Development of a Novel Electrochemical Biosensor Based on Carbon Nanofibers–Cobalt Phthalocyanine–Laccase for the Detection of p-Coumaric Acid in Phytoproducts
Abstract
:1. Introduction
2. Results and Discussion
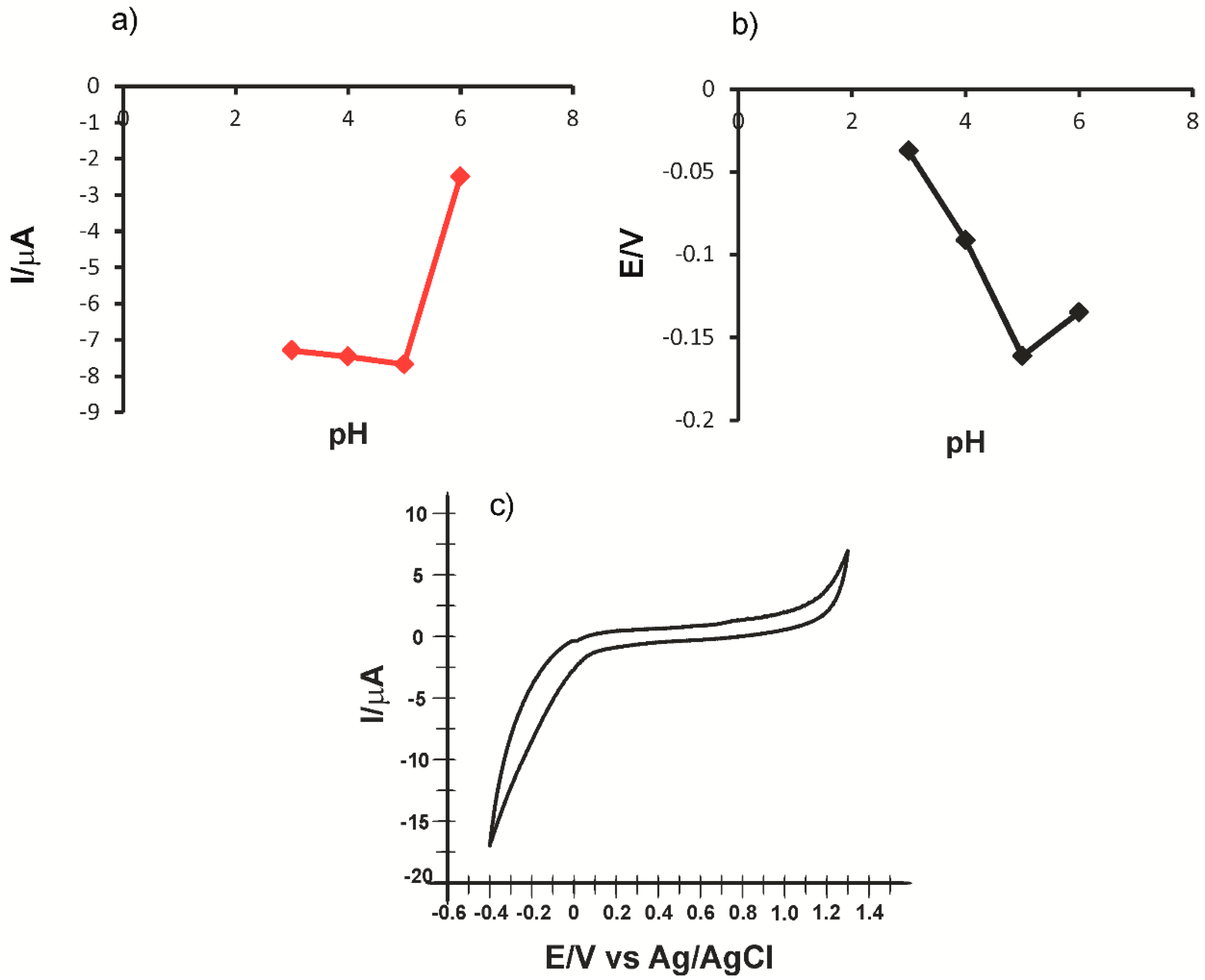
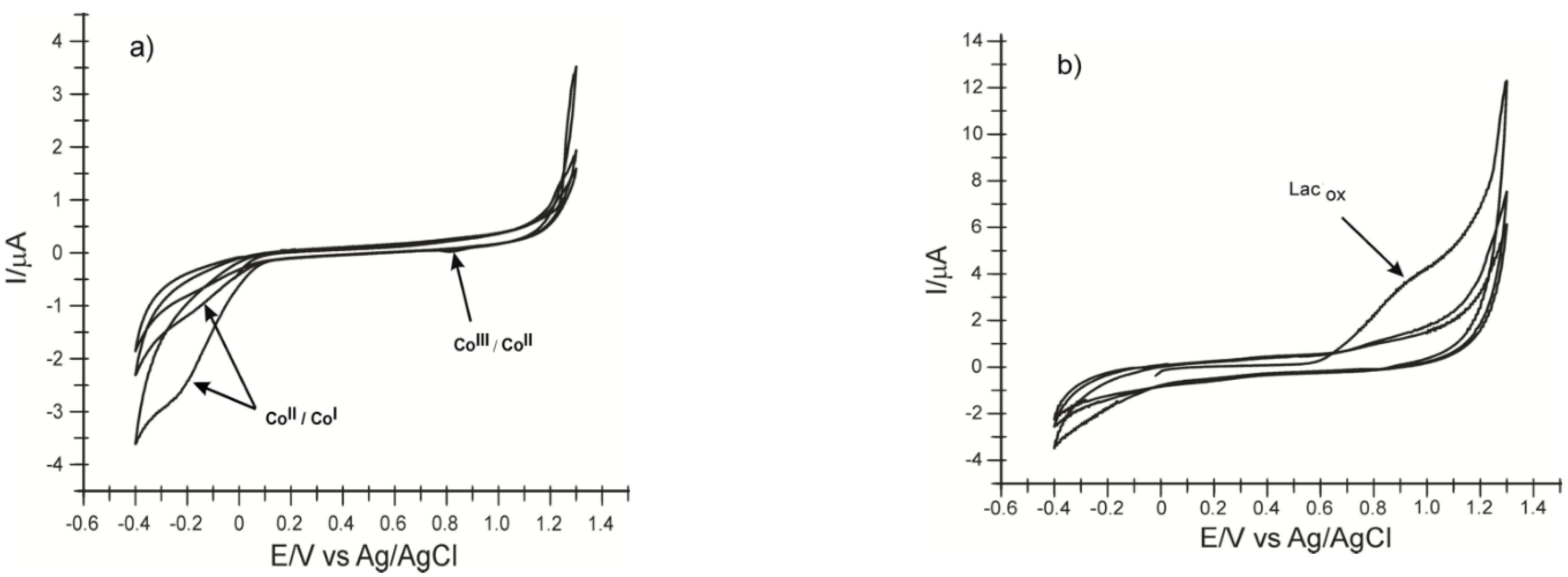
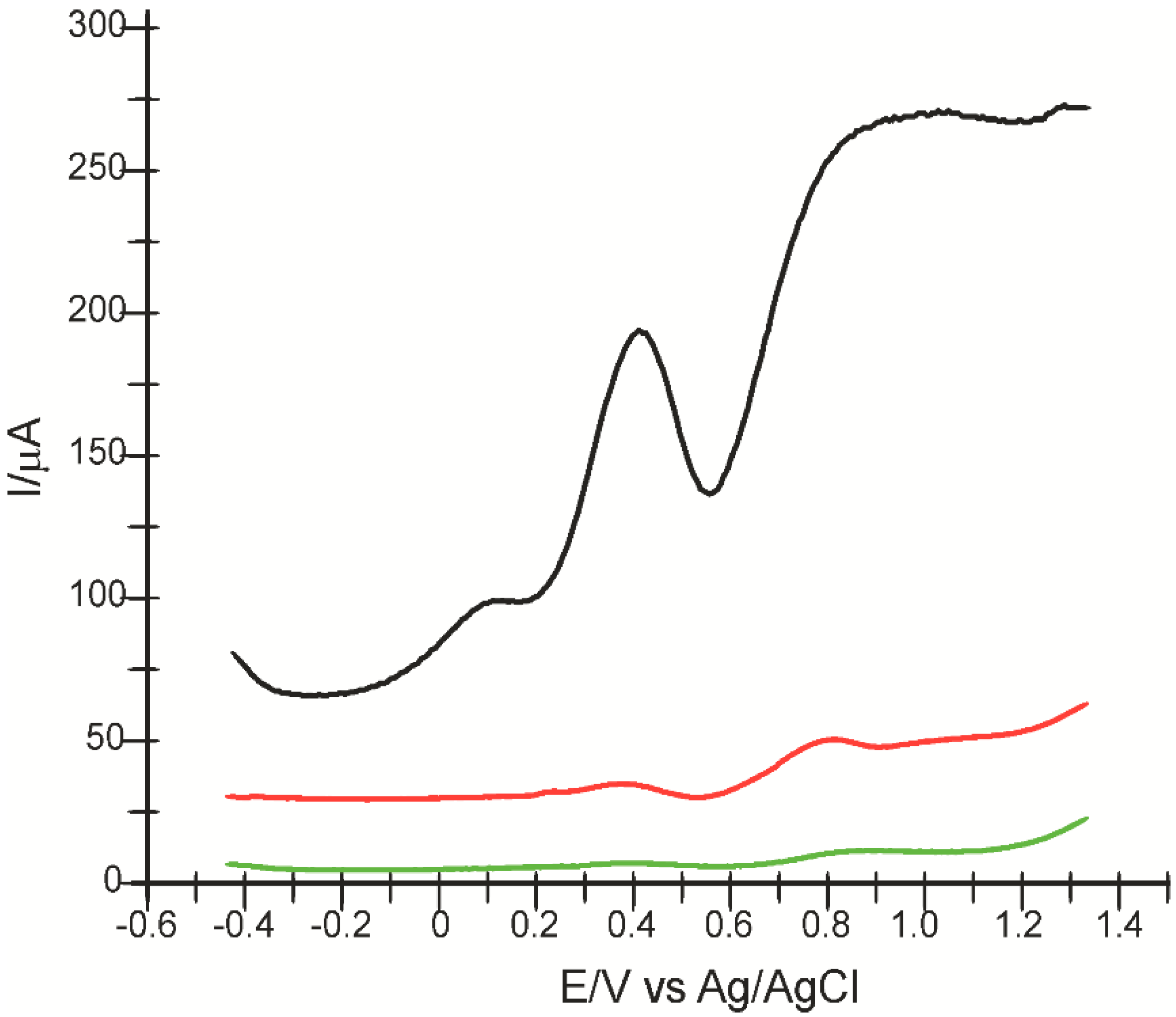
2.1. Preliminary Studies for Electrode Characterization
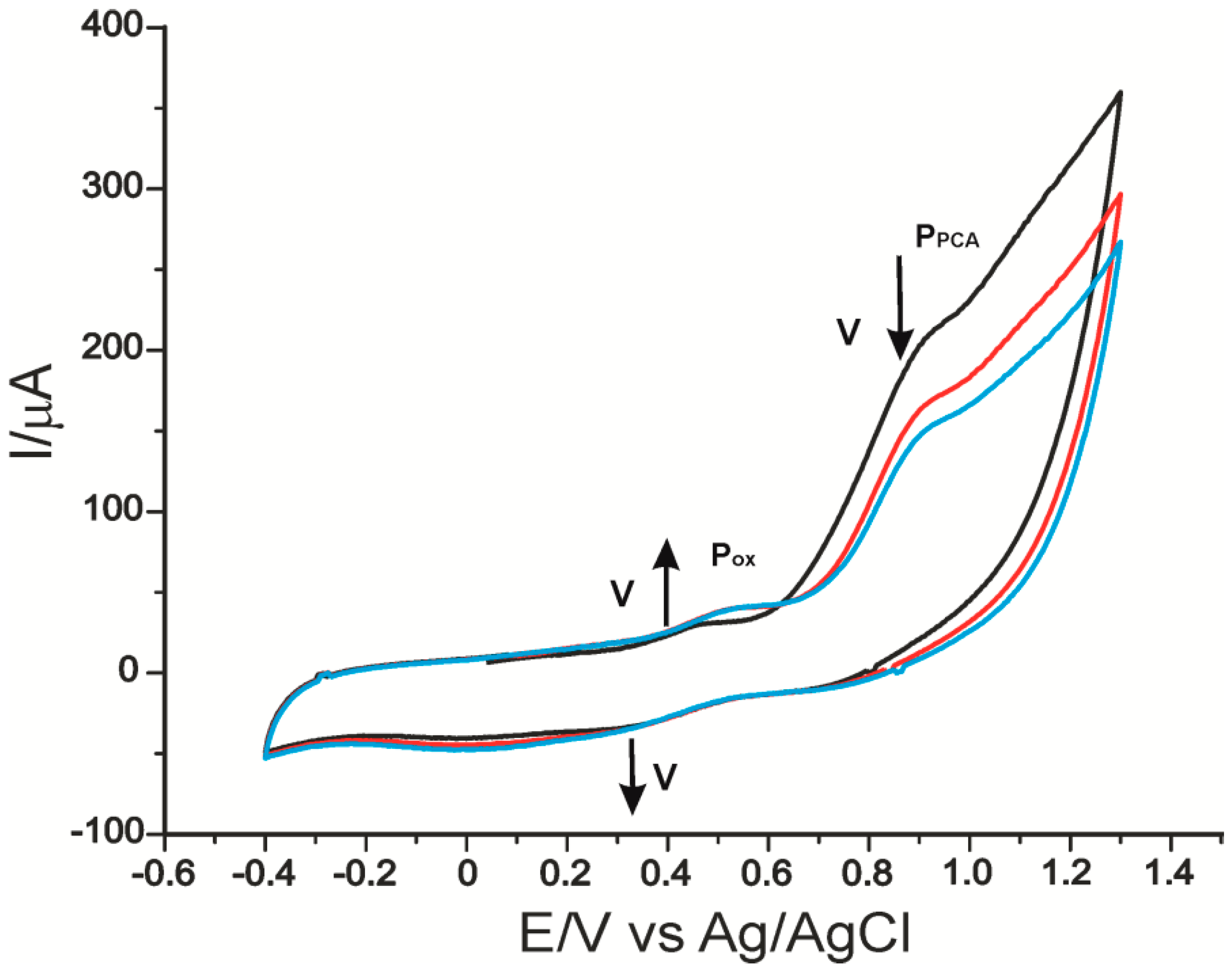
2.2. The Voltammetric Responses of Electrodes in p-Coumaric Acid Solution
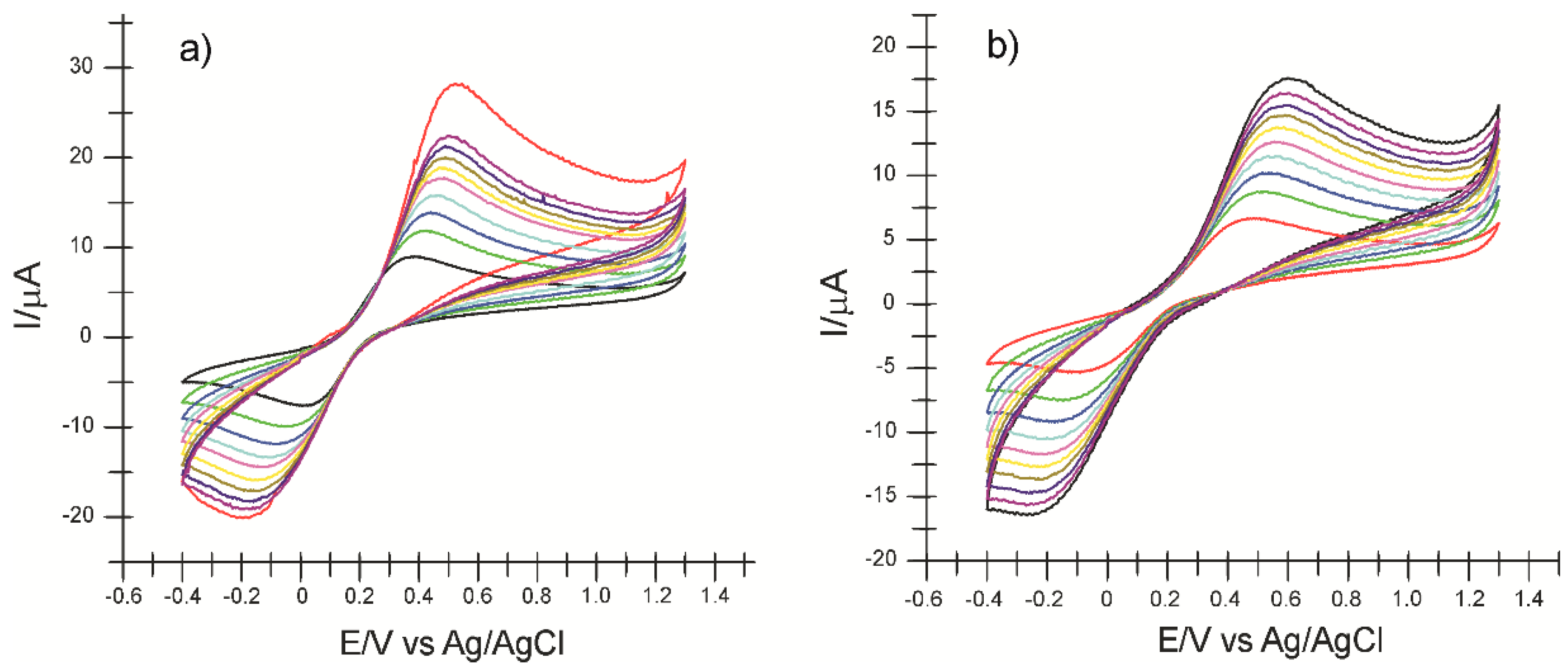
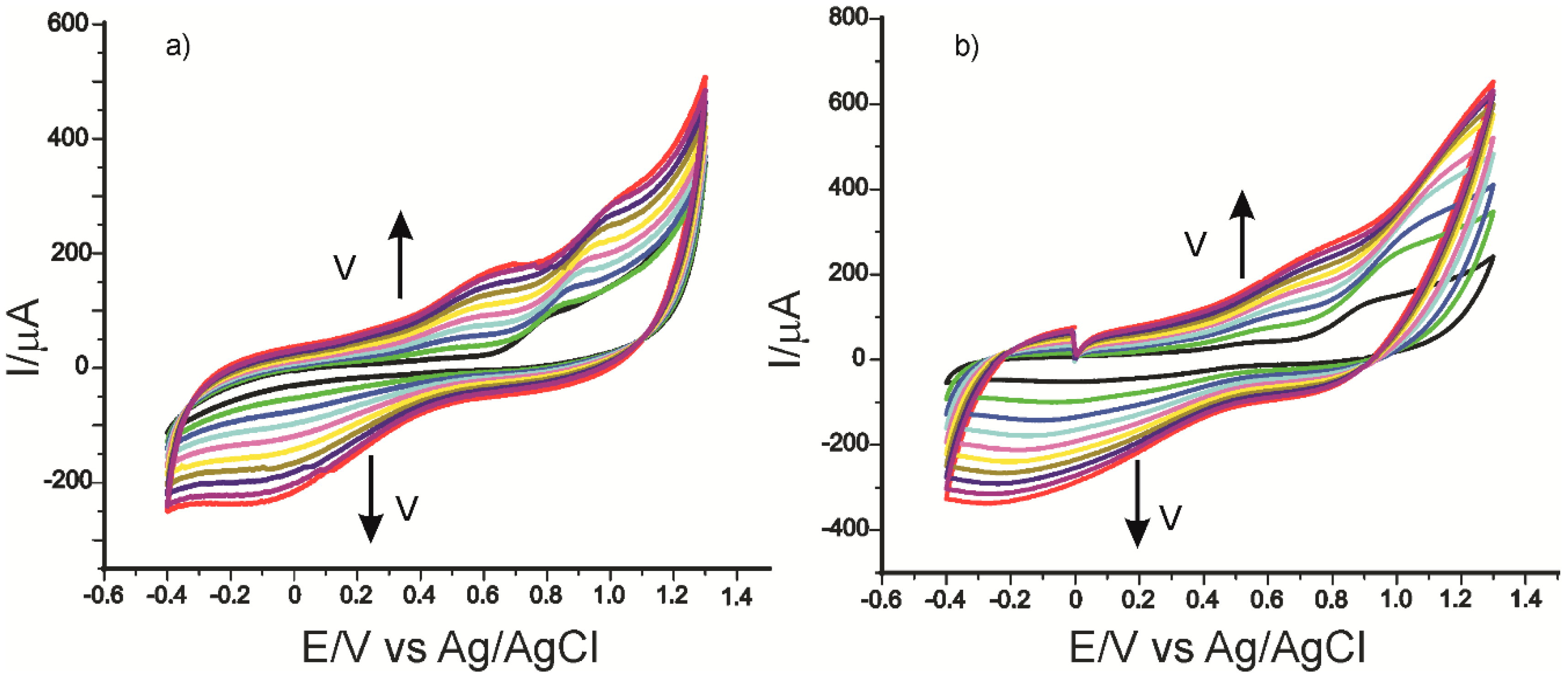
2.3. Influence of Scan Rate on the Voltammetric Response
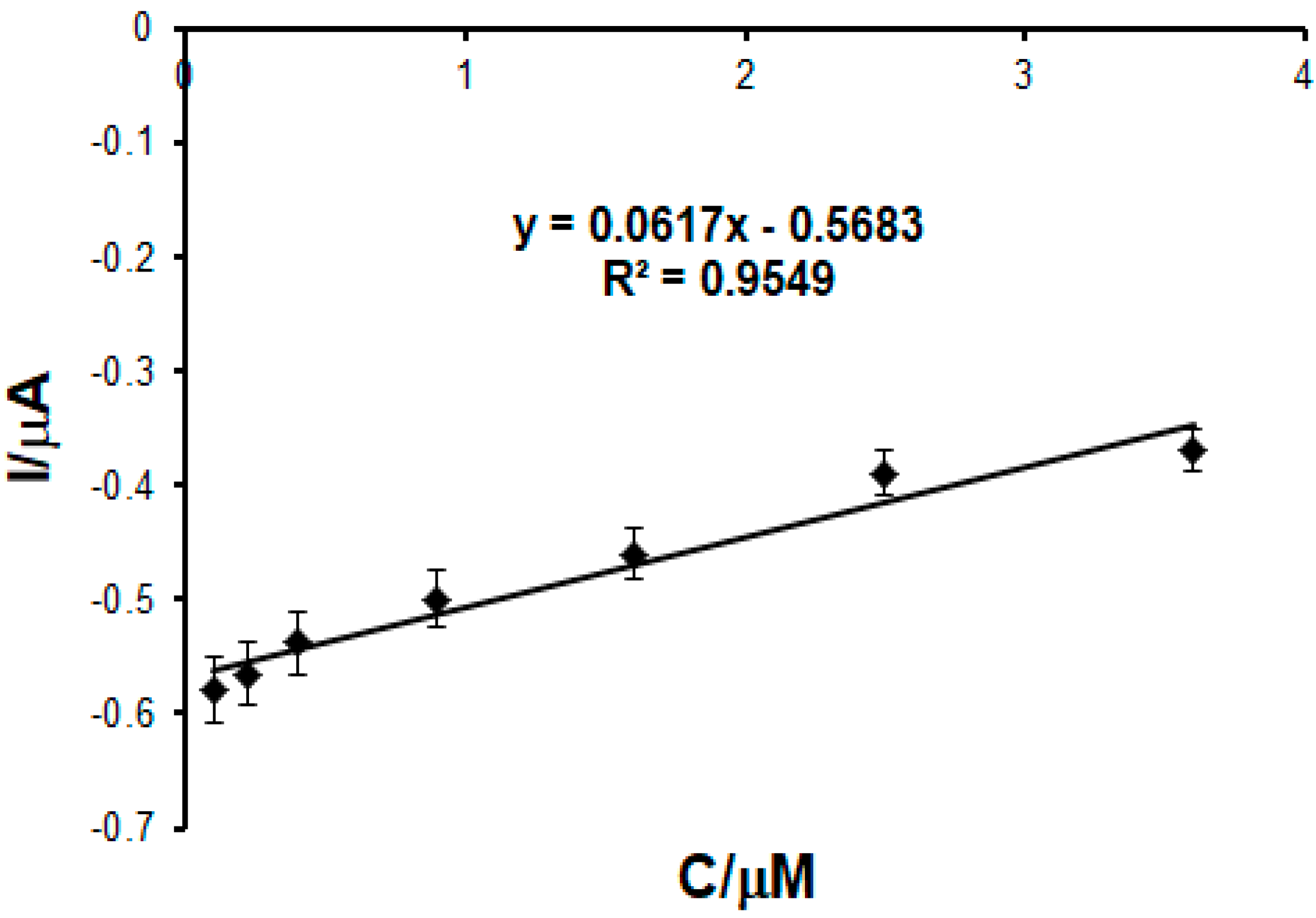
2.4. Calibration Curve
2.5. Characteristics of the Biosensor
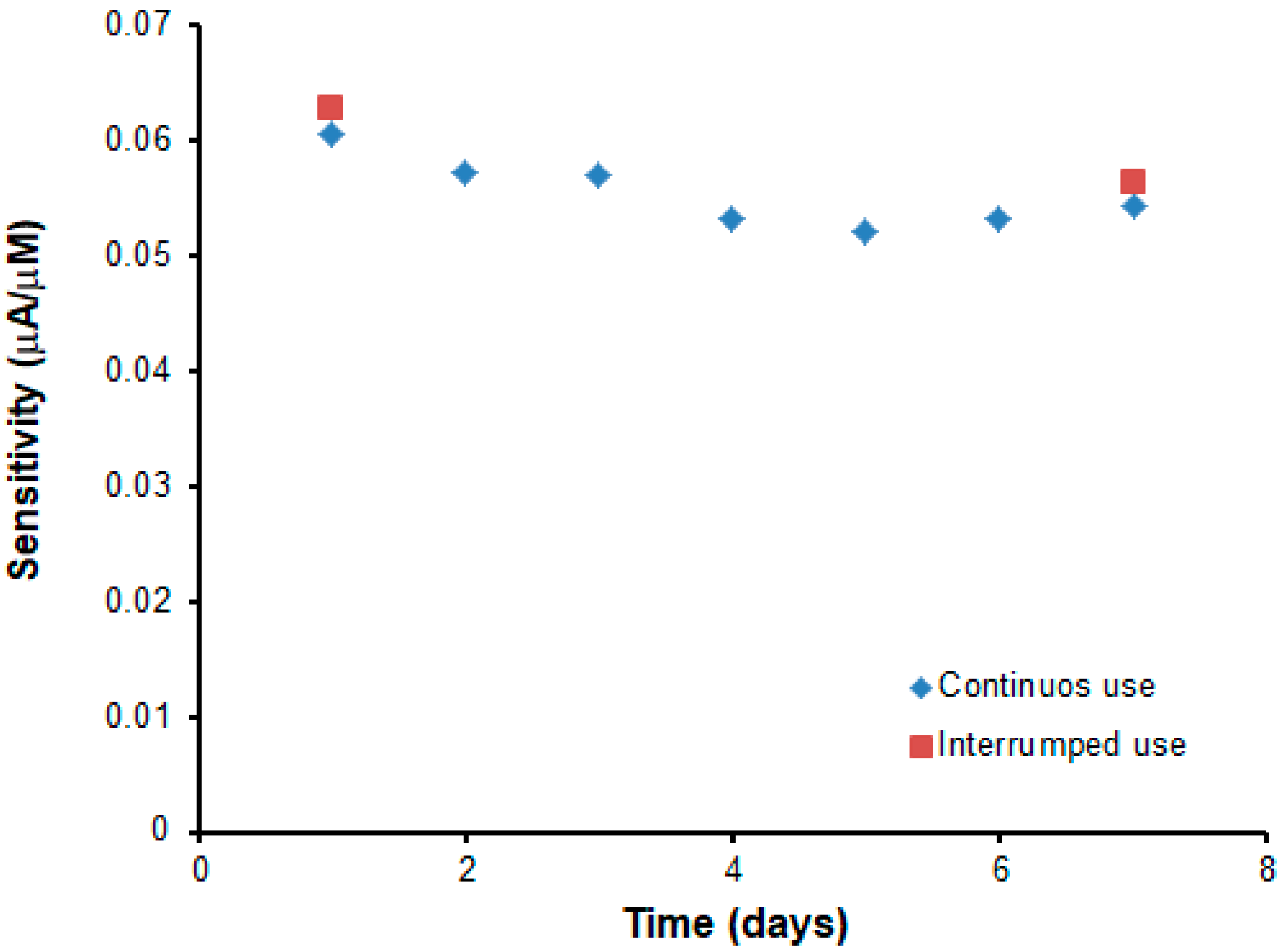
2.6. Stability, Reproducibility of Fabrication, and Repeatability of the Biosensor
2.7. Interference Studies
2.8. Determining PCA in Phytoproducts
3. Materials and Methods
3.1. Reagents and Solution
3.2. Electrodes and Devices Used
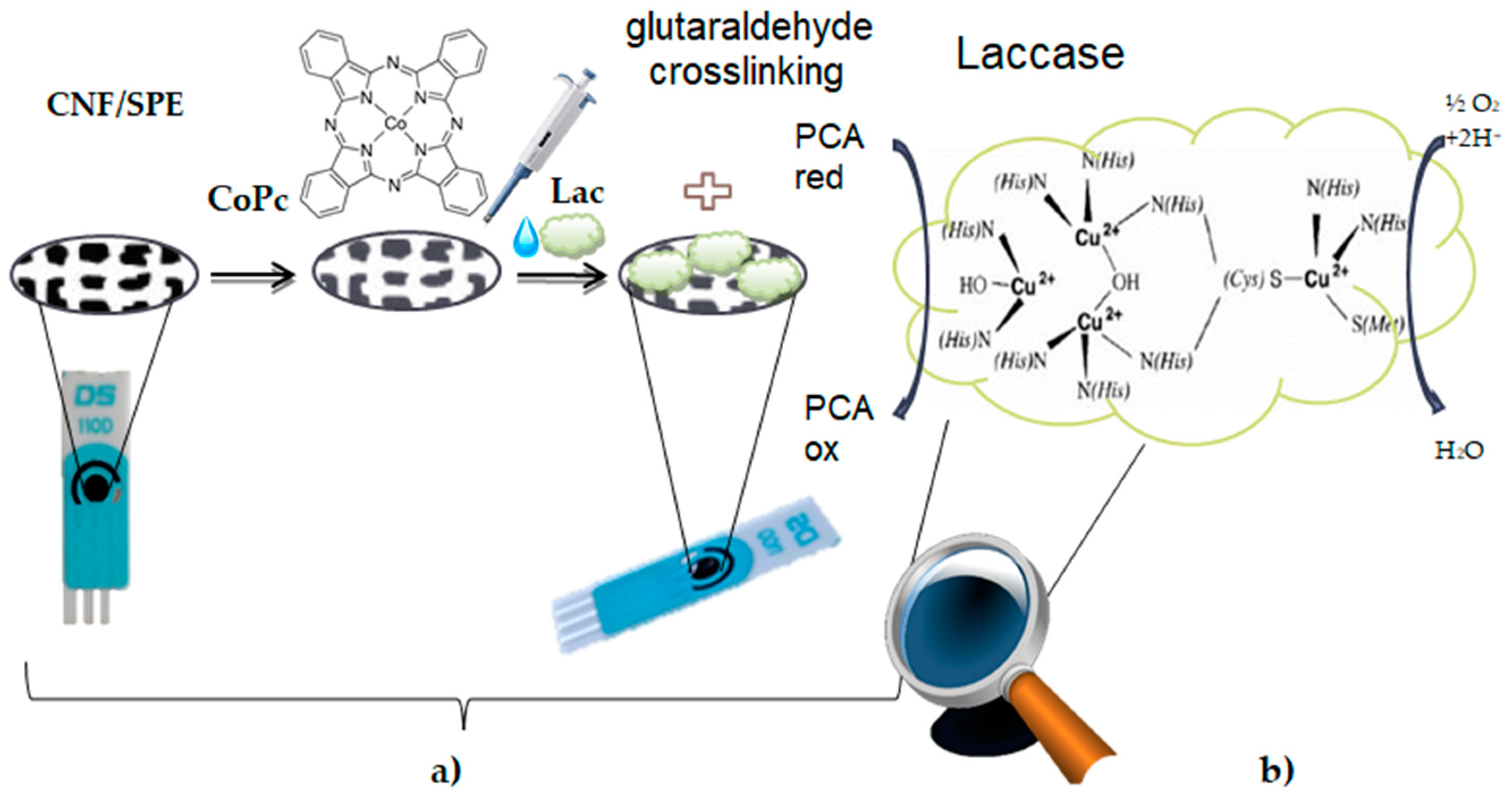
3.3. Preparation of the CNF-CoPc/SPE Biosensor
3.4. Preparation of the CNF-CoPc-Lac/SPE Biosensor
3.5. Methods of Analysis
3.5.1. Cyclic Voltammetry
3.5.2. Square Wave Voltammetry
3.5.3. Chronoamperometry
3.6. Real-Life Samples and Preparation of Testing Solutions
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boz, H. P-Coumaric Acid in Cereals: Presence, Antioxidant and Antimicrobial Effects. Int. J. Food Sci. Technol. 2015, 50, 2323–2328. [Google Scholar] [CrossRef]
- Pei, K.; Ou, J.; Huang, J.; Ou, S. P-Coumaric Acid and Its Conjugates: Dietary Sources, Pharmacokinetic Properties and Biological Activities: P -Coumaric Acid and Its Conjugates. J. Sci. Food Agric. 2016, 96, 2952–2962. [Google Scholar] [CrossRef] [PubMed]
- Vanholme, R.; Storme, V.; Vanholme, B.; Sundin, L.; Christensen, J.H.; Goeminne, G.; Halpin, C.; Rohde, A.; Morreel, K.; Boerjan, W. A Systems Biology View of Responses to Lignin Biosynthesis Perturbations in Arabidopsis. Plant Cell 2012, 24, 3506–3529. [Google Scholar] [CrossRef] [Green Version]
- Achnine, L.; Blancaflor, E.B.; Rasmussen, S.; Dixon, R.A. Colocalization of L-Phenylalanine Ammonia-Lyase and Cinnamate 4-Hydroxylase for Metabolic Channeling in Phenylpropanoid Biosynthesis. Plant Cell 2004, 16, 3098–3109. [Google Scholar] [CrossRef] [PubMed]
- Jendresen, C.B.; Stahlhut, S.G.; Li, M.; Gaspar, P.; Siedler, S.; Förster, J.; Maury, J.; Borodina, I.; Nielsen, A.T. Highly Active and Specific Tyrosine Ammonia-Lyases from Diverse Origins Enable Enhanced Production of Aromatic Compounds in Bacteria and Saccharomyces Cerevisiae. Appl. Environ. Microbiol. 2015, 81, 4458–4476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vargas-Tah, A.; Martínez, L.M.; Hernández-Chávez, G.; Rocha, M.; Martínez, A.; Bolívar, F.; Gosset, G. Production of Cinnamic and P-Hydroxycinnamic Acid from Sugar Mixtures with Engineered Escherichia Coli. Microb. Cell Fact. 2015, 14, 6. [Google Scholar] [CrossRef] [Green Version]
- Vannelli, T.; Wei Qi, W.; Sweigard, J.; Gatenby, A.A.; Sariaslani, F.S. Production of P-Hydroxycinnamic Acid from Glucose in Saccharomyces Cerevisiae and Escherichia Coli by Expression of Heterologous Genes from Plants and Fungi. Metab. Eng. 2007, 9, 142–151. [Google Scholar] [CrossRef]
- Kawai, Y.; Noda, S.; Ogino, C.; Takeshima, Y.; Okai, N.; Tanaka, T.; Kondo, A. P-Hydroxycinnamic Acid Production Directly from Cellulose Using Endoglucanase- and Tyrosine Ammonia Lyase-Expressing Streptomyces Lividans. Microb. Cell Fact. 2013, 12, 45. [Google Scholar] [CrossRef] [Green Version]
- Nijkamp, K.; Westerhof, R.G.M.; Ballerstedt, H.; de Bont, J.A.M.; Wery, J. Optimization of the Solvent-Tolerant Pseudomonas Putida S12 as Host for the Production of p-Coumarate from Glucose. Appl. Microbiol. Biotechnol. 2007, 74, 617–624. [Google Scholar] [CrossRef]
- Kiliç, I.; Yeşiloğlu, Y. Spectroscopic Studies on the Antioxidant Activity of P-Coumaric Acid. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 115, 719–724. [Google Scholar] [CrossRef]
- Lee, S.-J.; Mun, G.-I.; An, S.-M.; Boo, Y.-C. Evidence for the Association of Peroxidases with the Antioxidant Effect of P-Coumaric Acid in Endothelial Cells Exposed to High Glucose plus Arachidonic Acid. BMB Rep. 2009, 42, 561–567. [Google Scholar] [CrossRef] [Green Version]
- Peng, J.; Zheng, T.; Liang, Y.; Duan, L.; Zhang, Y.; Wang, L.-J.; He, G.; Xiao, H. Coumaric Acid Protects Human Lens Epithelial Cells against Oxidative Stress-Induced Apoptosis by MAPK Signaling. Oxid. Med. Cell. Longev. 2018, 2018, 8549052. [Google Scholar] [CrossRef] [Green Version]
- Lou, Z.; Wang, H.; Rao, S.; Sun, J.; Ma, C.; Li, J. P-Coumaric Acid Kills Bacteria through Dual Damage Mechanisms. Food Control 2012, 25, 550–554. [Google Scholar] [CrossRef]
- Boo, Y.C. P-Coumaric Acid as An Active Ingredient in Cosmetics: A Review Focusing on Its Antimelanogenic Effects. Antioxidants 2019, 8, 275. [Google Scholar] [CrossRef] [Green Version]
- Contardi, M.; Heredia-Guerrero, J.A.; Guzman-Puyol, S.; Summa, M.; Benítez, J.J.; Goldoni, L.; Caputo, G.; Cusimano, G.; Picone, P.; Carlo, M.D.; et al. Combining Dietary Phenolic Antioxidants with Polyvinylpyrrolidone: Transparent Biopolymer Films Based on p-Coumaric Acid for Controlled Release. J. Mater. Chem. B 2019, 7, 1384–1396. [Google Scholar] [CrossRef] [PubMed]
- Pragasam, S.J.; Venkatesan, V.; Rasool, M. Immunomodulatory and Anti-Inflammatory Effect of p-Coumaric Acid, a Common Dietary Polyphenol on Experimental Inflammation in Rats. Inflammation 2013, 36, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Kheiry, M.; Dianat, M.; Badavi, M.; Mard, S.A.; Bayati, V. P-Coumaric Acid Attenuates Lipopolysaccharide-Induced Lung Inflammation in Rats by Scavenging ROS Production: An In Vivo and In Vitro Study. Inflammation 2019, 42, 1939–1950. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Lim, D.; Kim, J. P-Coumaric Acid, a Major Active Compound of Bambusae Caulis in Taeniam, Suppresses Cigarette Smoke-Induced Pulmonary Inflammation. Am. J. Chin. Med. 2018, 46, 407–421. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, T.; Thi, T.H.; Shi, D.J.; Akashi, M. Environmentally Degradable, High-Performance Thermoplastics from Phenolic Phytomonomers. Nat. Mater. 2006, 5, 966–970. [Google Scholar] [CrossRef]
- Navaneethan, D.; Rasool, M. P-Coumaric Acid, a Common Dietary Polyphenol, Protects Cadmium Chloride-Induced Nephrotoxicity in Rats. Ren. Fail. 2014, 36, 244–251. [Google Scholar] [CrossRef]
- Scheepens, A.; Bisson, J.-F.; Skinner, M. P-Coumaric Acid Activates the GABA-A Receptor In Vitro and Is Orally Anxiolytic In Vivo: P-COUMARIC ACID IS GABAERGIC AND ANXIOLYTIC IN RODENTS. Phytother. Res. 2014, 28, 207–211. [Google Scholar] [CrossRef]
- Sakamula, R.; Thong-asa, W. Neuroprotective Effect of P-Coumaric Acid in Mice with Cerebral Ischemia Reperfusion Injuries. Metab. Brain Dis. 2018, 33, 765–773. [Google Scholar] [CrossRef]
- Cha, H.; Lee, S.; Lee, J.H.; Park, J.-W. Protective Effects of P-Coumaric Acid against Acetaminophen-Induced Hepatotoxicity in Mice. Food Chem. Toxicol. 2018, 121, 131–139. [Google Scholar] [CrossRef]
- Yoon, H.S.; Lee, N.-H.; Hyun, C.-G.; Shin, D.-B. Differential Effects of Methoxylated P-Coumaric Acids on Melanoma in B16/F10 Cells. JFN 2015, 20, 73–77. [Google Scholar] [CrossRef] [Green Version]
- Kawaguchi, H.; Katsuyama, Y.; Danyao, D.; Kahar, P.; Nakamura-Tsuruta, S.; Teramura, H.; Wakai, K.; Yoshihara, K.; Minami, H.; Ogino, C.; et al. Caffeic Acid Production by Simultaneous Saccharification and Fermentation of Kraft Pulp Using Recombinant Escherichia Coli. Appl. Microbiol. Biotechnol. 2017, 101, 5279–5290. [Google Scholar] [CrossRef]
- Zhao, S.; Jones, J.A.; Lachance, D.M.; Bhan, N.; Khalidi, O.; Venkataraman, S.; Wang, Z.; Koffas, M.A.G. Improvement of Catechin Production in Escherichia Coli through Combinatorial Metabolic Engineering. Metab. Eng. 2015, 28, 43–53. [Google Scholar] [CrossRef]
- Li, M.; Kildegaard, K.R.; Chen, Y.; Rodriguez, A.; Borodina, I.; Nielsen, J. De Novo Production of Resveratrol from Glucose or Ethanol by Engineered Saccharomyces Cerevisiae. Metab. Eng. 2015, 32, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zha, J.; Koffas, M.A.G. Production of Anthocyanins in Metabolically Engineered Microorganisms: Current Status and Perspectives. Synth. Syst. Biotechnol. 2017, 2, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.S.L.; Boyce, M.C.; Breadmore, M.C. A Rapid Quantitative Determination of Phenolic Acids in Brassica Oleracea by Capillary Zone Electrophoresis. Food Chem. 2011, 127, 797–801. [Google Scholar] [CrossRef] [PubMed]
- Martí, R.; Valcárcel, M.; Herrero-Martínez, J.M.; Cebolla-Cornejo, J.; Roselló, S. Simultaneous Determination of Main Phenolic Acids and Flavonoids in Tomato by Micellar Electrokinetic Capillary Electrophoresis. Food Chem. 2017, 221, 439–446. [Google Scholar] [CrossRef] [Green Version]
- Aguilar-Hernández, I.; Afseth, N.K.; López-Luke, T.; Contreras-Torres, F.F.; Wold, J.P.; Ornelas-Soto, N. Surface Enhanced Raman Spectroscopy of Phenolic Antioxidants: A Systematic Evaluation of Ferulic Acid, p-Coumaric Acid, Caffeic Acid and Sinapic Acid. Vib. Spectrosc. 2017, 89, 113–122. [Google Scholar] [CrossRef]
- Pironi, A.M.; de Araújo, P.R.; Fernandes, M.A.; Salgado, H.R.N.; Chorilli, M. Characteristics, Biological Properties and Analytical Methods of Ursolic Acid: A Review. Crit. Rev. Anal. Chem. 2018, 48, 86–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Apetrei, I.M.; Apetrei, C. Study of Different Carbonaceous Materials as Modifiers of Screen-Printed Electrodes for Detection of Catecholamines. IEEE Sens. J. 2015, 15, 3094–3101. [Google Scholar] [CrossRef]
- Apetrei, C.; Apetrei, I.M. Biosensor Based on Tyrosinase Immobilized on a Single-Walled Carbon Nanotube-Modified Glassy Carbon Electrode for Detection of Epinephrine. IJN 2013, 4391. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Apetrei, I.; Apetrei, C. Amperometric Biosensor Based on Diamine Oxidase/Platinum Nanoparticles/Graphene/Chitosan Modified Screen-Printed Carbon Electrode for Histamine Detection. Sensors 2016, 16, 422. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Masek, A.; Chrzescijanska, E.; Latos, M. Determination of Antioxidant Activity of Caffeic Acid and P-Coumaric Acid by Using Electrochemical and Spectrophotometric Assays. Int. J. Electrochem. Sci. 2016, 11, 10644–10658. [Google Scholar] [CrossRef]
- Ziyatdinova, G.; Aytuganova, I.; Nizamova, A.; Budnikov, H. Differential Pulse Voltammetric Assay of Coffee Antioxidant Capacity with MWNT-Modified Electrode. Food Anal. Methods 2013, 6, 1629–1638. [Google Scholar] [CrossRef]
- Şenocak, A. Fast, Simple and Sensitive Determination of Coumaric Acid in Fruit Juice Samples by Magnetite Nanoparticles-zeolitic Imidazolate Framework Material. Electroanalysis 2020, 32, 2330–2339. [Google Scholar] [CrossRef]
- Sanger, K.; Zór, K.; Bille Jendresen, C.; Heiskanen, A.; Amato, L.; Toftgaard Nielsen, A.; Boisen, A. Lab-on-a-Disc Platform for Screening of Genetically Modified E. Coli Cells via Cell-Free Electrochemical Detection of p-Coumaric Acid. Sens. Actuators B Chem. 2017, 253, 999–1005. [Google Scholar] [CrossRef] [Green Version]
- Giardina, P.; Faraco, V.; Pezzella, C.; Piscitelli, A.; Vanhulle, S.; Sannia, G. Laccases: A Never-Ending Story. Cell. Mol. Life Sci. 2010, 67, 369–385. [Google Scholar] [CrossRef]
- Salamanca-Neto, C.A.R.; Marcheafave, G.G.; Scremin, J.; Barbosa, E.C.M.; Camargo, P.H.C.; Dekker, R.F.H.; Scarminio, I.S.; Barbosa-Dekker, A.M.; Sartori, E.R. Chemometric-Assisted Construction of a Biosensing Device to Measure Chlorogenic Acid Content in Brewed Coffee Beverages to Discriminate Quality. Food Chem. 2020, 315, 126306. [Google Scholar] [CrossRef]
- Kunene, K.; Sabela, M.; Kanchi, S.; Bisetty, K. High Performance Electrochemical Biosensor for Bisphenol a Using Screen Printed Electrodes Modified with Multiwalled Carbon Nanotubes Functionalized with Silver-Doped Zinc Oxide. Waste Biomass Valor. 2020, 11, 1085–1096. [Google Scholar] [CrossRef]
- Romero-Arcos, M.; Garnica-Romo, M.; Martínez-Flores, H. Electrochemical Study and Characterization of an Amperometric Biosensor Based on the Immobilization of Laccase in a Nanostructure of TiO2 Synthesized by the Sol-Gel Method. Materials 2016, 9, 543. [Google Scholar] [CrossRef] [Green Version]
- Pavinatto, A.; Mercante, L.A.; Facure, M.H.M.; Pena, R.B.; Sanfelice, R.C.; Mattoso, L.H.C.; Correa, D.S. Ultrasensitive Biosensor Based on Polyvinylpyrrolidone/Chitosan/Reduced Graphene Oxide Electrospun Nanofibers for 17α—Ethinylestradiol Electrochemical Detection. Appl. Surf. Sci. 2018, 458, 431–437. [Google Scholar] [CrossRef]
- Arduini, F.; Cinti, S.; Caratelli, V.; Amendola, L.; Palleschi, G.; Moscone, D. Origami Multiple Paper-Based Electrochemical Biosensors for Pesticide Detection. Biosens. Bioelectron. 2019, 126, 346–354. [Google Scholar] [CrossRef]
- Chaiyo, S.; Mehmeti, E.; Siangproh, W.; Hoang, T.L.; Nguyen, H.P.; Chailapakul, O.; Kalcher, K. Non-Enzymatic Electrochemical Detection of Glucose with a Disposable Paper-Based Sensor Using a Cobalt Phthalocyanine–Ionic Liquid–Graphene Composite. Biosens. Bioelectron. 2018, 102, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Kunpatee, K.; Chamsai, P.; Mehmeti, E.; Stankovic, D.M.; Ortner, A.; Kalcher, K.; Samphao, A. A Highly Sensitive Fenobucarb Electrochemical Sensor Based on Graphene Nanoribbons-Ionic Liquid-Cobalt Phthalocyanine Composites Modified on Screen-Printed Carbon Electrode Coupled with a Flow Injection Analysis. J. Electroanal. Chem. 2019, 855, 113630. [Google Scholar] [CrossRef]
- Zhao, K.; Veksha, A.; Ge, L.; Lisak, G. Near Real-Time Analysis of Para-Cresol in Wastewater with a Laccase-Carbon Nanotube-Based Biosensor. Chemosphere 2021, 269, 128699. [Google Scholar] [CrossRef] [PubMed]
- Nazari, M.; Kashanian, S.; Rafipour, R. Laccase Immobilization on the Electrode Surface to Design a Biosensor for the Detection of Phenolic Compound Such as Catechol. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 145, 130–138. [Google Scholar] [CrossRef]
- Bounegru, A.V.; Apetrei, C. Development of a Novel Electrochemical Biosensor Based on Carbon Nanofibers–Gold Nanoparticles–Tyrosinase for the Detection of Ferulic Acid in Cosmetics. Sensors 2020, 20, 6724. [Google Scholar] [CrossRef]
- Apetrei, I.M.; Rodriguez-Mendez, M.L.; Apetrei, C.; de Saja, J.A. Enzyme Sensor Based on Carbon Nanotubes/Cobalt(II) Phthalocyanine and Tyrosinase Used in Pharmaceutical Analysis. Sens. Actuators B Chem. 2013, 177, 138–144. [Google Scholar] [CrossRef]
- Portaccio, M.; Di Tuoro, D.; Arduini, F.; Moscone, D.; Cammarota, M.; Mita, D.G.; Lepore, M. Laccase Biosensor Based on Screen-Printed Electrode Modified with Thionine–Carbon Black Nanocomposite, for Bisphenol A Detection. Electrochim. Acta 2013, 109, 340–347. [Google Scholar] [CrossRef]
- Caro, C. Electrocatalytic Oxidation of Nitrite on a Vitreous Carbon Electrode Modified with Cobalt Phthalocyanine. Electrochim. Acta 2002, 47, 1489–1494. [Google Scholar] [CrossRef]
- Bounegru, A.; Apetrei, C. Voltammetric Sensors Based on Nanomaterials for Detection of Caffeic Acid in Food Supplements. Chemosensors 2020, 8, 41. [Google Scholar] [CrossRef]
- Thakurathi, M.; Gurung, E.; Cetin, M.M.; Thalangamaarachchige, V.D.; Mayer, M.F.; Korzeniewski, C.; Quitevis, E.L. The Stokes-Einstein Equation and the Diffusion of Ferrocene in Imidazolium-Based Ionic Liquids Studied by Cyclic Voltammetry: Effects of Cation Ion Symmetry and Alkyl Chain Length. Electrochim. Acta 2018, 259, 245–252. [Google Scholar] [CrossRef]
- Cancelliere, R.; Carbone, K.; Pagano, M.; Cacciotti, I.; Micheli, L. Biochar from Brewers’ Spent Grain: A Green and Low-Cost Smart Material to Modify Screen-Printed Electrodes. Biosensors 2019, 9, 139. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Delgado, M.M.; Alemán-Nava, G.S.; Rodríguez-Delgado, J.M.; Dieck-Assad, G.; Martínez-Chapa, S.O.; Barceló, D.; Parra, R. Laccase-Based Biosensors for Detection of Phenolic Compounds. TrAC Trends Anal. Chem. 2015, 74, 21–45. [Google Scholar] [CrossRef] [Green Version]
- Casero, E.; Petit-Domínguez, M.D.; Vázquez, L.; Ramírez-Asperilla, I.; Parra-Alfambra, A.M.; Pariente, F.; Lorenzo, E. Laccase Biosensors Based on Different Enzyme Immobilization Strategies for Phenolic Compounds Determination. Talanta 2013, 115, 401–408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ribeiro, F.W.P.; Barroso, M.F.; Morais, S.; Viswanathan, S.; de Lima-Neto, P.; Correia, A.N.; Oliveira, M.B.P.P.; Delerue-Matos, C. Simple Laccase-Based Biosensor for Formetanate Hydrochloride Quantification in Fruits. Bioelectrochemistry 2014, 95, 7–14. [Google Scholar] [CrossRef]
- Oliveira-Neto, J.R.; Rezende, S.G.; de Fátima Reis, C.; Benjamin, S.R.; Rocha, M.L.; de Souza Gil, E. Electrochemical Behavior and Determination of Major Phenolic Antioxidants in Selected Coffee Samples. Food Chem. 2016, 190, 506–512. [Google Scholar] [CrossRef]
- Cadorin Fernandes, S.; Cruz Vieira, I.; Barbosa, A.M.J.; Souza Ferreira, V. Methomyl Detection by Inhibition of Laccase Using a Carbon Ceramic Biosensor. Electroanalysis 2011, 23, 1623–1630. [Google Scholar] [CrossRef]
- Zapp, E.; Brondani, D.; Vieira, I.C.; Scheeren, C.W.; Dupont, J.; Barbosa, A.M.J.; Ferreira, V.S. Biomonitoring of Methomyl Pesticide by Laccase Inhibition on Sensor Containing Platinum Nanoparticles in Ionic Liquid Phase Supported in Montmorillonite. Sens. Actuators B Chem. 2011, 155, 331–339. [Google Scholar] [CrossRef]
- Janeiro, P.; Novak, I.; Seruga, M.; Maria Oliveira-Brett, A. Electroanalytical Oxidation of p-Coumaric Acid. Anal. Lett. 2007, 40, 3309–3321. [Google Scholar] [CrossRef]
- Centeno, D.A.; Solano, X.H.; Castillo, J.J. A New Peroxidase from Leaves of Guinea Grass (Panicum Maximum): A Potential Biocatalyst to Build Amperometric Biosensors. Bioelectrochemistry 2017, 116, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Apetrei, R.-M.; Cârâc, G.; Bahrim, G.; Camurlu, P. Sensitivity Enhancement for Microbial Biosensors through Cell Self-Coating with Polypyrrole. Int. J. Polym. Mater. Polym. Biomater. 2019, 68, 1058–1067. [Google Scholar] [CrossRef]
- Vilian, A.T.E.; Chen, S.-M. Preparation of Carbon Nanotubes Decorated with Manganese Dioxide Nanoparticles for Electrochemical Determination of Ferulic Acid. Microchim. Acta 2015, 182, 1103–1111. [Google Scholar] [CrossRef]
- Mantzavinos, D.; Hellenbrand, R.; Metcalfe, I.S.; Livingston, A.G. Partial Wet Oxidation of P-Coumaric Acid: Oxidation Intermediates, Reaction Pathways and Implications for Wastewater Treatment. Water Res. 1996, 30, 2969–2976. [Google Scholar] [CrossRef]
- Yaqoob, H.; Kamran, M.; Ali, S. Innovative Roles of Laccase Mediator System in Nano Biotechnology, Bio Bleaching and Bioremediation. TrAC Trends Anal. Chem. 2015, 12, 21–45. [Google Scholar]
- Sousa, W.R.; da Rocha, C.; Cardoso, C.L.; Silva, D.H.S.; Zanoni, M.V.B. Determination of the Relative Contribution of Phenolic Antioxidants in Orange Juice by Voltammetric Methods. J. Food Compos. Anal. 2004, 17, 619–633. [Google Scholar] [CrossRef]
- Santos, D.P.; Bergamini, M.F.; Fogg, A.G.; Zanoni, M.V.B. Application of a Glassy Carbon Electrode Modified with Poly (Glutamic Acid) in Caffeic Acid Determination. Microchim. Acta 2005, 151, 127–134. [Google Scholar] [CrossRef]
- Kumar, S.V.S.; Phale, P.S.; Durani, S.; Wangikar, P.P. Combined Sequence and Structure Analysis of the Fungal Laccase Family. Biotechnol. Bioeng. 2003, 83, 386–394. [Google Scholar] [CrossRef]
- Mate, D.M.; Alcalde, M. Laccase Engineering: From Rational Design to Directed Evolution. Biotechnol. Adv. 2015, 33, 25–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Lv, Z.; Zhou, J.; Xin, F.; Ma, J.; Wu, H.; Fang, Y.; Jiang, M.; Dong, W. Application of Eukaryotic and Prokaryotic Laccases in Biosensor and Biofuel Cells: Recent Advances and Electrochemical Aspects. Appl. Microbiol. Biotechnol. 2018, 102, 10409–10423. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Gopal Khan, G. Synthesis of BiFeO3 Nanoparticle Anchored TiO2-BiFeO3 Nano-Heterostructure and Exploring Its Different Electrochemical Aspects as Electrode. Mater. Today Proc. 2018, 5, 10177–10184. [Google Scholar] [CrossRef]
- Dinu, A.; Apetrei, C. Voltammetric Determination of Phenylalanine Using Chemically Modified Screen-Printed Based Sensors. Chemosensors 2020, 8, 113. [Google Scholar] [CrossRef]
- Li, G.; Sun, K.; Li, D.; Lv, P.; Wang, Q.; Huang, F.; Wei, Q. Biosensor Based on Bacterial Cellulose-Au Nanoparticles Electrode Modified with Laccase for Hydroquinone Detection. Colloids Surf. A Physicochem. Eng. Asp. 2016, 509, 408–414. [Google Scholar] [CrossRef]
- Li, D.; Lv, P.; Zhu, J.; Lu, Y.; Chen, C.; Zhang, X.; Wei, Q. NiCu Alloy Nanoparticle-Loaded Carbon Nanofibers for Phenolic Biosensor Applications. Sensors 2015, 15, 29419–29433. [Google Scholar] [CrossRef] [Green Version]
- Sánchez-Calvo, A.; Costa-García, A.; Blanco-López, M.C. Paper-Based Electrodes Modified with Cobalt Phthalocyanine Colloid for the Determination of Hydrogen Peroxide and Glucose. Analyst 2020, 145, 2716–2724. [Google Scholar] [CrossRef]
- Zheng, L.; Song, J. Curcumin Multi-Wall Carbon Nanotubes Modified Glassy Carbon Electrode and Its Electrocatalytic Activity towards Oxidation of Hydrazine. Sens. Actuators B Chem. 2009, 135, 650–655. [Google Scholar] [CrossRef]
- Almeida, L.C.; Correia, R.D.; Squillaci, G.; Morana, A.; La Cara, F.; Correia, J.P.; Viana, A.S. Electrochemical Deposition of Bio-Inspired Laccase-Polydopamine Films for Phenolic Sensors. Electrochim. Acta 2019, 319, 462–471. [Google Scholar] [CrossRef]
- Albayati, S.A.R.; Kashanian, S.; Nazari, M.; Rezaei, S. Novel Fabrication of a Laccase Biosensor to Detect Phenolic Compounds Using a Carboxylated Multiwalled Carbon Nanotube on the Electropolymerized Support. Bull. Mater. Sci. 2019, 42, 187. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Li, D.; Fu, J.; Huang, F.; Wei, Q. TiO2-CuCNFs Based Laccase Biosensor for Enhanced Electrocatalysis in Hydroquinone Detection. J. Electroanal. Chem. 2016, 766, 16–23. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, D.; Li, Z.; Sun, X.; Gao, T.; Zhou, G. Laccase Biosensor Fabricated on Flower–Shaped Yolk–Shell SiO2 Nanospheres for Catechol Detection. Colloids Surf. A Physicochem. Eng. Asp. 2018, 538, 202–209. [Google Scholar] [CrossRef]
- Djurdjić, S.; Stanković, V.; Vlahović, F.; Ognjanović, M.; Kalcher, K.; Veličković, T.Ć.; Mutić, J.; Stanković, D.M. Laccase Polyphenolic Biosensor Supported on MnO2@ GNP Decorated SPCE: Preparation, Characterization, and Analytical Application. J. Electrochem. Soc. 2021, 168, 037510. [Google Scholar]
- Apetrei, I.; Apetrei, C. Development of a Novel Biosensor Based on Tyrosinase/Platinum Nanoparticles/Chitosan/Graphene Nanostructured Layer with Applicability in Bioanalysis. Materials 2019, 12, 1009. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Apetrei, I.M.; Rodriguez-Mendez, M.L.; Apetrei, C.; de Saja, J.A. Fish Freshness Monitoring Using an E-Tongue Based on Polypyrrole Modified Screen-Printed Electrodes. IEEE Sens. J. 2013, 13, 2548–2554. [Google Scholar] [CrossRef]
- Apetrei, I.M.; Apetrei, C. Voltammetric Determination of Melatonin Using a Graphene-Based Sensor in Pharmaceutical Products. Int. J. Nanomed. 2016, 11, 1859–1866. [Google Scholar] [CrossRef] [PubMed] [Green Version]


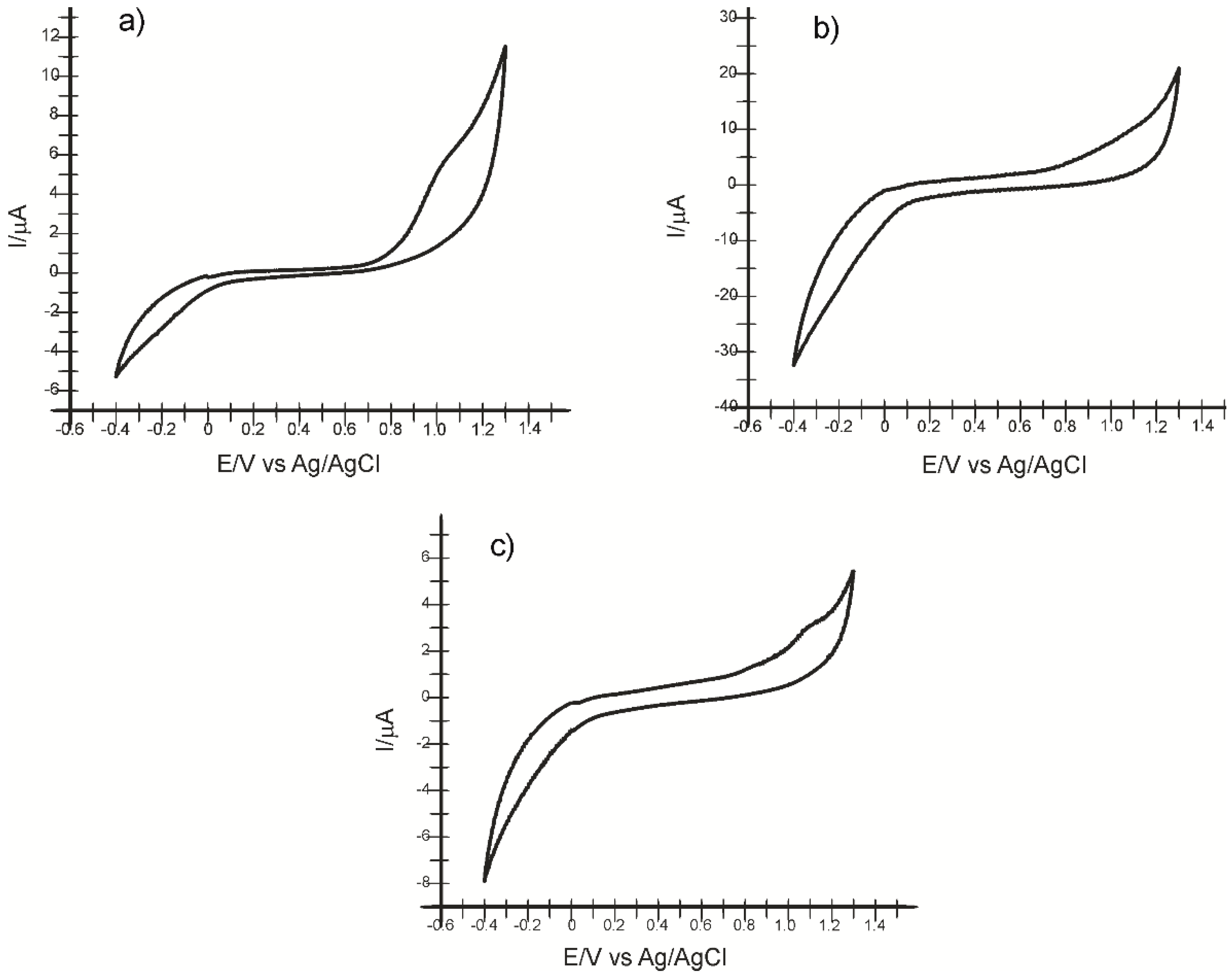

| Electrode | Epa1 (V) | Epox2 (V) | Epc3 (V) | E1/24 (V) | Ipa5 (µA) | Ipox6 (µA) | Ipc7 (µA) | Ipc/Ipox |
|---|---|---|---|---|---|---|---|---|
| CNF/SPE | 0.814 | 0.523 | 0.160 | 0.341 | 171.170 | 30.300 | −20.219 | 0.669 |
| CNF-CoPc/SPE | 0.818 | 0.489 | 0.079 | 0.284 | 96.208 | 19.650 | −25.354 | 1.290 |
| CNF-CoPc-Lac/SPE | 0.904 | 0.537 | 0.011 | 0.274 | 147.964 | 40.269 | −52.506 | 1.303 |
| Electrode | Equation | R2 | Γ (mol × cm−2) |
|---|---|---|---|
| CNF/SPE | Ipc = −197.46 × 10−6 v −7.5975 × 10−6 | 0.996 | 3.84 × 10−10 |
| CNF-CoPc/SPE | Ipc = −229.75 × 10−6 v −11.029 × 10−6 | 0.999 | 4.46 × 10−10 |
| CNF-CoPc-Lac/SPE | Ipc = −310.01 × 10−6 v −41.585 × 10−6 | 0.982 | 6.02 × 10−10 |
| Electrode | Linear Equation | R2 | LOD (M) | LOQ (M) |
|---|---|---|---|---|
| CNF-CoPc-Lac/SPE | Ipc = −0.0247c − 0.4188 | 0.9714 | 4.83 × 10−7 | 1.61 × 10−6 |
| Laccase Biosensor | Analyte | Detection Technique | Linearity Range (µM) | LOD (M) | Ref. |
|---|---|---|---|---|---|
| Lac/Ag-ZnONPs/MWCNTs/C-SPE | Bisphenol A | CV | 0.5–2.99 | 6.0×10−9 | [42] |
| ePDA-Lac | Caffeic acid | CV | 1–50 | 1.4 × 10−7 | [80] |
| Rosmarinic acid | 1–20 | 9.0 × 10−8 | |||
| Gallic acid | 1–150 | 2.9 × 10−7 | |||
| Lac/MWCNT-COOH/AuNPs-SDBS-PEDOT/GCE | Catechol | DPV | 0.1–0.5 11.99–94.11 | 1.1 × 10−7 1.22 × 10−5 | [81] |
| Nafion-TiO2/CuCNFs-Lac-GCE | Hydroquinone | Amp | 1–89.8 | 3.65 × 10−6 | [82] |
| FYSSns–2–Lac/GCE | Catechol | DPV | 12.5–450 | 1.6 × 10−6 | [83] |
| PtNPs-BOT-Lac/GO | 5-CQA | SWV | 0.56–7.3 | 1.8 × 10−7 | [41] |
| GNP@MnO2 (SPCE/GNP@MnO2) | caffeic acid | Amp | - | 1.9 × 10−6 | [84] |
| CNF-CoPc-Lac/SPE | p-coumaric acid | CV | 0.4–6.4 | 4.83 × 10−7 | This work |
| Interfering Compound | Ratio | Recovery/% | Ratio | Recovery/% | Ratio | Recovery/% |
|---|---|---|---|---|---|---|
| ascorbic acid | 1:1 | 101 ± 3.6 | 1:10 | 99 ± 2.6 | 1:20 | 98 ± 2.8 |
| ferrulic acid | 1:1 | 99 ± 2.7 | 1:10 | 98 ± 3.1 | 1:20 | 97 ± 3.3 |
| vanillic acid | 1:1 | 98 ± 3.9 | 1:10 | 96 ± 4.0 | 1:20 | 95 ± 3.6 |
| gallic acid | 1:1 | 102 ± 3.7 | 1:10 | 100 ± 2.2 | 1:20 | 99 ± 3.2 |
| Phytopreparations | FTIR Method mg/g PCA | Voltammetric Method mg/g PCA |
|---|---|---|
| Spirulin | 1.569 | 1.674 |
| Ghindazin | 0.644 | 0.783 |
| Tuiazin | 1.936 | 2.149 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bounegru, A.V.; Apetrei, C. Development of a Novel Electrochemical Biosensor Based on Carbon Nanofibers–Cobalt Phthalocyanine–Laccase for the Detection of p-Coumaric Acid in Phytoproducts. Int. J. Mol. Sci. 2021, 22, 9302. https://doi.org/10.3390/ijms22179302
Bounegru AV, Apetrei C. Development of a Novel Electrochemical Biosensor Based on Carbon Nanofibers–Cobalt Phthalocyanine–Laccase for the Detection of p-Coumaric Acid in Phytoproducts. International Journal of Molecular Sciences. 2021; 22(17):9302. https://doi.org/10.3390/ijms22179302
Chicago/Turabian StyleBounegru, Alexandra Virginia, and Constantin Apetrei. 2021. "Development of a Novel Electrochemical Biosensor Based on Carbon Nanofibers–Cobalt Phthalocyanine–Laccase for the Detection of p-Coumaric Acid in Phytoproducts" International Journal of Molecular Sciences 22, no. 17: 9302. https://doi.org/10.3390/ijms22179302
APA StyleBounegru, A. V., & Apetrei, C. (2021). Development of a Novel Electrochemical Biosensor Based on Carbon Nanofibers–Cobalt Phthalocyanine–Laccase for the Detection of p-Coumaric Acid in Phytoproducts. International Journal of Molecular Sciences, 22(17), 9302. https://doi.org/10.3390/ijms22179302








