Unlocking the Hidden Potential of Agave tequilana for the Green Synthesis of Antibacterial ZnO Nanomaterials: A Waste-to-Value Nanotechnology Approach
Abstract
1. Introduction
2. Results
Antibacterial Activity
3. Discussion
4. Materials and Methods
4.1. Chemicals
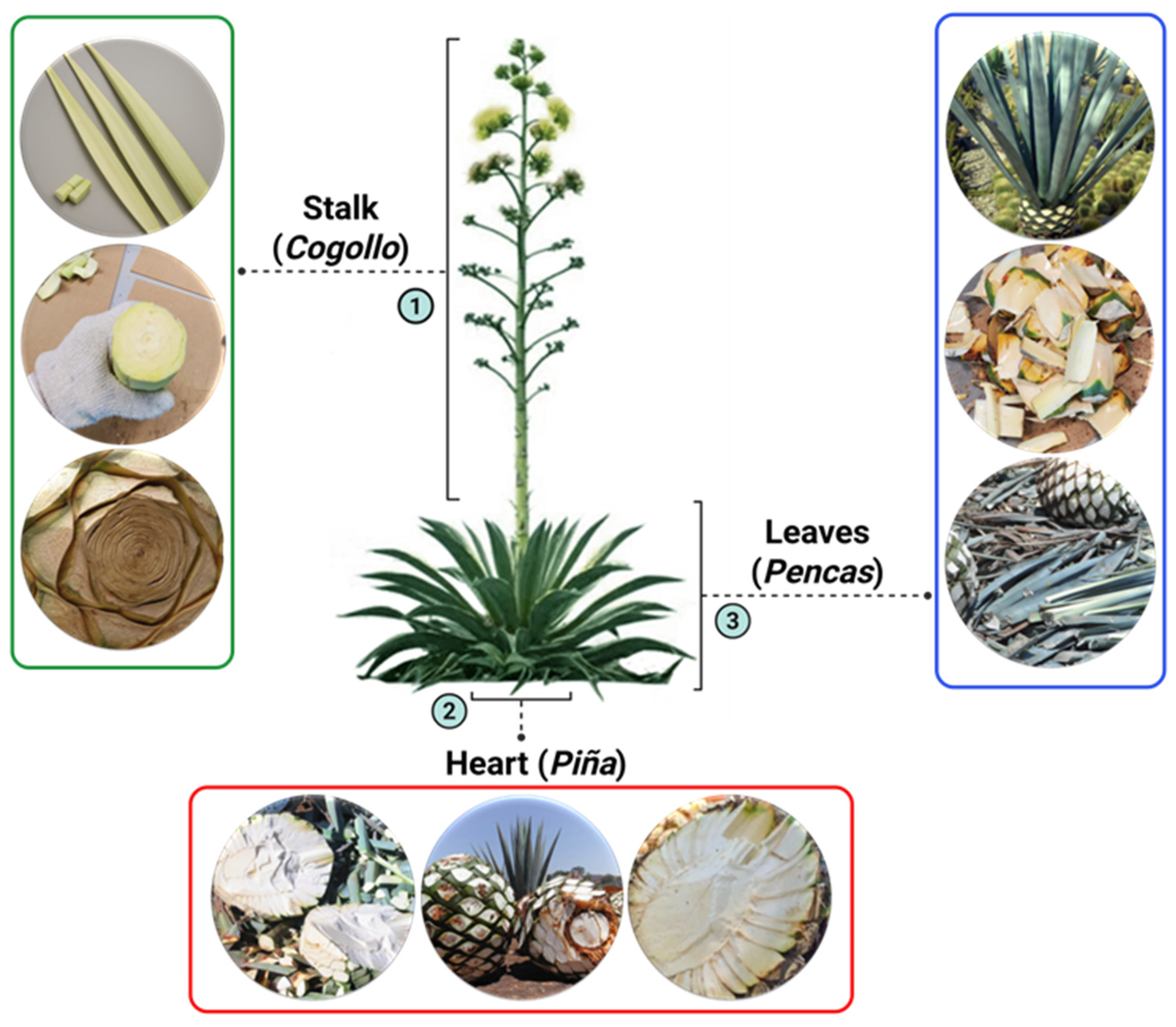
4.2. Collection of Plants (Agave tequilana)
4.3. Green Synthesis of ZnO-NPs Using Extracts of Different Parts of Agave tequilana
4.4. Materials Characterization
4.5. Antibacterial Activity Assessment
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AMR | Anti-Microbial Resistance |
| ZnO | Zinc Oxide |
| NPs | Nanoparticles |
| ZnO-NPs | Zinc Oxide Nanoparticles |
| MO | Metal Oxide |
| MO-NPs | Metal Oxide Nanoparticles |
| ZnO-S | Zinc Oxide Stalk |
| ZnO-H | Zinc Oxide Heart |
| ZnO-L | Zinc Oxide Leaves |
| UV | Ultraviolet |
| XRD | X-Ray Diffraction |
| ATR | Attenuated Total Reflectance |
| FTIR | Fourier-Transform Infrared Spectroscopy |
| FESEM | Field Emission Scanning Electron Microscopy |
| EDS | Energy-Dispersive X-Ray Spectroscopy |
| ROS | Reactive Oxygen Species |
| mL | Milliliter |
| g | Gram |
| mg | Milligram |
| µg | Microgram |
| Eg | Bandgap |
| eV | Electron Volt |
| NIR | Near-Infrared |
| nm | Nanometer |
| mm | Millimeter |
| cm | Centimeter |
| L | Liter |
| IZ | Inhibition Zones |
| FWHM | Full Width at Half Maximum |
| JCPDS | Joint Committee on Powder Diffraction Standards |
References
- Tiwari, A.; Shaik, A.H.; Brianna, B.; Anwar, A.; Chandan, M.R. Anti-cancer and antimicrobial efficacy of ZnO nanoparticles synthesized via green route using Amaranthus dubius (Spleen Amaranth) leaves extract. Green Chem. Lett. Rev. 2025, 18, 2453532. [Google Scholar]
- Aldeen, T.S.; Mohamed, H.E.A.; Maaza, M. ZnO nanoparticles prepared via a green synthesis approach: Physical properties, photocatalytic and antibacterial activity. J. Phys. Chem. Solids 2022, 160, 110313. [Google Scholar] [CrossRef]
- Soni, V.; Raizada, P.; Singh, P.; Cuong, H.N.; Saini, A.; Saini, R.V.; Van Le, Q.; Nadda, A.K.; Le, T.-T.; Nguyen, V.-H. Sustainable and green trends in using plant extracts for the synthesis of biogenic metal nanoparticles toward environmental and pharmaceutical advances: A review. Environ. Res. 2021, 202, 111622. [Google Scholar] [CrossRef]
- Faisal, S.; Jan, H.; Shah, S.A.; Shah, S.; Khan, A.; Akbar, M.T.; Rizwan, M.; Jan, F.; Wajidullah; Akhtar, N. Green synthesis of zinc oxide (ZnO) nanoparticles using aqueous fruit extracts of Myristica fragrans: Their characterizations and biological and environmental applications. ACS Omega 2021, 6, 9709–9722. [Google Scholar] [CrossRef]
- Arshi, N.; Prashanthi, Y.; Rao, T.N.; Ahmed, F.; Kumar, S.; Oves, M. Biosynthesis of ZnO nanostructures using Azadirachta indica leaf extract and their effect on seed germination and seedling growth of tomato: An eco-friendly approach. J. Nanoelectron. Optoelectron. 2020, 15, 1412–1422. [Google Scholar] [CrossRef]
- Sportelli, M.C.; Gaudiuso, C.; Volpe, A.; Izzi, M.; Picca, R.A.; Ancona, A.; Cioffi, N. Biogenic synthesis of ZnO nanoparticles and their application as bioactive agents: A critical overview. Reactions 2022, 3, 423–441. [Google Scholar] [CrossRef]
- Benitez-Salazar, M.I.; Niño-Castaño, V.E.; Dueñas-Cuellar, R.A.; Caldas-Arias, L.; Fernández, I.; Rodríguez-Páez, J.E. Chemical synthesis versus green synthesis to obtain ZnO powders: Evaluation of the antibacterial capacity of the nanoparticles obtained by the chemical method. J. Environ. Chem. Eng. 2021, 9, 106544. [Google Scholar] [CrossRef]
- Abdelbaky, A.S.; Abd El-Mageed, T.A.; Babalghith, A.O.; Selim, S.; Mohamed, A.M.J.A. Green synthesis and characterization of ZnO nanoparticles using Pelargonium odoratissimum (L.) aqueous leaf extract and their antioxidant, antibacterial and anti-inflammatory activities. Antioxidants 2022, 11, 1444. [Google Scholar] [CrossRef]
- Yusefi, M.; Shameli, K.; Ali, R.R.; Pang, S.-W.; Teow, S.-Y. Evaluating anticancer activity of plant-mediated synthesized iron oxide nanoparticles using Punica granatum fruit peel extract. J. Mol. Struct. 2020, 1204, 127539. [Google Scholar] [CrossRef]
- Ahamad Khan, M.; Lone, S.A.; Shahid, M.; Zeyad, M.T.; Syed, A.; Ehtram, A.; Elgorban, A.M.; Verma, M.; Danish, M. Phytogenically synthesized zinc oxide nanoparticles (ZnO-NPs) potentially inhibit the bacterial pathogens: In vitro studies. Toxics 2023, 11, 452. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Morshedi, M. Cutting-edge nanotechnology: Unveiling the role of zinc oxide nanoparticles in combating deadly gastrointestinal tumors. Front. Bioeng. Biotechnol. 2025, 13, 1547757. [Google Scholar] [CrossRef] [PubMed]
- Aigbe, U.O.; Osibote, O.A. Green synthesis of metal oxide nanoparticles, and their various applications. J. Hazard. Mater. Adv. 2024, 13, 100401. [Google Scholar] [CrossRef]
- Hamed, R.; Obeid, R.Z.; Abu-Huwaij, R. Plant mediated-green synthesis of zinc oxide nanoparticles: An insight into biomedical applications. Nanotechnol. Rev. 2023, 12, 20230112. [Google Scholar] [CrossRef]
- Murali, M.; Kalegowda, N.; Gowtham, H.G.; Ansari, M.A.; Alomary, M.N.; Alghamdi, S.; Shilpa, N.; Singh, S.B.; Thriveni, M.; Aiyaz, M. Plant-mediated zinc oxide nanoparticles: Advances in the new millennium towards understanding their therapeutic role in biomedical applications. Pharmaceutics 2021, 13, 1662. [Google Scholar] [CrossRef]
- Channa, G.M.; Iturbe-Ek, J.; Sustaita, A.O.; Melo-Maximo, D.V.; Bhatti, A.; Esparza-Sanchez, J.; Navarro-Lopez, D.E.; Lopez-Mena, E.R.; Sanchez-Lopez, A.L.; Lozano, L.M. Eco-Friendly Synthesis of ZnO Nanoparticles from Natural Agave, Chiku, and Soursop Extracts: A Sustainable Approach to Antibacterial Applications. Crystals 2025, 15, 470. [Google Scholar] [CrossRef]
- Alhujaily, M.; Albukhaty, S.; Yusuf, M.; Mohammed, M.K.; Sulaiman, G.M.; Al-Karagoly, H.; Alyamani, A.A.; Albaqami, J.; AlMalki, F.A. Recent advances in plant-mediated zinc oxide nanoparticles with their significant biomedical properties. Bioengineering 2022, 9, 541. [Google Scholar] [CrossRef]
- Bajwa, H.U.R.; Khan, M.K.; Abbas, Z.; Riaz, R.; Abbas, R.Z.; Aleem, M.T.; Abbas, A.; Almutairi, M.M.; Alshammari, F.A.; Alraey, Y.J.L. Nanoparticles: Synthesis and their role as potential drug candidates for the treatment of parasitic diseases. Life 2022, 12, 750. [Google Scholar] [CrossRef]
- Geioushy, R.A.; El-Sherbiny, S.; Mohamed, E.T.; Fouad, O.A.; Samir, M. Mechanical characteristics and antibacterial activity against Staphylococcus aureus of sustainable cellulosic paper coated with Ag and Cu modified ZnO nanoparticles. Sci. Rep. 2024, 14, 29722. [Google Scholar] [CrossRef]
- Priya, G.H.; Shaly, A.A.; Ragu, R.; Evangelin, G.; Mani, J.; Martin, A.; Linet, J.M. ZnO nanoparticles with altered structural and optical traits by Cu and Li doping for elevated photocatalytic activity toward organic pollutants. J. Dispers. Sci. Technol. 2024, 46, 21–28. [Google Scholar]
- Moosavi, F.; Neri, G. Effect of Pb doping on the structural, optical and electrical properties of sol–gel ZnO nanoparticles. Discov. Mater. 2023, 3, 30. [Google Scholar] [CrossRef]
- Qu, B.; Xiao, Z.; Luo, Y. Sustainable Nanotechnology for Food Preservation: Synthesis, Mechanisms, and Applications of Zinc Oxide Nanoparticles. J. Agric. Food Res. 2025, 19, 101743. [Google Scholar] [CrossRef]
- Haiouani, K.; Hegazy, S.; Alsaeedi, H.; Bechelany, M.; Barhoum, A. Green Synthesis of Hexagonal-like ZnO nanoparticles modified with phytochemicals of clove (Syzygium aromaticum) and Thymus capitatus extracts: Enhanced antibacterial, antifungal, and antioxidant activities. Materials 2024, 17, 4340. [Google Scholar] [CrossRef]
- Ilavenil, K.; Senthilkumar, V.; Kasthuri, A. Green synthesis of metal nanoparticles from three medicinal plants: A review of environmental and health applications. Discov. Catal. 2025, 2, 3. [Google Scholar] [CrossRef]
- Díaz-Ramos, D.I.; Ortiz-Basurto, R.I.; García-Barradas, O.; Chacón-López, M.A.; Montalvo-González, E.; Pascual-Pineda, L.A.; Valenzuela-Vázquez, U.; Jiménez-Fernández, M. Lauroylated, Acetylated, and Succinylated Agave tequilana Fructans Fractions: Structural Characterization, Prebiotic, Antibacterial Activity and Their Effect on Lactobacillus paracasei under Gastrointestinal Conditions. Polymers 2023, 15, 3115. [Google Scholar] [CrossRef]
- Pérez-López, A.V.; Simpson, J.; Clench, M.R.; Gomez-Vargas, A.D.; Ordaz-Ortiz, J.J. Localization and Composition of Fructans in Stem and Rhizome of Agave tequilana Weber var. azul. Front. Plant Sci. 2021, 11, 608850. [Google Scholar] [CrossRef]
- Riwayati, I.; Winardi, S.; Madhania, S.; Shimada, M. Green synthesis of ZnO nanoparticles using Cosmos caudatus: Effects of calcination temperature and precursor type on photocatalytic and antimicrobial activities. Results Eng. 2024, 24, 103594. [Google Scholar] [CrossRef]
- Khan, M.F.; Khan, M.A. Plant-derived metal nanoparticles (PDMNPs): Synthesis, characterization, and oxidative stress-mediated therapeutic actions. Future Pharmacol. 2023, 3, 252–295. [Google Scholar] [CrossRef]
- Swain, M.; Mishra, D.; Sahoo, G. A review on green synthesis of ZnO nanoparticles. Discov. Appl. Sci. 2025, 7, 997. [Google Scholar] [CrossRef]
- Panchal, P.; Paul, D.R.; Sharma, A.; Choudhary, P.; Meena, P.; Nehra, S. Biogenic mediated Ag/ZnO nanocomposites for photocatalytic and antibacterial activities towards disinfection of water. J. Colloid Interface Sci. 2020, 563, 370–380. [Google Scholar] [CrossRef]
- Panchal, P.; Paul, D.R.; Sharma, A.; Hooda, D.; Yadav, R.; Meena, P.; Nehra, S.P. Phytoextract mediated ZnO/MgO nanocomposites for photocatalytic and antibacterial activities. J. Photochem. Photobiol. A Chem. 2019, 385, 112049. [Google Scholar] [CrossRef]
- Coccia, F.; Mascitti, A.; Rastelli, G.; d’Alessandro, N.; Tonucci, L. Sustainable Photocatalytic Reduction of Maleic Acid: Enhancing CuxO/ZnO Stability with Polydopamine. Appl. Sci. 2025, 15, 1631. [Google Scholar] [CrossRef]
- Golzarnezhad, F.; Allahdou, M.; Mehravaran, L.; Naderi, S. Green synthesis of ZnO nanoparticles from the extract of Cymbopogon olivieri and investigation of their antimicrobial and anticancer effects. Discov. Appl. Sci. 2025, 7, 196. [Google Scholar] [CrossRef]
- Dey, A.; Somaiah, S. Green synthesis and characterization of zinc oxide nanoparticles using leaf extract of Thryallis glauca (Cav.) Kuntze and their role as antioxidant and antibacterial. Microsc. Res. Tech. 2022, 85, 2835–2847. [Google Scholar] [CrossRef]
- Kanimozhi, S.; Durga, R.; Sabithasree, M.; Kumar, A.V.; Sofiavizhimalar, A.; Kadam, A.A.; Rajagopal, R.; Sathya, R.; Azelee, N.I.W. Biogenic synthesis of silver nanoparticle using Cissus quadrangularis extract and its invitro study. J. King Saud Univ.-Sci. 2022, 34, 101930. [Google Scholar] [CrossRef]
- Naseer, M.; Aslam, U.; Khalid, B.; Chen, B. Green route to synthesize Zinc Oxide Nanoparticles using leaf extracts of Cassia fistula and Melia azadarach and their antibacterial potential. Sci. Rep. 2020, 10, 9055. [Google Scholar] [CrossRef]
- Hossain, A.; Abdallah, Y.; Ali, M.A.; Masum, M.M.I.; Li, B.; Sun, G.; Meng, Y.; Wang, Y.; An, Q. Lemon-fruit-based green synthesis of zinc oxide nanoparticles and titanium dioxide nanoparticles against soft rot bacterial pathogen Dickeya dadantii. Biomolecules 2019, 9, 863. [Google Scholar] [CrossRef]
- Rad, S.S.; Sani, A.M.; Mohseni, S. Biosynthesis, characterization and antimicrobial activities of zinc oxide nanoparticles from leaf extract of Mentha pulegium (L.). Microb. Pathog. 2019, 131, 239–245. [Google Scholar] [CrossRef]
- Manojkumar, U.; Kaliannan, D.; Srinivasan, V.; Balasubramanian, B.; Kamyab, H.; Mussa, Z.H.; Palaniyappan, J.; Mesbah, M.; Chelliapan, S.; Palaninaicker, S. Green synthesis of zinc oxide nanoparticles using Brassica oleracea var. botrytis leaf extract: Photocatalytic, antimicrobial and larvicidal activity. Chemosphere 2023, 323, 138263. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, H.; Venugopal, K.; Rajagopal, K.; De Britto, S.; Nandini, B.; Pushpalatha, H.G.; Konappa, N.; Udayashankar, A.C.; Geetha, N.; Jogaiah, S. Green synthesis and characterization of zinc oxide nanoparticles using Eucalyptus globules and their fungicidal ability against pathogenic fungi of apple orchards. Biomolecules 2020, 10, 425. [Google Scholar] [CrossRef]
- Miri, A.; Khatami, M.; Ebrahimy, O.; Sarani, M. Cytotoxic and antifungal studies of biosynthesized zinc oxide nanoparticles using extract of Prosopis farcta fruit. Green Chem. Lett. Rev. 2020, 13, 27–33. [Google Scholar] [CrossRef]
- Kavithaa, K.; Paulpandi, M.; Ponraj, T.; Murugan, K.; Sumathi, S. Induction of intrinsic apoptotic pathway in human breast cancer (MCF-7) cells through facile biosynthesized zinc oxide nanorods. Karbala Int. J. Mod. Sci. 2016, 2, 46–55. [Google Scholar] [CrossRef]
- Neamah, S.A.; Albukhaty, S.; Falih, I.Q.; Dewir, Y.H.; Mahood, H.B. Biosynthesis of zinc oxide nanoparticles using Capparis spinosa L. fruit extract: Characterization, biocompatibility, and antioxidant activity. Appl. Sci. 2023, 13, 6604. [Google Scholar] [CrossRef]
- Efati, Z.; Shahangian, S.S.; Darroudi, M.; Amiri, H.; Hashemy, S.I.; Aghamaali, M.R. Green chemistry synthesized zinc oxide nanoparticles in Lepidium sativum L. seed extract and evaluation of their anticancer activity in human colorectal cancer cells. Ceram. Int. 2023, 49, 32568–32576. [Google Scholar] [CrossRef]
- Bhagat, T.; Lokhande, R.; Khadke-Lokhande, L.; Chandorkar, J. Green synthesis, characterization, application and study of antimicrobial properties of zinc oxide nano particles using Cyathocline purpurea phytoextract. Pharma. Innov. 2023, 12, 2627–2633. [Google Scholar]
- Kumar, N.H.; Andia, J.D.; Manjunatha, S.; Murali, M.; Amruthesh, K.; Jagannath, S. Antimitotic and DNA-binding potential of biosynthesized ZnO-NPs from leaf extract of Justicia wynaadensis (Nees) Heyne-A medicinal herb. Biocatal. Agric. Biotechnol. 2019, 18, 101024. [Google Scholar]
- Kavya, J.; Murali, M.; Manjula, S.; Basavaraj, G.; Prathibha, M.; Jayaramu, S.; Amruthesh, K. Genotoxic and antibacterial nature of biofabricated zinc oxide nanoparticles from Sida rhombifolia Linn. J. Drug Deliv. Sci. Technol. 2020, 60, 101982. [Google Scholar] [CrossRef]
- Chunchegowda, U.A.; Shivaram, A.B.; Mahadevamurthy, M.; Ramachndrappa, L.T.; Lalitha, S.G.; Krishnappa, H.K.N.; Anandan, S.; Sudarshana, B.S.; Chanappa, E.G.; Ramachandrappa, N.S. Biosynthesis of Zinc oxide nanoparticles using leaf extract of Passiflora subpeltata: Characterization and antibacterial activity against Escherichia coli isolated from poultry faeces. J. Clust. Sci. 2021, 32, 1663–1672. [Google Scholar] [CrossRef]
- Melk, M.M.; El-Hawary, S.S.; Melek, F.R.; Saleh, D.O.; Ali, O.M.; El Raey, M.A.; Selim, N.M. Nano zinc oxide green-synthesized from Plumbago auriculata lam. alcoholic extract. Plants 2021, 10, 2447. [Google Scholar] [CrossRef]
- Al Awadh, A.A.; Shet, A.R.; Patil, L.R.; Shaikh, I.A.; Alshahrani, M.M.; Nadaf, R.; Mahnashi, M.H.; Desai, S.V.; Muddapur, U.M.; Achappa, S. Sustainable synthesis and characterization of zinc oxide nanoparticles using Raphanus sativus extract and its biomedical applications. Crystals 2022, 12, 1142. [Google Scholar] [CrossRef]
- Thomas, S.; Gunasangkaran, G.; Arumugam, V.A.; Muthukrishnan, S. Synthesis and characterization of zinc oxide nanoparticles of solanum nigrum and its anticancer activity via the induction of apoptosis in cervical cancer. Biol. Trace Element Res. 2022, 200, 2684–2697. [Google Scholar] [CrossRef]
- Vijayakumar, S.; Vaseeharan, B.; Malaikozhundan, B.; Shobiya, M. Laurus nobilis leaf extract mediated green synthesis of ZnO nanoparticles: Characterization and biomedical applications. Biomed. Pharmacother. 2016, 84, 1213–1222. [Google Scholar] [CrossRef]
- Khatami, M.; Varma, R.S.; Zafarnia, N.; Yaghoobi, H.; Sarani, M.; Kumar, V.G. Applications of green synthesized Ag, ZnO and Ag/ZnO nanoparticles for making clinical antimicrobial wound-healing bandages. Sustain. Chem. Pharm. 2018, 10, 9–15. [Google Scholar] [CrossRef]
- Sarwar, K.; Nazli, Z.-i.-H.; Munir, H.; Aslam, M.; Khalofah, A. Biosynthesis of zinc oxide nanoparticles using Moringa oleifera leaf extract, probing antibacterial and antioxidant activities. Sci. Rep. 2025, 15, 20413. [Google Scholar] [CrossRef]
- Bala, N.; Saha, S.; Chakraborty, M.; Maiti, M.; Das, S.; Basu, R.; Nandy, P. Green synthesis of zinc oxide nanoparticles using Hibiscus subdariffa leaf extract: Effect of temperature on synthesis, anti-bacterial activity and anti-diabetic activity. RSC Adv. 2015, 5, 4993–5003. [Google Scholar] [CrossRef]
- Fregoso-Zamorano, B.E.; Mancilla-Villa, O.R.; Guevara-Gutiérrez, R.D.; Moreno-Hernández, A.; Figueroa-Bautista, P.; Can-Chulim, Á.; Hernández-Vargas, O.; Cruz-Crespo, E.; Ortega-Escobar, H.M.; Khalil Gardezi, A.J.T.L. Caracterización edafológica con cultivo de agave azul (Agave tequilana Weber) en Tonaya y Tuxcacuesco, Jalisco, México. Terra Latinoam. 2023, 41, e1592. [Google Scholar] [CrossRef]
- Tetreault, D.; McCulligh, C.; Lucio, C. Distilling agro-extractivism: Agave and tequila production in Mexico. J. Agrar. Chang. 2021, 21, 219–241. [Google Scholar] [CrossRef]
- García-Villalba, W.G.; Rodríguez-Herrera, R.; Ochoa-Martínez, L.A.; Rutiaga-Quiñones, O.M.; Gallegos-Infante, J.A.; González-Herrera, S.M. Agave fructans: A review of their technological functionality and extraction processes. J. Food Sci. Technol. 2023, 60, 1265–1273. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Chávez, J.; Villamiel, M.; Santos-Zea, L.; Ramírez-Jiménez, A.K. Agave by-products: An overview of their nutraceutical value, current applications, and processing methods. Polysaccharides 2021, 2, 720–743. [Google Scholar] [CrossRef]
- Lachenmeier, D.W.; Sohnius, E.-M.; Attig, R.; López, M.G. Quantification of selected volatile constituents and anions in Mexican Agave spirits (Tequila, Mezcal, Sotol, Bacanora). J. Agric. Food Chem. 2006, 54, 3911–3915. [Google Scholar] [CrossRef]
- Jakhrani, M.A.; Tahira, A.; Bhatti, M.A.; Shah, A.A.; Shaikh, N.M.; Mari, R.H.; Vigolo, B.; Emo, M.; Albaqami, M.D.; Nafady, A. A green approach for the preparation of ZnO@C nanocomposite using agave americana plant extract with enhanced photodegradation. Nanotechnology 2022, 33, 505202. [Google Scholar] [CrossRef]
- Gutiérrez-Hernández, J.M.; Escalante, A.; Murillo-Vázquez, R.N.; Delgado, E.; González, F.J.; Toríz, G. Use of Agave tequilana-lignin and zinc oxide nanoparticles for skin photoprotection. J. Photochem. Photobiol. B Biol. 2016, 163, 156–161. [Google Scholar] [CrossRef]
- Chiu, R.; Tinel, L.; Gonzalez, L.; Ciuraru, R.; Bernard, F.; George, C.; Volkamer, R. UV photochemistry of carboxylic acids at the air-sea boundary: A relevant source of glyoxal and other oxygenated VOC in the marine atmosphere. Geophys. Res. Lett. 2017, 44, 1079–1087. [Google Scholar] [CrossRef]
- Boulet, J.C.; Ducasse, M.A.; Cheynier, V. Ultraviolet spectroscopy study of phenolic substances and other major compounds in red wines: Relationship between astringency and the concentration of phenolic substances. Aust. J. Grape Wine Res. 2017, 23, 193–199. [Google Scholar] [CrossRef]
- Taniguchi, M.; LaRocca, C.A.; Bernat, J.D.; Lindsey, J.S. Digital database of absorption spectra of diverse flavonoids enables structural comparisons and quantitative evaluations. J. Nat. Prod. 2023, 86, 1087–1119. [Google Scholar] [CrossRef] [PubMed]
- Shamsipur, M.; Roushani, M.; Pourmortazavi, S.M. Electrochemical synthesis and characterization of zinc oxalate nanoparticles. Mater. Res. Bull. 2013, 48, 1275–1280. [Google Scholar] [CrossRef]
- Mousa, S.A.; Wissa, D.; Hassan, H.; Ebnalwaled, A.; Khairy, S. Enhanced photocatalytic activity of green synthesized zinc oxide nanoparticles using low-cost plant extracts. Sci. Rep. 2024, 14, 16713. [Google Scholar] [CrossRef]
- Zou, Y.; Zhang, Y.; Hu, Y.; Gu, H. Ultraviolet detectors based on wide bandgap semiconductor nanowire: A review. Sensors 2018, 18, 2072. [Google Scholar] [CrossRef]
- Hameed, H.; Waheed, A.; Sharif, M.S.; Saleem, M.; Afreen, A.; Tariq, M.; Kamal, A.; Al-Onazi, W.A.; Al Farraj, D.A.; Ahmad, S. Green synthesis of zinc oxide (ZnO) nanoparticles from green algae and their assessment in various biological applications. Micromachines 2023, 14, 928. [Google Scholar] [CrossRef]
- Phoohinkong, W.; Foophow, T.; Pecharapa, W. Synthesis and characterization of copper zinc oxide nanoparticles obtained via metathesis process. Adv. Nat. Sci. Nanosci. Nanotechnol. 2017, 8, 035003. [Google Scholar] [CrossRef]
- Kaur, H.; Sharma, A.; Anand, K.; Panday, A.; Tagotra, S.; Kakran, S.; Singh, A.K.; Alam, M.W.; Kumar, S.; Bouzid, G. Green synthesis of ZnO nanoparticles using E. cardamomum and zinc nitrate precursor: A dual-functional material for water purification and antibacterial applications. RSC Adv. 2025, 15, 16742–16765. [Google Scholar] [CrossRef]
- Ali, M.B.; Iftikhar, T.; Majeed, H. Green synthesis of zinc oxide nanoparticles for the industrial biofortification of (Pleurotus pulmonarius) mushrooms. Heliyon 2024, 10, e37927. [Google Scholar] [CrossRef] [PubMed]
- Kalaba, M.H.; El-Sherbiny, G.M.; Ewais, E.A.; Darwesh, O.M.; Moghannem, S.A. Green synthesis of zinc oxide nanoparticles (ZnO-NPs) by Streptomyces baarnensis and its active metabolite (Ka): A promising combination against multidrug-resistant ESKAPE pathogens and cytotoxicity. BMC Microbiol. 2024, 24, 254. [Google Scholar] [CrossRef]
- Fatima, K.; Asif, M.; Farooq, U.; Gilani, S.J.; Bin Jumah, M.N.; Ahmed, M.M. Antioxidant and anti-inflammatory applications of Aerva persica aqueous-root extract-mediated synthesis of ZnO nanoparticles. ACS Omega 2024, 9, 15882–15892. [Google Scholar] [CrossRef]
- Fouda, A.; Saied, E.; Eid, A.M.; Kouadri, F.; Alemam, A.M.; Hamza, M.F.; Alharbi, M.; Elkelish, A.; Hassan, S.E.-D. Green synthesis of zinc oxide nanoparticles using an aqueous extract of Punica granatum for antimicrobial and catalytic activity. J. Funct. Biomater. 2023, 14, 205. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, M.; Kumar, S.; Gaur, J.; Kaushal, S.; Dalal, J.; Singh, G.; Misra, M.; Ahlawat, D.S. Green synthesis of ZnO nanoparticles using Justicia adhatoda for photocatalytic degradation of malachite green and reduction of 4-nitrophenol. RSC Adv. 2025, 15, 2958–2980. [Google Scholar] [CrossRef]
- Ríos-Ramírez, S.d.C.; Valle, E.-d.; Raymundo, J.; Rodríguez-Ortiz, G.; Ruíz-Luna, J.; Velasco-Velasco, V.A. Growth of Agave angustifolia Haw. In relation to its nutritional condition. Rev. Mex. Cienc. Agríc. 2021, 12, 865–873. [Google Scholar]
- Babayevska, N.; Przysiecka, Ł.; Iatsunskyi, I.; Nowaczyk, G.; Jarek, M.; Janiszewska, E.; Jurga, S. ZnO size and shape effect on antibacterial activity and cytotoxicity profile. Sci. Rep. 2022, 12, 8148, Erratum in Sci. Rep. 2023, 13, 12465. http://doi.org/10.1038/s41598-023-39615-3. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- El-Habib, I.; Maatouk, H.; Lemarchand, A.; Dine, S.; Roynette, A.; Mielcarek, C.; Traoré, M.; Azouani, R. Antibacterial size effect of ZnO nanoparticles and their role as additives in emulsion waterborne paint. J. Funct. Biomater. 2024, 15, 195. [Google Scholar] [CrossRef]
- Irede, E.L.; Awoyemi, R.F.; Owolabi, B.; Aworinde, O.R.; Kajola, R.O.; Hazeez, A.; Raji, A.A.; Ganiyu, L.O.; Onukwuli, C.O.; Onivefu, A.P. Cutting-edge developments in zinc oxide nanoparticles: Synthesis and applications for enhanced antimicrobial and UV protection in healthcare solutions. RSC Adv. 2024, 14, 20992–21034, Erratum in RSC Adv. 2025, 15, 35727. http://doi.org/10.1039/d5ra90105g. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nan, J.; Chu, Y.; Guo, R.; Chen, P. Research on the antibacterial properties of nanoscale zinc oxide particles comprehensive review. Front. Mater. 2024, 11, 1449614, Erratum in Front. Mater. 2024, 11, 1449614. http://doi.org/10.3389/fmats.2024.1449614. [Google Scholar] [PubMed]
- Natural Resources; Species Survival Commission. IUCN Red List Categories and Criteria; IUCN: Gland, Switzerland, 2001. [Google Scholar]









| Plant Source | Part Used | Shape of NPs | Size (nm) | Application | Reference |
|---|---|---|---|---|---|
| Lemongrass (Cymbopogon olivieri) | Leaves | Spherical | 28 | Antimicrobial and Anticancer Activity | [32] |
| Rain of Gold (Thryallis glauca) | Leaves | Hexagonal Wurtzite | 50 | Antioxidants and Antibacterial Activity | [33] |
| Veldt Grape (Cissus quadrangularis) | Stem | Spherical | 75–90 | Antibacterial and Anticancer Activity | [34] |
| Golden Shower Tree (Cassia fistula) | Leaves | Spherical | 68 | Antibacterial Activity | [35] |
| Lemon (Citrus limon) | Fruit | Cuboid, Hexagonal Prism, Thin Rods | 60.8 | Antibacterial and Antihemolytic Activity | [36] |
| Pennyroyal (Mentha pulegium) | Leaves | Semi Spherical | 40 | Antimicrobial Activity | [37] |
| Cauliflower (Brassica Var. botrytis) | Leaves | Flower Like | 52 | Antimicrobial Larvicidal Activity | [38] |
| Tasmanian Blue Gum (Eucalyptus globules) | Leaves | Spherical | 52–70 | Antifungal Activity | [39] |
| Syrian Mesquite (Prosopis farcta) | Aerial | Hexagonal | 40–80 | Antifungal and Breast Cancer (MCF-7) Activity | [40] |
| Sandalwood (Santalum album) | Leaves | Nanorods | 100 | Brest Cancer (MCF-7) Activity | [41] |
| Caper Bush (C. spinosa L.) | Fruit | Spherical | 37.49 | Antioxidant Activity | [42] |
| Garden Cress (Lepidium sativum) | Seeds | Spherical | 37–45 | Anticancer Activity | [43] |
| Gangotra (Cyathocline purpurea) | Leaves | Spherical | 80–120 | Antimicrobial Activity | [44] |
| Maddu Toppu (Justicia wynaadensis) | Leaves | Hexagonal Wurtzite | 39 | Antimitotic and DNA-Binding | [45] |
| Arrowleaf Sida (Sida rhombifolia Linn) | Leaves | Spherical | 30.23 | Genotoxic and Antibacterial Activity | [46] |
| White Passionflower (Passiflora subpeltata) | Leaves | Hexagonal | 45–50 | Antibacterial Activity | [47] |
| Cape Leadwort (Plumbago auriculata) | Aerial | Hexagonal | 38.3 | Antiviral Activity | [48] |
| Radish (Raphanus sativus) | Leaves | Spherical/Hexagonal | 66.47 | Breast Cancer Cells Antibacterial Activity | [49] |
| Black Nightshade (Solanum nigrum) | Leaves | Quasi-Spherical | 30 | Anticancer Activity | [50] |
| Bay Laurel (Laurus nobilis) | Leaves | Flower | 47.27 | Antibacterial Activity | [51] |
| Sweet Leaf (Stevia) | Leaves | Rectangular | 50 | Antimicrobial Wound-healing Bandages | [52] |
| Horseradish Tree (Moringa oleifera) | Leaves | Spherical | 52.24 | Antibacterial and Antioxidant Activity | [53] |
| Roselle (Hibiscus subdariffa) | Leaves | Dumbbell | 190 | Antibacterial and Antidiabetic Activity | [54] |
| Nanomaterials Concentrations (μg/mL) | Zone of Inhibition (mm) | |||||
|---|---|---|---|---|---|---|
| ZnO-S | ZnO-H | ZnO-L | ||||
| S. aureus | E. coli | S. aureus | E. coli | S. aureus | E. coli | |
| 5 | 14.52 ± 0.06 | 13.27 ± 0.30 | 10.93 ± 0.64 | 9.37 ± 0.23 | 9.48 ± 0.31 | 8.76 ± 0.78 |
| 10 | 15.48 ± 0.13 | 14.55 ± 0.73 | 11.70 ± 0.68 | 10.48 ± 0.47 | 10.88 ± 0.21 | 9.69 ± 0.32 |
| 20 | 16.05 ± 0.27 | 15.21 ± 0.44 | 12.44 ± 0.43 | 12.21 ± 0.59 | 11.44 ± 0.30 | 10.92 ± 0.50 |
| 30 | 17.37 ± 0.17 | 17.45 ± 0.48 | 13.57 ± 0.45 | 13.62 ± 0.53 | 11.83 ± 0.35 | 12.69 ± 0.82 |
| 40 | 18.34 ± 0.18 | 18.55 ± 0.46 | 14.41 ± 0.26 | 15.03 ± 0.61 | 12.27 ± 0.39 | 14.50 ± 0.27 |
| 50 | 21.49 ± 0.52 | 20.18 ± 0.76 | 18.02 ± 0.55 | 17.96 ± 0.53 | 14.24 ± 0.68 | 15.92 ± 0.19 |
| Control (Kanamycin) | 23.14 ± 0.38 | 22.53 ± 0.30 | 23.40 ± 0.56 | 19.19 ± 0.72 | 22.78 ± 0.69 | 17.87 ± 0.69 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Channa, G.M.; Bhatti, A.; Sotelo, J.G.; Obregón, S.; Sánchez-Arreola, E.; Mejía-Méndez, J.L.; Navarro-López, D.E.; López-Mena, E.R.; Sánchez-López, A.L.; Lozano, L.M. Unlocking the Hidden Potential of Agave tequilana for the Green Synthesis of Antibacterial ZnO Nanomaterials: A Waste-to-Value Nanotechnology Approach. Int. J. Mol. Sci. 2025, 26, 11545. https://doi.org/10.3390/ijms262311545
Channa GM, Bhatti A, Sotelo JG, Obregón S, Sánchez-Arreola E, Mejía-Méndez JL, Navarro-López DE, López-Mena ER, Sánchez-López AL, Lozano LM. Unlocking the Hidden Potential of Agave tequilana for the Green Synthesis of Antibacterial ZnO Nanomaterials: A Waste-to-Value Nanotechnology Approach. International Journal of Molecular Sciences. 2025; 26(23):11545. https://doi.org/10.3390/ijms262311545
Chicago/Turabian StyleChanna, Ghulam Mustafa, Atiya Bhatti, Juan G. Sotelo, Sergio Obregón, Eugenio Sánchez-Arreola, Jorge L. Mejía-Méndez, Diego E. Navarro-López, Edgar R. López-Mena, Angélica Lizeth Sánchez-López, and Luis Marcelo Lozano. 2025. "Unlocking the Hidden Potential of Agave tequilana for the Green Synthesis of Antibacterial ZnO Nanomaterials: A Waste-to-Value Nanotechnology Approach" International Journal of Molecular Sciences 26, no. 23: 11545. https://doi.org/10.3390/ijms262311545
APA StyleChanna, G. M., Bhatti, A., Sotelo, J. G., Obregón, S., Sánchez-Arreola, E., Mejía-Méndez, J. L., Navarro-López, D. E., López-Mena, E. R., Sánchez-López, A. L., & Lozano, L. M. (2025). Unlocking the Hidden Potential of Agave tequilana for the Green Synthesis of Antibacterial ZnO Nanomaterials: A Waste-to-Value Nanotechnology Approach. International Journal of Molecular Sciences, 26(23), 11545. https://doi.org/10.3390/ijms262311545











