Particle Size Modulates Silver Nanoparticle Toxicity during Embryogenesis of Urchins Arbacia lixula and Paracentrotus lividus
Abstract
1. Introduction
2. Results
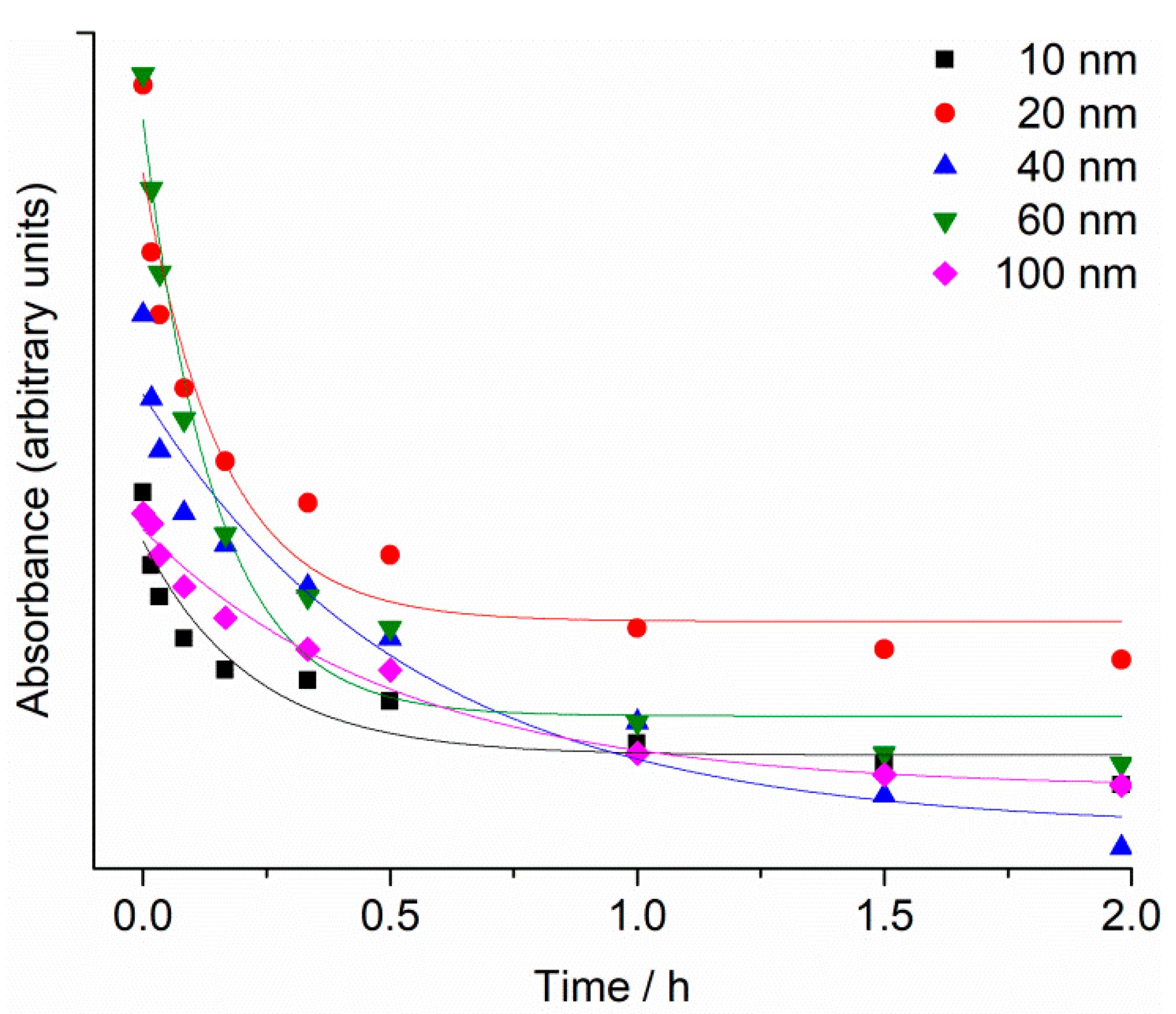
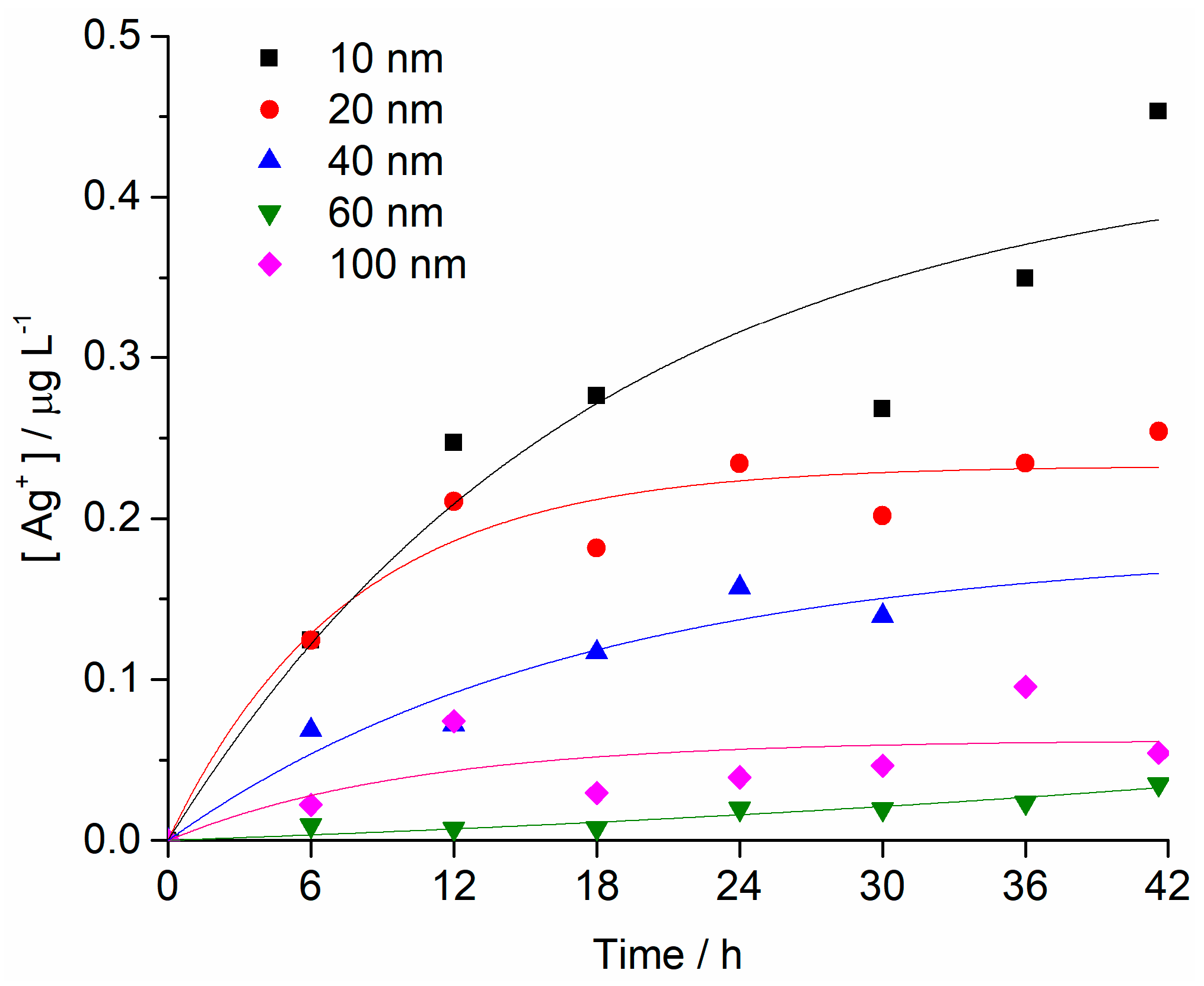
2.1. Nanoparticle Characterisation
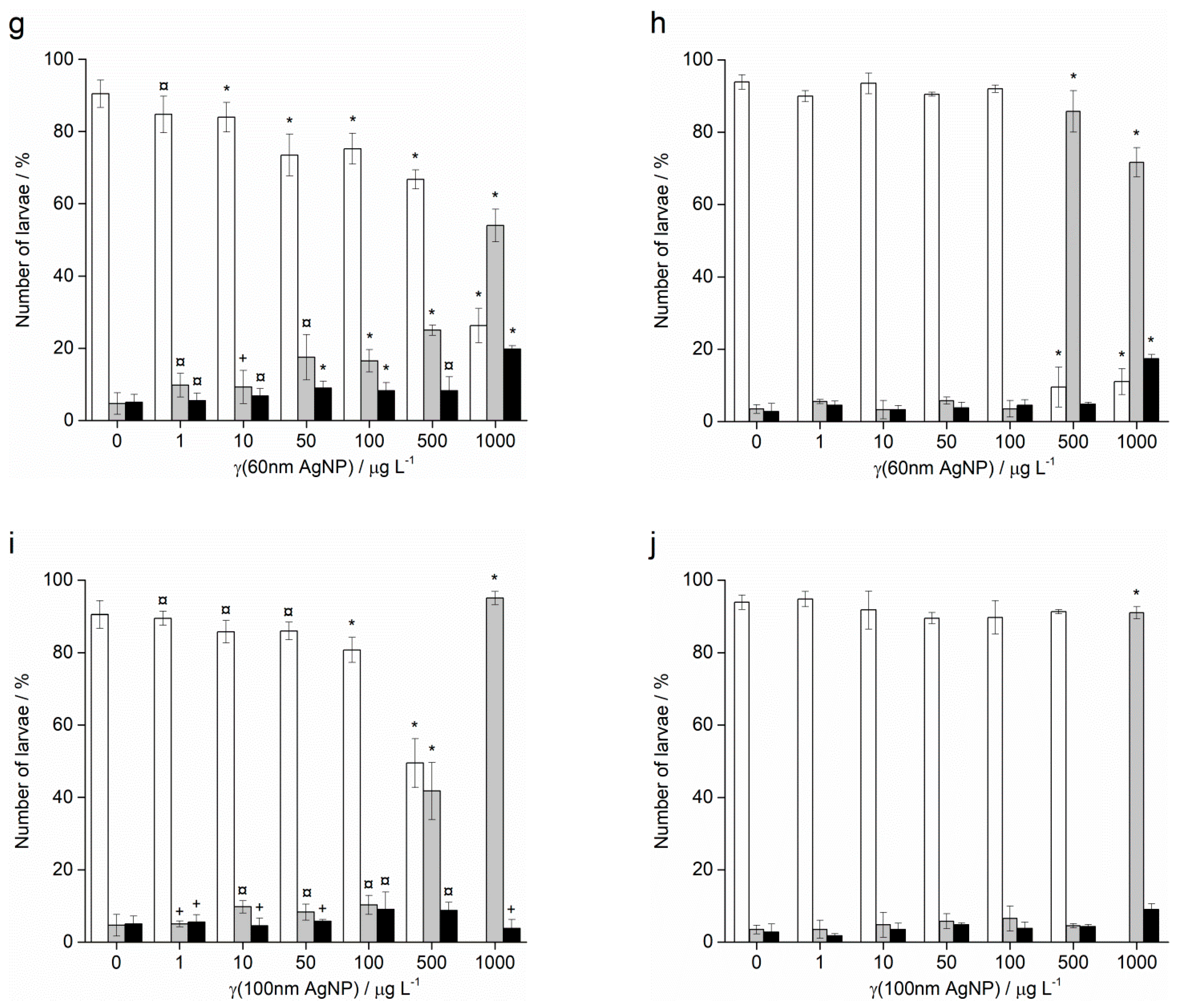
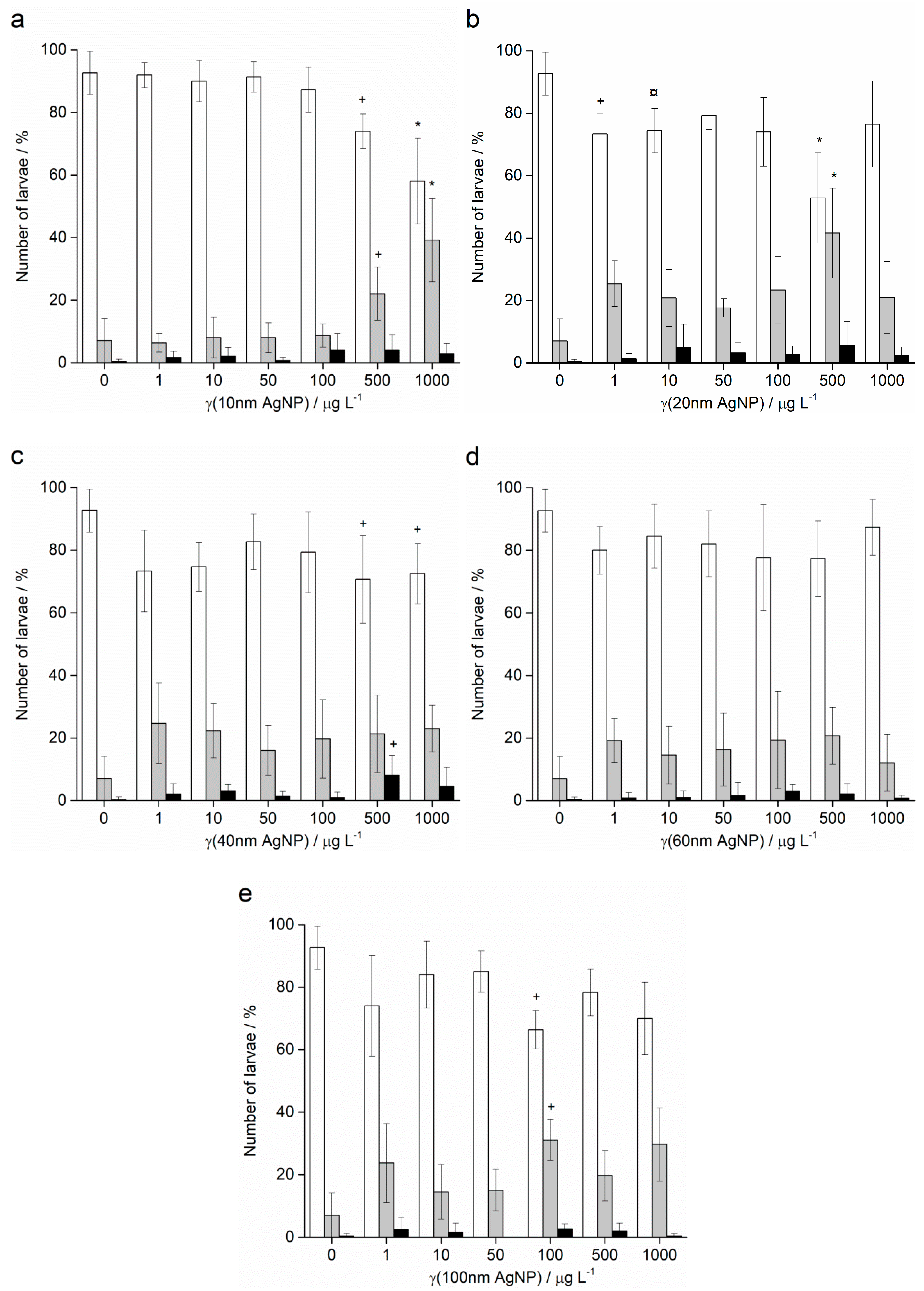
2.2. Embryotoxicity
2.3. Spermiotoxicity
3. Discussion
4. Materials & Methods
4.1. Materials
4.2. Nanoparticle Characterisation
4.3. Embryotoxicity
4.4. Spermiotoxocity
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bruna, T.; Maldonado-Bravo, F.; Jara, P.; Caro, N. Silver nanoparticles and their antibacterial applications. Int. J. Mol. Sci. 2021, 22, 7202. [Google Scholar] [CrossRef] [PubMed]
- Kuskov, A.N.; Kulikov, P.P.; Goryachaya, A.V.; Tzatzarakis, M.N.; Docea, A.O.; Velonia, K.; Shtilman, M.I.; Tsatsakis, A.M. Amphiphilic poly-N-vinylpyrrolidone nanoparticles as carriers for non-steroidal, anti-inflammatory drugs: In vitro cytotoxicity and in vivo acute toxicity study. Nanomedicine 2017, 13, 1021–1030. [Google Scholar] [CrossRef] [PubMed]
- Pinzaru, I.; Coricovac, D.; Dehelean, C.; Moacă, E.-A.; Mioc, M.; Baderca, F.; Sizemore, I.; Brittle, S.; Marti, D.; Calina, C.D.; et al. Stable PEG-coated silver nanoparticles—A comprehensive toxicological profile. Food Chem. Toxicol. 2018, 111, 546–556. [Google Scholar] [CrossRef]
- Chinnapongse, S.L.; MacCuspie, R.I.; Hackley, V.A. Persistence of singly dispersed silver nanoparticles in natural freshwaters, synthetic seawater, and simulated estuarine waters. Sci. Total Environ. 2011, 409, 2443–2450. [Google Scholar] [CrossRef] [PubMed]
- António, D.C.; Cascio, C.; Jakšić, Ž.; Jurašin, D.; Lyons, D.M.; Nogueira, A.J.A.; Rossi, F.; Calzolai, L. Assessing silver nanoparticles behaviour in artificial sea water by mean of AF4 and sp-ICP-MS. Mar. Environ. Res. 2015, 111, 162–169. [Google Scholar] [CrossRef]
- Levak, M.; Burić, P.; Sikirić, M.D.; Jurašin, D.D.; Mikac, N.; Bačić, N.; Drexel, R.; Meier, F.; Jakšić, Ž.; Lyons, D.M. Effect of protein corona on silver nanoparticle stabilization and ion release kinetics in artificial seawater. Environ. Sci. Technol. 2017, 51, 1259–1266. [Google Scholar] [CrossRef]
- Bondarenko, O.; Juganson, K.; Ivask, A.; Kasemets, K.; Mortimer, M.; Kahru, A. Toxicity of Ag, CuO and ZnO nanoparticles to selected environmentally relevant test organisms and mammalian cells in vitro: A critical review. Arch. Toxicol. 2013, 87, 1181–1200. [Google Scholar] [CrossRef]
- Fabrega, J.; Luoma, S.N.; Tyler, C.R.; Galloway, T.S.; Lead, J.R. Silver nanoparticles: Behaviour and effects in the aquatic environment. Environ. Int. 2011, 37, 517–531. [Google Scholar] [CrossRef]
- Lapresta-Fernández, A.; Fernández, A.; Blasco, J. Nanoecotoxicity effects of engineered silver and gold nanoparticles in aquatic organisms. TrAC Trend. Anal. Chem. 2012, 32, 40–59. [Google Scholar] [CrossRef]
- Schultz, A.G.; Boyle, D.; Chamot, D.; Ong, K.J.; Wilkinson, K.J.; McGeer, J.C.; Sunahara, G.; Goss, G.G. Aquatic toxicity of manufactured nanomaterials: Challenges and recommendations for future toxicity testing. Environ. Chem. 2014, 11, 207–226. [Google Scholar] [CrossRef]
- Matranga, V.; Corsi, I. Toxic effects of engineered nanoparticles in the marine environment: Model organisms and molecular approaches. Mar. Environ. Res. 2012, 76, 32–40. [Google Scholar] [CrossRef]
- Gomes, T.; Pereira, C.G.; Cardoso, C.; Sousa, V.S.; Teixeira, M.R.; Pinheiro, J.P.; Bebianno, M.J. Effects of silver nanoparticles exposure in the mussel Mytilus galloprovincialis. Mar. Environ. Res. 2014, 101, 208–214. [Google Scholar] [CrossRef]
- Ringwood, A.H.; McCarthy, M.; Bates, T.C.; Carroll, D.L. The effects of silver nanoparticles on oyster embryos. Mar. Environ. Res. 2010, 69, S49–S51. [Google Scholar] [CrossRef]
- Gambardella, C.; Gallus, L.; Gatti, A.M.; Faimali, M.; Carbone, S.; Antisari, L.V.; Falugi, C.; Ferrando, S. Toxicity and transfer of metal oxide nanoparticles from microalgae to sea urchin larvae. Chem. Ecol. 2014, 30, 308–316. [Google Scholar] [CrossRef]
- Schiavo, S.; Duroudier, N.; Bilbao, E.; Mikolaczyk, M.; Schäfer, J.; Cajaraville, M.P.; Manzo, S. Effects of PVP/PEI coated and uncoated silver NPs and PVP/PEI coating agent on three species of marine microalgae. Sci. Total Environ. 2017, 577, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Gambardella, C.; Ferrando, S.; Morgana, S.; Gallus, L.; Ramoino, P.; Ravera, S.; Bramini, M.; Diaspro, A.; Faimali, M.; Falugi, C. Exposure of Paracentrotus lividus male gametes to engineered nanoparticles affects skeletal bio-mineralization processes and larval plasticity. Aquat. Toxicol. 2015, 158, 181–191. [Google Scholar] [CrossRef]
- Gambardella, C.; Marcellini, F.; Falugi, C.; Varrella, S.; Corinaldesi, C. Early-stage anomalies in the sea urchin (Paracentrotus lividus) as bioindicators of multiple stressors in the marine environment: Overview and future perspectives. Environ. Pollut. 2021, 287, 117608. [Google Scholar] [CrossRef] [PubMed]
- Carballeira, C.; Ramos-Gómez, J.; Martín-Díaz, L.; DelValls, T.A. Identification of specific malformations of sea urchin larvae for toxicity assessment: Application to marine pisciculture effluents. Mar. Environ. Res. 2012, 77, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Burić, P.; Jakšić, Ž.; Štajner, L.; Sikirić, M.D.; Jurašin, D.; Cascio, C.; Calzolai, L.; Lyons, D.M. Effect of silver nanoparticles on Mediterranean sea urchin embryonal development is species specific and depends on moment of first exposure. Mar. Environ. Res. 2015, 111, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Falugi, C.; Aluigi, M.G.; Chiantore, M.C.; Privitera, D.; Ramoino, P.; Gatti, M.A.; Fabrizi, A.; Pinsino, A.; Matranga, V. Toxicity of metal oxide nanoparticles in immune cells of the sea urchin. Mar. Environ. Res. 2012, 76, 114–121. [Google Scholar] [CrossRef]
- Torre, C.D.; Bergami, E.; Salvati, A.; Faleri, C.; Dawson, K.A.; Corsi, I. Accumulation and embryotoxicity of polystyrene nanoparticles at early stage of development of sea urchin embryos Paracentrotus lividus. Environ. Sci. Technol. 2014, 48, 12302–12311. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Torres-Duarte, C.; Cole, B.J.; Cherr, G.N. Copper oxide and zinc oxide nanomaterials act as inhibitors of multidrug resistance transport in sea urchin embryos: Their role as chemosensitizers. Environ. Sci. Technol. 2015, 49, 5760–5770. [Google Scholar] [CrossRef] [PubMed]
- Genevière, A.-M.; Derelle, E.; Escande, M.-L.; Grimsley, N.; Klopp, C.; Ménager, C.; Michel, A.; Moreau, H. Responses to iron oxide and zinc oxide nanoparticles in echinoderm embryos and microalgae: Uptake, growth, morphology, and transcriptomic analysis. Nanotoxicology 2020, 14, 1342–1361. [Google Scholar] [CrossRef]
- Šiller, L.; Lemloh, M.L.; Piticharoenphun, S.; Mendis, B.G.; Horrocks, B.R.; Brümmer, F.; Medaković, D. Silver nanoparticle toxicity in sea urchin Paracentrotus lividus. Environ. Pollut. 2013, 178, 498–502. [Google Scholar] [CrossRef] [PubMed]
- Urić, P.; Pfannkuchen, M.; Jakšić, Ž.; Stipić, F.; Lyons, D.M. Uptake and impact of engineered nanoparticles on embryonal development and stress response in selected marine organisms. Arh. Hig. Rada Toksikol. 2012, 63, 21. [Google Scholar]
- Manno, D.; Serra, A.; Buccolieri, A.; Panzarini, E.; Carata, E.; Tenuzzo, B.; Izzo, D.; Vergallo, C.; Rossi, M.; Dini, L. Silver and carbon nanoparticles toxicity in sea urchin Paracentrotus lividus embryos. BioNanoMaterials 2013, 14, 229–236. [Google Scholar] [CrossRef]
- Magesky, A.; De Olivieira Riberio, C.A.; Beaulieu, L.; Pelletier, E. Silver nanoparticles and dissolved silver activate contrasting immune responses and stress-induced Hsp expression in sea urchin. Environ. Toxicol. Chem. 2017, 36, 1872–1886. [Google Scholar] [CrossRef]
- Mesarič, T.; Sepčić, K.; Drobne, D.; Makoved, D.; Faimali, M.; Morgana, S.; Falugi, C.; Gambardella, C. Sperm exposure to carbon-based nanomaterials causes abnormalities in early development of purple sea urchin (Paracentrotus lividus). Aquat. Toxicol. 2015, 163, 158–166. [Google Scholar] [CrossRef]
- Pikula, K.; Mintcheva, N.; Kulinich, S.A.; Zakharenko, A.; Markina, Z.; Chaika, V.; Orlova, T.; Mezhuev, Y.; Kokkinakis, E.; Tsatsakis, A.; et al. Aquatic toxicity and mode of action of CdS and ZnS nanoparticles in four microalgae species. Environ. Res. 2020, 186, 109513. [Google Scholar] [CrossRef]
- Tajima, K.; da Silva, J.R.M.C.; Lawrence, J.M. Chapter 9—Disease in sea urchins. Dev. Aquacult. Fish. Sci. 2007, 37, 167–182. [Google Scholar]
- Jevanandam, J.; Barhoum, A.; Chan, Y.S.; Dufresne, A.; Danquah, M.K. Review on nanoparticles and nanostructured materials: History, sources, toxicity and regulations. Beilstein J. Nanotechnol. 2018, 9, 1050–1074. [Google Scholar] [CrossRef]
- Gilles, K.W.; Pearse, J.S. Disease in sea urchins Strongylocentrotus purpuratus: Experimental infection and bacterial virulence. Dis. Aquat. Organ. 1985, 1, 105–114. [Google Scholar] [CrossRef]
- Ivask, A.; Kurvet, I.; Kasemets, K.; Blinova, I.; Aruoja, V.; Suppi, S.; Vija, H.; Käkinen, A.; Titma, T.; Heinlaan, M.; et al. Size-dependent toxicity of silver nanoparticles to bacteria, yeast, algae, crustaceans and mammalian cells In Vitro. PLoS ONE 2014, 9, e102108. [Google Scholar] [CrossRef] [PubMed]
- Farkas, J.; Christianc, P.; Gallego-Urread, J.A.; Roose, N.; Hassellövd, M.; Tollefsena, K.E.; Thomasa, K.V. Uptake and effects of manufactured silver nanoparticles in rainbow trout (Oncorhynchus mykiss) gill cells. Aquat. Toxicol. 2011, 101, 117–125. [Google Scholar] [CrossRef]
- Bouallegui, Y.; Ben Younes, R.; Turki, F.; Mezni, A.; Oueslati, R. Effect of exposure time, particle size and uptake pathways in immune cell lysosomal cytotoxicity of mussels exposed to silver nanoparticles. Drug. Chem. Toxicol. 2017, 6, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, K.; Suematsu, H.; Kiyomiya, E.; Aoki, M.; Sato, M.; Moritoki, N. Size-dependent toxicity of silica nano-particles to Chlorella kessleri. J. Environ. Sci. Health. 2008, 43, 1167–1173. [Google Scholar] [CrossRef]
- Passagne, I.; Morille, M.; Rousset, M.; Pujalté, I.; L’azou, B. Implication of oxidative stress in size-dependent toxicity of silica nanoparticles in kidney cells. Toxicology 2012, 229, 112–124. [Google Scholar] [CrossRef]
- Pinsino, A.; Russo, R.; Bonaventura, R.; Brunelli, A.; Marcomini, A.; Matranga, V. Titanium dioxide nanoparticles stimulate sea urchin immune cell phagocytic activity involving TLR/p38 MAPK-mediated signalling pathway. Sci. Rep. 2015, 5, 14492. [Google Scholar] [CrossRef]
- Choi, S.; Kim, S.; Bae, Y.J.; Park, J.W.; Jung, J. Size-dependent toxicity of silver nanoparticles to Glyptotendipes tokunagai. Environ. Health Toxicol. 2015, 30, e2015003. [Google Scholar] [CrossRef]
- Jimeno-Romero, A.; Izagirre, U.; Gilliland, D.; Warley, A.; Cajaraville, M.P.; Marigómez, I.; Soto, M. Lysosomal responses to different gold forms (nanoparticles, aqueous, bulk) in mussel digestive cells: A trade-off between the toxicity of the capping agent and form, size and exposure concentration. Nanotoxicology 2017, 11, 658–670. [Google Scholar] [CrossRef]
- Ward, J.E.; Kach, D.J. Marine aggregates facilitate ingestion of nanoparticles by suspension-feeding bivalves. Mar. Environ. Res. 2009, 68, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Bruneau, A.; Turcotte, P.; Pilote, M.; Gagné, F.; Gagnon, C. Fate of silver nanoparticles in wastewater and immunotoxic effects on rainbow trout. Aquat. Toxicol. 2016, 174, 70–81. [Google Scholar] [CrossRef]
- Čarapar, I.; Jurković, L.; Pavičić-Hamer, D.; Hamer, B.; Lyons, D.M. Simultaneous influence of gradients in natural organic matter and abiotic parameters on the behavior of silver nanoparticles in the transition zone from freshwater to saltwater environments. Nanomaterials 2022, 12, 296. [Google Scholar] [CrossRef] [PubMed]
- Aerle, R.V.; Lange, A.; Moorhouse, A.; Paszkiewicz, K.; Ball, K.; Johnston, B.D.; De-Bastos, E.; Booth, T.; Tyler, C.R.; Santos, E.M. Molecular mechanisms of toxicity of silver nanoparticles in zebrafish embryos. Environ. Sci. Technol. 2013, 47, 8005–8014. [Google Scholar] [CrossRef]
- Magesky, A.; Pelletier, E. Toxicity of ionic silver and polymer-coated silver nanoparticles with interactions of functionalized carbon nanotubes on early development stages of sea urchin. Aquat. Toxicol. 2015, 167, 106–123. [Google Scholar] [CrossRef]
- Magesky, A.; Ribeiro, C.A.O.; Pelletier, É. Physiological effects and cellular responses of metamorphic larvae and juveniles of sea urchin exposed to ionic and nanoparticulate silver. Aquat. Toxicol. 2016, 174, 208–227. [Google Scholar] [CrossRef] [PubMed]
- George, S.; Lin, S.; Ji, Z.; Thomas, C.R.; Li, L.; Mecklenburg, M.; Meng, H.; Wang, X.; Zhang, H.; Xia, T.; et al. Surface defects on plate-shaped silver nanoparticles contribute to its hazard potential in a fish cell line and zebrafish embryos. ACS Nano 2012, 6, 3745–3759. [Google Scholar] [CrossRef] [PubMed]
- Yoisungnern, T.; Choi, Y.-J.; Han, J.W.; Kang, M.-H.; Das, J.; Gurunathan, S.; Kwon, D.-N.; Cho, S.-G.; Park, C.; Chang, W.K.; et al. Internalization of silver nanoparticles into mouse spermatozoa results in poor fertilization and compromised embryo development. Sci. Rep. 2015, 5, 11170. [Google Scholar] [CrossRef]
- Martino, C.; Bonaventura, R.; Byrne, M.; Roccheri, M.; Matranga, V. Effects of exposure to gadolinium on the development of geographically and phylogenetically distant sea urchins species. Mar. Environ. Res. 2017, 128, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Pucelik, B.; Sułek, A.; Borkowski, M.; Barzowska, A.; Kobielusz, M.; Dąbrowski, J.M. Synthesis and characterization of size- and charge-tunable silver nanoparticles for selective anticancer and antibacterial treatment. ACS Appl. Mater. Interfaces 2022, 14, 14981–14996. [Google Scholar] [CrossRef]
- Camacho-Jiménez, L.; Álvarez-Sánchez, A.R.; Mejía-Ruíz, C.H. Silver nanoparticles (AgNPs) as antimicrobials in marine shrimp farming: A review. Aquac. Rep. 2020, 18, 100512. [Google Scholar] [CrossRef]
- Marcuello, C.; Chambel, L.; Rodrigues, M.S.; Ferreira, L.P.; Cruz, M.M. Magnetotactic bacteria: Magnetism beyond magnetosomes. IEEE Trans. Nanobioscience 2018, 17, 555–559. [Google Scholar] [CrossRef] [PubMed]
- Pagano, G.; Esposito, A.; Bove, P.; De Angelis, M.; Rota, A.; Giordano, G.G. The effects of hexavalent and trivalent chromium on fertilization and development in sea urchins. Environ. Res. 1983, 30, 442–452. [Google Scholar] [CrossRef] [PubMed]
- Trifuoggi, M.; Pagano, G.; Guida, M.; Palumbo, A.; Siciliano, A.; Gravina, M.; Lyons, D.M.; Burić, P.; Levak, M.; Thomas, P.J.; et al. Comparative toxicity of seven rare earth elements in sea urchin early life stages. Environ. Sci. Pollut. Res. 2017, 24, 20803–20810. [Google Scholar] [CrossRef]







| Diameter/nm | EC50/μg L−1 | R2 | LC50/μg L−1 | R2 | |
|---|---|---|---|---|---|
| A. lixula | 10 | 48.59 | 0.99 | 387.34 ± 10.13 | 0.98 |
| 20 | 74.90 ± 2.90 | 0.99 | 397.67 ± 2.00 | 0.99 | |
| 40 | 485.57 | 0.99 | 903.08 ± 17.61 | 0.99 | |
| 60 | 846.77 ± 67.63 | 0.98 | |||
| 100 | 529.11 ± 21.54 | 0.99 | 1433.97 | 0.99 | |
| Ag+ | 39.50 ± 2.33 | 0.99 | 87.95 | 0.99 | |
| P. lividus | 10 | 66.87 ± 0.95 | 0.99 | 327.25 | 0.99 |
| 20 | 91.25 ± 10.43 | 0.97 | 376.34 ± 10.00 | 0.99 | |
| 40 | 322.45 | 0.99 | 724.06 ± 25.48 | 0.98 | |
| 60 | 360.88 ± 189.72 | 0.99 | |||
| 100 | 662.26 | 0.99 | |||
| Ag+ | 29.29 ± 0.80 | 0.99 | 73.79 ± 1.26 | 0.99 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burić, P.; Čarapar, I.; Pavičić-Hamer, D.; Kovačić, I.; Jurković, L.; Dutour Sikirić, M.; Domazet Jurašin, D.; Mikac, N.; Bačić, N.; Lyons, D.M. Particle Size Modulates Silver Nanoparticle Toxicity during Embryogenesis of Urchins Arbacia lixula and Paracentrotus lividus. Int. J. Mol. Sci. 2023, 24, 745. https://doi.org/10.3390/ijms24010745
Burić P, Čarapar I, Pavičić-Hamer D, Kovačić I, Jurković L, Dutour Sikirić M, Domazet Jurašin D, Mikac N, Bačić N, Lyons DM. Particle Size Modulates Silver Nanoparticle Toxicity during Embryogenesis of Urchins Arbacia lixula and Paracentrotus lividus. International Journal of Molecular Sciences. 2023; 24(1):745. https://doi.org/10.3390/ijms24010745
Chicago/Turabian StyleBurić, Petra, Ivana Čarapar, Dijana Pavičić-Hamer, Ines Kovačić, Lara Jurković, Maja Dutour Sikirić, Darija Domazet Jurašin, Nevenka Mikac, Niko Bačić, and Daniel Mark Lyons. 2023. "Particle Size Modulates Silver Nanoparticle Toxicity during Embryogenesis of Urchins Arbacia lixula and Paracentrotus lividus" International Journal of Molecular Sciences 24, no. 1: 745. https://doi.org/10.3390/ijms24010745
APA StyleBurić, P., Čarapar, I., Pavičić-Hamer, D., Kovačić, I., Jurković, L., Dutour Sikirić, M., Domazet Jurašin, D., Mikac, N., Bačić, N., & Lyons, D. M. (2023). Particle Size Modulates Silver Nanoparticle Toxicity during Embryogenesis of Urchins Arbacia lixula and Paracentrotus lividus. International Journal of Molecular Sciences, 24(1), 745. https://doi.org/10.3390/ijms24010745












