Marine Omega-3 (N-3) Fatty Acids for Cardiovascular Health: An Update for 2020
Abstract
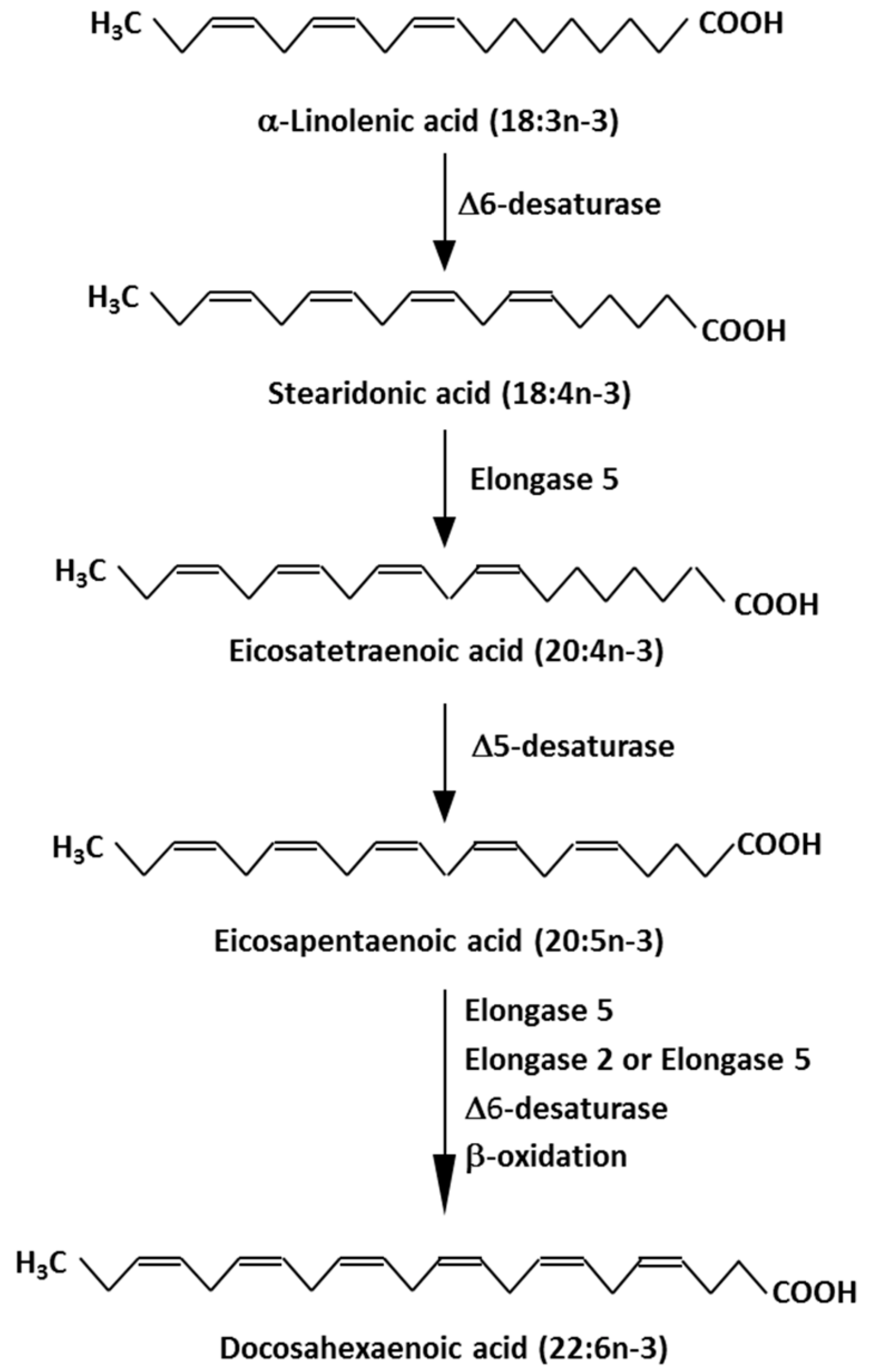
1. Marine Omega-3 Fatty Acids: Sources and Intakes
2. Strong Evidence for a Protective Effect of EPA and DHA Towards Cardiovascular Disease Emerges from Ecological, Case Control and Cohort Studies
3. Mechanisms by which EPA and DHA Reduce the Risk of Cardiovascular Disease
4. RCTs of Primary Prevention of Cardiovascular Disease with Marine n-3 Fatty Acids
5. RCTs of Secondary Prevention of Cardiovascular Disease with Marine n-3 Fatty Acids
5.1. Secondary Prevention Trials and Meta-Analyses Published Prior to 2010
5.2. RCTs with Marine n-3 Fatty Acids in High-Risk Patients Published in the Period 2010–2013
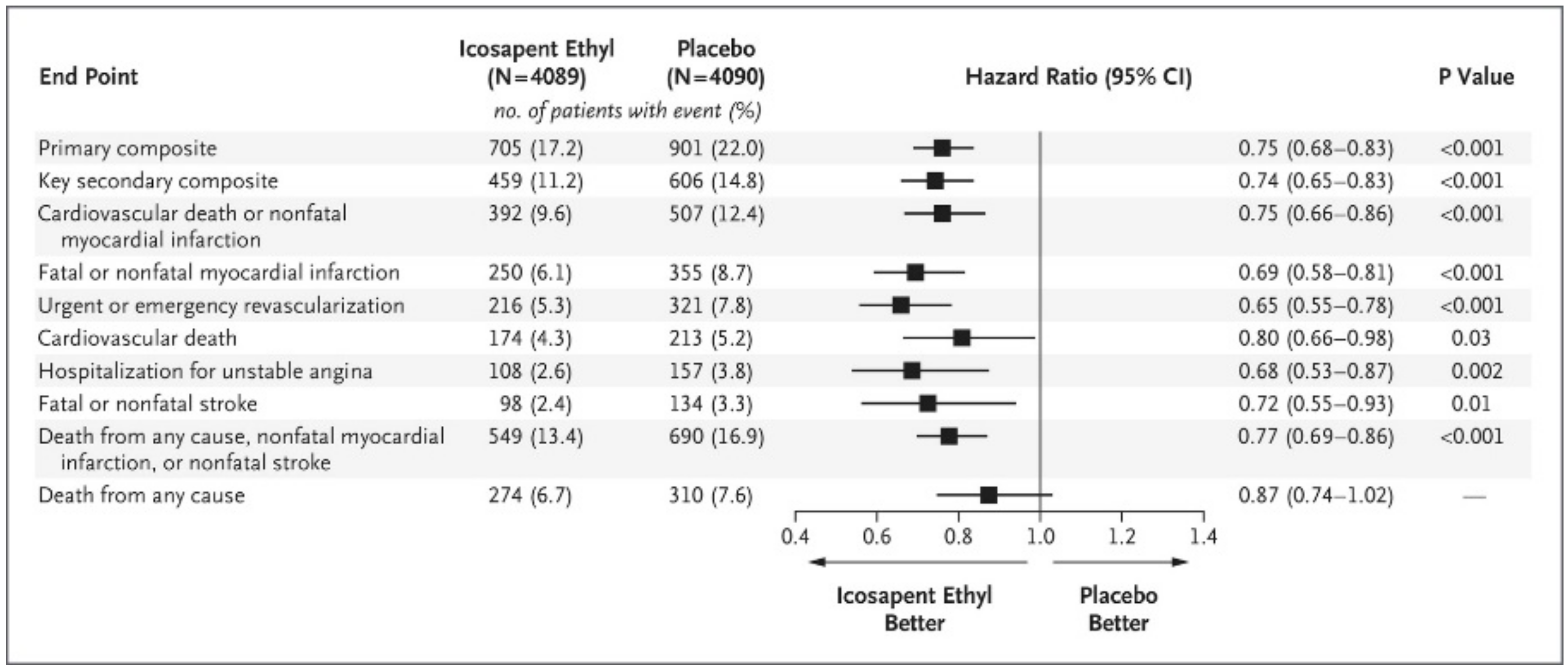
5.3. An Important RCT and a New Meta-Analysis were Published in 2019
6. Trusted Authority Views on Marine n-3 Fatty Acids and Cardiovascular Disease
7. Summary, Discussion and Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AHA | American Heart Association |
| ALA | alpha-linolenic acid |
| CHD | coronary heart disease |
| CI | confidence interval |
| CRP | C-reactive protein |
| CVD | cardiovascular disease |
| DHA | docosahexaenoic acid |
| DPA | docosapentaenoic acid |
| EFSA | European Food Safety Authority |
| EPA | eicosapentaenoic acid |
| HDL | high-density lipoprotein |
| IL | interleukin |
| LDL | low-density lipoprotein |
| MD | mean difference |
| MI | myocardial infarction |
| NS | not significant |
| RCT | randomised controlled trial |
| SMD | standard mean difference |
| TNF | tumour necrosis factor |
| WMD | weighted mean difference |
References
- Calder, P.C. Very long-chain n-3 fatty acids and human health: Fact, fiction and the future. Proc. Nutr. Soc. 2018, 77, 52–72. [Google Scholar] [CrossRef]
- Kaur, G.; Cameron-Smith, D.; Garg, M.; Sinclair, A.J. Docosapentaenoic acid (22:5n-3): A review of its biological effects. Prog. Lipid Res. 2011, 50, 28–34. [Google Scholar] [CrossRef]
- Arterburn, L.M.; Hall, E.B.; Oken, H. Distribution, interconversion, and dose response of n-3 fatty acids in humans. Am. J. Clin. Nutr. 2006, 83, 1467S. [Google Scholar] [CrossRef]
- Baker, E.J.; Miles, E.A.; Burdge, G.C.; Yaqoob, P.; Calder, P.C. Metabolism and functional effects of plant-derived omega-3 fatty acids in humans. Prog. Lipid Res. 2016, 64, 30–56. [Google Scholar] [CrossRef]
- Meyer, B.J.; Mann, N.J.; Lewis, J.L.; Milligan, G.C.; Sinclair, A.J.; Howe, P.R. Dietary intakes and food sources of omega-6 and omega-3 polyunsaturated fatty acids. Lipids 2003, 38, 391–398. [Google Scholar] [CrossRef]
- Howe, P.; Meyer, B.; Record, S.; Baghurst, K. Dietary intake of long-chain omega-3 polyunsaturated fatty acids: Contribution of meat sources. Nutrition 2006, 22, 47–53. [Google Scholar] [CrossRef]
- Scientific Advisory Committee on Nutrition/Committee on Toxicity. Advice on Fish Consumption: Benefits and Risks; TSO: London, UK, 2004. [Google Scholar]
- Food and Agricultural Organisation of the United Nations. Fat and Fatty Acids in Human Nutrition: Report of an Expert Consultation; Food and Agricultural Organisation of the United Nations: Rome, Italy, 2010. [Google Scholar]
- European Food Safety Authority. Panel on Dietetic Products, Nutrition and Allergies (NDA); Scientific Opinion on the substantiation of health claims related to eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA), docosapentaenoic acid (DPA) and maintenance. EFSA J. 2010, 8, 1796. [Google Scholar] [CrossRef]
- Kromann, N.; Green, A. Epidemiological studies in the Upernavik district, Greenland. J. Intern. Med. 1980, 208, 401–406. [Google Scholar] [CrossRef]
- Bjerregaard, P.; Dyerberg, J. Mortality from ischaemic heart disease and cerebrovascular disease in Greenland. Int. J. Epidemiol. 1988, 17, 514–519. [Google Scholar] [CrossRef]
- Bang, H.; Dyerberg, J.; Hjørne, N. The composition of food consumed by Greenland Eskimos. J. Intern. Med. 1976, 200, 69–73. [Google Scholar] [CrossRef]
- Newman, W.; Middaugh, J.; Propst, M.; Rogers, D. Atherosclerosis in Alaska Natives and non-Natives. Lancet 1993, 341, 1056–1057. [Google Scholar] [CrossRef]
- Yano, K.; MacLean, C.J.; Reed, D.M.; Shimizu, Y.; Sasaki, H.; Kodama, K.; Kato, H.; Kagan, A. A comparison of the 12-year mortality and predictive factors of coronary heart disease among Japanese men in Japan and Hawaii. Am. J. Epidemiol. 1988, 127, 476–487. [Google Scholar] [CrossRef]
- Calder, P.C. n-3 Fatty acids and cardiovascular disease: Evidence explained and mechanisms explored. Clin. Sci. 2004, 107, 1–11. [Google Scholar] [CrossRef]
- Hu, F.; Bronner, L.; Willett, W.; Stampfer, M.; Rexrode, K.M.; Albert, C.; Hunter, D.; Manson, J. Fish and omega-3 fatty acid intake and risk of coronary heart disease in women. JAMA 2002, 287, 1815–1821. [Google Scholar] [CrossRef]
- Browning, L.M.; Walker, C.G.; Mander, A.P.; West, A.L.; Madden, J.; Gambell, J.M.; Young, S.; Wang, L.; Jebb, S.A.; Calder, P.C. Incorporation of eicosapentaenoic and docosahexaenoic acids into lipid pools when given as supplements providing doses equivalent to typical intakes of oily fish. Am. J. Clin. Nutr. 2012, 96, 748–758. [Google Scholar] [CrossRef]
- Albert, C.; Campos, H.; Stampfer, M.; Ridker, P.; Manson, J.; Willett, W.; Ma, J. Blood levels of long-chain n–3 fatty acids and the risk of sudden death. N. Engl. J. Med. 2002, 346, 1113–1118. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhuang, P.; He, W.; Chen, J.N.; Wang, W.Q.; Freedman, N.D.; Abnet, C.C.; Wang, J.B.; Jiao, J.J. Association of fish and long-chain omega-3 fatty acids intakes with total and cause-specific mortality: Prospective analysis of 421 309 individuals. J. Intern. Med. 2018, 284, 399–417. [Google Scholar] [CrossRef]
- Djoussé, L.; Akinkuolie, A.; Wu, J.; Ding, E.; Gaziano, J. Fish consumption, omega-3 fatty acids and risk of heart failure: A meta-analysis. Clin. Nutr. 2012, 31, 846–853. [Google Scholar] [CrossRef]
- Chowdhury, R.; Warnakula, S.; Kunutsor, S.; Crowe, F.; Ward, H.; Johnson, L.; Franco, O.; Butterworth, A.; Forouhi, N.; Thompson, S.; et al. Association of dietary, circulating, and supplement fatty acids with coronary risk: A systematic review and meta-analysis. Ann. Intern. Med. 2014, 160, 398–406. [Google Scholar] [CrossRef]
- Alexander, D.D.; Miller, P.E.; Van Elswyk, M.E.; Kuratko, C.N.; Bylsma, L.C. A meta-analysis of randomized controlled trials and prospective cohort studies of eicosapentaenoic and docosahexaenoic long-chain omega-3 fatty acids and coronary heart disease risk. Mayo Clin. Proc. 2017, 92, 15–29. [Google Scholar] [CrossRef]
- Del Gobbo, L.C.; Imamura, F.; Aslibekyan, S.; Marklund, M.; Virtanen, J.K.; Wennberg, M.; Yakoob, M.Y.; Chiuve, S.E.; Dela Cruz, L.; Frazier-Wood, A.C.; et al. ω-3 Polyunsaturated fatty acid biomarkers and coronary heart disease: Pooling project of 19 cohort studies. JAMA Intern. Med. 2016, 176, 1155–1166. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Von Schacky, C. The Omega-3 Index: A new risk factor for death from coronary heart disease? Prev. Med. 2004, 39, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Del Gobbo, L.; Tintle, N.L. The omega-3 index and relative risk for coronary heart disease mortality: Estimation from 10 cohort studies. Atherosclerosis 2017, 262, 51–54. [Google Scholar] [CrossRef] [PubMed]
- AbuMweis, S.; Jew, S.; Tayyem, R.; Agraib, L. Eicosapentaenoic acid and docosahexaenoic acid containing supplements modulate risk factors for cardiovascular disease: A meta-analysis of randomised placebo-control human clinical trials. J. Hum. Nutr. Diet. 2018, 31, 67–84. [Google Scholar] [CrossRef]
- Miller, P.E.; Van Elswyk, M.; Alexander, D.D. Long-chain omega-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid and blood pressure: A meta-analysis of randomized controlled trials. Am. J. Hypertens. 2014, 27, 885–896. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Geelen, A.; Brouwer, I.A.; Geleijnse, J.M.; Zock, P.L.; Katan, M.B. Effect of fish oil on heart rate in humans: A meta-analysis of randomized controlled trials. Circulation 2005, 112, 1945–1952. [Google Scholar] [CrossRef]
- Xin, W.; Wei, W.; Li, X. Short-term effects of fish-oil supplementation on heart rate variability in humans: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2013, 97, 926–935. [Google Scholar] [CrossRef]
- Hidayat, K.; Yang, J.; Zhang, Z.; Chen, G.-C.; Qin, L.-Q.; Eggersdorfer, M.; Zhang, W. Effect of omega-3 long-chain polyunsaturated fatty acid supplementation on heart rate: A meta-analysis of randomized controlled trials. Eur. J. Clin. Nutr. 2017. [Google Scholar] [CrossRef]
- Gao, L.; Cao, J.; Mao, Q.X.; Lu, X.X.; Zhou, X.L.; Fan, L. Influence of omega-3 polyunsaturated fatty acid-supplementation on platelet aggregation in humans: A meta-analysis of randomized controlled trials. Atherosclerosis 2013, 226, 328–334. [Google Scholar] [CrossRef]
- Xin, W.; Wei, W.; Li, X. Effect of fish oil supplementation on fasting vascular endothelial function in humans: A meta-analysis of randomized controlled trials. PLoS ONE 2012, 7, e46028. [Google Scholar] [CrossRef]
- Wang, Q.; Liang, X.; Wang, L.; Lu, X.; Huang, J.; Cao, J.; Li, H.; Gu, D. Effect of omega-3 fatty acids supplementation on endothelial function: A meta-analysis of randomized controlled trials. Atherosclerosis 2012, 221, 536–543. [Google Scholar] [CrossRef]
- Pase, M.P.; Grima, N.A.; Sarris, J. Do long-chain n-3 fatty acids reduce arterial stiffness? A meta-analysis of randomised controlled trials. Br. J. Nutr. 2011, 106, 974–980. [Google Scholar] [CrossRef]
- Li, K.; Huang, T.; Zheng, J.; Wu, K.; Li, D. Effect of marine-derived n-3 polyunsaturated fatty acids on C-reactive protein, interleukin-6 and tumor necrosis factor alpha: A meta-analysis. PLoS ONE 2014, 9, e88103. [Google Scholar] [CrossRef]
- Jiang, J.; Li, K.; Wang, F.; Yang, B.; Fu, Y.; Zheng, J.; Li, D. Effect of marine-derived n-3 polyunsaturated fatty acids on major eicosanoids: A systematic review and meta-analysis from 18 randomized controlled trials. PLoS ONE 2016, 11, e0147351. [Google Scholar] [CrossRef] [PubMed]
- Innes, J.K.; Calder, P.C. The differential effects of eicosapentaenoic acid and docosahexaenoic acid on cardiometabolic risk factors: A systematic review. Int. J. Mol. Sci. 2018, 19, 532. [Google Scholar] [CrossRef] [PubMed]
- Grimsgaard, S.; Bonna, K.H.; Hansen, J.-B.; Nordøy, A. Highly purified eicosapentaenoic acid and docosahexaenoic acid in humans have similar triacylglycerol-lowering effects but divergent effects on serum fatty acids. Am. J. Clin. Nutr. 1997, 66, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Allaire, J.; Couture, P.; Leclerc, M.; Charest, A.; Marin, J.; Lépine, M.-C.; Talbot, D.; Tchernof, A.; Lamarche, B. A randomized, crossover, head-to-head comparison of eicosapentaenoic acid and docosahexaenoic acid supplementation to reduce inflammation markers in men and women: The Comparing EPA to DHA (ComparED) Study. Am. J. Clin. Nutr. 2016, 104, 280–287. [Google Scholar] [CrossRef]
- Woodman, R.J.; Mori, T.A.; Burke, V.; Puddey, I.B.; Watts, G.F.; Beilin, L.J. Effects of purified eicosapentaenoic and docosahexaenoic acids on glycemic control, blood pressure, and serum lipids in type 2 diabetic patients with treated hypertension. Am. J. Clin. Nutr. 2002, 76, 1007–1015. [Google Scholar] [CrossRef]
- Mori, T.A.; Burke, V.; Puddey, I.B.; Watts, G.F.; O’Neal, D.N.; Best, J.D.; Beilin, L.J. Purified eicosapentaenoic and docosahexaenoic acids have differential effects on serum lipids and lipoproteins, LDL particle size, glucose, and insulin in mildly hyperlipidemic men. Am. J. Clin. Nutr. 2000, 71, 1085–1094. [Google Scholar] [CrossRef]
- Grimsgaard, S.; Bonaa, K.; Hansen, J.; Myhre, E. Effects of highly purified eicosapentaenoic acid and docosahexaenoic acid on hemodynamics in humans. Am. J. Clin. Nutr. 1998, 68, 52–59. [Google Scholar] [CrossRef]
- Mori, T.; Bao, D.; Burke, V.; Puddey, I.; Beilin, L. Docosahexaenoic acid but not eicosapentaenoic acid lowers ambulatory blood pressure and heart rate in humans. Hypertension 1999, 34, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.A.; Watts, G.F.; Burke, V.; Hilme, E.; Puddey, I.B.; Beilin, L.J. Differential effects of eicosapentaenoic acid and docosahexaenoic acid on vascular reactivity of the forearm microcirculation in hyperlipidaemic, overweight men. Circulation 2000, 102, 1264–1269. [Google Scholar] [CrossRef] [PubMed]
- Nestel, P.; Shige, H.; Pomeroy, S.; Cehun, M.; Abbey, M.; Raederstorff, D. The n-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid increase systemic arterial compliance in humans. Am. J. Clin. Nutr. 2002, 76, 326–330. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Harris, W. EPA, but not DHA, decreases mean platelet volume in normal subjects. Lipids 2002, 37, 941–946. [Google Scholar] [CrossRef] [PubMed]
- Woodman, R.J.; Mori, T.A.; Burke, V.; Puddey, I.B.; Barden, A.; Watts, G.F.; Beilin, L.J. Effects of purified eicosapentaenoic acid and docosahexaenoic acid on platelet, fibrinolytic and vascular function in hypertensive type 2 diabetic patients. Atherosclerosis 2003, 166, 85–93. [Google Scholar] [CrossRef]
- Vors, C.; Allaire, J.; Marin, J.; Lepine, M.C.; Charest, A.; Tchernof, A.; Couture, P.; Lamarche, B. Inflammatory gene expression in whole blood cells after EPA vs. DHA supplementation: Results from the ComparED study. Atherosclerosis 2017, 257, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.A.; Puddey, I.B.; Burke, V.; Croft, K.D.; Dunstan, D.W.; Rivera, J.H.; Beilin, L.J. Effect of omega 3 fatty acids on oxidative stress in humans: GC-MS measurement of urinary F-2-isoprostane excretion. Redox Rep. 2000, 5, 45–46. [Google Scholar] [CrossRef]
- Mas, E.; Woodman, R.J.; Burke, V.; Puddey, I.B.; Beilin, L.J.; Durand, T.; Mori, T.A. The omega-3 fatty acids EPA and DHA decrease plasma F2-isoprostanes: Results from two placebo-controlled interventions. Free Radic. Res. 2010, 44, 983–990. [Google Scholar] [CrossRef]
- Mori, T.A.; Woodman, R.J.; Burke, V.; Puddey, I.B.; Croft, K.D.; Beilin, L.J. Effect of eicosapentaenoic acid and docosahexaenoic acid on oxidative stress and inflammatory markers in treated-hypertensive type 2 diabetic subjects. Free Radic. Biol. Med. 2003, 35, 772–781. [Google Scholar] [CrossRef]
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsuzawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): A randomised open- label, blinded endpoint analysis. Lancet 2007, 369, 1090–1098. [Google Scholar] [CrossRef]
- Bowman, L.; Mafham, M.; Wallendszus, K.; Stevens, W.; Buck, G.; Barton, J.; Murphy, K.; Aung, T.; Haynes, R.; Cox, J.; et al. Effects of n-3 fatty acid supplements in diabetes mellitus. N. Engl. J. Med. 2018, 379, 1540–1550. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.A.E.; Cook, N.R.; Lee, I.M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Albert, C.M.; Gordon, D.; Copeland, T.; et al. Marine n-3 fatty acids and prevention of cardiovascular disease and cancer. N. Engl. J. Med. 2019, 380, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Burr, M.L.; Gilbert, J.F.; Holliday, R.M.; Elwood, P.C.; Fehily, A.M.; Rogers, S.; Sweetnam, P.M.; Deadman, N.M. Effects of changes in fat, fish, and fibre intakes on death and myocardial reinfarction: Diet and reinfarction trial (DART). Lancet 1989, 344, 757–761. [Google Scholar] [CrossRef]
- The GISSI-Prevenzione Investigators. Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E after myocardial infarction: Results of the GISSI-Prevenzione trial. Lancet 1999, 354, 447–455. [Google Scholar] [CrossRef]
- Marchioli, R.; Barzi, F.; Bomba, E.; Chieffo, C.; Di Gregorio, D.; Di Mascio, R.; Franzosi, M.G.; Geraci, E.; Levantesi, G.; Maggioni, A.; et al. Early protection against sudden death by n-3 polyunsaturated fatty acids after myocardial infarction: Time-course analysis of the results of the Gruppo Italiano per lo Studio della Sopravvivenza nell’Infarto Miocardico (GISSI)-Prevenzione. Circulation 2002, 105, 1897–1903. [Google Scholar] [CrossRef]
- Romagna, E. The GISSI-HF Investigators Effect of n-3 polyunsaturated fatty acids in patients with chronic heart failure (the GISSI-HF trial): A randomised, double-blind, placebo-controlled trial. Lancet 2008, 372, 1223–1230. [Google Scholar] [CrossRef]
- Itakura, H.; Yokoyama, M.; Matsuzaki, M.; Saito, Y.; Origasa, H.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Kita, T.; et al. Relationships between plasma fatty acid composition and coronary artery disease. J. Atheroscler. Thromb. 2011, 18, 99–107. [Google Scholar] [CrossRef]
- Bucher, H.C.; Hengstler, P.; Schindler, C.; Meier, G. N-3 polyunsaturated fatty acids in coronary heart disease: A meta-analysis of randomized controlled trials. Am. J. Med. 2002, 112, 298–304. [Google Scholar] [CrossRef]
- Studer, M.; Briel, M.; Leimenstoll, B.; Glass, T.R.; Bucher, H.C. Effect of different antilipidemic agents and diets on mortality: A systematic review. Arch. Intern. Med. 2005, 165, 725–730. [Google Scholar] [CrossRef]
- Zhao, Y.-T.; Chen, Q.; Sun, Y.-X.; Li, X.-B.; Zhang, P.; Xu, Y.; Guo, J.-H. Prevention of sudden cardiac death with omega-3 fatty acids in patients with coronary heart disease: A meta-analysis of randomized controlled trials. Ann. Med. 2009, 41, 301–310. [Google Scholar] [CrossRef]
- Marik, P.E.; Varon, J. Omega-3 dietary supplements and the risk of cardiovascular events: A systematic review. Clin. Cardiol. 2009, 32, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Rauch, B.; Schiele, R.; Schneider, S.; Diller, F.; Victor, N.; Gohlke, H.; Gottwik, M.; Steinbeck, G.; Del Castillo, U.; Sack, R.; et al. OMEGA, a randomized, placebo-controlled trial to test the effect of highly purified omega-3 fatty acids on top of modern guideline-adjusted therapy after myocardial infarction. Circulation 2010, 122, 2152–2159. [Google Scholar] [CrossRef] [PubMed]
- Galan, P.; Kesse-Guyot, E.; Czernichow, S.; Briancon, S.; Blacher, J.; Hercberg, S. Effects of B vitamins and omega 3 fatty acids on cardiovascular diseases: A randomised placebo controlled trial. BMJ 2010, 341, c6273. [Google Scholar] [CrossRef] [PubMed]
- Kromhout, D.; Giltay, E.J.; Geleijnse, J.M. n–3 Fatty acids and cardiovascular events after myocardial infarction. N. Engl. J. Med. 2010, 363, 2015–2026. [Google Scholar] [CrossRef]
- Bosch, J.; Gerstein, H.; Dagenais, G.; Diaz, R.; Dyal, L.; Jung, H.; Maggiono, A.; Probstfield, J.; Ramachandran, A.; Riddle, M.; et al. n–3 Fatty acids and cardiovascular outcomes in patients with dysglycemia (ORIGIN). N. Engl. J. Med. 2012, 367, 309–318. [Google Scholar] [CrossRef]
- Roncaglioni, M.; Tombesi, M.; Avanzini, F.; Barlera, S.; Caimi, V.; Longoni, P.; Marzona, I.; Milani, V.; Silletta, M.; Tognoni, G.; et al. n–3 Fatty acids in patients with multiple cardiovascular risk factors (Risk and Prevention Study). N. Engl. J. Med. 2013, 368, 1800–1808. [Google Scholar] [CrossRef]
- Calder, P.C.; Yaqoob, P. Marine omega-3 fatty acids and coronary heart disease. Curr. Opin. Cardiol. 2012, 27, 412–419. [Google Scholar] [CrossRef]
- Calder, P.C. Limited impact of omega-3 fatty acids in patients with multiple cardiovascular risk factors. Evid. Based Med. 2014, 19, 18. [Google Scholar] [CrossRef]
- Kotwal, S.; Jun, M.; Sullivan, D.; Perkovic, V.; Neal, B. Omega 3 fatty acids and cardiovascular outcomes: Systematic review and meta-analysis. Circ. Cardiovasc. Qual. Outcomes 2012, 5, 808–818. [Google Scholar] [CrossRef]
- Kwak, S.M.; Myung, S.K.; Lee, Y.J.; Seo, H.G. Efficacy of omega-3 fatty acid supplements (eicosapentaenoic acid and docosahexaenoic acid) in the secondary prevention of cardiovascular disease: A meta-analysis of randomized, double-blind, placebo-controlled trials. Arch. Intern. Med. 2012, 172, 686–694. [Google Scholar] [CrossRef]
- Trikalinos, T.; Lee, J.; Moorthy, D.; Yu, W.; Lau, J.; Lichtenstein, A.; Chung, M. Effects of eicosapentanoic acid and docosahexanoic acid on mortality across diverse settings: Systematic review and meta-analysis of randomized trials and prospective cohorts. Tech. Rev. 2012, 17, 4. [Google Scholar]
- Rizos, E.; Ntzani, E.; Bika, E.; Kostapanos, M.; Elisaf, M. Association between omega-3 fatty acid supplementation and risk of major cardiovascular disease events: A systematic review and meta-analysis. JAMA 2012, 308, 1024–1033. [Google Scholar] [CrossRef] [PubMed]
- Casula, M.; Soranna, D.; Catapano, A.L.; Corrao, G. Long-term effect of high dose omega-3 fatty acid supplementation for secondary prevention of cardiovascular outcomes: A meta-analysis of randomized, double blind, placebo controlled trials. Atheroscler. Suppl. 2013, 14, 243–251. [Google Scholar] [CrossRef]
- Wen, Y.T.; Dai, J.H.; Gao, Q. Effects of omega-3 fatty acid on major cardiovascular events and mortality in patients with coronary heart disease: A meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 470–475. [Google Scholar] [CrossRef]
- Maki, K.C.; Palacios, O.M.; Bell, M.; Toth, P.P. Use of supplemental long-chain omega-3 fatty acids and risk for cardiac death: An updated meta-analysis and review of research gaps. J. Clin. Lipidol. 2017, 11, 1152–1160.e2. [Google Scholar] [CrossRef]
- Aung, T.; Halsey, J.; Kromhout, D.; Gerstein, H.C.; Marchioli, R.; Tavazzi, L.; Geleijnse, J.M.; Rauch, B.; Ness, A.; Galan, P.; et al. Associations of omega-3 fatty acid supplement use with cardiovascular disease risks. JAMA Cardiol. 2018, 3, 225–234. [Google Scholar] [CrossRef]
- Abdelhamid, A.; Brown, T.; Brainard, J.; Biswas, P.; Thorpe, G.; Moore, H.; Deane, K.; AlAbdulghafoor, F.; Summerbell, C.; Worthington, H.; et al. Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2018, CD003177. [Google Scholar] [CrossRef]
- Bhatt, D.L.; Steg, P.G.; Miller, M.; Brinton, E.A.; Jacobson, T.A.; Ketchum, S.B.; Doyle, R.T.; Juliano, R.A.; Jiao, L.; Granowitz, C.; et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N. Engl. J. Med. 2019, 380, 11–22. [Google Scholar] [CrossRef]
- Hu, Y.; Hu, F.B.; Manson, J.E. Marine omega-3 supplementation and cardiovascular disease: An updated meta-analysis of 13 randomized controlled trials involving 127,477 participants. J. Am. Heart Assoc. 2019, 8, e013543. [Google Scholar] [CrossRef]
- Einvik, G.; Ole Klemsdal, T.; Sandvik, L.; Hjerkinn, E.M. A randomized clinical trial on n-3 polyunsaturated fatty acids supplementation and all-cause mortality in elderly men at high cardiovascular risk. Eur. J. Cardiovasc. Prev. Rehabil. 2010, 17, 588–592. [Google Scholar] [CrossRef]
- Rimm, E.B.; Appel, L.J.; Chiuve, S.E.; Djoussé, L.; Engler, M.B.; Kris-Etherton, P.M.; Mozaffarian, D.; Siscovick, D.S.; Lichtenstein, A.H. Seafood long-chain n-3 polyunsaturated fatty acids and cardiovascular disease: A science advisory from the American Heart Association. Circulation 2018, 138, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Skulas-Ray, A.C.; Wilson, P.W.F.; Harris, W.S.; Brinton, E.A.; Kris-Etherton, P.M.; Richter, C.K.; Jacobson, T.A.; Engler, M.B.; Miller, M.; Robinson, J.G.; et al. Omega-3 fatty acids for the management of hypertriglyceridemia: A science advisory from the American Heart Association. Circulation 2019, 140, e673–e691. [Google Scholar] [CrossRef] [PubMed]
- Mach, F.; Baigent, C.; Carapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; de Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Eur. Heart J. 2020, 41, 111–188. [Google Scholar] [CrossRef] [PubMed]
- The Living Standards of Medical Care in Diabetes—Updates to the standards of medical care in diabetes. Available online: https://care.diabetesjournals.org/living-standards#March%2027,%202019 (accessed on 7 February 2020).
- Orringer, C.E.; Jacobson, T.A.; Maki, K.C. National Lipid Association Scientific Statement on the use of icosapent ethyl in statin-treated patients with elevated triglycerides and high or very-high ASCVD risk. J. Clin. Lipidol. 2019, 13, 860–872. [Google Scholar] [CrossRef]
- Kris-Etherton, P.M.; Harris, W.S.; Appel, L.J. Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Circulation 2002, 106, 2747–2757. [Google Scholar] [CrossRef]
- Siscovick, D.S.; Barringer, T.A.; Fretts, A.M.; Wu, J.H.Y.; Lichtenstein, A.H.; Costello, R.B.; Kris-Etherton, P.M.; Jacobson, T.A.; Engler, M.B.; Alger, H.M.; et al. Omega-3 polyunsaturated fatty acid (fish oil) supplementation and the prevention of clinical cardiovascular disease: A Science Advisory from the American Heart Association. Circulation 2017, 135, e867–e884. [Google Scholar] [CrossRef]
- NICE Cardiovascular disease: Risk assessment and reduction, including lipid modification (clinical guideline CG181). Available online: https://www.nice.org.uk/guidance/cg181 (accessed on 14 July 2014).
- Von Schacky, C. Omega-3 fatty acids in cardiovascular disease—An uphill battle. Prostagland. Leukot. Essent Fatty Acids 2015, 92, 41–47. [Google Scholar] [CrossRef]
- Rice, H.B.; Bernasconi, A.; Maki, K.C.; Harris, W.S.; von Schacky, C.; Calder, P.C. Conducting omega-3 clinical trials with cardiovascular outcomes: Proceedings of a Workshop held at ISSFAL 2014. Prostagland. Leukot. Essent. Fatty Acids 2016, 107, 30–42. [Google Scholar] [CrossRef]
- Schuchardt, J.P.; Hahn, A. Bioavailability of long-chain omega-3 fatty acids. Prostagland. Leukot. Essent. Fatty Acids 2013, 89, 1–9. [Google Scholar] [CrossRef]
| Fish Type | Typical EPA + DHA per Adult Serving | Comment |
|---|---|---|
| Fatty (e.g., salmon, trout, mackerel, sardines and herring) | 1–3.5 g | Usually more EPA than DHA; content depends on the type of fish, season, water temperature, diet, stage of life cycle, wild or farmed and method of cooking |
| Lean (e.g., cod, plaice, haddock and sea bass) | 0.1–0.3 g | Usually more EPA than DHA |
| Supplement Type | Typical EPA + DHA Content per g of oil | |
| Cod liver oil | 200 mg | Usually more EPA than DHA |
| Standard “fish oil” | 300 mg | Usually more EPA than DHA |
| Fish oil concentrate | 450–600 mg | Usually more EPA than DHA |
| Tuna oil | 460 mg | More DHA than EPA |
| Krill oil | 205 mg | Usually more EPA than DHA; some in phospholipid form |
| Algal oil | 400 mg | Mainly DHA |
| Flaxseed oil | 0 mg | Contains α-linolenic acid, but not EPA or DHA |
| Pharmaceuticals | Typical EPA + DHA Content per g of oil | |
| Omacor/Lovaza | 460 mg EPA + 380 mg DHA | In ethyl ester form |
| Omtryg | 465 mg EPA + 375 mg DHA | In ethyl ester form |
| Epanova | 550 mg EPA + 200 mg DHA | In free fatty acid form |
| Vascepa/icosapent ethyl | 900 mg EPA | In ethyl ester form |
| Study | Cardiovascular Risk Factors Assessed | Study Design | Form and Dosage of n-3 Fatty Acids | Duration of n-3 Fatty Acid Treatment | Pooled Effects of n-3 Fatty Acids Versus Placebo |
|---|---|---|---|---|---|
| AbuMweis et al., 2018 [26] | Blood lipids, heart rate, blood pressure, inflammatory markers, platelet function and flow-mediated dilatation | Meta-analysis of 171 RCTs (up to Feb 2013) in participants in various states of health (note: the number of studies used for the analysis of different outcomes varied from 110 for triglycerides and HDL-cholesterol to 9 for flow-mediated dilatation) | Oral marine n-3 fatty acid supplements providing 0.18–15 g/d EPA+DHA | 4–240 weeks | Significant dose-dependent decrease in triglycerides |
| (MD = −0.368 mmol/L; 95% CI: −0.427–−0.309) | |||||
| Significant decrease in systolic blood pressure | |||||
| (MD = −2.195 mmHg; 95% CI: −3.171–−1.217) | |||||
| Significant decrease in diastolic blood pressure | |||||
| (MD = −1.37 mmHg; 95% CI: −2.415–−0.325) | |||||
| Significant decrease in heart rate (MD = −1.37 bpm; 95% CI: −2.41–−0.325) | |||||
| Significant decrease in CRP (MD = −0.343 mg/L; 95% CI: −0.454–−0.232) | |||||
| Significant increase in LDL-cholesterol (MD = 0.150 mmol/L; 95% CI: 0.058–0.243) and HDL-cholesterol (MD = 0.039 mmol/L; 95% CI: 0.024–0.054) | |||||
| No significant effect on total cholesterol, TNF-α, fibrinogen, platelet count, soluble intercellular adhesion molecule 1, soluble vascular cell adhesion molecule 1 or flow-mediated dilatation | |||||
| Gao et al., 2013 [31] | Platelet aggregation | Meta-analysis of 15 RCTs (up to Jul 2011) including 742 participants in various states of health | Oral marine n-3 fatty acid supplements providing 0.84–6.8 g/d EPA+DHA | 2–16 weeks | Significant decrease in adenosine diphosphate-induced platelet aggregation (SMD = −1.23; 95% CI: −2.24–−0.23; p = 0.02) |
| Significant decrease in platelet aggregation units (SMD = −6.78; 95% CI: −12.58–−0.98; p = 0.02) | |||||
| Non-significant trend towards decreased collagen-induced and arachidonic acid-induced platelet aggregation | |||||
| Greater effect observed in non-healthy participants | |||||
| Hidayat et al., 2017 [30] | Heart rate | Meta-analysis of 51 RCTs (up to May 2017) including ~3000 participants in various states of health | Oral marine n-3 fatty acid supplements providing 0.5–15.0 g/d EPA+DHA | 2–52 weeks | Significant decrease in heart rate (WMD = −2.23 bpm; 95% CI: −3.07–−1.40); observed to be due to DHA, not EPA |
| Jiang et al., 2016 [36] | Pro-inflammatory eicosanoids | Meta-analysis of 18 RCTS (up to November 2015) including 826 subjects in various states of health | Oral marine n-3 fatty acid supplements providing 0.18–4.05 g/d EPA+DHA or EPA alone | 4–24 weeks | Significant decrease in serum/plasma thromboxane B2 in participants with high risk of CVD (SMD = −1.26; 95% CI: −1.65–−0.86) |
| Significant decrease in neutrophil leukotriene B4 in unhealthy subjects (SMD = −0.59; 95% CI: −1.02–−0.16) | |||||
| Li et al., 2014 [35] | Pro-inflammatory cytokines | Meta-analysis of 68 RCTs (up to 2013) including 4601 participants in various states of health | Oral marine n-3 fatty acid supplements or dietary intake providing 0.3–6.6 g/d EPA+DHA | 4–12 months | Participants with chronic disease: |
| Significant decrease in CRP (WMD = −0.20 mg/L; 95% CI: −0.28–−0.12) and IL-6 (WMD = −0.22 pg/mL; 95%CI: −0.38–−0.06) | |||||
| No significant effect on TNF-α | |||||
| Healthy participants: Significant decrease in CRP (WMD = −0.18 mg/L; 95% CI: −0.28–−0.08) and TNF-α (WMD = −0.12 pg/mL; 95% CI: −0.16–−0.07) | |||||
| No significant effect on IL-6 | |||||
| Miller et al., 2014 [27] | Blood pressure | Meta-analysis of 70 RCTs (up to February 2013) in normotensive and hypertensive subjects | Oral marine n-3 fatty acids from seafood, fortified foods, fish oil, algal oil and purified ethyl esters; mean EPA+DHA dose: 3.8 g/d | >3 weeks (mean study duration: 69 days) | Significant decrease in systolic blood pressure (WMD = −1.52 mmHg; 95% CI: −2.25–−0.79) |
| Significant decrease in diastolic blood pressure (WMD = −0.99 mmHg; 95% CI: −1.54–−0.44) | |||||
| Significant decrease in systolic blood pressure (WMD = −4.51 mmHg; 95% CI: −6.12–−2.83) and diastolic blood pressure (WMD = −3.05 mmHg; 95% CI: −4.35–−1.74) in hypertensive individuals | |||||
| Mozaffarian et al., 2005 [28] | Heart rate | Meta-analysis of 30 RCTs (up to January 2005) including 1678 healthy participants | Oral marine n-3 fatty acid supplements; median EPA+DHA intake: 3.5 g/d | >2 weeks (median study duration: 8 weeks) | Significant decrease in heart rate (WMD = −1.6 bpm; 95% CI: 0.6–2.5) |
| In those with baseline heart rate ≥ 69 bpm, heart rate decreased by 2.5 bpm (95% CI: 1.4–3.5) | |||||
| Pase et al., 2011 [34] | Arterial stiffness | Meta-analysis of 10 RCTs (up to September 2010) including 550 participants in various states of health | Oral marine n-3 fatty acid supplements providing 0.64–3 g/d EPA+DHA | 6–105 weeks | Significant improvement in pulse wave velocity (SMD = 0.33; 95% CI: 0.12–0.56) |
| Significant improvement in arterial compliance (SMD = 0.48; 95% CI: 0.24–0.72) | |||||
| Wang et al., 2012 [33] | Vascular endothelial function | Meta-analysis of 16 RCTs (up to August 2011) including 901 participants in various states of health | Oral marine n-3 fatty acid supplements and dietary intake providing 0.45–4.7 g/d EPA+DHA | 2 weeks to 12 months (median: 56 days) | Significant increase in flow-mediated dilatation (WMD = 2.3%; 95% CI: 0.89–3.72) |
| No significant change in endothelium-independent vasodilation | |||||
| Xin et al., 2012 [32] | Vascular endothelial function | Meta-analysis of 16 RCTs (up to February 2012) including 1385 participants in various states of health | Oral marine n-3 fatty acid supplements providing 0.45–4.53 g/d EPA+DHA | 2-52 weeks | Significant increase in flow-mediated dilatation (WMD = 1.49%; 95% CI: 0.48–2.5) |
| Xin et al., 2013 [29] | Heart rate variability | Meta-analysis of 15 RCTS including 692 participants in various states of health | Oral marine n-3 fatty acid supplements providing 0.64–5.9 g/d EPA+DHA | 6-24 weeks | Significant increase in high frequency power value of heart rate variability (SMD = 0.30). A sensitivity analysis demonstrated a significant reduction in low frequency power/high frequency power ratio with >1 g/d EPA+DHA |
| Study | Study Design | Form & Dosage of Marine-3 Fatty Acids | Duration of Treatment with Marine n-3 Fatty Acids | Pooled Effects of Marine n-3 Fatty Acids Versus Placebo |
|---|---|---|---|---|
| Bucher et al., 2002 [60] | 11 RCTs (up to August 1999) representing 15,806 patients with CHD | Dietary (2 RCTs) and supplemental (9 RCTs) marine n-3 fatty acids with a dose range of 0.3–6.0 g/d EPA and 0.6–3.7 g/d DHA | 6–46 months (mean: 20 months) | 30% reduction in fatal MI |
| 30% reduction in sudden death | ||||
| 20% reduction in overall mortality | ||||
| Studer et al., 2005 [61] | 14 RCTs (up to June 2003) representing 20,260 participants in primary and secondary prevention settings | Supplemental marine n-3 fatty acids; dose range not given | Mean: 1.9 ± 1.2 years | 23% reduction in overall mortality |
| 32% reduction in cardiovascular mortality | ||||
| Zhao et al., 2009 [62] | 8 RCTs (up to June 2008) representing 20,997 patients with CHD | Dietary (3 RCTs) and supplemental (5 RCTs) marine | 9–108 months (mean: 33 months) | 57% reduction in sudden death in patients with prior MI |
| 39% increased risk of sudden death in patients with angina | ||||
| n-3 fatty acids with a dose range of 0.3–4.1 g/d EPA and 0.4–2.8 g/d DHA | 29% reduction in cardiac death (NS) | |||
| 23% reduction in all-cause mortality (NS) | ||||
| Marik and Varon, 2009 [63] | 11 RCTs (up to December 2008) representing 39,044 patients with all stages of CVD including high-risk and low-risk subjects | Supplemental marine n-3 fatty acids with a dose range of 0.7–4.8 g/d EPA + DHA (mean: 1.8 ± 1.2 g/d) | 1–4.6 years (mean: 2.2 ± 1.2 years) | 13% reduction in cardiovascular death in high-risk patients |
| 13% reduction in sudden cardiac death in high-risk patients | ||||
| 8% reduction in all-cause mortality in high-risk patients | ||||
| 8% reduction in non-fatal cardiovascular events in moderate-risk patients. | ||||
| Kotwal et al., 2012 [71] | 20 RCTs (up to March 2011) representing 62,851 patients in primary and secondary prevention settings | Diet (3 RCTs) and supplemental (17 RCTs) marine n-3 fatty acids with a dose range of 0.8–3.4 g/d EPA + DHA | 6 months–6 years | 14% reduction in vascular death |
| No effect on cardiovascular events, total mortality, coronary events, arrhythmia or cerebrovascular events | ||||
| Kwak et al., 2012 [72] | 14 RCTs (up to April 2011) representing 20,485 patients with CVD | Supplemental marine n-3 fatty acids with a dose range of 0.4–4.8 g/d EPA + DHA (mean: 1.7 g/d EPA + DHA) | 1–4.7 years (mean: 2 years) | 9% reduction in cardiovascular death |
| No effect on cardiovascular events, all-cause mortality, sudden cardiac death, MI, congestive heart failure or stroke | ||||
| Trikalinos et al., 2012 [73] | 18 RCTs (up to May 2011) representing 51,264 patients | Supplemental marine n-3 fatty acids with a dose range of 0.27–6.0 g/d EPA + DHA | 1–5 years | 11% reduction in cardiovascular mortality |
| Rizos et al., 2012 [74] | 20 RCTs (up to August 2012) representing 68,680 patients in primary and secondary prevention settings | Diet (2 RCTs) and supplemental (18 RCTs) marine n-3 fatty acids with a dose range of 0.53–1.80 g/d EPA + DHA (median EPA + DHA dose: 1 g/d) | 1–6.2 years (median: 2 years) | No effect on all-cause mortality, cardiac death, sudden death, MI or stroke |
| Casula et al., 2013 [75] | 11 RCTs (up to March 2013) representing 15,348 patients with CVD | Supplemental marine n-3 fatty acids with a dose range of 1–6 g/d EPA + DHA | ≥ 1 year (duration ranged from 1–3.5 years) | 32% reduction in cardiac death |
| 33% reduction in sudden death | ||||
| 25% reduction in MI | ||||
| 11% reduction in all-cause mortality (NS) | ||||
| No effect on stroke | ||||
| Wen et al., 2014 [76] | 14 RCTs (up to May 2013) representing 32,656 patients with CHD | Supplemental marine n-3 fatty acids with a dose range of 0.4–6.9 g/d EPA + DHA | < 3 months to 4.6 years | 12% reduction in death from cardiac causes |
| 14% reduction in sudden cardiac death | ||||
| 8% reduction in all-cause mortality | ||||
| 7% reduction in cardiovascular events (NS) | ||||
| Chowdhury et al., 2014 [21] | 17 RCTs (up to June 2013) representing 76,580 participants | Supplemental marine n-3 fatty acids with a dose range of 0.3 g/d EPA to 6 g/d EPA + DHA. | 0.1–8 years | 7% reduction in coronary outcomes (NS) |
| Alexander et al., 2017 [22] | 18 RCTs (up to November 2015) | Supplemental marine n-3 fatty acids with a dose range of 0.4–5.0 g/d EPA + DHA | 0.5–7 years | 14%–16% reduction in CHD in high-risk subgroups i.e., those with elevated triglycerides and LDL-cholesterol |
| Maki et al., 2017 [77] | 14 RCTs (up to December 2016) representing 71,899 patients in a mixed/secondary prevention setting | Supplemental marine n-3 fatty acids with a dose range of 0.27–5.0 g/d EPA + DHA | ≥ 6 months (range 0.5–6.2 years) | 8% reduction in cardiac death |
| ~13%–29% reduction in cardiac death in the subgroup with high-risk individuals (secondary prevention, high triglycerides, high LDL-cholesterol and <40% statin use) and with EPA+DHA > 1 g/d | ||||
| Aung et al., 2018 [78] | 10 RCTs representing 77,917 high-risk patients (prior CHD or stroke) | Supplemental marine n-3 fatty acids with a dose range of 0.2–1.8 g/d EPA and 0–1.7 g/d DHA | 1–6.2 years (mean: 4.4 years) | 7% reduction in CHD death (NS) |
| No effect on non-fatal MI, CHD events or major vascular events | ||||
| Abdelhamid et al., 2018 [79] | 79 RCTs (up to April 2017) representing 112,059 participants in primary and secondary prevention settings | Dietary or supplemental marine n-3 fatty acids with a dose range from 0.5 g/d to ~5 g/d EPA + DHA | 1–7 years | 7% reduction in CHD events |
| No effect on all-cause mortality, cardiovascular mortality, cardiovascular events, CHD mortality, stroke or arrhythmia. |
| Outcome | Number of Studies | Finding for Marine n-3 Fatty Acids Versus Placebo | Finding if Data from REDUCE-IT Removed |
|---|---|---|---|
| Rate Ratio [95% Confidence Interval] (p) | Rate Ratio [95% Confidence Interval] (p) | ||
| Myocardial infarction | 13 | 0.88 [0.83, 0.94] (<0.001) | 0.92 [0.86, 0.99] (0.020) |
| CHD death | 12 | 0.92 [0.86, 0.98] (0.014) | Outcome not reported in REDUCE-IT |
| Total CHD | 13 | 0.93 [0.89, 0.96] (<0.001) | 0.95 [0.91, 0.99] (0.008) |
| Total stroke | 13 | 1.02 [0.95, 1.10] (0.569) | 1.05 [0.98, 1.14] (0.183) |
| CVD Death | 12 | 0.92 [0.88, 0.97] (0.003) | 0.93 [0.88, 0.99] (0.013) |
| Total CVD | 13 | 0.95 [0.82, 0.98] (<0.001) | 0.97 [0.94, 0.99] (0.015) |
| Major vascular events | 13 | 0.95 [0.93, 0.98] (<0.001) | 0.97 [0.94, 1.00] 90.058) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Innes, J.K.; Calder, P.C. Marine Omega-3 (N-3) Fatty Acids for Cardiovascular Health: An Update for 2020. Int. J. Mol. Sci. 2020, 21, 1362. https://doi.org/10.3390/ijms21041362
Innes JK, Calder PC. Marine Omega-3 (N-3) Fatty Acids for Cardiovascular Health: An Update for 2020. International Journal of Molecular Sciences. 2020; 21(4):1362. https://doi.org/10.3390/ijms21041362
Chicago/Turabian StyleInnes, Jacqueline K., and Philip C. Calder. 2020. "Marine Omega-3 (N-3) Fatty Acids for Cardiovascular Health: An Update for 2020" International Journal of Molecular Sciences 21, no. 4: 1362. https://doi.org/10.3390/ijms21041362
APA StyleInnes, J. K., & Calder, P. C. (2020). Marine Omega-3 (N-3) Fatty Acids for Cardiovascular Health: An Update for 2020. International Journal of Molecular Sciences, 21(4), 1362. https://doi.org/10.3390/ijms21041362





