Novel Insight into Neuroimmune Regulatory Mechanisms and Biomarkers Linking Major Depression and Vascular Diseases: The Dilemma Continues
Abstract
1. Introduction
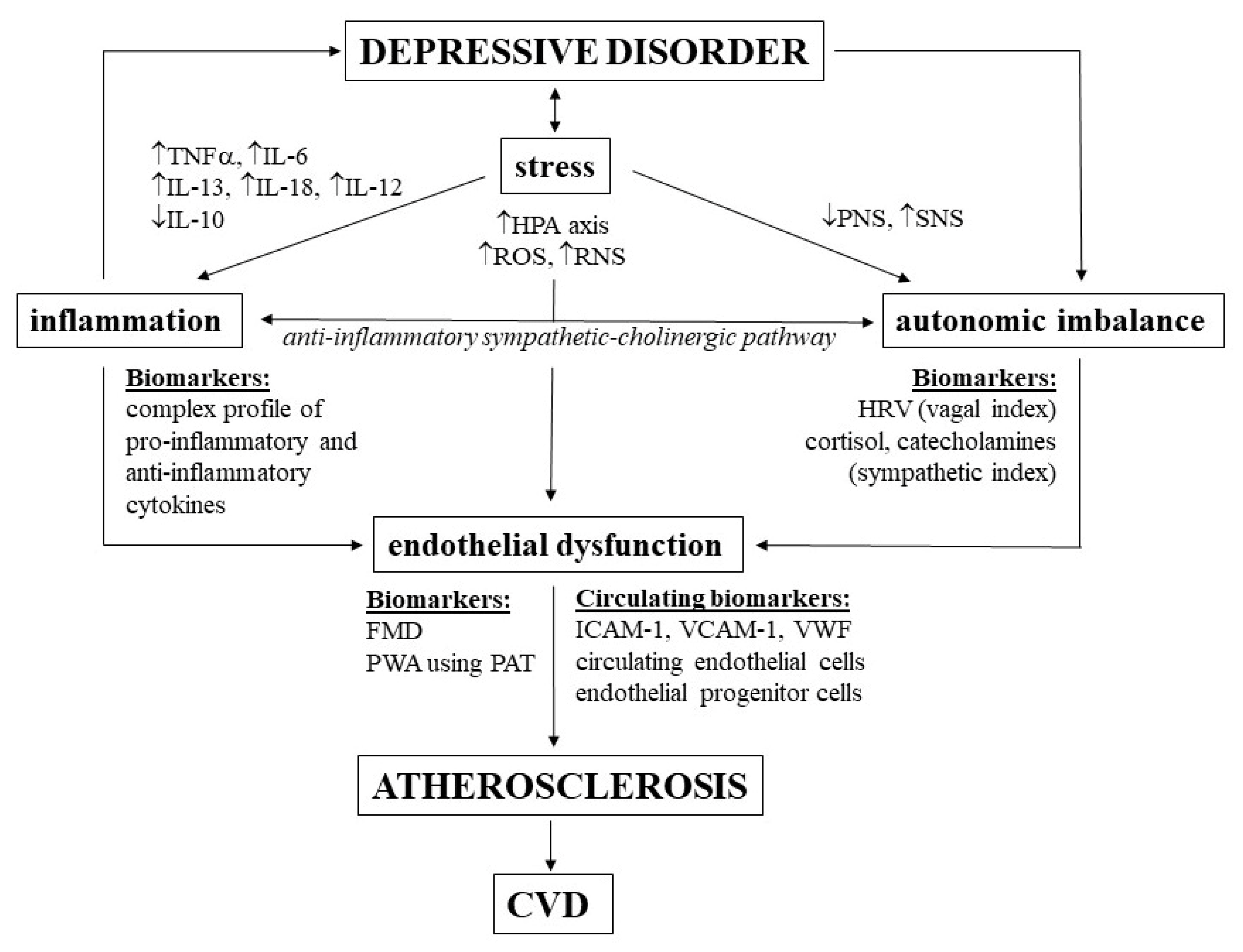
2. Role of Inflammation in Depressive Disorder
2.1. Cytokines in MDD Pathophysiology
2.1.1. Tryptophan/Kynurenine Pathway
2.1.2. Hypothalamus–Pituitary–Adrenal (HPA) Axis Pathway
3. Role of Endothelial Function in Depressive Disorder
3.1. Evaluation of Endothelial Function
3.2. Endothelial Dysfunction—Role in MDD-Linked Vascular Pathology
3.2.1. Endothelial Dysfunction and Oxidative Stress
3.2.2. Endothelial Dysfunction and Inflammation
3.2.3. Endothelial Dysfunction and Glucocorticoids
4. Role of Autonomic Nervous System in Depressive Disorder
5. Concluding Remarks and Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| MDD | Major depressive disorder |
| CVD | Cardiovascular disease |
| IL | Interleukin |
| TNF | Tumor necrosis factor |
| WHO | World Health Organization |
| IFN | Interferon |
| TGF | Transforming growth factor |
| CNS | Central nervous system |
| MCP | Monocyte chemoattractant protein |
| IDO | Indoleamine 2 |
| NMDA | N-methyl-D-aspartate |
| HPA | Hypothalamic-pituitary-adrenal |
| GRs | Glucocorticoid receptors |
| ROS | Reactive oxygen species |
| RNS | Reactive nitrogen species |
| NO | Nitric oxide |
| FMD | Flow-mediated dilation |
| PAT | Peripheral-artery tonometry |
| VWF | von Willebrand factor |
| ICAM-1 | Intercellular adhesion molecule-1 |
| VCAM-1 | Vascular cell adhesion molecule-1 |
| LDL | Low-density lipoprotein |
| HDL | High-density lipoprotein |
| cGMP | Cyclic guanosine monophosphate |
| NADPH | Reduced form of nicotinamide adenine dinucleotide phosphate |
| CRP | C-reactive protein |
| EPCs | Endothelial progenitor cells |
| VEGF | Vascular endothelial growth factor |
| ANS | Autonomic nervous system |
| SNS | Sympathetic nervous system |
| PNS | Parasympathetic nervous system |
| HRV | Heart rate variability |
References
- Depression. Available online: https://www.who.int/en/news-room/fact-sheets/detail/depression (accessed on 13 January 2020).
- Polanczyk, G.V.; Salum, G.A.; Sugaya, L.S.; Caye, A.; Rohde, L.A. Annual Research Review: A meta-analysis of the worldwide prevalence of mental disorders in children and adolescents. J. Child Psychol. Psychiatry 2015, 56, 345–365. [Google Scholar] [CrossRef] [PubMed]
- Halaris, A. Inflammation-associated co-morbidity between depression and cardiovascular disease. Curr. Top. Behav. Neurosci. 2017, 31, 45–70. [Google Scholar] [CrossRef] [PubMed]
- Tracey, K.J. The inflammatory reflex. Nature 2002, 420, 853–859. [Google Scholar] [CrossRef] [PubMed]
- Benarroch, E.E. Autonomic nervous system and neuroimmune interactions: New insights and clinical implications. Neurology 2019, 92, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Femenia, T.; Giménez-Cassina, A.; Codeluppi, S.; Fernández-Zafra, T.; Katsu-Jiménez, Y.; Terrando, N.; Eriksson, L.I.; Gómez-Galán, M. Disrupted neuroglial metabolic coupling after peripheral surgery. J. Neurosci. 2018, 38, 452–464. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Na, K.S.; Myint, A.M.; Leonard, B.E. The role of pro-inflammatory cytokines in neuroinflammation, neurogenesis and the neuroendocrine system in major depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 64, 277–284. [Google Scholar] [CrossRef]
- Himmerich, H.; Patsalos, O.; Lichtblau, N.; Ibrahim, M.A.; Dalton, B. Cytokine research in depression: Principles, challenges, and open questions. Front. Psychiatry 2019, 10, 1–16. [Google Scholar] [CrossRef]
- Jeon, S.W.; Kim, Y.K. Neuroinflammation and cytokine abnormality in major depression: Cause or consequence in that illness? World J. Psychiatry 2016, 6, 283–293. [Google Scholar] [CrossRef]
- Sochocka, M.; Diniz, B.S.; Leszek, J. Inflammatory response in the CNS: Friend or foe? Mol. Neurobiol. 2017, 54, 8071–8089. [Google Scholar] [CrossRef]
- Smith, R.S. Cytokines and Depression: How Your Immune System Causes Depression. Available online: http://www.cytokines-and-depression.com/ (accessed on 13 January 2020).
- Maes, M.; Yirmyia, R.; Noraberg, J.; Brene, S.; Hibbeln, J.; Perini, G.; Kubera, M.; Bob, P.; Lerer, B.; Maj, M. The inflammatory & neurodegenerative (I&ND) hypothesis of depression: Leads for future research and new drug developments in depression. Metab. Brain Dis. 2009, 24, 27–53. [Google Scholar] [CrossRef]
- Dowlati, Y.; Herrmann, N.; Swardfager, W.; Liu, H.; Sham, L.; Reim, E.K.; Lanctôt, K.L. A meta-analysis of cytokines in major depression. Biol. Psychiatry 2010, 67, 446–457. [Google Scholar] [CrossRef] [PubMed]
- Köhler, C.A.; Freitas, T.H.; Maes, M.; de Andrade, N.Q.; Liu, C.S.; Fernandes, B.S.; Stubbs, B.; Solmi, M.; Veronese, N.; Herrmann, N.; et al. Peripheral cytokine and chemokine alterations in depression: A meta-analysis of 82 studies. Acta Psychiatr. Scand. 2017, 135, 373–387. [Google Scholar] [CrossRef] [PubMed]
- D’Acunto, G.; Nageye, F.; Zhang, J.; Masi, G.; Cortese, S. Inflammatory cytokines in children and adolescents with depressive disorders: A systematic review and meta-analysis. J. Child Adolesc. Psychopharmacol. 2019, 29, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Myint, A.M.; Kim, Y.K. Cytokine-serotonin interaction through IDO: A neurodegeneration hypothesis of depression. Med. Hypotheses 2003, 61, 519–525. [Google Scholar] [CrossRef]
- Won, E.; Kim, Y.K. Stress, the autonomic nervous system, and the immune-kynurenine pathway in the etiology of depression. Curr. Neuropharmacol. 2016, 14, 665–673. [Google Scholar] [CrossRef]
- Baranyi, A.; Amouzadeh-Ghadikolai, O.; von Lewinski, D.; Breitenecker, R.J.; Rothenhäusler, H.B.; Robier, C.; Baranyi, M.; Theokas, S.; Meinitzer, A. Revisiting the tryptophan-serotonin deficiency and the inflammatory hypotheses of major depression in a biopsychosocial approach. PeerJ 2017, 5, 1–22. [Google Scholar] [CrossRef]
- Slavich, G.M.; Irwin, M.R. From stress to inflammation and major depressive disorder: A social signal transduction theory of depression. Psychol. Bull. 2014, 140, 774–815. [Google Scholar] [CrossRef]
- Johnson, J.D.; Barnard, D.F.; Kulp, A.C.; Mehta, D.M. Neuroendocrine regulation of brain cytokines after psychological stress. J. Endocr. Soc. 2019, 3, 1302–1320. [Google Scholar] [CrossRef]
- Raison, C.L.; Capuron, L.; Miller, A.H. Cytokines sing the blues: Inflammation and the pathogenesis of depression. Trends Immunol. 2006, 27, 24–31. [Google Scholar] [CrossRef]
- Shishkina, G.T.; Dygalo, N.N. The glucocorticoid hypothesis of depression: History and prospects. Russ. J. Genet. Appl. Res. 2017, 7, 128–133. [Google Scholar] [CrossRef]
- Moncada, S. The Vascular Endothelium. In Endothelium and Cardiovascular Diseases, 1st ed.; Luz, P.L., Libby, P., Chagas, A.C., Laurindo, F.R., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 5–10. [Google Scholar]
- Barthelmes, J.; Nägele, M.P.; Ludovici, V.; Ruschitzka, F.; Sudano, I.; Flammer, A.J. Endothelial dysfunction in cardiovascular disease and Flammer syndrome-similarities and differences. EPMA J. 2017, 8, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Goncharov, N.V.; Nadeev, A.D.; Jenkins, R.O.; Avdonin, P.V. Markers and biomarkers of endothelium: When something is rotten in the state. Oxid. Med. Cell. Longev. 2017, 2017, 1–27. [Google Scholar] [CrossRef]
- Flammer, A.J.; Anderson, T.; Celermajer, D.S.; Creager, M.A.; Deanfield, J.; Ganz, P.; Hamburg, N.M.; Lüscher, T.F.; Shechter, M.; Taddei, S.; et al. The assessment of endothelial function: From research into clinical practice. Circulation 2012, 126, 753–767. [Google Scholar] [CrossRef] [PubMed]
- Tonhajzerova, I.; Bona Olexova, L.; Jurko, A.; Spronck, B.; Jurko, T.; Sekaninova, N.; Visnovcova, Z.; Mestanikova, A.; Kudela, E.; Mestanik, M. Novel biomarkers of early atherosclerotic changes for personalised prevention of cardiovascular disease in cervical cancer and human papillomavirus infection. Int. J. Mol. Sci. 2019, 20, 720. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Li, C.; Shao, R.; Yu, H.; Zhang, Q. The role of biomarkers of endothelial activation in predicting morbidity and mortality in patients with severe sepsis and septic shock in intensive care: A prospective observational study. Thromb. Res. 2018, 171, 149–154. [Google Scholar] [CrossRef]
- Lopez-Vilchez, I.; Diaz-Ricart, M.; Navarro, V.; Torramade, S.; Zamorano-Leon, J.; Lopez-Farre, A.; Galan, A.M.; Gasto, C.; Escolar, G. Endothelial damage in major depression patients is modulated by SSRI treatment, as demonstrated by circulating biomarkers and an in vitro cell model. Transl. Psychiatry 2016, 6, 1–8. [Google Scholar] [CrossRef]
- Regueiro, A.; Cuadrado-Godia, E.; Bueno-Betí, C.; Diaz-Ricart, M.; Oliveras, A.; Novella, S.; Gené, G.G.; Jung, C.; Subirana, I.; Ortiz-Pérez, J.T.; et al. Mobilization of endothelial progenitor cells in acute cardiovascular events in the PROCELL study: Time-course after acute myocardial infarction and stroke. J. Mol. Cell. Cardiol. 2015, 80, 146–155. [Google Scholar] [CrossRef]
- Schmitz, S.; Abosi, O.; Persons, J.; Sinkey, C.; Fiedorowicz, J. Impact of mood on endothelial function and arterial stiffness in bipolar disorder. Hear. Mind 2018, 2, 78–84. [Google Scholar] [CrossRef]
- Shi, H.; Feng, G.; Wang, Z.; Zhou, C.; Zhong, G.; Hu, Y.; Wang, G. Relationships between depressive symptoms and endothelial function among outpatients of a general hospital in China. Med. Sci. Monit. 2015, 21, 1812–1819. [Google Scholar] [CrossRef][Green Version]
- van Dooren, F.E.; Verhey, F.R.; Pouwer, F.; Schalkwijk, C.G.; Sep, S.J.; Stehouwer, C.D.; Henry, R.M.; Dagnelie, P.C.; Schaper, N.C.; van der Kallen, C.J.; et al. Association of Type D personality with increased vulnerability to depression: Is there a role for inflammation or endothelial dysfunction?—The Maastricht Study. J. Affect. Disord. 2016, 189, 118–125. [Google Scholar] [CrossRef]
- Marchio, P.; Guerra-Ojeda, S.; Vila, J.M.; Aldasoro, M.; Victor, V.M.; Mauricio, M.D. Targeting early atherosclerosis: A focus on oxidative stress and inflammation. Oxid. Med. Cell. Longev. 2019, 2019, 1–32. [Google Scholar] [CrossRef] [PubMed]
- Pothineni, N.V.; Subramany, S.; Kuriakose, K.; Shirazi, L.F.; Romeo, F.; Shah, P.K.; Mehta, J.L. Infections, atherosclerosis, and coronary heart disease. Eur. Heart J. 2017, 38, 3195–3201. [Google Scholar] [CrossRef] [PubMed]
- Sessa, R.; Di Pietro, M.; Filardo, S.; Turriziani, O. Infectious burden and atherosclerosis: A clinical issue. World J. Clin. Cases 2014, 2, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Gimbrone, M.A.; García-Cardeña, G. Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circ. Res. 2016, 118, 620–636. [Google Scholar] [CrossRef] [PubMed]
- Ormonde do Carmo, M.B.; Mendes-Ribeiro, A.C.; Matsuura, C.; Pinto, V.L.; Mury, W.V.; Pinto, N.O.; Moss, M.B.; Ferraz, M.R.; Brunini, T.M. Major depression induces oxidative stress and platelet hyperaggregability. J. Psychiatr. Res. 2015, 61, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, B.I.; Carnethon, M.R.; Matthews, K.A.; McIntyre, R.S.; Miller, G.E.; Raghuveer, G.; Stoney, C.M.; Wasiak, H.; McCrindle, B.W. Major depressive disorder and bipolar disorder predispose youth to accelerated atherosclerosis and early cardiovascular disease: A scientific statement from the American Heart Association. Circulation 2015, 132, 965–986. [Google Scholar] [CrossRef]
- Förstermann, U.; Xia, N.; Li, H. Roles of vascular oxidative stress and nitric oxide in the pathogenesis of atherosclerosis. Circ. Res. 2017, 120, 713–735. [Google Scholar] [CrossRef]
- Brooks, S.; Brnayan, K.W.; DeVallance, E.; Skinner, R.; Lemaster, K.; Sheets, J.W.; Pitzer, C.R.; Asano, S.; Bryner, R.W.; Olfert, I.M.; et al. Psychological stress-induced cerebrovascular dysfunction: The role of metabolic syndrome and exercise. Exp. Physiol. 2018, 103, 761–776. [Google Scholar] [CrossRef]
- Greaney, J.L.; Saunders, E.F.; Santhanam, L.; Alexander, L.M. Oxidative stress contributes to microvascular endothelial dysfunction in men and women with major depressive disorder. Circ. Res. 2019, 124, 564–574. [Google Scholar] [CrossRef]
- Palta, P.; Samuel, L.J.; Miller, E.R.; Szanton, S.L. Depression and oxidative stress: Results from a meta-analysis of observational studies. Psychosom. Med. 2014, 76, 12–19. [Google Scholar] [CrossRef]
- Black, C.N.; Bot, M.; Scheffer, P.G.; Cuijpers, P.; Penninx, B.W. Is depression associated with increased oxidative stress? A systematic review and meta-analysis. Psychoneuroendocrinology 2015, 51, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Shihata, W.A.; Michell, D.L.; Andrews, K.L.; Chin-Dusting, J.P. Caveolae: A role in endothelial inflammation and mechanotransduction? Front. Physiol. 2016, 7, 628. [Google Scholar] [CrossRef] [PubMed]
- Danesh, J.; Collins, R.; Peto, R. Chronic infections and coronary heart disease: Is there a link? Lancet 1997, 350, 430–436. [Google Scholar] [CrossRef]
- Kop, W.J.; Gottdiener, J.S. The role of immune system parameters in the relationship between depression and coronary artery disease. Psychosom. Med. 2005, 67, 37–41. [Google Scholar] [CrossRef]
- Tarantino, G.; Costantini, S.; Finelli, C.; Capone, F.; Guerriero, E.; La Sala, N.; Gioia, S.; Castello, G. Carotid intima-media thickness is predicted by combined eotaxin levels and severity of hepatic steatosis at ultrasonography in obese patients with nonalcoholic fatty liver disease. PLoS ONE 2014, 9, 1–8. [Google Scholar] [CrossRef]
- Burford, N.G.; Webster, N.A.; Cruz-Topete, D. Hypothalamic-pituitary-adrenal axis modulation of glucocorticoids in the cardiovascular system. Int. J. Mol. Sci. 2017, 18, 2150. [Google Scholar] [CrossRef]
- Goodwin, J.E.; Feng, Y.; Velazquez, H.; Sessa, W.C. Endothelial glucocorticoid receptor is required for protection against sepsis. Proc. Natl. Acad. Sci. USA 2013, 110, 306–311. [Google Scholar] [CrossRef]
- Goodwin, J.E.; Zhang, X.; Rotllan, N.; Feng, Y.; Zhou, H.; Fernández-Hernando, C.; Yu, J.; Sessa, W.C. Endothelial glucocorticoid receptor suppresses atherogenesis--brief report. Arterioscler. Thromb. Vasc. Biol. 2015, 35, 779–782. [Google Scholar] [CrossRef]
- Luo, M.J.; Thieringer, R.; Springer, M.S.; Wright, S.D.; Hermanowski-Vosatka, A.; Plump, A.; Balkovec, J.M.; Cheng, K.; Ding, G.J.; Kawka, D.W.; et al. 11β-HSD1 inhibition reduces atherosclerosis in mice by altering proinflammatory gene expression in the vasculature. Physiol. Genomics 2013, 45, 47–57. [Google Scholar] [CrossRef]
- Kipari, T.; Hadoke, P.W.; Iqbal, J.; Man, T.Y.; Miller, E.; Coutinho, A.E.; Zhang, Z.; Sullivan, K.M.; Mitic, T.; Livingstone, D.E.; et al. 11β-hydroxysteroid dehydrogenase type 1 deficiency in bone marrow-derived cells reduces atherosclerosis. FASEB J. 2013, 27, 1519–1531. [Google Scholar] [CrossRef]
- Deuchar, G.A.; McLean, D.; Hadoke, P.; Brownstein, D.G.; Webb, D.J.; Mullins, J.J.; Chapman, K.; Seckl, J.R.; Kotelevtsev, Y.V. 11β-hydroxysteroid dehydrogenase type 2 deficiency accelerates atherogenesis and causes proinflammatory changes in the endothelium in apoe-/- mice. Endocrinology 2011, 152, 236–246. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, F.M.; Lichtblau, N.; Minkwitz, J.; Chittka, T.; Thormann, J.; Kirkby, K.C.; Sander, C.; Mergl, R.; Faßhauer, M.; Stumvoll, M.; et al. Cytokine levels in depressed and non-depressed subjects, and masking effects of obesity. J. Psychiatr. Res. 2014, 55, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Haapakoski, R.; Mathieu, J.; Ebmeier, K.P.; Alenius, H.; Kivimäki, M. Cumulative meta-analysis of interleukins 6 and 1β, tumour necrosis factor α and C-reactive protein in patients with major depressive disorder. Brain. Behav. Immun. 2015, 49, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Al-Hakeim, H.K.; Al-Rammahi, D.A.; Al-Dujaili, A.H. IL-6, IL-18, sIL-2R, and TNFα proinflammatory markers in depression and schizophrenia patients who are free of overt inflammation. J. Affect. Disord. 2015, 182, 106–114. [Google Scholar] [CrossRef]
- Muthuramalingam, A.; Menon, V.; Rajkumar, R.P.; Negi, V. Is depression an inflammatory disease? Findings from a cross-sectional study at a tertiary care center. Indian J. Psychol. Med. 2016, 38, 114–119. [Google Scholar] [CrossRef]
- Goldsmith, D.R.; Rapaport, M.H.; Miller, B.J. A meta-analysis of blood cytokine network alterations in psychiatric patients: Comparisons between schizophrenia, bipolar disorder and depression. Mol. Psychiatry 2016, 21, 1696–1709. [Google Scholar] [CrossRef]
- Zou, W.; Feng, R.; Yang, Y. Changes in the serum levels of inflammatory cytokines in antidepressant drug-naïve patients with major depression. PLoS ONE 2018, 13, 1–10. [Google Scholar] [CrossRef]
- Ng, A.; Tam, W.W.; Zhang, M.W.; Ho, C.S.; Husain, S.F.; McIntyre, R.S.; Ho, R.C. IL-1β, IL-6, TNF- α and CRP in elderly patients with depression or Alzheimer’s disease: Systematic review and meta-analysis. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef]
- Gariup, M.; Gonzalez, A.; Lázaro, L.; Torres, F.; Serra-Pagès, C.; Morer, A. IL-8 and the innate immunity as biomarkers in acute child and adolescent psychopathology. Psychoneuroendocrinology 2015, 62, 233–242. [Google Scholar] [CrossRef]
- Pallavi, P.; Sagar, R.; Mehta, M.; Sharma, S.; Subramanium, A.; Shamshi, F.; Sengupta, U.; Pandey, R.M.; Mukhopadhyay, A.K. Serum cytokines and anxiety in adolescent depression patients: Gender effect. Psychiatry Res. 2015, 229, 374–380. [Google Scholar] [CrossRef]
- Miklowitz, D.J.; Portnoff, L.C.; Armstrong, C.C.; Keenan-Miller, D.; Breen, E.C.; Muscatell, K.A.; Eisenberger, N.I.; Irwin, M.R. Inflammatory cytokines and nuclear factor-kappa B activation in adolescents with bipolar and major depressive disorders. Psychiatry Res. 2016, 241, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Sánchez, G.; Becerril-Villanueva, E.; Arreola, R.; Martínez-Levy, G.; Hernández-Gutiérrez, M.E.; Velasco-Velásquez, M.A.; Alvarez-Herrera, S.; Cruz-Fuentes, C.; Palacios, L.; De La Peña, F.; et al. Inflammatory profiles in depressed adolescents treated with fluoxetine: An 8-week follow-up open study. Mediators Inflamm. 2018, 2018, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Van Agtmaal, M.J.; Houben, A.J.; Pouwer, F.; Stehouwer, C.D.; Schram, M.T. Association of microvascular dysfunction with late-life depression: A systematic review and meta-analysis. JAMA Psychiatry 2017, 74, 729–739. [Google Scholar] [CrossRef] [PubMed]
- Blum, A.; Pastuck, N.; Zaroura, I.; Rotem, J.; Kamal, F. Impaired ability to grow colonies of endothelial stem cells could be the mechanism explaining the high cardiovascular morbidity and mortality of patients with depression. QJM 2017, 110, 501–506. [Google Scholar] [CrossRef]
- Baghai, T.C.; Varallo-Bedarida, G.; Born, C.; Häfner, S.; Schüle, C.; Eser, D.; Zill, P.; Manook, A.; Weigl, J.; Jooyandeh, S.; et al. Classical risk factors and inflammatory biomarkers: One of the missing biological links between cardiovascular disease and major depressive disorder. Int. J. Mol. Sci. 2018, 19, 1740. [Google Scholar] [CrossRef]
- Saleptsis, V.; Papaliaga, M.; Spanos, K.; Kouvelos, G.; Labropoulos, N.; Halaris, A.; Giannoukas, A. Carotid intima-media thickness and circulation markers of inflammation in patients with depression. J. Diagnostic Med. Sonogr. 2019, 36, 111–119. [Google Scholar] [CrossRef]
- Shi, S.; Liang, J.; Liu, T.; Yuan, X.; Ruan, B.; Sun, L.; Tang, Y.; Yang, B.; Hu, D.; Huang, C. Depression increases sympathetic activity and exacerbates myocardial remodeling after myocardial infarction: Evidence from an animal experiment. PLoS ONE 2014, 9, 1–12. [Google Scholar] [CrossRef]
- Ngampramuan, S.; Tungtong, P.; Mukda, S.; Jariyavilas, A.; Sakulisariyaporn, C. Evaluation of autonomic nervous system, saliva cortisol levels, and cognitive function in major depressive disorder patients. Depress. Res. Treat. 2018, 2018, 1–8. [Google Scholar] [CrossRef]
- Mestanikova, A.; Mestanik, M.; Ondrejka, I.; Hrtanek, I.; Cesnekova, D.; Jurko, A.; Visnovcova, Z.; Sekaninova, N.; Tonhajzerova, I. Complex cardiac vagal regulation to mental and physiological stress in adolescent major depression. J. Affect. Disord. 2019, 249, 234–241. [Google Scholar] [CrossRef]
- Koch, C.; Wilhelm, M.; Salzmann, S.; Rief, W.; Euteneuer, F. A meta-analysis of heart rate variability in major depression. Psychol. Med. 2019, 49, 1948–1957. [Google Scholar] [CrossRef]
- An, H.; Han, J.W.; Jeong, H.G.; Kim, T.H.; Lee, J.J.; Lee, S.B.; Park, J.H.; Kim, K.W. Parasympathetic predominance is a risk factor for future depression: A prospective cohort study. J. Affect. Disord. 2020, 260, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Shahane, A.D.; LeRoy, A.S.; Denny, B.T.; Fagundes, C.P. Connecting cognition, cardiology, and chromosomes: Cognitive reappraisal impacts the relationship between heart rate variability and telomere length in CD8+CD28– cells. Psychoneuroendocrinology 2020, 112, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Bratton, B.O.; Martelli, D.; McKinley, M.J.; Trevaks, D.; Anderson, C.R.; McAllen, R.M. Neural regulation of inflammation: No neural connection from the vagus to splenic sympathetic neurons. Exp. Physiol. 2012, 97, 1180–1185. [Google Scholar] [CrossRef] [PubMed]
- Martelli, D.; McKinley, M.J.; McAllen, R.M. The cholinergic anti-inflammatory pathway: A critical review. Auton. Neurosci. Basic Clin. 2014, 182, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.P.; Koenig, J.; Carnevali, L.; Sgoifo, A.; Jarczok, M.N.; Sternberg, E.M.; Thayer, J.F. Heart rate variability and inflammation: A meta-analysis of human studies. Brain. Behav. Immun. 2019, 80, 219–226. [Google Scholar] [CrossRef]
- Kadoya, M.; Koyama, H. Sleep, autonomic nervous function and atherosclerosis. Int. J. Mol. Sci. 2019, 20, 794. [Google Scholar] [CrossRef]
- Pizzi, C.; Manzoli, L.; Mancini, S.; Bedetti, G.; Fontana, F.; Costa, G.M. Autonomic nervous system, inflammation and preclinical carotid atherosclerosis in depressed subjects with coronary risk factors. Atherosclerosis 2010, 212, 292–298. [Google Scholar] [CrossRef]
- Ulleryd, M.A.; Prahl, U.; Börsbo, J.; Schmidt, C.; Nilsson, S.; Bergström, G.; Johansson, M.E. The association between autonomic dysfunction, inflammation and atherosclerosis in men under investigation for carotid plaques. PLoS ONE 2017, 12, e0174974. [Google Scholar] [CrossRef]
- Pagano, G.; Talamanca, A.A.; Castello, G.; Cordero, M.D.; d’Ischia, M.; Gadaleta, M.N.; Pallardó, F.V.; Petrović, S.; Tiano, L.; Zatterale, A. Oxidative stress and mitochondrial dysfunction across broad-ranging pathologies: Toward mitochondria-targeted clinical strategies. Oxid. Med. Cell. Longev. 2014, 2014, 1–27. [Google Scholar] [CrossRef]
- Caruso, G.; Fresta, C.G.; Grasso, M.; Santangelo, R.; Lazzarino, G.; Lunte, S.M.; Caraci, F. Inflammation as the common biological link between depression and cardiovascular diseases: Can carnosine exert a protective role? Curr. Med. Chem. 2019, 26, 1. [Google Scholar] [CrossRef]
- Köhler-Forsberg, O.; N. Lydholm, C.; Hjorthøj, C.; Nordentoft, M.; Mors, O.; Benros, M.E. Efficacy of anti-inflammatory treatment on major depressive disorder or depressive symptoms: Meta-analysis of clinical trials. Acta Psychiatr. Scand. 2019, 139, 404–419. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.J.; Wei, Y.B.; Strawbridge, R.; Bao, Y.; Chang, S.; Shi, L.; Que, J.; Gadad, B.S.; Trivedi, M.H.; Kelsoe, J.R.; et al. Peripheral cytokine levels and response to antidepressant treatment in depression: A systematic review and meta-analysis. Mol. Psychiatry 2020, 25, 339–350. [Google Scholar] [CrossRef] [PubMed]
| Recent Studies | Measured Markers | Main Findings |
|---|---|---|
| Inflammation-Activity Biomarkers | ||
| Schmidt et al., 2014 [55] | IL-2, IL-4, IL-5, IL-10, IL-12, IL-13, TNF-γ, TNF-α | Higher levels of IL-2, IL-5, IL-12, IL-13, INF-γ, and TNF-α were found in depressive patients compared with those in non-depressive subjects. |
| Haapakoski et al., 2015 [56] | IL-6, IL-1β, TNF-α | Meta-analysis confirmed higher serum IL-6 levels in depressive patients compared with those in non-depressive controls. |
| Al-Hakeim et al., 2015 [57] | IL-6, IL-18, TNF-α | Serum levels of IL-6, IL-18, and TNFα were significantly increased in depressive patients compared with those of the control group. |
| Muthuramalingam et al., 2016 [58] | IL-6, TNF-α, TGF-β | Depressive patients demonstrated significantly raised baseline levels of TNF-α and IL-6, but no difference in levels of TGF-β compared with healthy controls. |
| Goldsmith et al., 2016 [59] | IL-1β, IL-2, IL-4, IL-6, IL-8, IL-10, IL-12, TNF-α, INF-γ | IL-6, IL-10, IL-12, and TNF-α levels were significantly increased, and levels of IFN-γ and IL-4 were significantly decreased without significant differences in IL-1β and IL-2 levels in patients with MDD compared with controls. |
| Zou et al., 2018 [60] | IL-1β, IL-6, IL-8, IL-10, TNF-α, TGF-β1 | Increased levels of IL-1β, IL-10, and TNF-α, and decreased IL-8 levels, were found in MDD patients compared with healthy controls. |
| Ng et al., 2018 [61] | IL-1β, IL-6, TNF- α | Meta-analysis found elevated peripheral levels of IL-1β and IL-6 without TNF-α in depressive patients compared with controls. |
| Gariup et al., 2015 [62] | IL-1β, IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IFN-γ, and TNF-a | Depressive patients (aged 8–17 years) had significantly higher levels of IL-6, IL-8, and IL-1β compared with controls. |
| Pallavi et al., 2015 [63] | IL-1β, IL-2, IL-6, IL-10, IL-17, TNF-α, IFN-γ, and TGF-β | Male MDD adolescents had significantly higher levels of IL-2 compared with controls; female MDD adolescents had significantly elevated serum IL-2 and IL-6 compared with their healthy female counterparts. |
| Miklowitz et al., 2016 [64] | TNF-α, IL-1β, IL-6, IL-8, and IL-10 | No significant differences in measured cytokines between MDD and controls. |
| Perez-Sanchez et al., 2018 [65] | IL-2, IFN-γ, IL-1β, TNF-α, IL-6, IL-12, IL-4, IL-5, IL-13, and IL-10 | Adolescents with MDD at baseline showed significant increases in all mentioned cytokines, except for IL-10, compared with healthy subjects. |
| Endothelial-Function Biomarkers | ||
| Van Agtmaal et al., 2017 [66] | VCAM, ICAM, E-selectin, VWF | Meta-analysis revealed association between increased levels of all measured peripheral markers and depression. |
| Blum et al., 2017 [67] | VCAM-1, VEGF, EPCs | MDD patients had high levels of VCAM-1 and VEGF; significant inhibition of EPCs colonies. |
| Baghai et al., 2018 [68] | ICAM-1, P-selectin, E-Selectin | ICAM-1 was significantly elevated in MDD group compared with healthy controls. |
| Saleptsis et al., 2019 [69] | P-selectin | Depressive patients had higher levels of P-selectin compared with individuals free of depression. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tonhajzerova, I.; Sekaninova, N.; Bona Olexova, L.; Visnovcova, Z. Novel Insight into Neuroimmune Regulatory Mechanisms and Biomarkers Linking Major Depression and Vascular Diseases: The Dilemma Continues. Int. J. Mol. Sci. 2020, 21, 2317. https://doi.org/10.3390/ijms21072317
Tonhajzerova I, Sekaninova N, Bona Olexova L, Visnovcova Z. Novel Insight into Neuroimmune Regulatory Mechanisms and Biomarkers Linking Major Depression and Vascular Diseases: The Dilemma Continues. International Journal of Molecular Sciences. 2020; 21(7):2317. https://doi.org/10.3390/ijms21072317
Chicago/Turabian StyleTonhajzerova, Ingrid, Nikola Sekaninova, Lucia Bona Olexova, and Zuzana Visnovcova. 2020. "Novel Insight into Neuroimmune Regulatory Mechanisms and Biomarkers Linking Major Depression and Vascular Diseases: The Dilemma Continues" International Journal of Molecular Sciences 21, no. 7: 2317. https://doi.org/10.3390/ijms21072317
APA StyleTonhajzerova, I., Sekaninova, N., Bona Olexova, L., & Visnovcova, Z. (2020). Novel Insight into Neuroimmune Regulatory Mechanisms and Biomarkers Linking Major Depression and Vascular Diseases: The Dilemma Continues. International Journal of Molecular Sciences, 21(7), 2317. https://doi.org/10.3390/ijms21072317





