich, J.E.; Hittinger, C.T.; Rokas, A. Toward a Fully Resolved Fungal Tree of Life. Annu. Rev. Microbiol. 2020, 74, 291–313. [Google Scholar] [CrossRef]
- Gonçalves, S.C.; Haelewaters, D.; Furci, G.; Mueller, G.M. Include All Fungi in Biodiversity Goals. Science 2021, 373, 403. [Google Scholar] [CrossRef]
- GBIF. New Feature Enables Search of Occurrence Data by Global IUCN Red List Category; GBIF: Copenhagen, Denmark, 2022. [Google Scholar]
- Baillie, J.E.M.; Collen, B.; Amin, R.; Akcakaya, H.R.; Butchart, S.H.M.; Brummitt, N.; Meagher, T.R.; Ram, M.; Hilton-Taylor, C.; Mace, G.M. Toward Monitoring Global Biodiversity. Conserv. Lett. 2008, 1, 18–26. [Google Scholar] [CrossRef]
- Brummitt, N.A.; Bachman, S.P.; Griffiths-Lee, J.; Lutz, M.; Moat, J.F.; Farjon, A.; Donaldson, J.S.; Hilton-Taylor, C.; Meagher, T.R.; Albuquerque, S.; et al. Green Plants in the Red: A Baseline Global Assessment for the IUCN Sampled Red List Index for Plants. PLoS ONE 2015, 10, e0135152. [Google Scholar] [CrossRef]
- Henriques, S.; Böhm, M.; Collen, B.; Luedtke, J.; Hoffmann, M.; Hilton-Taylor, C.; Cardoso, P.; Butchart, S.H.M.; Freeman, R. Accelerating the Monitoring of Global Biodiversity: Revisiting the Sampled Approach to Generating Red List Indices. Conserv. Lett. 2020, 13, e12703. [Google Scholar] [CrossRef]
- Minter, D.W. The Ascomycota and the Sampled Red List Index Scheme. Fungal Conserv. 2011, 1, 45–53. [Google Scholar]
- May, T.W. Use of Target Species in Citizen Science Fungi Recording Schemes. Biodivers. Inf. Sci. Stand. 2021, 5, e73960. [Google Scholar] [CrossRef]
- Yan, D.; Mills, J.G.; Gellie, N.J.C.; Bissett, A.; Lowe, A.J.; Breed, M.F. High-Throughput EDNA Monitoring of Fungi to Track Functional Recovery in Ecological Restoration. Biol. Conserv. 2018, 217, 113–120. [Google Scholar] [CrossRef]
- Frøslev, T.G.; Kjøller, R.; Bruun, H.H.; Ejrnæs, R.; Hansen, A.J.; Læssøe, T.; Heilmann-Clausen, J. Man against Machine: Do Fungal Fruitbodies and EDNA Give Similar Biodiversity Assessments across Broad Environmental Gradients? Biol. Conserv. 2019, 233, 201–212. [Google Scholar] [CrossRef]
- Ruppert, K.M.; Kline, R.J.; Rahman, M.S. Past, Present, and Future Perspectives of Environmental DNA (EDNA) Metabarcoding: A Systematic Review in Methods, Monitoring, and Applications of Global EDNA. Glob. Ecol. Conserv. 2019, 17, e00547. [Google Scholar] [CrossRef]
- Norman, K.J.V.; Gordon, M. Landscape Scale Environmental DNA Sampling for a Rare Fungal Species: Implications for Forest Management. For. Ecol. Manag. 2021, 480, 118741. [Google Scholar] [CrossRef]
- Bachman, S.; Walker, B.E.; Barrios, S.; Copeland, A.; Moat, J. Rapid Least Concern: Towards Automating Red List Assessments. Biodivers. Data J. 2020, 8, e47018. [Google Scholar] [CrossRef] [Green Version]
- May, T.W. Where Are the Short-Range Endemics among Western Australian Macrofungi? Aust. Syst. Bot. 2002, 15, 501–511. [Google Scholar] [CrossRef]
- FNAI. Status Survey of Perforate Reindeer Lichen (Cladonia Perforata), a Federally Endangered Lichen Species throughout Its Known Range in Florida; Final Report to the Florida Department of Agriculture and Florida Forest Service; FNAI: Tallahassee, FL, USA, 2021. [Google Scholar]
- COSEWIC. COSEWIC Assessment and Status Report on the Boreal Felt Lichen Erioderma Pedicellatum, Boreal Population and Atlantic Population, in Canada; Committee on the Status of Endangered Wildlife in Canada: Ottawa, ON, Canada, 2014; Available online: https://www.canada.ca/en/environment-climate-change/services/species-risk-public-registry/cosewic-assessments-status-reports/boreal-felt-lichen.html (accessed on 21 July 2022).
- Bosanquet, S.; Ainsworth, M.; Cooch, S.; Genney, D.; Wilkins, T. Non-Lichenised Fungi. In Guidelines for the Selection of Biological SSSIs. Part 2: Detailed Guidelines for Habitats and Species Groups; Joint Nature Conservation Committee: Peterborough, UK, 2018. [Google Scholar]
- Sanderson, N.; Wilkins, T.; Bosanquet, S.; Genney, D. Lichens and Associated Microfungi. In Guidelines for the Selection of Biological SSSIs. Part 2; JNCC: Peterborough, UK, 2018; 31p. [Google Scholar]
- IUCN. IUCN Red List Categories and Criteria: Version 3.1, 2nd ed.; IUCN: Gland, Switzerland; Cambridge, UK, 2012. [Google Scholar]
- Rondinini, C.; Di Marco, M.; Visconti, P.; Butchart, S.H.M.; Boitani, L. Update or Outdate: Long-Term Viability of the IUCN Red List. Conserv. Lett. 2014, 7, 126–130. [Google Scholar] [CrossRef]
- May, T.W.; Cooper, J.A.; Dahlberg, A.; Furci, G.; Minter, D.W.; Mueller, G.M.; Pouliot, A.; Yang, Z. Recognition of the Discipline of Conservation Mycology. Conserv. Biol. 2018, 33, 733–736. [Google Scholar] [CrossRef]
- Nordén, J.; Abrego, N.; Boddy, L.; Bässler, C.; Dahlberg, A.; Halme, P.; Hällfors, M.; Maurice, S.; Menkis, A.; Miettinen, O. Ten Principles for Conservation Translocations of Threatened Wood-Inhabiting Fungi. Fungal Ecol. 2020, 44, 100919. [Google Scholar] [CrossRef]



| Trophic Mode | Number of Assessed Species | Number of Threatened Species |
|---|---|---|
| Mutualists | 369 | 143 |
| Parasites | 20 | 11 |
| Saprotrophs | 174 | 100 |
| Unknown | 34 | 30 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mueller, G.M.; Cunha, K.M.; May, T.W.; Allen, J.L.; Westrip, J.R.S.; Canteiro, C.; Costa-Rezende, D.H.; Drechsler-Santos, E.R.; Vasco-Palacios, A.M.; Ainsworth, A.M.; et al. What Do the First 597 Global Fungal Red List Assessments Tell Us about the Threat Status of Fungi? Diversity 2022, 14, 736. https://doi.org/10.3390/d14090736
Mueller GM, Cunha KM, May TW, Allen JL, Westrip JRS, Canteiro C, Costa-Rezende DH, Drechsler-Santos ER, Vasco-Palacios AM, Ainsworth AM, et al. What Do the First 597 Global Fungal Red List Assessments Tell Us about the Threat Status of Fungi? Diversity. 2022; 14(9):736. https://doi.org/10.3390/d14090736
Chicago/Turabian StyleMueller, Gregory M., Kelmer Martins Cunha, Tom W. May, Jessica L. Allen, James R. S. Westrip, Cátia Canteiro, Diogo Henrique Costa-Rezende, Elisandro Ricardo Drechsler-Santos, Aída M. Vasco-Palacios, Antony Martyn Ainsworth, and et al. 2022. "What Do the First 597 Global Fungal Red List Assessments Tell Us about the Threat Status of Fungi?" Diversity 14, no. 9: 736. https://doi.org/10.3390/d14090736
APA StyleMueller, G. M., Cunha, K. M., May, T. W., Allen, J. L., Westrip, J. R. S., Canteiro, C., Costa-Rezende, D. H., Drechsler-Santos, E. R., Vasco-Palacios, A. M., Ainsworth, A. M., Alves-Silva, G., Bungartz, F., Chandler, A., Gonçalves, S. C., Krisai-Greilhuber, I., Iršėnaitė, R., Jordal, J. B., Kosmann, T., Lendemer, J., ... Dahlberg, A. (2022). What Do the First 597 Global Fungal Red List Assessments Tell Us about the Threat Status of Fungi? Diversity, 14(9), 736. https://doi.org/10.3390/d14090736









