The chemicals were procured from commercial suppliers and were used without further purification. Melting points (M.p.) were determined on the Electrothermal IA9200 digital melting point apparatus (Staffordshire, UK) and were uncorrected. Thin Layer Chromatography (TLC) was performed on TLC Silica gel 60 F254 aluminum sheets (Merck, Darmstadt, Germany) using petroleum ether:ethyl acetate solvent system (1:1). IR spectra were recorded on the IRPrestige-21 Fourier Transform Infrared spectrophotometer (Shimadzu, Tokyo, Japan). 1H and 13C NMR spectra were recorded on the Varian Mercury 400 NMR spectrometer (Agilent, Palo Alto, CA, USA). HRMS spectra were recorded on the LC/MS IT-TOF system (Shimadzu, Tokyo, Japan) using the electrospray ionization (ESI) technique.
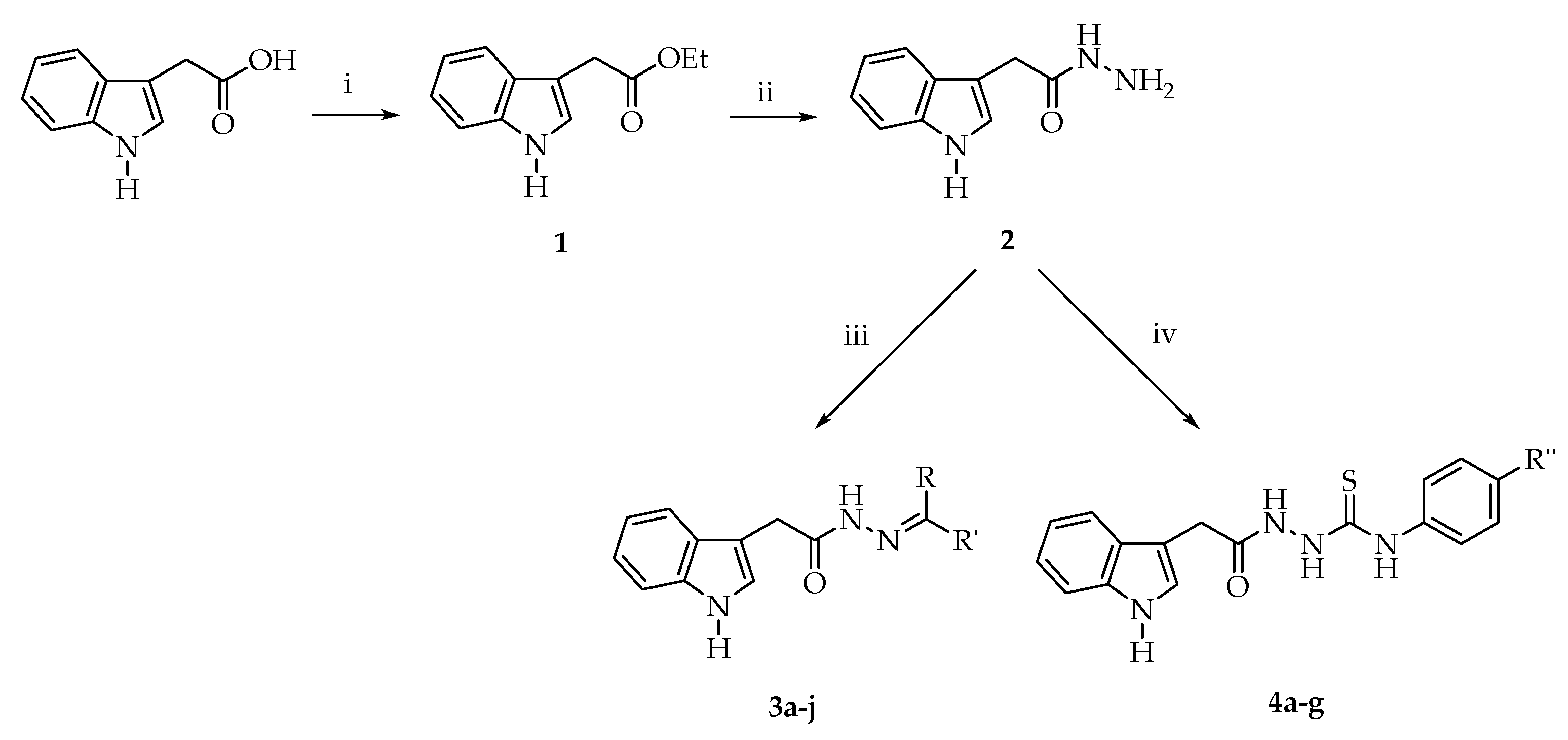
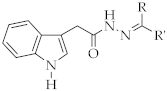
4.1.3. General Method for the Preparation of N′-benzylidene/(1-arylethylidene)-2-(1H-indol-3-yl)acetohydrazide Derivatives (3a-j)
A mixture of compound 2 and aromatic aldehyde or ketone in ethanol was heated under reflux for 15 h. At the end of this period, the precipitate was filtered off and dried. The product was crystallized from ethanol.
2-(1H-Indol-3-yl)-N′-[4-(pyrrolidin-1-yl)benzylidene]acetohydrazide (3a)
Yield: 78%. M.p.: 302–303 °C. IR νmax (cm−1): 3196.05, 3074.53, 3043.67, 2966.52, 2914.44, 2873.94, 2848.86, 1668.43, 1595.13, 1546.91, 1521.84, 1487.12, 1460.11, 1431.18, 1386.82, 1350.17, 1323.17, 1292.31, 1249.87, 1224.80, 1174.65, 1163.08, 1118.71, 1047.35, 1001.06, 983.70, 958.62, 929.69, 914.26, 856.39, 804.32, 719.45, 682.80. 1H NMR (400 MHz, DMSO-d6): 2.02–2.05 (m, 4H), 3.40–3.42 (m, 4H), 3.60 and 4.02 (2s, 2H), 6.92–7.07 (m, 4H), 7.21 (dd, J = 2.4 Hz, 12.8 Hz, 1H), 7.31–7.35 (m, 1H), 7.48–7.59 (m, 3H), 7.88 and 8.08 (2s, 1H), 10.85 and 10.88 (2s, 1H), 11.05 and 11.28 (2s, 1H). 13C NMR (100 MHz, DMSO-d6): 25.41 (2CH2), 32.13 (CH2), 47.70 (2CH2), 108.79 (C), 111.74 (CH), 115.13 (2CH), 118.74 (CH), 119.19 (CH), 121.33 (CH), 124.32 (CH), 124.75 (C), 127.90 (C), 128.27 (2CH), 136.45 (C), 146.90 (CH), 152.26 (C), 172.72 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C21H22N4O: 347.1866, found: 347.1864.
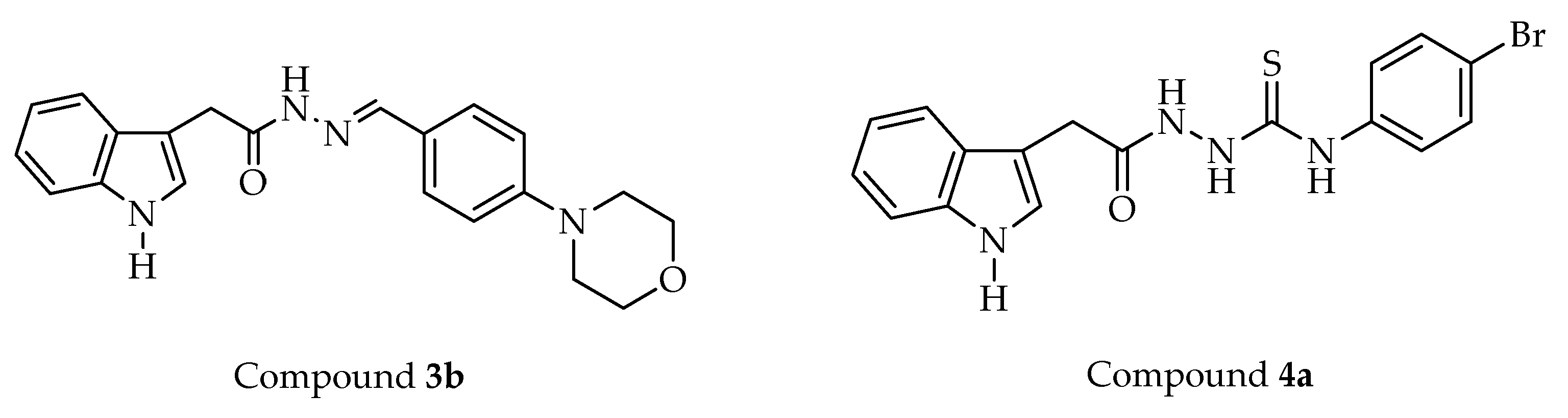
2-(1H-Indol-3-yl)-N′-(4-morpholinobenzylidene)acetohydrazide (3b)
Yield: 85%. M.p.: 306–307 °C. IR νmax (cm−1): 3275.13, 3178.69, 3055.24, 2964.59, 2922.16, 2870.08, 2825.72, 1660.71, 1604.77, 1558.48, 1541.12, 1519.91, 1506.41, 1489.05, 1456.26, 1446.61, 1425.40, 1392.61, 1375.25, 1338.60, 1313.52, 1301.95, 1259.52, 1224.80, 1186.22, 1176.58, 1159.22, 1109.07, 1095.57, 1062.78, 1045.42, 1006.84, 958.62, 921.97, 875.68, 858.32, 846.75, 823.60, 798.53, 786.96, 742.59, 682.80. 1H NMR (400 MHz, DMSO-d6): 3.19 (t, J = 4.41 Hz, 4.62 Hz, 4H), 3.72–3.75 (m, 4H), 3.61 and 4.02 (2s, 2H), 6.92–7.08 (m, 4H), 7.21 (dd, J = 2.4 Hz, 12.8 Hz, 1H), 7.31–7.35 (m, 1H), 7.49–7.60 (m, 3H), 7.88 and 8.07 (2s, 1H), 10.85 and 10.89 (2s, 1H), 11.05 and 11.28 (2s, 1H). 13C NMR (100 MHz, DMSO-d6): 32.13 (CH2), 53.79 (2CH2), 66.40 (2CH2), 108.79 (C), 111.74 (CH), 115.13 (2CH), 118.74 (CH), 119.19 (CH), 121.33 (CH), 124.32 (CH), 124.75 (C), 127.91 (C), 128.27 (2CH), 136.45 (C), 146.90 (CH), 152.29 (C), 172.73 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C21H22N4O2: 363.1816, found: 363.1824.
2-(1H-Indol-3-yl)-N′-[4-(piperidin-1-yl)benzylidene]acetohydrazide (3c)
Yield: 80%. M.p.: 265–266 °C. IR νmax (cm−1): 3203.76, 3082.25, 3034.03, 2972.31, 2935.66, 2856.58, 2825.72, 1668.43, 1598.99, 1552.70, 1514.12, 1448.54, 1427.32, 1384.89, 1350.17, 1282.66, 1247.94, 1220.94, 1182.36, 1124.50, 1024.20, 962.48, 914.26, 858.32, 804.32, 721.38, 651.94. 1H NMR (400 MHz, DMSO-d6): 1.58 (brs, 6H), 3.32 (brs, 4H), 3.60 and 4.02 (2s, 2H), 6.92–7.08 (m, 4H), 7.21 (dd, J = 2.4 Hz, 12.8 Hz, 1H), 7.31–7.35 (m, 1H), 7.48–7.59 (m, 3H), 7.88 and 8.08 (2s, 1H), 10.85 and 10.89 (2s, 1H), 11.05 and 11.28 (2s, 1H). 13C NMR (100 MHz, DMSO-d6): 24.40 (CH2), 25.42 (2CH2), 32.12 (CH2), 48.95 (2CH2), 108.79 (C), 111.74 (CH), 115.13 (2CH), 118.74 (CH), 119.19 (CH), 121.33 (CH), 124.32 (CH), 124.75 (C), 127.91 (C), 128.27 (2CH), 136.45 (C), 146.90 (CH), 152.26 (C), 172.73 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C22H24N4O: 361.2023, found: 361.2031.
2-(1H-Indol-3-yl)-N′-[4-(4-methylpiperazin-1-yl)benzylidene]acetohydrazide (3d)
Yield: 81%. M.p.: 218–220 °C. IR νmax (cm−1): 3398.57, 3205.69, 3165.19, 3111.18, 3043.67, 2939.52, 2883.58, 2831.50, 1649.49, 1602.85, 1517.98, 1446.61, 1427.32, 1409.96, 1377.17, 1340.53, 1286.52, 1232.51, 1184.29, 1159.22, 1141.86, 1124.50, 1105.21, 1080.14, 1001.06, 956.69, 943.19, 921.97, 806.25, 794.67, 742.59, 686.66. 1H NMR (400 MHz, DMSO-d6): 2.21 (s, 3H), 2.40–2.44 (m, 4H), 3.20–3.22 (m, 4H), 3.59 and 4.02 (2s, 2H), 6.92–7.08 (m, 4H), 7.21 (dd, J = 2.4 Hz, 12.8 Hz, 1H), 7.31–7.35 (m, 1H), 7.48–7.59 (m, 3H), 7.88 and 8.08 (2s, 1H), 10.85 and 10.89 (2s, 1H), 11.05 and 11.28 (2s, 1H). 13C NMR (100 MHz, DMSO-d6): 32.13 (CH2), 46.21 (CH3), 47.69 (2CH2), 54.89 (2CH2), 108.79 (C), 111.74 (CH), 115.13 (2CH), 118.74 (CH), 119.19 (CH), 121.33 (CH), 124.32 (CH), 124.75 (C), 127.91 (C), 128.27 (2CH), 136.45 (C), 146.90 (CH), 152.28 (C), 172.73 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C22H25N5O: 376.2132, found: 376.2148.
2-(1H-Indol-3-yl)-N′-(4-methylsulfonylbenzylidene)acetohydrazide (3e)
Yield: 86%. M.p.: 264–265 °C. IR νmax (cm−1): 3344.57, 3205.69, 3055.24, 2929.87, 2897.08, 1668.43, 1604.77, 1556.55, 1489.05, 1454.33, 1408.04, 1365.60, 1328.95, 1313.52, 1290.38, 1242.16, 1222.87, 1199.72, 1145.72, 1089.78, 1055.06, 1018.41, 983.70, 972.12, 956.69, 943.19, 869.90, 835.18, 792.74, 769.60, 750.31, 729.09, 686.66, 651.94. 1H NMR (400 MHz, DMSO-d6): 3.24 (s, 3H), 3.67 and 4.09 (2s, 2H), 6.94–7.09 (m, 2H), 7.25 (dd, J = 2.4 Hz, 9.6 Hz, 1H), 7.34 (t, J = 8.0 Hz, 8.4 Hz, 1H), 7.56–7.60 (m, 1H), 7.90–7.96 (m, 4H), 8.07 and 8.31 (2s, 1H), 10.87 and 10.93 (2s, 1H), 11.52 and 11.76 (2s, 1H). 13C NMR (100 MHz, DMSO-d6): 31.87 (CH2), 43.64 (CH3), 108.10 (C), 111.51 (CH), 118.53 (CH), 118.87 (CH), 121.12 (CH), 124.18 (CH), 127.31 (C), 127.50 (2CH), 127.70 (2CH), 136.19 (C), 139.35 (C), 141.21 (C), 144.37 (CH), 173.17 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C18H17N3O3S: 356.1063, found: 356.1071.
2-(1H-Indol-3-yl)-N′-[1-(4-methylsulfonylphenyl)ethylidene]acetohydrazide (3f)
Yield: 81%. M.p.: 204–205 °C. IR νmax (cm−1): 3342.64, 3190.26, 3088.03, 3032.10, 3005.10, 2924.09, 2848.86, 1668.43, 1585.49, 1562.34, 1489.05, 1456.26, 1417.68, 1394.53, 1338.60, 1296.16, 1280.73, 1226.73, 1188.15, 1145.72, 1093.64, 1070.49, 1008.77, 977.91, 964.41, 852.54, 839.03, 788.89, 758.02, 740.67, 717.52, 700.16, 688.59. 1H NMR (400 MHz, DMSO-d6): 2.30 (s, 3H), 3.26 (s, 3H), 3.81 and 4.12 (2s, 2H), 6.95–7.07 (m, 2H), 7.20–7.36 (m, 2H), 7.54–7.61 (m, 1H), 8.01 (d, J = 8.0 Hz, 2H), 8.15 (d, J = 8.8 Hz, 2H), 10.63 and 10.66 (2s, 1H), 10.84 and 10.89 (2s, 1H). 13C NMR (100 MHz, DMSO-d6): 14.96 (CH3), 31.87 (CH2), 43.42 (CH3), 108.10 (C), 111.26 (CH), 118.25 (CH), 118.64 (CH), 121.12 (CH), 123.84 (CH), 126.70 (C), 127.11 (2CH), 127.39 (2CH), 141.55 (C), 142.06 (C), 142.97 (C), 156.20 (C), 173.17 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C19H19N3O3S: 370.1220, found: 370.1202.
2-(1H-Indol-3-yl)-N′-[1-(4-morpholinophenyl)ethylidene]acetohydrazide (3g)
Yield: 79%. M.p.: 198–199 °C. IR νmax (cm−1): 3269.34, 3080.32, 3047.53, 2966.52, 2916.37, 2848.86, 1668.43, 1608.63, 1593.20, 1546.91, 1516.05, 1454.33, 1444.68, 1417.68, 1379.10, 1361.74, 1340.53, 1301.95, 1263.37, 1236.37, 1197.79, 1118.71, 1068.56, 1051.20, 1026.13, 937.40, 923.90, 864.11, 821.68, 798.53, 742.59, 729.09, 648.08. 1H NMR (400 MHz, DMSO-d6): 2.31 (s, 3H), 3.19 (t, J = 4.41 Hz, 4.62 Hz, 4H), 3.72–3.75 (m, 4H), 3.60 and 4.02 (2s, 2H), 6.92–7.08 (m, 4H), 7.21 (dd, J = 2.4 Hz, 12.8 Hz, 1H), 7.31–7.35 (m, 1H), 7.48–7.59 (m, 3H), 10.85 and 10.89 (2s, 1H), 11.05 and 11.27 (2s, 1H). 13C NMR (100 MHz, DMSO-d6): 14.96 (CH3), 32.13 (CH2), 53.79 (2CH2), 66.40 (2CH2), 108.79 (C), 111.74 (CH), 115.13 (2CH), 118.74 (CH), 119.19 (CH), 121.33 (CH), 124.32 (CH), 124.75 (C), 127.91 (C), 128.27 (2CH), 136.45 (C), 143.30 (C), 156.18 (C), 172.73 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C22H24N4O2: 377.1972, found: 377.1982.
2-(1H-Indol-3-yl)-N′-[4-(2-morpholinoethoxy)benzylidene]acetohydrazide (3h)
Yield: 83%. M.p.: 132–135 °C. IR νmax (cm−1): 3383.14, 3319.49, 3196.05, 3045.60, 2958.80, 2918.30, 2850.79, 1664.57, 1604.77, 1548.84, 1510.26, 1456.26, 1421.54, 1355.96, 1340.53, 1303.88, 1240.23, 1201.65, 1170.79, 1116.78, 1049.28, 1010.70, 983.70, 952.84, 925.83, 860.25, 831.32, 742.59, 646.15. 1H NMR (400 MHz, DMSO-d6): 2.45–2.47 (m, 4H), 2.66–2.70 (m, 2H), 3.55–3.58 (m, 4H), 4.04 (s, 2H), 4.08–4.12 (m, 2H), 6.95–7.07 (m, 4H), 7.23 (dd, J = 2.4 Hz, 12.4 Hz, 1H), 7.34 (t, J = 8.0 Hz, 1H), 7.57–7.65 (m, 3H), 7.94 and 8.16 (2s, 1H), 10.86 and 10.91 (2s, 1H), 11.15 and 11.39 (2s, 1H). 13C NMR (100 MHz, DMSO-d6): 31.85 (CH2), 53.79 (2CH2), 57.12 (CH2), 65.64 (CH2), 66.35 (2CH2), 108.44 (C), 111.48 (CH), 115.05 (2CH), 118.47 (CH), 118.91 (CH), 121.07 (CH), 124.07 (CH), 127.18 (C), 127.63 (C), 128.41 (2CH), 136.19 (C), 146.18 (CH), 159.90 (C), 172.64 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C23H26N4O3: 407.2078, found: 407.2071.
2-(1H-Indol-3-yl)-N′-[(1-methyl-1H-indol-3-yl)methylene]acetohydrazide (3i)
Yield: 84%. M.p.: 221–224 °C. IR νmax (cm−1): 3414.00, 3147.83, 3101.54, 3061.03, 2980.02, 2945.30, 2908.65, 2819.93, 1651.07, 1612.49, 1570.06, 1539.20, 1502.55, 1462.04, 1452.40, 1421.54, 1404.18, 1377.17, 1346.31, 1332.81, 1321.24, 1253.73, 1244.09, 1197.79, 1157.29, 1139.93, 1120.64, 1087.85, 1072.42, 1045.42, 1008.77, 948.98, 933.55, 900.76, 856.39, 808.17, 785.03, 744.52, 734.88, 673.16. 1H NMR (400 MHz, DMSO-d6): 3.64 and 4.13 (2s, 2H), 3.79 (s, 3H), 6.94–7.18 (m, 3H), 7.22–7.28 (m, 2H), 7.36 (t, J = 8.4 Hz, 8.8 Hz, 1H), 7.47 (t, J = 8.4 Hz, 9.2 Hz, 1H), 7.64 (t, J = 7.6 Hz, 1H), 7.73 (d, J = 2.4 Hz, 1H), 8.20–8.38 (m, 2H), 10.86 and 10.92 (2s, 1H), 10.99 and 11.19 (2s, 1H). 13C NMR (100 MHz, DMSO-d6): 31.94 (CH2), 32.90 (CH3), 108.74 (C), 110.42 (CH), 110.83 (CH), 111.49 (C), 118.50 (CH), 118.93 (CH), 120.89 (CH), 121.10 (CH), 121.86 (CH), 122.80 (CH), 123.96 (CH), 124.70 (C), 127.70 (C), 133.81 (CH), 136.23 (C), 137.76 (C), 142.98 (CH), 172.09 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C20H18N4O: 331.1553, found: 331.1538.
2-(1H-Indol-3-yl)-N′-[(5-methoxy-1H-indol-3-yl)methylene]acetohydrazide (3j)
Yield: 82%. M.p.: 231–233 °C. IR νmax (cm−1): 3415.93, 3373.50, 3049.46, 3012.81, 2958.80, 2931.80, 2877.79, 2829.57, 1654.92, 1614.42, 1577.77, 1539.20, 1487.12, 1456.26, 1421.54, 1396.46, 1354.03, 1342.46, 1307.74, 1292.31, 1261.45, 1213.23, 1182.36, 1176.58, 1130.29, 1105.21, 1087.85, 1072.42, 1049.28, 1022.27, 1006.84, 950.91, 923.90, 856.39, 810.10, 744.52, 725.23, 671.23, 651.94. 1H NMR (400 MHz, DMSO-d6): 3.59 (s, 3H), 3.74 and 4.16 (2s, 2H), 6.83 (dd, J = 2.4 Hz, 8.8 Hz, 1H), 6.93–7.10 (m, 2H), 7.27–7.38 (m, 3H), 7.64 (t, J = 8.8 Hz, 9.2 Hz, 1H), 7.72–7.80 (m, 2H), 8.23 and 8.41 (2s, 1H), 10.85 and 10.91 (2s, 1H), 11.02 and 11.18 (2s, 1H), 11.40 (s, 1H). 13C NMR (100 MHz, DMSO-d6): 31.90 (CH2), 55.01 (CH3), 103.47 (CH), 108.71 (C), 111.50 (C), 111.59 (CH), 112.39 (CH), 112.64 (CH), 118.52 (CH), 118.83 (CH), 121.14 (CH), 123.88 (CH), 124.83 (C), 127.73 (C), 130.56 (C), 132.19 (CH), 136.19 (C), 143.76 (CH), 154.55 (C), 172.04 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C20H18N4O2: 347.1503, found: 347.1505.
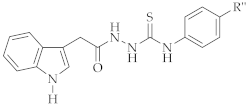
4.1.4. General Method for the Preparation of 4-aryl-1-[2-(1H-indol-3-yl)acetyl]thiosemicarbazide Derivatives (4a-g)
A mixture of compound 2 and aryl isothiocyanate in ethanol was stirred at room temperature for 8 h. The precipitate was filtered off. The product was crystallized from ethanol.
4-(4-Bromophenyl)-1-[2-(1H-indol-3-yl)acetyl]thiosemicarbazide (4a)
Yield: 87%. M.p.: 187–189 °C. IR νmax (cm−1): 3390.86, 3311.78, 3286.70, 3207.62, 3143.97, 3057.17, 2997.38, 2927.94, 1680.00, 1647.21, 1620.21, 1589.34, 1544.98, 1506.41, 1485.19, 1452.40, 1419.61, 1352.10, 1309.67, 1282.66, 1247.94, 1207.44, 1138.00, 1087.85, 1074.35, 1049.28, 1004.91, 987.55, 871.82, 823.60, 792.74, 736.81, 715.59, 669.30. 1H NMR (400 MHz, DMSO-d6): 3.63 (s, 2H), 6.97 (t, J = 6.8 Hz, 1H), 7.07 (t, J = 6.8 Hz, 1H), 7.25 (d, J = 2.4 Hz, 1H), 7.34 (d, J = 8.0 Hz, 1H), 7.42 (d, J = 8.0 Hz, 2H), 7.51 (d, J = 8.4 Hz, 2H), 7.59 (d, J = 7.6 Hz, 1H), 9.59 (brs, 1H), 9.73 (s, 1H), 10.10 (brs, 1H), 10.89 (s, 1H). 13C NMR (100 MHz, DMSO-d6): 31.18 (CH2), 108.39 (C), 111.75 (CH), 118.80 (CH), 119.26 (CH), 121.46 (CH), 122.70 (C), 124.44 (CH), 127.70 (C), 129.40 (2CH), 131.37 (2CH), 136.51 (C), 139.05 (C), 170.35 (C), 181.10 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C17H15BrN4OS: 403.0223, found: 403.0204.
4-(4-Trifluoromethylphenyl)-1-[2-(1H-indol-3-yl)acetyl]thiosemicarbazide (4b)
Yield: 80%. M.p.: 184–186 °C. IR νmax (cm−1): 3392.79, 3315.63, 3292.49, 3223.05, 3163.26, 3070.68, 2995.45, 2927.94, 1681.93, 1649.14, 1616.35, 1568.13, 1544.98, 1504.48, 1454.33, 1419.61, 1357.89, 1321.24, 1246.02, 1224.80, 1209.37, 1184.29, 1163.08, 1132.21, 1120.64, 1112.93, 1085.92, 1070.49, 1012.63, 985.62, 846.75, 788.89, 736.81, 711.73, 665.44. 1H NMR (400 MHz, DMSO-d6): 3.64 (s, 2H), 6.97 (t, J = 6.8 Hz, 1H), 7.07 (t, J = 6.8 Hz, 1H), 7.26 (d, J = 2.0 Hz, 1H), 7.34 (d, J = 8.0 Hz, 1H), 7.60 (d, J = 7.6 Hz, 1H), 7.67–7.75 (m, 4H), 9.75 (brs, 1H), 9.88 (s, 1H), 10.14 (brs, 1H), 10.89 (s, 1H). 13C NMR (100 MHz, DMSO-d6): 31.16 (CH2), 108.36 (C), 111.76 (CH), 118.80 (CH), 119.25 (CH), 121.45 (CH), 123.45 (CH), 124.46 (2CH), 125.64 (C), 126.15 (C), 127.69 (2CH), 132.50 (C), 136.51 (C), 143.45 (C), 170.35 (C), 181.10 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C18H15F3N4OS: 393.0991, found: 393.0989.
4-(4-Cyanophenyl)-1-[2-(1H-indol-3-yl)acetyl]thiosemicarbazide (4c)
Yield: 89%. M.p.: 180–182 °C. IR νmax (cm−1): 3425.58, 3313.71, 3284.77, 3201.83, 3145.90, 3059.10, 2995.45, 2956.87, 2914.44, 2223.92, 1680.00, 1651.07, 1620.21, 1602.85, 1541.12, 1510.26, 1475.54, 1454.33, 1409.96, 1334.74, 1290.38, 1244.09, 1226.73, 1203.58, 1174.65, 1136.07, 1093.64, 1060.85, 1012.63, 975.98, 837.11, 790.81, 769.60, 734.88, 692.44. 1H NMR (400 MHz, DMSO-d6): 3.64 (s, 2H), 6.97 (t, J = 7.2 Hz, 7.6 Hz, 1H), 7.07 (t, J = 7.2 Hz, 7.6 Hz, 1H), 7.26 (s, 1H), 7.35 (d, J = 7.6 Hz, 1H), 7.60 (d, J = 7.2 Hz, 1H), 7.78 (s, 4H), 9.76 (brs, 1H), 9.97 (s, 1H), 10.16 (brs, 1H), 10.90 (s, 1H). 13C NMR (100 MHz, DMSO-d6): 31.17 (CH2), 108.04 (C), 109.58 (C), 111.49 (CH), 118.53 (C), 118.97 (CH), 119.16 (CH), 121.20 (CH), 124.20 (CH), 127.40 (C), 129.32 (2CH), 132.55 (2CH), 136.24 (C), 143.87 (C), 170.35 (C), 181.10 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C18H15N5OS: 350.1070, found: 350.1063.
4-[4-(Piperidin-1-ylsulfonyl)phenyl]-1-[2-(1H-indol-3-yl)acetyl]thiosemicarbazide (4d)
Yield: 85%. M.p.: 182–184 °C. IR νmax (cm−1): 3390.86, 3288.63, 3197.98, 3089.96, 2939.52, 2850.79, 1645.28, 1595.13, 1550.77, 1496.76, 1467.83, 1404.18, 1336.67, 1315.45, 1276.88, 1244.09, 1226.73, 1215.15, 1149.57, 1093.64, 1053.13, 1028.06, 1012.63, 983.70, 929.69, 860.25, 839.03, 819.75, 777.31, 752.24, 738.74, 719.45, 698.23, 667.37. 1H NMR (400 MHz, DMSO-d6): 1.33–1.36 (m, 2H), 1.50–1.54 (m, 4H), 2.87 (t, J = 4.8 Hz, 5.2 Hz, 4H), 3.66 (s, 2H), 6.98 (t, J = 7.2 Hz, 1H), 7.08 (t, J = 7.2 Hz, 1H), 7.27 (d, J = 2.0 Hz, 1H), 7.36 (d, J = 7.6 Hz, 1H), 7.61 (d, J = 8.0 Hz, 1H), 7.67 (d, J = 8.4 Hz, 2H), 7.83 (d, J = 8.4 Hz, 2H), 9.75 (brs, 1H), 9.93 (s, 1H), 10.16 (brs, 1H), 10.90 (s, 1H). 13C NMR (100 MHz, DMSO-d6): 23.37 (CH2), 25.14 (2CH2), 31.18 (CH2), 47.08 (2CH2), 108.35 (C), 111.78 (CH), 118.83 (CH), 119.26 (CH), 121.48 (CH), 124.49 (CH), 125.05 (2CH), 127.68 (C), 128.11 (2CH), 135.47 (C), 136.53 (C), 143.89 (C), 170.34 (C), 181.11 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C22H25N5O3S2: 472.1472, found: 472.1452.
4-[4-(1H-Pyrazol-1-yl)phenyl]-1-[2-(1H-indol-3-yl)acetyl]thiosemicarbazide (4e)
Yield: 85%. M.p.: 196–198 °C. IR νmax (cm−1): 3305.99, 3223.05, 3167.12, 3134.33, 3095.75, 3061.03, 2999.31, 2933.73, 1680.00, 1647.21, 1622.13, 1573.91, 1546.91, 1523.76, 1454.33, 1421.54, 1396.46, 1359.82, 1332.81, 1317.38, 1305.81, 1249.87, 1222.87, 1199.72, 1159.22, 1136.07, 1124.50, 1089.78, 1043.49, 1033.85, 1008.77, 985.62, 935.48, 840.96, 792.74, 758.02, 744.52, 717.52, 667.37. 1H NMR (400 MHz, DMSO-d6): 3.65 (s, 2H), 6.53 (t, J = 2.4 Hz, 1H), 6.99 (t, J = 7.2 Hz, 1H), 7.08 (t, J = 7.2 Hz, 1H), 7.27 (d, J = 2.0 Hz, 1H), 7.36 (d, J = 8.0 Hz, 1H), 7.55 (d, J = 8.0 Hz, 1H), 7.62 (d, J = 8.0 Hz, 2H), 7.74 (d, J = 1.6 Hz, 1H), 7.80 (d, J = 8.8 Hz, 2H), 8.46 (d, J = 2.4 Hz, 1H), 9.65 (brs, 1H), 9.71 (s, 1H), 10.13 (s, 1H), 10.90 (s, 1H). 13C NMR (100 MHz, DMSO-d6): 31.20 (CH2), 108.23 (C), 108.45 (CH), 111.77 (CH), 118.56 (CH), 118.82 (CH), 119.28 (2CH), 121.48 (CH), 124.47 (CH), 126.80 (CH), 127.73 (C), 128.09 (2CH), 136.53 (C), 137.64 (2C), 141.30 (CH), 170.32 (C), 181.10 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C20H18N6OS: 391.1336, found: 391.1334.
4-(1,3-Benzodioxol-5-yl)-1-[2-(1H-indol-3-yl)acetyl]thiosemicarbazide (4f)
Yield: 81%. M.p.: 168–170 °C. IR νmax (cm−1): 3300.20, 3209.55, 3149.76, 3057.17, 2929.87, 2897.08, 1678.07, 1643.35, 1591.27, 1539.20, 1500.62, 1481.33, 1454.33, 1419.61, 1334.74, 1282.66, 1240.23, 1197.79, 1122.57, 1089.78, 1037.70, 981.77, 923.90, 850.61, 815.89, 808.17, 790.81, 731.02, 698.23. 1H NMR (400 MHz, DMSO-d6): 3.62 (s, 2H), 6.02 (s, 2H), 6.72 (d, J = 8.4 Hz, 1H), 6.87 (d, J = 8.0 Hz, 1H), 6.96–7.09 (m, 3H), 7.26 (s, 1H), 7.34 (d, J = 8.4 Hz, 1H), 7.60 (d, J = 8.0 Hz, 1H), 9.47 (brs, 1H), 9.55 (s, 1H), 10.06 (s, 1H), 10.89 (s, 1H). 13C NMR (100 MHz, DMSO-d6): 30.65 (CH2), 101.21 (CH2), 107.38 (C), 107.97 (CH), 111.25 (CH), 118.29 (2CH), 118.78 (CH), 120.95 (CH), 123.95 (CH), 127.24 (CH), 133.08 (C), 136.02 (2C), 144.59 (C), 146.56 (C), 170.35 (C), 181.10 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C18H16N4O3S: 369.1016, found: 369.0998.
4-[4-(Benzyloxy)phenyl]-1-[2-(1H-indol-3-yl)acetyl]thiosemicarbazide (4g)
Yield: 88%. M.p.: 198–200 °C. IR νmax (cm−1): 3394.72, 3290.56, 3213.41, 3155.54, 3059.10, 3032.10, 2939.52, 2873.94, 1681.93, 1649.14, 1618.28, 1564.27, 1546.91, 1504.48, 1456.26, 1417.68, 1381.03, 1359.82, 1294.24, 1244.09, 1219.01, 1170.79, 1138.00, 1089.78, 1051.20, 999.13, 912.33, 879.54, 829.39, 790.81, 734.88, 702.09, 646.15. 1H NMR (400 MHz, DMSO-d6): 3.64 (s, 2H), 5.10 (s, 2H), 6.97–7.01 (m, 3H), 7.06–7.10 (m, 1H), 7.27–7.29 (m, 3H), 7.33–7.47 (m, 6H), 7.61 (d, J = 8.0 Hz, 1H), 9.47 (brs, 1H), 9.53 (s, 1H), 10.07 (s, 1H), 10.90 (s, 1H). 13C NMR (100 MHz, DMSO-d6): 30.68 (CH2), 69.34 (CH2), 107.99 (C), 111.27 (CH), 114.22 (2CH), 118.31 (2CH), 118.79 (CH), 120.96 (CH), 123.95 (CH), 127.25 (C), 127.67 (2CH), 127.80 (2CH), 128.41 (2CH), 132.15 (C), 136.03 (C), 137.08 (C), 155.77 (C), 170.39 (C), 181.17 (C). HRMS (ESI) (m/z): [M + H]+ calcd. for C24H22N4O2S: 431.1536, found: 431.1554.