Neuroprotective Effect of a Novel ATP-Synthase Inhibitor Bedaquiline in Cerebral Ischemia-Reperfusion Injury
Abstract
1. Introduction
2. Results
2.1. Effect of BDQ Treatment on the Infarct Size and Neurological Impairment of Ischemic Rats
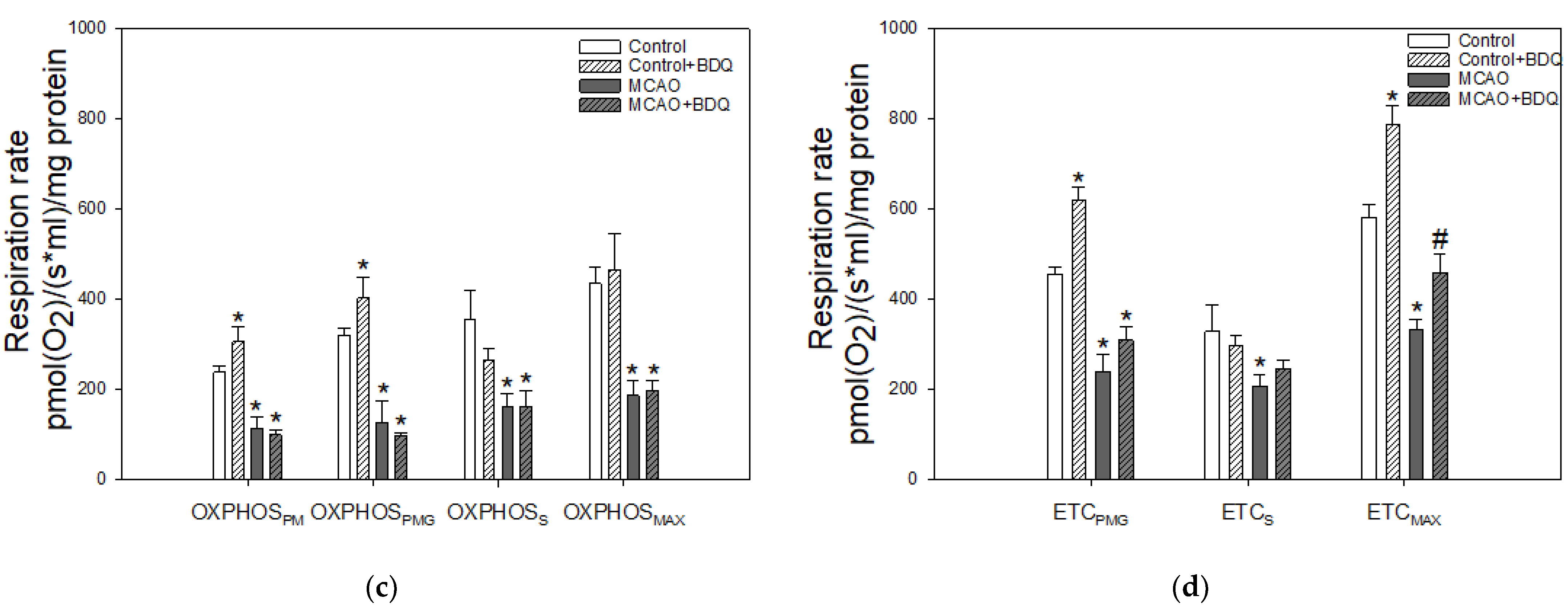
2.2. Effect of MCAO and BDQ Treatment on Mitochondrial Respiration
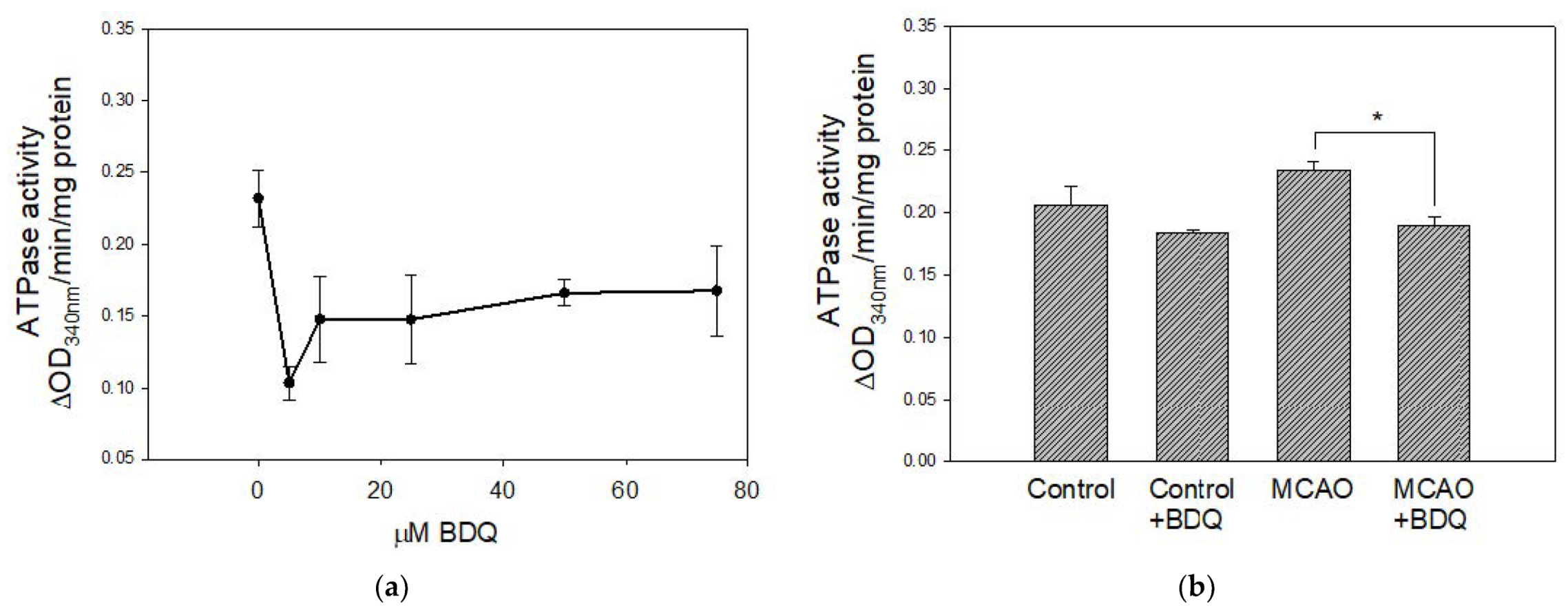
2.3. BDQ Effects on the Activity of ATPase
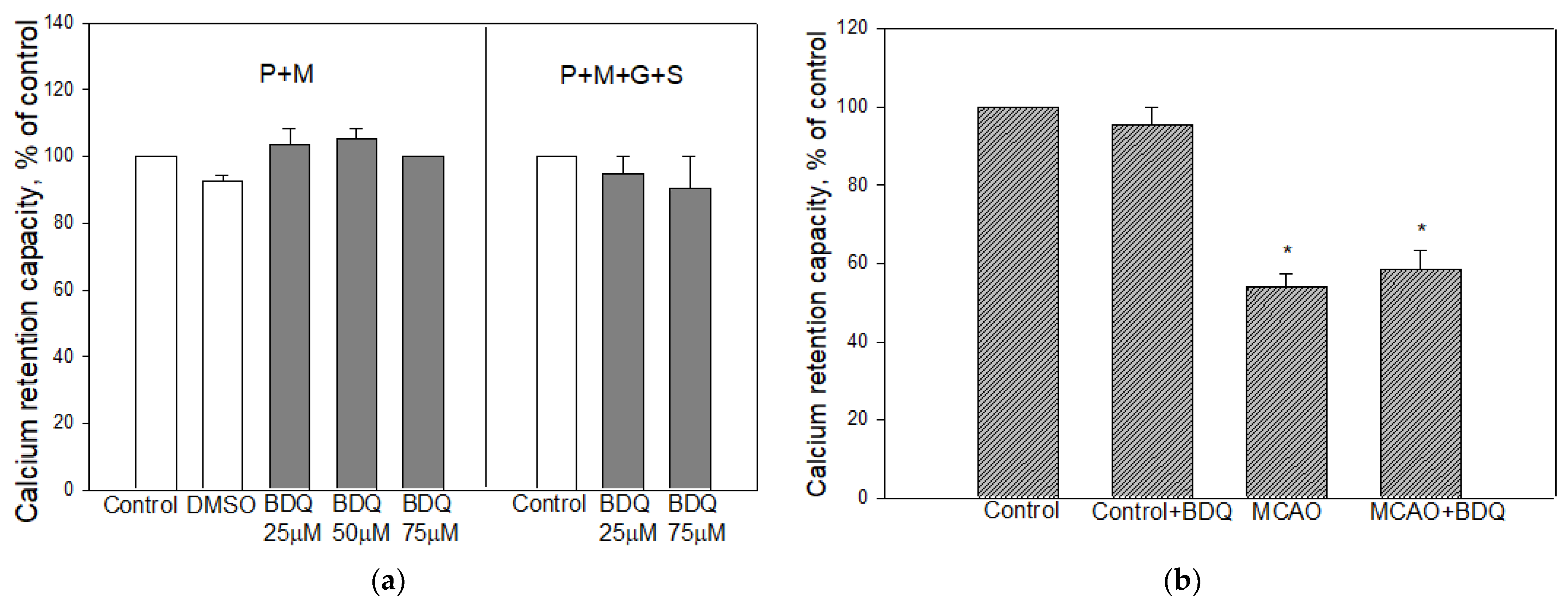
2.4. Effect of BDQ on Mitochondrial Calcium Retention Capacity
3. Discussion
4. Materials and Methods
4.1. Animals and Treatment
4.2. Transient Focal Ischemia Model
4.3. Neurological Evaluation
4.4. Evaluation of Infarct Size
4.5. Isolation of Mitochondria
4.6. Measurement of Mitochondrial Respiration
4.7. Measurement of Mitochondrial Calcium Retention Capacity
4.8. Measurement of the Enzymatic Activity of ATPase
4.9. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Anderson, C.S. The global burden of stroke: Persistent and disabling. Lancet Neurol. 2019, 18, 417–418. [Google Scholar] [CrossRef]
- Campbell, B.C.; Silva, D.A.D.; Macleod, M.R.; Coutts, S.B.; Schwamm, L.H.; Davis, S.M.; Donnan, G.A. Ischaemic stroke. Nat. Rev. Dis. Prim. 2019, 5, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Grefkes, C.; Fink, G.R. Recovery from stroke: Current concepts and future perspectives. Neurol. Res. Pract. 2020, 2, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Goyal, M.; Menon, B.K.; Zwam, W.H.V.; Dippel, D.W.J.; Mitchell, P.J.; Demchuk, A.M.; Dávalos, A.; Majoie, C.B.L.M.; Berg, L.A.V.D.; Levy, E.I.; et al. Endovascular thrombectomy after large-vessel ischaemic stroke: A meta-analysis of individual patient data from fi ve randomised trials. Lancet 2016, 387, 1723–1731. [Google Scholar] [CrossRef]
- Fiehler, J.; Ford, I.; Galinovic, I.; Gellissen, S.; Golsari, A.; Gregori, J.; Günther, M.; Guibernau, J.; Häusler, K.G.; Hennerici, M.; et al. MRI-Guided Thrombolysis for Stroke with Unknown Time of Onset. N. Engl. J. Med. 2018, 379, 611–622. [Google Scholar] [CrossRef]
- Whiteley, W.N.; Emberson, J.; Lees, K.R.; Blackwell, L.; Albers, G.; Bluhmki, E.; Brott, T.; Cohen, G. Risk of intracerebral haemorrhage with alteplase after acute ischaemic stroke: A secondary analysis of an individual patient data meta-analysis. Lancet 2016, 15, 925–933. [Google Scholar] [CrossRef]
- Behme, D.; Gondecki, L.; Fiethen, S. Complications of mechanical thrombectomy for acute ischemic stroke—A retrospective single-center study of 176 consecutive cases. Neuroradiology 2014, 56, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Warach, S.; Latour, L.L. Evidence of Reperfusion Injury, Exacerbated by Thrombolytic Therapy, in Human Focal Brain Ischemia Using a Novel Imaging Marker of Early Blood-Brain Barrier Disruption. Stroke 2004, 35, 2659–2661. [Google Scholar] [CrossRef]
- Zhang, R.; Chopp, M.; Chen, H.; Garcia, J.H. Temporal profile of ischemic tissue damage, neutrophil response, and vascular plugging following permanent and transient (2H) middle cerebral artery occlusion in the rat. J. Neurol. Sci. 1994, 125, 3–10. [Google Scholar] [CrossRef]
- Lin, L.; Wang, X.; Yu, Z. Ischemia-reperfusion Injury in the Brain: Mechanisms and Potential Therapeutic Strategies. Biochem. Pharmacol. 2018, 5, 1–16. [Google Scholar] [CrossRef]
- Andrabi, S.S.; Parvez, S.; Tabassum, H. Ischemic stroke and mitochondria: Mechanisms and targets. Protoplasma 2020, 257, 335–343. [Google Scholar] [CrossRef]
- Jordan, J.; Groot, P.W.J.D.; Galindo, M.F. Mitochondria: The Headquarters in Ischemia-Induced Neuronal Death. Agents Med. Chem. 2011, 11, 98–106. [Google Scholar] [CrossRef]
- Grover, G.J.; Atwal, K.S.; Sleph, P.G.; Wang, F.; Monshizadegan, H.; Monticello, T.; Green, D.W.; Gary, J.; Atwal, K.S.; Sleph, P.G.; et al. Excessive ATP hydrolysis in ischemic myocardium by mitochondrial F1F0-ATPase: Effect of selective pharmacological inhibition of mitochondrial ATPase hydrolase activity. Am. J. Physiol. Hear. Circ. Physiol. 2004, 287, 1747–1755. [Google Scholar] [CrossRef]
- Gao, Y.Q.; Yang, W.; Karplus, M.; Biophysique, L.D.C. A Structure-Based Model for the Synthesis and Hydrolysis of ATP by F1-ATPase. Cell 2005, 123, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Iijima, T. Mitochondrial membrane potential and ischemic neuronal death. Neurosci. Res. 2006, 55, 234–243. [Google Scholar] [CrossRef] [PubMed]
- Berkich, D.A.; Salama, G.; Lanoue, K.F. Mitochondrial membrane potentials in ischemic hearts. Arch. Biochem. Biophys. 2003, 420, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Chinopoulos, C.; Adam-vizi, V. Mitochondria as ATP consumers in cellular pathology. BBA 2010, 1802, 221–227. [Google Scholar] [CrossRef]
- Matsuyama, S.; Xu, Q.; Velours, J.; Reed, J.C. The Mitochondrial F0F1-ATPase Proton Pump Is Required for Function of the Proapoptotic Protein Bax in Yeast and Mammalian Cells. Mol. Cell 1998, 1, 327–336. [Google Scholar] [CrossRef]
- Formentini, L.; Cuezva, M. Mitochondria-Mediated Energy Adaption in Cancer: The H+-ATP Synthase-Geared Switch of Metabolism in Human Tumors. Antioxid. Redox Signal. 2013, 19, 285–298. [Google Scholar] [CrossRef]
- Formentini, L.; Pereira, M.P.; Sánchez-Cenizo, L.; Santacatterina, F.; Lucas, J.J.; Navarro, C.; Martínez-Serrano, A.; Cuezva, J.M. In vivo inhibition of the mitochondrial H+-ATP synthase in neurons promotes metabolic preconditioning. EMBO J. 2014, 33, 762–778. [Google Scholar] [CrossRef]
- Long, Q.; Yang, K.; Yang, Q. Regulation of mitochondrial ATP synthase in cardiac pathophysiology. Am. J. Cardiovasc. Dis. 2015, 5, 19–32. [Google Scholar]
- Nesci, S.; Trombetti, F.; Algieri, C.; Pagliarani, A. A Therapeutic Role for the F1F0-ATP Synthase. SLAS Discov. 2019, 24, 893–903. [Google Scholar] [CrossRef]
- Bosetti, F.; Yu, G.; Zucchi, R.; Solaini, G. Myocardial ischemic preconditioning and mitochondrial F1F0-ATPase activity. Mol. Cell. Biochem. 2000, 215, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Matic, I.; Cocco, S.; Ferraina, C.; Martin-jimenez, R.; Florenzano, F.; Crosby, J.; Lupi, R.; Amadoro, G.; Russell, C.; Pignataro, G.; et al. Neuroprotective coordination of cell mitophagy by the F1F0-ATPase inhibitory factor 1 (IF1). Pharmacol. Res. 2016, 103, 56–68. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Maierean, S.; Serban, M.; Rizzo, M.; Lippi, G.; Sahebkar, A. The potential role of mitochondrial ATP synthase inhibitory factor 1 (IF1) in coronary heart disease: A literature review. Lipids Health Dis. 2017, 16, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Chuang, I.; Dong, H.; Yang, R. Hypoxia-Inducible Factor 1a Regulates the Expression of the Mitochondrial ATPase Inhibitor Protein (IF1) in Rat Liver. Shock 2011, 36, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Bernardi, P.; Rasola, A.; Forte, M.; Lippe, G. The Mitochondrial Permeability Transition Pore: Channel Formation by F-ATP Synthase, Integration in Signal Transduction, and Role in Pathophysiology. Physiol. Rev. 2015, 95, 1111–1155. [Google Scholar] [CrossRef]
- Bonora, M.; Wieckowski, M.R.; Chinopoulos, C.; Kepp, O.; Kroemer, G.; Galluzzi, L.; Pinton, P. Molecular mechanisms of cell death: Central implication of ATP synthase in mitochondrial permeability transition This article has been corrected since Advance Online Publication and a corrigendum is also printed in this issue. Oncogene 2015, 34, 1475–1486. [Google Scholar] [CrossRef]
- Hurst, S.; Hoek, J.; Sheu, S.-S. Mitochondrial Ca2+ and Regulation of the Permeability Transition Pore. J. Bioenerg. Biomembr. 2018, 49, 27–47. [Google Scholar] [CrossRef]
- Li, B.; Chauvin, C.; De Paulis, D.; De Oliveira, F.; Gharib, A.; Vial, G.; Lablanche, S.; Leverve, X.; Bernardi, P.; Ovize, M.; et al. Inhibition of complex I regulates the mitochondrial permeability transition through a phosphate-sensitive inhibitory site masked by cyclophilin D. BBA 2012, 1817, 1628–1634. [Google Scholar] [CrossRef] [PubMed]
- Urbani, A.; Giorgio, V.; Carrer, A.; Franchin, C.; Arrigoni, G.; Jiko, C.; Abe, K.; Maeda, S.; Shinzawa-itoh, K.; Bogers, J.F.M.; et al. Purified F-ATP synthase forms a Ca2+-dependent high-conductance channel matching the mitochondrial permeability transition pore. Nat. Commun. 2019, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Neginskaya, M.A.; Solesio, M.E.; Berezhnaya, E.V.; Amodeo, G.F.; Mnatsakanyan, N.; Jonas, E.A.; Pavlov, E.V. ATP Synthase C-Subunit-Deficient Mitochondria Have a Small Cyclosporine A-Sensitive Channel, but Lack the Permeability Transition Pore. Cell Rep. 2019, 26, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Giorgio, V.; von Stockum, S.; Antoniel, M.; Fabbro, A.; Fogolari, F.; Forte, M.; Glick, G.D.; Petronilli, V.; Zoratti, M.; Szabo, I.; et al. Dimers of mitochondrial ATP synthase form the permeability transition pore. Proc. Natl. Acad. Sci. USA 2013, 110, 5887–5892. [Google Scholar] [CrossRef] [PubMed]
- Worley, M.V.; Estrada, S.J. Bedaquiline: A Novel Antitubercular Agent for the Treatment of Multidrug-Resistant Tuberculosis. Pharmacotherapy 2014. [Google Scholar] [CrossRef]
- Haagsma, A.C.; Podasca, I.; Koul, A.; Andries, K.; Guillemont, J.; Lill, H.; Bald, D. Probing the Interaction of the Diarylquinoline TMC207 with Its Target Mycobacterial ATP Synthase. PLoS ONE 2011, 6, 1–7. [Google Scholar] [CrossRef]
- Koul, A.; Vranckx, L.; Dendouga, N.; Balemans, W.; Van Den Wyngaert, I.; Vergauwen, K.; Go, H.W.H.; Willebrords, R.; Poncelet, A.; Guillemont, J.; et al. Diarylquinolines Are Bactericidal for Dormant Mycobacteria as a Result of Disturbed ATP Homeostasis. J. Biol. Chem. 2008, 283, 25273–25280. [Google Scholar] [CrossRef]
- Haagsma, A.C.; Abdillahi-ibrahim, R.; Wagner, M.J.; Krab, K.; Vergauwen, K.; Guillemont, J.; Andries, K.; Lill, H.; Koul, A.; Bald, D. Selectivity of TMC207 towards Mycobacterial ATP Synthase Compared with That towards the Eukaryotic Homologue. Antimicrob. Agents Chemother. 2009, 53, 1290–1292. [Google Scholar] [CrossRef]
- Andries, K.; Verhasselt, P.; Guillemont, J.; Gohlmann, H.W.H.; Neefs, J.; Winkler, H.; Van Gestel, J.; Timmerman, P.; Zhu, M.; Lee, E.; et al. A Diarylquinoline Drug Active on the ATP Synthase of Mycobacterium tuberculosis. Science 2005, 307, 223–227. [Google Scholar] [CrossRef]
- Luo, M.; Zhou, W.; Patel, H.; Srivastava, A.P.; Symersky, J.; Bonar, M.M.; Faraldo-gómez, J.D.; Liao, M.; Mueller, D.M. Bedaquiline inhibits the yeast and human mitochondrial ATP synthases. Commun. Biol. 2020, 3, 1–10. [Google Scholar] [CrossRef]
- Shchepina, L.A.; Pletjushkina, O.Y.; Avetisyan, A.V.; Bakeeva, L.E.; Fetisova, E.K.; Izyumov, D.S.; Saprunova, V.B.; Vyssokikh, M.Y.; Chernyak, B.V.; Skulachev, V.P. Oligomycin, inhibitor of the F0 part of H+-ATP-synthase, suppresses the TNF-induced apoptosis. Oncogene 2002, 21, 8149–8157. [Google Scholar] [CrossRef] [PubMed]
- Jennings, R.B.; Reimer, K.A.; Steenbergen, C. Effect of Inhibition of the Mitochondrial ATPase on Net Myocardial ATP in Total Ischemia. J. Mol. Cell. Cardiol. 1991, 23, 1383–1395. [Google Scholar] [CrossRef]
- Mnatsakanyan, N.; Jonas, E.A. ATP synthase c-subunit ring as the channel of mitochondrial permeability transition: Regulator of metabolism in development and degeneration. J. Mol. Cell. Cardiol. 2020, 144, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Belosludtsev, K.N.; Belosludtseva, N.V.; Talanov, E.Y.; Tenkov, K.S.; Starinets, V.S.; Agafonov, A.V.; Pavlik, L.L.; Dubinin, M. V Effect of bedaquiline on the functions of rat liver mitochondria. Biochim. Biophys. Acta Biomembr. 2019, 1861, 288–297. [Google Scholar] [CrossRef] [PubMed]
- Pamreddy, A.; Baijnath, S.; Naicker, T.; Ntshangase, S.; Mdanda, S.; Lubanyana, H.; Kruger, H.G.; Govender, T. Bedaquiline has potential for targeting tuberculosis reservoirs in the central nervous system. RSC Adv. 2018, 8, 11902–11907. [Google Scholar] [CrossRef]
- Penna, C.; Pagliaro, P.; Rastaldo, R.; Di Pancrazio, F.; Lippe, G.; Gattullo, D.; Mancardi, D.; Samaja, M.; Losano, G.; Mavelli, I. F0F1 ATP synthase activity is differently modulated by coronary reactive hyperemia before and after ischemic preconditioning in the goat. Am. J. Physiol. Hear. Circ. Physiol. 2004, 287, 2192–2200. [Google Scholar] [CrossRef]
- Djafarzadeh, S.; Jakob, S.M. High-resolution respirometry to assess mitochondrial function in permeabilized and intact cells. J. Vis. Exp. 2017, 2017, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hards, K.; Robson, J.R.; Berney, M.; Shaw, L.; Bald, D.; Koul, A.; Andries, K.; Cook, G.M. Bactericidal mode of action of bedaquiline. J. Antimicrob. Chemother. 2015, 70, 2028–2037. [Google Scholar] [CrossRef]
- Nath, S. Interpretation of the mechanism of action of antituberculosis drug bedaquiline based on a novel two-ion theory of energy coupling in ATP synthesis. Bioeng. Transl. Med. 2019, 4, 164–170. [Google Scholar] [CrossRef]
- Hards, K.; McMillan, D.G.G.; Schurig-Briccio, L.A.; Gennis, R.B.; Lill, H.; Bald, D.; Cook, G.M. Ionophoric effects of the antitubercular drug bedaquiline. Proc. Natl. Acad. Sci. USA 2018, 115, 7326–7331. [Google Scholar] [CrossRef]
- Fiorillo, M.; Lamb, R.; Tanowitz, H.B.; Cappello, A.R.; Martinez-Outschoorn, U.E.; Sotgia, F.; Lisanti, M.P. Bedaquiline, an FDA-approved antibiotic, inhibits mitochondrial function and potently blocks the proliferative expansion of stem-like cancer cells (CSCs). Aging (Albany N. Y.) 2016, 8, 1593–1607. [Google Scholar] [CrossRef]
- Klingenberg, M. The ADP and ATP transport in mitochondria and its carrier. Biochim. Biophys. Acta Biomembr. 2008, 1778, 1978–2021. [Google Scholar] [CrossRef] [PubMed]
- Brennan, J.P.; Southworth, R.; Medina, R.A.; Davidson, S.M.; Duchen, M.R.; Shattock, M.J. Mitochondrial uncoupling, with low concentration FCCP, induces ROS-dependent cardioprotection independent of KATP channel activation. Cardiovasc. Res. 2006, 72, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Pandya, J.D.; Pauly, J.R.; Sullivan, P.G. The optimal dosage and window of opportunity to maintain mitochondrial homeostasis following traumatic brain injury using the uncoupler FCCP. Exp. Neurol. 2009, 218, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Hüttemann, M.; Helling, S.; Sanderson, T.H.; Sinkler, C.; Samavati, L.; Mahapatra, G.; Varughese, A.; Lu, G.; Liu, J.; Ramzan, R.; et al. Regulation of mitochondrial respiration and apoptosis through cell signaling: Cytochrome c oxidase and cytochrome c in ischemia/reperfusion injury and inflammation. Biochim. Biophys. Acta Bioenerg. 2012, 1817, 598–609. [Google Scholar] [CrossRef] [PubMed]
- Racay, P.; Tatarkova, Z.; Chomova, M.; Hatok, J.; Kaplan, P.; Dobrota, D. Mitochondrial calcium transport and mitochondrial dysfunction after global brain ischemia in rat hippocampus. Neurochem. Res. 2009, 34, 1469–1478. [Google Scholar] [CrossRef]
- Christensen, T.; Diemer, N.H. Reduction of Mitochondrial Electron Transport Complex Activity is Restricted to the Ischemic Focus after Transient Focal Cerebral Ischemia in Rats: A Histochemical Volumetric Analysis. Neurochem. Res. 2003, 28, 1805–1812. [Google Scholar] [CrossRef] [PubMed]
- Giorgio, V.; Bisetto, E.; Soriano, M.E.; Dabbeni-Sala, F.; Basso, E.; Petronilli, V.; Forte, M.A.; Bernardi, P.; Lippe, G. Cyclophilin D modulates mitochondrial F0F1-ATP synthase by interacting with the lateral stalk of the complex. J. Biol. Chem. 2009, 284, 33982–33988. [Google Scholar] [CrossRef]
- Giorgio, V.; Fogolari, F.; Lippe, G.; Bernardi, P. OSCP subunit of mitochondrial ATP synthase: Role in regulation of enzyme function and of its transition to a pore. Br. J. Pharmacol. 2019, 176, 4247–4257. [Google Scholar] [CrossRef]
- Sheu, J.J.; Chiou, H.Y.; Kang, J.H.; Chen, Y.H.; Lin, H.C. Tuberculosis and the risk of ischemic stroke: A 3-year follow-up study. Stroke 2010, 41, 244–249. [Google Scholar] [CrossRef]
- Longa, E.Z.; Weinstein, P.R.; Carlson, S.; Cummins, R. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 1989, 20, 84–91. [Google Scholar] [CrossRef]
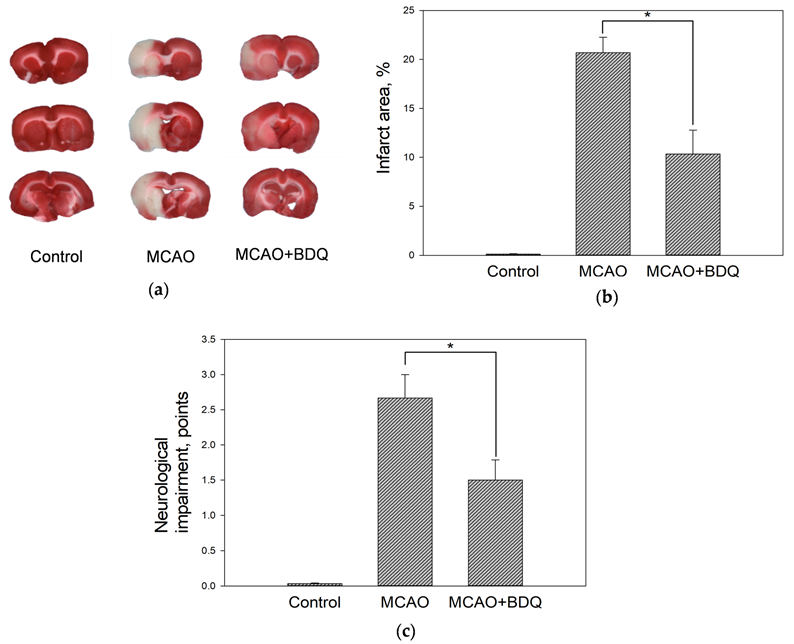


| Test | Neurological Deficit | Score |
|---|---|---|
| Forelimb extension (flexion) | Mild | 1 |
| Twisting of the torso | Mild-moderate | 2 |
| Circling to the right | Moderate | 3 |
| Falling to the right | Severe | 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Umbrasas, D.; Arandarcikaite, O.; Grigaleviciute, R.; Stakauskas, R.; Borutaite, V. Neuroprotective Effect of a Novel ATP-Synthase Inhibitor Bedaquiline in Cerebral Ischemia-Reperfusion Injury. Int. J. Mol. Sci. 2021, 22, 9717. https://doi.org/10.3390/ijms22189717
Umbrasas D, Arandarcikaite O, Grigaleviciute R, Stakauskas R, Borutaite V. Neuroprotective Effect of a Novel ATP-Synthase Inhibitor Bedaquiline in Cerebral Ischemia-Reperfusion Injury. International Journal of Molecular Sciences. 2021; 22(18):9717. https://doi.org/10.3390/ijms22189717
Chicago/Turabian StyleUmbrasas, Danielius, Odeta Arandarcikaite, Ramune Grigaleviciute, Rimantas Stakauskas, and Vilmante Borutaite. 2021. "Neuroprotective Effect of a Novel ATP-Synthase Inhibitor Bedaquiline in Cerebral Ischemia-Reperfusion Injury" International Journal of Molecular Sciences 22, no. 18: 9717. https://doi.org/10.3390/ijms22189717
APA StyleUmbrasas, D., Arandarcikaite, O., Grigaleviciute, R., Stakauskas, R., & Borutaite, V. (2021). Neuroprotective Effect of a Novel ATP-Synthase Inhibitor Bedaquiline in Cerebral Ischemia-Reperfusion Injury. International Journal of Molecular Sciences, 22(18), 9717. https://doi.org/10.3390/ijms22189717






