BCG-Unresponsive Non-Muscle-Invasive Bladder Cancer: Current Treatment Landscape and Novel Emerging Molecular Targets
Abstract
1. Introduction
2. Intravesical Chemotherapy
3. Chemo-Hyperthermia
4. Immunotherapy and Inflammation-Targeted Agents
5. Gene Therapy
6. Other Therapies
7. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer Statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef]
- Beijert, I.J.; Hentschel, A.E.; Bründl, J.; Compérat, E.M.; Plass, K.; Rodríguez, O.; Subiela Henríquez, J.D.; Hernández, V.; de la Peña, E.; Alemany, I.; et al. Prognosis of Primary Papillary Ta Grade 3 Bladder Cancer in the Non-Muscle-Invasive Spectrum. Eur. Urol. Oncol. 2023, 6, 214–221. [Google Scholar] [CrossRef]
- Claps, F.; van de Kamp, M.W.; Mayr, R.; Bostrom, P.J.; Boormans, J.L.; Eckstein, M.; Mertens, L.S.; Boevé, E.R.; Neuzillet, Y.; Burger, M.; et al. Risk Factors Associated with Positive Surgical Margins’ Location at Radical Cystectomy and Their Impact on Bladder Cancer Survival. World J. Urol. 2021, 39, 4363–4371. [Google Scholar] [CrossRef] [PubMed]
- Claps, F.; van de Kamp, M.W.; Mayr, R.; Bostrom, P.J.; Shariat, S.F.; Hippe, K.; Bertz, S.; Neuzillet, Y.; Sanders, J.; Otto, W.; et al. Prognostic Impact of Variant Histologies in Urothelial Bladder Cancer Treated with Radical Cystectomy. BJU Int. 2023, 132, 170–180. [Google Scholar] [CrossRef] [PubMed]
- Babjuk, M.; Burger, M.; Capoun, O.; Cohen, D.; Compérat, E.M.; Dominguez Escrig, J.L.; Gontero, P.; Liedberg, F.; Masson-Lecomte, A.; Mostafid, A.H.; et al. European Association of Urology Guidelines on Non-Muscle-Invasive Bladder Cancer (Ta, T1, and Carcinoma In Situ). Eur. Urol. 2022, 81, 75–94. [Google Scholar] [CrossRef] [PubMed]
- Nazmifar, M.; Williams, C.; Naser-Tavakolian, A.; Heard, J.; Rosser, C.; Theodorescu, D.; Ahdoot, M. Clinical and Preclinical Therapies for Bladder Cancer Following Bacillus Calmette-Guérin Failure. J. Urol. 2023, 209, 32–48. [Google Scholar] [CrossRef]
- Chang, S.S.; Boorjian, S.A.; Chou, R.; Clark, P.E.; Daneshmand, S.; Konety, B.R.; Pruthi, R.; Quale, D.Z.; Ritch, C.R.; Seigne, J.D.; et al. Diagnosis and Treatment of Non-Muscle Invasive Bladder Cancer: AUA/SUO Guideline. J. Urol. 2016, 196, 1021–1029. [Google Scholar] [CrossRef]
- Kamat, A.M.; Colombel, M.; Sundi, D.; Lamm, D.; Boehle, A.; Brausi, M.; Buckley, R.; Persad, R.; Palou, J.; Soloway, M.; et al. BCG-Unresponsive Non-Muscle-Invasive Bladder Cancer: Recommendations from the IBCG. Nat. Rev. Urol. 2017, 14, 244–255. [Google Scholar] [CrossRef]
- Cambier, S.; Sylvester, R.J.; Collette, L.; Gontero, P.; Brausi, M.A.; Van Andel, G.; Kirkels, W.J.; Silva, F.C.; Da Oosterlinck, W.; Prescott, S.; et al. EORTC Nomograms and Risk Groups for Predicting Recurrence, Progression, and Disease-Specific and Overall Survival in Non-Muscle-Invasive Stage Ta-T1 Urothelial Bladder Cancer Patients Treated with 1-3 Years of Maintenance Bacillus Calmette-Guérin. Eur. Urol. 2016, 69, 60–69. [Google Scholar] [CrossRef]
- Balar, A.V.; Kamat, A.M.; Kulkarni, G.S.; Uchio, E.M.; Boormans, J.L.; Roumiguié, M.; Krieger, L.E.M.; Singer, E.A.; Bajorin, D.F.; Grivas, P.; et al. Pembrolizumab Monotherapy for the Treatment of High-Risk Non-Muscle-Invasive Bladder Cancer Unresponsive to BCG (KEYNOTE-057): An Open-Label, Single-Arm, Multicentre, Phase 2 Study. Lancet Oncol. 2021, 22, 919–930. [Google Scholar] [CrossRef]
- Witjes, J.A.; Bruins, H.M.; Cathomas, R.; Compérat, E.M.; Cowan, N.C.; Gakis, G.; Hernández, V.; Linares Espinós, E.; Lorch, A.; Neuzillet, Y.; et al. European Association of Urology Guidelines on Muscle-Invasive and Metastatic Bladder Cancer: Summary of the 2020 Guidelines. Eur. Urol. 2021, 79, 82–104. [Google Scholar] [CrossRef] [PubMed]
- Satkunasivam, R.; Tallman, C.T.; Taylor, J.M.; Miles, B.J.; Klaassen, Z.; Wallis, C.J.D. Robot-Assisted Radical Cystectomy Versus Open Radical Cystectomy: A Meta-Analysis of Oncologic, Perioperative, and Complication-Related Outcomes. Eur. Urol. Oncol. 2019, 2, 443–447. [Google Scholar] [CrossRef] [PubMed]
- Claps, F.; Mir, M.C.; van Rhijn, B.W.G.; Mazzon, G.; Soria, F.; D’Andrea, D.; Marra, G.; Boltri, M.; Traunero, F.; Massanova, M.; et al. Impact of the Controlling Nutritional Status (CONUT) Score on Perioperative Morbidity and Oncological Outcomes in Patients with Bladder Cancer Treated with Radical Cystectomy. Urol. Oncol. 2023, 41, e13–e49. [Google Scholar] [CrossRef]
- Wessels, F.; Lenhart, M.; Kowalewski, K.F.; Braun, V.; Terboven, T.; Roghmann, F.; Michel, M.S.; Honeck, P.; Kriegmair, M.C. Early Recovery after Surgery for Radical Cystectomy: Comprehensive Assessment and Meta-Analysis of Existing Protocols. World J. Urol. 2020, 38, 3139–3153. [Google Scholar] [CrossRef] [PubMed]
- Zehnder, P.; Studer, U.E.; Skinner, E.C.; Thalmann, G.N.; Miranda, G.; Roth, B.; Cai, J.; Birkhäuser, F.D.; Mitra, A.P.; Burkhard, F.C.; et al. Unaltered Oncological Outcomes of Radical Cystectomy with Extended Lymphadenectomy over Three Decades. BJU Int. 2013, 112, E51–E58. [Google Scholar] [CrossRef]
- Psutka, S.P.; Barocas, D.A.; Catto, J.W.F.; Gore, J.L.; Lee, C.T.; Morgan, T.M.; Master, V.A.; Necchi, A.; Rouprêt, M.; Boorjian, S.A. Staging the Host: Personalizing Risk Assessment for Radical Cystectomy Patients. Eur. Urol. Oncol. 2018, 1, 292–304. [Google Scholar] [CrossRef]
- Vetterlein, M.W.; Klemm, J.; Gild, P.; Bradtke, M.; Soave, A.; Dahlem, R.; Fisch, M.; Rink, M. Improving Estimates of Perioperative Morbidity after Radical Cystectomy Using the European Association of Urology Quality Criteria for Standardized Reporting and Introducing the Comprehensive Complication Index. Eur. Urol. 2020, 77, 55–65. [Google Scholar] [CrossRef]
- Soria, F.; Gontero, P. Re: Intravesical Nadofaragene Firadenovec Gene Therapy for BCG-Unresponsive Non-Muscle-Invasive Bladder Cancer: A Single-Arm, Open-Label, Repeat-Dose Clinical Trial. Eur. Urol. 2021, 79, 891–892. [Google Scholar] [CrossRef]
- Claps, F.; Mir, M.C.; Zargar, H. Molecular Markers of Systemic Therapy Response in Urothelial Carcinoma. Asian J. Urol. 2021, 8, 376–390. [Google Scholar] [CrossRef]
- Mertens, L.S.; Claps, F.; Mayr, R.; Bostrom, P.J.; Shariat, S.F.; Zwarthoff, E.C.; Boormans, J.L.; Abas, C.; van Leenders, G.J.L.H.; Götz, S.; et al. Prognostic Markers in Invasive Bladder Cancer: FGFR3 Mutation Status versus P53 and KI-67 Expression: A Multi-Center, Multi-Laboratory Analysis in 1058 Radical Cystectomy Patients. Urol. Oncol. 2022, 40, e1–e110. [Google Scholar] [CrossRef]
- Mir, M.C.; Campi, R.; Loriot, Y.; Puente, J.; Giannarini, G.; Necchi, A.; Rouprêt, M. Adjuvant Systemic Therapy for High-Risk Muscle-Invasive Bladder Cancer after Radical Cystectomy: Current Options and Future Opportunities. Eur. Urol. Oncol. 2021, 5, 726–731. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, M.A.; Boehle, A. Treatment Options for BCG Failures. World J. Urol. 2006, 24, 481–487. [Google Scholar] [CrossRef]
- Lerner, S.P.; Dinney, C.; Kamat, A.; Bivalacqua, T.J.; Nielsen, M.; O’Donnell, M.; Schoenberg, M.P.; Steinberg, G. Clarification of Bladder Cancer Disease States Following Treatment of Patients with Intravesical BCG. Bladder Cancer 2015, 1, 29–30. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Kates, M.; Fujita, K.; Bivalacqua, T.J.; McConkey, D.J. Predictive Biomarkers for Drug Response in Bladder Cancer. Int. J. Urol. 2019, 26, 1044–1053. [Google Scholar] [CrossRef] [PubMed]
- Chevuru, P.T.; McElree, I.M.; Mott, S.L.; Steinberg, R.L.; O’Donnell, M.A.; Packiam, V.T. Long-Term Follow-up of Sequential Intravesical Gemcitabine and Docetaxel Salvage Therapy for Non-Muscle Invasive Bladder Cancer. Urol. Oncol. 2023, 41, 148.e1–148.e7. [Google Scholar] [CrossRef]
- Hurle, R.; Casale, P.; Morenghi, E.; Saita, A.; Buffi, N.; Lughezzani, G.; Colombo, P.; Contieri, R.; Frego, N.; Guazzoni, G.; et al. Intravesical Gemcitabine as Bladder-Preserving Treatment for BCG Unresponsive Non-Muscle-Invasive Bladder Cancer. Results from a Single-Arm, Open-Label Study. BJUI Compass 2020, 1, 126–132. [Google Scholar] [CrossRef]
- Steinberg, R.L.; Thomas, L.J.; Brooks, N.; Mott, S.L.; Vitale, A.; Crump, T.; Rao, M.Y.; Daniels, M.J.; Wang, J.; Nagaraju, S.; et al. Multi-Institution Evaluation of Sequential Gemcitabine and Docetaxel as Rescue Therapy for Nonmuscle Invasive Bladder Cancer. J. Urol. 2020, 203, 902–908. [Google Scholar] [CrossRef] [PubMed]
- DeCastro, G.J.; Sui, W.; Pak, J.S.; Lee, S.M.; Holder, D.; Kates, M.M.; Virk, R.K.; Drake, C.G.; Anderson, C.B.; James, B.; et al. A Phase I Trial of Intravesical Cabazitaxel, Gemcitabine and Cisplatin for the Treatment of Nonmuscle Invasive Bacillus Calmette-Guérin Unresponsive or Recurrent/Relapsing Urothelial Carcinoma of the Bladder. J. Urol. 2020, 204, 247–253. [Google Scholar] [CrossRef]
- Milbar, N.; Kates, M.; Chappidi, M.R.; Pederzoli, F.; Yoshida, T.; Sankin, A.; Pierorazio, P.M.; Schoenberg, M.P.; Bivalacqua, T.J. Oncological Outcomes of Sequential Intravesical Gemcitabine and Docetaxel in Patients with Non-Muscle Invasive Bladder Cancer. Bladder Cancer 2017, 3, 293–303. [Google Scholar] [CrossRef]
- Dalbagni, G.; Benfante, N.; Sjoberg, D.D.; Bochner, B.H.; Machele Donat, S.; Herr, H.W.; McCoy, A.S.; Fahrner, A.J.; Retinger, C.; Rosenberg, J.E.; et al. Single Arm Phase I/II Study of Everolimus and Intravesical Gemcitabine in Patients with Primary or Secondary Carcinoma In Situ of the Bladder Who Failed Bacillus Calmette Guerin (NCT01259063). Bladder Cancer 2017, 3, 113–119. [Google Scholar] [CrossRef]
- Robins, D.J.; Sui, W.; Matulay, J.T.; Ghandour, R.; Anderson, C.B.; DeCastro, G.J.; McKiernan, J.M. Long-Term Survival Outcomes with Intravesical Nanoparticle Albumin-Bound Paclitaxel for Recurrent Non-Muscle-Invasive Bladder Cancer after Previous Bacillus Calmette-Guérin Therapy. Urology 2017, 103, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Cockerill, P.A.; Knoedler, J.J.; Frank, I.; Tarrell, R.; Karnes, R.J. Intravesical Gemcitabine in Combination with Mitomycin C as Salvage Treatment in Recurrent Non-Muscle-Invasive Bladder Cancer. BJU Int. 2016, 117, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, R.L.; Thomas, L.J.; O’Donnell, M.A.; Nepple, K.G. Sequential Intravesical Gemcitabine and Docetaxel for the Salvage Treatment of Non-Muscle Invasive Bladder Cancer. Bladder Cancer 2015, 1, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Lightfoot, A.J.; Breyer, B.N.; Rosevear, H.M.; Erickson, B.A.; Konety, B.R.; O’Donnell, M.A. Multi-Institutional Analysis of Sequential Intravesical Gemcitabine and Mitomycin C Chemotherapy for Non-Muscle Invasive Bladder Cancer. Urol. Oncol. 2014, 32, 35.e15–35.e19. [Google Scholar] [CrossRef]
- McKiernan, J.M.; Holder, D.D.; Ghandour, R.A.; Barlow, L.J.; Ahn, J.J.; Kates, M.; Badalato, G.M.; Roychoudhury, A.; Decastro, G.J.; Benson, M.C. Phase II Trial of Intravesical Nanoparticle Albumin Bound Paclitaxel for the Treatment of Nonmuscle Invasive Urothelial Carcinoma of the Bladder after Bacillus Calmette-Guérin Treatment Failure. J. Urol. 2014, 192, 1633–1638. [Google Scholar] [CrossRef]
- Barlow, L.J.; McKiernan, J.M.; Benson, M.C. Long-Term Survival Outcomes with Intravesical Docetaxel for Recurrent Nonmuscle Invasive Bladder Cancer after Previous Bacillus Calmette-Guérin Therapy. J. Urol. 2013, 189, 834–839. [Google Scholar] [CrossRef]
- Skinner, E.C.; Goldman, B.; Sakr, W.A.; Petrylak, D.P.; Lenz, H.J.; Lee, C.T.; Wilson, S.S.; Benson, M.; Lerner, S.P.; Tangen, C.M.; et al. SWOG S0353: Phase II Trial of Intravesical Gemcitabine in Patients with Nonmuscle Invasive Bladder Cancer and Recurrence after 2 Prior Courses of Intravesical Bacillus Calmette-Guérin. J. Urol. 2013, 190, 1200–1204. [Google Scholar] [CrossRef]
- Sternberg, I.A.; Dalbagni, G.; Chen, L.Y.; Donat, S.M.; Bochner, B.H.; Herr, H.W. Intravesical Gemcitabine for High Risk, Nonmuscle Invasive Bladder Cancer after Bacillus Calmette-Guérin Treatment Failure. J. Urol. 2013, 190, 1686–1691. [Google Scholar] [CrossRef]
- Steinberg, G.D.; Smith, N.D.; Ryder, K.; Strangman, N.M.; Slater, S.J. Factors Affecting Valrubicin Response in Patients with Bacillus Calmette-Guérin-Refractory Bladder Carcinoma In Situ. Postgrad. Med. 2011, 123, 28–36. [Google Scholar] [CrossRef]
- McKiernan, J.M.; Barlow, L.J.; Laudano, M.A.; Mann, M.J.; Petrylak, D.P.; Benson, M.C. A Phase I Trial of Intravesical Nanoparticle Albumin-Bound Paclitaxel in the Treatment of Bacillus Calmette-Guérin Refractory Nonmuscle Invasive Bladder Cancer. J. Urol. 2011, 186, 448–451. [Google Scholar] [CrossRef]
- Bassi, P.F.; Volpe, A.; D’Agostino, D.; Palermo, G.; Renier, D.; Franchini, S.; Rosato, A.; Racioppi, M. Paclitaxel-Hyaluronic Acid for Intravesical Therapy of Bacillus Calmette-Guérin Refractory Carcinoma In Situ of the Bladder: Results of a Phase I Study. J. Urol. 2011, 185, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, G.; Perdonà, S.; Damiano, R.; Faiella, A.; Cantiello, F.; Pignata, S.; Ascierto, P.; Simeone, E.; De Sio, M.; Autorino, R. Gemcitabine versus Bacille Calmette-Guérin after Initial Bacille Calmette-Guérin Failure in Non-Muscle-Invasive Bladder Cancer: A Multicenter Prospective Randomized Trial. Cancer 2010, 116, 1893–1900. [Google Scholar] [CrossRef] [PubMed]
- Perdonà, S.; Di Lorenzo, G.; Cantiello, F.; Damiano, R.; De Sio, M.; Masala, D.; Bruni, G.; Gallo, L.; Federico, P.; Quattrone, C.; et al. Is Gemcitabine an Option in BCG-Refractory Nonmuscle-Invasive Bladder Cancer? A Single-Arm Prospective Trial. Anticancer Drugs 2010, 21, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Laudano, M.A.; Barlow, L.J.; Murphy, A.M.; Petrylak, D.P.; Desai, M.; Benson, M.C.; McKiernan, J.M. Long-Term Clinical Outcomes of a Phase I Trial of Intravesical Docetaxel in the Management of Non-Muscle-Invasive Bladder Cancer Refractory to Standard Intravesical Therapy. Urology 2010, 75, 134–137. [Google Scholar] [CrossRef]
- Addeo, R.; Caraglia, M.; Bellini, S.; Abbruzzese, A.; Vincenzi, B.; Montella, L.; Miragliuolo, A.; Guarrasi, R.; Lanna, M.; Cennamo, G.; et al. Randomized Phase III Trial on Gemcitabine versus Mytomicin in Recurrent Superficial Bladder Cancer: Evaluation of Efficacy and Tolerance. J. Clin. Oncol. 2010, 28, 543–548. [Google Scholar] [CrossRef]
- Ignatoff, J.M.; Chen, Y.H.; Greenberg, R.E.; Pow-Sang, J.M.; Messing, E.M.; Wilding, G. Phase II Study of Intravesical Therapy with AD32 in Patients with Papillary Urothelial Carcinoma or Carcinoma In Situ (CIS) Refractory to Prior Therapy with Bacillus Calmette-Guerin (E3897): A Trial of the Eastern Cooperative Oncology Group. Urol. Oncol. 2009, 27, 496–501. [Google Scholar] [CrossRef]
- Mohanty, N.K.; Nayak, R.L.; Vasudeva, P.; Arora, R.P. Intravesicle Gemcitabine in Management of BCG Refractory Superficial TCC of Urinary Bladder-Our Experience. Urol. Oncol. 2008, 26, 616–619. [Google Scholar] [CrossRef]
- Gunelli, R.; Bercovich, E.; Nanni, O.; Ballardini, M.; Frassineti, G.L.; Giovannini, N.; Fiori, M.; Pasquini, E.; Ulivi, P.; Pappagallo, G.L.; et al. Activity of Endovesical Gemcitabine in BCG-Refractory Bladder Cancer Patients: A Translational Study. Br. J. Cancer 2007, 97, 1499–1504. [Google Scholar] [CrossRef]
- Dalbagni, G.; Russo, P.; Bochner, B.; Ben-Porat, L.; Sheinfeld, J.; Sogani, P.; Donat, M.S.; Herr, H.W.; Bajorin, D. Phase II Trial of Intravesical Gemcitabine in Bacille Calmette-Guérin-Refractory Transitional Cell Carcinoma of the Bladder. J. Clin. Oncol. 2006, 24, 2729–2734. [Google Scholar] [CrossRef]
- McKiernan, J.M.; Masson, P.; Murphy, A.M.; Goetzl, M.; Olsson, C.A.; Petrylak, D.P.; Desai, M.; Benson, M.C. Phase I Trial of Intravesical Docetaxel in the Management of Superficial Bladder Cancer Refractory to Standard Intravesical Therapy. J. Clin. Oncol. 2006, 24, 3075–3080. [Google Scholar] [CrossRef]
- Bartoletti, R.; Cai, T.; Gacci, M.; Giubilei, G.; Viggiani, F.; Santelli, G.; Repetti, F.; Nerozzi, S.; Ghezzi, P.; Sisani, M. Intravesical Gemcitabine Therapy for Superficial Transitional Cell Carcinoma: Results of a Phase II Prospective Multicenter Study. Urology 2005, 66, 726–731. [Google Scholar] [CrossRef] [PubMed]
- Bassi, P.; De Marco, V.; Tavolini, I.M.; Longo, F.; Pinto, F.; Zucchetti, M.; Crucitta, E.; Marini, L.; Dal Moro, F. Pharmacokinetic Study of Intravesical Gemcitabine in Carcinoma In Situ of the Bladder Refractory to Bacillus Calmette-Guérin Therapy. Urol. Int. 2005, 75, 309–313. [Google Scholar] [CrossRef]
- Dalbagni, G.; Russo, P.; Sheinfeld, J.; Mazumdar, M.; Tong, W.; Rabbani, F.; Donat, M.S.; Herr, H.W.; Sogani, P.; dePalma, D.; et al. Phase I Trial of Intravesical Gemcitabine in Bacillus Calmette-Guérin-Refractory Transitional-Cell Carcinoma of the Bladder. J. Clin. Oncol. 2002, 20, 3193–3198. [Google Scholar] [CrossRef] [PubMed]
- Rampersaud, E.N.; Vujaskovic, Z.; Inman, B.A. Hyperthermia as a Treatment for Bladder Cancer. Oncology 2010, 24, 1149. [Google Scholar]
- Seynhaeve, A.L.B.; Amin, M.; Haemmerich, D.; van Rhoon, G.C.; Ten Hagen, T.L.M. Hyperthermia and Smart Drug Delivery Systems for Solid Tumor Therapy. Adv. Drug Deliv. Rev. 2020, 163–164, 125–144. [Google Scholar] [CrossRef] [PubMed]
- Peer, A.J.; Grimm, M.J.; Zynda, E.R.; Repasky, E.A. Diverse Immune Mechanisms May Contribute to the Survival Benefit Seen in Cancer Patients Receiving Hyperthermia. Immunol. Res. 2010, 46, 137–154. [Google Scholar] [CrossRef] [PubMed]
- Liem, E.I.M.L.; Crezee, H.; de la Rosette, J.J.; de Reijke, T.M. Chemohyperthermia in Non-Muscle-Invasive Bladder Cancer: An Overview of the Literature and Recommendations. Int. J. Hyperth. 2016, 32, 363–373. [Google Scholar] [CrossRef]
- Zargar, H.; Aning, J.; Ischia, J.; So, A.; Black, P. Optimizing Intravesical Mitomycin C Therapy in Non-Muscle-Invasive Bladder Cancer. Nat. Rev. Urol. 2014, 11, 220–230. [Google Scholar] [CrossRef]
- Marquette, T.; Walz, J.; Rybikowski, S.; Maubon, T.; Branger, N.; Fakhfakh, S.; Verde, K.L.; Dermeche, S.; Gravis, G.; Pignot, G. Safety of Hyperthermic IntraVEsical Chemotherapy (HIVEC) for BCG Unresponsive Non-Muscle Invasive Bladder Cancer Patients. Prog. Urol. 2020, 30, 35–40. [Google Scholar] [CrossRef]
- De Jong, J.J.; Hendricksen, K.; Rosier, M.; Mostafid, H.; Boormans, J.L. Hyperthermic Intravesical Chemotherapy for BCG Unresponsive Non-Muscle Invasive Bladder Cancer Patients. Bladder Cancer 2018, 4, 395–401. [Google Scholar] [CrossRef]
- Tan, W.S.; Panchal, A.; Buckley, L.; Devall, A.J.; Loubière, L.S.; Pope, A.M.; Feneley, M.R.; Cresswell, J.; Issa, R.; Mostafid, H.; et al. Radiofrequency-Induced Thermo-Chemotherapy Effect Versus a Second Course of Bacillus Calmette-Guérin or Institutional Standard in Patients with Recurrence of Non-Muscle-Invasive Bladder Cancer Following Induction or Maintenance Bacillus Calmette-Guérin Therapy (HYMN): A Phase III, Open-Label, Randomised Controlled Trial. Eur. Urol. 2019, 75, 63–71. [Google Scholar] [CrossRef]
- Soria, F.; Milla, P.; Fiorito, C.; Pisano, F.; Sogni, F.; Di Marco, M.; Pagliarulo, V.; Dosio, F.; Gontero, P. Efficacy and Safety of a New Device for Intravesical Thermochemotherapy in Non-Grade 3 BCG Recurrent NMIBC: A Phase I–II Study. World J. Urol. 2016, 34, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Inman, B.A.; Stauffer, P.R.; Craciunescu, O.A.; Maccarini, P.F.; Dewhirst, M.W.; Vujaskovic, Z. A Pilot Clinical Trial of Intravesical Mitomycin-C and External Deep Pelvic Hyperthermia for Non-Muscle-Invasive Bladder Cancer. Int. J. Hyperth. 2014, 30, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Witjes, J.A.; Hendricksen, K.; Gofrit, O.; Risi, O.; Nativ, O. Intravesical Hyperthermia and Mitomycin-C for Carcinoma In Situ of the Urinary Bladder: Experience of the European Synergo Working Party. World J. Urol. 2009, 27, 319–324. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nativ, O.; Witjes, J.A.; Hendricksen, K.; Cohen, M.; Kedar, D.; Sidi, A.; Colombo, R.; Leibovitch, I. Combined Thermo-Chemotherapy for Recurrent Bladder Cancer after Bacillus Calmette-Guerin. J. Urol. 2009, 182, 1313–1317. [Google Scholar] [CrossRef] [PubMed]
- Rouprêt, M.; Seisen, T.; Birtle, A.J.; Capoun, O.; Compérat, E.M.; Dominguez-Escrig, J.L.; Gürses Andersson, I.; Liedberg, F.; Mariappan, P.; Hugh Mostafid, A.; et al. European Association of Urology Guidelines on Upper Urinary Tract Urothelial Carcinoma: 2023 Update. Eur. Urol. 2023, 84, 49–64. [Google Scholar] [CrossRef] [PubMed]
- Alanee, S.; Sana, S.; El-Zawahry, A.; Peabody, J.; Pearce, T.; Adams, N.; Deebajah, M.; Crabtree, J.; Delfino, K.; McVary, K.; et al. Phase I Trial of Intravesical Bacillus Calmette-Guérin Combined with Intravenous Pembrolizumab in Recurrent or Persistent High-Grade Non-Muscle-Invasive Bladder Cancer after Previous Bacillus Calmette-Guérin Treatment. World J. Urol. 2021, 39, 3807–3813. [Google Scholar] [CrossRef]
- Chamie, K.; Chang, S.S.; Kramolowsky, E.; Gonzalgo, M.L.; Agarwal, P.K.; Bassett, J.C.; Bjurlin, M.; Cher, M.L.; Clark, W.; Cowan, B.E.; et al. IL-15 Superagonist NAI in BCG-Unresponsive Non–Muscle-Invasive Bladder Cancer. NEJM Evid. 2022, 2, EVIDoa2200167. [Google Scholar] [CrossRef]
- Meghani, K.; Cooley, L.F.; Choy, B.; Kocherginsky, M.; Swaminathan, S.; Munir, S.S.; Svatek, R.S.; Kuzel, T.; Meeks, J.J. First-in-Human Intravesical Delivery of Pembrolizumab Identifies Immune Activation in Bladder Cancer Unresponsive to Bacillus Calmette-Guérin. Eur. Urol. 2022, 82, 602–610. [Google Scholar] [CrossRef]
- Li, R.; Amrhein, J.; Cohen, Z.; Champagne, M.; Kamat, A.M. Efficacy of Mycobacterium Phlei Cell Wall-Nucleic Acid Complex (MCNA) in BCG-Unresponsive Patients. Bladder Cancer 2017, 3, 65–71. [Google Scholar] [CrossRef]
- Huang, J.; Schisler, J.; Wong, H.C.; Rosser, C.J.; Sterbis, J. Intravesical ALT-803 for BCG-Unresponsive Bladder Cancer—A Case Report. Urol. Case Rep. 2017, 14, 15–17. [Google Scholar] [CrossRef]
- Morales, A.; Herr, H.; Steinberg, G.; Given, R.; Cohen, Z.; Amrhein, J.; Kamat, A.M. Efficacy and Safety of MCNA in Patients with Nonmuscle Invasive Bladder Cancer at High Risk for Recurrence and Progression after Failed Treatment with Bacillus Calmette-Guérin. J. Urol. 2015, 193, 1135–1143. [Google Scholar] [CrossRef] [PubMed]
- Kowalski, M.; Guindon, J.; Brazas, L.; Moore, C.; Entwistle, J.; Cizeau, J.; Jewett, M.A.S.; MacDonald, G.C. A Phase II Study of Oportuzumab Monatox: An Immunotoxin Therapy for Patients with Noninvasive Urothelial Carcinoma In Situ Previously Treated with Bacillus Calmette-Guérin. J. Urol. 2012, 188, 1712–1718. [Google Scholar] [CrossRef] [PubMed]
- Morales, A.; Phadke, K.; Steinhoff, G. Intravesical Mycobacterial Cell Wall-DNA Complex in the Treatment of Carcinoma In Situ of the Bladder after Standard Intravesical Therapy Has Failed. J. Urol. 2009, 181, 1040–1045. [Google Scholar] [CrossRef]
- Joudi, F.N.; Smith, B.J.; O’Donnell, M.A. Final Results from a National Multicenter Phase II Trial of Combination Bacillus Calmette-Guérin plus Interferon Alpha-2B for Reducing Recurrence of Superficial Bladder Cancer. Urol. Oncol. 2006, 24, 344–348. [Google Scholar] [CrossRef]
- O’Donnell, M.A.; Lilli, K.; Leopold, C. Interim Results from a National Multicenter Phase II Trial of Combination Bacillus Calmette-Guerin plus Interferon Alfa-2b for Superficial Bladder Cancer. J. Urol. 2004, 172, 888–893. [Google Scholar] [CrossRef]
- Lam, J.S.; Benson, M.C.; O’Donnell, M.A.; Sawczuk, A.; Gavazzi, A.; Wechsler, M.H.; Sawczuk, I.S. Bacillus Calmete-Guérin plus Interferon-A2B Intravesical Therapy Maintains an Extended Treatment Plan for Superficial Bladder Cancer with Minimal Toxicity. Urol. Oncol. Semin. Orig. Investig. 2003, 21, 354–360. [Google Scholar] [CrossRef]
- O’Donnell, M.A.; Krohn, J.; Dewolf, W.C. Salvage Intravesical Therapy with Interferon-A2b Plus Low Dose Bacillus Calmette-Guerin Is Effective in Patients with Superficial Bladder Cancer in Whom Bacillus Calmette-Guerin Alone Previously Failed. J. Urol. 2001, 166, 1300–1305. [Google Scholar] [CrossRef]
- Sarosdy, M.F.; Manyak, M.J.; Sagalowsky, A.I.; Belldegrun, A.; Benson, M.C.; Bihrle, W.; Carroll, P.R.; Ellis, W.J.; Hudson, M.A.; Sharkey, F.E. Oral Bropirimine Immunotherapy of Bladder Carcinoma In Situ after Prior Intravesical Bacille Calmette-Guérin. Urology 1998, 51, 226–231. [Google Scholar] [CrossRef]
- Belldegrun, A.S.; Franklin, J.R.; O’Donnell, M.A.; Gomella, L.G.; Klein, E.; Neri, R.; Nseyo, U.O.; Ratliff, T.L.; Williams, R.D. Superficial Bladder Cancer: The Role of Interferon-Alpha. J. Urol. 1998, 159, 1793–1801. [Google Scholar] [CrossRef]
- Kamat, A.M.; Lamm, D.L. Immunotherapy for Bladder Cancer. Curr. Urol. Rep. 2001, 2, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, A.R.H.; Shepherd, E.; Brook, N.R. Intravesical Bacillus Calmette-Guérin with Interferon-Alpha versus Intravesical Bacillus Calmette-Guérin for Treating Non-Muscle-Invasive Bladder Cancer. Cochrane Database Syst. Rev. 2017, 3, CD012112. [Google Scholar] [CrossRef] [PubMed]
- Filion, M.C.; Phillips, N.C. Therapeutic Potential of Mycobacterial Cell Wall-DNA Complexes. Expert. Opin. Investig. Drugs 2001, 10, 2157–2165. [Google Scholar] [CrossRef]
- Sarosdy, M.F. Bropirimine in Bladder Cancer: Clinical Studies. Ann. N. Y. Acad. Sci. 1993, 685, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Zappavigna, S.; Cossu, A.M.; Grimaldi, A.; Bocchetti, M.; Ferraro, G.A.; Nicoletti, G.F.; Filosa, R.; Caraglia, M. Anti-Inflammatory Drugs as Anticancer Agents. Int. J. Mol. Sci. 2020, 21, 2605. [Google Scholar] [CrossRef]
- Catalano, M.; Roviello, G.; Santi, R.; Villari, D.; Spatafora, P.; Galli, I.C.; Sessa, F.; Conte, F.L.; Mini, E.; Cai, T.; et al. Inflammation in Urological Malignancies: The Silent Killer. Int. J. Mol. Sci. 2023, 24, 866. [Google Scholar] [CrossRef]
- Cantiello, F.; Russo, G.I.; Vartolomei, M.D.; Farhan, A.R.A.; Terracciano, D.; Musi, G.; Lucarelli, G.; Di Stasi, S.M.; Hurle, R.; Serretta, V.; et al. Systemic Inflammatory Markers and Oncologic Outcomes in Patients with High-Risk Non-Muscle-Invasive Urothelial Bladder Cancer. Eur. Urol. Oncol. 2018, 1, 403–410. [Google Scholar] [CrossRef]
- Neganova, M.; Liu, J.; Aleksandrova, Y.; Klochkov, S.; Fan, R. Therapeutic Influence on Important Targets Associated with Chronic Inflammation and Oxidative Stress in Cancer Treatment. Cancers 2021, 13, 6062. [Google Scholar] [CrossRef]
- Biot, C.; Rentsch, C.A.; Gsponer, J.R.; Birkhäuser, F.D.; Jusforgues-Saklani, H.; Lemaître, F.; Auriau, C.; Bachmann, A.; Bousso, P.; Demangel, C.; et al. Preexisting BCG-Specific T Cells Improve Intravesical Immunotherapy for Bladder Cancer. Sci. Transl. Med. 2012, 4, 137ra72. [Google Scholar] [CrossRef]
- Han, K.P.; Zhu, X.; Liu, B.; Jeng, E.; Kong, L.; Yovandich, J.L.; Vyas, V.V.; Marcus, W.D.; Chavaillaz, P.A.; Romero, C.A.; et al. IL-15:IL-15 Receptor Alpha Superagonist Complex: High-Level Co-Expression in Recombinant Mammalian Cells, Purification and Characterization. Cytokine 2011, 56, 804–810. [Google Scholar] [CrossRef]
- Kamat, A.M.; Sylvester, R.J.; Böhle, A.; Palou, J.; Lamm, D.L.; Brausi, M.; Soloway, M.; Persad, R.; Buckley, R.; Colombel, M.; et al. Definitions, End Points, and Clinical Trial Designs for Non-Muscle-Invasive Bladder Cancer: Recommendations from the International Bladder Cancer Group. J. Clin. Oncol. 2016, 34, 1935–1944. [Google Scholar] [CrossRef]
- Sonpavde, G.; Rosser, C.J.; Pan, C.; Parikh, R.A.; Nix, J.; Gingrich, J.R.; Hernandez, L.; Huang, B.-Y.; Wong, H.C. Phase I Trial of ALT-801, a First-in-Class T-Cell Receptor (TCR)-Interleukin (IL)-2 Fusion Molecule, plus Gemcitabine (G) for Bacillus Calmette Guerin (BCG)-Resistant Non-Muscle-Invasive Bladder Cancer (NMIBC). J. Clin. Oncol. 2015, 33, e15509. [Google Scholar] [CrossRef]
- Wadhwa, N.; Mathew, B.B.; Jatawa, S.K.; Tiwari, A. Genetic Instability in Urinary Bladder Cancer: An Evolving Hallmark. J. Postgrad. Med. 2013, 59, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Boorjian, S.A.; Alemozaffar, M.; Konety, B.R.; Shore, N.D.; Gomella, L.G.; Kamat, A.M.; Bivalacqua, T.J.; Montgomery, J.S.; Lerner, S.P.; Busby, J.E.; et al. Intravesical Nadofaragene Firadenovec Gene Therapy for BCG-Unresponsive Non-Muscle-Invasive Bladder Cancer: A Single-Arm, Open-Label, Repeat-Dose Clinical Trial. Lancet Oncol. 2021, 22, 107–117. [Google Scholar] [CrossRef]
- Shore, N.D.; Boorjian, S.A.; Canter, D.J.; Ogan, K.; Karsh, L.I.; Downs, T.M.; Gomella, L.G.; Kamat, A.M.; Lotan, Y.; Svatek, R.S.; et al. Intravesical RAd-IFNα/Syn3 for Patients with High-Grade, Bacillus Calmette-Guerin-Refractory or Relapsed Non-Muscle-Invasive Bladder Cancer: A Phase II Randomized Study. J. Clin. Oncol. 2017, 35, 3410–3416. [Google Scholar] [CrossRef] [PubMed]
- Packiam, V.T.; Lamm, D.L.; Barocas, D.A.; Trainer, A.; Fand, B.; Davis, R.L.; Clark, W.; Kroeger, M.; Dumbadze, I.; Chamie, K.; et al. An Open Label, Single-Arm, Phase II Multicenter Study of the Safety and Efficacy of CG0070 Oncolytic Vector Regimen in Patients with BCG-Unresponsive Non-Muscle-Invasive Bladder Cancer: Interim Results. Urol. Oncol. 2018, 36, 440–447. [Google Scholar] [CrossRef]
- Navai, N.; Benedict, W.F.; Zhang, G.; Abraham, A.; Ainslie, N.; Shah, J.B.; Grossman, H.B.; Kamat, A.M.; Dinney, C.P.N. Phase 1b Trial to Evaluate Tissue Response to a Second Dose of Intravesical Recombinant Adenoviral Interferon A2b Formulated in Syn3 for Failures of Bacillus Calmette-Guerin (BCG) Therapy in Nonmuscle Invasive Bladder Cancer. Ann. Surg. Oncol. 2016, 23, 4110–4114. [Google Scholar] [CrossRef]
- Dinney, C.P.N.; Fisher, M.B.; Navai, N.; O’Donnell, M.A.; Cutler, D.; Abraham, A.; Young, S.; Hutchins, B.; Caceres, M.; Kishnani, N.; et al. Phase I Trial of Intravesical Recombinant Adenovirus Mediated Interferon-A2b Formulated in Syn3 for Bacillus Calmette-Guérin Failures in Nonmuscle Invasive Bladder Cancer. J. Urol. 2013, 190, 850–856. [Google Scholar] [CrossRef]
- Burke, J.M.; Lamm, D.L.; Meng, M.V.; Nemunaitis, J.J.; Stephenson, J.J.; Arseneau, J.C.; Aimi, J.; Lerner, S.; Yeung, A.W.; Kazarian, T.; et al. A First in Human Phase 1 Study of CG0070, a GM-CSF Expressing Oncolytic Adenovirus, for the Treatment of Nonmuscle Invasive Bladder Cancer. J. Urol. 2012, 188, 2391–2397. [Google Scholar] [CrossRef]
- Sidi, A.A.; Ohana, P.; Benjamin, S.; Shalev, M.; Ransom, J.H.; Lamm, D.; Hochberg, A.; Leibovitch, I. Phase I/II Marker Lesion Study of Intravesical BC-819 DNA Plasmid in H19 over Expressing Superficial Bladder Cancer Refractory to Bacillus Calmette-Guerin. J. Urol. 2008, 180, 2379–2383. [Google Scholar] [CrossRef]
- Lamm, D.; Brausi, M.; O’Donnell, M.A.; Witjes, J.A. Interferon Alfa in the Treatment Paradigm for Non-Muscle-Invasive Bladder Cancer. Urol. Oncol. 2014, 32, 35.e21–35.e30. [Google Scholar] [CrossRef]
- Benedict, W.F.; Tao, Z.; Kim, C.S.; Zhang, X.; Zhou, J.H.; Adam, L.; McConkey, D.J.; Papageorgiou, A.; Munsell, M.; Philopena, J.; et al. Intravesical Ad-IFNalpha Causes Marked Regression of Human Bladder Cancer Growing Orthotopically in Nude Mice and Overcomes Resistance to IFN-Alpha Protein. Mol. Ther. 2004, 10, 525–532. [Google Scholar] [CrossRef]
- Connor, R.J.; Anderson, J.M.; Machemer, T.; Maneval, D.C.; Engler, H. Sustained Intravesical Interferon Protein Exposure Is Achieved Using an Adenoviral-Mediated Gene Delivery System: A Study in Rats Evaluating Dosing Regimens. Urology 2005, 66, 224–229. [Google Scholar] [CrossRef]
- Yamashita, M.; Rosser, C.J.; Zhou, J.H.; Zhang, X.Q.; Connor, R.J.; Engler, H.; Maneval, D.C.; Karashima, T.; Czerniak, B.A.; Dinney, C.P.N.; et al. Syn3 Provides High Levels of Intravesical Adenoviral-Mediated Gene Transfer for Gene Therapy of Genetically Altered Urothelium and Superficial Bladder Cancer. Cancer Gene Ther. 2002, 9, 687–691. [Google Scholar] [CrossRef] [PubMed]
- Moussa, M.; Papatsoris, A.G.; Dellis, A.; Abou Chakra, M.; Saad, W. Novel Anticancer Therapy in BCG Unresponsive Non-Muscle-Invasive Bladder Cancer. Expert. Rev. Anticancer Ther. 2020, 20, 965–983. [Google Scholar] [CrossRef]
- Kulkarni, G.S.; Lilge, L.; Nesbitt, M.; Dumoulin-White, R.J.; Mandel, A.; Jewett, M.A.S. A Phase 1b Clinical Study of Intravesical Photodynamic Therapy in Patients with Bacillus Calmette-Guérin-Unresponsive Non-Muscle-Invasive Bladder Cancer. Eur. Urol. Open Sci. 2022, 41, 105–111. [Google Scholar] [CrossRef]
- Lee, J.Y.; Diaz, R.R.; Cho, K.S.; Lim, M.S.; Chung, J.S.; Kim, W.T.; Ham, W.S.; Choi, Y.D. Efficacy and Safety of Photodynamic Therapy for Recurrent, High Grade Nonmuscle Invasive Bladder Cancer Refractory or Intolerant to Bacille Calmette-Guérin Immunotherapy. J. Urol. 2013, 190, 1192–1199. [Google Scholar] [CrossRef] [PubMed]
- Zahoor, H.; Mir, M.C.; Barata, P.C.; Stephenson, A.J.; Campbell, S.C.; Fergany, A.; Dreicer, R.; Garcia, J.A. Phase II Trial of Continuous Treatment with Sunitinib in Patients with High-Risk (BCG-Refractory) Non-Muscle Invasive Bladder Cancer. Investig. New Drugs 2019, 37, 1231–1238. [Google Scholar] [CrossRef]
- Hahn, N.M.; Bivalacqua, T.J.; Ross, A.E.; Netto, G.J.; Baras, A.; Park, J.C.; Chapman, C.; Masterson, T.A.; Koch, M.O.; Bihrle, R.; et al. A Phase II Trial of Dovitinib in BCG-Unresponsive Urothelial Carcinoma with FGFR3 Mutations or Overexpression: Hoosier Cancer Research Network Trial HCRN 12-157. Clin. Cancer Res. 2017, 23, 3003–3011. [Google Scholar] [CrossRef] [PubMed]
- Berger, A.P.; Steiner, H.; Stenzl, A.; Akkad, T.; Bartsch, G.; Holtl, L. Photodynamic Therapy with Intravesical Instillation of 5-Aminolevulinic Acid for Patients with Recurrent Superficial Bladder Cancer: A Single-Center Study. Urology 2003, 61, 338–341. [Google Scholar] [CrossRef]
- Shore, N.D.; Gupta, S.; Kulkarni, G.S.; Necchi, A.; Dave, H.K.; Kapadia, E.; Zhao, Q.; Kamat, A.M. Phase 2 KEYNOTE-057 Cohort C: Pembrolizumab (Pembro) with Vibostolimab or Favezelimab for Patients (Pts) with High-Risk (HR) Bacillus Calmette-Guérin (BCG)-Unresponsive Non–Muscle-Invasive Bladder Cancer (NMIBC). J. Clin. Oncol. 2023, 41, TPS591. [Google Scholar] [CrossRef]
- Harjunpää, H.; Guillerey, C. TIGIT as an Emerging Immune Checkpoint. Clin. Exp. Immunol. 2020, 200, 108–119. [Google Scholar] [CrossRef] [PubMed]
- Long, L.; Zhang, X.; Chen, F.; Pan, Q.; Phiphatwatchara, P.; Zeng, Y.; Chen, H. The Promising Immune Checkpoint LAG-3: From Tumor Microenvironment to Cancer Immunotherapy. Genes Cancer 2018, 9, 176. [Google Scholar] [CrossRef]
- Catto, J.W.F.; Tran, B.; Master, V.A.; Roupret, M.; Pignot, G.; Tubaro, A.; Shimizu, N.; Vasdev, N.; Gschwend, J.E.; Loriot, Y.; et al. Phase 2 Study of the Efficacy and Safety of Erdafitinib in Patients (Pts) with Bacillus Calmette-Guérin (BCG)-Unresponsive, High-Risk Non–Muscle-Invasive Bladder Cancer (HR-NMIBC) with FGFR3/2 Alterations (Alt) in THOR-2: Cohort 2 Interim Analysis Results. J. Clin. Oncol. 2023, 41, 503. [Google Scholar] [CrossRef]
- Ye, D.; Liu, J.; Zhou, A.; Zou, Q.; Li, H.; Fu, C.; Hu, H.; Huang, J.; Zhu, S.; Jin, J.; et al. Tislelizumab in Asian Patients with Previously Treated Locally Advanced or Metastatic Urothelial Carcinoma. Cancer Sci. 2021, 112, 305–313. [Google Scholar] [CrossRef]
- Li, Z.; He, L.; Li, X.; Yao, K.; Liu, Y.; Qin, Z.; Ye, Y.; Zhou, F.; Liu, Z. Preliminary Results from a Phase II Study of Tislelizumab Combined with Radiotherapy as Bladder-Preserving Treatment for Patients with High-Risk Non-Muscle-Invasive Bladder Cancer (HR NMIBC) Unresponsive to Bacillus Calmette-Guerin (BCG). J. Clin. Oncol. 2023, 41, 510. [Google Scholar] [CrossRef]
- Powles, T.; Rosenberg, J.E.; Sonpavde, G.P.; Loriot, Y.; Durán, I.; Lee, J.-L.; Matsubara, N.; Vulsteke, C.; Castellano, D.; Wu, C.; et al. Enfortumab Vedotin in Previously Treated Advanced Urothelial Carcinoma. N. Engl. J. Med. 2021, 384, 1125–1135. [Google Scholar] [CrossRef]
- Kamat, A.M.; Steinberg, G.D.; Inman, B.A.; Kates, M.R.; Uchio, E.M.; Porten, S.P.; Roupret, M.; Redorta, J.; Catto, J.W.F.; Kulkarni, G.S.; et al. Study EV-104: Phase 1 Study of Intravesical Enfortumab Vedotin for Treatment of Patients with Non-Muscle Invasive Bladder Cancer (NMIBC)—Trial in Progress. J. Clin. Oncol. 2023, 41, TPS582. [Google Scholar] [CrossRef]
- Study of HX008 for the Treatment of BCG-Unresponsive Non-Muscle Invasive Bladder Cancer—Full Text View—ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT04738630 (accessed on 21 June 2023).
- Zhang, J.; Huang, Y.; Xi, G.; Zhang, F. HX008: A Humanized PD-1 Blocking Antibody with Potent Antitumor Activity and Superior Pharmacologic Properties. MAbs 2020, 12, 1724751. [Google Scholar] [CrossRef]
- EG-70 in NMIBC Patients Who Are BCG-Unresponsive and High-Risk NMIBC Patients Who Have Been Incompletely Treated with BCG or Are BCG-Naïve (LEGEND STUDY)—Full Text View—ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/show/NCT04752722?cond=bcg+bladder+cancer&draw=8 (accessed on 21 June 2023).
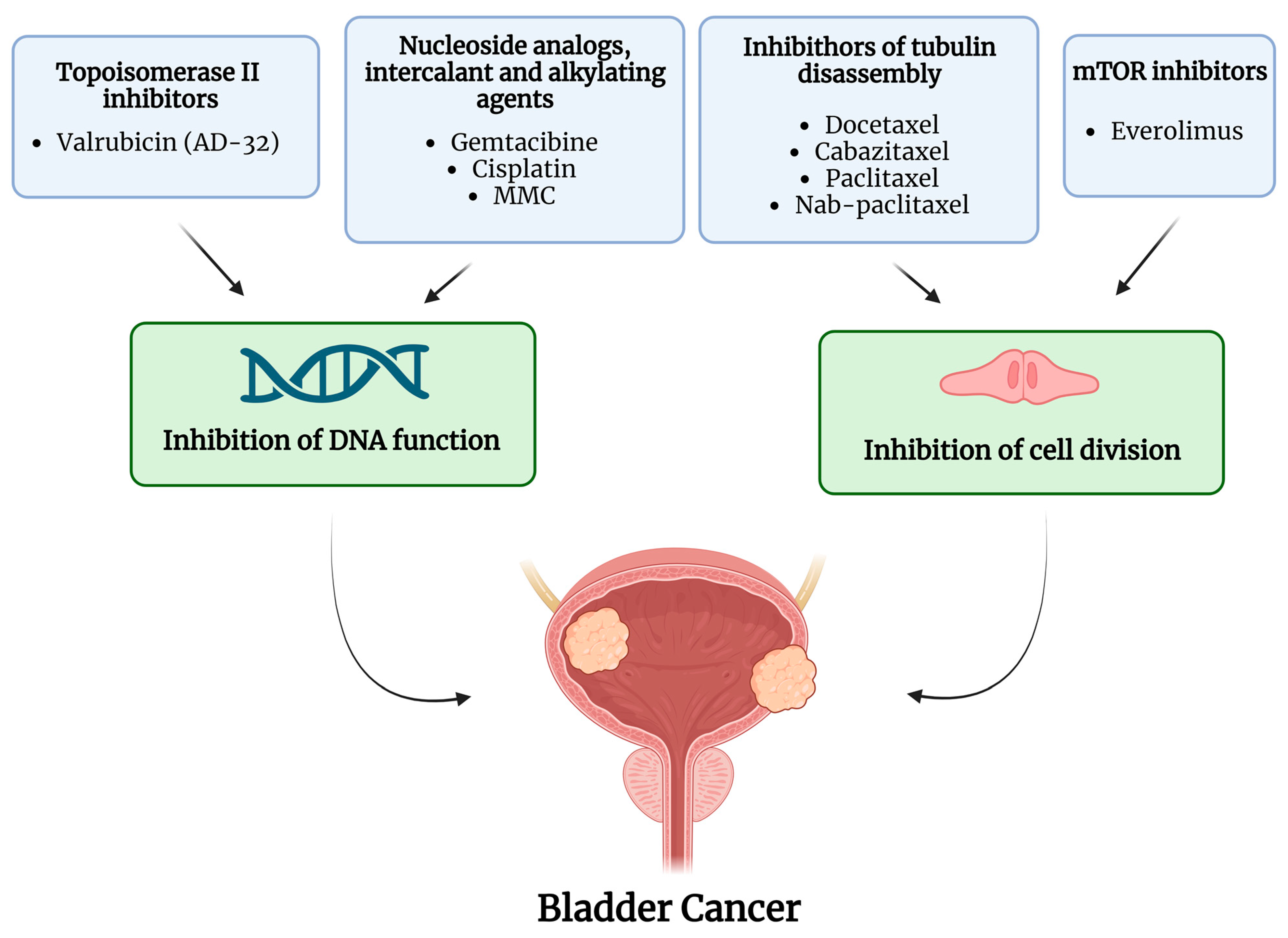
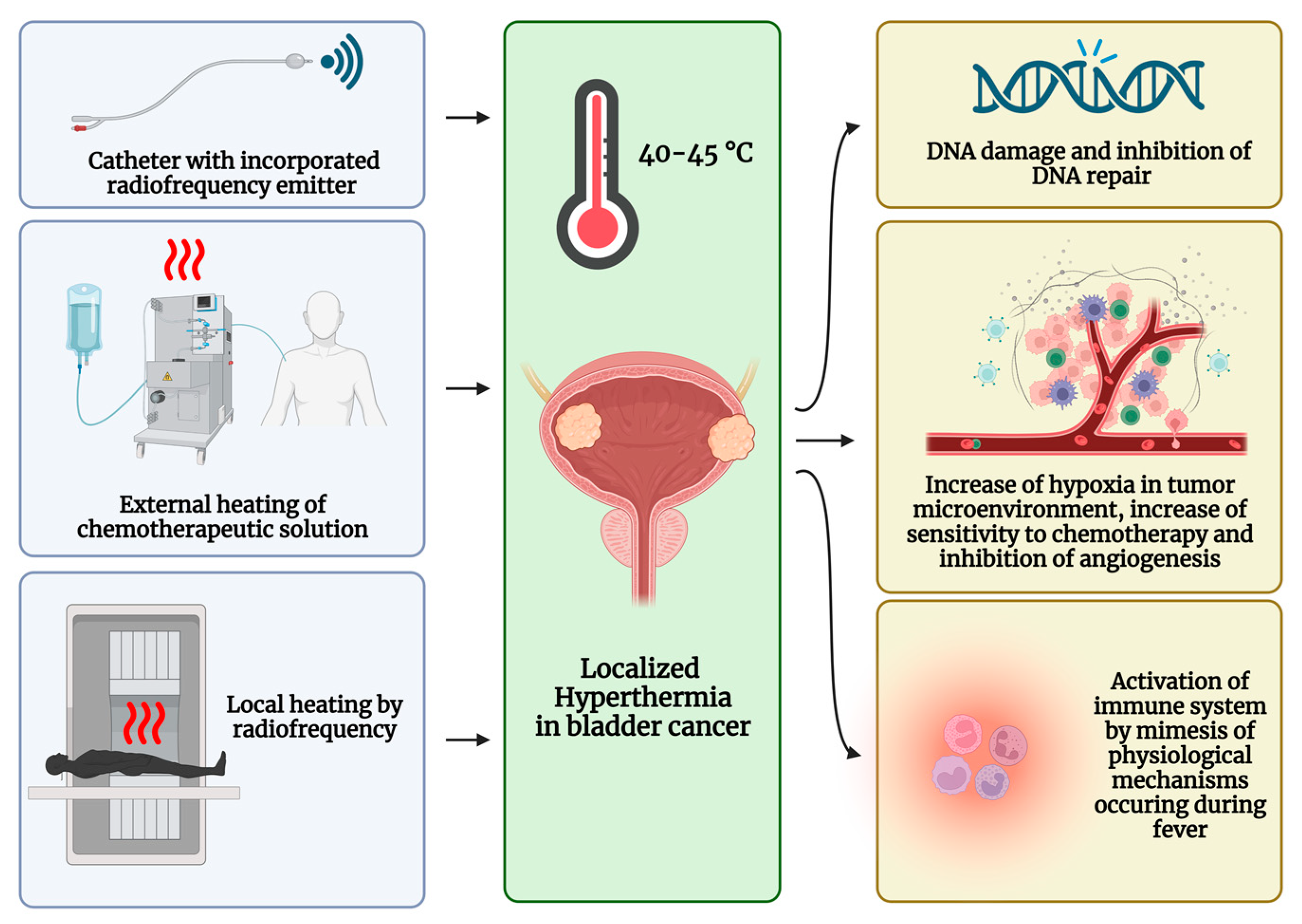
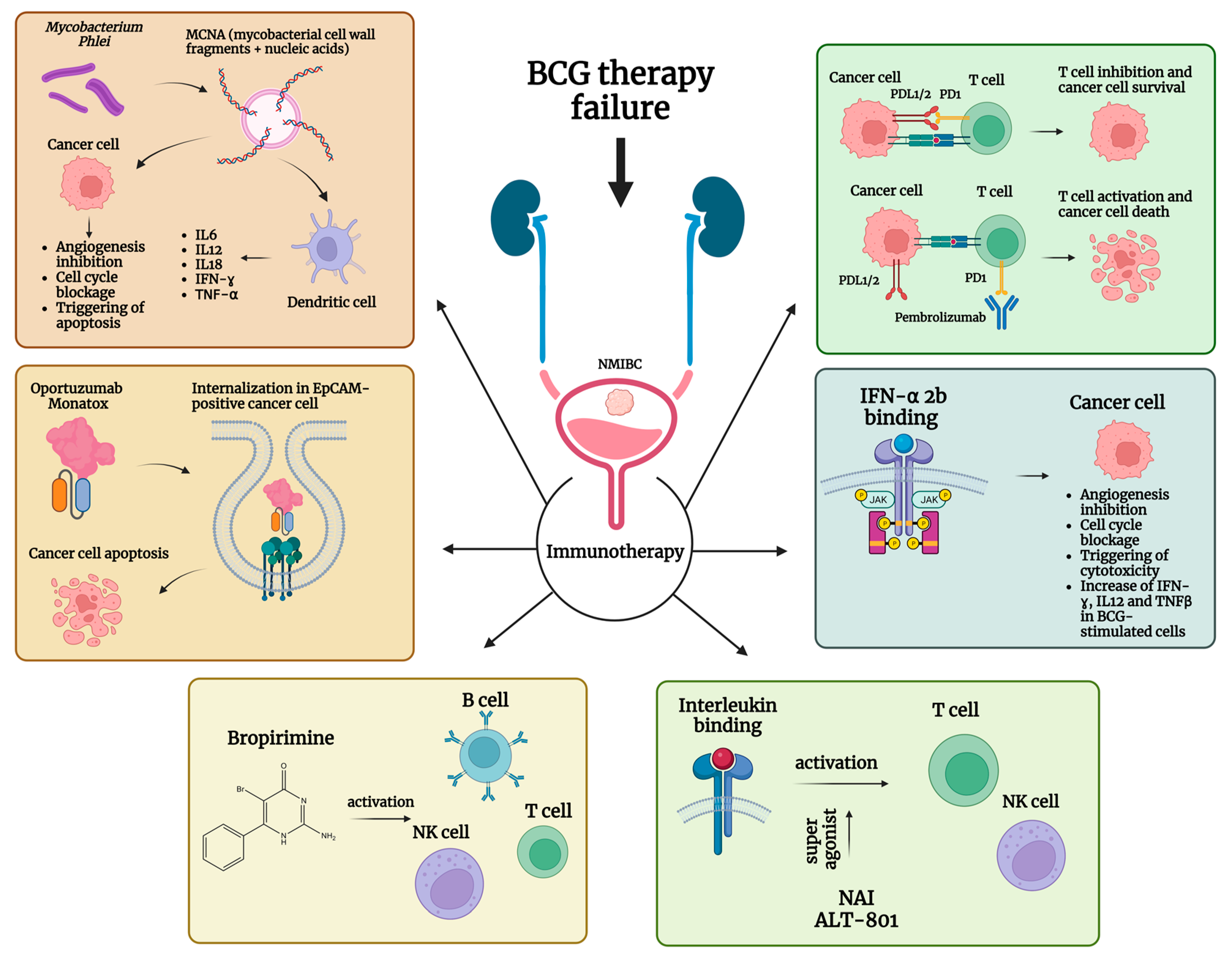
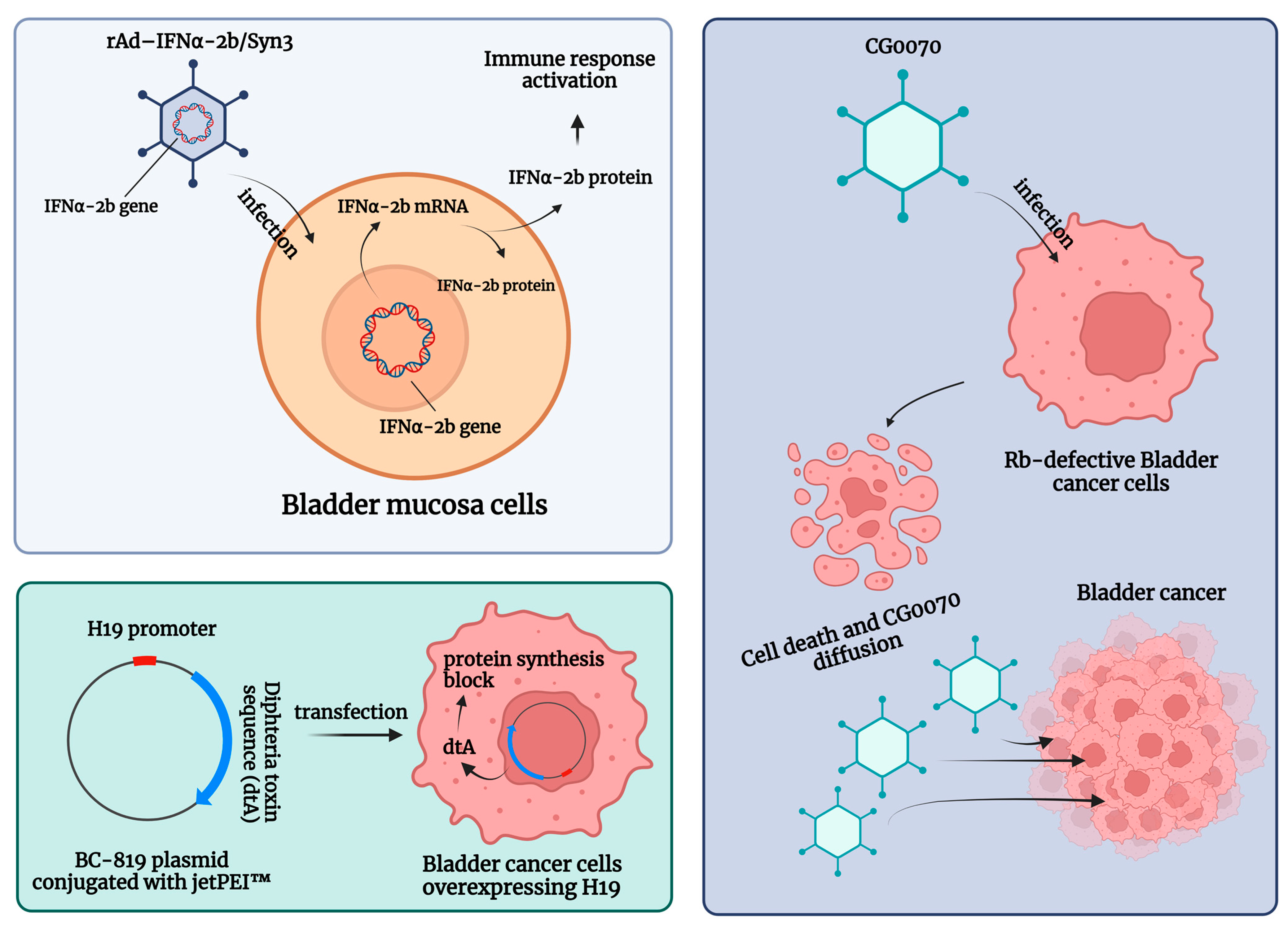
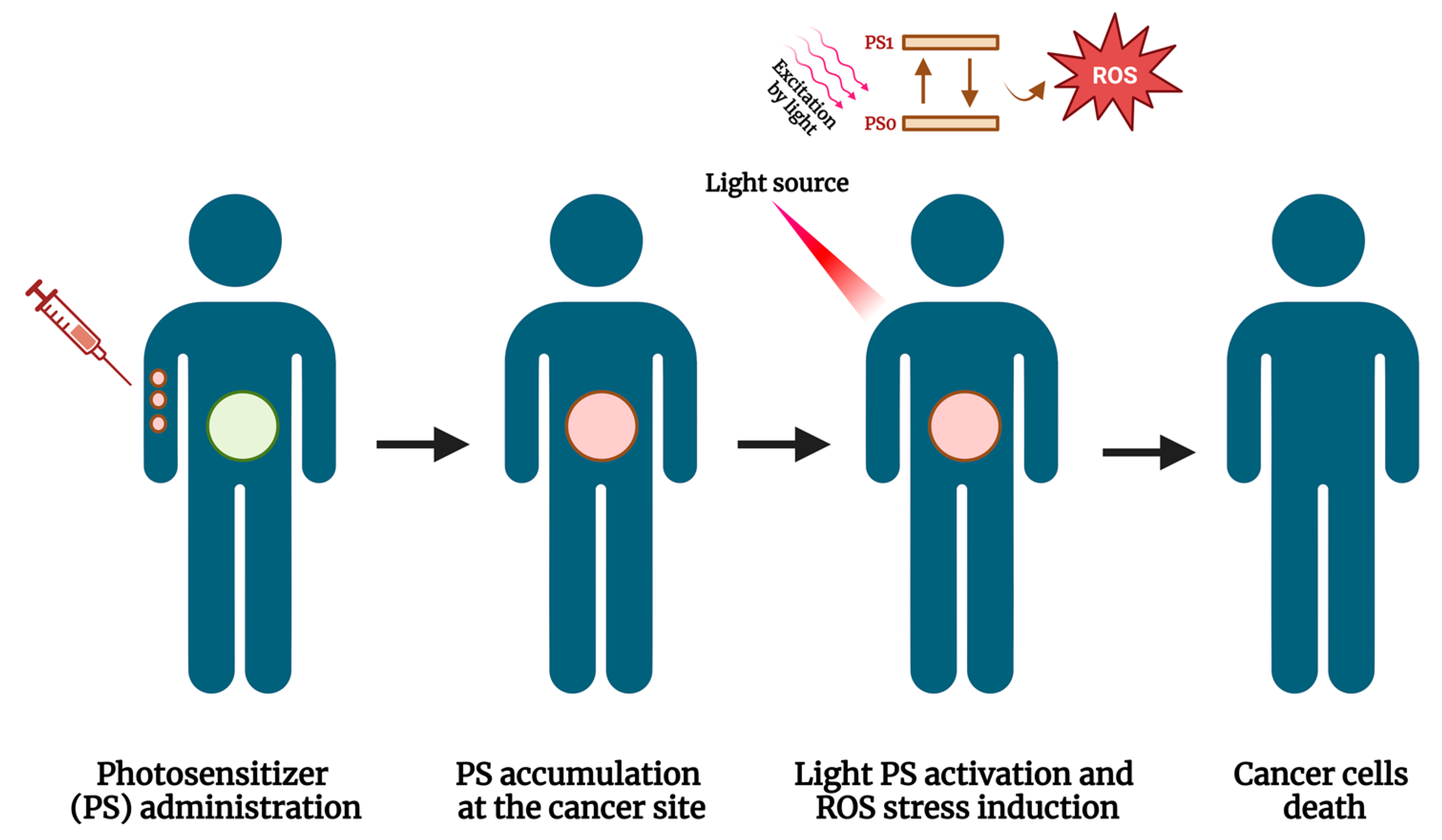
| Author, Year | Patients Number | Study Design | Treatment | Regimen | Effects of Treatment | Main Findings |
|---|---|---|---|---|---|---|
| Chevuru et al., 2023 [25] | 97 | Retrospective | Gemcitabine Docetaxel | Induction +/− maintenance | Gemcitabine: inhibition of DNA synthesis Docetaxel: inhibition of cell division | 12, 24, and 60 months RFS 57%, 44%, and 24% (12, 24, and 60 months HG-RFS 60%, 50%, and 30%, respectively); BCG unresponsive: 12, 24, and 60 months RFS 67%, 50%, and 28%; CIS-only: 1-, 2-, and 5-year RFS 48%, 38%, and 22%, papillary only: 1-, 2-, and 5-year RFS 64%, 49%, and 25% (p = 0.3); 12, 24, and 60 months RC-free survival 89%, 86%, and 75% |
| Hurle et al., 2020 [26] | 36 | Open-label, single-arm | Gemcitabine | Induction +/− maintenance | Gemcitabine: inhibition of DNA synthesis | 12 and 24 months DFS 44.44% (95% CI 28.02–59.64%) and 31.66% (95% CI 16.97–47.43%); 12 and 24 months PFS 80.13% (95% CI: 62.78–90.00%) and 69.55% (95% CI: 50.33% 82.52%), 24 months CSS 80.68% (95% CI 61.49–90.96%), 24 months OS 77.9% (95% CI 58.78–88.92%) |
| Steinberg et al., 2020 [27] | 276 | Retrospective | Gemcitabine Docetaxel | Induction +/− maintenance | Gemcitabine: inhibition DNA synthesis Docetaxel: inhibition of cell division | 1 and 2 years RFS 60% and 46% (43% if CIS present); HG 1 and 2 years RFS 65% and 52% (50% if CIS present) |
| DeCastro et al., 2020 [28] | 18 | Phase I trial | Cabazitaxel Gemcitabin Cisplatin | Induction +/− maintenance (cabazitaxel + gemcitabine) | Cabazitaxel: inhibition of cell division Gemcitabine: inhibition of DNA synthesis Cisplatin: inhibition of DNA replication and transcription | CR rate: 89%; PR: 94% (negative biopsy but positive cytology). One and two years RFS 0.83 (range 0.57 to 0.94) and 0.64 (0.32 to 0.84), median 27 months. One- and two-years RC-free survival 0.94 (0.67 to 0.99) and 0.81 (0.52 to 0.94) |
| Milbar et al., 2017 [29] | 33 (22 BCG unresponsive/relapsing) | Retrospective | Gemcitabine Docetaxel | Induction +/− maintenance | Gemcitabine: inhibition DNA synthesis Docetaxel: inhibition of cell division | 1 and 2 years DFS38% and 24%. One and two years HG-RFS 49% and 34%. |
| Dalbagni et al., 2017 [30] | 19 | Single-arm, phase I/II trial | Gemcitabine Everolimus (oral) | Gemcitabine induction + everolimus maintenance | Gemcitabine: inhibition of DNA synthesis Everolimus: mTOR inhibition | 3, 6, and 9, 12 months RFS 58% (95% CI 33–76%), 27% (95% CI 9–49%) and 20% (95% CI 5–42%) |
| Robins et al., 2017 [31] | 22 | Single-arm, open-label, phase II trial | Nab-paclitaxel | Induction +/− maintenance | Nab-paclitaxel: inhibition of cell division | Overall CR 36%, non-CIS CR 63, CIS CR 25%. One and three-year RFS 32% and 18% (no CIS CR 40%, CIS CR 10%), |
| Cockerill et al., 2016 [32] | 27 | Retrospective | Gemcitabine MMC | Induction, no standardized maintenance | Gemcitabine: inhibition of DNA synthesis MMC: inhibition of DNA functions. | 63% recurrence rate, median RFS 15.2 months (range 1.7–32). RFS 37% (median follow-up 22 months) |
| Steinberg et al., 2015 [33] | 45 | Retrospective | Gemcitabine Docetaxel | Induction +/− maintenance | Gemcitabine: inhibition of DNA synthesis Docetaxel: inhibition of cell division | Treatment success (no recurrence + no cystectomy) 66% at 12 weeks, 54% at 1 year, 34% at 2 years; median time to failure 3.1 months (range 2.2–25.9) |
| Lightfoot et al., 2014 [34] | 52 (10 BCG naive) | Retrospective | Gemcitabine MMC | Induction +/− maintenance | Gemcitabine: inhibition of DNA synthesis MMC: inhibition of DNA functions. | CR 68%, 1- and 2-year RFS, 48%, and 38% |
| McKiernan et al., 2014 [35] | 28 | Single-arm, phase II trial | Nab-paclitaxel | Induction +/− maintenance | Nab-paclitaxel: inhibition of cell division | 35.7% CR, 1 and 2-year RFS 35.7% and RFS 30.6%. 12, 24, and 36 months CFS 74%, 74%, and 55% |
| Barlow et al., 2013 [36] | 54 | Retrospective | Docetaxel | Induction +/− maintenance | Docetaxel: inhibition of cell division | 59% CR (cystoscopy with biopsy + cytology); 1- and 3-year RFS 40% and 25%; 31% RC rate |
| Skinner et al., 2013 [37] | 58 | Single-arm, open-label, phase II trial | Gemcitabine | Induction +/− maintenance | Gemcitabine: inhibition of DNA synthesis | 3 months CR (negative cystoscopy, urinary cytology +/− biopsy) 47%. Median RFS 6.1 months (95% CI 16–43), 21% at 24 months. Progression/RC rate 36%. |
| Sternberg et al., 2013 [38] | 69 | Retrospective | Gemcitabine | Induction | Gemcitabine: inhibition of DNA synthesis | 5 years progression rate 19% for BCG-refractory pts, 22% for pts with other types of BCG failure (HR 1.09, 95% CI 0.34–3.50). CR (negative cystoscopy and cytology) in 27 pts, PR (negative cystoscopy and positive cytology in 19 with), NR (positive cystoscopy) 20 pts. Subsequent RC in 20 pts |
| Steinberg et al., 2011 [39] | 90 | Single-arm, pivotal phase III open-label study | Valrubicin | Induction | Valrubicin: inhibition of DNA and RNA synthesis | CR in 18 pts at 3 and 6 months (negative cytology, cystoscopy, and biopsy), NR 64 pts |
| McKiernan et al., 2011 [40] | 18 | Phase I trial | Nab-paclitaxel | Induction | Nab-paclitaxel: inhibition of cell division | CR in 5 patients (28%), 13 NR (stage progression in 1) |
| Bassi et al., 2011 [41] | 16 | Single-arm, open-label, phase I trial | Paclitaxel-hyaluronic acid | Induction | Paclitaxel: inhibition of cell division | 6 NR (40%), 9 disease-free pts (60%) |
| Di Lorenzo et al., 2010 [42] | 80 | Multicentric, phase II trial, randomized | Gemcitabine vs. BCG | Induction + maintenance for both arms | Gemcitabine: inhibition of DNA synthesis BCG: stimulating cellular and humoral immune response | 2-year RFS 19% for Gem (95% CI, 5–39), 3% for BCG (95% CI, 0–21; HR, 0.15; 95% CI, 0.1–0.3.008). Progression rate 33% for Gem, 37.5% for BCG |
| Perdonà et al., 2010 [43] | 20 | Single-arm, phase II trial | Gemcitabine | Induction + maintenance | Gemcitabine: inhibition of DNA synthesis | 3 months CR at the first 75%; 55% recurrence rate (11 of 20 pts); 45% progression rate (5 of 11 pts) |
| Laudano et al., 2010 [44] | 18 | Single-arm, phase I trial | Docetaxel | Induction | Docetaxel: inhibition of cell division | 22% CR, 17% PR (NMIBC recurrence requiring TURBT with no further treatment), 61% NR (RC or further pharmacologic therapy). PFS 89%. |
| Addeo et al., 2010 [45] | 109 | Phase III trial randomized | Gemcitabine vs. MMC | Induction +/− maintenance for both arms | Gemcitabine: inhibition of DNA synthesis MMC: inhibition of DNA functions. | RFS in gemcitabine arm, 72% (39 of 54 pts), in MMC arm 61% (33 of 55 pts). Stage progression in 10 pts in the MMC arm and 6 in the gem arm |
| Ignatoff et al., 2009 [46] | 38 | Multicentric, single-arm, phase II trial | AD32 (doxorubicin analog with limited systemic exposure) | Induction | AD32: inhibition of DNA functions and induction of apoptosis | CR 42.9% (90% CI: 24.5%, 62.8%), CIS CR 23.8% (90% CI: 9.9%, 43.7%). 12 and 24 RFS months 20% (90% CI: 7.8–36.1%) and 15% (CI, 4.9%, 30.2%),12 and 24 CIS RFS 80% (90% CI, 31.4%, 95.8%) if previous CR. PFS 22.4 months, CIS PFS 8.7 months |
| Mohanty et al., 2008 [47] | 35 | Single-arm, non-randomized, phase I trial | Gemcitabine | Induction | Gemcitabine: inhibition of DNA synthesis | At 18 months follow-up 21 disease free pts (60%), 11 pts (31.4%) with superficial recurrences, 3 (8.75%) with MIBC. Average RFS 12 months, average time to progression 16 months. |
| Gunelli et al., 2007 [48] | 40 | Single-arm, phase II trial | Gemcitabine | Induction | Gemcitabine: inhibition of DNA synthesis | 95% (38 of 40 pts) CR at 6 months (cystoscopy + cytology); overall event-free survival rate 80% at 1 year and 66% at 2.5 years. At a median follow-up of 28 months, 35% relapse rate (NMIBC). RC in 2 pts |
| Dalbagni et al., 2006 [49] | 30 | Single-arm, phase II trial | Gemcitabine | Induction | Gemcitabine: inhibition of DNA synthesis | 50% CR; median RFS 3.6 months (95% CI, 2.9 to 11.0 months); 21% 1-year RFS in pevious CR (95% CI, 0% to 43%). RC rate 37% |
| McKiernan et al., 2006 [50] | 18 | Single-arm, phase I trial | Docetaxel | Induction | Docetaxel: inhibition of cell division | CR in 56% (10 pts) |
| Bartoletti et al., 2005 [51] | 40 BCG refractory (total population 116) | Multicentric, single-arm, phase II trial | Gemcitabine | Induction | Gemcitabine: inhibition of DNA synthesis | Recurrence rate 32.5%, relapse in 6 (25%) of 24 intermediate-risk BCG refractory pts and 7 (43.7%) of 16 BCG refractory high-risk pts |
| Bassi et al., 2005 [52] | 9 | Single-arm, phase I trial | Gemcitabine | Induction +/− maintenance | Gemcitabine: inhibition of DNA synthesis | CR in 4/9 pts |
| Dalbagni et al., 2002 [53] | 14 | Single-arm, phase I trial | Gemcitabine | Induction (dose levels 500 mg, 1.000 mg, 1.500 mg, and 2.000 mg. | Gemcitabine: inhibition DNA synthesis | CR (defined as a negative posttreatment cystoscopy with biopsy of the urothelium + negative cytology) in 7, failure in 11 (negative bladder biopsy + persistent positive cytology), RC rate 1/11 pts |
| Author, Year | Patients Number | Study Design | Treatment | Regimen | Effects of Treatment | Main Findings |
|---|---|---|---|---|---|---|
| Marquette et al., 2020 [59] | 22 | Retrospective | HIVEC MMC | Induction | MMC: inhibition of DNA functions. Hyperthermia: DNA damage, ROS generation, cell death induction, damage to tumor vascular system, activation of immune response. | Progression to MIBC: 2 pts (9.1%) Time to to RC: 4 pts (18.2%) |
| De Jong et al., 2018 [60] | 55 | Post hoc analysis of prospective data | MMC + hyperthermia | Induction + maintenance | MMC: inhibition of DNA functions. Hyperthermia: DNA damage, ROS generation, cell death induction, damage to tumor vascular system, activation of immune response. | Median DFS 17.7 months, papillary only DFS 28.8 months, papillary + CIS DFS 17.7 months, T1/ T1 + CIS DFS 12.1 months CR 70% at 3 months if concomitant CIS. One-year disease recurrence/progression 53%. Overall CR 50%, recurrence rate 42% and progression rate 8%. RC rate 50% |
| Tan et al., 2019 [61] | 104 (48 randomized to MMC + hyperthermia) | Phase III trial, open-label, randomized with control | MMC + hyperthermia vs. BCG or institutional standard of care defined at randomization | Induction +/− maintenance | MMC: Inhibition of DNA functions. Hyperthermia: DNA damage, ROS generation, cell death induction, damage to tumor vascular system, activation of immune response. BCG: stimulating cellular and humoral immune response. | No statistically significant differences between the 2 arms in 24 months DFS (35% vs. 41%, HR 1.33, 95% CI 0.84–2.10, p = 0.23; adjusted p = 0.49) and 3 months CIS CR (30% vs. 47%, OR 0.43, 95% CI 0.18–1.28, p = 0.15). Lower DFS of experimental arm lower if CIS (HR 2.06, 95% CI 1.17–3.62, p = 0.01). 24 months DFS in non-CIS pts for experimental and control arms 53% and 24%,. worse DFS in papillary tumor + CIS than in CIS only. No differential treatment effect in CIS only pts (HR 1.53, 95% CI 0.77–3.05, p = 0.22). No difference between the arms in PFS (24 month rates 83% vs. 87%, p = 0.16), and RFS (24 month rates 23% vs. 40%; p = 0.98), borderline difference in DSS (24 month rates 89% vs. 96%; p = 0.04). Progression in 4 pts in both arms. |
| Soria et al., 2016 [62] | 34 | Multicentric, single-arm, phase I–II trial | MMC + hyperthermia | Induction | MMC: inhibition of DNA functions. Hyperthermia: DNA damage, ROS generation, cell death induction, damage to tumor vascular system, activation of immune response. | Response rate 59%, 42 NR, 1-year progression rate 18%; 41 months DFS 44.1%. RFS 10.5 months, PFS 29.5 months, RC-free survival 20 months (range 8–60). At median follow-up of 41 months, recurrence and progression rates 35.3 and 23.5% |
| Inman et al., 2014 [63] | 15 | Pilot prospective trial | MMC + hyperthermia | Induction +/− maintenance | MMC: inhibition of DNA functions. Hyperthermia: DNA damage, ROS generation, cell death induction, damage to tumor vascular system, activation of immune response. | 67% recurrence rate, RFS 15.4 months, 60% RC rate, RC-free survival 20.1 months |
| Witjes et al., 2009 [64] | 51 (17 BCG refractory, 15 BCG relapsing) | Retrospective | MMC + hyperthermia | Induction + maintenance | MMC: inhibition of DNA functions. Hyperthermia: DNA damage, ROS generation, cell death induction, damage to tumor vascular system, activation of immune response. | CR (negative cytology and biopsy) 92%, 50% at 2 years. 49% recurrence rate, mean RFS 27 months (median 22 months). |
| Nativ et al., 2009 [65] | 111 | Retrospective | MMC + hyperthermia | Induction + maintenance | MMC: inhibition of DNA functions. Hyperthermia: DNA damage, ROS generation, cell death induction, damage to tumor vascular system, activation of immune response. | 1 and 2 year DFS 85% and 56%, respectively; average DFS 16 months; 3% progression rate to MIBC. Two-year recurrence rate of 61% if maintenance not administered, 39% if administered. |
| Author, Year | Patients Number | Study Design | Treatment | Regimen | Effects of Treatment | Main Findings |
|---|---|---|---|---|---|---|
| Meghani et al., 2022 [69] | 9 | Single-arm, phase I trial | BCG + pembrolizumab (intravesical) | Induction + maintenance (pembrolizumab only) | BCG: stimulating cellular and humoral immune response. Pembrolizumab: stimulation of immune response. | 3, 6, and 12 months RFS 100%, 67% (95% CI: 42–100%) and 22% (95% CI: 7–75%), respectively. Median RFS 6.2 months. Progression in 5 pts, 6 and 12 months PFS 100% and 56% (95% CI: 31–100%) |
| Chamie et al., 2022 [68] | 171 (cohorts A and C: pts with BCG-unresponsive CIS with or without Ta/T1 papillary NMIBC; cohort B pts with BCG-unresponsive high-grade Ta/T1 papillary NMIBC) | Pivotal trial, multicentric open-label, single-arm, three- cohort | IL-15 superagonist NAI (intravesical) + BCG vs. NAI (intravesical) alone | Induction +/− maintenance | NAI: stimulation of immune response. BCG: stimulating cellular and humoral immune response. | Cohort A: 71% 3, 6, and 12 months CR 55% (45 of 82 patients; 95% CI = 43.5% to 65.9%), 56% (46 of 82 patients; 95% CI = 44.7% to 67.0%), and 45% (37 of 82 patients; 95% CI = 34.1% to 56.5%). 24 months PFS 84.7%%. 7% RC rate in CR pts, 33% in NR Cohort B: 12, 18 and 24 months DFS 55.4% (95% CI = 42.0% to 66.8%), 51.1% (95% CI = 37.6% to 63.1%) and 48.3% (95% CI = 34.5% to 60.7%). RC rate 7%. Cohort C: 3 months CR 20%, 10% of pts with CR maintained at 6 months |
| Alanee et al., 2021 [67] | 18 | Single-arm, phase I trial | BCG + intravenous pembrolizumab | BCG induction + 6 doses of pembrolizumab every 3 weeks concurrently with BCG | BCG: stimulating cellular and humoral immune response Pembrolizumab: stimulation of immune response. | CR 69% at 3 months following BCG treatment. One patient progressed to MIBC. |
| Balar et al., 2021 [10] | 96 | Open-label, single-arm, multicentric, phase II trial | Pembrolizumab (intravenous) | Every 3 weeks up to 24 months | Pembrolizumab: stimulation of immune response. | 39 pts (41%; 95% CI 30∙7–51∙1) achieved CR at 3 months, median duration of CR 16.2 months (95% CI 6∙7–36∙2), 51% recurrence after CR. At 12 months PFS to worsening of grade or stage or death 83% (95% CI 70.22–90.4), PFS to MIBC or metastatic disease or death 97% (86.0–99.2). |
| Li et al., 2017 [70] | 94 | Retrospective | MCNA | Induction +/− maintenance | MCNA: stimulation of immune response, apoptosis induction. | 6, 12, and 24 months DFS 48.9% (95% CI 38.0–59.0%), 34.8% (95% CI 24.7–45%) and 28.3% (15.7–34.3%). Papillary-only 6, 12, and 24 months DFS 61.2% (95% CI 38.2–77.8%), 61.2% (95% CI 38.2–77.8%) and 50.1% (95% CI 27.5–69.0%) at 2 years. In pts with CIS +/− papillary disease 6, 12 and 24 months DFS 44.8% (95% CI 32.3–56.4%), 26.5% (95% CI 16.3–37.9%) and 16.6% (95% CI 8.6–26.9%). 47.9% RC rate, 16% progression rate to MIBC and 11.7% to metastatic disease. |
| Huang et al., 2017 [71] | 1 | Case report | ALT-803 (NAI) + BCG | Induction | ALT-803 (NAI): stimulating immune response. BCG: stimulating cellular and humoral immune response. | No evidence of recurrence at 19 months after treatment. |
| Morales et al., 2015 [72] | 129 | Open-label multicentric, single-arm phase II trial | MCNA | Induction +/− maintenance | MCNA: stimulation of immune response, apoptosis induction. | 6, 12, and 24 months CR 34.1%, 22.5% and 14.7% at (34%, 195%, and 11% in CIS pts). 21.7% progression rate, 6, 12, 24, and 36 months PFS 95%, 87.3%, 79.8%, and 77.7%. RC rate 43%. |
| Kowalski et al., 2012 [73] | 46 | Open-label, multicentric, 2-arm phase I trial | Oportuzumab monatox | Induction +/− maintenance | Oportuzumab monatox: apoptosis induction | CR 44%, 16% at 1 year. One-year recurrence rate 65%. Two pts progressed to MIBC. |
| Morales et al., 2009 [74] | 55 | Single-arm, phase II trial | MCNA | Induction +/− maintenance | MCNA: stimulation of immune response, apoptosis induction. | CR 27.3% at 12 and 26 weeks, 31.8% and 22.7% at 12 and 18 months in the group receiving 4 mg, 46.4% at 12 and 26 weeks, 25% and 28.6% at 12 and 18 months in the group receiving 8 mg. |
| Joudi et al., 2006 [75] | 1106 (467 BCG failure) | Multicentric, single-arm, phase II trial | BCG + IFNα2b (intravesical) | Induction +/− maintenance | BCG: stimulating cellular and humoral immune response. Interferon alpha-2b: induction of cytotoxic effects | 24 months disease-free rate 45% |
| O’Donnel et al., 2004 [76] | 490 (231 BCG failure) | Phase II trial | BCG + IFNα2b (intravesical) | Induction + maintenance | BCG: stimulating cellular and humoral immune response. Interferon alpha 2 b: induction of cytotoxic effects | In BCG failure group, recurrence rate 51.5%, 20.8% at 3 months. 24 months disease-free rate 42%, median RFS 16 months. Progression rate to MIBC 4.3%, to metastatic disease 2.6%. RC rate 3.9% |
| Lam et al., 2003 [77] | 32 (20 BCG failure) | Retrospective | BCG + IFNα2b (intravesical) | Induction +/− maintenance | BCG: stimulating cellular and humoral immune response Interferon alpha 2 b: induction of cytotoxic effects. | In BCG failure group, 60% disease-free rate, 40% 3-month failure rate. Three pts underwent RC. |
| O’Donnell et al., 2001 [78] | 40 | Multicentric phase II trial | BCG + IFNα2b (intravesical) | Induction + maintenance | BCG: stimulating cellular and humoral immune response Interferon alpha-2b: induction of cytotoxic effects | 12, 24, and 30 months RFS 63%, 53%, and 55% |
| Sarosdy et al., 1998 [79] | 86 (60 BCG-resistant CIS, 26 with BCG-intolerant CIS) | Single-arm, phase II trial | Bropirimine | Oral self-administration, 3.0 g/day for 3 consecutive days, weekly, up to 1 year | Bropirimine: stimulation of immune response | CR in 14 pts (30%, 95% CI 16.7 to 42.9) of 47 evaluable BCG-resistant pts, 2 additional pts free of CIS (negative biopsy and negative cytology) but recurrent papillary tumor. Overall CR in 21 pts (24%). RFS in CR pts 65–810 days. RC in 26 pts. Progression to MIBC or metastatic disease in 4 pts (6%). |
| Author, Year | Patients Number | Study Design | Treatment | Regimen | Effects of Treatment | Main Findings |
|---|---|---|---|---|---|---|
| Boorjian et al., 2021 [94] | 151 | Multicentric open-label repeat-dose phase III trial | rAd-IFNa/Syn3 (intravesical) | Induction +/− maintenance | rAd–IFNa2b/Syn3: cytotoxic effects | 3-month CR: 55 (53.4%) CIS pts (with or without a high-grade Ta or T1 tumor). 12-month maintained CR among 25/55 (45.5%). |
| Shore et al., 2017 [95] | 40 | Phase II trial, randomized, | rAd–IFNα-2b/Syn3 (intravesical) | Induction +/− maintenance | rAd–IFNa2b/Syn3: cytotoxic effects | 12-month RFS: 35% Time to HG recurrence: 6.5 months. RC rate: 35%. 11 pts were disease-free for 15 to more than 36 months. |
| Packiam et al., 2018 [96] | 45 | Open-label, multicentric, single-arm, interventional phase II trial | CG0070 (replication selective serotype-5 oncolytic adenovirus) | Induction +/− maintenance | CG0070: stimulation of immune response for Rb-defective tumor cells | Overall CR: 47% (95% CI: 32–62) CR in CIS: 58% (95% CI: 37–78) CR in CIS ± Ta/T1: 50% (95% CI: 33–67) CR CIS + Ta/T1: 33% (95% CI: 10–65) CR in pure Ta/T1: 33% (95% CI: 8–70) 4 pts developed MIBC over 12-month follow-up 3 cancer-specific deaths |
| Navai et al., 2016 [97] | 7 | Single-arm, phase Ib trial | rAd–IFNα-2b/Syn3 (intravesical) | Two administrations +/− second treatment | rAd–IFNa2b/Syn3: cytotoxic effects in selected bladder mucosa cells. | CR in 2 pts (29%). Thereafter, RC in 6 pts (1 was lost to follow-up), and 1 pts died of the disease. |
| Dinney et al., 2013 [98] | 17 | Open-label, dose-escalating, non-randomized, multicentric phase I trial | rAd–IFNα-2b/Syn3 (intravesical) | Single treatment (3 patients received a second dose) | rAd–IFNa2b/Syn3: cytotoxic effects in selected bladder mucosa cells. | CR in 7 patients at 3 months, RFS up to 39.2 months. RC performed in 10 pts. |
| Burke et al., 2012 [99] | 35 | Single-arm, observational, phase I trial | CG0070 (replication selective serotype-5 oncolytic adenovirus) | Induction +/− maintenance | CG0070: stimulation of immune response for Rb-defective tumor cells | Overall RR: 48.6% CR duration: 10.4 months in responders (with some responses ongoing at 17.0 months) CR 50.0% in pts with CIS only CR in pts with CIS alone or CIS + Ta or T1 tumors: 41.2% Higher CR (58.3%) in Rp-positive pts |
| Sidi et al., 2008 [100] | 18 | Single-arm, phase I/II trial | BC-819 DNA plasmid (intravesical) | Induction +/− maintenance | BC-8129 DNA plasmid: inhibition of protein synthesis and induction of cell death. | Overall CR: 22% 35 weeks DFS: 4 pts 49 weeks DFS: 1 pt |
| Author, Year | Patients Number | Study Design | Treatment | Regimen | Effects of Treatment | Main Findings |
|---|---|---|---|---|---|---|
| Kulkarni et al., 2022 [106] | 6 | Single-arm, phase Ib trial | PDT: green light laser (wavelength 520 nm) + intravesical TLD–1433 (ruthenium-based photosensitizer) | Single treatment | Oxygen-reactive species production: cell death induction. | Disease persistence in 3 pts treated with 0.35 mg/cm2 TLD–1433 dose, CR at 3 and 6 months in 2 pts treated with 0.70 mg/cm2 (DFS 18 months), metastatic progression in 1 case |
| Zahoor et al., 2018 [108] | 19 | Single-arm, phase II trial | Sunitinib | 12 weeks therapy | Multi-targeted receptor tyrosine kinase (RTK) inhibition: inhibition of cell growth. | DFS 44% at 12 weeks, 50% progression rate, one recurrence. At 12 months 4 pts remained disease free, overall 78% progression rate, median time to progression 4.7 months |
| Hahn et al., 2017 [109] | 13 | Single-arm, phase II trial | Dovitinib | 4 weeks therapy cycles (median 4 cycles, range 1–19) | Multi-targeted receptor tyrosine kinase (RTK) inhibition: inhibition of cell growth. | CR in 1 case out of 3 pts with FGFR3 mutation (33%), no response in 11 pts (85%), and progression to MIBC in 1 case (8%). DFS in the CR patient 19 months. RC rate 62% (11 pts) |
| Lee et al., 2013 [107] | 34 (BCG refractory + intolerant) | Retrospective | PDT: semiconductor laser (power 3 W, wavelength 662 ± 2 nm) + intravenous Radachlorin | Single treatment | Caspase-3 activation and induction of cell death. | 12, 24, and 30 months DFS 90.9%, 64.4%, and 60.1%, respectively. RC performed in 2 pts |
| Berger et al., 2003 [110] | 31 (10 with previous BCG treatment) | Retrospective | PDT: laser (maximal power 7 W, wavelength of 633 nm—red light) + intravesical 5ALA | 1 to 6 treatment sessions | Mitochondrial cytochrome C translocation and induction of cell apoptosis. | 40% RFS at 11.8 months (range 1–26), 60% recurrence rate after a mean follow-up of 12.7 months. Three patients receiving repeated treatment (3 sessions) were disease free at 6.3 months (range 1–11) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Claps, F.; Pavan, N.; Ongaro, L.; Tierno, D.; Grassi, G.; Trombetta, C.; Tulone, G.; Simonato, A.; Bartoletti, R.; Mertens, L.S.; et al. BCG-Unresponsive Non-Muscle-Invasive Bladder Cancer: Current Treatment Landscape and Novel Emerging Molecular Targets. Int. J. Mol. Sci. 2023, 24, 12596. https://doi.org/10.3390/ijms241612596
Claps F, Pavan N, Ongaro L, Tierno D, Grassi G, Trombetta C, Tulone G, Simonato A, Bartoletti R, Mertens LS, et al. BCG-Unresponsive Non-Muscle-Invasive Bladder Cancer: Current Treatment Landscape and Novel Emerging Molecular Targets. International Journal of Molecular Sciences. 2023; 24(16):12596. https://doi.org/10.3390/ijms241612596
Chicago/Turabian StyleClaps, Francesco, Nicola Pavan, Luca Ongaro, Domenico Tierno, Gabriele Grassi, Carlo Trombetta, Gabriele Tulone, Alchiede Simonato, Riccardo Bartoletti, Laura S. Mertens, and et al. 2023. "BCG-Unresponsive Non-Muscle-Invasive Bladder Cancer: Current Treatment Landscape and Novel Emerging Molecular Targets" International Journal of Molecular Sciences 24, no. 16: 12596. https://doi.org/10.3390/ijms241612596
APA StyleClaps, F., Pavan, N., Ongaro, L., Tierno, D., Grassi, G., Trombetta, C., Tulone, G., Simonato, A., Bartoletti, R., Mertens, L. S., van Rhijn, B. W. G., Mir, M. C., & Scaggiante, B. (2023). BCG-Unresponsive Non-Muscle-Invasive Bladder Cancer: Current Treatment Landscape and Novel Emerging Molecular Targets. International Journal of Molecular Sciences, 24(16), 12596. https://doi.org/10.3390/ijms241612596










