Genome-Wide Characterization of High-Affinity Nitrate Transporter 2 (NRT2) Gene Family in Brassica napus
Abstract
1. Introduction
2. Results
2.1. Identification of NRT2 Genes in B. napus
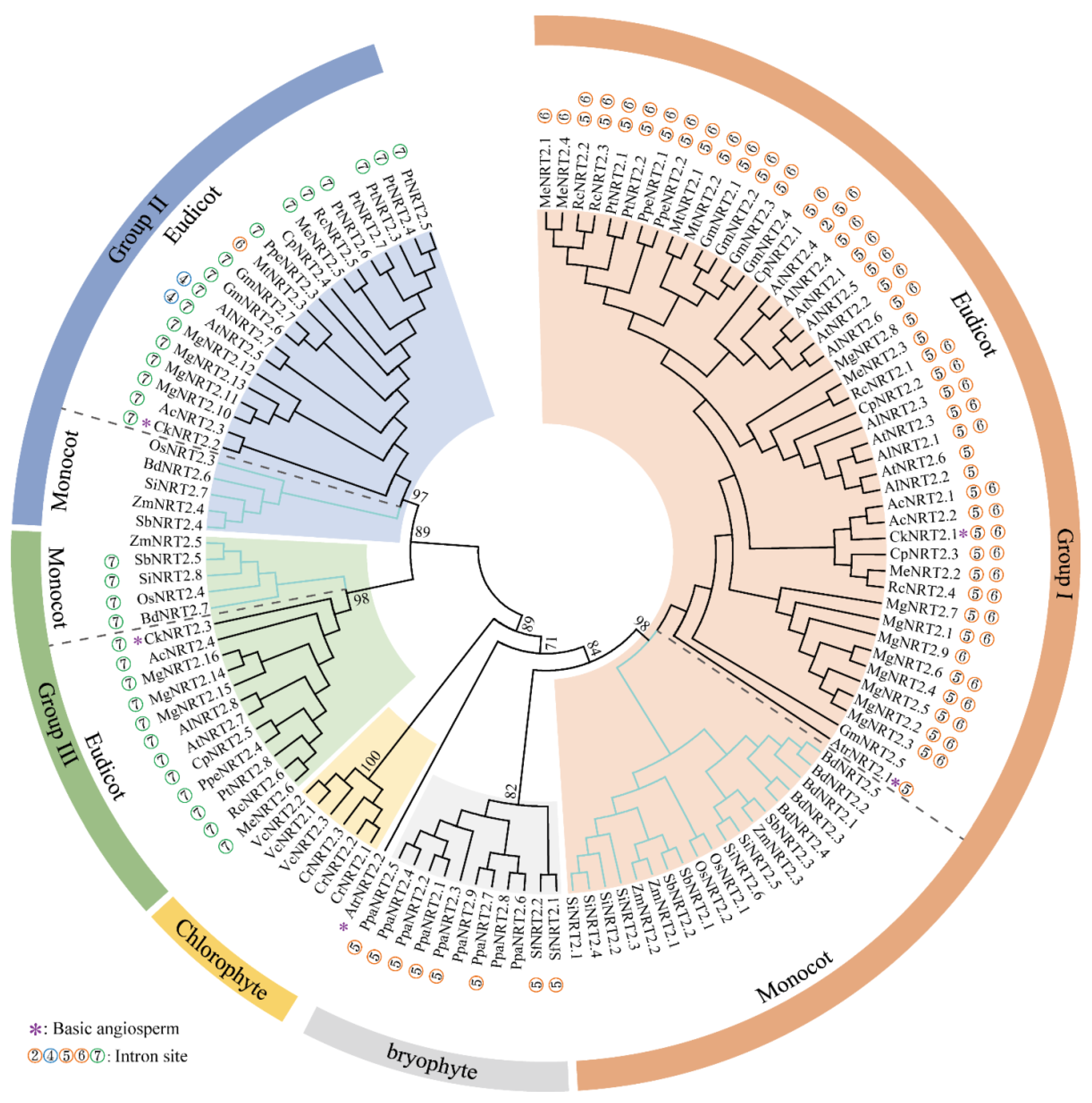
2.2. Phylogenetic and Sequence Structure Analysis of B. napus NRT2 Gene Family
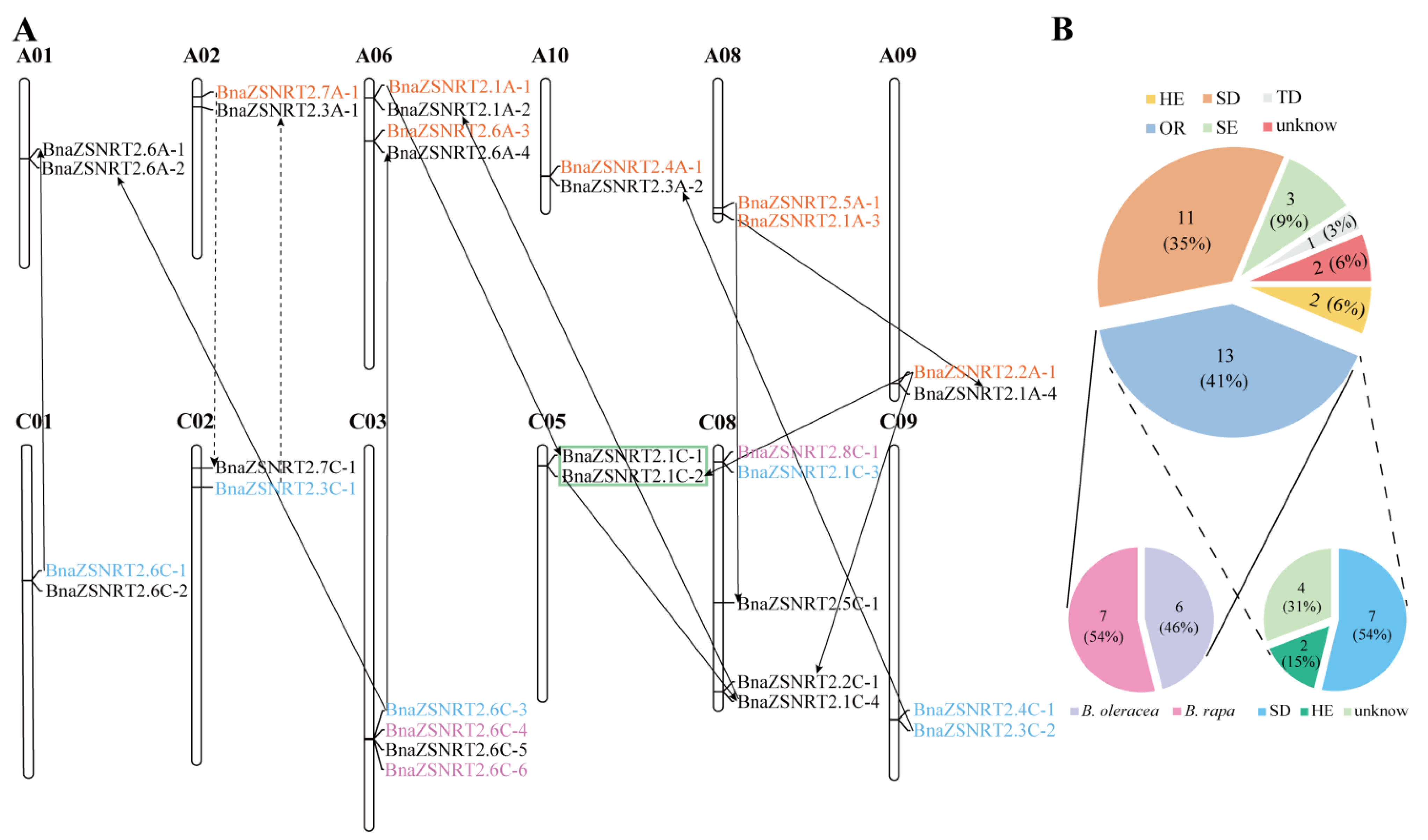
2.3. Chromosomal Location and Collinearity Relationship in BnaZSNRT2s
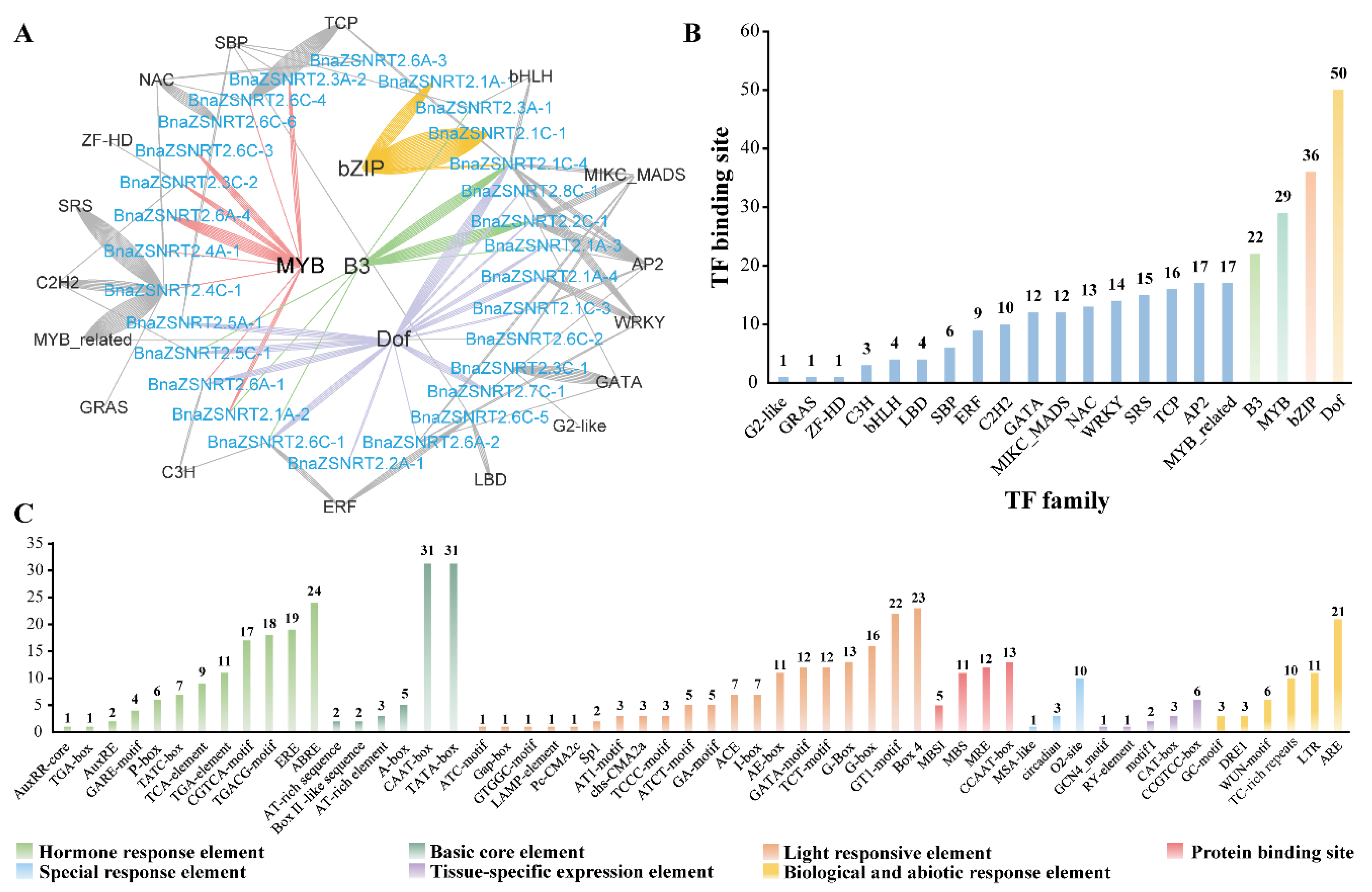
2.4. Potential Regulatory Mechanism in the Promoter Regions of BnaZSNRT2s
2.5. Potential miRNAs Targets of BnaZSNRT2s
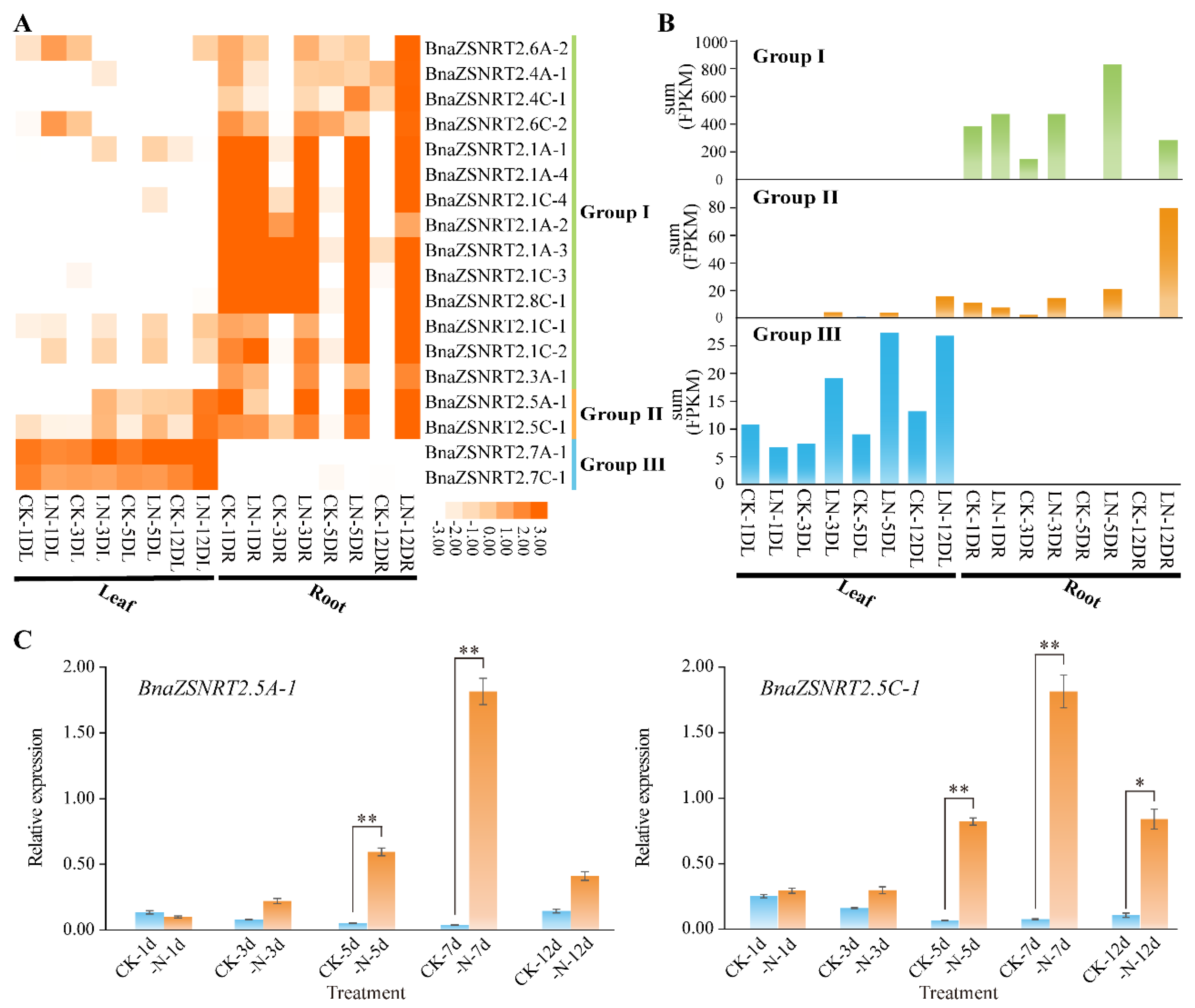
2.6. Spatial-Temporal Expressions of BnaZSNRT2s in Different Developmental Stages of B. napus
2.7. Expression Profile of BnaZSNRT2s under LN Stress
3. Discussion
3.1. A Conserved Patterns of NRT2s Intron Insertion Patterns and Phylogeny
3.2. Expression and Function Characteristics of NRT2s in Plants
4. Materials and Methods
4.1. Identification of NRT2 Genes in Plants
4.2. Phylogenetic and Sequence Structure Analysis of B. napus NRT2 Gene Family
4.3. Chromosomal Location and Collinearity Relationship of BnaZSNRT2s
4.4. The Potential Regulatory Mechanism in the Promoter Regions of BnaZSNRT2s
4.5. Spatial-Temporal and LN Stress Expression Profile Analysis of BnaZSNRT2s by RNA-Seq Data
4.6. RT-qPCR Analysis of BnaZSNRT2s under LN Conditions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Evans, J.R.; Clarke, V.C. The nitrogen cost of photosynthesis. J. Exp. Bot. 2019, 70, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Frink, C.R.; Waggoner, P.E.; Ausubel, J.H. Nitrogen fertilizer: Retrospect and prospect. Proc. Natl. Acad. Sci. USA 1999, 96, 1175–1180. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.Q.; Shen, R.F. Aluminum-Nitrogen Interactions in the Soil-Plant System. Front. Plant. Sci. 2018, 9, 807. [Google Scholar] [CrossRef] [PubMed]
- Vitousek, P.M.; Howarth, R.W. Nitrogen limitation on land and in the sea: How can it occur? Biogeochemistry 1991, 13, 87–115. [Google Scholar] [CrossRef]
- Alvarez, J.M.; Vidal, E.A.; Gutierrez, R.A. Integration of local and systemic signaling pathways for plant N responses. Curr. Opin. Plant Biol. 2012, 15, 185–191. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Cheng, Y.H.; Chen, K.E.; Tsay, Y.F. Nitrate Transport, Signaling, and Use Efficiency. Annu. Rev. Plant. Biol. 2018, 69, 85–122. [Google Scholar] [CrossRef]
- Robertson, G.P.; Vitousek, P.M. Nitrogen in Agriculture: Balancing the Cost of an Essential Resource. Annu. Rev. Environ. Resour. 2009, 34, 97–125. [Google Scholar] [CrossRef]
- Gutiérrez, R.A. Systems biology for enhanced plant nitrogen nutrition. Science 2012, 336, 1673–1675. [Google Scholar] [CrossRef]
- Fixen, P.E.; West, F.B. Nitrogen fertilizers: Meeting contemporary challenges. Ambio 2002, 31, 169–176. [Google Scholar] [CrossRef]
- Ju, X.T.; Xing, G.X.; Chen, X.P.; Zhang, S.L.; Zhang, L.J.; Liu, X.J.; Cui, Z.L.; Yin, B.; Christie, P.; Zhu, Z.L.; et al. Reducing environmental risk by improving N management in intensive Chinese agricultural systems. Proc. Natl. Acad. Sci. USA 2009, 106, 3041–3046. [Google Scholar] [CrossRef]
- Khan, F.A.; Ansari, A.A. Eutrophication: An Ecological Vision. Bot. Rev. 2005, 71, 449–482. [Google Scholar] [CrossRef]
- Yang, X.E.; Wu, X.; Hao, H.L.; He, Z.L. Mechanisms and assessment of water eutrophication. J. Zhejiang Univ. Sci. B 2008, 9, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Sieling, K.; Kage, H. Apparent fertilizer N recovery and the relationship between grain yield and grain protein concentration of different winter wheat varieties in a long-term field trial. Eur. J. Agron. 2021, 124, 126246. [Google Scholar] [CrossRef]
- Dimkpa, C.O.; Fugice, J.; Singh, U.; Lewis, T.D. Development of fertilizers for enhanced nitrogen use efficiency—Trends and perspectives. Sci. Total Environ. 2020, 731, 139113. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Diamond, J. China’s environment in a globalizing world. Nature 2005, 435, 1179–1186. [Google Scholar] [CrossRef] [PubMed]
- Yan, N. Structural Biology of the Major Facilitator Superfamily Transporters. Annu. Rev. Biophys. 2015, 44, 257–283. [Google Scholar] [CrossRef] [PubMed]
- Galvan, A.; Fernández, E. Eukaryotic nitrate and nitrite transporters. Cell. Mol. Life Sci. 2001, 58, 225–233. [Google Scholar] [CrossRef]
- Tong, J.; Walk, T.C.; Han, P.; Chen, L.; Shen, X.; Li, Y.; Gu, C.; Xie, L.; Hu, X.; Liao, X.; et al. Genome-wide identification and analysis of high-affinity nitrate transporter 2 (NRT2) family genes in rapeseed (Brassica napus L.) and their responses to various stresses. BMC Plant Biol. 2020, 20, 464. [Google Scholar] [CrossRef]
- Castro-Rodríguez, V.; Cañas, R.A.; de la Torre, F.N.; Pascual, M.B.; Avila, C.; Cánovas, F.M. Molecular fundamentals of nitrogen uptake and transport in trees. J. Exp. Bot. 2017, 68, 2489–2500. [Google Scholar] [CrossRef]
- Guan, M.; Chen, M.; Cao, Z. NRT2.1, a major contributor to cadmium uptake controlled by high-affinity nitrate transporters. Ecotoxicol. Environ. Saf. 2021, 218, 112269. [Google Scholar] [CrossRef]
- Kiba, T.; Feria-Bourrellier, A.B.; Lafouge, F.; Lezhneva, L.; Boutet-Mercey, S.; Orsel, M.; Brehaut, V.; Miller, A.; Daniel-Vedele, F.; Sakakibara, H.; et al. The Arabidopsis nitrate transporter NRT2.4 plays a double role in roots and shoots of nitrogen-starved plants. Plant Cell 2012, 24, 245–258. [Google Scholar] [CrossRef] [PubMed]
- Dechorgnat, J.; Patrit, O.; Krapp, A.; Fagard, M.; Daniel-Vedele, F. Characterization of the Nrt2.6 gene in Arabidopsis thaliana: A link with plant response to biotic and abiotic stress. PLoS ONE 2012, 7, e42491. [Google Scholar] [CrossRef] [PubMed]
- Lezhneva, L.; Kiba, T.; Feria-Bourrellier, A.B.; Lafouge, F.; Boutet-Mercey, S.; Zoufan, P.; Sakakibara, H.; Daniel-Vedele, F.; Krapp, A. The Arabidopsis nitrate transporter NRT2.5 plays a role in nitrate acquisition and remobilization in nitrogen-starved plants. Plant J. 2014, 80, 230–241. [Google Scholar] [CrossRef] [PubMed]
- Chopin, F.; Orsel, M.; Dorbe, M.F.; Chardon, F.; Truong, H.N.; Miller, A.J.; Krapp, A.; Daniel-Vedele, F. The Arabidopsis ATNRT2.7 nitrate transporter controls nitrate content in seeds. Plant Cell 2007, 19, 1590–1602. [Google Scholar] [CrossRef] [PubMed]
- Kotur, Z.; Mackenzie, N.; Ramesh, S.; Tyerman, S.D.; Kaiser, B.N.; Glass, A.D.M. Nitrate transport capacity of the Arabidopsis thaliana NRT2 family members and their interactions with AtNAR2.1. New Phytol. 2012, 194, 724–731. [Google Scholar] [CrossRef]
- Chen, J.; Liu, X.; Liu, S.; Fan, X.; Zhao, L.; Song, M.; Fan, X.; Xu, G. Co-Overexpression of OsNAR2.1 and OsNRT2.3a Increased Agronomic Nitrogen Use Efficiency in Transgenic Rice Plants. Front. Plant Sci. 2020, 11, 1245. [Google Scholar] [CrossRef]
- Liu, R.; Jia, T.; Cui, B.; Song, J. The expression patterns and putative function of nitrate transporter 2.5 in plants. Plant Signal. Behav. 2020, 15, 1815980. [Google Scholar] [CrossRef]
- Lupini, A.; Mercati, F.; Araniti, F.; Miller, A.J.; Sunseri, F.; Abenavoli, M.R. NAR2.1/NRT2.1 functional interaction with NO3(-) and H(+) fluxes in high-affinity nitrate transport in maize root regions. Plant Physiol. Biochem. 2016, 102, 107–114. [Google Scholar] [CrossRef]
- Song, J.M.; Guan, Z.; Hu, J.; Guo, C.; Yang, Z.; Wang, S.; Liu, D.; Wang, B.; Lu, S.; Zhou, R.; et al. Eight high-quality genomes reveal pan-genome architecture and ecotype differentiation of Brassica napus. Nat. Plants 2020, 6, 34–45. [Google Scholar] [CrossRef]
- Chalhoub, B.; Denoeud, F.; Liu, S.; Parkin, I.A.; Tang, H.; Wang, X.; Chiquet, J.; Belcram, H.; Tong, C.; Samans, B.; et al. Plant genetics. Early allopolyploid evolution in the post-Neolithic Brassica napus oilseed genome. Science 2014, 345, 950–953. [Google Scholar] [CrossRef]
- Stahl, A.; Vollrath, P.; Samans, B.; Frisch, M.; Wittkop, B.; Snowdon, R.J. Effect of breeding on nitrogen use efficiency-associated traits in oilseed rape. J. Exp. Bot. 2019, 70, 1969–1986. [Google Scholar] [CrossRef] [PubMed]
- Bouchet, A.S.; Laperche, A.; Bissuel-Belaygue, C.; Baron, C.; Morice, J.; Rousseau-Gueutin, M.; Dheu, J.E.; George, P.; Pinochet, X.; Foubert, T.; et al. Genetic basis of nitrogen use efficiency and yield stability across environments in winter rapeseed. BMC Genet. 2016, 17, 131. [Google Scholar] [CrossRef] [PubMed]
- Sorin, C.; Leport, L.; Cambert, M.; Bouchereau, A.; Mariette, F.; Musse, M. Nitrogen deficiency impacts on leaf cell and tissue structure with consequences for senescence associated processes in Brassica napus. Bot. Stud. 2016, 57, 11. [Google Scholar] [CrossRef] [PubMed]
- Avice, J.C.; Etienne, P. Leaf senescence and nitrogen remobilization efficiency in oilseed rape (Brassica napus L.). J. Exp. Bot. 2014, 65, 3813–3824. [Google Scholar] [CrossRef]
- Orsel, M.; Krapp, A.; Daniel-Vedele, F. Analysis of the NRT2 nitrate transporter family in Arabidopsis. Structure and gene expression. Plant Physiol. 2002, 129, 886–896. [Google Scholar] [CrossRef]
- Bai, H.; Euring, D.; Volmer, K.; Janz, D.; Polle, A. The nitrate transporter (NRT) gene family in poplar. PLoS ONE 2013, 8, e72126. [Google Scholar] [CrossRef]
- Guo, B.; Li, Y.; Wang, S.; Li, D.; Lv, C.; Xu, R. Characterization of the Nitrate Transporter gene family and functional identification of HvNRT2.1 in barley (Hordeum vulgare L.). PLoS ONE 2020, 15, e0232056. [Google Scholar] [CrossRef]
- Cheng, F.; Wu, J.; Wang, X. Genome triplication drove the diversification of Brassica plants. Hortic. Res. 2014, 1, 14024. [Google Scholar] [CrossRef]
- Wen, J.; Guo, P.; Ke, Y.; Liu, M.; Li, P.; Wu, Y.; Ran, F.; Wang, M.; Li, J.; Du, H. The auxin response factor gene family in allopolyploid Brassica napus. PLoS ONE 2019, 14, e0214885. [Google Scholar] [CrossRef]
- Du, H.; Liang, Z.; Zhao, S.; Nan, M.G.; Tran, L.S.; Lu, K.; Huang, Y.B.; Li, J.N. The Evolutionary History of R2R3-MYB Proteins Across 50 Eukaryotes: New Insights Into Subfamily Classification and Expansion. Sci. Rep. 2015, 5, 11037. [Google Scholar] [CrossRef]
- Guo, P.; Wen, J.; Yang, J.; Ke, Y.; Wang, M.; Liu, M.; Ran, F.; Wu, Y.; Li, P.; Li, J.; et al. Genome-wide survey and expression analyses of the GRAS gene family in Brassica napus reveals their roles in root development and stress response. Planta 2019, 250, 1051–1072. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Hüner, N.; Tian, L. Identification and molecular characterization of the Brachypodium distachyon NRT2 family, with a major role of BdNRT2.1. Physiol. Plant. 2019, 165, 498–510. [Google Scholar] [CrossRef] [PubMed]
- Cai, C.; Wang, J.Y.; Zhu, Y.G.; Shen, Q.R.; Li, B.; Tong, Y.P.; Li, Z.S. Gene structure and expression of the high-affinity nitrate transport system in rice roots. J. Integr. Plant Biol. 2008, 50, 443–451. [Google Scholar] [CrossRef] [PubMed]
- You, L.; Wang, Y.; Zhang, T.; Zhu, Y.; Ren, N.; Jiang, X.; Zhou, Y. Genome-wide identification of nitrate transporter 2 (NRT2) gene family and functional analysis of MeNRT2.2 in cassava (Manihot esculenta Crantz). Gene 2022, 809, 146038. [Google Scholar] [CrossRef]
- Slot, J.C.; Hallstrom, K.N.; Matheny, P.B.; Hibbett, D.S. Diversification of NRT2 and the origin of its fungal homolog. Mol. Biol. Evol. 2007, 24, 1731–1743. [Google Scholar] [CrossRef]
- von Wittgenstein, N.J.; Le, C.H.; Hawkins, B.J.; Ehlting, J. Evolutionary classification of ammonium, nitrate, and peptide transporters in land plants. BMC Evol. Biol. 2014, 14, 11. [Google Scholar] [CrossRef]
- Pellizzaro, A.; Clochard, T.; Planchet, E.; Limami, A.M.; Morère-Le Paven, M.C. Identification and molecular characterization of Medicago truncatula NRT2 and NAR2 families. Physiol. Plant. 2015, 154, 256–269. [Google Scholar] [CrossRef]
- Feng, H.; Yan, M.; Fan, X.; Li, B.; Shen, Q.; Miller, A.J.; Xu, G. Spatial expression and regulation of rice high-affinity nitrate transporters by nitrogen and carbon status. J. Exp. Bot. 2011, 62, 2319–2332. [Google Scholar] [CrossRef]
- Gu, C.; Song, A.; Zhang, X.; Wang, H.; Li, T.; Chen, Y.; Jiang, J.; Chen, F.; Chen, S. Cloning of chrysanthemum high-affinity nitrate transporter family (CmNRT2) and characterization of CmNRT2.1. Sci. Rep. 2016, 6, 23462. [Google Scholar] [CrossRef]
- Remans, T.; Nacry, P.; Pervent, M.; Girin, T.; Tillard, P.; Lepetit, M.; Gojon, A. A central role for the nitrate transporter NRT2.1 in the integrated morphological and physiological responses of the root system to nitrogen limitation in Arabidopsis. Plant Physiol. 2006, 140, 909–921. [Google Scholar] [CrossRef]
- Tang, Z.; Fan, X.; Li, Q.; Feng, H.; Miller, A.J.; Shen, Q.; Xu, G. Knockdown of a rice stelar nitrate transporter alters long-distance translocation but not root influx. Plant Physiol. 2012, 160, 2052–2063. [Google Scholar] [CrossRef]
- Li, W.; He, X.; Chen, Y.; Jing, Y.; Shen, C.; Yang, J.; Teng, W.; Zhao, X.; Hu, W.; Hu, M.; et al. A wheat transcription factor positively sets seed vigour by regulating the grain nitrate signal. New Phytol. 2020, 225, 1667–1680. [Google Scholar] [CrossRef] [PubMed]
- Goodstein, D.M.; Shu, S.; Howson, R.; Neupane, R.; Hayes, R.D.; Fazo, J.; Mitros, T.; Dirks, W.; Hellsten, U.; Putnam, N.; et al. Phytozome: A comparative platform for green plant genomics. Nucleic Acids Res. 2012, 40, D1178–D1186. [Google Scholar] [CrossRef] [PubMed]
- Artimo, P.; Jonnalagedda, M.; Arnold, K.; Baratin, D.; Csardi, G.; de Castro, E.; Duvaud, S.; Flegel, V.; Fortier, A.; Gasteiger, E.; et al. ExPASy: SIB bioinformatics resource portal. Nucleic Acids Res. 2012, 40, W597–W603. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Hu, B.; Jin, J.; Guo, A.Y.; Zhang, H.; Luo, J.; Gao, G. GSDS 2.0: An upgraded gene feature visualization server. Bioinformatics 2015, 31, 1296–1297. [Google Scholar] [CrossRef]
- Li, P.; Wen, J.; Chen, P.; Guo, P.; Ke, Y.; Wang, M.; Liu, M.; Tran, L.P.; Li, J.; Du, H. MYB Superfamily in Brassica napus: Evidence for Hormone-Mediated Expression Profiles, Large Expansion, and Functions in Root Hair Development. Biomolecules 2020, 10, 875. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef]
- de Hoon, M.J.; Imoto, S.; Nolan, J.; Miyano, S. Open source clustering software. Bioinformatics 2004, 20, 1453–1454. [Google Scholar] [CrossRef]
- Saldanha, A.J. Java Treeview--extensible visualization of microarray data. Bioinformatics 2004, 20, 3246–3248. [Google Scholar] [CrossRef]
- Zhou, J.; Zhou, H.J.; Chen, P.; Zhang, L.L.; Zhu, J.T.; Li, P.F.; Yang, J.; Ke, Y.Z.; Zhou, Y.H.; Li, J.N.; et al. Genome-Wide Survey and Expression Analysis of the KT/HAK/KUP Family in Brassica napus and Its Potential Roles in the Response to K(+) Deficiency. Int. J. Mol. Sci. 2020, 21, 9487. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.H.; Huang, C.Y.; Tsay, Y.F. CHL1 is a dual-affinity nitrate transporter of Arabidopsis involved in multiple phases of nitrate uptake. Plant Cell 1999, 11, 865–874. [Google Scholar] [CrossRef] [PubMed]



| Gene Name | Genome ID | Chromosome | Protein Length (aa) | CDS Length (bp) | DNA Length (bp) | pI | Molecular Weight (kDa) | Subcellular Localization | |
|---|---|---|---|---|---|---|---|---|---|
| Predicted by Cell-PLoc2.0 | Predicted by WoLF PSORT | ||||||||
| BnaZSNRT2.6A-1 | BnaA01G0234100ZS | chrA01 | 546 | 1641 | 2624 | 7.23 | 59.39 | Cell membrane | plasmalemma |
| BnaZSNRT2.6A-2 | BnaA01G0234200ZS | chrA01 | 541 | 1626 | 1721 | 8.80 | 58.48 | Cell membrane | plasmalemma |
| BnaZSNRT2.7A-1 | BnaA02G0054200ZS | chrA02 | 484 | 1455 | 1525 | 8.14 | 52.06 | Cell membrane | plasmalemma |
| BnaZSNRT2.3A-1 | BnaA02G0096600ZS | chrA02 | 502 | 1509 | 6544 | 9.06 | 53.76 | Cell membrane | plasmalemma |
| BnaZSNRT2.1A-1 | BnaA06G0047500ZS | chrA06 | 530 | 1593 | 2163 | 9.07 | 57.70 | Cell membrane | plasmalemma |
| BnaZSNRT2.1A-2 | BnaA06G0047600ZS | chrA06 | 530 | 1593 | 1804 | 9.03 | 57.73 | Cell membrane | plasmalemma |
| BnaZSNRT2.6A-3 | BnaA06G0186600ZS | chrA06 | 543 | 1632 | 2940 | 6.89 | 59.12 | Cell membrane | plasmalemma |
| BnaZSNRT2.6A-4 | BnaA06G0186700ZS | chrA06 | 538 | 1617 | 1720 | 9.04 | 58.37 | Cell membrane | plasmalemma |
| BnaZSNRT2.5A-1 | BnaA08G0276500ZS | chrA08 | 499 | 1500 | 1773 | 9.01 | 54.17 | Cell membrane | plasmalemma |
| BnaZSNRT2.1A-3 | BnaA08G0300800ZS | chrA08 | 530 | 1593 | 2129 | 8.79 | 57.61 | Cell membrane | plasmalemma |
| BnaZSNRT2.2A-1 | BnaA09G0667700ZS | chrA09 | 506 | 1521 | 1983 | 9.02 | 54.83 | Cell membrane | plasmalemma |
| BnaZSNRT2.1A-4 | BnaA09G0667800ZS | chrA09 | 529 | 1590 | 1860 | 8.90 | 57.34 | Cell membrane | plasmalemma |
| BnaZSNRT2.4A-1 | BnaA10G0160100ZS | chrA10 | 527 | 1584 | 2368 | 8.90 | 57.61 | Cell membrane | plasmalemma |
| BnaZSNRT2.3A-2 | BnaA10G0160300ZS | chrA10 | 536 | 1611 | 1964 | 9.14 | 58.21 | Cell membrane | plasmalemma |
| BnaZSNRT2.6C-1 | BnaC01G0301400ZS | chrC01 | 546 | 1641 | 2618 | 7.66 | 59.40 | Cell membrane | plasmalemma |
| BnaZSNRT2.6C-2 | BnaC01G0301600ZS | chrC01 | 541 | 1626 | 1721 | 8.85 | 58.56 | Cell membrane | plasmalemma |
| BnaZSNRT2.7C-1 | BnaC02G0063100ZS | chrC02 | 502 | 1509 | 1579 | 7.61 | 53.45 | Cell membrane | plasmalemma |
| BnaZSNRT2.3C-1 | BnaC02G0116100ZS | chrC02 | 536 | 1611 | 2105 | 9.04 | 58.06 | Cell membrane | plasmalemma |
| BnaZSNRT2.6C-3 | BnaC03G0602800ZS | chrC03 | 538 | 1617 | 1720 | 9.13 | 58.13 | Cell membrane | plasmalemma |
| BnaZSNRT2.6C-4 | BnaC03G0603000ZS | chrC03 | 154 | 465 | 1109 | 9.86 | 17.09 | Cell membrane | cytoplasm |
| BnaZSNRT2.6C-5 | BnaC03G0603300ZS | chrC03 | 162 | 489 | 17582 | 6.91 | 17.55 | Chlo Cyto | E.R. |
| BnaZSNRT2.6C-6 | BnaC03G0603600ZS | chrC03 | 184 | 555 | 652 | 9.93 | 19.86 | Cell membrane | chloroplast |
| BnaZSNRT2.1C-1 | BnaC05G0059500ZS | chrC05 | 475 | 1428 | 1806 | 9.03 | 51.83 | Cell membrane | plasmalemma |
| BnaZSNRT2.1C-2 | BnaC05G0059600ZS | chrC05 | 530 | 1593 | 2059 | 9.03 | 57.76 | Cell membrane | plasmalemma |
| BnaZSNRT2.8C-1 | BnaC08G0033200ZS | chrC08 | 113 | 342 | 342 | 6.02 | 12.19 | Cell membrane | cytoplasm |
| BnaZSNRT2.1C-3 | BnaC08G0033300ZS | chrC08 | 395 | 1188 | 1579 | 9.00 | 43.15 | Cell membrane | chloroplast |
| BnaZSNRT2.5C-1 | BnaC08G0220400ZS | chrC08 | 498 | 1497 | 1799 | 9.11 | 54.13 | Cell membrane | plasmalemma |
| BnaZSNRT2.2C-1 | BnaC08G0532700ZS | chrC08 | 502 | 1509 | 1967 | 8.91 | 54.40 | Cell membrane | plasmalemma |
| BnaZSNRT2.1C-4 | BnaC08G0532800ZS | chrC08 | 529 | 1590 | 1915 | 8.95 | 57.38 | Cell membrane | plasmalemma |
| BnaZSNRT2.4C-1 | BnaC09G0443000ZS | chrC09 | 527 | 1584 | 4336 | 8.90 | 57.52 | Cell membrane | plasmalemma |
| BnaZSNRT2.3C-2 | BnaC09G0443100ZS | chrC09 | 567 | 1704 | 1987 | 9.08 | 61.87 | Cell membrane | plasmalemma |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Du, R.-J.; Wu, Z.-X.; Yu, Z.-X.; Li, P.-F.; Mu, J.-Y.; Zhou, J.; Li, J.-N.; Du, H. Genome-Wide Characterization of High-Affinity Nitrate Transporter 2 (NRT2) Gene Family in Brassica napus. Int. J. Mol. Sci. 2022, 23, 4965. https://doi.org/10.3390/ijms23094965
Du R-J, Wu Z-X, Yu Z-X, Li P-F, Mu J-Y, Zhou J, Li J-N, Du H. Genome-Wide Characterization of High-Affinity Nitrate Transporter 2 (NRT2) Gene Family in Brassica napus. International Journal of Molecular Sciences. 2022; 23(9):4965. https://doi.org/10.3390/ijms23094965
Chicago/Turabian StyleDu, Run-Jie, Ze-Xuan Wu, Zhao-Xi Yu, Peng-Feng Li, Jian-Yu Mu, Jie Zhou, Jia-Na Li, and Hai Du. 2022. "Genome-Wide Characterization of High-Affinity Nitrate Transporter 2 (NRT2) Gene Family in Brassica napus" International Journal of Molecular Sciences 23, no. 9: 4965. https://doi.org/10.3390/ijms23094965
APA StyleDu, R.-J., Wu, Z.-X., Yu, Z.-X., Li, P.-F., Mu, J.-Y., Zhou, J., Li, J.-N., & Du, H. (2022). Genome-Wide Characterization of High-Affinity Nitrate Transporter 2 (NRT2) Gene Family in Brassica napus. International Journal of Molecular Sciences, 23(9), 4965. https://doi.org/10.3390/ijms23094965







