Tryptophan and Substance Abuse: Mechanisms and Impact
Abstract
1. Introduction
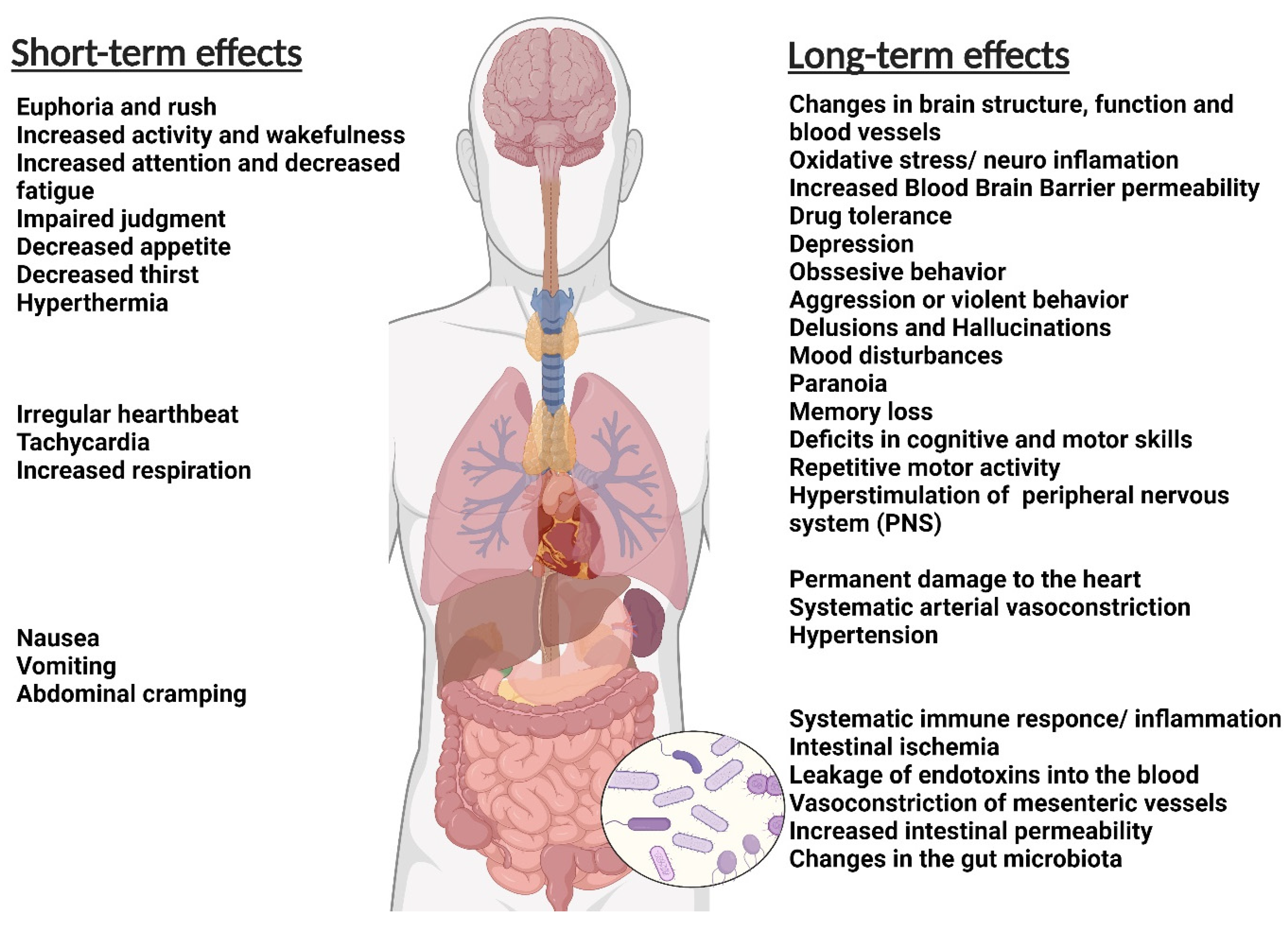
2. General Effects of Addiction on the Body
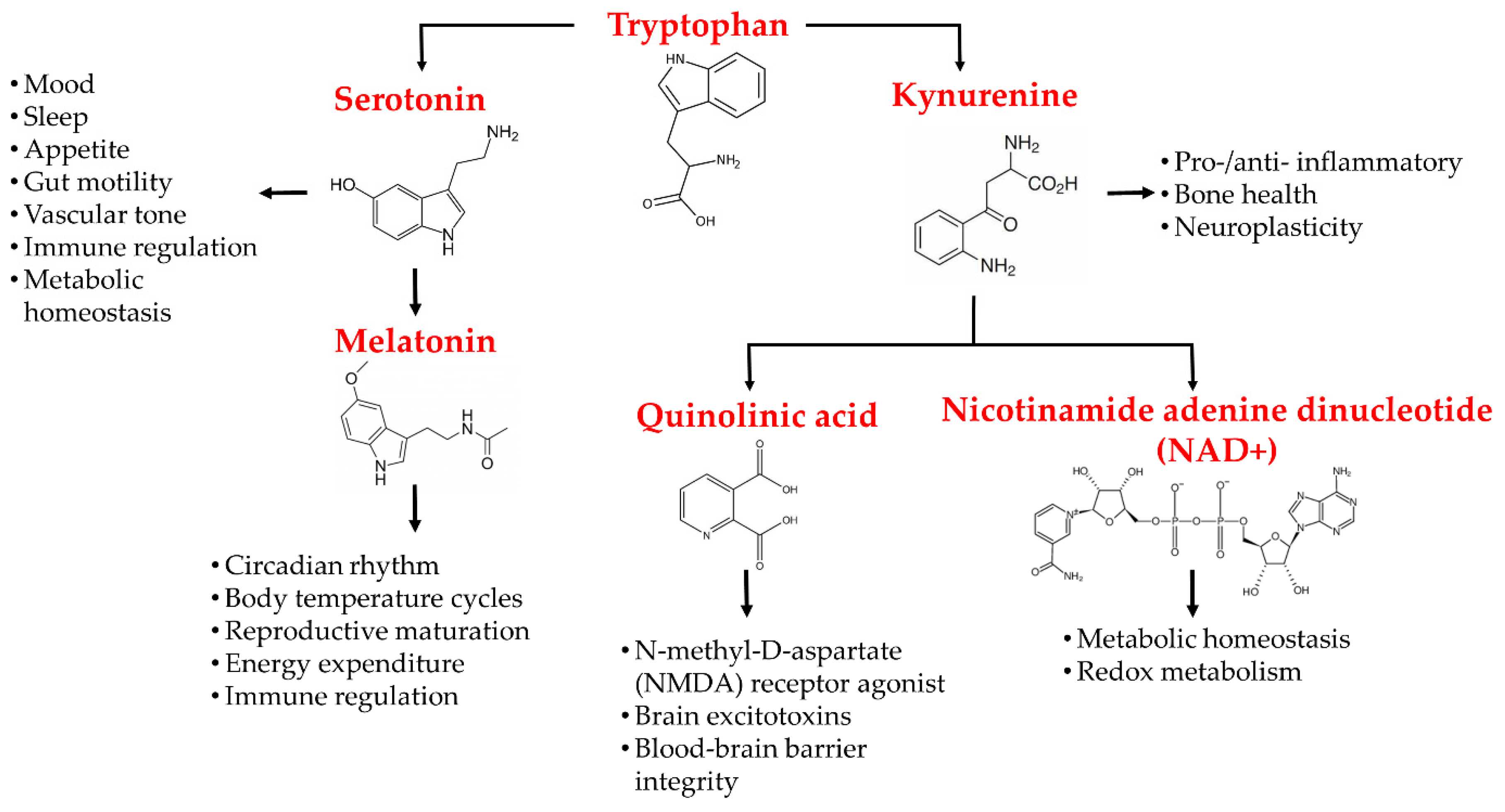
3. Tryptophan and Its Metabolites
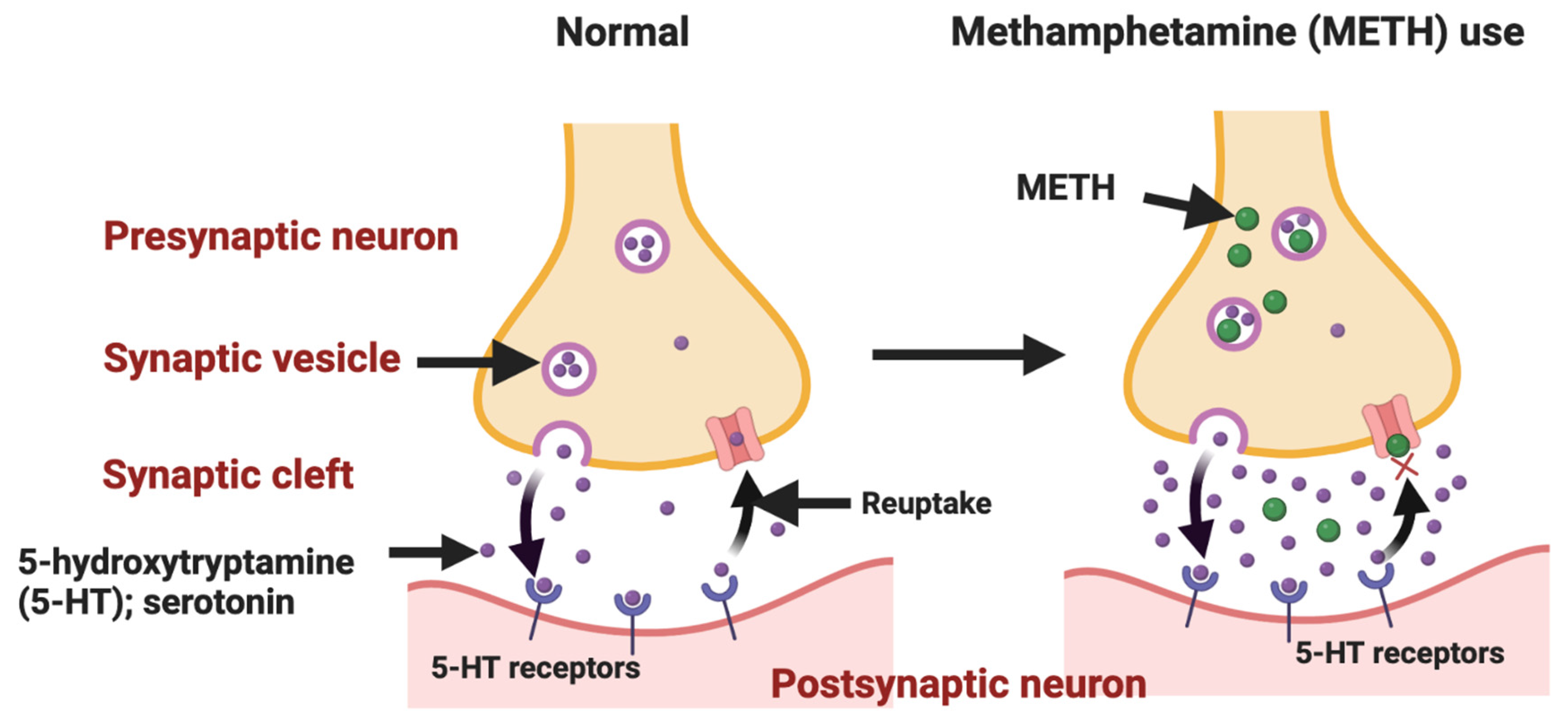
4. Tryptophan and Addiction: METH Addiction as an Example
4.1. Serotonin (5-hydroxytryptophan)
4.2. Melatonin
4.3. Kynurenine
4.4. Kynurenic Acid
4.5. Xanthurenic Acid
4.6. 3-hydroxyanthranillic Acid
4.7. Quinolinic Acid
4.8. Picolinic Acid
4.9. Nicotinamide Adenine Dinucleotide (NAD+)
4.10. Reactive Oxygen and Nitrogen Species
5. Tryptophan Metabolites and Treatment Options for Addiction
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goldstein, R.; Volkow, N. Dysfunktion av prefrontal cortex i missbruk: Neuroimaging fynd och kliniska konsekvenser. Nat. Rev. Neurosci. 2011, 12, 652–669. [Google Scholar] [CrossRef]
- Volkow, N.D.; Li, T.-K. Drug addiction: The neurobiology of behaviour gone awry. Nat. Rev. Neurosci. 2004, 5, 963–970. [Google Scholar] [CrossRef]
- Barnard, M. Drug Addiction and Families; Jessica Kingsley Publishers: London, UK, 2006. [Google Scholar]
- Ihezie, S.A.; Dafny, N. Prevention of Opioid Addiction. J. ISSN 2021, 2766, 2276. [Google Scholar] [CrossRef]
- Pantoni, M.M.; Kim, J.L.; Van Alstyne, K.R.; Anagnostaras, S.G. MDMA and memory, addiction, and depression: Dose-effect analysis. Psychopharmacology 2022, 239, 935–949. [Google Scholar] [CrossRef] [PubMed]
- Heilig, M.; Augier, E.; Pfarr, S.; Sommer, W.H. Developing neuroscience-based treatments for alcohol addiction: A matter of choice? Transl. Psychiatry 2019, 9, 255. [Google Scholar] [CrossRef]
- Adams, M.K.; Chong, E.W.; Williamson, E.; Aung, K.Z.; Makeyeva, G.A.; Giles, G.G.; English, D.R.; Hopper, J.; Guymer, R.H.; Baird, P.N. 20/20—Alcohol and age-related macular degeneration: The Melbourne Collaborative Cohort Study. Am. J. Epidemiol. 2012, 176, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Nwokike, M.; Anusiem, C.; Arinze, C.; Ogbonna, A. The Abuse of Central Nervous System Stimulants and its Impact on the Youth of Eastern Nigeria. Int. Neuropsychiatr. Dis. J. 2021, 15, 20–33. [Google Scholar] [CrossRef]
- White, E.; Comiskey, C. Heroin epidemics, treatment and ODE modelling. Math. Biosci. 2007, 208, 312–324. [Google Scholar] [CrossRef] [PubMed]
- Carmack, S.A.; Keeley, R.J.; Vendruscolo, J.C.; Lowery-Gionta, E.G.; Lu, H.; Koob, G.F.; Stein, E.A.; Vendruscolo, L.F. Heroin addiction engages negative emotional learning brain circuits in rats. J. Clin. Investig. 2019, 129, 2480–2484. [Google Scholar] [CrossRef]
- Ritter, A.; Cao, V.L.; Shanahan, M.; Shukla, N.; Perez, P.; Farrell, M. Modelling heroin careers over 40 years: Social costs. Drug Alcohol Depend. 2017, 100, e177. [Google Scholar] [CrossRef]
- Walsh, J.M. Are we there yet? Measuring Progress in the US War on Drugs in Latin America. WOLA Drug War Monit. 2004, 8. [Google Scholar]
- Hser, Y.-I. Predicting long-term stable recovery from heroin addiction: Findings from a 33-year follow-up study. J. Addict. Dis. 2007, 26, 51–60. [Google Scholar] [CrossRef]
- Dani, J.A.; Harris, R.A. Nicotine addiction and comorbidity with alcohol abuse and mental illness. Nat. Neurosci. 2005, 8, 1465–1470. [Google Scholar] [CrossRef] [PubMed]
- Scarlata, M.; Keeley, R.; Stein, E. Nicotine addiction: Translational insights from circuit neuroscience. Pharmacol. Biochem. Behav. 2021, 204, 173171. [Google Scholar] [CrossRef] [PubMed]
- Fagerström, K. Nicotine: Pharmacology, toxicity and therapeutic use. J. Smok. Cessat. 2014, 9, 53–59. [Google Scholar] [CrossRef]
- Jetton, J.A.; Ding, K.; Kim, Y.; Stone, D.U. Effects of tobacco smoking on human corneal wound healing. Cornea 2014, 33, 453–456. [Google Scholar] [CrossRef]
- Antonelli, M.; Ferrulli, A.; Sestito, L.; Vassallo, G.A.; Tarli, C.; Mosoni, C.; Rando, M.M.; Mirijello, A.; Gasbarrini, A.; Addolorato, G. Alcohol addiction-the safety of available approved treatment options. Expert Opin. Drug Saf. 2018, 17, 169–177. [Google Scholar] [CrossRef]
- Gibbins, R.J. Chronic Alcoholism and Alcohol Addiction; University of Toronto Press: Toronto, ON, USA, 2019. [Google Scholar]
- Behling, M.; Winters, B. Methods to Stop Caffeine Use and Minimize Caffeine Withdrawal Symptoms in the State of Caffeine Dependence: A Literature Review. Stud. Works 2021. Available online: https://scholarsarchive.byu.edu/cgi/viewcontent.cgi?article=1363&context=studentpub (accessed on 3 January 2023).
- Favrod-Coune, T.; Broers, B. Addiction to caffeine and other xanthines. In Textbook of Addiction Treatment; Springer: Berlin/Heidelberg, Germany, 2021; pp. 215–228. [Google Scholar]
- Alasmari, F. Caffeine induces neurobehavioral effects through modulating neurotransmitters. Saudi Pharm. J. 2020, 28, 445–451. [Google Scholar] [CrossRef]
- Chandio, Z.A.; Sidiqua, A.; Khaskheli, M.I.; Waghani, A.; Metlo, W.A. Review Effect of Caffeine Overdose. RADS J. Biol. Res. Appl. Sci. 2020, 11, 152–156. [Google Scholar] [CrossRef]
- Riezzo, I.; Fiore, C.; De Carlo, D.; Pascale, N.; Neri, M.; Turillazzi, E.; Fineschi, V. Side effects of cocaine abuse: Multiorgan toxicity and pathological consequences. Curr. Med. Chem. 2012, 19, 5624–5646. [Google Scholar] [CrossRef]
- Pierce, R.C.; Fant, B.; Swinford-Jackson, S.E.; Heller, E.A.; Berrettini, W.H.; Wimmer, M.E. Environmental, genetic and epigenetic contributions to cocaine addiction. Neuropsychopharmacology 2018, 43, 1471–1480. [Google Scholar] [CrossRef]
- Valencia-Alfonso, C.-E.; Luigjes, J.; Smolders, R.; Cohen, M.X.; Levar, N.; Mazaheri, A.; van den Munckhof, P.; Schuurman, P.R.; van den Brink, W.; Denys, D. Effective deep brain stimulation in heroin addiction: A case report with complementary intracranial electroencephalogram. Biol. Psychiatry 2012, 71, e35–e37. [Google Scholar] [CrossRef] [PubMed]
- Sarajlija, M.; Raketic, D.; Nesic, N. Heroin addiction in Serbian patients with Tourette syndrome. J. Psychiatr. Pract. 2018, 24, 424–427. [Google Scholar] [CrossRef]
- Sabet, K. Marijuana and legalization impacts. Berkeley J. Crim. L. 2018, 23, 84. [Google Scholar]
- Ethan, X.; Logan, A.; Liam, M.; Leonard, J. Impact of Marijuana (Cannabis) on Health, Safety and Economy. Int. Digit. Organ. Sci. Res. (IDOSR) J. Exp. Sci. 2020, 5, 43–52. [Google Scholar]
- Sun, X.; Xu, C.S.; Chadha, N.; Chen, A.; Liu, J. Focus: Addiction: Marijuana for Glaucoma: A Recipe for Disaster or Treatment? Yale J. Biol. Med. 2015, 88, 265–269. [Google Scholar]
- Temmingh, H.S.; van den Brink, W.; Howells, F.; Sibeko, G.; Stein, D.J. Methamphetamine use and antipsychotic-related extrapyramidal side-effects in patients with psychotic disorders. J. Dual Diagn. 2020, 16, 208–217. [Google Scholar] [CrossRef]
- Dhein, S.; Schmelmer, K.; Guenther, J.; Salameh, A. Aspects of methamphetamine abuse in adolescents and young adults in a Thuringian County. Eur. Addict. Res. 2018, 24, 98–105. [Google Scholar] [CrossRef]
- Naji, L.; Dennis, B.; Rosic, T.; Wiercioch, W.; Paul, J.; Worster, A.; Thabane, L.; Samaan, Z. Mirtazapine for the treatment of amphetamine and methamphetamine use disorder: A systematic review and meta-analysis. Drug Alcohol Depend. 2022, 232, 109295. [Google Scholar] [CrossRef]
- Escobar-Chávez, J.J.; Domínguez-Delgado, C.L.; Rodríguez-Cruz, I.M. Targeting nicotine addiction: The possibility of a therapeutic vaccine. Drug Des. Dev. Ther. 2011, 5, 211–224. [Google Scholar] [CrossRef]
- Sliwińska-Mossoń, M.; Zieleń, I.; Milnerowicz, H. New trends in the treatment of nicotine addiction. Acta Pol. Pharm. 2014, 71, 525–530. [Google Scholar] [PubMed]
- Jamali, Q. Galantamine as a Treatment Option for Nicotine Addiction. J. Smok. Cessat. 2021, 2021, 9975811. [Google Scholar] [CrossRef] [PubMed]
- Stewart, S.H.; Walitzer, K.S.; Blanco, J.; Swiatek, D.; Paine Hughes, L.; Quiñones-Lombraña, A.; Shyhalla, K. Medication-enhanced behavior therapy for alcohol use disorder: Naltrexone, Alcoholics Anonymous Facilitation, and OPRM1 genetic variation. J. Subst. Abus. Treat. 2019, 104, 7–14. [Google Scholar] [CrossRef]
- Saunders, E.C.; McGovern, M.P.; Lambert-Harris, C.; Meier, A.; McLeman, B.; Xie, H. The impact of addiction medications on treatment outcomes for persons with co-occurring PTSD and opioid use disorders. Am. J. Addict. 2015, 24, 722–731. [Google Scholar] [CrossRef]
- Kałużna-Czaplińska, J.; Gątarek, P.; Chirumbolo, S.; Chartrand, M.S.; Bjørklund, G. How important is tryptophan in human health? Crit. Rev. Food Sci. Nutr. 2019, 59, 72–88. [Google Scholar] [CrossRef] [PubMed]
- Araos, P.; Vidal, R.; O’Shea, E.; Pedraz, M.; García-Marchena, N.; Serrano, A.; Suárez, J.; Castilla-Ortega, E.; Ruiz, J.J.; Campos-Cloute, R.; et al. Serotonin is the main tryptophan metabolite associated with psychiatric comorbidity in abstinent cocaine-addicted patients. Sci. Rep. 2019, 9, 16842. [Google Scholar] [CrossRef]
- Bamia, C.; Trichopoulou, A.; Lenas, D.; Trichopoulos, D. Tobacco smoking in relation to body fat mass and distribution in a general population sample. Int. J. Obes. 2004, 28, 1091–1096. [Google Scholar] [CrossRef]
- Harris, K.K.; Zopey, M.; Friedman, T.C. Metabolic effects of smoking cessation. Nat. Rev. Endocrinol. 2016, 12, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Arterberry, B.J.; Treloar, H.R.; Smith, A.E.; Martens, M.P.; Pedersen, S.L.; McCarthy, D.M. Marijuana use, driving, and related cognitions. Psychol. Addict. Behav. 2013, 27, 854–860. [Google Scholar] [CrossRef]
- Gottlieb, E. Cannabis: A Danger to the Adolescent Brain—How Pediatricians Can Address Marijuana Use; Citeseer: University Park, PA, USA, 2012. [Google Scholar]
- Roerecke, M.; Vafaei, A.; Hasan, O.S.M.; Chrystoja, B.R.; Cruz, M.; Lee, R.; Neuman, M.G.; Rehm, J. Alcohol Consumption and Risk of Liver Cirrhosis: A Systematic Review and Meta-Analysis. Am. J. Gastroenterol. 2019, 114, 1574–1586. [Google Scholar] [CrossRef]
- Nawrot, P.; Jordan, S.; Eastwood, J.; Rotstein, J.; Hugenholtz, A.; Feeley, M. Effects of caffeine on human health. Food Addit. Contam. 2003, 20, 1–30. [Google Scholar] [CrossRef]
- Saimaiti, A.; Zhou, D.D.; Li, J.; Xiong, R.G.; Gan, R.Y.; Huang, S.Y.; Shang, A.; Zhao, C.N.; Li, H.Y.; Li, H.B. Dietary sources, health benefits, and risks of caffeine. Crit. Rev. Food Sci. Nutr. 2022, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Hswen, Y.; Zhang, A.; Brownstein, J.S. Estimating the incidence of cocaine use and mortality with music lyrics about cocaine. NPJ Digit. Med. 2021, 4, 100. [Google Scholar] [CrossRef] [PubMed]
- Won, S.; Hong, R.A.; Shohet, R.V.; Seto, T.B.; Parikh, N.I. Methamphetamine-Associated Cardiomyopathy. Clin. Cardiol. 2013, 36, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Davidson, M.; Mayer, M.; Habib, A.; Rashidi, N.; Filippone, R.T.; Fraser, S.; Prakash, M.D.; Sinnayah, P.; Tangalakis, K.; Mathai, M.L.; et al. Methamphetamine Induces Systemic Inflammation and Anxiety: The Role of the Gut–Immune–Brain Axis. Int. J. Mol. Sci. 2022, 23, 11224. [Google Scholar] [CrossRef]
- Sulzer, D.; Sonders, M.S.; Poulsen, N.W.; Galli, A. Mechanisms of neurotransmitter release by amphetamines: A review. Prog. Neurobiol. 2005, 75, 406–433. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.; Wang, C.; Zhang, X.; Pestell, R.G. Acetylation of nuclear receptors in cellular growth and apoptosis. Biochem. Pharmacol. 2004, 68, 1199–1208. [Google Scholar] [CrossRef]
- Prakash, M.D.; Tangalakis, K.; Antonipillai, J.; Stojanovska, L.; Nurgali, K.; Apostolopoulos, V. Methamphetamine: Effects on the brain, gut and immune system. Pharmacol. Res. 2017, 120, 60–67. [Google Scholar] [CrossRef]
- Papageorgiou, M.; Raza, A.; Fraser, S.; Nurgali, K.; Apostolopoulos, V. Methamphetamine and its immune-modulating effects. Maturitas 2019, 121, 13–21. [Google Scholar] [CrossRef]
- Surratt, C.K.; Ukairo, O.T.; Ramanujapuram, S. Recognition of psychostimulants, antidepressants, and other inhibitors of synaptic neurotransmitter uptake by the plasma membrane monoamine transporters. AAPS J. 2005, 7, E739–E751. [Google Scholar] [CrossRef]
- Darke, S.; Kaye, S.; McKetin, R.; Duflou, J. Major physical and psychological harms of methamphetamine use. Drug Alcohol Rev. 2009, 27, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Teixeira-Gomes, A.; Costa, V.M.; Feio-Azevedo, R.; de Lourdes Bastos, M.; Carvalho, F.; Capela, J.P. The neurotoxicity of amphetamines during the adolescent period. Int. J. Dev. Neurosci. 2015, 41, 44–62. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, C.; Rose Childress, A.; Thomas Mclellan, A.; Ehrman, R. Classical Conditioning in Drug-Dependent Humans. Ann. N. Y. Acad. Sci. 1992, 654, 400–415. [Google Scholar] [CrossRef]
- Danaceau, J.P.; Deering, C.E.; Day, J.E.; Smeal, S.J.; Johnson-Davis, K.L.; Fleckenstein, A.E.; Wilkins, D.G. Persistence of tolerance to methamphetamine-induced monoamine deficits. Eur. J. Pharmacol. 2007, 559, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Carrico, A.; Johnson, M.; Morin, S.F.; Remien, R.; Riley, E.D.; Hecht, F.; Fuchs, D. Stimulant Use is Associated with Immune Activation and Depleted Tryptophan Among HIV-Positive Persons on Anti-Retroviral Therapy. Brain Behav. Immun. 2008, 22, 1257–1262. [Google Scholar] [CrossRef] [PubMed]
- Bradley, K.; Case, J.A.C.; Khan, O.; Ricart, T.; Hanna, A.; Alonso, C.; Gabbay, V. The role of the kynurenine pathway in suicidality in adolescent major depressive disorder. Psychiatry Res. 2015, 227, 206–212. [Google Scholar] [CrossRef]
- Fleckenstein, A.E.; Volz, T.J.; Riddle, E.L.; Gibb, J.W.; Hanson, G.R. New Insights into the Mechanism of Action of Amphetamines. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 681–698. [Google Scholar] [CrossRef]
- Halpin, L.E.; Collins, S.A.; Yamamoto, B.K. Neurotoxicity of methamphetamine and 3,4-methylenedioxymethamphetamine. Life Sci. 2014, 97, 37–44. [Google Scholar] [CrossRef]
- Fiore, A.; Murray, P.J. Tryptophan and indole metabolism in immune regulation. Curr. Opin. Immunol. 2021, 70, 7–14. [Google Scholar] [CrossRef]
- Davidson, M.; Rashidi, N.; Nurgali, K.; Apostolopoulos, V. The Role of Tryptophan Metabolites in Neuropsychiatric Disorders. Int. J. Mol. Sci. 2022, 23, 9968. [Google Scholar] [CrossRef]
- Bryleva, E.; Brundin, L. Kynurenine Pathway Metabolites and Suicidality. Neuropharmacology 2017, 112, 324–330. [Google Scholar] [CrossRef]
- Engin, A.; Engin, A. Tryptophan Metabolism: Implications for Biological Processes, Health and Disease; Humana Press: Totowa, NJ, USA, 2015. [Google Scholar] [CrossRef]
- Petit, A.; Karila, L.; Chalmin, F.; Lejoyeux, M. Methamphetamine addiction: A review of the literature. J. Addict. Res. Ther. S 2012, 1, 1–6. [Google Scholar] [CrossRef]
- Savitz, J. The kynurenine pathway: A finger in every pie. Mol. Psychiatry 2020, 25, 131–147. [Google Scholar] [CrossRef] [PubMed]
- Platten, M.; Nollen, E.A.; Röhrig, U.F.; Fallarino, F.; Opitz, C.A. Tryptophan metabolism as a common therapeutic target in cancer, neurodegeneration and beyond. Nat. Rev. Drug Discov. 2019, 18, 379–401. [Google Scholar] [CrossRef]
- Cheong, J.E.; Ekkati, A.; Sun, L. A patent review of IDO1 inhibitors for cancer. Expert Opin. Ther. Pat. 2018, 28, 317–330. [Google Scholar] [CrossRef] [PubMed]
- Andersen, M.H. The targeting of tumor-associated macrophages by vaccination. Cell Stress 2019, 3, 139. [Google Scholar] [CrossRef] [PubMed]
- Borish, L.C.; Steinke, J.W. 2. Cytokines and chemokines. J. Allergy Clin. Immunol. 2003, 111, S460–S475. [Google Scholar] [CrossRef] [PubMed]
- Van der Meide, P.H.; Schellekens, H. Cytokines and the immune response. Biotherapy 1996, 8, 243–249. [Google Scholar] [CrossRef]
- Li, Y.; Hu, N.; Yang, D.; Oxenkrug, G.; Yang, Q. Regulating the balance between the kynurenine and serotonin pathways of tryptophan metabolism. FEBS J. 2017, 284, 948–966. [Google Scholar] [CrossRef]
- Maddison, D.C.; Giorgini, F. The kynurenine pathway and neurodegenerative disease. Semin. Cell Dev. Biol. 2015, 40, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Boyen, G.; Steinkamp, M.; Reinshagen, M.; Schäfer, K.-H.; Adler, G.; Kirsch, J. Proinflammatory cytokines increase glial fibrillary acidic protein expression in enteric glia. Gut 2004, 53, 222–228. [Google Scholar] [CrossRef]
- Gibney, G.T.; Hamid, O.; Lutzky, J.; Olszanski, A.J.; Mitchell, T.C.; Gajewski, T.F.; Chmielowski, B.; Hanks, B.A.; Zhao, Y.; Newton, R.C. Phase 1/2 study of epacadostat in combination with ipilimumab in patients with unresectable or metastatic melanoma. J. Immunother. Cancer 2019, 7, 80. [Google Scholar] [CrossRef] [PubMed]
- Gomes, B.; Driessens, G.; Bartlett, D.; Cai, D.; Cauwenberghs, S.; Crosignani, S.; Dalvie, D.; Denies, S.; Dillon, C.P.; Fantin, V.R. Characterization of the selective indoleamine 2, 3-dioxygenase-1 (IDO1) catalytic inhibitor EOS200271/PF-06840003 supports IDO1 as a critical resistance mechanism to PD-(L) 1 blockade therapy. Mol. Cancer Ther. 2018, 17, 2530–2542. [Google Scholar] [CrossRef]
- Hassanzadeganroudsari, M.; Soltani, M.; Heydarinasab, A.; Nakhjiri, A.T.; Hossain, M.D.K.; Khiyavi, A.A. Mathematical modeling and simulation of molecular mass transfer across blood brain barrier in brain capillary. J. Mol. Liq. 2020, 310, 113254. [Google Scholar] [CrossRef]
- Gobaille, S.; Kemmel, V.; Brumaru, D.; Dugave, C.; Aunis, D.; Maitre, M. Xanthurenic acid distribution, transport, accumulation and release in the rat brain. J. Neurochem. 2008, 105, 982–993. [Google Scholar] [CrossRef] [PubMed]
- Dostal, C.; Carson Sulzer, M.; Kelley, K.W.; Freund, G.; McCusker, R.H. Glial and tissue-specific regulation of Kynurenine Pathway dioxygenases by acute stress of mice. Neurobiol. Stress 2017, 7, 1–15. [Google Scholar] [CrossRef]
- Koshiguchi, M.; Komazaki, H.; Hirai, S.; Egashira, Y. Ferulic acid suppresses expression of tryptophan metabolic key enzyme indoleamine 2, 3-dioxygenase via NFκB and p38 MAPK in lipopolysaccharide-stimulated microglial cells. Biosci. Biotechnol. Biochem. 2017, 81, 966–971. [Google Scholar] [CrossRef] [PubMed]
- Badawy, A.A.B. Tryptophan metabolism in alcoholism. Nutr. Res. Rev. 2002, 15, 123–152. [Google Scholar] [CrossRef]
- Merz, F. United Nations Office on Drugs and Crime: World Drug Report 2017. 2017. SIRIUS-Z. Strateg. Anal. 2018, 2, 85–86. [Google Scholar] [CrossRef]
- Azmi, A.; Jai, J.; Zamanhuri, N.; Yahya, A. Precious metals recovery from electroplating wastewater: A review. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2018; p. 012024. [Google Scholar]
- Vrajová, M.; Šlamberová, R.; Hoschl, C.; Ovsepian, S.V. Methamphetamine and sleep impairments: Neurobehavioral correlates and molecular mechanisms. Sleep 2021, 44, zsab001. [Google Scholar] [CrossRef] [PubMed]
- Rawson, R.A.; Gonzales, R.; Marinelli-Casey, P.; Ang, A. Methamphetamine dependence: A closer look at treatment response and clinical characteristics associated with route of administration in outpatient treatment. Am. J. Addict. 2007, 16, 291–299. [Google Scholar] [CrossRef]
- McKetin, R.; Voce, A.; Burns, R.; Shanahan, M. Health-related quality of life among people who use methamphetamine. Drug Alcohol Rev. 2019, 38, 503–509. [Google Scholar] [CrossRef] [PubMed]
- McKetin, R.; Lubman, D.I.; Lee, N.M.; Ross, J.E.; Slade, T.N. Major depression among methamphetamine users entering drug treatment programs. Med. J. Aust. 2011, 195, S51–S55. [Google Scholar] [CrossRef]
- Hoffman, W.F.; Jacobs, M.B.; Dennis, L.E.; McCready, H.D.; Hickok, A.W.; Smith, S.B.; Kohno, M. Psychopathy and corticostriatal connectivity: The link to criminal behavior in methamphetamine dependence. Front. Psychiatry 2020, 11, 90. [Google Scholar] [CrossRef]
- Newton, T.F.; De La Garza, R.; Kalechstein, A.D.; Tziortzis, D.; Jacobsen, C.A. Theories of addiction: Methamphetamine users’ explanations for continuing drug use and relapse. Am. J. Addict. 2009, 18, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Filip, M.; Bader, M. Overview on 5-HT receptors and their role in physiology and pathology of the central nervous system. Pharmacol. Rep. 2009, 61, 761–777. [Google Scholar] [CrossRef]
- Haughey, H.; Fleckenstein, A.E.; Hanson, G.R.; Haughey, H.M.; Fleckenstein, A.E.; Hanson, G.R. Differential regional effects of methampetamine on the activities of tryptophan and tyrosine hydroxylase. J. Neurochem. 1999, 72, 661–668. [Google Scholar] [CrossRef]
- Kobayashi, H.; Ujike, H.; Iwata, N.; Inada, T.; Yamada, M.; Sekine, Y.; Uchimura, N.; Iyo, M.; Ozaki, N.; Itokawa, M.; et al. Association Analysis of the Tryptophan Hydroxylase 2 Gene Polymorphisms in Patients with Methamphetamine Dependence/Psychosis. Curr. Neuropharmacol. 2011, 9, 176–182. [Google Scholar] [CrossRef]
- Sitte, H.; Freissmuth, M. Amphetamines, new psychoactive drugs and the monoamine transporter cycle. Trends Pharmacol. Sci. 2015, 36, 41–50. [Google Scholar] [CrossRef]
- Hasler, B.; Smith, L.J.; Cousins, J.; Bootzin, R. Circadian Rhythms, Sleep, and Substance Abuse. Sleep Med. Rev. 2012, 16, 67–81. [Google Scholar] [CrossRef]
- Andersen, M.; Perez Diaz, M.; Murnane, K.; Howell, L.L. Effects of methamphetamine self-administration on actigraphy-based sleep parameters in rhesus monkeys. Psychopharmacology 2013, 227, 101–107. [Google Scholar] [CrossRef]
- Kuwano, N.; Kato, T.; Setoyama, D.; Sato-Kasai, M.; Shimokawa, N.; Hayakawa, K.; Ohgidani, M.; Sagata, N.; Kubo, H.; Kishimoto, J.; et al. Tryptophan-kynurenine and lipid related metabolites as blood biomarkers for first-episode drug-naïve patients with major depressive disorder: An exploratory pilot case-control study. J. Affect. Disord. 2018, 231, 74–82. [Google Scholar] [CrossRef]
- Stone, T.; Stoy, N.; Gail Darlington, L. An expanding range of targets for kynurenine metabolites of tryptophan. Trends Pharmacol. Sci. 2013, 34, 136–143. [Google Scholar] [CrossRef]
- Rassoulpour, A.; Wu, H.Q.; Poeggeler, B.; Schwarcz, R. Systemic d-amphetamine administration causes a reduction of kynurenic acid levels in rat brain. Brain Res. 1998, 802, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Carlborg, A.; Jokinen, J.; Jönsson, E.; Erhardt, S.; Nordstrom, P. CSF kynurenic acid and suicide risk in schizophrenia spectrum psychosis. Psychiatry Res. 2013, 205, 165–167. [Google Scholar] [CrossRef] [PubMed]
- Shimosato, K.; Nagao, N.; Watanabe, S.; Kitayama, S. Suppressive effects of trihexyphenidyl on methamphetamine-induced DA release as measured by in vivo microdialysis. Synapse 2003, 49, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Scofield, M. Similitude in Methamphetamine-Induced Neuroadaptations Across Susceptibility and Chronic Drug Exposure Paradigms. Biol. Psychiatry 2017, 81, e83–e84. [Google Scholar] [CrossRef]
- Haruki, H.; Hovius, R.; Grønlund Pedersen, M.; Johnsson, K. Tetrahydrobiopterin Biosynthesis as a Potential Target of the Kynurenine Pathway Metabolite Xanthurenic Acid. J. Biol. Chem. 2016, 291, 652–657. [Google Scholar] [CrossRef]
- Stone, T. Kynurenines in the CNS: From endogenous obscurity to therapeutic importance. Prog. Neurobiol. 2001, 64, 185–218. [Google Scholar]
- Fazio, F.; Lionetto, L.; Curto, M.; Iacovelli, L.; Copeland, C.S.; Neale, S.A.; Bruno, V.; Battaglia, G.; Salt, T.; Nicoletti, F. Cinnabarinic acid and xanthurenic acid: Two kynurenine metabolites that interact with metabotropic glutamate receptors. Neuropharmacology 2017, 112, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Stone, T. Inhibitors of the Kynurenine Pathway. Eur. J. Med. Chem. 2000, 35, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, C.; Grando, V.; Dutra Filho, C.S.; Wannmacher, C.; Wajner, M. Evidence that quinolinic acid severely impairs energy metabolism through activation of NMDA receptors in striatum from developing rats. J. Neurochem. 2006, 99, 1531–1542. [Google Scholar] [CrossRef] [PubMed]
- Guillemin, G. Quinolinic acid: Neurotoxicity. FEBS J. 2012, 279, 1355. [Google Scholar] [CrossRef]
- Erhardt, S.; Lim, C.; Linderholm, K.; Janelidze, S.; Lindqvist, D.; Samuelsson, M.; Lundberg, K.; Postolache, T.; Träskman-Bendz, L.; Guillemin, G.; et al. Connecting Inflammation with Glutamate Agonism in Suicidality. Neuropsychopharmacology 2012, 38, 743–752. [Google Scholar] [CrossRef]
- Bay-Richter, C.; Linderholm, K.; Lim, C.; Samuelsson, M.; Träskman-Bendz, L.; Guillemin, G.; Erhardt, S.; Brundin, L. A role for inflammatory metabolites as modulators of the glutamate N-methyl-D-aspartate receptor in depression and suicidality. Brain Behav. Immun. 2015, 43, 110–117. [Google Scholar] [CrossRef]
- Van Heeringen, K.; Bijttebier, S.; Desmyter, S.; Vervaet, M.; Baeken, C. Is there a neuroanatomical basis of the vulnerability to suicidal behavior? A coordinate-based meta-analysis of structural and functional MRI studies. Front. Hum. Neurosci. 2014, 8, 824. [Google Scholar] [CrossRef]
- Zarate, C.; Brutsche, N.; Ibrahim, L.; Franco-Chaves, J.; Diazgranados, N.; Cravchik, A.; Selter, J.; Marquardt, C.; Liberty, V.; Luckenbaugh, D. Replication of Ketamine’s Antidepressant Efficacy in Bipolar Depression: A Randomized Controlled Add-On Trial. Biol. Psychiatry 2012, 71, 939–946. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Chen, T.; Wang, J.; Jia, Y.; Ren, H.; Wu, F.; Hu, M.; Chen, Y. Methamphetamine modulates the production of interleukin-6 and tumor necrosis factor-alpha via the cAMP/PKA/CREB signaling pathway in lipopolysaccharide-activated microglia. Int. Immunopharmacol. 2018, 56, 168–178. [Google Scholar] [CrossRef]
- Grant, R.; Coggan, S.; Smythe, G. The Physiological Action of Picolinic Acid in the Human Brain. Int. J. Tryptophan Res. 2009, 2, 71–79. [Google Scholar] [CrossRef]
- Davidson, J.; Abraham, K.; Connor, K.; McLeod, M.N. Effectiveness of Chromium in atypical depression: A placebo-controlled trial. Biol. Psychiatry 2003, 53, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Rex, A.; Schickert, R.; Fink, H. Antidepressant-like effect of nicotinamide adenine dinucleotide in the forced swim test in rats. Pharmacol. Biochem. Behav. 2004, 77, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Oka, S.-I.; Hsu, C.-P.; Sadoshima, J. Regulation of Cell Survival and Death by Pyridine Nucleotides. Circ. Res. 2012, 111, 611–627. [Google Scholar] [CrossRef] [PubMed]
- Salim, S. Oxidative Stress and Psychological Disorders. Curr. Neuropharmacol. 2014, 12, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Gawryluk, J.; Wang, J.-F.; Andreazza, A.C.; Shao, L.; Young, L.T. Decreased levels of glutathione, the major brain antioxidant, in post-mortem prefrontal cortex from patients with psychiatric disorders. Int. J. Neuropsychopharmacol. 2011, 14, 123–130. [Google Scholar] [CrossRef]
- Lee, S.-Y.; Lee, S.-J.; Han, C.; A Patkar, A.; S Masand, P.; Pae, C.-U. Oxidative/nitrosative stress and antidepressants: Targets for novel antidepressants. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2013, 46, 224–235. [Google Scholar] [CrossRef] [PubMed]
- Badawy, A.A.-B. Tryptophan metabolism: A versatile area providing multiple targets for pharmacological intervention. Egypt. J. Basic Clin. Pharmacol. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Saberi, B. Alcoholism and Serotonin. Psychopharmacology 2019, 171, 340–348. [Google Scholar]
- Kelley, K.W.; Dantzer, R. Alcoholism and inflammation: Neuroimmunology of behavioral and mood disorders. Brain Behav. Immun. 2011, 25, S13–S20. [Google Scholar] [CrossRef]
- Degenhardt, L.; Baxter, A.J.; Lee, Y.Y.; Hall, W.; Sara, G.E.; Johns, N.; Flaxman, A.; Whiteford, H.A.; Vos, T. The global epidemiology and burden of psychostimulant dependence: Findings from the Global Burden of Disease Study 2010. Drug Alcohol Depend. 2014, 137, 36–47. [Google Scholar] [CrossRef]
- Araos, P.; Vergara-Moragues, E.; González-Saiz, F.; Pedraz, M.; García-Marchena, N.; Romero-Sanchiz, P.; Ruiz, J.J.; Campos-Cloute, R.; Serrano, A.; Pavón, F.J. Differences in the rates of drug polyconsumption and psychiatric comorbidity among patients with cocaine use disorders according to the mental health service. J. Psychoact. Drugs 2017, 49, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Shorter, D.; Domingo, C.B.; Kosten, T.R. Emerging drugs for the treatment of cocaine use disorder: A review of neurobiological targets and pharmacotherapy. Expert Opin. Emerg. Drugs 2015, 20, 15–29. [Google Scholar] [CrossRef]
- Myint, A.M.; Kim, Y.K. Cytokine–serotonin interaction through IDO: A neurodegeneration hypothesis of depression. Med. Hypotheses 2003, 61, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Justinova, Z.; Mascia, P.; Wu, H.-Q.; Secci, M.E.; Redhi, G.H.; Panlilio, L.V.; Scherma, M.; Barnes, C.; Parashos, A.; Zara, T. Reducing cannabinoid abuse and preventing relapse by enhancing endogenous brain levels of kynurenic acid. Nat. Neurosci. 2013, 16, 1652–1661. [Google Scholar] [CrossRef]
- Vengeliene, V.; Cannella, N.; Takahashi, T.; Spanagel, R. Metabolic shift of the kynurenine pathway impairs alcohol and cocaine seeking and relapse. Psychopharmacology 2016, 233, 3449–3459. [Google Scholar] [CrossRef]
- Wrona, M.Z.; Dryhurst, G. A putative metabolite of serotonin, tryptamine-4, 5-dione, is an irreversible inhibitor of tryptophan hydroxylase: Possible relevance to the serotonergic neurotoxicity of methamphetamine. Chem. Res. Toxicol. 2001, 14, 1184–1192. [Google Scholar] [CrossRef]
- Zheng, T.; Liu, L.; Shi, J.; Yu, X.; Xiao, W.; Sun, R.; Zhou, Y.; Aa, J.; Wang, G. The metabolic impact of methamphetamine on the systemic metabolism of rats and potential markers of methamphetamine abuse. Mol. BioSyst. 2014, 10, 1968–1977. [Google Scholar] [CrossRef] [PubMed]
- Jenny, M.; Schröcksnadel, S.; Überall, F.; Fuchs, D. The potential role of cannabinoids in modulating serotonergic signaling by their influence on tryptophan metabolism. Pharmaceuticals 2010, 3, 2647–2660. [Google Scholar] [CrossRef]
- McDew-White, M.; Lee, E.; Alvarez, X.; Sestak, K.; Ling, B.J.; Byrareddy, S.N.; Okeoma, C.M.; Mohan, M. Cannabinoid control of gingival immune activation in chronically SIV-infected rhesus macaques involves modulation of the indoleamine-2,3-dioxygenase-1 pathway and salivary microbiome. EBioMedicine 2022, 75, 103769. [Google Scholar] [CrossRef]
- Ishak, N.; Ahmad, A.H.; Noor, S.A.M.; Ahmad, A. Detection of heroin metabolites at different developmental stages of Lucilia cuprina (Diptera: Calliphoridae) reared in heroin-treated meat: A preliminary analysis. Egypt. J. Forensic Sci. 2019, 9, 65. [Google Scholar] [CrossRef]
- Gaynor, C.M.; Handley, S.L. Effects of nicotine on head-shakes and tryptophan metabolites. Psychopharmacology 2001, 153, 327–333. [Google Scholar] [CrossRef]
- Fang, C.; Hayashi, S.; Du, X.; Cai, X.; Deng, B.; Zheng, H.; Ishido, S.; Tsutsui, H.; Sheng, J. Caffeine protects against stress-induced murine depression through activation of PPARγC1α-mediated restoration of the kynurenine pathway in the skeletal muscle. Sci. Rep. 2021, 11, 7287. [Google Scholar] [CrossRef]
- Varma, S.D.; Hegde, K.R. Kynurenine-induced photo oxidative damage to lens in vitro: Protective effect of caffeine. Mol. Cell. Biochem. 2010, 340, 49–54. [Google Scholar] [CrossRef]
- Giménez-Gómez, P.; Ballestín, R.; de Biedma-Elduayen, L.G.; Vidal, R.; Ferrer-Pérez, C.; Reguilón, M.D.; O’Shea, E.; Miñarro, J.; Colado, M.I.; Rodríguez-Arias, M. Decreased kynurenine pathway potentiate resilience to social defeat effect on cocaine reward. Neuropharmacology 2021, 197, 108753. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, F.; Mestre-Pintó, J.-I.; Gómez-Gómez, À.; Martinez-Sanvisens, D.; Rodríguez-Minguela, R.; Papaseit, E.; Pérez-Mañá, C.; Langohr, K.; Valverde, O.; Pozo, Ó.J. The tryptophan system in cocaine-induced depression. J. Clin. Med. 2020, 9, 4103. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.K.; Davidson, M.; Kypreos, E.; Feehan, J.; Muir, J.A.; Nurgali, K.; Apostolopoulos, V. Immunotherapies for the Treatment of Drug Addiction. Vaccines 2022, 10, 1778. [Google Scholar] [CrossRef]
- Kamal Hossain, M.; Davidson, M.; Feehan, J.; Deraos, G.; Nurgali, K.; Matsoukas, J.; Apostolopoulos, V. Development and characterization of a novel conjugated methamphetamine vaccine. Vaccine 2022, 40, 5882–5891. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.K.; Hassanzadeganroudsari, M.; Kypreos, E.; Feehan, J.; Apostolopoulos, V. Immune to addiction: How immunotherapies can be used to combat methamphetamine addiction. Expert Rev. Vaccines 2021, 20, 707–715. [Google Scholar] [CrossRef]
| Drug | Mechanism of Action | Chemical Structure | Mode of Administration | Side Effects | Ref. |
|---|---|---|---|---|---|
| Alcohol | Promotion of inhibitory neurotransmission | 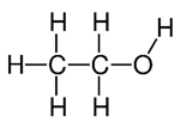 | Oral | Liver cirrhosis Neurotoxicity Drowsiness Loss of consciousness Gaps in memory | [6,18,19] |
| Caffeine | Release of dopamine, serotonin, and noradrenaline | 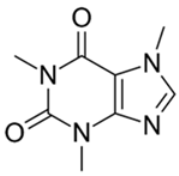 | Oral | Increased vigilance and metabolic activity Insomnia Nervousness Restlessness Nausea | [20,21,22,23] |
| Cocaine | Potentiation of dopamine and norepinephrine | 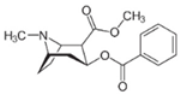 | Injection Inhalation | Euphoria Increased concentration Pupillary dilation | [24,25] |
| Heroin | Stimulation of opioid receptor | 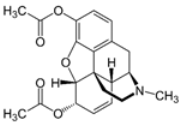 | Injection Inhalation | Analgesia Depression Pupillary constriction Pneumonia Increased heart rate Difficulty sleeping Difficulty breathing Dry mouth | [10,26,27] |
| Marijuana | Stimulation of cannabinoid receptor type 1 and cannabinoid receptor type 2 | 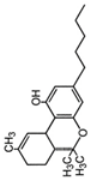 | Oral Smoke | Tachycardia High blood pressure Decrease in saccadic accuracy Difficulty concentrating Hallucinations Abnormal happiness Depression | [28,29,30] |
| Methamphetamine | Catecholamine potentiation | 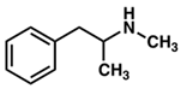 | Injection Inhalation Oral Intranasal sniffing | Depression Euphoria Pupillary dilation Cardiovascular diseases | [31,32,33] |
| Nicotine | Nicotinic acetylcholine agonist | 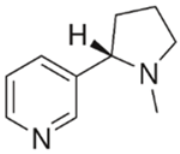 | Oral Smoke | Delayed coronary healing Hypertension Tachycardia Smoke-associated dryness | [34,35,36] |
| Drug | Tryptophan Metabolite | Ref. |
|---|---|---|
| METH | 5-HT, KA, and QA | [132,133] |
| Alcohol | 5-HT, KA, 3-HK, and 3-HAA | [84,123] |
| Marijuana | IDO 1, KA, and QA | [134,135] |
| Heroin | QA and 5-HIAA | [136] |
| Nicotine | KA, 5-HT, KYN, HKY, and 3-HAA | [137] |
| Caffeine | KA, 5-HT, 5-HIAA, and QA | [138,139] |
| Cocaine | KA, KYN, 5-HT, and 5-HIAA | [140,141] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Davidson, M.; Rashidi, N.; Hossain, M.K.; Raza, A.; Nurgali, K.; Apostolopoulos, V. Tryptophan and Substance Abuse: Mechanisms and Impact. Int. J. Mol. Sci. 2023, 24, 2737. https://doi.org/10.3390/ijms24032737
Davidson M, Rashidi N, Hossain MK, Raza A, Nurgali K, Apostolopoulos V. Tryptophan and Substance Abuse: Mechanisms and Impact. International Journal of Molecular Sciences. 2023; 24(3):2737. https://doi.org/10.3390/ijms24032737
Chicago/Turabian StyleDavidson, Majid, Niloufar Rashidi, Md Kamal Hossain, Ali Raza, Kulmira Nurgali, and Vasso Apostolopoulos. 2023. "Tryptophan and Substance Abuse: Mechanisms and Impact" International Journal of Molecular Sciences 24, no. 3: 2737. https://doi.org/10.3390/ijms24032737
APA StyleDavidson, M., Rashidi, N., Hossain, M. K., Raza, A., Nurgali, K., & Apostolopoulos, V. (2023). Tryptophan and Substance Abuse: Mechanisms and Impact. International Journal of Molecular Sciences, 24(3), 2737. https://doi.org/10.3390/ijms24032737









