Cell Therapy Based on Gingiva-Derived Mesenchymal Stem Cells Seeded in a Xenogeneic Collagen Matrix for Root Coverage of RT1 Gingival Lesions: An In Vivo Experimental Study
Abstract
:1. Introduction
2. Results
2.1. Cell Expansion and Differentiation
2.2. Histological Observations
2.2.1. Two Weeks of Healing
2.2.2. Six Weeks of Healing
2.3. Histometric Analysis
- M: The most coronal marginal mucosa;
- aJE: The most apical extent of the junctional epithelium;
- cC: The most coronal extent of the cementum;
- cN: The most apical portion of the coronal notch at the gingival margin level;
- aN: The most apical portion of the apical notch;
- Bc: The most coronal level of the Bc.
| Outcome Variable | Healing Period | CAF (n = 14) | CAF+CMX (n = 14) | CAF+CMX+GMSCs (n = 13) | CAF+CMX+FBs (n = 14) | p-Value * |
|---|---|---|---|---|---|---|
| M–aJE in mm, mean (SD) | 2 wk | 4.97 (1.45) | 4.37 (1.55) | 5.32 (1.49) | 4.87 (1.90) | 0.766 ‡ |
| 6 wk | 3.51 (2.09) | 3.24 (1.47) | 3.91 (2.29) | 4.57 (1.19) | 0.593 ‡ | |
| cN–M in mm, mean (SD) | 2 wk | 2.54 (0.92) * | 0.61 (1.09) * | 1.31 (0.54) | 1.09 (0.72) * | 0.003 ‡ |
| 6 wk | 0.93 (0.82) | 1.02 (0.99) | 0.88 (1.94) | 1.22 (0.56) | 0.950 ‡ | |
| aN–aJE in mm, mean (SD) | 2 wk | 1.62 (3.44) | 0.61 (1.20) | −0.031 (0.062) | 0.96 (1.25) | 0.177 † |
| 6 wk | 1.52 (1.59) | 1.69 (0.99) | 1.46 (1.04) | 1.30 (2.13) | 0.498 ‡ | |
| aN–cC in mm, mean (SD) | 2 wk | 0.08 (0.21) | 0.59 (1.21) | 0.00 (0.00) | 1.09 (0.72) | 0.701 † |
| 6 wk | 1.43 (1.58) | 1.58 (1.07) | 1.35 (1.17) | 0.40 (0.65) | 0.252 ‡ | |
| aN–Bc in mm, mean (SD) | 2 wk | −0.48 (0.61) | −0.41 (0.84) | −0.50 (0.75) | −0.034 (1.11) | 0.825 † |
| 6 wk | −0.14 (0.62) | −0.018 (0.99) | −0.207 (0.50) | 0.03 (0.74) | 0.948 ‡ |
2.3.1. Epithelial Length (M–aJE)
2.3.2. Histological Root Coverage (cN–M)
2.3.3. Length of the Connective Tissue Attachment (aN–aJe)
2.3.4. Length of Newly Formed Cementum (aN–cC)
2.3.5. Dimensions of New Bone (aN–Bc)
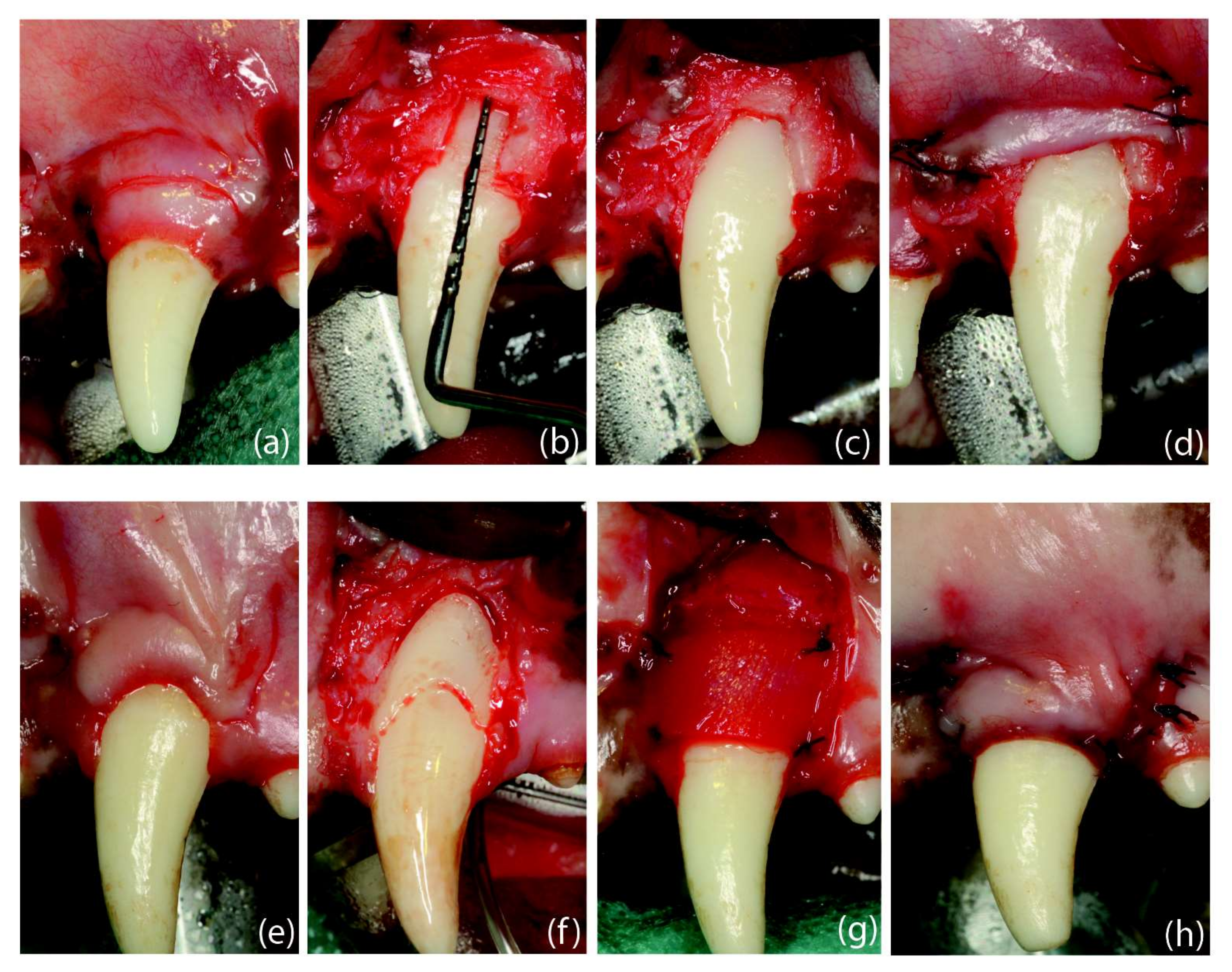
2.4. Clinical Observations
3. Discussion
4. Materials and Methods
4.1. Study Design and Randomization
4.2. Experimental Sample
4.3. Treatment Protocols
4.3.1. Phase 1: Tooth Extraction and Cell Isolation
4.3.2. Phase 2: Experimental Dehiscence-Type Defects
4.3.3. Phase 3. Root Coverage Surgeries
4.3.4. Phase 4: Euthanasia
4.4. Histological Processing
4.5. Histological and Histometric Analysis
4.6. Clinical Evaluation
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Treatment Group | Mean | SD | 95% CI of the Difference | p-Value | |
|---|---|---|---|---|---|
| Lower | Upper | ||||
| CAF | 2.00 | 1.10 | 1.35 | 2.64 | 0.000 # |
| CAF+CMX | 0.92 | 1.14 | 0.26 | 1.58 | 0.009 # |
| CAF+CMX+GMSCs | 1.46 | 1.65 | 0.50 | 2.42 | 0.006 # |
| CAF+CMX+FBs | 1.53 | 0.88 | 1.02 | 2.04 | 0.000 # |
Appendix B
| Recession Reduction 0–6 Weeks (%) | CAF | CAF+CMX | CAF+CMX+GMSC | CAF+CMX+FBs | p-Value * |
|---|---|---|---|---|---|
| ≥1 mm | 86 | 43 | 71 | 86 | 0.241 |
| ≥2 mm | 0 | 29 | 57 | 14 | 0.086 |
Appendix C
| Variable | Upper Canine (n = 14) | Upper 2nd Incisor (n = 13) | Lower 1st Molar (n = 14) | Lower Canine (n = 14) | p-Value |
|---|---|---|---|---|---|
| Recession length in mm mean (SD) | 0.25 (0.58) | 0.42 (0.53) | 0.35 (0.49) | 0.71 (0.87) | 0.358 † |
| RecRed in mm mean (SD) | |||||
| Baseline–2 weeks | 2.35 (1.31) | 1.08 (1.11) | 1.71 (1.28) | 2.28 (1.46) | 0.295 ‡ |
| Baseline–6 weeks | 1.35 (1.54) | 0.92 (0.53) | 1.5 (0.70) | 0.78 (1.25) | 0.572 ‡ |
| Root coverage in % mean (SD) | |||||
| Baseline–2 weeks | 96.66 (8.16) | 50.00 (63.24) | 92.85 (18.89) | 79.76 (34.64) | 0.330 † |
| Baseline–6 weeks | 72.42 (48.79) | 72.61 (40.45) | 67.38 (34.61) | 28.57 (85.91) | 0.508 † |
| Specimens with CRC in % | |||||
| 2 weeks | 83.3 | 50 | 85.7 | 71.4 | 0.471 # |
| 6 weeks | 71.4 | 57.1 | 28.6 | 28.6 | 0.275 # |
References
- Tavelli, L.; Barootchi, S.; Cairo, F.; Rasperini, G.; Shedden, K.; Wang, H.L. The Effect of Time on Root Coverage Outcomes: A Network Meta-analysis. J. Dent. Res. 2019, 98, 1195–1203. [Google Scholar] [CrossRef] [PubMed]
- Zuhr, O.; Baumer, D.; Hurzeler, M. The addition of soft tissue replacement grafts in plastic periodontal and implant surgery: Critical elements in design and execution. J. Clin. Periodontol. 2014, 41, S123–S142. [Google Scholar] [CrossRef]
- Cairo, F.; Barootchi, S.; Tavelli, L.; Barbato, L.; Wang, H.L.; Rasperini, G.; Graziani, F.; Tonetti, M. Aesthetic-And patient-related outcomes following root coverage procedures: A systematic review and network meta-analysis. J. Clin. Periodontol. 2020, 47, 1403–1415. [Google Scholar] [CrossRef] [PubMed]
- Nunez, J.; Caffesse, R.; Vignoletti, F.; Guerra, F.; San Roman, F.; Sanz, M. Clinical and histological evaluation of an acellular dermal matrix allograft in combination with the coronally advanced flap in the treatment of Miller class I recession defects: An experimental study in the mini-pig. J. Clin. Periodontol. 2009, 36, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Vignoletti, F.; Nunez, J.; Discepoli, N.; De Sanctis, F.; Caffesse, R.; Munoz, F.; Lopez, M.; Sanz, M. Clinical and histological healing of a new collagen matrix in combination with the coronally advanced flap for the treatment of Miller class-I recession defects: An experimental study in the minipig. J. Clin. Periodontol. 2011, 38, 847–855. [Google Scholar] [CrossRef]
- Jepsen, K.; Jepsen, S.; Zucchelli, G.; Stefanini, M.; de Sanctis, M.; Baldini, N.; Greven, B.; Heinz, B.; Wennstrom, J.; Cassel, B.; et al. Treatment of gingival recession defects with a coronally advanced flap and a xenogeneic collagen matrix: A multicenter randomized clinical trial. J. Clin. Periodontol. 2013, 40, 82–89. [Google Scholar] [CrossRef]
- Moreira, A.R.O.; Santamaria, M.P.; Silverio, K.G.; Casati, M.Z.; Nociti Junior, F.H.; Sculean, A.; Sallum, E.A. Coronally advanced flap with or without porcine collagen matrix for root coverage: A randomized clinical trial. Clin. Oral Investig. 2016, 20, 2539–2549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rotundo, R.; Genzano, L.; Patel, D.; D’Aiuto, F.; Nieri, M. Adjunctive benefit of a xenogenic collagen matrix associated with coronally advanced flap for the treatment of multiple gingival recessions: A superiority, assessor-blind, randomized clinical trial. J. Clin. Periodontol. 2019, 46, 1013–1023. [Google Scholar] [CrossRef]
- Aroca, S.; Molnar, B.; Windisch, P.; Gera, I.; Salvi, G.E.; Nikolidakis, D.; Sculean, A. Treatment of multiple adjacent Miller class I and II gingival recessions with a Modified Coronally Advanced Tunnel (MCAT) technique and a collagen matrix or palatal connective tissue graft: A randomized, controlled clinical trial. J. Clin. Periodontol. 2013, 40, 713–720. [Google Scholar] [CrossRef] [PubMed]
- McGuire, M.K.; Scheyer, E.T. Long-Term Results Comparing Xenogeneic Collagen Matrix and Autogenous Connective Tissue Grafts With Coronally Advanced Flaps for Treatment of Dehiscence-Type Recession Defects. J. Periodontol. 2016, 87, 221–227. [Google Scholar] [CrossRef]
- Tonetti, M.S.; Cortellini, P.; Pellegrini, G.; Nieri, M.; Bonaccini, D.; Allegri, M.; Bouchard, P.; Cairo, F.; Conforti, G.; Fourmousis, I.; et al. Xenogenic collagen matrix or autologous connective tissue graft as adjunct to coronally advanced flaps for coverage of multiple adjacent gingival recession: Randomized trial assessing non-inferiority in root coverage and superiority in oral health-related quality of life. J. Clin. Periodontol. 2018, 45, 78–88. [Google Scholar] [CrossRef]
- Cha, J.K.; Sun, Y.K.; Lee, J.S.; Choi, S.H.; Jung, U.W. Root coverage using porcine collagen matrix with fibroblast growth factor-2: A pilot study in dogs. J. Clin. Periodontol. 2017, 44, 96–103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simion, M.; Rocchietta, I.; Fontana, F.; Dellavia, C. Evaluation of a resorbable collagen matrix infused with rhPDGF-BB in peri-implant soft tissue augmentation: A preliminary report with 3.5 years of observation. Int. J. Periodontics Restor. Dent. 2012, 32, 273–282. [Google Scholar]
- Zanwar, K.; Kumar Ganji, K.; Bhongade, M.L. Efficacy of Human Umbilical Stem Cells Cultured on Polylactic/ Polyglycolic Acid Membrane in the Treatment of Multiple Gingival Recession Defects: A Randomized Controlled Clinical Study. J. Dent. 2017, 18, 95–103. [Google Scholar]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, X.; Cao, W.; Shi, Y. Plasticity of mesenchymal stem cells in immunomodulation: Pathological and therapeutic implications. Nat. Immunol. 2014, 15, 1009–1016. [Google Scholar] [CrossRef] [PubMed]
- Wada, N.; Gronthos, S.; Bartold, P.M. Immunomodulatory effects of stem cells. Periodontology 2000 2013, 63, 198–216. [Google Scholar] [CrossRef]
- Kuo, T.F.; Lee, S.Y.; Wu, H.D.; Poma, M.; Wu, Y.W.; Yang, J.C. An in vivo swine study for xeno-grafts of calcium sulfate-based bone grafts with human dental pulp stem cells (hDPSCs). Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 50, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yin, X.; Ye, Q.; He, W.; Ge, M.; Zhou, X.; Hu, J.; Zou, S. Periodontal regeneration with stem cells-seeded collagen-hydroxyapatite scaffold. J. Biomater. Appl. 2016, 31, 121–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nunez, J.; Sanz-Blasco, S.; Vignoletti, F.; Munoz, F.; Arzate, H.; Villalobos, C.; Nunez, L.; Caffesse, R.G.; Sanz, M. Periodontal regeneration following implantation of cementum and periodontal ligament-derived cells. J. Periodontal. Res. 2012, 47, 33–44. [Google Scholar] [CrossRef]
- Sanchez, N.; Fierravanti, L.; Nunez, J.; Vignoletti, F.; Gonzalez-Zamora, M.; Santamaria, S.; Suarez-Sancho, S.; Fernandez-Santos, M.E.; Figuero, E.; Herrera, D.; et al. Periodontal regeneration using a xenogeneic bone substitute seeded with autologous periodontal ligament-derived mesenchymal stem cells: A 12-month quasi-randomized controlled pilot clinical trial. J. Clin. Periodontol. 2020, 47, 1391–1402. [Google Scholar] [CrossRef] [PubMed]
- Shanbhag, S.; Suliman, S.; Pandis, N.; Stavropoulos, A.; Sanz, M.; Mustafa, K. Cell therapy for orofacial bone regeneration: A systematic review and meta-analysis. J. Clin. Periodontol. 2019, 46, 162–182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santamaria, S.; Sanchez, N.; Sanz, M.; Garcia-Sanz, J.A. Comparison of periodontal ligament and gingiva-derived mesenchymal stem cells for regenerative therapies. Clin. Oral Investig. 2017, 21, 1095–1102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrukhov, O.; Behm, C.; Blufstein, A.; Rausch-Fan, X. Immunomodulatory properties of dental tissue-derived mesenchymal stem cells: Implication in disease and tissue regeneration. World J. Stem Cells 2019, 11, 604–617. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Wang, Y.; Li, Q.; Liu, K.; Hou, J.; Shao, C.; Wang, Y. Immunoregulatory mechanisms of mesenchymal stem and stromal cells in inflammatory diseases. Nat. Rev. Nephrol. 2018, 14, 493–507. [Google Scholar] [CrossRef]
- Wada, N.; Bartold, P.M.; Gronthos, S. Human foreskin fibroblasts exert immunomodulatory properties by a different mechanism to bone marrow stromal/stem cells. Stem Cells Dev. 2011, 20, 647–659. [Google Scholar] [CrossRef]
- Jhaveri, H.M.; Chavan, M.S.; Tomar, G.B.; Deshmukh, V.L.; Wani, M.R.; Miller, P.D., Jr. Acellular dermal matrix seeded with autologous gingival fibroblasts for the treatment of gingival recession: A proof-of-concept study. J. Periodontol. 2010, 81, 616–625. [Google Scholar] [CrossRef]
- Koseoglu, S.; Duran, I.; Saglam, M.; Bozkurt, S.B.; Kirtiloglu, O.S.; Hakki, S.S. Efficacy of collagen membrane seeded with autologous gingival fibroblasts in gingival recession treatment: A randomized, controlled pilot study. J. Periodontol. 2013, 84, 1416–1424. [Google Scholar] [CrossRef]
- Milinkovic, I.; Aleksic, Z.; Jankovic, S.; Popovic, O.; Bajic, M.; Cakic, S.; Lekovic, V. Clinical application of autologous fibroblast cell culture in gingival recession treatment. J. Periodontal. Res. 2015, 50, 363–370. [Google Scholar] [CrossRef]
- Murata, M.; Okuda, K.; Momose, M.; Kubo, K.; Kuroyanagi, Y.; Wolff, L.F. Root coverage with cultured gingival dermal substitute composed of gingival fibroblasts and matrix: A case series. Int. J. Periodontics Restor. Dent. 2008, 28, 461–467. [Google Scholar]
- Wilson, T.G., Jr.; McGuire, M.K.; Nunn, M.E. Evaluation of the safety and efficacy of periodontal applications of a living tissue-engineered human fibroblast-derived dermal substitute. II. Comparison to the subepithelial connective tissue graft: A randomized controlled feasibility study. J. Periodontol. 2005, 76, 881–889. [Google Scholar] [CrossRef]
- Zanwar, K.; Bhongade, M.L.; Ganji, K.K.; Koudale, S.B.; Gowda, P. Comparative evaluation of efficacy of stem cells in combination with PLA/PGA membrane versus sub-epithelial connective tissue for the treatment of multiple gingival recession defects: A clinical study. J. Stem Cells 2014, 9, 253–267. [Google Scholar] [PubMed]
- Miller, P.D., Jr. A classification of marginal tissue recession. Int. J. Periodontics Restor. Dent. 1985, 5, 8–13. [Google Scholar]
- Cortellini, P.; Bissada, N.F. Mucogingival conditions in the natural dentition: Narrative review, case definitions, and diagnostic considerations. J. Clin. Periodontol. 2018, 45, S190–S198. [Google Scholar] [CrossRef] [Green Version]
- de Sanctis, M.; Zucchelli, G. Coronally advanced flap: A modified surgical approach for isolated recession-type defects: Three-year results. J. Clin. Periodontol. 2007, 34, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Lindl, T.; Lewandowski, B.; Schreyogg, S.; Staudte, A. An evaluation of the in vitro cytotoxicities of 50 chemicals by using an electrical current exclusion method versus the neutral red uptake and MTT assays. Altern. Lab. Anim. 2005, 33, 591–601. [Google Scholar] [CrossRef]
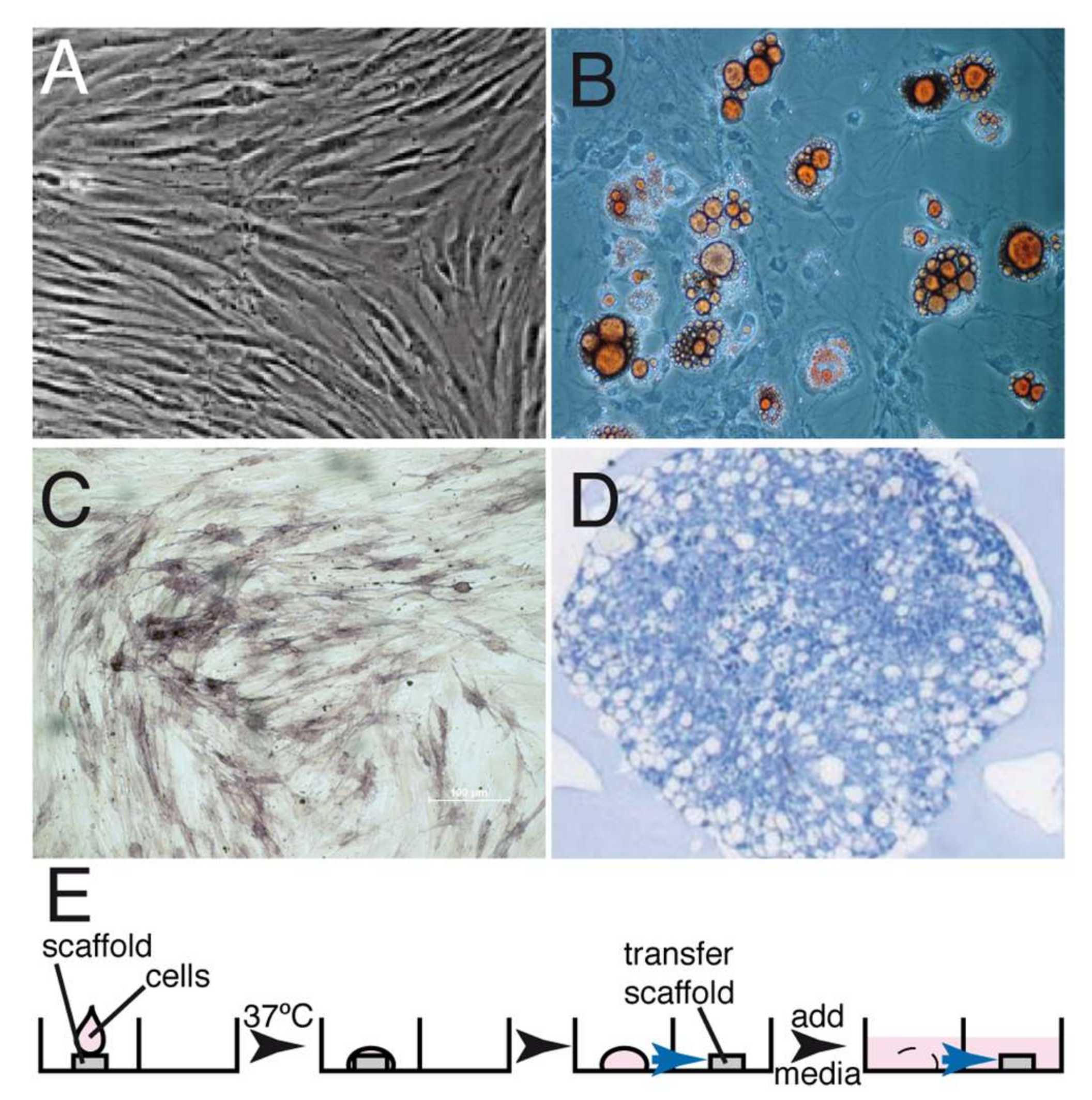
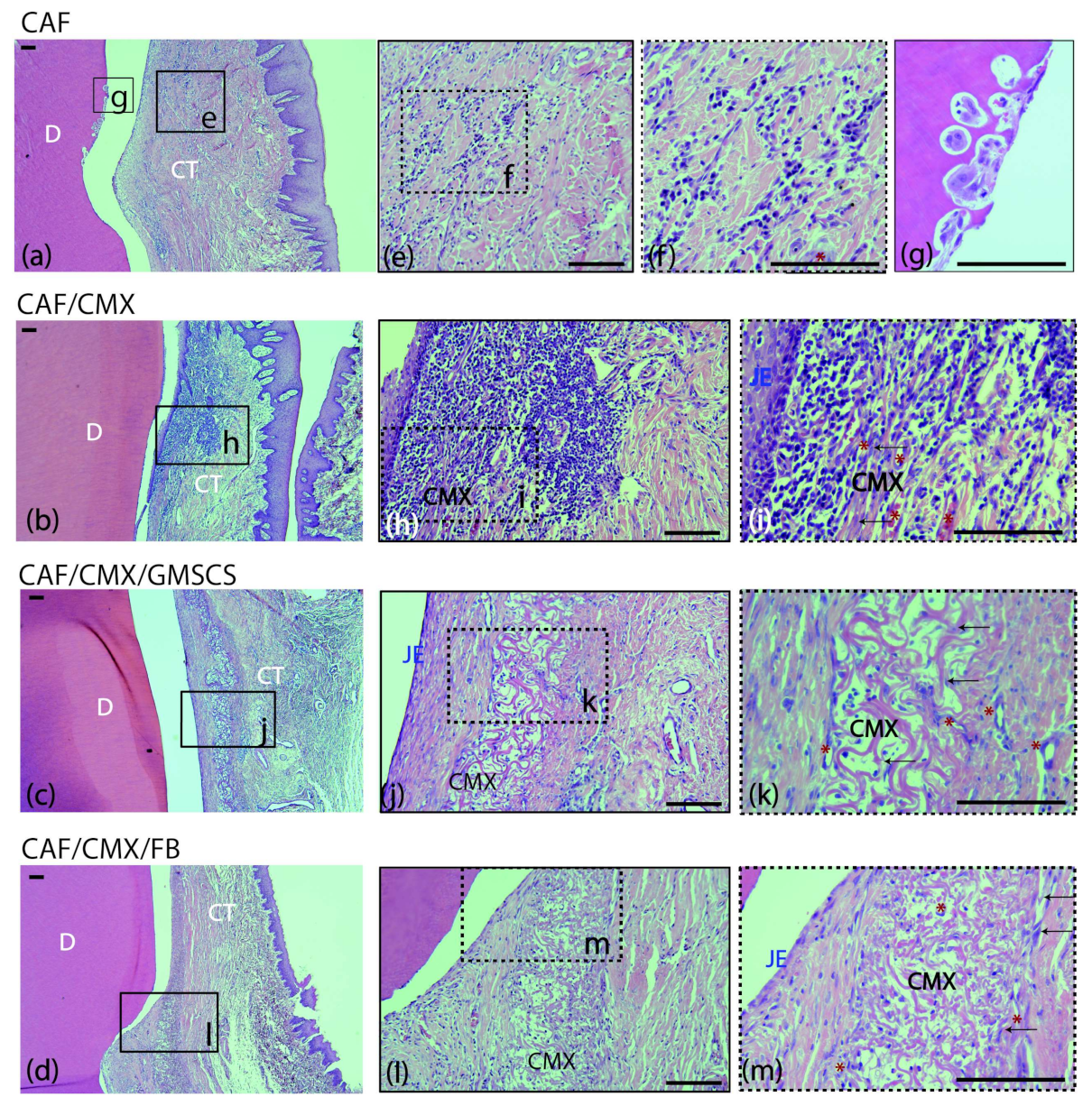
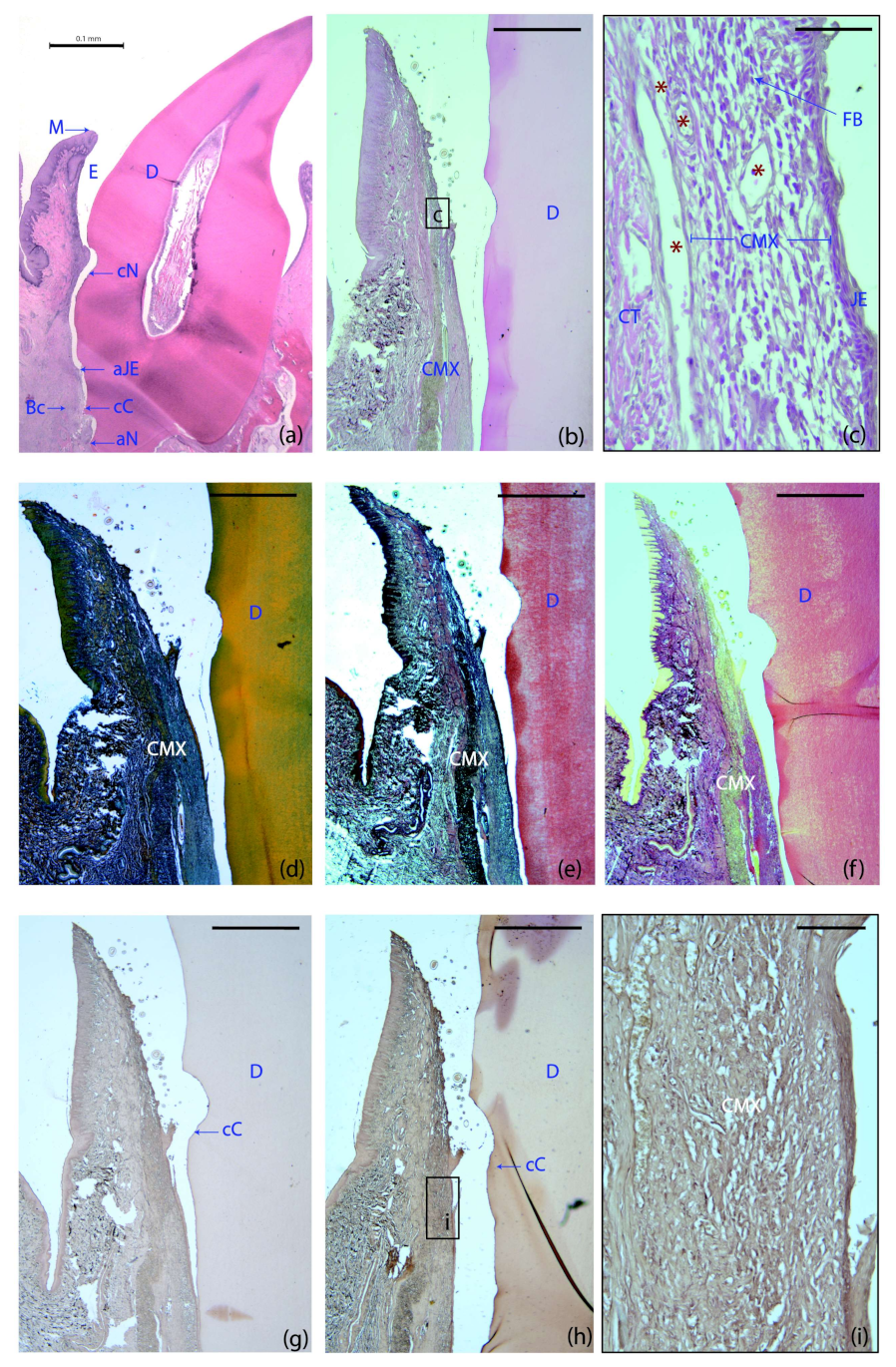
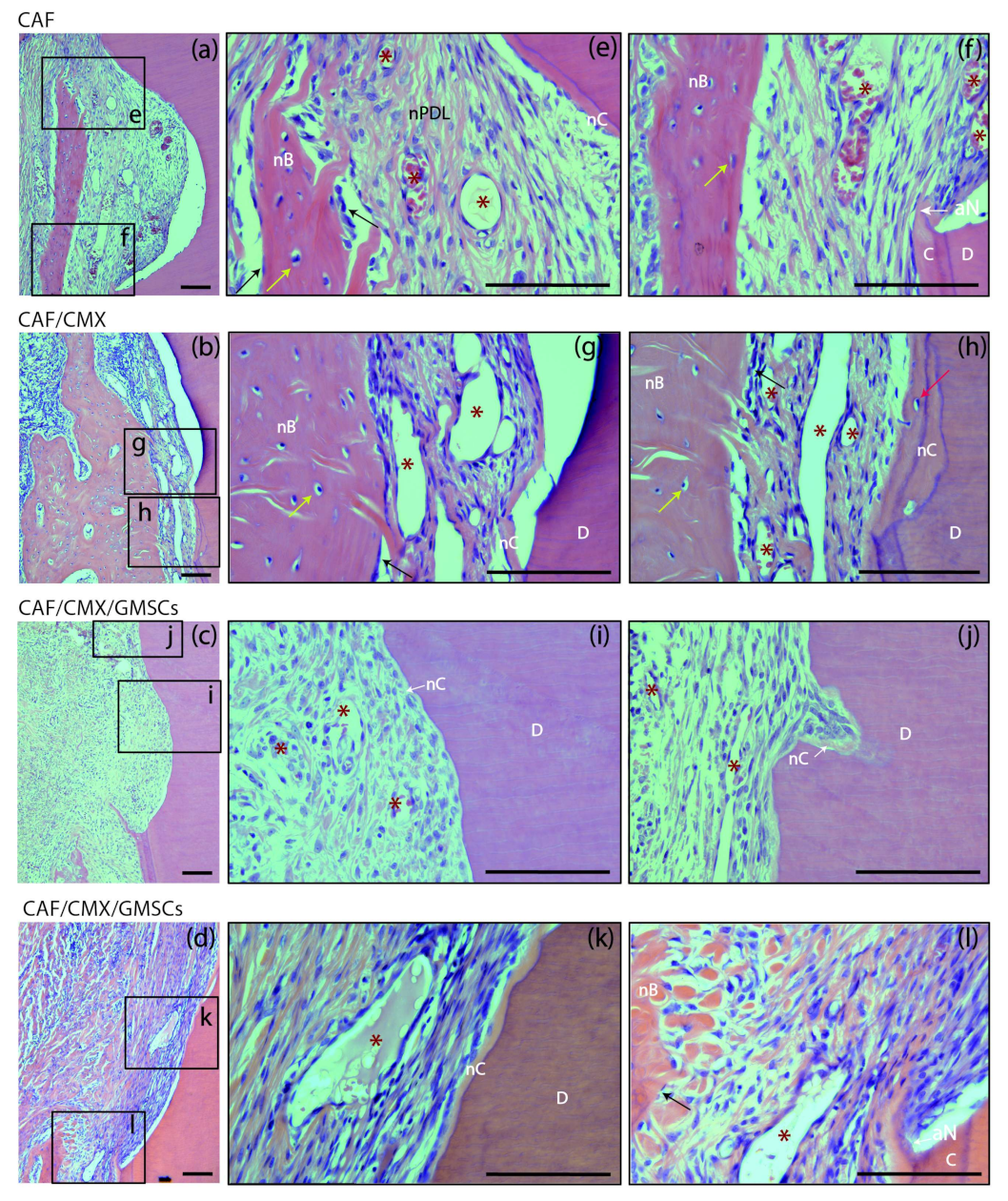
| Variable | CAF (n = 14) | CAF+CMX (n = 14) | CAF+CMX+GMSCs (n = 13) | CAF+CMX+FBs (n = 14) | p-Value |
|---|---|---|---|---|---|
| Recession length in mm mean (SD) | |||||
| Baseline | 2.28 (0.97) | 1.60 (0.88) | 2.07 (1.26) | 1.89 (0.81) | 0.289 † |
| 2 weeks | 0 | 0.57 (0.67) | 0.25 (0.41) | 0.14 (0.37) | 0.095 † |
| 6 weeks | 0.57 (0.60) | 0.78 (0.69) | 0.57 (0.93) | 0.57 (0.83) | 0.816 † |
| RecRed in mm mean (SD) | |||||
| Baseline–2 weeks | 2.85 (0.89) | 1.07 (1.39) | 1.75 (1.54) | 1.85 (1.02) | 0.085 ‡ |
| Baseline–6 weeks | 1.14 (0.37) | 0.78 (0.90) | 1.42 (1.88) | 1.21 (0.63) | 0.749 ‡ |
| Root coverage in % mean (SD) | |||||
| Baseline–2 weeks | 100 (0.0) | 46.42 (58,50) | 82.66 (28.90) | 92.85 (18.89) | 0.062 † |
| Baseline–6 weeks | 72.61 (28.34) | 45.23 (45.86) | 47.14 (94.28) | 75.00 (38.18) | 0.586 † |
| Specimens with CRC in % | |||||
| 2 weeks | 100 | 42.9 | 60 | 85.7 | 0.077 * |
| 6 weeks | 42.9 | 28.6 | 57.1 | 57.1 | 0.664 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanchez, N.; Vignoletti, F.; Sanz-Martin, I.; Coca, A.; Nuñez, J.; Maldonado, E.; Sanz-Esporrin, J.; Hernando-Pradíes, I.; Santamaría, S.; Herrera, D.; et al. Cell Therapy Based on Gingiva-Derived Mesenchymal Stem Cells Seeded in a Xenogeneic Collagen Matrix for Root Coverage of RT1 Gingival Lesions: An In Vivo Experimental Study. Int. J. Mol. Sci. 2022, 23, 3248. https://doi.org/10.3390/ijms23063248
Sanchez N, Vignoletti F, Sanz-Martin I, Coca A, Nuñez J, Maldonado E, Sanz-Esporrin J, Hernando-Pradíes I, Santamaría S, Herrera D, et al. Cell Therapy Based on Gingiva-Derived Mesenchymal Stem Cells Seeded in a Xenogeneic Collagen Matrix for Root Coverage of RT1 Gingival Lesions: An In Vivo Experimental Study. International Journal of Molecular Sciences. 2022; 23(6):3248. https://doi.org/10.3390/ijms23063248
Chicago/Turabian StyleSanchez, Nerea, Fabio Vignoletti, Ignacio Sanz-Martin, Alejandro Coca, Javier Nuñez, Estela Maldonado, Javier Sanz-Esporrin, Irene Hernando-Pradíes, Silvia Santamaría, David Herrera, and et al. 2022. "Cell Therapy Based on Gingiva-Derived Mesenchymal Stem Cells Seeded in a Xenogeneic Collagen Matrix for Root Coverage of RT1 Gingival Lesions: An In Vivo Experimental Study" International Journal of Molecular Sciences 23, no. 6: 3248. https://doi.org/10.3390/ijms23063248
APA StyleSanchez, N., Vignoletti, F., Sanz-Martin, I., Coca, A., Nuñez, J., Maldonado, E., Sanz-Esporrin, J., Hernando-Pradíes, I., Santamaría, S., Herrera, D., Garcia-Sanz, J. A., & Sanz, M. (2022). Cell Therapy Based on Gingiva-Derived Mesenchymal Stem Cells Seeded in a Xenogeneic Collagen Matrix for Root Coverage of RT1 Gingival Lesions: An In Vivo Experimental Study. International Journal of Molecular Sciences, 23(6), 3248. https://doi.org/10.3390/ijms23063248








