Microglia and the Aging Brain: Are Geriatric Microglia Linked to Poor Sleep Quality?
Abstract
1. Introduction
2. Microglia and Their Role in Normal Adult Brain
3. Sleep–Wake Cycle Impact on Microglia–Synapse Interaction
4. Diurnal Rhythmicity in Microglial Structure
5. Diurnal Rhythmicity in Microglial Interactions with Neurons and Neuronal Elements
6. Diurnal Rhythmicity in the Expression of Genes Related to Microglial Phagocytosis
7. Diurnal Rhythmicity in the Expression of Genes Related to Microglial Inflammation
8. Extrinsic Cellular Factors behind Microglial Rhythmicity
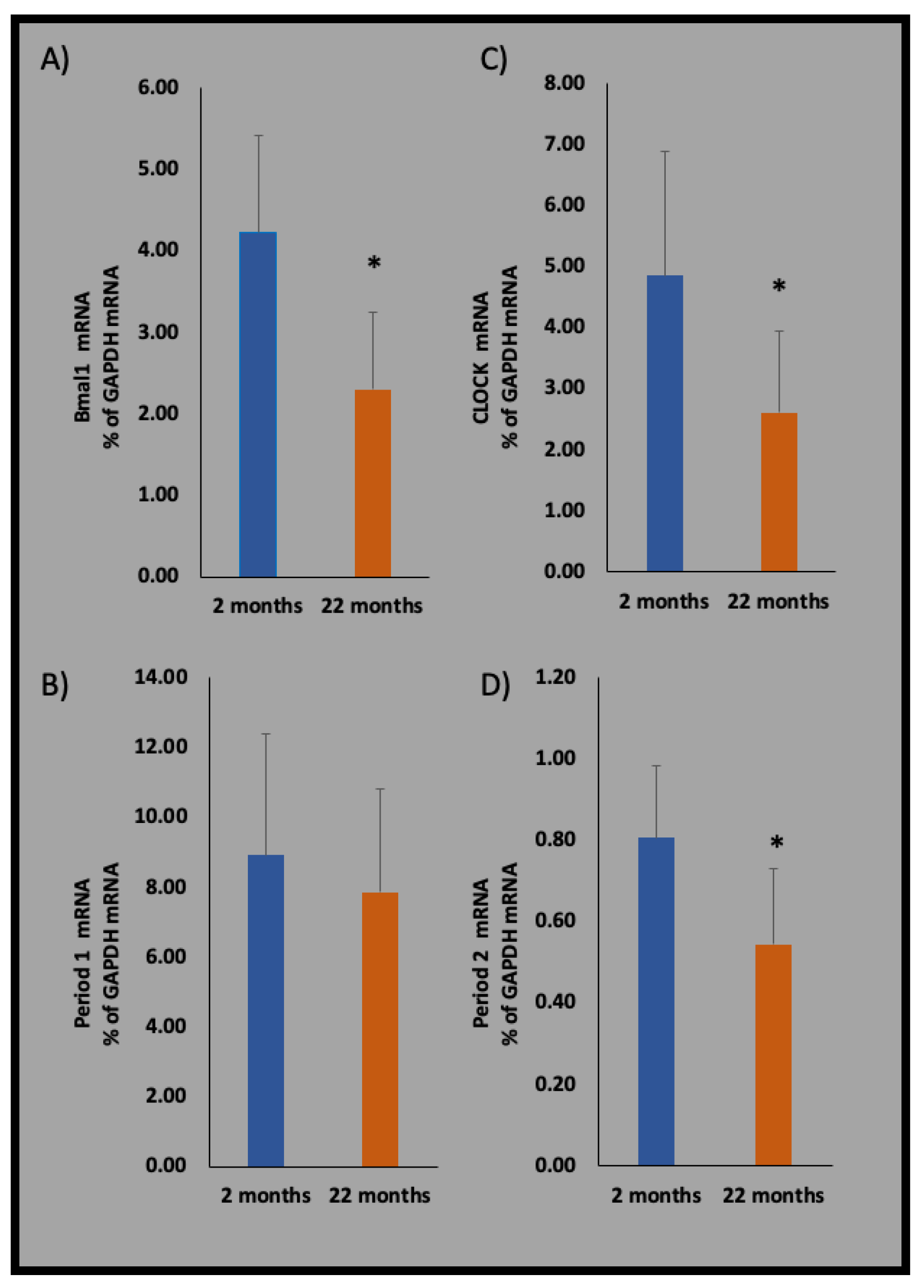
9. Intrinsic Molecular Clocks behind Microglial Rhythmicity
10. Alteration of Clock Genes Changes the Microglial Phenotype
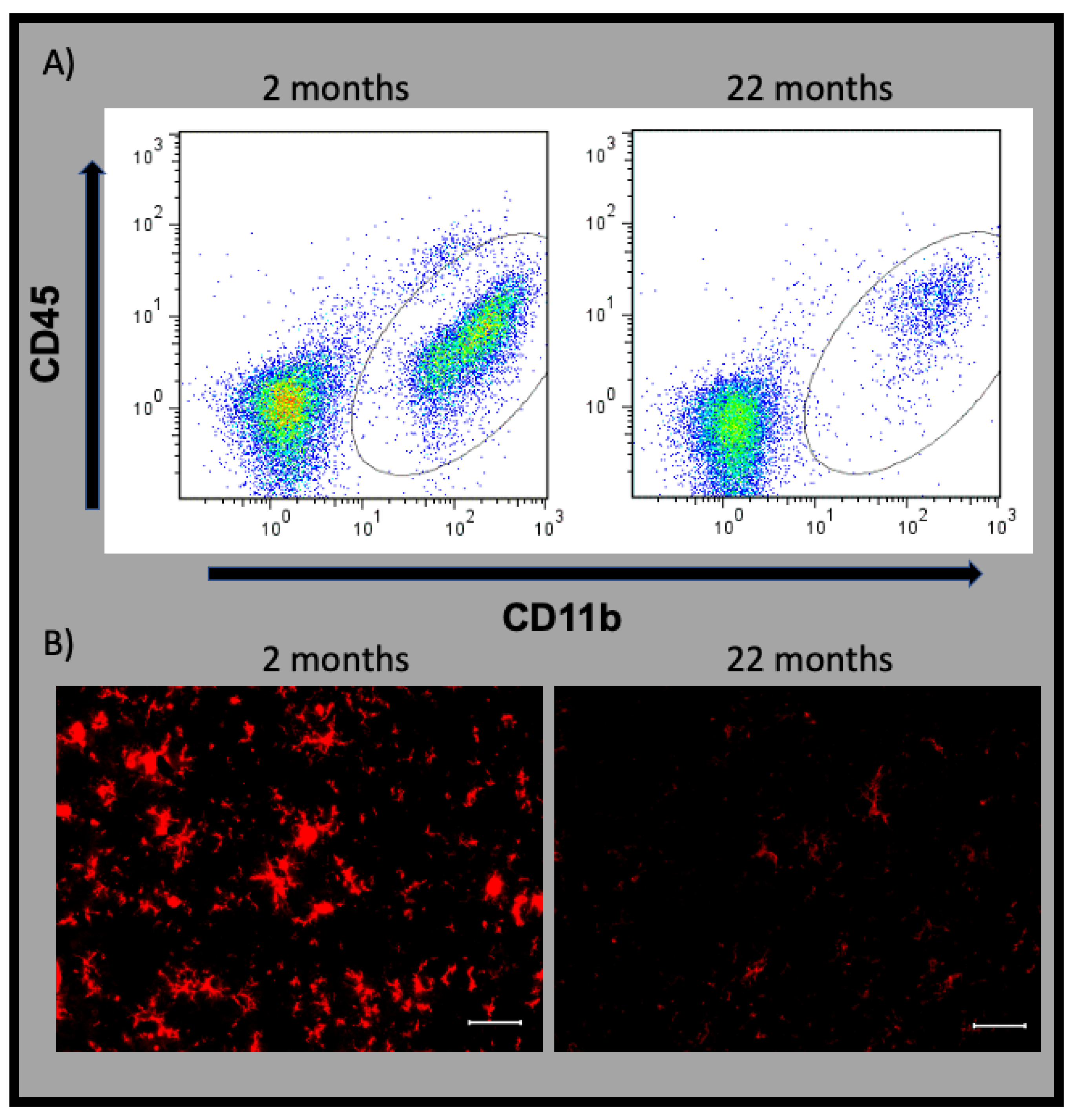
11. Senescent Microglia
12. Diurnal Rhythmicity of Senescent Microglia
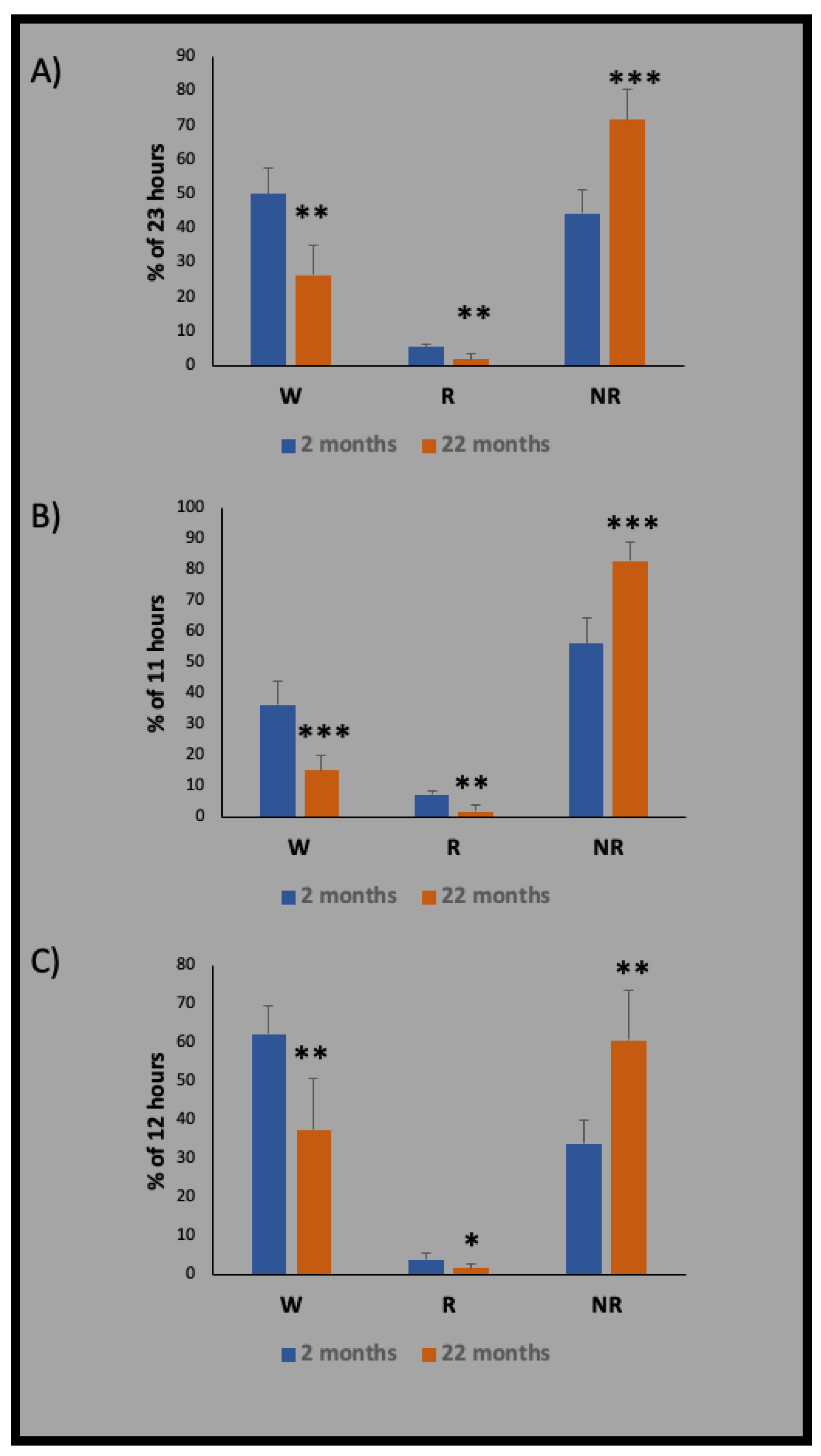
13. Sleep Characteristics in Aged Rodents
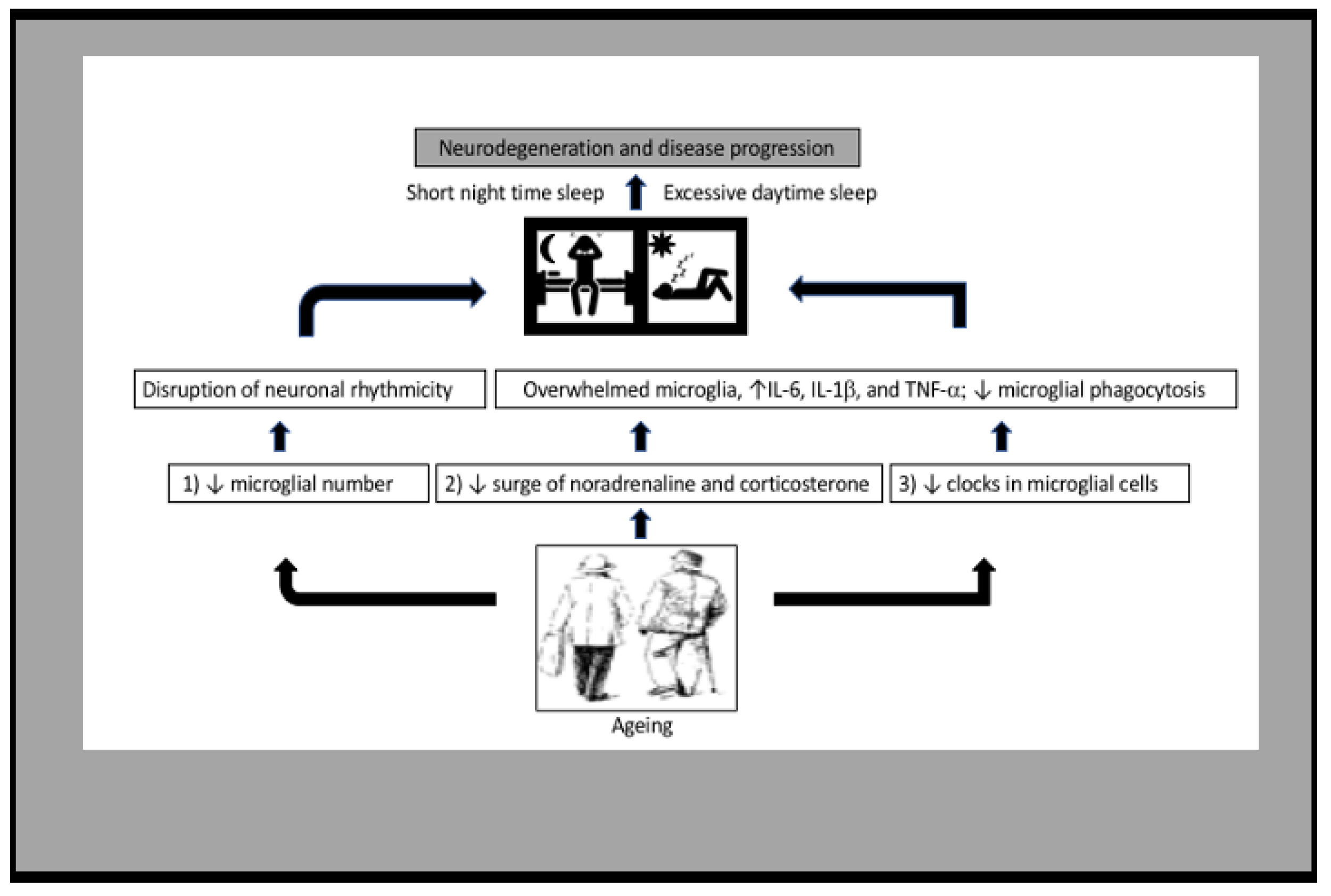
14. Senescent Microglia and Their Impact on Sleep
15. Effect of Sleep Loss on Microglial Senescence
16. Effects of Commonly Used Drugs for Sleep Disorders on Microglial Performances
17. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Damoiseaux, J.S. Effects of aging on functional and structural brain connectivity. Neuroimage 2017, 160, 32–40. [Google Scholar] [CrossRef]
- Pierce, A.L.; Bullain, S.S.; Kawas, C.H. Late-Onset Alzheimer Disease. Neurol. Clin. 2017, 35, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Tworoger, S.S.; Lee, S.; Schernhammer, E.S.; Grodstein, F. The association of self-reported sleep duration, difficulty sleeping, and snoring with cognitive function in older women. Alzheimer Dis. Assoc. Disord. 2006, 20, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; McAuliffe, S.; Swain, C.A.; Ward, S.A.; Crosby, C.A.; Zheng, H.; Sherman, J.; Dong, Y.; Zhang, Y.; Sunder, N.; et al. Cerebrospinal fluid abeta to tau ratio and postoperative cognitive change. Ann. Surg. 2013, 258, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Stokholm, M.G.; Iranzo, A.; Ostergaard, K.; Serradell, M.; Otto, M.; Svendsen, K.B.; Garrido, A.; Vilas, D.; Borghammer, P.; Santamaria, J.; et al. Assessment of neuroinflammation in patients with idiopathic rapid-eye-movement sleep behaviour disorder: A case-control study. Lancet Neurol. 2017, 16, 789–796. [Google Scholar] [CrossRef]
- Mander, B.A.; Winer, J.R.; Walker, M.P. Sleep and Human Aging. Neuron 2017, 94, 19–36. [Google Scholar] [CrossRef]
- Petralia, R.S.; Mattson, M.P.; Yao, P.J. Communication breakdown: The impact of ageing on synapse structure. Ageing Res. Rev. 2014, 14, 31–42. [Google Scholar] [CrossRef]
- Converse, A.K.; Larsen, E.C.; Engle, J.W.; Barnhart, T.E.; Nickles, R.J.; Duncan, I.D. 11C-(R)-PK11195 PET imaging of microglial activation and response to minocycline in zymosan-treated rats. J. Nucl. Med. 2011, 52, 257–262. [Google Scholar] [CrossRef]
- Norden, D.M.; Godbout, J.P. Review: Microglia of the aged brain: Primed to be activated and resistant to regulation. Neuropathol. Appl. Neurobiol. 2013, 39, 19–34. [Google Scholar] [CrossRef]
- Lull, M.E.; Block, M.L. Microglial activation and chronic neurodegeneration. Neurotherapeutics 2010, 7, 354–365. [Google Scholar] [CrossRef]
- Tremblay, M.E.; Lowery, R.L.; Majewska, A.K. Microglial interactions with synapses are modulated by visual experience. PLoS Biol. 2010, 8, e1000527. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, M.E.; Miyanishi, K.; Takeda, H.; Islam, A.; Matsuoka, N.; Kubo, M.; Matsumoto, S.; Kunieda, T.; Nomoto, M.; Yano, H.; et al. Phagocytic elimination of synapses by microglia during sleep. Glia 2020, 68, 44–59. [Google Scholar] [CrossRef] [PubMed]
- Sominsky, L.; Dangel, T.; Malik, S.; De Luca, S.N.; Singewald, N.; Spencer, S.J. Microglial ablation in rats disrupts the circadian system. FASEB J. 2021, 35, e21195. [Google Scholar] [CrossRef]
- Zoller, T.; Attaai, A.; Potru, P.S.; Russ, T.; Spittau, B. Aged Mouse Cortical Microglia Display an Activation Profile Suggesting Immunotolerogenic Functions. Int. J. Mol. Sci. 2018, 19, 706. [Google Scholar] [CrossRef]
- Streit, W.J.; Sammons, N.W.; Kuhns, A.J.; Sparks, D.L. Dystrophic microglia in the aging human brain. Glia 2004, 45, 208–212. [Google Scholar] [CrossRef]
- Kettenmann, H.; Hanisch, U.K.; Noda, M.; Verkhratsky, A. Physiology of microglia. Physiol. Rev. 2011, 91, 461–553. [Google Scholar] [CrossRef]
- Nimmerjahn, A.; Kirchhoff, F.; Helmchen, F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 2005, 308, 1314–1318. [Google Scholar] [CrossRef]
- Wake, H.; Moorhouse, A.J.; Jinno, S.; Kohsaka, S.; Nabekura, J. Resting microglia directly monitor the functional state of synapses in vivo and determine the fate of ischemic terminals. J. Neurosci. 2009, 29, 3974–3980. [Google Scholar] [CrossRef]
- Li, Y.; Du, X.F.; Liu, C.S.; Wen, Z.L.; Du, J.L. Reciprocal regulation between resting microglial dynamics and neuronal activity in vivo. Dev. Cell 2012, 23, 1189–1202. [Google Scholar] [CrossRef]
- Dworak, M.; McCarley, R.W.; Kim, T.; Kalinchuk, A.V.; Basheer, R. Sleep and brain energy levels: ATP changes during sleep. J. Neurosci. 2010, 30, 9007–9016. [Google Scholar] [CrossRef]
- Haynes, S.E.; Hollopeter, G.; Yang, G.; Kurpius, D.; Dailey, M.E.; Gan, W.B.; Julius, D. The P2Y12 receptor regulates microglial activation by extracellular nucleotides. Nat. Neurosci. 2006, 9, 1512–1519. [Google Scholar] [CrossRef]
- Badimon, A.; Strasburger, H.J.; Ayata, P.; Chen, X.; Nair, A.; Ikegami, A.; Hwang, P.; Chan, A.T.; Graves, S.M.; Uweru, J.O.; et al. Negative feedback control of neuronal activity by microglia. Nature 2020, 586, 417–423. [Google Scholar] [CrossRef]
- Hayashi, Y.; Satoru, K.; Naoki, K.; Fumiko, T.; Ryo, O.; Zhou, W.; Shigehiro, O.; Hiroshi, N. Diurnal Spatial Rearrangement of Microglial Processes through the Rhythmic Expression of P2Y12 Receptors. J. Neurol. Disord. 2013, 1, 120. [Google Scholar] [CrossRef]
- Krueger, J.M.; Taishi, P.; De, A.; Davis, C.J.; Winters, B.D.; Clinton, J.; Szentirmai, E.; Zielinski, M.R. ATP and the purine type 2 X7 receptor affect sleep. J. Appl. Physiol. 2010, 109, 1318–1327. [Google Scholar] [CrossRef] [PubMed]
- Stowell, R.D.; Sipe, G.O.; Dawes, R.P.; Batchelor, H.N.; Lordy, K.A.; Whitelaw, B.S.; Stoessel, M.B.; Bidlack, J.M.; Brown, E.; Sur, M.; et al. Noradrenergic signaling in the wakeful state inhibits microglial surveillance and synaptic plasticity in the mouse visual cortex. Nat. Neurosci. 2019, 22, 1782–1792. [Google Scholar] [CrossRef] [PubMed]
- Wisor, J.P.; Clegern, W.C. Quantification of short-term slow wave sleep homeostasis and its disruption by minocycline in the laboratory mouse. Neurosci. Lett. 2011, 490, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, K.; Nakazawa, Y.; Kotorii, T. Effects of antibiotics, minocycline and ampicillin, on human sleep. Brain Res. 1983, 288, 253–259. [Google Scholar] [CrossRef]
- Hayashi, Y.; Koyanagi, S.; Kusunose, N.; Okada, R.; Wu, Z.; Tozaki-Saitoh, H.; Ukai, K.; Kohsaka, S.; Inoue, K.; Ohdo, S.; et al. The intrinsic microglial molecular clock controls synaptic strength via the circadian expression of cathepsin S. Sci. Rep. 2013, 3, 2744. [Google Scholar] [CrossRef] [PubMed]
- Griffin, P.; Sheehan, P.W.; Dimitry, J.M.; Guo, C.; Kanan, M.F.; Lee, J.; Zhang, J.; Musiek, E.S. REV-ERBalpha mediates complement expression and diurnal regulation of microglial synaptic phagocytosis. Elife 2020, 9, e58765. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.U.; Ying, Y.; Li, Y.; Eyo, U.B.; Chen, T.; Zheng, J.; Umpierre, A.D.; Zhu, J.; Bosco, D.B.; Dong, H.; et al. Neuronal network activity controls microglial process surveillance in awake mice via norepinephrine signaling. Nat. Neurosci. 2019, 22, 1771–1781. [Google Scholar] [CrossRef]
- Esser, S.K.; Hill, S.L.; Tononi, G. Sleep homeostasis and cortical synchronization: I. Modeling the effects of synaptic strength on sleep slow waves. Sleep 2007, 30, 1617–1630. [Google Scholar] [CrossRef]
- Tononi, G.; Cirelli, C. Sleep function and synaptic homeostasis. Sleep Med. Rev. 2006, 10, 49–62. [Google Scholar] [CrossRef]
- Diekelmann, S.; Born, J. The memory function of sleep. Nat. Rev. Neurosci. 2010, 11, 114–126. [Google Scholar] [CrossRef]
- Lee, E.; Chung, W.S. Glial Control of Synapse Number in Healthy and Diseased Brain. Front. Cell Neurosci. 2019, 13, 42. [Google Scholar] [CrossRef]
- Paolicelli, R.C.; Bolasco, G.; Pagani, F.; Maggi, L.; Scianni, M.; Panzanelli, P.; Giustetto, M.; Ferreira, T.A.; Guiducci, E.; Dumas, L.; et al. Synaptic pruning by microglia is necessary for normal brain development. Science 2011, 333, 1456–1458. [Google Scholar] [CrossRef] [PubMed]
- Curtis, A.M.; Bellet, M.M.; Sassone-Corsi, P.; O’Neill, L.A. Circadian clock proteins and immunity. Immunity 2014, 40, 178–186. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, J.E.; Blaikley, J.; Beesley, S.; Matthews, L.; Simpson, K.D.; Boyce, S.H.; Farrow, S.N.; Else, K.J.; Singh, D.; Ray, D.W.; et al. The nuclear receptor REV-ERBalpha mediates circadian regulation of innate immunity through selective regulation of inflammatory cytokines. Proc. Natl. Acad. Sci. USA 2012, 109, 582–587. [Google Scholar] [CrossRef] [PubMed]
- Keller, M.; Mazuch, J.; Abraham, U.; Eom, G.D.; Herzog, E.D.; Volk, H.D.; Kramer, A.; Maier, B. A circadian clock in macrophages controls inflammatory immune responses. Proc. Natl. Acad. Sci. USA 2009, 106, 21407–21412. [Google Scholar] [CrossRef] [PubMed]
- Fonken, L.K.; Frank, M.G.; Kitt, M.M.; Barrientos, R.M.; Watkins, L.R.; Maier, S.F. Microglia inflammatory responses are controlled by an intrinsic circadian clock. Brain Behav. Immun. 2015, 45, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Fonken, L.K.; Weber, M.D.; Daut, R.A.; Kitt, M.M.; Frank, M.G.; Watkins, L.R.; Maier, S.F. Stress-induced neuroinflammatory priming is time of day dependent. Psychoneuroendocrinology 2016, 66, 82–90. [Google Scholar] [CrossRef]
- Ishii, Y.; Yamaizumi, A.; Kawakami, A.; Islam, A.; Choudhury, M.E.; Takahashi, H.; Yano, H.; Tanaka, J. Anti-inflammatory effects of noradrenaline on LPS-treated microglial cells: Suppression of NFkappaB nuclear translocation and subsequent STAT1 phosphorylation. Neurochem. Int. 2015, 90, 56–66. [Google Scholar] [CrossRef]
- Fonken, L.K.; Kitt, M.M.; Gaudet, A.D.; Barrientos, R.M.; Watkins, L.R.; Maier, S.F. Diminished circadian rhythms in hippocampal microglia may contribute to age-related neuroinflammatory sensitization. Neurobiol. Aging 2016, 47, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.; Debono, M. Replication of cortisol circadian rhythm: New advances in hydrocortisone replacement therapy. Ther. Adv. Endocrinol. Metab. 2010, 1, 129–138. [Google Scholar] [CrossRef]
- Gong, S.; Miao, Y.L.; Jiao, G.Z.; Sun, M.J.; Li, H.; Lin, J.; Luo, M.J.; Tan, J.H. Dynamics and correlation of serum cortisol and corticosterone under different physiological or stressful conditions in mice. PLoS ONE 2015, 10, e0117503. [Google Scholar] [CrossRef] [PubMed]
- Bunger, M.K.; Wilsbacher, L.D.; Moran, S.M.; Clendenin, C.; Radcliffe, L.A.; Hogenesch, J.B.; Simon, M.C.; Takahashi, J.S.; Bradfield, C.A. Mop3 is an essential component of the master circadian pacemaker in mammals. Cell 2000, 103, 1009–1017. [Google Scholar] [CrossRef]
- Man, K.; Loudon, A.; Chawla, A. Immunity around the clock. Science 2016, 354, 999–1003. [Google Scholar] [CrossRef] [PubMed]
- Musiek, E.S.; Holtzman, D.M. Mechanisms linking circadian clocks, sleep, and neurodegeneration. Science 2016, 354, 1004–1008. [Google Scholar] [CrossRef]
- Scheiermann, C.; Kunisaki, Y.; Frenette, P.S. Circadian control of the immune system. Nat. Rev. Immunol. 2013, 13, 190–198. [Google Scholar] [CrossRef]
- Nakao, A. Temporal regulation of cytokines by the circadian clock. J. Immunol. Res. 2014, 2014, 614529. [Google Scholar] [CrossRef]
- Huang, R.C. The discoveries of molecular mechanisms for the circadian rhythm: The 2017 Nobel Prize in Physiology or Medicine. Biomed. J. 2018, 41, 5–8. [Google Scholar] [CrossRef]
- Labrecque, N.; Cermakian, N. Circadian Clocks in the Immune System. J. Biol. Rhythm. 2015, 30, 277–290. [Google Scholar] [CrossRef]
- Mohawk, J.A.; Green, C.B.; Takahashi, J.S. Central and peripheral circadian clocks in mammals. Annu. Rev. Neurosci. 2012, 35, 445–462. [Google Scholar] [CrossRef] [PubMed]
- Duez, H.; Staels, B. The nuclear receptors Rev-erbs and RORs integrate circadian rhythms and metabolism. Diabetes Vasc. Dis. Res. 2008, 5, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Silver, A.C.; Arjona, A.; Hughes, M.E.; Nitabach, M.N.; Fikrig, E. Circadian expression of clock genes in mouse macrophages, dendritic cells, and B cells. Brain Behav. Immun. 2012, 26, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Nakazato, R.; Takarada, T.; Yamamoto, T.; Hotta, S.; Hinoi, E.; Yoneda, Y. Selective upregulation of Per1 mRNA expression by ATP through activation of P2X7 purinergic receptors expressed in microglial cells. J. Pharmacol. Sci. 2011, 116, 350–361. [Google Scholar] [CrossRef]
- Nguyen, K.D.; Fentress, S.J.; Qiu, Y.; Yun, K.; Cox, J.S.; Chawla, A. Circadian gene Bmal1 regulates diurnal oscillations of Ly6C(hi) inflammatory monocytes. Science 2013, 341, 1483–1488. [Google Scholar] [CrossRef]
- Nakazato, R.; Hotta, S.; Yamada, D.; Kou, M.; Nakamura, S.; Takahata, Y.; Tei, H.; Numano, R.; Hida, A.; Shimba, S.; et al. The intrinsic microglial clock system regulates interleukin-6 expression. Glia 2017, 65, 198–208. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.L.; Wolff, S.E.C.; Korpel, N.; Milanova, I.; Sandu, C.; Rensen, P.C.N.; Kooijman, S.; Cassel, J.C.; Kalsbeek, A.; Boutillier, A.L.; et al. Deficiency of the Circadian Clock Gene Bmal1 Reduces Microglial Immunometabolism. Front. Immunol. 2020, 11, 586399. [Google Scholar] [CrossRef]
- Spengler, M.L.; Kuropatwinski, K.K.; Comas, M.; Gasparian, A.V.; Fedtsova, N.; Gleiberman, A.S.; Gitlin, I.I.; Artemicheva, N.M.; Deluca, K.A.; Gudkov, A.V.; et al. Core circadian protein CLOCK is a positive regulator of NF-kappaB-mediated transcription. Proc. Natl. Acad. Sci. USA 2012, 109, E2457–E2465. [Google Scholar] [CrossRef]
- Griffin, P.; Dimitry, J.M.; Sheehan, P.W.; Lananna, B.V.; Guo, C.; Robinette, M.L.; Hayes, M.E.; Cedeno, M.R.; Nadarajah, C.J.; Ezerskiy, L.A.; et al. Circadian clock protein Rev-erbalpha regulates neuroinflammation. Proc. Natl. Acad. Sci. USA 2019, 116, 5102–5107. [Google Scholar] [CrossRef]
- Lee, J.; Kim, D.E.; Griffin, P.; Sheehan, P.W.; Kim, D.H.; Musiek, E.S.; Yoon, S.Y. Inhibition of REV-ERBs stimulates microglial amyloid-beta clearance and reduces amyloid plaque deposition in the 5XFAD mouse model of Alzheimer’s disease. Aging Cell 2020, 19, e13078. [Google Scholar] [CrossRef]
- Wolff, S.E.C.; Wang, X.L.; Jiao, H.; Sun, J.; Kalsbeek, A.; Yi, C.X.; Gao, Y. The Effect of Rev-erbalpha Agonist SR9011 on the Immune Response and Cell Metabolism of Microglia. Front. Immunol. 2020, 11, 550145. [Google Scholar] [CrossRef]
- Eichenfield, D.Z.; Troutman, T.D.; Link, V.M.; Lam, M.T.; Cho, H.; Gosselin, D.; Spann, N.J.; Lesch, H.P.; Tao, J.; Muto, J.; et al. Tissue damage drives co-localization of NF-kappaB, Smad3, and Nrf2 to direct Rev-erb sensitive wound repair in mouse macrophages. Elife 2016, 5. [Google Scholar] [CrossRef]
- Li, S.C.; Lindenberger, U.; Sikstrom, S. Aging cognition: From neuromodulation to representation. Trends Cogn. Sci. 2001, 5, 479–486. [Google Scholar] [CrossRef]
- Mahncke, H.W.; Bronstone, A.; Merzenich, M.M. Brain plasticity and functional losses in the aged: Scientific bases for a novel intervention. Prog. Brain Res. 2006, 157, 81–109. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, M.E.; Zettel, M.L.; Ison, J.R.; Allen, P.D.; Majewska, A.K. Effects of aging and sensory loss on glial cells in mouse visual and auditory cortices. Glia 2012, 60, 541–558. [Google Scholar] [CrossRef]
- Choudhury, M.E.; Kigami, Y.; Tanaka, J. Dual Roles of Microglia in the Basal Ganglia in Parkinson’s Disease. Int. J. Mol. Sci. 2021, 22, 3907. [Google Scholar] [CrossRef]
- Damani, M.R.; Zhao, L.; Fontainhas, A.M.; Amaral, J.; Fariss, R.N.; Wong, W.T. Age-related alterations in the dynamic behavior of microglia. Aging Cell 2011, 10, 263–276. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.; Martins, S.; Ferreira, P.A.; Cardoso, A.M.S.; Guedes, J.R.; Peca, J.; Cardoso, A.L. The old guard: Age-related changes in microglia and their consequences. Mech. Ageing Dev. 2021, 197, 111512. [Google Scholar] [CrossRef]
- Brawek, B.; Skok, M.; Garaschuk, O. Changing Functional Signatures of Microglia along the Axis of Brain Aging. Int. J. Mol. Sci. 2021, 22, 1091. [Google Scholar] [CrossRef]
- Greenwood, E.K.; Brown, D.R. Senescent Microglia: The Key to the Ageing Brain? Int. J. Mol. Sci. 2021, 22, 4402. [Google Scholar] [CrossRef] [PubMed]
- Lupi, D.; Semo, M.; Foster, R.G. Impact of age and retinal degeneration on the light input to circadian brain structures. Neurobiol. Aging 2012, 33, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Farajnia, S.; Michel, S.; Deboer, T.; vanderLeest, H.T.; Houben, T.; Rohling, J.H.; Ramkisoensing, A.; Yasenkov, R.; Meijer, J.H. Evidence for neuronal desynchrony in the aged suprachiasmatic nucleus clock. J. Neurosci. 2012, 32, 5891–5899. [Google Scholar] [CrossRef] [PubMed]
- Kallo, I.; Kalamatianos, T.; Piggins, H.D.; Coen, C.W. Ageing and the diurnal expression of mRNAs for vasoactive intestinal peptide and for the VPAC2 and PAC1 receptors in the suprachiasmatic nucleus of male rats. J. Neuroendocrinol. 2004, 16, 758–766. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.J.; Nakamura, W.; Yamazaki, S.; Kudo, T.; Cutler, T.; Colwell, C.S.; Block, G.D. Age-related decline in circadian output. J. Neurosci. 2011, 31, 10201–10205. [Google Scholar] [CrossRef]
- Pazo, D.; Cardinali, D.P.; Cano, P.; Reyes Toso, C.A.; Esquifino, A.I. Age-related changes in 24-hour rhythms of norepinephrine content and serotonin turnover in rat pineal gland: Effect of melatonin treatment. Neurosignals 2002, 11, 81–87. [Google Scholar] [CrossRef]
- Heneka, M.T.; Nadrigny, F.; Regen, T.; Martinez-Hernandez, A.; Dumitrescu-Ozimek, L.; Terwel, D.; Jardanhazi-Kurutz, D.; Walter, J.; Kirchhoff, F.; Hanisch, U.K.; et al. Locus ceruleus controls Alzheimer’s disease pathology by modulating microglial functions through norepinephrine. Proc. Natl. Acad. Sci. USA 2010, 107, 6058–6063. [Google Scholar] [CrossRef]
- Tanaka, J.; Fujita, H.; Matsuda, S.; Toku, K.; Sakanaka, M.; Maeda, N. Glucocorticoid- and mineralocorticoid receptors in microglial cells: The two receptors mediate differential effects of corticosteroids. Glia 1997, 20, 23–37. [Google Scholar] [CrossRef]
- Aono, H.; Choudhury, M.E.; Higaki, H.; Miyanishi, K.; Kigami, Y.; Fujita, K.; Akiyama, J.I.; Takahashi, H.; Yano, H.; Kubo, M.; et al. Microglia may compensate for dopaminergic neuron loss in experimental Parkinsonism through selective elimination of glutamatergic synapses from the subthalamic nucleus. Glia 2017, 65, 1833–1847. [Google Scholar] [CrossRef] [PubMed]
- Cano, P.; Cardinali, D.P.; Spinedi, E.; Esquifino, A.I. Effect of aging on 24-hour pattern of stress hormones and leptin in rats. Life Sci. 2008, 83, 142–148. [Google Scholar] [CrossRef]
- Ohayon, M.M.; Carskadon, M.A.; Guilleminault, C.; Vitiello, M.V. Meta-analysis of quantitative sleep parameters from childhood to old age in healthy individuals: Developing normative sleep values across the human lifespan. Sleep 2004, 27, 1255–1273. [Google Scholar] [CrossRef] [PubMed]
- Van Cauter, E.; Leproult, R.; Plat, L. Age-related changes in slow wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA 2000, 284, 861–868. [Google Scholar] [CrossRef] [PubMed]
- Panagiotou, M.; Vyazovskiy, V.V.; Meijer, J.H.; Deboer, T. Differences in electroencephalographic non-rapid-eye movement sleep slow-wave characteristics between young and old mice. Sci. Rep. 2017, 7, 43656. [Google Scholar] [CrossRef] [PubMed]
- Hasan, S.; Dauvilliers, Y.; Mongrain, V.; Franken, P.; Tafti, M. Age-related changes in sleep in inbred mice are genotype dependent. Neurobiol. Aging 2012, 33, 195.e13–195.e26. [Google Scholar] [CrossRef] [PubMed]
- Banks, G.; Heise, I.; Starbuck, B.; Osborne, T.; Wisby, L.; Potter, P.; Jackson, I.J.; Foster, R.G.; Peirson, S.N.; Nolan, P.M. Genetic background influences age-related decline in visual and nonvisual retinal responses, circadian rhythms, and sleep. Neurobiol. Aging 2015, 36, 380–393. [Google Scholar] [CrossRef]
- Wimmer, M.E.; Rising, J.; Galante, R.J.; Wyner, A.; Pack, A.I.; Abel, T. Aging in mice reduces the ability to sustain sleep/wake states. PLoS ONE 2013, 8, e81880. [Google Scholar] [CrossRef]
- Welsh, D.K.; Richardson, G.S.; Dement, W.C. Effect of age on the circadian pattern of sleep and wakefulness in the mouse. J. Gerontol. 1986, 41, 579–586. [Google Scholar] [CrossRef]
- Soltani, S.; Chauvette, S.; Bukhtiyarova, O.; Lina, J.M.; Dube, J.; Seigneur, J.; Carrier, J.; Timofeev, I. Sleep-Wake Cycle in Young and Older Mice. Front. Syst. Neurosci. 2019, 13, 51. [Google Scholar] [CrossRef]
- Rosenberg, R.S.; Zepelin, H.; Rechtschaffen, A. Sleep in young and old rats. J. Gerontol. 1979, 34, 525–532. [Google Scholar] [CrossRef]
- Zepelin, H.; Whitehead, W.E.; Rechtschaffen, A. Aging and sleep in the albino rat. Behav. Biol. 1972, 7, 65–74. [Google Scholar] [CrossRef]
- Mendelson, W.B.; Bergmann, B.M. Age-related changes in sleep in the rat. Sleep 1999, 22, 145–150. [Google Scholar] [CrossRef]
- Kaneshwaran, K.; Olah, M.; Tasaki, S.; Yu, L.; Bradshaw, E.M.; Schneider, J.A.; Buchman, A.S.; Bennett, D.A.; De Jager, P.L.; Lim, A.S.P. Sleep fragmentation, microglial aging, and cognitive impairment in adults with and without Alzheimer’s dementia. Sci. Adv. 2019, 5, eaax7331. [Google Scholar] [CrossRef] [PubMed]
- Bredow, S.; Guha-Thakurta, N.; Taishi, P.; Obal, F., Jr.; Krueger, J.M. Diurnal variations of tumor necrosis factor alpha mRNA and alpha-tubulin mRNA in rat brain. Neuroimmunomodulation 1997, 4, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Cearley, C.; Churchill, L.; Krueger, J.M. Time of day differences in IL1beta and TNFalpha mRNA levels in specific regions of the rat brain. Neurosci. Lett. 2003, 352, 61–63. [Google Scholar] [CrossRef] [PubMed]
- Ingiosi, A.M.; Raymond, R.M., Jr.; Pavlova, M.N.; Opp, M.R. Selective contributions of neuronal and astroglial interleukin-1 receptor 1 to the regulation of sleep. Brain Behav. Immun. 2015, 48, 244–257. [Google Scholar] [CrossRef]
- Taishi, P.; Bredow, S.; Guha-Thakurta, N.; Obal, F., Jr.; Krueger, J.M. Diurnal variations of interleukin-1 beta mRNA and beta-actin mRNA in rat brain. J. Neuroimmunol. 1997, 75, 69–74. [Google Scholar] [CrossRef]
- Krueger, J.M.; Obal, F.J.; Fang, J.; Kubota, T.; Taishi, P. The role of cytokines in physiological sleep regulation. Ann. N. Y. Acad. Sci. 2001, 933, 211–221. [Google Scholar] [CrossRef]
- Taishi, P.; Chen, Z.; Obal, F., Jr.; Hansen, M.K.; Zhang, J.; Fang, J.; Krueger, J.M. Sleep-associated changes in interleukin-1beta mRNA in the brain. J. Interferon Cytokine Res. 1998, 18, 793–798. [Google Scholar] [CrossRef]
- Takahashi, S.; Fang, J.; Kapas, L.; Wang, Y.; Krueger, J.M. Inhibition of brain interleukin-1 attenuates sleep rebound after sleep deprivation in rabbits. Am. J. Physiol. 1997, 273, R677–R682. [Google Scholar] [CrossRef]
- Friedman, E.M.; Boinski, S.; Coe, C.L. Interleukin-1 induces sleep-like behavior and alters call structure in juvenile rhesus macaques. Am. J. Primatol. 1995, 35, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Krueger, J.M.; Walter, J.; Dinarello, C.A.; Wolff, S.M.; Chedid, L. Sleep-promoting effects of endogenous pyrogen (interleukin-1). Am. J. Physiol. 1984, 246, R994–R999. [Google Scholar] [CrossRef] [PubMed]
- Lancel, M.; Mathias, S.; Faulhaber, J.; Schiffelholz, T. Effect of interleukin-1 beta on EEG power density during sleep depends on circadian phase. Am. J. Physiol. 1996, 270, R830–R837. [Google Scholar] [CrossRef] [PubMed]
- Opp, M.R.; Krueger, J.M. Interleukin 1-receptor antagonist blocks interleukin 1-induced sleep and fever. Am. J. Physiol. 1991, 260, R453–R457. [Google Scholar] [CrossRef]
- Susic, V.; Totic, S. Effects of purified human interleukin-1 on sleep and febrile response of cats. Arch. Int. Physiol. Biochim. 1989, 97, 293–302. [Google Scholar] [CrossRef]
- Kapas, L.; Bohnet, S.G.; Traynor, T.R.; Majde, J.A.; Szentirmai, E.; Magrath, P.; Taishi, P.; Krueger, J.M. Spontaneous and influenza virus-induced sleep are altered in TNF-alpha double-receptor deficient mice. J. Appl. Physiol. 2008, 105, 1187–1198. [Google Scholar] [CrossRef]
- Davis, C.J.; Dunbrasky, D.; Oonk, M.; Taishi, P.; Opp, M.R.; Krueger, J.M. The neuron-specific interleukin-1 receptor accessory protein is required for homeostatic sleep and sleep responses to influenza viral challenge in mice. Brain Behav. Immun. 2015, 47, 35–43. [Google Scholar] [CrossRef]
- Kitchen, G.B.; Cunningham, P.S.; Poolman, T.M.; Iqbal, M.; Maidstone, R.; Baxter, M.; Bagnall, J.; Begley, N.; Saer, B.; Hussell, T.; et al. The clock gene Bmal1 inhibits macrophage motility, phagocytosis, and impairs defense against pneumonia. Proc. Natl. Acad. Sci. USA 2020, 117, 1543–1551. [Google Scholar] [CrossRef] [PubMed]
- Broman, J.E.; Lundh, L.G.; Hetta, J. Insufficient sleep in the general population. Neurophysiol. Clin. 1996, 26, 30–39. [Google Scholar] [CrossRef]
- Palma, J.A.; Urrestarazu, E.; Iriarte, J. Sleep loss as risk factor for neurologic disorders: A review. Sleep Med. 2013, 14, 229–236. [Google Scholar] [CrossRef]
- Abel, M.; Bauml, K.H. Sleep can reduce proactive interference. Memory 2014, 22, 332–339. [Google Scholar] [CrossRef]
- Gronli, J.; Soule, J.; Bramham, C.R. Sleep and protein synthesis-dependent synaptic plasticity: Impacts of sleep loss and stress. Front. Behav. Neurosci. 2013, 7, 224. [Google Scholar] [CrossRef]
- Krause, A.J.; Simon, E.B.; Mander, B.A.; Greer, S.M.; Saletin, J.M.; Goldstein-Piekarski, A.N.; Walker, M.P. The sleep-deprived human brain. Nat. Rev. Neurosci. 2017, 18, 404–418. [Google Scholar] [CrossRef]
- McCoy, J.G.; Strecker, R.E. The cognitive cost of sleep lost. Neurobiol. Learn. Mem. 2011, 96, 564–582. [Google Scholar] [CrossRef]
- Tononi, G.; Cirelli, C. Sleep and the price of plasticity: From synaptic and cellular homeostasis to memory consolidation and integration. Neuron 2014, 81, 12–34. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Grone, B.; Colas, D.; Appelbaum, L.; Mourrain, P. Synaptic plasticity in sleep: Learning, homeostasis and disease. Trends Neurosci. 2011, 34, 452–463. [Google Scholar] [CrossRef]
- Pace-Schott, E.F.; Spencer, R.M. Sleep-dependent memory consolidation in healthy aging and mild cognitive impairment. Curr. Top. Behav. Neurosci. 2015, 25, 307–330. [Google Scholar] [CrossRef] [PubMed]
- Barrientos, R.M.; Kitt, M.M.; Watkins, L.R.; Maier, S.F. Neuroinflammation in the normal aging hippocampus. Neuroscience 2015, 309, 84–99. [Google Scholar] [CrossRef]
- Feinberg, I. The ontogenesis of human sleep and the relationship of sleep variables to intellectual function in the aged. Compr. Psychiatry 1968, 9, 138–147. [Google Scholar] [CrossRef]
- Deurveilher, S.; Golovin, T.; Hall, S.; Semba, K. Microglia dynamics in sleep/wake states and in response to sleep loss. Neurochem. Int. 2021, 143, 104944. [Google Scholar] [CrossRef]
- Hsu, J.C.; Lee, Y.S.; Chang, C.N.; Chuang, H.L.; Ling, E.A.; Lan, C.T. Sleep deprivation inhibits expression of NADPH-d and NOS while activating microglia and astroglia in the rat hippocampus. Cells Tissues Organs 2003, 173, 242–254. [Google Scholar] [CrossRef]
- Kaur, H.; Bhoday, H.S. Changing Adolescent Sleep Patterns: Factors Affecting them and the Related Problems. J. Assoc. Physicians India 2017, 65, 73–77. [Google Scholar]
- Mishra, R.; Manchanda, S.; Gupta, M.; Kaur, T.; Saini, V.; Sharma, A.; Kaur, G. Tinospora cordifolia ameliorates anxiety-like behavior and improves cognitive functions in acute sleep deprived rats. Sci. Rep. 2016, 6, 25564. [Google Scholar] [CrossRef] [PubMed]
- Dominguini, D.; Steckert, A.V.; Michels, M.; Spies, M.B.; Ritter, C.; Barichello, T.; Thompson, J.; Dal-Pizzol, F. The effects of anaesthetics and sedatives on brain inflammation. Neurosci. Biobehav. Rev. 2021, 127, 504–513. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, U.; Crestani, F.; Benke, D.; Brunig, I.; Benson, J.A.; Fritschy, J.M.; Martin, J.R.; Bluethmann, H.; Mohler, H. Benzodiazepine actions mediated by specific gamma-aminobutyric acid(A) receptor subtypes. Nature 1999, 401, 796–800. [Google Scholar] [CrossRef]
- Veiga, S.; Carrero, P.; Pernia, O.; Azcoitia, I.; Garcia-Segura, L.M. Translocator protein 18 kDa is involved in the regulation of reactive gliosis. Glia 2007, 55, 1426–1436. [Google Scholar] [CrossRef] [PubMed]
- Rupprecht, R.; Papadopoulos, V.; Rammes, G.; Baghai, T.C.; Fan, J.; Akula, N.; Groyer, G.; Adams, D.; Schumacher, M. Translocator protein (18 kDa) (TSPO) as a therapeutic target for neurological and psychiatric disorders. Nat. Rev. Drug Discov. 2010, 9, 971–988. [Google Scholar] [CrossRef] [PubMed]
- Wilms, H.; Claasen, J.; Rohl, C.; Sievers, J.; Deuschl, G.; Lucius, R. Involvement of benzodiazepine receptors in neuroinflammatory and neurodegenerative diseases: Evidence from activated microglial cells in vitro. Neurobiol. Dis. 2003, 14, 417–424. [Google Scholar] [CrossRef]
- Bae, K.R.; Shim, H.J.; Balu, D.; Kim, S.R.; Yu, S.W. Translocator protein 18 kDa negatively regulates inflammation in microglia. J. Neuroimmune Pharmacol. 2014, 9, 424–437. [Google Scholar] [CrossRef]
- Jazvinscak Jembrek, M.; Radovanovic, V.; Vlainic, J.; Vukovic, L.; Hanzic, N. Neuroprotective effect of zolpidem against glutamate-induced toxicity is mediated via the PI3K/Akt pathway and inhibited by PK11195. Toxicology 2018, 406–407, 58–69. [Google Scholar] [CrossRef]
- Schroeck, J.L.; Ford, J.; Conway, E.L.; Kurtzhalts, K.E.; Gee, M.E.; Vollmer, K.A.; Mergenhagen, K.A. Review of Safety and Efficacy of Sleep Medicines in Older Adults. Clin. Ther. 2016, 38, 2340–2372. [Google Scholar] [CrossRef]
- Wang, J.; Jiang, C.; Zhang, K.; Lan, X.; Chen, X.; Zang, W.; Wang, Z.; Guan, F.; Zhu, C.; Yang, X.; et al. Melatonin receptor activation provides cerebral protection after traumatic brain injury by mitigating oxidative stress and inflammation via the Nrf2 signaling pathway. Free Radic. Biol. Med. 2019, 131, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Sugishita, H.; Kuwabara, Y.; Toku, K.; Doi, L.; Yang, L.; Mitoma, J.; Furuya, S.; Hirabayashi, Y.; Maeda, N.; Sakanaka, M.; et al. L-Serine regulates the activities of microglial cells that express very low level of 3-phosphoglycerate dehydrogenase, an enzyme for L-Serine biosynthesis. J. Neurosci. Res. 2001, 64, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Takahashi, S.; Shen, M.; Yamaguchi, K.; Satoh, M. Effects of L-serine ingestion on human sleep. Springerplus 2014, 3, 456. [Google Scholar] [CrossRef] [PubMed][Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choudhury, M.E.; Miyanishi, K.; Takeda, H.; Tanaka, J. Microglia and the Aging Brain: Are Geriatric Microglia Linked to Poor Sleep Quality? Int. J. Mol. Sci. 2021, 22, 7824. https://doi.org/10.3390/ijms22157824
Choudhury ME, Miyanishi K, Takeda H, Tanaka J. Microglia and the Aging Brain: Are Geriatric Microglia Linked to Poor Sleep Quality? International Journal of Molecular Sciences. 2021; 22(15):7824. https://doi.org/10.3390/ijms22157824
Chicago/Turabian StyleChoudhury, Mohammed E., Kazuya Miyanishi, Haruna Takeda, and Junya Tanaka. 2021. "Microglia and the Aging Brain: Are Geriatric Microglia Linked to Poor Sleep Quality?" International Journal of Molecular Sciences 22, no. 15: 7824. https://doi.org/10.3390/ijms22157824
APA StyleChoudhury, M. E., Miyanishi, K., Takeda, H., & Tanaka, J. (2021). Microglia and the Aging Brain: Are Geriatric Microglia Linked to Poor Sleep Quality? International Journal of Molecular Sciences, 22(15), 7824. https://doi.org/10.3390/ijms22157824





