Abstract
Cyclodextrins (CDs) are cyclic oligomers broadly used in food manufacturing as food additives for different purposes, e.g., to improve sensorial qualities, shelf life, and sequestration of components. In this review, the latest advancements of their applications along with the characteristics of the uses of the different CDs (α, β, γ and their derivatives) were reviewed. Their beneficial effects can be achieved by mixing small amounts of CDs with the target material to be stabilized. Essentially, they have the capacity to form stable inclusion complexes with sensitive lipophilic nutrients and constituents of flavor and taste. Their toxicity has been also studied, showing that CDs are innocuous in oral administration. A review of the current legislation was also carried out, showing a general trend towards a wider acceptance of CDs as food additives. Suitable and cost-effective procedures for the manufacture of CDs have progressed, and nowadays it is possible to obtain realistic prices and used them in foods. Therefore, CDs have a promising future due to consumer demand for healthy and functional products.
1. Introduction
Numerous investigations have been focused on developing relatively simple organic compounds able to catalyze organic reactions, like enzymes. Since the discovery of enzymes’ active sites, new models of synthesis of nonpeptide organic systems that could simulate the enzymatic behavior have been developed. Chemists have been developing more sophisticated molecular structures within the nanometric order. However, even though apparently the easiest way to solve it would be to assemble individual molecules directly, the most efficient alternative is to generate molecules with a complementary form capable of spontaneously self-organizing, resulting in orderly assemblies. This can be achieved, for example, with host and guest molecules [1].
Host–guest complexes are molecular aggregates stabilized via noncovalent bonds (for example, van der Waals, hydrogen bonds, and hydrophobic interactions), but never by complete covalent bonds. Host molecules are characterized by having an inner cavity where another molecule can be incorporated, this is, the guest molecule. Therefore, hosts will act as receptors and guests as substrates, inhibitors, or cofactors [2]. The resulting molecular inclusion complex can easily break under determined physiological environments [3].
Therefore, the establishment of these systems can improve physicochemical properties of the guest molecule. Different types of host molecules have been developed, but they are all characterized by acting as non-natural receptors capable of partial or total enclosure of the compounds of interest (drugs, active compounds, etc.). Likewise, numerous hosts-guest complexes have already been synthesized. Among them, crown ethers, cryptands, spherands, carcerands, and cyclodextrins (CDs) can be highlighted.
CDs are a group of relatively recent discovered compounds, which are also identified as cycloamyloses, cyclomaltoses, and Schardinger dextrins [4]. Their first reference dates from 1891. In this year, a crystalline product was isolated during a starch fermentation carried out by Bacillus amylobacter. Some authors reflected that this production was probably due to impurities in cultures and, consequently, it was supposed that CDs might be produced by cross-contamination with another species (B. macerans). Later, in 1903, two crystalline components were isolated: dextrins A and B, which were characterized by their lack of reducing power. Even so, the bacterial strain responsible of its production was unfortunately not preserved [5]. A year later, the same authors were able to isolate an organism capable of producing acetone and ethyl alcohol from plant materials that contained sugar and starch. In 1911, it was discovered that a strain of B. macerans produced large amounts of crystalline dextrins (25–30%) from starch too. These resulting products were named as crystallized dextrin α and crystallized dextrin β [4]. It was not until 1935 that the third type of dextrin (dextrin γ) was isolated. Several fractionation processes were also developed in order to produce CDs [5]. At that point, the structure of said molecules was still a mystery. It was not fully discovered until 1942, when the structure of two CDs (α and β) was established through X-ray crystallography. A few years later (1948), the same method was applied to γ-CDs. Later, in 1961, more types of CDs (9–12 residues) were discovered [6].
CDs are one of the most used hosts-guest complexes for organic molecules. Their structure consists of a cyclic arrangement of D (+)-glucopyranose units joined by α-(1 → 4) glycosidic links. They can be differentiated according to their number of glucoses in α-, β- and γ-CDs (6, 7, and 8 glucose units, respectively) (Table 1) [7,8]. Regarding its form, it is a truncated cone characterized as bearing primary and secondary OH groups, respectively, in the slim and wide rim. It is characterized by having cavities with hydrophobic properties. In this case, as in other complexes, Van der Waals and hydrophobic forces are responsible for keeping guest and host together with a partial or total adjustment of the cavity. Due to the establishment of an inclusion complex, guest reactivity can vary, making possible its use in a wide variety of fields [9]. Furthermore, as these receptors can improve bioavailability, they are suitable for functional delivery systems [10]. They can be found in food, pharmaceuticals, cosmetics, the textile industry, conversion and fermentation processes, and environmental and other chemical systems and applications. However, the development of hosts is still ongoing, the development of processes not only simple and reproducible, but also economically profitable is fundamental so that their widespread acceptance would be guaranteed in diverse fields such as food and drug production [11].

Table 1.
Main cyclodextrins (CDs) properties [5,17].
Regarding the importance and new advances on the field of CDs and food science, some recent reviews have been focused on the latest updates of the use of CDs in food products such as nanoparticles, nanosensors, extraction enhancers, active or smart packaging, and their most widespread use as carriers and complexes formers to protect and stabilize bioactive compounds with the aim of improving the final product [12,13]. However, future research is also focused on CDs properties and their interaction with other materials (e.g., durability, stability, or dispersion), new applications (e.g., CDs immobilization for wastewater treatment) or the development of green chemistry processes [14]. They have also been used for sequester certain molecules (i.e., cholesterol), as modulators for medicinal biomaterials, as elicitors or as synergistic agents for the production of secondary metabolites in plants [15,16].
This review will be focused on the main applications of CDs in the food industry as well as in other applications, revising toxicity and legislation aspects and performing a discussion about the advantages and disadvantages of their use. Nevertheless, some applications such as nanoparticles, nanosensors, or active packaging have not been reviewed in this manuscript. Complementary information on these fields can be found along the most recent bibliography cited in this article.
2. Cyclodextrins
2.1. Definition
Industrially, CDs are enzymatically produced with cyclodextrin glucosyltransferase from modified starch. This is an economic process, whose production is estimated in 150 tons per year [7]. The resulting product is nontoxic, characterized by not being absorbed in the upper gastrointestinal tract and being entirely processed by the colon microflora. [18].
Natural CDs are also characterized by poor solubility. At the beginning, this was a major inconvenience since it prevented the use of CDs as effective complexing agents. It was not until the end of the 1960s when it was revealed that, by means of chemical replacements at positions 2, 3, and 6-hydroxyl, the solubility was highly increased. In addition, depending on the grade of chemical replacement and the type of the substituents, the maximum concentration of CDs in aqueous mediums was improved. The maximum concentration that most chemically modified CDs can reach is 50% (w/v) in water by carrying out substitutions at the 2, 3, and 6 hydroxyl like (2-Hydroxypropyl)-β-CDs [8].
2.2. Mechanism
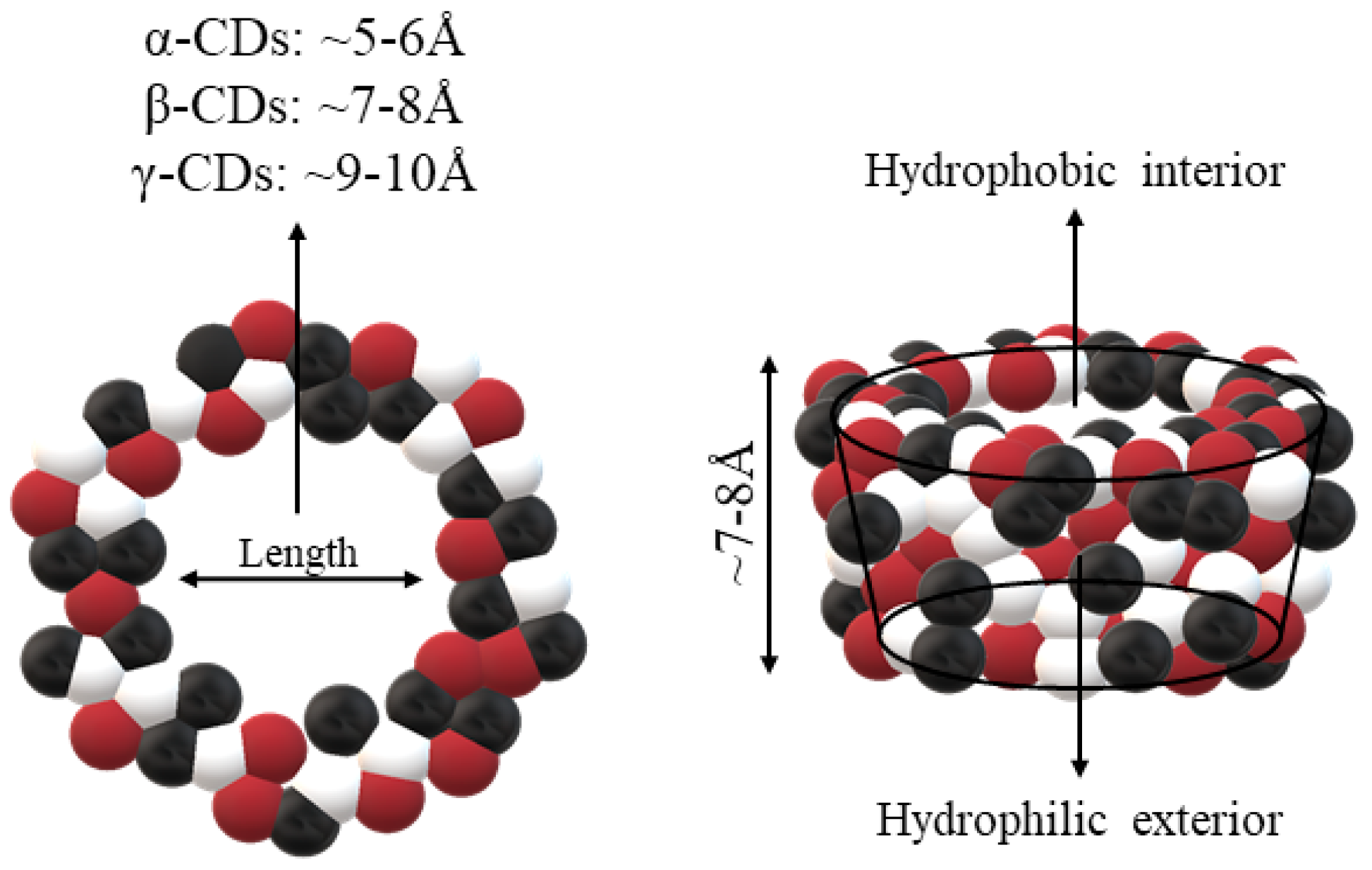
Some parameters must be considered for the study of CDs mechanism of action. The size of the cavity is essential to choose which type of CDs is more suitable to use in the interaction. The size is related to the degree of adjustment, which is a critical parameter to achieve optimal CDs incorporation. Therefore, each type of CDs will have different types of cavities. The α-CDs possess small cavities, so that they are not able to accept many molecules. The γ-CDs are larger in size compared to most molecules that can be complexed. In addition, hydrophobic CDs loads cannot efficiently interact to simplify complex formation. Therefore, it is considered that the diameter of the cavity of β-CDs is the most appropriate for molecules such as hormones, vitamins, and other compounds commonly used in tissue and cell culture applications. All these features make β-CDs the CDs of choice as complexing agent in most cases [19].
The structure of CDs allows the enclosure of hydrophobic molecules, such as vitamins and lipid soluble hormones, improving their solubility in aqueous systems. The process can be reversed by diluting the complex in a larger volume of solvent. As a result, the molecule of interest is released into the environment. The structure of the resulting complexes (Figure 1) is like a cage, with the same characteristics as those formed by cryptands, calixarenes, cyclophanes, spheres, and crown ethers. As mentioned before, all these molecules are involved in chemical reactions due to non-covalent bonds, with most of the reactions happening as the “host–guest” type [19,20].
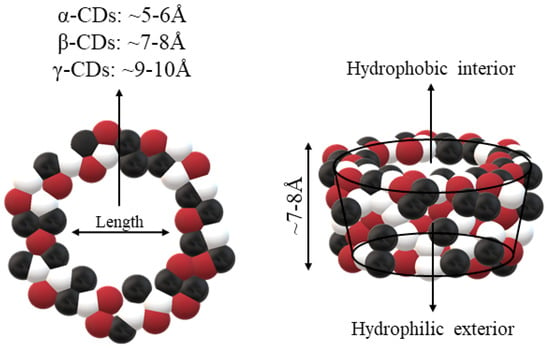
Figure 1.
Schematic representations of cyclodextrins (CDs). Adapted from [13,21,22].
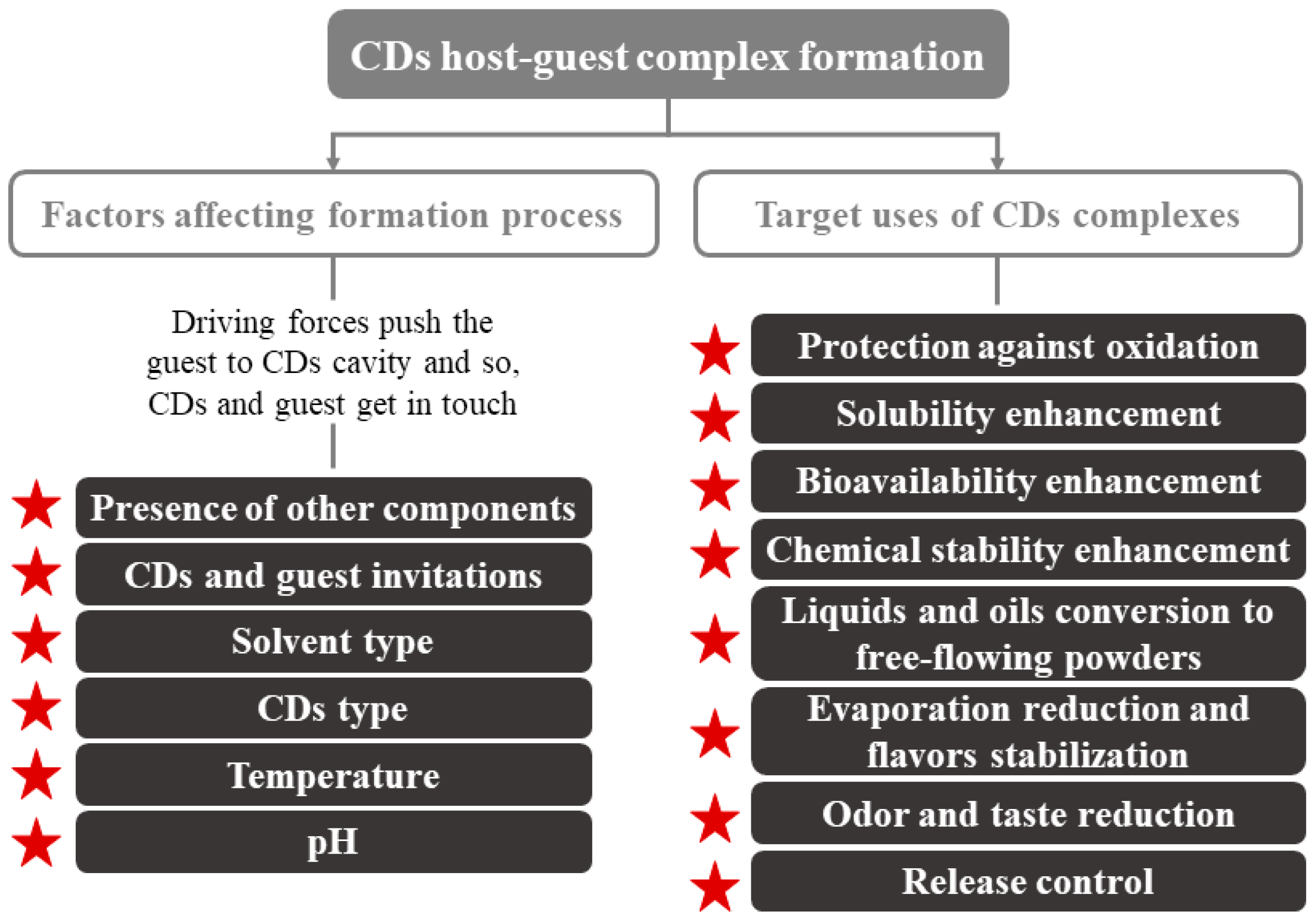
Regarding their physical characteristics, CDs complexes can get wet; they are almost odorless and non-hygroscopic powders [18,23]. Their mechanical properties (crystallization, flow, etc.) depend on the complex formation process. The moisture content and temperature present an enormous importance in controlling the deformation or release of the CDs-guest. Other parameters to consider can be seen in Figure 2. CDs are crystallized by two main mechanisms that, depending on the type of compounds that make up the complex (CDs and guest), turn into to two key categories of crystal packing (channel or cage structures) [5]. An example of these molecules is the flavor/β-CDs complexes, which are formed after a co-crystallization, kneading, and suspension process [18].
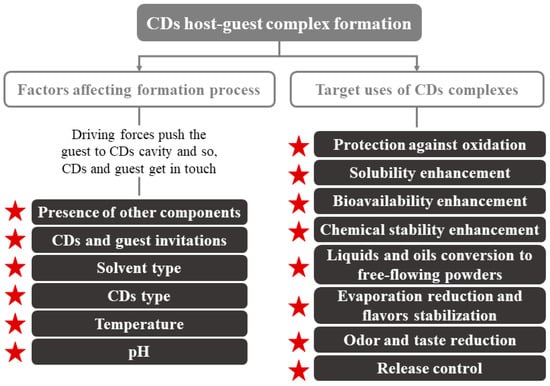
Figure 2.
Factors affecting CDs inclusion complex formation and main application of CDs. Adapted from [22].
3. Applications in the Food Industry
CDs are used primarily in foods for the encapsulation of compounds of interest and the improvement of water retention, since they are hygroscopic compounds [18]. Their use can enhance several technological advantages, such as more homogeneous compositions that are easier to be standardized [18,24]. Numerous applications of CDs have been described, e.g., improving the organoleptic quality (total or partial elimination of undesired flavors/odors), increasing food shelf life, component sequestration, and pickering emulsions, among others (Figure 2).
3.1. Improving Sensorial Qualities
3.1.1. Color
Food color is the first quality parameter assessed by customers, so it is a key parameter of food quality [25]. CDs can be applied to modulate food color by increasing solubility and chemical stability of coloring compounds (natural ones and coloring components produced during food processing). They can provoke the inhibition of pro-browning polyphenol-oxidase reactions by complexing with several substrates or cofactors (e.g., chlorogenic acid, polyphenols, cinnamic acid, Cu2+) [22]. Several studies have proven the utility of CDs in food science. For instance, a study showed that the natural pigments curcumin and lycopene can form a complex with CDs, improving their solubility and reducing the degree of oxidation compared to the compounds separately [26]. Another study, conducted with chopped ginger root, showed that by adding 1–4% of CDs, the sample could be enzymatic browning stabilized for four weeks at 5 °C while being vacuum sealed. A similar inhibitory effect was observed in another study carried out with maltosyl-β-CDs in apple and pear juices. The mechanism consists of preventing ascorbic acid oxidation by an antioxidant effect, which maintains the color and the food quality [5]. α-, β-, and γ-CDs (the only CDs allowed in food industry by the U.S. Food and Drug Administration and EU) are commonly used in the elaboration of different juices to improve the color of the final product. The addition of these molecules has other effects as they can also change the concentration of individual volatile molecules as well as their chemical grouping. In the case of pear juice, adding α-CDs was recommended. By adding this molecule, the overall quality of the juice was increased since browning reactions were reduced and no significant loss of aroma quality is produced [27].
3.1.2. Flavor
Flavoring substances are historic in food, although their direct use presents a series of disadvantages like having high volatility and sensitivity to light and heat. Part of these inconveniences can be solved with the CDs based encapsulation of food flavors, which is a frequent and simple solution to maintain the stability [22,24,28]. The price of the resulting β-CDs encapsulated flavor (USD $5–6 per kg) would not be much higher than other microencapsulated flavors price. CDs encapsulation provides an effective protection of each flavor component found in a multicomponent food system to whichever process it has been subjected (freezing, thawing, and/or microwaving). This factor is very important since the substances responsible for flavor usually involve numerous compounds, so it is interesting that all these molecules become part of the complex without seeing their organoleptic properties altered [18,29,30]. This method can be also used in oils to achieve a manipulability powder that can be added to food [22,24]. The liberation of aroma in these complexes can be controlled by slow guest liberation, mask off-notes of aromatic components by affinity with CDs cavity, and increase food flavor by water dissociation of aroma due to the polar external part of CDs [22]. Due to the properties of CDs-complexes, they can be applied to enhance flavor before extrusion, being a promising alternative for applying during the process [31]. As for the evolution of aromas over time, α-, β-, and γ-CDs are the best for initial flavor retention, α being better than γ for avoiding the loss of volatiles after storage [32,33]. Some examples of the application of CDs in the flavor and taste can be seen in Table 2.

Table 2.
Studies of cyclodextrins (CDs) for food flavor and taste improvement.
3.1.3. Taste
Bitterness is one of the factors that can generate the rejection of a food product. However, there are exceptions to this rule, as some products are expected to have a certain degree of bitterness, like coffee, beer, or wine [29]. By using appropriate CDs, the bitter taste of certain substances may be totally or partially eliminated since complexed compounds cannot react in the oral cavity with the taste buds. This type of taste is not perceived as only dissolved substances have flavor. This system has been applied to the bitter and astringent compounds of foods (e.g., soy), beverages (e.g., naringin in citrus juice or chlorogenic acid and polyphenols in coffee), cigarette smoke (nicotine), or oral care products or drugs [49].
The mechanism of action consists of forming complexes of enough stability with the selected CDs to make the substance, that gives the unwanted taste, insoluble in water and, therefore, in saliva, and do not cause a bad taste sensation. The effectiveness of the process will depend on the value of the complex association constant (usually 101–104 molK−1), pH (less stable complexes with ionized guest molecules), and guest/host ratio (the higher possible molar excess, the better) [49]. Finally, the complex is released throughout the digestive system. Considering this, CDs are one of the best methods for masking the unpleasant taste [22]. The most relevant publications dealing with the elimination of unwanted tastes focus on the positive effect of β-CDs, the possibilities of α-, γ-, hydroxypropyl-CDs, and maltosyl-CDs having not yet been explored. CDs can also be used in seafood and meat products, to improve texture [29]. Other cases of study of CDs for food taste improvement can be seen in Table 2.
3.2. Improving Shelf Life
CDs can protect several compounds present in foods from reactions such as oxidation, light induced reactions, heat promoted decomposition, self-decomposition, and loss through volatility or sublimation [24]. The encapsulation of CDs with lipophilic food ingredients, physically and chemically, improves the stability of flavors, vitamins, dyes, and unsaturated fats, among others. Consequently, the shelf life of the product will be increased [18]. As an example, an in vitro study demonstrated that CDs encapsulation improved the stability of rosemary bioactive compounds [24]. Different accelerated and long-term storage stability tests showed that ingredients complexed with CDs have a longer life than those traditionally formulated. Another study tested twelve different complexes with β-CDs stored for 14 years. The results showed that encapsulation resulted in a notable improvement of the stability during long-term storage. Its preserving power depends on factors that affect the host–guest union such as the structure, the polarity of the compounds, or their geometry. Different studies showed that the greatest protective effect is observed in flavors with terpenoid, phenylpropane, and alkylsulfide structures [18]. In a recent study, different nonalcoholic beverages used limonene complexes with α-, β-, and γ-CDs to improve flavor and shelf life. The study showed that although limonene content diminished in all cases, it did so to a lesser extent once β-CDs/limonene complexes were adjoined. After 10 days, which mimic nine months of storage, 40% of limonene complexed remained in the model drink [50].
3.2.1. Against Oxidation
CDs can form complexes with ingredients (flavors, unsaturated fatty acids, dyes, etc.) sensitive to oxygen or oxidizing substances, which in most cases leads to an improvement in the stability of encapsulated substrates. Several studies have shown that complexing by means of this type of compounds almost completely prevents these oxidizable substances from undergoing chemical modifications, even when warehoused in an atmosphere of 100% oxygen [18]. Another study showed that using cinnamon-CDs complexes in the manufacture of dried apple slices with cinnamon flavor not only prevented a decrease in the concentration of this compound due to evaporation, but also protected it from oxidation [51].
3.2.2. Against Light-Induced Decomposition
CDs can be also used to protect compounds of interest from deterioration factors such as light, heat, or oxidation. In addition, if the CDs cavity is filled, the entry of other molecules is prevented, so no unwanted reactions occur. Another mechanism of action is preventing reactive molecules from approaching the active sites of the host molecule. For instance, CDs have been used to protect vitamins and pharmaceutical products that contain easily oxidizable double bonds (e.g., prostaglandins). It has been demonstrated that hydroxypropyl-β-CDs protected peptides from hydrolysis and their consequent loss of ability [51].
3.2.3. Against Heat-Induced Changes
Another important problematic event is thermal degradation of natural compounds. In most cases, the application of heat causes the volatilization of less stable compounds, which might have interesting biological properties. One possible solution can be the encapsulation of bioactive compounds with CDs, resulting in a complex that would provide a barrier for preventing their loss [52]. Several studies have shown that these complexes are very useful when it comes to protecting volatile flavor compounds and essential oils against heat, generally achieving better flavor retention with CDs than with various traditional formulations [53]. For example, this system has been used in vitro for the encapsulation of vitamin A palmitate to produce enriched foods using β-CDs. As a result, there is an increase of both, in its solubility in aqueous media and in its stability against different external factors (temperature, light, and oxygen) [54]. Other examples of the application of these complexes to protect several compounds can be observed in Table 3.

Table 3.
Studies of cyclodextrins (CDs) for heat protection of food ingredients.
3.3. Modifying Solubility
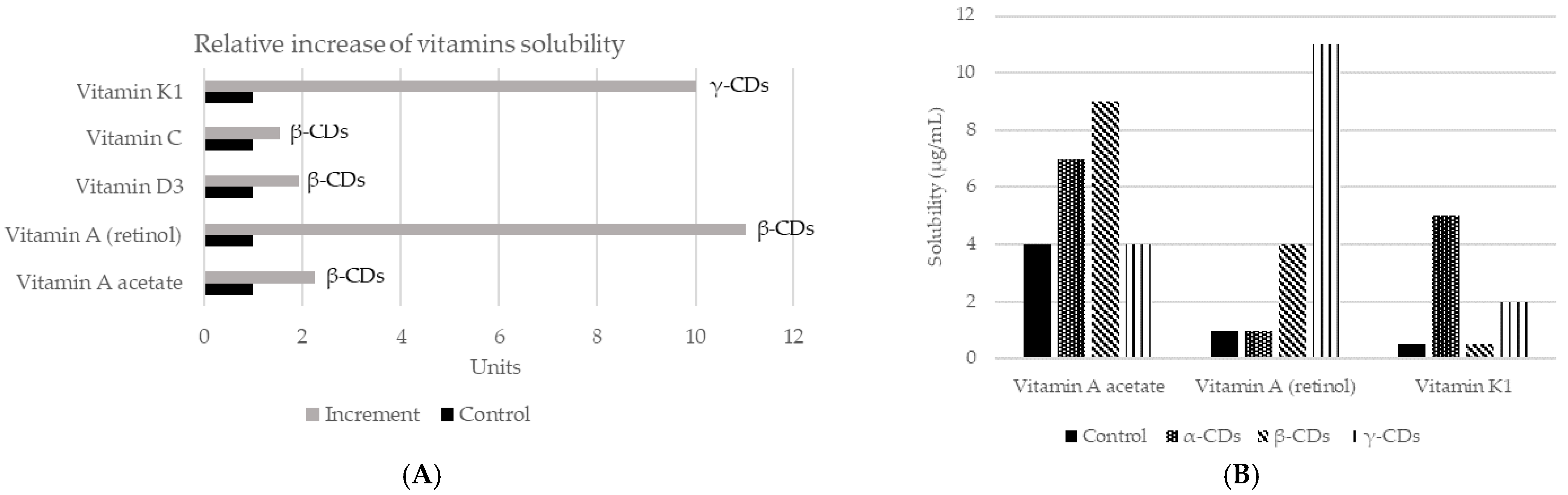
As mentioned before, CDs are capable of changing the solubility of a compound [67]. They have the ability to form stable emulsions of water in oil (e.g., mayonnaise, salad dressings), due to differences in polarity between the inside and outside of the molecule [18]. In addition, CDs can also increase the solubility of certain compounds in water, by forming dynamic, noncovalent, water-soluble inclusion complexes [68]. However, in many cases, the solubility of the complex is not appropriate, so it is necessary to modify the external surface of CDs. Neutral (hydroxypropyl) or ionic groups (hydroxy, carboxymethyl, tertiary amine, or quaternary amine) can be used to increase the solubility up to 60%. On the other hand, to improve solubility in organic solvents, the modification is carried out with aliphatic groups or smaller groups (hexyl, acetyl). Thus, complexation with CDs is a mechanism to increase or decrease the solubility of a guest component. A common example of its application is to reduce the bitterness in citrus juices, by creating naringin-β-CDs complex. In fact, the rate of transformation of naringin to naringenin in inclusion complexes or free can reach 98.7% and 56.2%, respectively. It might be concluded that β-CDs can improve the aqueous solubility, which also means that the rate of enzymatic hydrolysis of naringin will be increased [69]. The increase in solubility will also affect aromatic compounds, such as vanillin, used at the vanillin/β-CDs inclusion complex. This complex increased solubility in water with respect to the free compound. Moreover, the formation of this complex protects vanillin inside the CDs cavity, which avoids damage from several factors such as oxidation according to the results obtained in differential scanning calorimetry (DSC) studies. Therefore, the vanillin/β-CDs complex can be used as a food additive for its higher antioxidant activity [70]. CDs may be also applied to improve the solubility of vitamins (Figure 3). The union of β-CDs with the essential oil from guava leaves also increases solubility and stability. The resulting complex presented antioxidant (against light) and antibacterial (Staphylococcus aureus and Escherichia coli) activities [71]. Similar effects were achieved by encapsulation of black pepper essential oil or yarrow essential oil [72,73].
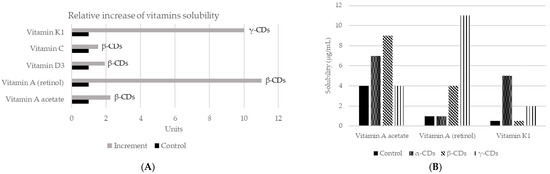
Figure 3.
Changes in solubility in water of different vitamins according to the use of different CDs. (A) Relative increase of the solubility of vitamins using different CDs complexes. Adapted from [74,75]. (B) Vitamins solubility (µg/mL) when the three main types of CDs applied in a concentration of 5%, using saline solution as control and measured by mass spectrometry [74]. More recently, new studies have assessed the solubility of vitamin E + large ring cyclodextrins [76] and vitamin A together with β-CDs [77].
3.4. Sequestration of Selected Components
Among the most recent CDs applications, reduction of unwanted compounds (flavor, trans-fats, allergens, toxins) is included [24]. Allergens can be avoided with β-CDs. It has been demonstrated that they form host–guest systems with allergenic aroma molecules (eugenol, isoeugenol, benzyl alcohol, or anisyl alcohol) and proteins [78,79]. An example of this possible application is the preparation of soybean milk of low allergenicity. They are also able of forming complexes with mycotoxins like ochratoxin A from cereals, coffee, beer, wine, and cocoa [80]. CDs have been demonstrated to sequestrate other mycotoxins, such as aflatoxin, ochratoxin, patulin, zearalenone, zearalenol, and citrinin [79]. A study used 1% β-CDs during apple juice processing to reduce mycotoxins (patulin) and inhibit enzymatic browning by 70% and 75%, respectively [81].
Other application of CDs is the sequestration of cholesterol from food products [24,82]. They can be applied in food with high content of these fatty acids to make it healthier. This is the case of milk, butter, and egg yolks [83]. In fact, this property is widely used in the industry to produce products without cholesterol since this compound is retained in the β-CDs cavity. CDs did also sequester reducing sugars capable of reacting with proteins (Maillard reaction) and that could give an undesirable color and adverse effects in the nervous system and fertility, being a possible carcinogen. The mechanism is based on the complexation of proteins with CDs, which protect them from reaction. They can also be applied to decrease acrylamide content in food products and food intermediates [84].
3.5. Pickering Emulsions
Pickering emulsions are strictly defined as emulsions that are stabilized by an adsorbed layer of solid particles at the emulsion drop surface [85]. Their properties are usually determined by particle size, particle wettability, particle concentration, oil/water ratio, pH, salt concentration, and solvent type [86]. CDs can form CDs-oil complexes, however, in most cases, high concentration of CDs is needed. To solve this inconvenience, modified CDs can be used such as soft colloidal CDs polymer (CDs nanogel) [87]. Thermal stability in water of this type of emulsions with different oils has been investigated, observing activity at room temperature and the dissolution/fusion of inclusion complexes with high melting temperatures (near to or higher than 100 °C) [86]. Another study has considered the union between β-CDs and octadecenylsuccinic anhydride (ODS) under alkaline conditions. ODS-β-CDs particles exhibited a higher emulsifying capacity compared to β-CDs. The resulting pickering emulsions formed by ODS-β-CDs particles were more stable during storage [88].
3.6. Other Food Applications
Apart from food processing, CDs also have other technological advantages such as improving nutritional properties and for developing nutraceutical products [18]. Several studies have been conducted in this aspect. α-CDs complex can be used to keep certain products (cereal, snacks) crunchy after storage and also as soluble dietary fiber in beverages and foods [30]. Other CDs complexes can be used in the preservation of food. They can be used indirectly in the prevention of microbial growth, being added in plastic packaging films. In this way, they preserve food during storage and also prevent loss of aroma [18]. Finally, CDs have been demonstrated to be useful to remove contaminants from food products, including herbicides, insecticides, fungicides, repellents, pheromones, and growth regulators [5].
4. Other Applications
4.1. Pharmaceutical Applications
CDs are used for numerous purposes in pharmaceutical applications (Table 4). In drug formulation, CDs have been described to increase the bioavailability, solubility, stability, reduce hemolysis and adverse effects, prevent admixture incompatibilities and act as excipients, among other uses [89,90,91] (Table 5). Improving the solubility of drugs is interesting, since the compound will have greater therapeutic efficacy, and lower doses will be necessary [68]. Numerous anticancer CDs-based drugs are being clinically evaluated [91]. CDs have been also used to delivery oligosaccharides, proteins, and oligonucleotides due to their capacity to interact with cellular membranes, improving cellular uptake. Another application is the delivery of gene-therapeutic agents, such as plasmids, viral vectors, and antisense constructs [92]. CDs have been demonstrated to protect carbamates in vitro, increasing their half-life [93]. Its ability to sequestrate determined compounds is also useful for pharmaceutical applications. CDs can sequester neuroactive steroids, which are potent modulators of GABAA receptors [94]. They can be used to elaborate pickering emulsions that can be used for topical applications in the formulation of antifungal econazole derivatives delivery [95].

Table 4.
Examples of some cyclodextrins (CDs) studied in pharmaceutical applications. Adapted from [87,96,97,98,99,100].

Table 5.
Example of some improved drug functions achieved by CDs complexation. Adapted from [89,101]
4.2. Cosmetics and Personal Care
CDs are also used is cosmetics [96]. Its application has numerous advantages, such as stabilizing compounds, obtaining odors and flavors of greater acceptability, improving the action of the compound by transforming a liquid constituent to a solid form, reducing vapor pressure, changing the solubility in water, and improving the thermal stability of oils, among others [6]. They are used in the suppression of the volatility of perfumes, air fresheners, and detergents since they allow a controlled release of fragrances from the host–guest complex, producing more doubtful fragrances [92]. They are also used in the formulation of toothpaste, skin creams, fabric softeners (liquid and solid), paper towels, tissues, and underarm shields. Therefore, CDs represent a valid formulation support, since they can improve the performance of the resulting product and solving problems that may arise during its formulation. Several studies have been performed to study different cosmetical applications of CDs. For instance, an in vitro study has proven that CDs are a useful delivery vehicle of ferulic acid (a compound with well-known antioxidant and photoprotective properties), improving its photo-stability, which could be an interesting property for cosmetic formulations [102].
4.3. Packing and Textile Industry
In recent years, the textile industry has directed its research towards making functional and sustainable fabrics [89]. In this field, the β-CDs can play a fundamental role since it can form complexes with different types of compounds, which makes a new wide variety of textile products and applications with advanced properties, such as antimicrobial or photoprotective. The incorporation of CDs to the textiles may also serve to deliver aromas and capture malodors (sweat, smoke) or increase the ability of fabrics to retain dyes with the consequential benefit of decreasing the amount lost in wastewater [89,92,103]. Moreover, they can also be flame-retardants [103]. In medicine, medical tissues containing CDs are used to release chemical compounds (both topically or inside the body) with beneficial properties, such as antibacterial, anti-allergic, antifungal, anti-inflammatory, and protection against insects [23]. Different studies seem to indicate that the most promising way to bond CDs in fabrics would be to generate complexes of monochlorotriazinyl-CDs by binding CDs with trichlorotriazines [104]. β-CDs can also be used as a novel molecular phosphorescent material as light sensitive phosphorescent color changes have been detected, which makes it a promising candidate as dynamically photo-functional material [105]. Other applications of CDs in fabrics and textiles can be seen in Table 6.

Table 6.
Examples of studies using cyclodextrins (CDs) system on textile surface.
4.4. Bioconversion and Fermentation
Bioconversion and fermentation processes are frequently limited due to the toxic or inflammatory effect produced by the substrate or product in the catalyst. In addition, the medium is also of great importance given that most of the organic substrates are lipophilic and, thus, they have low water solubility and the catalyst is usually more active. As a result, only a small part of the substrate is reachable to the biocatalyst [92]. Different techniques have been carried out to overcome these problems. Among them, there are the addition of the inhibitory substrate to the fed batch, in situ recovery of the inhibitory product or the solution of the lipophilic substrate with surfactants and organic solvents. The solution may be achieved by the use of CDs [120]. Some studies have used CDs to improve the production efficiency of different compounds. For instance, production efficiency of spiramycin was enhanced by CDs [121]. Modified β-CDs also increase the rate of deacetylation of spironolactone [122].
4.5. Environment
The role of this type of compounds in the environmental field is mainly due to its ability to solubilize organic pollutants, enhance and eliminate organic contaminants and heavy metals from the environment. In this field, CDs studies and new applications are expected to grow in the following years. β-CDs have been used in the adsorption of contaminants since they do not generate additional pollution [123]. Regarding water treatment, several possible applications have been described. An example of this application would be the treatment of textile wastewater contaminated with dyes [103]. CDs nanosponge adsorbents have been studied for water treatment. To achieve this, CDs nanosponges are modified with adsorbent nanomaterials (nanotubes made with carbon, titanium oxide, and silver nanoparticles). The obtained results proved the efficiency of these systems for removing contaminants from water [124].
4.6. Catalytic
In recent years, research efforts have been focused on the development of CDs capable of catalyzing organic reactions since they can bind to the substrate and form inclusion complexes with small compounds. CDs have been considered as artificial enzymes, generally characterized by having substrate specificity due to the structure and properties of the CDs cavity, even showing stereospecificity [125]. These complexes have been used for advanced homogeneous or heterogeneous catalytic processes. Moreover, they can be used to make alternative reaction media, such as hydrogels or low melting mixtures. These mixtures are capable of stabilizing active catalytic species, resulting in an increase of the catalytic activities and selectivity in transition metal reactions. In addition, after the catalysis is processed, artificial enzymes can be recovered by only a phase separation. With this catalytic systems process, safety is greatly improved [126]. Some advantages of CDs-based molecular catalysts are their easy preparation and isolation, economical and effortlessly obtainable starting materials, possibility of reuse and likelihood of acting in mild aqueous conditions (ecologically sustainable technology) [127].
A recent advance in this field consists of using CDs as an enzymatic mimic, since a molecular recognition phenomenon occurs due to the groups substituted in the CDs [92]. For example, β- or γ-CDs can be used in benzoin condensation with a rate increase of 7-fold [128]. Another example is a molecular catalyst of Pd/β-CDs, which shapes inclusion complexes with small organic compounds. It was utilized with remarkable results to reduce toxic aromatic components and the degradation of harmful dyes [129].
4.7. Analytical
CDs and their derivatives are utilized in a wide diversity of analytical chemistry fields, especially in analytical separations as they could distinguish between positional isomers, functional groups, homologues, and enantiomers [92]. They can be used in different areas such as chromatography, waste-water treatment, and other separation techniques (extraction, complex formation) [130]. For instance, they are used in chromatographic separations as they increase the selectivity in comparison to separations carried out with an eluent and stationary phase without any additional help [131]. Nevertheless, further development is necessary to reduce preparation costs, particularly for environmental applications. CDs could be also used as a reagent in several methods, including UV-visible spectrophotometry, photoluminescence and nuclear magnetic resonance [132]. This use is related to the ability of CDs to increase the emission intensity of the reactions that are taking place due to several aspects. Higher reaction rates and a greater efficiency in the process of excitation and protection of species that emit the quenching phenomena are some examples of these aspects [132].
5. Advantages and Disadvantages of Their Use
Using CDs complexes in food systems has several advantages, which have been explained relating them to their application in the industry. These advantages can be grouped in blocks. The first of these would be the increase of shelf life of the resulting products, due to the ability of CDs to protect compounds against different factors such as oxidation, light-induced reactions, decomposition, and thermal decomposition. Therefore, these types of molecules act as a potential stabilizing agent. For example, making possible its application to target orally administered water-insoluble drugs. They can also increase the stability of emulsions. The second block is related to physical properties changes, which has advantages such as increasing the solubility of different compounds. They also eliminate hygroscopicity, which is the ability to absorb moisture from the surrounding environment. Rheological properties as well as viscosity are also modified by the physical characteristics of these compounds. Other advantage is that its structure allows the union with a great variety of compounds, achieving even high order complexes. Moreover, as CDs have a well-defined chemical structure, they possess many potential sites for chemical modification or conjugation, giving rise to a great variety of complexes. In terms of their advantages at technological level, CDs are characterized for being stable, having simple dosing and being able to handle dry powders. This makes the reduction of packing and storage costs possible, resulting in more economical technological processes and manpower saving. Standardization allows products that do not vary over time to enter in the market, which means that they are more accepted by consumers. The ability to sequester unwanted compounds is also a big factor in terms of flavor. This quality is closely related to its application when it comes to improve sensory quality since CDs are able to eliminate or reduce undesired tastes and odors and preserve the desirable ones protecting them from loss by evaporation and sublimation. They can also form complexes that allow the development of new additives in order to improve important factors in the food industry such as color and flavor. In addition, CDs would satisfy consumers’ demand for more natural products, being one of the most promising compounds to use as additives to improve sensory quality.
Despite everything, they also present a series of drawbacks. Even though they are characterized for having a low toxicity, in some cases, it has been shown that they can have an irritating effect. Thus, there are safety concerns, which limit their use for parenteral administration showing renal toxicity in most species. However, despite these adverse effects, their toxicity as well as its immunogenicity remains low, which is the reason that CDs have extremely appealing pharmaceutical applications. In many cases, they can accelerate reactions, for example, the hydrolysis of esters, amides and organophosphates, decarboxylation and oxidation reactions. Furthermore, royalty payments may be required as many patents are still enforceable. In all cases, binding constants of the complex need to be optimum, especially in drugs for pharmacokinetics.
6. Toxicity and Legislation
When CDs were discovered, they were considered poisonous substances, so their application in complex formation was considered an anecdote. Afterwards, it was shown that they had no toxic effects and that they could also be very versatile for their protective properties [29]. β-CDs have been the most studied within the CDs, having proved its safety. At the maximum dose of β-CDs orally presented, no mortality has been observed. As a result, a nontoxic quantity of 650 mg/Kg/day (1.25% of diet) of β-CDs was designated in rats. In contrast, the injection of β-CDs resulted in the decease of the animals, as it caused kidney damage due to crystallization of the complex in the kidneys [51]. It is worth mentioning that only very little β-CDs were completely absorbed as salivary or pancreatic amylases had no effect. Therefore, the complex reached the colon without having undergone any modification and enzymes from the intestinal flora opened CDs ring generating a maltoheptaose. This compound is catabolized like the other starch fragments that reached the colon [51]. CDs are widely used in food since its applications include acting as a vehicle of ingredients (flavor, vitamins, polyunsaturated fatty acids, etc.) and as a stabilizer. The available data estimates an acceptable daily intake (ADI) for all its applications in foods of 4.1 g/person/day for consumers of foods containing γ-CDs. Several studies have been conducted to verify the risks of this consumption [133]. Different studies concluded that γ-CDs had no toxic effects and were well tolerated by the body. This may be because their similar metabolism to starch and linear dextrin, according to the results obtained in metabolic studies in rats (rapidly and digested by amylases present in saliva and pancreatic juice). Moreover, γ-CDs will not affect the absorption of lipophilic nutrients since inclusion complexes are reversible and they have a similar gastrointestinal tolerance of maltodextrin. All these studies included γ-CDs as a product generally recognized as safe (GRAS) for its use in food [134]. The acute oral LD50 was 8000 mg/kg for rats [51]. It should also be added that CDs, as well as their hydrophilic derivatives, can only penetrate lipophilic membranes with considerable difficulty. Consequently, its absorption in the gastrointestinal tract is very low, thus, its oral administration presents almost no toxicity [103]. The World Health Organization (WHO) concluded that, according to all available studies on α-, β-, and γ-CDs and their allocated ADIs, these food components contained an almost insignificant toxicity. Therefore, according to the available information, their consumption levels (total dietary intake) and their acceptable background levels in food, CDs do not entail a threat to health. Not showing cytotoxic effects is vitally important in applications such as food and flavors but also in cosmetics, packaging, textiles, separation processes, environmental protection, fermentation, and catalysis [5]. These types of compounds have not shown any allergic impact according to OECD experiments. Since 2000, β-CDs are commercialized in Germany as a food additive [103].
Many studies concerning the toxicity of CDs are based on their medical applications. Even though the safety aspects should have been considered during the development, and safety assessment of each medication should be clearly established on the data sheet, in practice, it does not occur. However, a high dose of CDs has adverse effects. Although its oral availability is very low, high doses can lead to reversible diarrhea and caecum enlargement. In addition, depending on the dose, the permeability of the tissues and, consequently, the bioavailability of the active substances administered, may also be altered. In cases of high systematic exposures in animals, nephrotoxic effects have been observed. So far, there is no evidence of these effects in humans [135,136,137]. Moreover, as there is not enough information about the effects of CDs in children under two years old, a case-by-case judgment regarding the risk/benefit to the patient must be performed. Nevertheless, CDs are not currently included in the European Commission Guide of excipients on the label and the package leaflet of medicinal products for anthropological use [138]. For example, the toxicity of hydroxypropyl-β-CDs (HP-β-CDs) was studied, i.e., a substance that has been extensively used as a solubilizing agent in the pharmaceutical industry for many years. Despite this, no studies on solubilizing capacity and toxicity have been conducted according to the degree of substitution. The obtained results showed that the best option for both solubility and toxicity would be HP-β-CDs with low degree of substitution (DS). However, further studies are still necessary since the comparison of HP-β-CDs toxicity with diverse DS must be executed in humans considering its dependence on the species.
The current legislation regarding CDs varies between Europe and other countries. Within Europe, as they are considered as food additives, they are subjected to the same legislation. In the USA α-, β-, and γ-CDs are considered a GRAS food additive. However, in Japan the three CDs are recognized as natural products. Therefore, their commercialization in the food sector will be only restricted by considerations of purity. Other countries in which α- and γ-CDs are considered food (novel foods) are Australia and New Zealand since more than fifteen years ago [30]. Despite α-, β-, and γ- CDs are being used as food additives in those countries, in Europe β-CDs has been approved as an additive (E-459) and lately α-CDs as a novel food. γ-CDs can be also used in the food industry; however, its use is restricted to those countries in which it has been approved. An outline of the state of the legislation worldwide can be seen in Table 7.

Table 7.
Summary of existing legislation.
According to different reports (1996) from the Scientific Committee on Food (SCF), the ADI of β-CDs (E 459) is 5 mg/kg of body weight per day. Giving the available reported use and use levels, the EFSA Panel also determined that the ADI was surpassed in the refined brand-loyal scenario (contemplated as the most relevant scenario) on average in whole population groups excluding babies and in all population groups in the 95th percentile [137]. According to data, FAO/WHO Expert Committee on Food Additives (JECFA) proposed a maximum level of β-CDs in foods of 5 mg/kg per day. Owing to their positive toxicological profiles, neither α- nor γ-CDs have a defined ADI. Furthermore, in July 2005 the U.S. Environmental Protection Agency (EPA) eliminated the need to create a maximum allowed level for residues of α-, β- and γ-CDs on several food supplies. Regarding the assessment of the levels of exposure on animal and human studies and associated adverse effects, it is considered that α-CDs have very low toxicity and that, at the proposed levels of dietary exposure when used as a food ingredient, would not have toxicological effects.
7. Future Perspectives and Conclusions
CDs and their derivatives have an extensive diversity of uses in various fields (food, cosmetics, and drugs), but especially on the food industry and, thus, their use have increased in recent years. These applications are mainly due to the capacity they possess to form host–guest complexes with a wide diversity of compounds. This type of molecular encapsulation improves the stability of flavors, vitamins, colorants, unsaturated fats, and other lipophilic molecules in physical and chemical senses leading to extended product shelf life. Furthermore, by using this technology, sensorial qualities can be improved, and microbiological contaminations can be avoided. In addition, due to their low toxicity, they can be applied without risk to human health, not only resulting in healthier and more functional products, but also less perishable. Cases of sold CDs-based food products for demonstration of the importance of CDs technology in the food industry were reviewed.
CDs are an exceptional type of building blocks in innovative molecular architecture due to their low toxicity, their capacity to hold, orient, conceal, modify their chirality, and isolate their guest compounds. All these properties make possible to use them not only as excipients, but also as extenders, chelating agents, or other multipurpose technological tools. Another application of great interest is the sequestration of toxic compounds, which makes them capable of modifying the toxicity of the substrate [24]. Their use in food, agriculture, pharmaceutical products, and chromatographic techniques has increased considerably in the last decade. Therefore, the unique architecture of CDs makes them a significant option in drug advance, in chiral separations and as complexing agents in food, cosmetics, and pharmaceutical manufactures. Among them, it is worth mentioning the scope of food since consumers’ demand more natural and healthy products with the consequent expansion of the market of functional foods and nutraceutical products. This can be achieved with the application of CDs, which has a promising future.
Another emerging use of CDs is as encapsulation agents at molecular level due to their ability to absorb whole molecules or part of them into their cavity. They can be the key to many future encapsulated formulation solutions. It is also expected that they will continue to be applied due to many other advantages, including increased solubility, stability against light, heat and oxidizing conditions, and decreased volatility. It must also be considered that not only pure CDs but also their derivatives can be used, which increases their possible applications. However, to carry out all these processes, a few main concerns are cost reduction and the efficiency of manufacturing. In recent years, there have been great advances in this regard, which makes it likely to increase their applications. In addition, several studies showed that the formulation of CDs derivatives have better stability than those of the traditionally formulated ones. The number of publications carried out in recent decades is a clear example of the growing interest in the potential applications of CDs.
Funding
The research leading to these results was funded by FEDER under the program Interreg V-A Spain-Portugal (POPTEC) 2014-2020 ref. 0377_IBERPHENOL_6_E and ref. 0181_NANOEATERS_01_E; to Xunta de Galicia supporting with the Axudas Conecta Peme the IN852A 2018/58 NeuroFood Project and the program EXCELENCIA-ED431F 2020/12; to Ibero-American Program on Science and Technology (CYTED - AQUA-CIBUS, P317RT0003) and by the Bio Based Industries Joint Undertaking (JU) under grant agreement No 888003 UP4HEALTH Project (H2020-BBI-JTI-2019), the JU receives support from the European Union’s Horizon 2020 research and innovation program and the Bio Based Industries Consortium.
Acknowledgments
The research leading to these results was supported by MICINN supporting the Ramón&Cajal grant for M.A.P. (RYC-2017-22891); by Xunta de Galicia and University of Vigo supporting the pre-doctoral grant for P.G.O. (ED481A-2019/295) and A.G.P. (ED481A-2019/0228) and to EcoChestnut Project (Erasmus+ KA202) that supports the work of M.C.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Wenz, G. An overview of host-guest chemistry and its application to nonsteroidal anti-inflammatory drugs. Clin. Drug Investig. 2000, 19, 21–25. [Google Scholar] [CrossRef]
- Cram, D.J.; Cram, J.M. Host-Guest Chemistry. Science 1974, 183, 803–809. [Google Scholar] [CrossRef] [PubMed]
- Lehn, J.M. Supramolecular chemistry: Receptors, catalysts, and carriers. Science 1985, 227, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Eastburn, S.D.; Tao, B.Y. Applications of modified cyclodextrins. Biotechnol. Adv. 1994, 12, 325–339. [Google Scholar] [CrossRef]
- Del Valle, E.M.M. Cyclodextrins and their uses: A review. Process Biochem. 2004, 39, 1033–1046. [Google Scholar] [CrossRef]
- Buschmann, H.J.; Schollmeyer, E. Applications of cyclodextrins in cosmetic products: A review. J. Cosmet. Sci. 2002, 53, 185–191. [Google Scholar]
- Shibatani, T. Industrial Application of Immobilized Biocatalysts in Japan. In Progress in Biotechnology; Elsevier: Amsterdam, The Netherlands, 1996; Volume 11, pp. 585–591. [Google Scholar]
- Chaudhari, P.; Ghate, V.M.; Lewis, S.A. Supramolecular cyclodextrin complex: Diversity, safety, and applications in ocular therapeutics. Exp. Eye Res. 2019, 189, 107829. [Google Scholar] [CrossRef]
- Bortolus, P.; Grabner, G.; Köhler, G.; Monti, S. Photochemistry of cyclodextrin host-guest complexes. Coord. Chem. Rev. 1993, 125, 261–268. [Google Scholar] [CrossRef]
- Da Hu, Q.; Tang, G.P.; Chu, P.K. Cyclodextrin-based host-guest supramolecular nanoparticles for delivery: From design to applications. Acc. Chem. Res. 2014, 47, 2017–2025. [Google Scholar] [CrossRef]
- Wenz, G. Cyclodextrins as Building Blocks for Supramolecular Structures and Functional Units. Angew. Chem. Int. Ed. Engl. 1994, 33, 803–822. [Google Scholar] [CrossRef]
- Matencio, A.; Navarro-Orcajada, S.; García-Carmona, F.; López-Nicolás, J.M. Applications of cyclodextrins in food science. A review. Trends Food Sci. Technol. 2020, 104, 132–143. [Google Scholar] [CrossRef]
- Astray, G.; Mejuto, J.C.; Simal-Gandara, J. Latest developments in the application of cyclodextrin host-guest complexes in beverage technology processes. Food Hydrocoll. 2020, 106, 105882. [Google Scholar] [CrossRef]
- Tian, B.; Xiao, D.; Hei, T.; Ping, R.; Hua, S.; Liu, J. The application and prospects of cyclodextrin inclusion complexes and polymers in the food industry: A review. Polym. Int. 2020, 69, 597–603. [Google Scholar] [CrossRef]
- Braga, S.S. Cyclodextrins: Emerging medicines of the new millennium. Biomolecules 2019, 9, 801. [Google Scholar] [CrossRef] [PubMed]
- Almagro, L.; Pedreño, M.Á. Use of cyclodextrins to improve the production of plant bioactive compounds. Phytochem. Rev. 2020, 19, 1061–1080. [Google Scholar] [CrossRef]
- Szejtli, J. Introduction and general overview of cyclodextrin chemistry. Chem. Rev. 1998, 98, 1743–1754. [Google Scholar] [CrossRef]
- Szente, L.; Szejtli, J. Cyclodextrins as food ingredients. Trends Food Sci. Technol. 2004, 15, 137–142. [Google Scholar] [CrossRef]
- De Menezes, P.P.; de Andrade, T.A.; Frank, L.A.; de Souza, E.P.B.S.S.; das Trindade, G.G.G.; Trindade, I.A.S.; Serafini, M.R.; Guterres, S.S.; de Araújo, A.A.S. Advances of nanosystems containing cyclodextrins and their applications in pharmaceuticals. Int. J. Pharm. 2019, 559, 312–328. [Google Scholar] [CrossRef]
- Kurkov, S.V.; Loftsson, T. Cyclodextrins. Int. J. Pharm. 2013, 453, 167–180. [Google Scholar] [CrossRef]
- Bruns, C.J. Exploring and exploiting the symmetry-breaking effect of cyclodextrins in mechanomolecules. Symmetry 2019, 11, 1249. [Google Scholar] [CrossRef]
- Linde, G.A.; Laverde, A.; Colauto, N.B. Changes to Taste Perception in the Food Industry: Use of Cyclodextrins. In Handbook of Behavior, Food and Nutrition; Preedy, V.R., Watson, R.R., Martin, C.R., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 99–118. [Google Scholar]
- Radu, C.-D.; Parteni, O.; Ochiuz, L. Applications of cyclodextrins in medical textiles. J. Control. Release 2016, 224, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, C.; Buera, P.; Mazzobre, F. Novel trends in cyclodextrins encapsulation. Applications in food science. Curr. Opin. Food Sci. 2017, 16, 106–113. [Google Scholar] [CrossRef]
- Roriz, C.L.; Barros, L.; Prieto, M.A.; Barreiro, M.F.; Morales, P.; Ferreira, I.C.F.R. Modern extraction techniques optimized to extract betacyanins from Gomphrena globosa L. Ind. Crops Prod. 2017, 105, 29–40. [Google Scholar] [CrossRef]
- Blanch, G.P.; Ruiz del Castillo, M.L.; del Mar Caja, M.; Pérez-Méndez, M.; Sánchez-Cortés, S. Stabilization of all-trans-lycopene from tomato by encapsulation using cyclodextrins. Food Chem. 2007, 105, 1335–1341. [Google Scholar] [CrossRef]
- Andreu-Sevilla, A.J.; López-Nicolás, J.M.; Carbonell-Barrachina, Á.A.; García-Carmona, F. Comparative Effect of the Addition of α-, β-, or γ-Cyclodextrin on Main Sensory and Physico-Chemical Parameters. J. Food Sci. 2011, 76, S347–S353. [Google Scholar] [CrossRef] [PubMed]
- Reineccius, T.A.; Reineccius, G.A.; Peppard, T.L. Flavor release from cyclodextrin complexes: Comparison of alpha, beta, and gamma types. J. Food Sci. 2003, 68, 1234–1239. [Google Scholar] [CrossRef]
- Astray, G.; Gonzalez-Barreiro, C.; Mejuto, J.C.; Rial-Otero, R.; Simal-Gándara, J. A review on the use of cyclodextrins in foods. Food Hydrocoll. 2009, 23, 1631–1640. [Google Scholar] [CrossRef]
- Cravotto, G.; Binello, A.; Baranelli, E.; Carraro, P.; Trotta, F. Cyclodextrins as food additives and in food processing. Curr. Nutr. Sci. 2006, 2, 343–350. [Google Scholar] [CrossRef]
- Yuliani, S.; Bhandari, B.; Rutgers, R.; D’Arcy, B. Application of microencapsulated flavor to extrusion product. Food Rev. Int. 2004, 20, 163–185. [Google Scholar] [CrossRef]
- Reineccius, T.A.; Reineccius, G.A.; Peppard, T.L. Encapsulation of flavors using cyclodextrins: Comparison of flavor retention in alpha, beta, and gamma types. J. Food Sci. 2002, 67, 3271–3279. [Google Scholar] [CrossRef]
- Astray, G.; Mejuto, J.C.; Morales, J.; Rial-Otero, R.; Simal-Gándara, J. Factors controlling flavors binding constants to cyclodextrins and their applications in foods. Food Res. Int. 2010, 43, 1212–1218. [Google Scholar] [CrossRef]
- Yoshii, H.; Yasuda, M.; Furuta, T.; Kuwahara, H.; Ohkawara, M.; Linko, P. Retention of cyclodextrin complexed shiitake (Lentinus edodes) flavors with spray drying. Dry. Technol. 2005, 23, 1205–1215. [Google Scholar] [CrossRef]
- Tobitsuka, K.; Miura, M.; Kobayashi, S. Retention of a European pear aroma model mixture using different types of saccharides. J. Agric. Food Chem. 2006, 54, 5069–5076. [Google Scholar] [CrossRef] [PubMed]
- Pagington, J.S. β-cyclodextrin and its uses in the flavour industry. In Developments in Food Flavours; Birch, G.G., Lindley, M.G., Eds.; Elsevier: Amsterdam, The Netherlands, 1986. [Google Scholar]
- Hedges, A.R.; Shieh, W.J.; Sikorski, C.T. Use of Cyclodextrins for Encapsulation in the Use and Treatment of Food Products. In Encapsulation and Controlled Release of Food Ingredients; ACS Publications: Washington, DC, USA, 1995; Volume 590, pp. 60–71. [Google Scholar]
- Bhandari, B.; D’Arcy, B.; Young, G. Flavour retention during high temperature short time extrusion cooking process: A review. Int. J. Food Sci. Technol. 2001, 36, 453–461. [Google Scholar] [CrossRef]
- Jouquand, C.; Ducruet, V.; Giampaoli, P. Partition coefficients of aroma compounds in polysaccharide solutions by the phase ratio variation method. Food Chem. 2004, 85, 467–474. [Google Scholar] [CrossRef]
- Kollengode, A.N.R.; Hanna, M.A. Cyclodextrin complexed flavors retention in extruded starches. J. Food Sci. 1997, 62, 1057–1060. [Google Scholar] [CrossRef]
- Young, O.A.; Gupta, R.B.; Sadooghy-Saraby, S. Effect of cyclodextrins on the flavour of goat milk and its yoghurt. J. Food Sci. 2012, 77, 122–127. [Google Scholar] [CrossRef]
- Reineccius, T.A.; Reineccius, G.A.; Peppard, T.L. Utilization of β-Cyclodextrin for Improved Flavor Retention in Thermally Processed Foods. J. Food Sci. 2004, 69, FCT58–FCT62. [Google Scholar] [CrossRef]
- Linde, G.A.; Junior, A.L.; de Faria, E.V.; Colauto, N.B.; de Moraes, F.F.; Zanin, G.M. Taste modification of amino acids and protein hydrolysate by α-cyclodextrin. Food Res. Int. 2009, 42, 814–818. [Google Scholar] [CrossRef]
- Binello, A.; Robaldo, B.; Barge, A.; Cavalli, R.; Cravotto, G. Synthesis of cyclodextrin-based polymers and their use as debittering agents. J. Appl. Polym. Sci. 2008, 107, 2549–2557. [Google Scholar] [CrossRef]
- Choi, M.J.; Ruktanonchai, U.; Soottitantawat, A.; Min, S.G. Morphological characterization of encapsulated fish oil with β-cyclodextrin and polycaprolactone. Food Res. Int. 2009, 42, 989–997. [Google Scholar] [CrossRef]
- Lee, S.H.; Yu, H.J.; Cho, N.S.; Park, J.H.; Kim, T.H.; Kim, K.H.; Lee, S.K. A method for preparing the inclusion complex of ginseng extract with gamma-cyclodextrin, and the composition comprising the same. In International Patent Classification IPC A23L; IPC Publication: Geneva, Switzerland, 2006; pp. 1–212. [Google Scholar]
- Shaw, P.E.; Tatum, J.H.; Wilson, C.W. Improved Flavor of Navel Orange and Grapefruit Juices by Removal of Bitter Components with β-Cyclodextrin Polymer. J. Agric. Food Chem. 1984, 32, 832–836. [Google Scholar] [CrossRef]
- Binello, A.; Cravotto, G.; Nano, G.M.; Spagliardi, P. Synthesis of chitosan-cyclodextrin adducts and evaluation of their bitter-masking properties. Flavour Fragr. J. 2004, 19, 394–400. [Google Scholar] [CrossRef]
- Szejtli, J.; Szente, L. Elimination of bitter, disgusting tastes of drugs and foods by cyclodextrins. Eur. J. Pharm. Biopharm. 2005, 61, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Saldanha do Carmo, C.; Pais, R.; Simplício, A.L.; Mateus, M.; Duarte, C.M.M. Improvement of Aroma and Shelf-Life of Non-alcoholic Beverages Through Cyclodextrins-Limonene Inclusion Complexes. Food Bioprocess Technol. 2017, 10, 1297–1309. [Google Scholar] [CrossRef]
- Hedges, A.R. Cyclodextrins: Properties and Applications. In Starch; Academic Press: Cambridge, MA, USA, 2009; pp. 833–851. ISBN 9780127462752. [Google Scholar]
- Capelezzo, A.P.; Mohr, L.C.; Dalcanton, F.; de Mello, J.M.M.; Fiori, M.A. β-Cyclodextrins as Encapsulating Agents of Essential Oils. In Cyclodextrin—A Versatile Ingredient; IntechOpen: London, UK, 2018. [Google Scholar]
- Furuta, T.; Yoshii, H.; Fujimoto, T.; Yasunishi, A.; Linko, Y.-Y.; Linko, P. A Short-Cut Method for Estimating l-Menthol Retention Included in Cyclodextrin During Drying a Single Drop. In Proceedings of the Eighth International Symposium on Cyclodextrins; Springer: Berlin/Heidelberg, Germany, 1996; pp. 583–586. [Google Scholar]
- Vilanova, N.; Solans, C. Vitamin A Palmitate-β-cyclodextrin inclusion complexes: Characterization, protection and emulsification properties. Food Chem. 2015, 175, 529–535. [Google Scholar] [CrossRef]
- Abarca, R.L.; Rodríguez, F.J.; Guarda, A.; Galotto, M.J.; Bruna, J.E. Characterization of beta-cyclodextrin inclusion complexes containing an essential oil component. Food Chem. 2016, 196, 968–975. [Google Scholar] [CrossRef]
- Fernandes, A.; Rocha, M.A.A.; Santos, L.M.N.B.F.; Brás, J.; Oliveira, J.; Mateus, N.; de Freitas, V. Blackberry anthocyanins: β-Cyclodextrin fortification for thermal and gastrointestinal stabilization. Food Chem. 2018, 245, 426–431. [Google Scholar] [CrossRef]
- Zhou, Q.; Wei, X.; Dou, W.; Chou, G.; Wang, Z. Preparation and characterization of inclusion complexes formed between baicalein and cyclodextrins. Carbohydr. Polym. 2013, 95, 733–739. [Google Scholar] [CrossRef]
- Hǎdǎruga, D.I.; Ünlüsayin, M.; Gruia, A.T.; Birǎu, C.; Rusu, G.; Hǎdǎruga, N.G. Thermal and oxidative stability of Atlantic salmon oil (Salmo salar L.) and complexation with β-cyclodextrin. Beilstein J. Org. Chem. 2016, 12, 179–191. [Google Scholar] [CrossRef]
- Li, W.; Liu, X.; Yang, Q.; Zhang, N.; Du, Y.; Zhu, H. Preparation and characterization of inclusion complex of benzyl isothiocyanate extracted from papaya seed with β-cyclodextrin. Food Chem. 2015, 184, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Hădărugă, D.; Hădărugă, N.; Costescu, C.; David, I.; Gruia, A. Thermal and oxidative stability of the Ocimum basilicum L. essential oil/β-cyclodextrin supramolecular system. Beilstein J. Org. Chem. 2014, 10, 2809–2820. [Google Scholar] [CrossRef] [PubMed]
- Kalogeropoulos, N.; Yannakopoulou, K.; Gioxari, A.; Chiou, A.; Makris, D.P. Polyphenol characterization and encapsulation in β-cyclodextrin of a flavonoid-rich Hypericum perforatum (St John’s wort) extract. LWT—Food Sci. Technol. 2010, 43, 882–889. [Google Scholar] [CrossRef]
- Hadaruga, D.I.; Hadaruga, N.G.; Rivis, A.; Gruia, A.; Pinzaru, I.A. Thermal and oxidative stability of the Allium sativum L. bioactive compounds/α- and β-cyclodextrin nanoparticles. Rev. Chim. 2007, 58, 1009–1015. [Google Scholar]
- Wang, J.; Cao, Y.; Sun, B.; Wang, C. Physicochemical and release characterisation of garlic oil-β- cyclodextrin inclusion complexes. Food Chem. 2011, 127, 1680–1685. [Google Scholar] [CrossRef]
- Hill, L.E.; Gomes, C.; Taylor, T.M. Characterization of beta-cyclodextrin inclusion complexes containing essential oils (trans-cinnamaldehyde, eugenol, cinnamon bark, and clove bud extracts) for antimicrobial delivery applications. LWT—Food Sci. Technol. 2013, 51, 86–93. [Google Scholar] [CrossRef]
- Cetin Babaoglu, H.; Bayrak, A.; Ozdemir, N.; Ozgun, N. Encapsulation of clove essential oil in hydroxypropyl beta-cyclodextrin for characterization, controlled release, and antioxidant activity. J. Food Process. Preserv. 2017, 41, e13202. [Google Scholar] [CrossRef]
- Kayaci, F.; Sen, H.S.; Durgun, E.; Uyar, T. Functional electrospun polymeric nanofibers incorporating geraniol-cyclodextrin inclusion complexes: High thermal stability and enhanced durability of geraniol. Food Res. Int. 2014, 62, 424–431. [Google Scholar] [CrossRef]
- Rakmai, J.; Cheirsilp, B.; Cid, A.; Torrado-Agrasar, A.; Mejuto, J.C.; Simal-Gandara, J. Encapsulation of Essential Oils by Cyclodextrins: Characterization and Evaluation. In Cyclodextrin—A Versatile Ingredient; IntechOpen: London, UK, 2018. [Google Scholar]
- Sharma, N.; Baldi, A. Exploring versatile applications of cyclodextrins: An overview. Drug Deliv. 2016, 23, 739–757. [Google Scholar] [CrossRef]
- Cui, L.; Zhang, Z.H.; Sun, E.; Jia, X. Bin Effect of β-cyclodextrin complexation on solubility and enzymatic conversion of naringin. Int. J. Mol. Sci. 2012, 13, 14251–14261. [Google Scholar] [CrossRef]
- Karathanos, V.T.; Mourtzinos, I.; Yannakopoulou, K.; Andrikopoulos, N.K. Study of the solubility, antioxidant activity and structure of inclusion complex of vanillin with β-cyclodextrin. Food Chem. 2007, 101, 652–658. [Google Scholar] [CrossRef]
- Rakmai, J.; Cheirsilp, B.; Mejuto, J.C.; Simal-Gándara, J.; Torrado-Agrasar, A. Antioxidant and antimicrobial properties of encapsulated guava leaf oil in hydroxypropyl-beta-cyclodextrin. Ind. Crops Prod. 2018, 111, 219–225. [Google Scholar] [CrossRef]
- Rakmai, J.; Cheirsilp, B.; Mejuto, J.C.; Torrado-Agrasar, A.; Simal-Gándara, J. Physico-chemical characterization and evaluation of bio-efficacies of black pepper essential oil encapsulated in hydroxypropyl-beta-cyclodextrin. Food Hydrocoll. 2017, 65, 157–164. [Google Scholar] [CrossRef]
- Rakmai, J.; Cheirsilp, B.; Torrado-Agrasar, A.; Simal-Gándara, J.; Mejuto, J.C. Encapsulation of yarrow essential oil in hydroxypropyl-beta-cyclodextrin: Physiochemical characterization and evaluation of bio-efficacies. CyTA—J. Food 2017, 15, 409–417. [Google Scholar] [CrossRef]
- Alpha-Cyclodextrin as a Novel Food. Available online: https://www.foodstandards.gov.au/code/applications/documents/A494_Alpha_cylclodextrin_FAR_FINAL.pdf (accessed on 26 January 2021).
- Braithwaite, M.C.; Kumar, P.; Choonara, Y.E.; du Toit, L.C.; Tomar, L.K.; Tyagi, C.; Pillay, V. A novel multi-tiered experimental approach unfolding the mechanisms behind cyclodextrin-vitamin inclusion complexes for enhanced vitamin solubility and stability. Int. J. Pharm. 2017, 532, 90–104. [Google Scholar] [CrossRef] [PubMed]
- Kuttiyawong, K.; Saehu, S.; Ito, K.; Pongsawasdi, P. Synthesis of large-ring cyclodextrin from tapioca starch by amylomaltase and complex formation with Vitamin E acetate for solubility enhancement. Process Biochem. 2015, 50, 2168–2176. [Google Scholar] [CrossRef]
- Xu, X.; Peng, S.; Bao, G.; Zhang, H.; Yin, C. β-cyclodextrin inclusion complexes with vitamin A and its esters: A comparative experimental and molecular modeling study. J. Mol. Struct. 2021, 1223, 129001. [Google Scholar] [CrossRef]
- Decock, G.; Fourmentin, S.; Surpateanu, G.G.; Landy, D.; Decock, P.; Surpateanu, G. Experimental and theoretical study on the inclusion compounds of aroma components with β-cyclodextrins. Supramol. Chem. 2006, 18, 477–482. [Google Scholar] [CrossRef]
- Fenyvesi, E.; Vikmon, M.; Szente, L. Cyclodextrins in Food Technology and Human Nutrition: Benefits and Limitations. Crit. Rev. Food Sci. Nutr. 2016, 56, 1981–2004. [Google Scholar] [CrossRef]
- Verrone, R.; Catucci, L.; Cosma, P.; Fini, P.; Agostiano, A.; Lippolis, V.; Pascale, M. Effect of β-cyclodextrin on spectroscopic properties of ochratoxin a in aqueous solution. J. Incl. Phenom. Macrocycl. Chem. 2007, 57, 475–479. [Google Scholar] [CrossRef]
- Essa, H.A.; Ayesh, A.M. Mycotoxins Reduction and Inhibition of Enzymatic Browning during Apple Juice Processing. Available online: https://www.kau.edu.sa/Files/857/Researches/58183_28330.pdf (accessed on 26 January 2021).
- Dos Santos, C.; Buera, M.P.; Mazzobre, M.F. Phase solubility studies and stability of cholesterol/β-cyclodextrin inclusion complexes. J. Sci. Food Agric. 2011, 91, 2551–2557. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.M.; Awad, A.C.; Bennink, M.R.; Gill, J.L. Cholesterol Reduction in Liquid Egg Yolk using β-Cyclodextrin. J. Food Sci. 1995, 60, 691–694. [Google Scholar] [CrossRef]
- Plank, D.W.; Novak, D.J. Method for Reducing Acrylamide Levels in Food Products and Food Products Produced Thereby. U.S. Patent No. 7,264,838, 4 September 2007. [Google Scholar]
- Murray, B.S. Pickering emulsions for food and drinks. Curr. Opin. Food Sci. 2019, 27, 57–63. [Google Scholar] [CrossRef]
- Diaz-Salmeron, R.; Chaab, I.; Carn, F.; Djabourov, M.; Bouchemal, K. Pickering emulsions with α-cyclodextrin inclusions: Structure and thermal stability. J. Colloid Interface Sci. 2016, 482, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Kawano, S.; Kida, T.; Akashi, M.; Sato, H.; Shizuma, M.; Ono, D. Preparation of Pickering emulsions through interfacial adsorption by soft cyclodextrin nanogels. Beilstein J. Org. Chem. 2015, 11, 2355–2364. [Google Scholar] [CrossRef] [PubMed]
- Xi, Y.; Luo, Z.; Lu, X.; Peng, X. Modulation of Cyclodextrin Particle Amphiphilic Properties to Stabilize Pickering Emulsion. J. Agric. Food Chem. 2018, 66, 228–237. [Google Scholar] [CrossRef]
- Singh, N.; Sahu, O. Sustainable cyclodextrin in textile applications. In The Impact and Prospects of Green Chemistry for Textile Technology; Elsevier Ltd.: Amsterdam, The Netherlands, 2019; pp. 83–105. ISBN 9780081024911. [Google Scholar]
- Davis, M.E.; Brewster, M.E. Cyclodextrin-based pharmaceutics: Past, present and future. Nat. Rev. Drug Discov. 2004, 3, 1023–1035. [Google Scholar] [CrossRef]
- Tian, B.; Hua, S.; Liu, J. Cyclodextrin-based delivery systems for chemotherapeutic anticancer drugs: A review. Carbohydr. Polym. 2020, 232, 115805. [Google Scholar] [CrossRef]
- Sing, M.; Sharma, R.; Banerjee, U.C. Biotechnological applications of cyclodextrins. Biotechnol. Adv. 2002, 20, 341–359. [Google Scholar] [CrossRef]
- Cid, A.; Astray, G.; Morales, J.; Mejuto, J.C.; Simal-Gándara, J. Influence of b-Cyclodextrins upon the Degradation of Carbofuran Derivatives. J. Pestic. Biofertil. 2018, 1, 1–4. [Google Scholar] [CrossRef]
- Shu, H.J.; Zeng, C.M.; Wang, C.; Covey, D.F.; Zorumski, C.F.; Mennerick, S. Cyclodextrins sequester neuroactive steroids and differentiate mechanisms that rate limit steroid actions. Br. J. Pharmacol. 2007, 150, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Leclercq, L.; Nardello-Rataj, V. Pickering emulsions based on cyclodextrins: A smart solution for antifungal azole derivatives topical delivery. Eur. J. Pharm. Sci. 2016, 82, 126–137. [Google Scholar] [CrossRef] [PubMed]
- Challa, R.; Ahuja, A.; Ali, J.; Khar, R.K. Cyclodextrins in drug delivery: An updated review. AAPS PharmSciTech 2005, 6, E329–E357. [Google Scholar] [CrossRef] [PubMed]
- Vikas, Y.; Sandeep, K.; Braham, D.; Manjusha, C.; Budhwar, V. Cyclodextrin Complexes: An Approach to Improve the Physicochemical Properties of Drugs and Applications of Cyclodextrin Complexes. Asian J. Pharm. 2018, 12, 394. [Google Scholar]
- Palem, C.R.; Siva Chaitanya Chopparapu, K.; Subrahmanyam, P.V.R.S.; Yamsani, M.R. Cyclodextrins and their derivatives in drug delivery: A review. Curr. Trends Biotechnol. Pharm. 2012, 6, 255–279. [Google Scholar]
- Lakkakula, J.R.; Maçedo Krause, R.W. A vision for cyclodextrin nanoparticles in drug delivery systems and pharmaceutical applications. Nanomedicine 2014, 9, 877–894. [Google Scholar] [CrossRef]
- Brewster, M.E.; Loftsson, T. Cyclodextrins as pharmaceutical solubilizers. Adv. Drug Deliv. Rev. 2007, 59, 645–666. [Google Scholar] [CrossRef]
- Tiwari, G.; Tiwari, R.; Rai, A. Cyclodextrins in delivery systems: Applications. J. Pharm. Bioallied Sci. 2010, 2, 72. [Google Scholar] [CrossRef]
- Centini, M.; Maggiore, M.; Casolaro, M.; Andreassi, M.; Maffei Facino, R.; Anselmi, C. Cyclodextrins as cosmetic delivery systems. J. Incl. Phenom. Macrocycl. Chem. 2007, 57, 109–112. [Google Scholar] [CrossRef]
- Voncina, B. Application of Cyclodextrins in Textile Dyeing. In Textile Dyeing; IntechOpen: London, UK, 2011. [Google Scholar]
- Grechin, A.G.; Buschmann, H.J.; Schollmeyer, E. Quantification of Cyclodextrins Fixed onto Cellulose Fibers. Text. Res. J. 2007, 77, 161–164. [Google Scholar] [CrossRef]
- Yong, G.P.; Zhang, B.; Zhang, Y.M.; Li, G.S. Excitation-light-responsive phosphorescent color changes in a β-cyclodextrin inclusion complex. J. Mater. Chem. 2012, 22, 13481–13483. [Google Scholar] [CrossRef]
- Ibrahim, N.A.; Abdalla, W.A.; El-Zairy, E.M.R.; Khalil, H.M. Utilization of monochloro-triazine β-cyclodextrin for enhancing printability and functionality of wool. Carbohydr. Polym. 2013, 92, 1520–1529. [Google Scholar] [CrossRef] [PubMed]
- Ursache, M.; Loghin, C.; Mureşan, R.; Cerempei, A.; Mureşan, A. Investigation on the effects of antibacterial finishes on dyed cotton knitted fabrics. Tekst. Konfeksiyon 2011, 21, 249–256. [Google Scholar]
- Wang, J.H.; Cai, Z. Incorporation of the antibacterial agent, miconazole nitrate into a cellulosic fabric grafted with β-cyclodextrin. Carbohydr. Polym. 2008, 72, 695–700. [Google Scholar] [CrossRef]
- Popescu, O.; Dunca, S.; Grigoriu, A. Antibacterial action of silver applied on cellulose fibers grafted with monochlorotriazinyl-β-cyclodextrin. Cellul. Chem. Technol. 2013, 47, 247–255. [Google Scholar]
- Abdel-Halim, E.S.; Al-Deyab, S.S.; Alfaifi, A.Y.A. Cotton fabric finished with β-cyclodextrin: Inclusion ability toward antimicrobial agent. Carbohydr. Polym. 2014, 102, 550–556. [Google Scholar] [CrossRef]
- Bajpai, M.; Gupta, P.; Bajpai, S.K. Silver(I) ions loaded cyclodextrin-grafted-cotton fabric with excellent antimicrobial property. Fibers Polym. 2010, 11, 8–13. [Google Scholar] [CrossRef]
- Racu, C.; Cogeanu, A.M.; Diaconescu, R.M.; Grigoriu, A. Antimicrobial treatments of hemp fibers grafted with β-cyclodextrin derivatives. Text. Res. J. 2012, 82, 1317–1328. [Google Scholar] [CrossRef]
- Uekama, K. Recent aspects of pharmaceutical application of cyclodextrins. J. Incl. Phenom. 2002, 44, 3–7. [Google Scholar] [CrossRef]
- Masajtis, J.; Dutkiewicz, J.; Członka, R.; Broniarczyk-Dyła, G.; Arkuszewska, C.; Skwarczyńska-Banyś, E.; Omulecki, A.; Kot, P. Correlations between the properties and effects of clinical therapy using textile material with antipsoriatic action. Polim. Med. 1992, 22, 26–41. [Google Scholar]
- Nichifor, M.; Constantin, M.; Mocanu, G.; Fundueanu, G.; Branisteanu, D.; Costuleanu, M.; Radu, C.D. New multifunctional textile biomaterials for the treatment of leg venous insufficiency. J. Mater. Sci. Mater. Med. 2009, 20, 975–982. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.Z.; Williams, G.R.; Hou, X.X.; Zhu, L.M. Electrospun curcumin-loaded fibers with potential biomedical applications. Carbohydr. Polym. 2013, 94, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Mohdy, F.A.; Fouda, M.M.G.; Rehan, M.F.; Aly, A.S. Repellency of controlled-release treated cotton fabrics based on cypermethrin and prallethrin. Carbohydr. Polym. 2008, 73, 92–97. [Google Scholar] [CrossRef]
- Romi, R.; Lo Nostro, P.; Bocci, E.; Ridi, F.; Baglioni, P. Bioengineering of a cellulosic fabric for insecticide delivery via grafted cyclodextrin. Biotechnol. Prog. 2005, 21, 1724–1730. [Google Scholar] [CrossRef]
- Hebeish, A.; Fouda, M.M.G.; Hamdy, I.A.; EL-Sawy, S.M.; Abdel-Mohdy, F.A. Preparation of durable insect repellent cotton fabric: Limonene as insecticide. Carbohydr. Polym. 2008, 74, 268–273. [Google Scholar] [CrossRef]
- Bar, R. Cyclodextrin-aided bioconversions and fermentations. Trends Biotechnol. 1989, 7, 2–4. [Google Scholar] [CrossRef]
- Calcagnile, M.; Bettini, S.; Damiano, F.; Talà, A.; Tredici, S.M.; Pagano, R.; Di Salvo, M.; Siculella, L.; Fico, D.; De Benedetto, G.E.; et al. Stimulatory Effects of Methyl-β-cyclodextrin on Spiramycin Production and Physical-Chemical Characterization of Nonhost@Guest Complexes. ACS Omega 2018, 3, 2470–2478. [Google Scholar] [CrossRef]
- Jarho, P.; Vander Velde, D.; Stella, V.J. Cyclodextrin-catalyzed deacetylation of spironolactone is pH and cyclodextrin dependent. J. Pharm. Sci. 2000, 89, 241–249. [Google Scholar] [CrossRef]
- Han, Y.; Zhou, W.; Shen, H.; Liu, Q.; Yu, W.; Ji, H.; She, Y. Progress in the immobilization of β-cyclodextrin and their application in adsorption of environmental pollutants. Chin. J. Org. Chem. 2016, 36, 248–257. [Google Scholar] [CrossRef][Green Version]
- Taka, A.L.; Pillay, K.; Yangkou Mbianda, X. Nanosponge cyclodextrin polyurethanes and their modification with nanomaterials for the removal of pollutants from waste water: A review. Carbohydr. Polym. 2017, 159, 94–107. [Google Scholar] [CrossRef]
- Breslow, R.; Dong, S.D. Biomimetic reactions catalyzed by cyclodextrins and their derivatives. Chem. Rev. 1998, 98, 1997–2011. [Google Scholar] [CrossRef] [PubMed]
- Hapiot, F.; Menuel, S.; Ferreira, M.; Léger, B.; Bricout, H.; Tilloy, S.; Monflier, E. Catalysis in Cyclodextrin-Based Unconventional Reaction Media: Recent Developments and Future Opportunities. ACS Sustain. Chem. Eng. 2017, 5, 3598–3606. [Google Scholar] [CrossRef]
- Bai, C.C.; Tian, B.R.; Zhao, T.; Huang, Q.; Wang, Z.Z. Cyclodextrin-catalyzed organic synthesis: Reactions, mechanisms, and applications. Molecules 2017, 22, 1475. [Google Scholar] [CrossRef]
- Leclercq, L.; Douyère, G.; Nardello-Rataj, V. Supramolecular chemistry and self-organization: A veritable playground for catalysis. Catalysts 2019, 9, 163. [Google Scholar] [CrossRef]
- Zare Asadabadi, A.; Hoseini, S.J.; Bahrami, M.; Nabavizadeh, S.M. Catalytic applications of β-cyclodextrin/palladium nanoparticle thin film obtained from oil/water interface in the reduction of toxic nitrophenol compounds and the degradation of azo dyes. New J. Chem. 2019, 43, 6513–6522. [Google Scholar] [CrossRef]
- Crini, G.; Morcellet, M. Synthesis and applications of adsorbents containing cyclodextrins. J. Sep. Sci. 2002, 25, 789–813. [Google Scholar] [CrossRef]
- Zhu, Q.; Scriba, G.K.E. Advances in the Use of Cyclodextrins as Chiral Selectors in Capillary Electrokinetic Chromatography: Fundamentals and Applications. Chromatographia 2016, 79, 1403–1435. [Google Scholar] [CrossRef]
- Martin, J.; Díaz-Montaña, E.J.; Asuero, A.G. Cyclodextrins: Past and Present. In Cyclodextrin—A Versatile Ingredient; Arora, P., Dhingra, N., Eds.; IntechOpen: London, UK, 2018. [Google Scholar]
- Harangi, J.; Béke, G.; Harangi, M.; Mótyán, J.A. The digestable parent cyclodextrin. J. Incl. Phenom. Macrocycl. Chem. 2012, 73, 335–339. [Google Scholar] [CrossRef]
- Munro, I.C.; Newberne, P.M.; Young, V.R.; Bär, A. Safety assessment of γ-cyclodextrin. Regul. Toxicol. Pharmacol. 2004, 39, 3–13. [Google Scholar] [CrossRef]
- Gidwani, B.; Vyas, A. A Comprehensive Review on Cyclodextrin-Based Carriers for Delivery of Chemotherapeutic Cytotoxic Anticancer Drugs. Biomed Res. Int. 2015, 2015, 198268. [Google Scholar] [CrossRef]
- Lina, B.A.R.; Bär, A. Subchronic oral toxicity studies with α-cyclodextrin in rats. Regul. Toxicol. Pharmacol. 2004, 39, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, A.; Aguilar, F.; Crebelli, R.; Di Domenico, A.; Dusemund, B.; Frutos, M.J.; Galtier, P.; Gott, D.; Gundert-Remy, U.; Leblanc, J.; et al. Re-evaluation of β-cyclodextrin (E 459) as a food additive. EFSA J. 2016, 14, e04628. [Google Scholar] [CrossRef]
- European Medicines Agency. Cyclodextrins Used as Excipients; European Medicines Agency: Amsterdam, The Netherlands, 2017; Volume 44. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).