The Palladium(II) Complex of Aβ4−16 as Suitable Model for Structural Studies of Biorelevant Copper(II) Complexes of N-Truncated Beta-Amyloids
Abstract
1. Introduction
2. Results
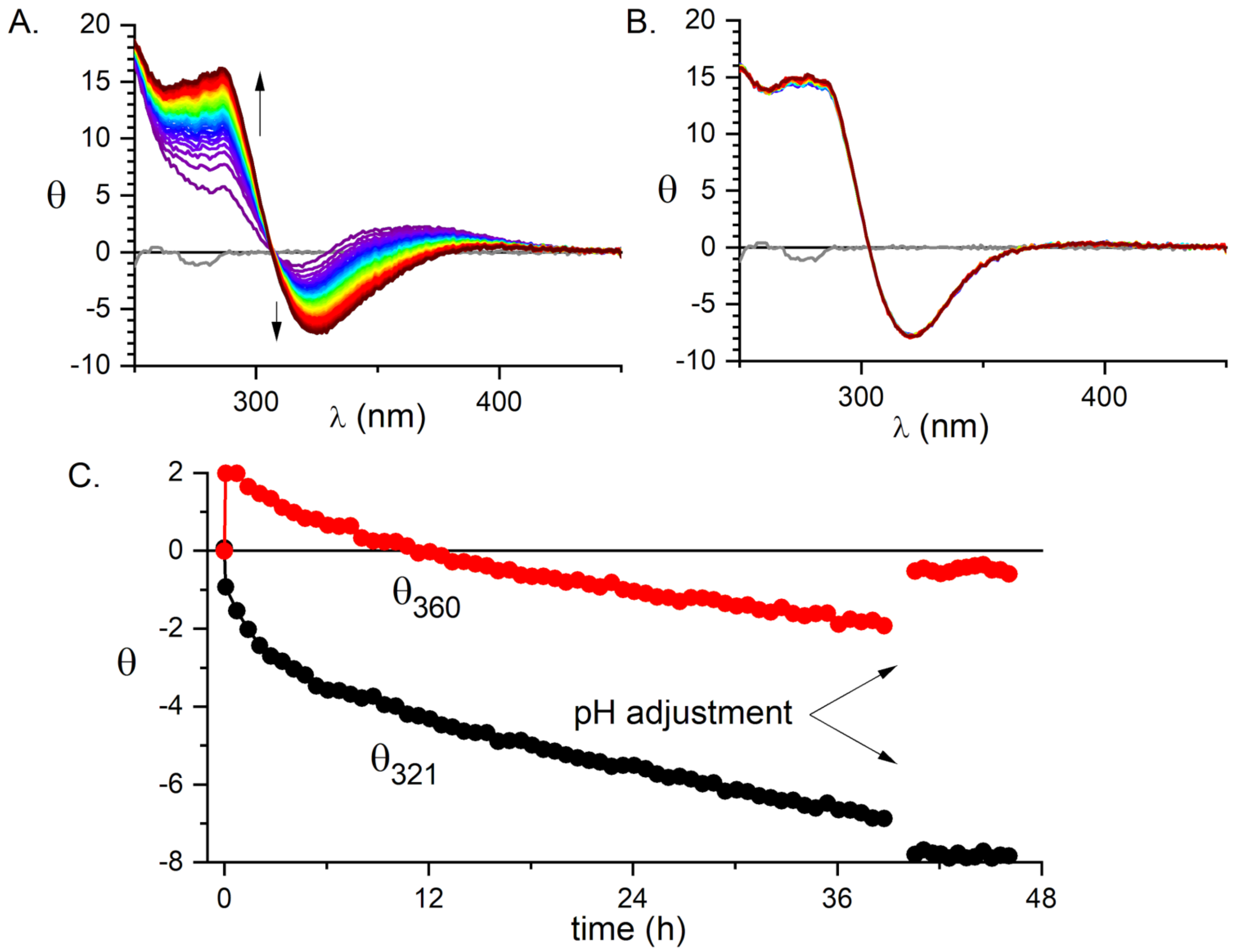
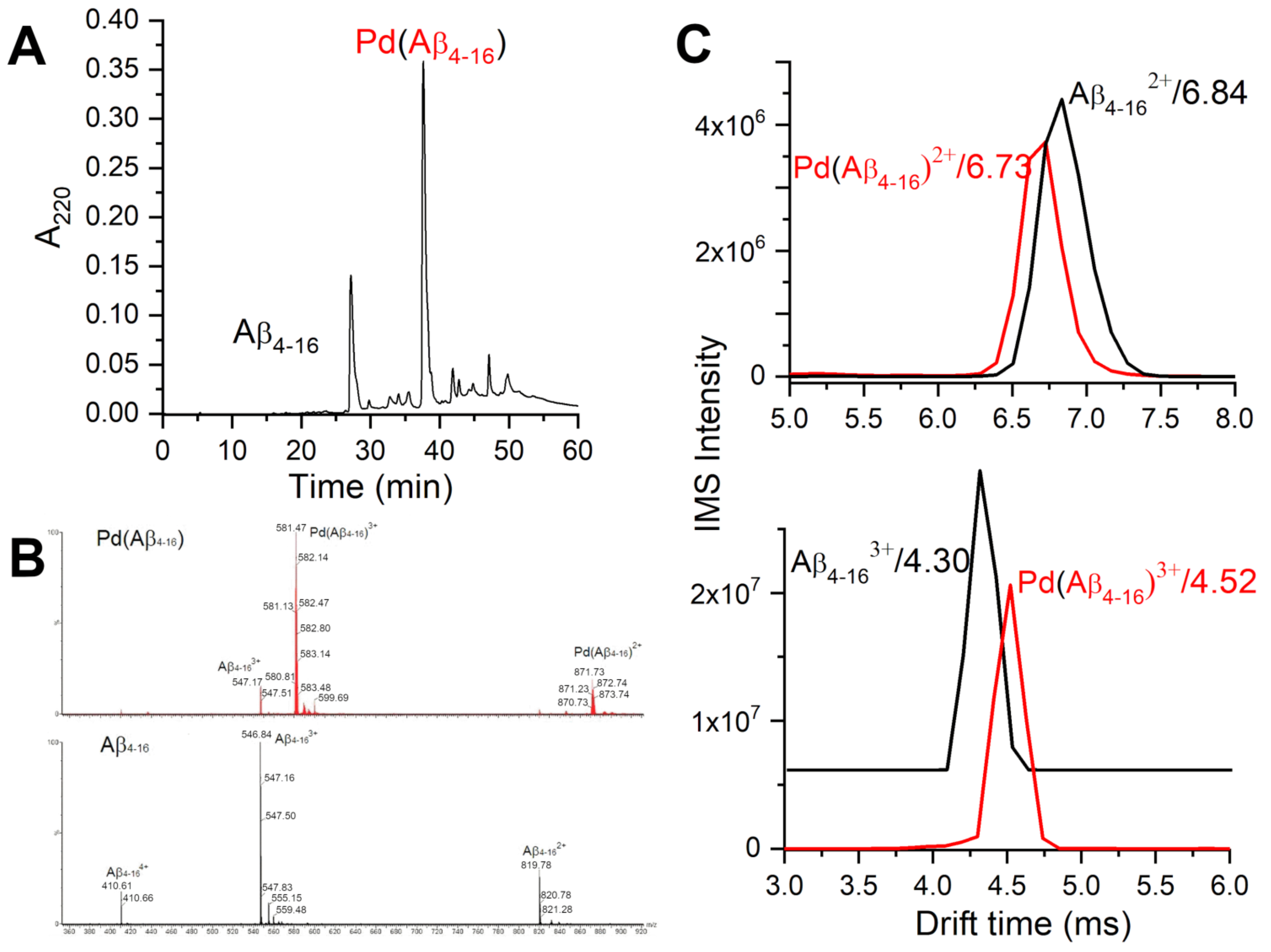
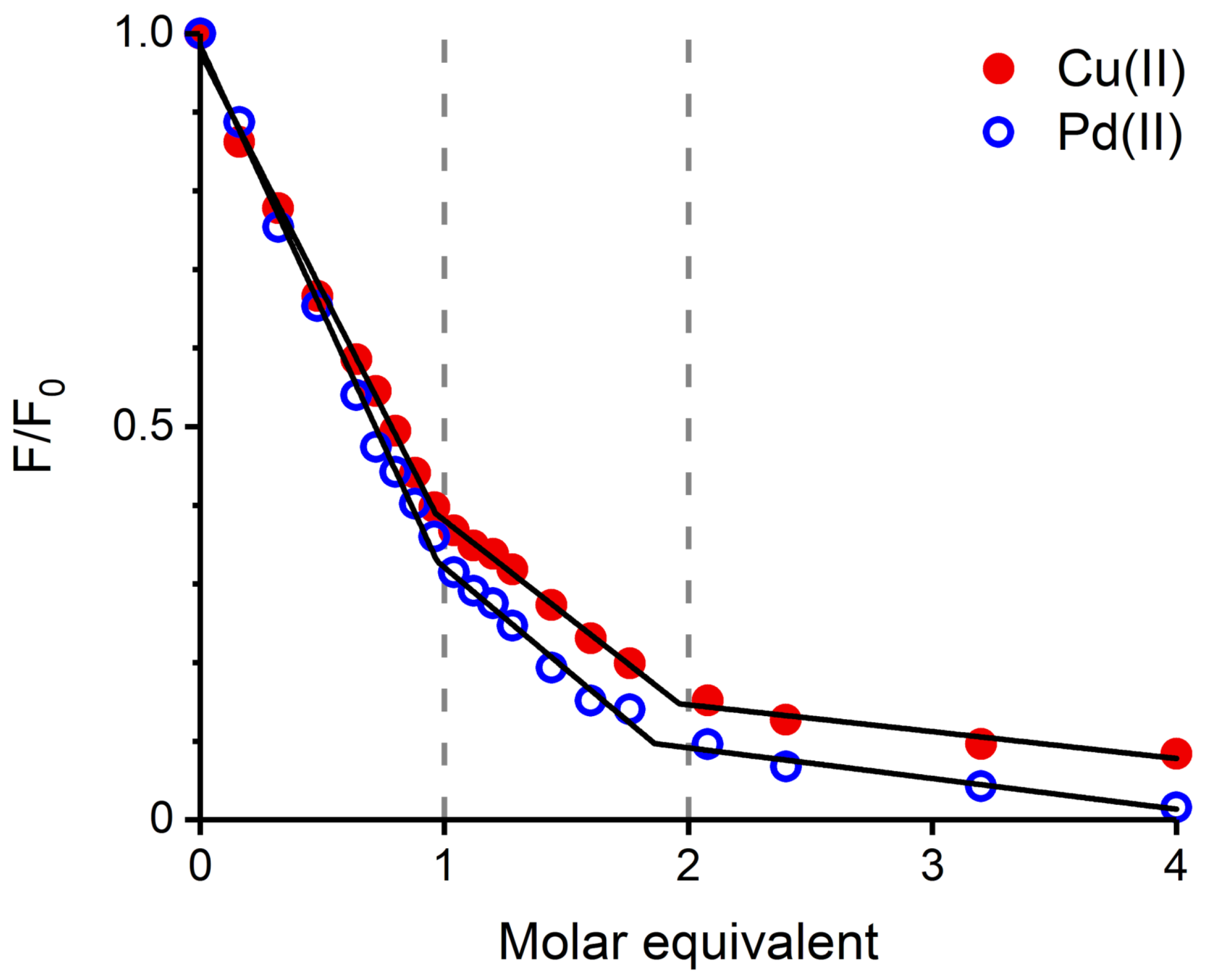
2.1. Validation of the Pd(II) Complex as a Model for the Cu(II) Complex
2.2. Structural Analysis of the A Peptide as a Simple Model of the ATCUN/NTS Site in A
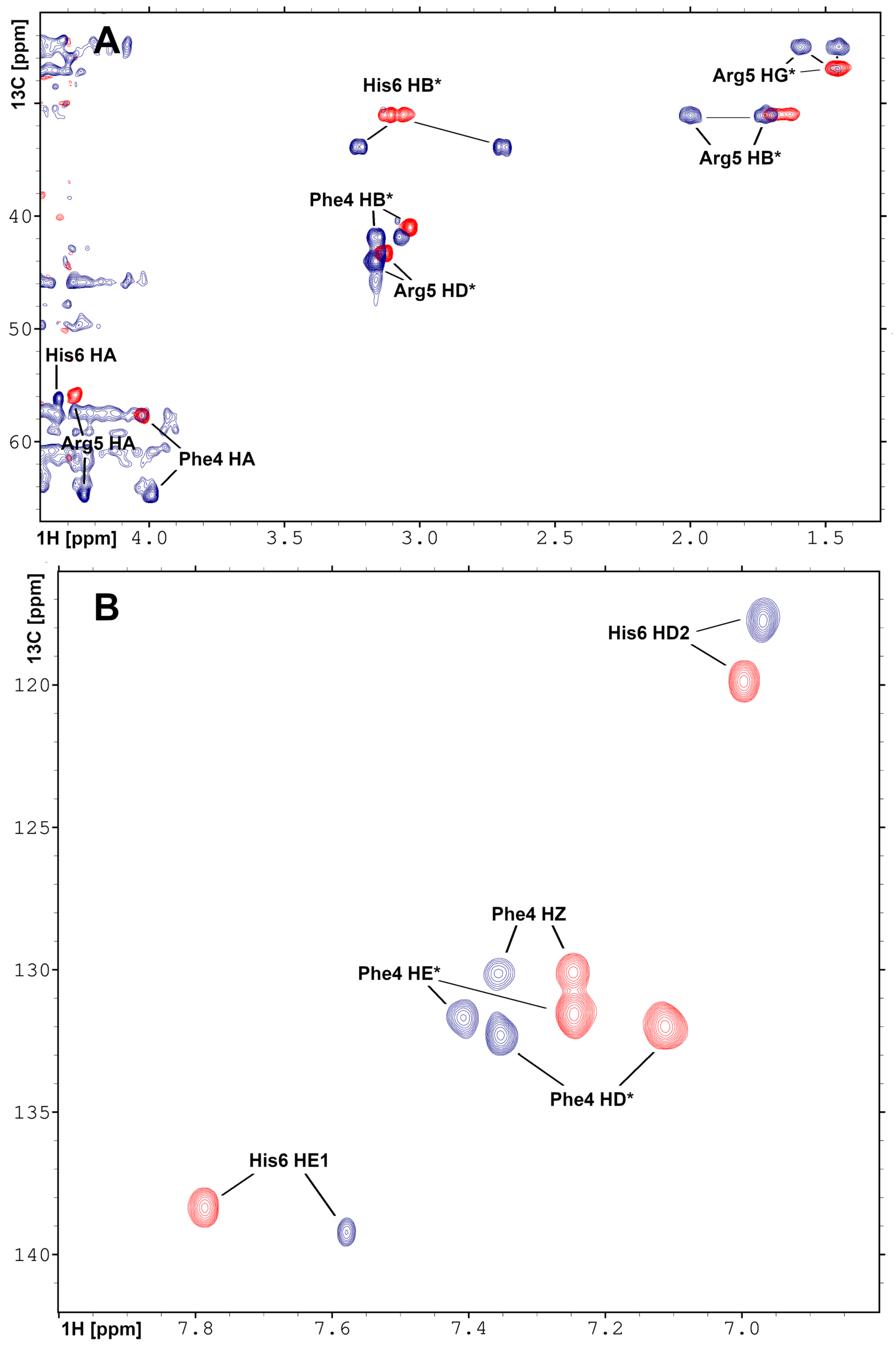
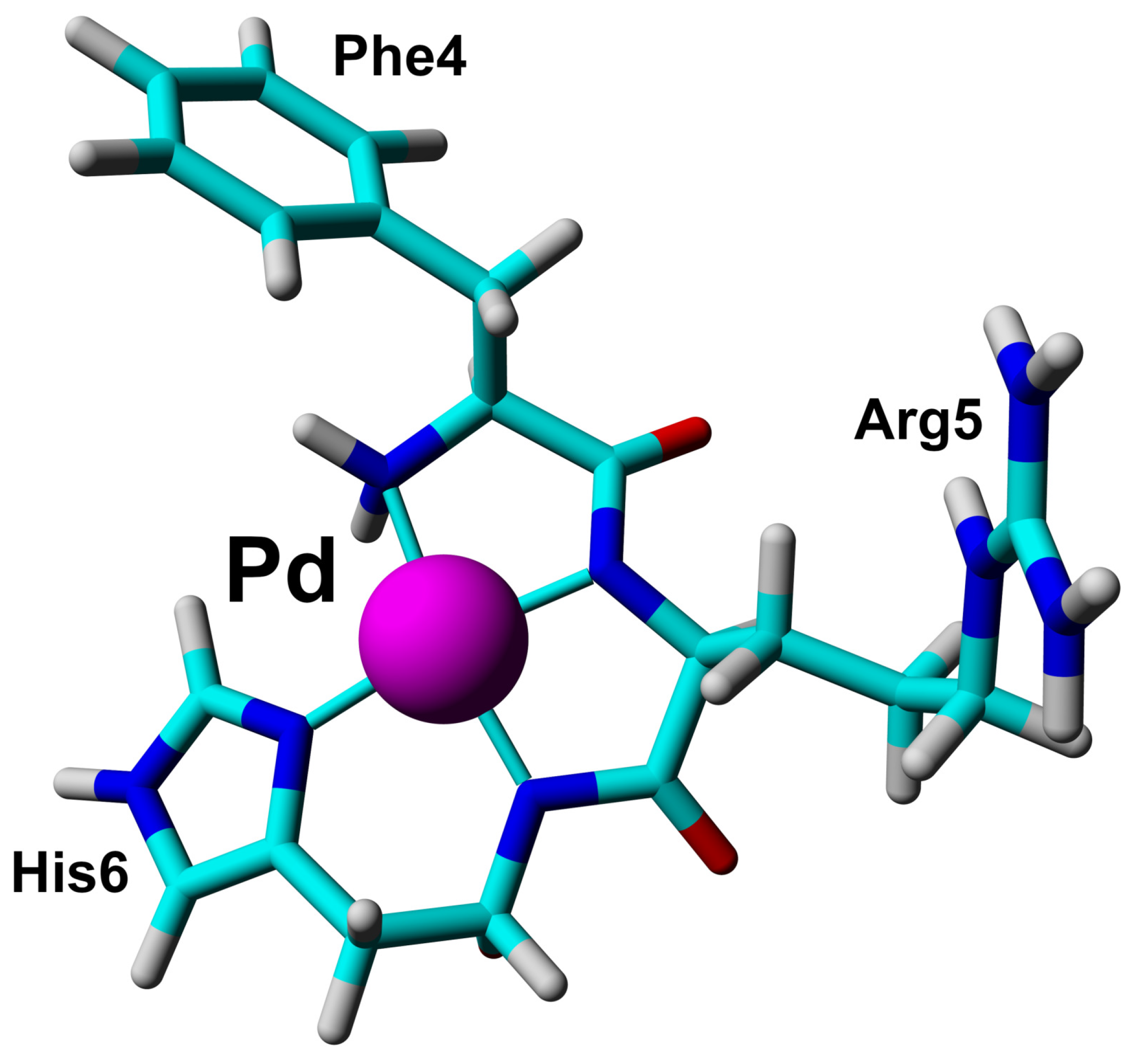
2.3. Solution Structure of the Pd(II) Complex with the A Peptide
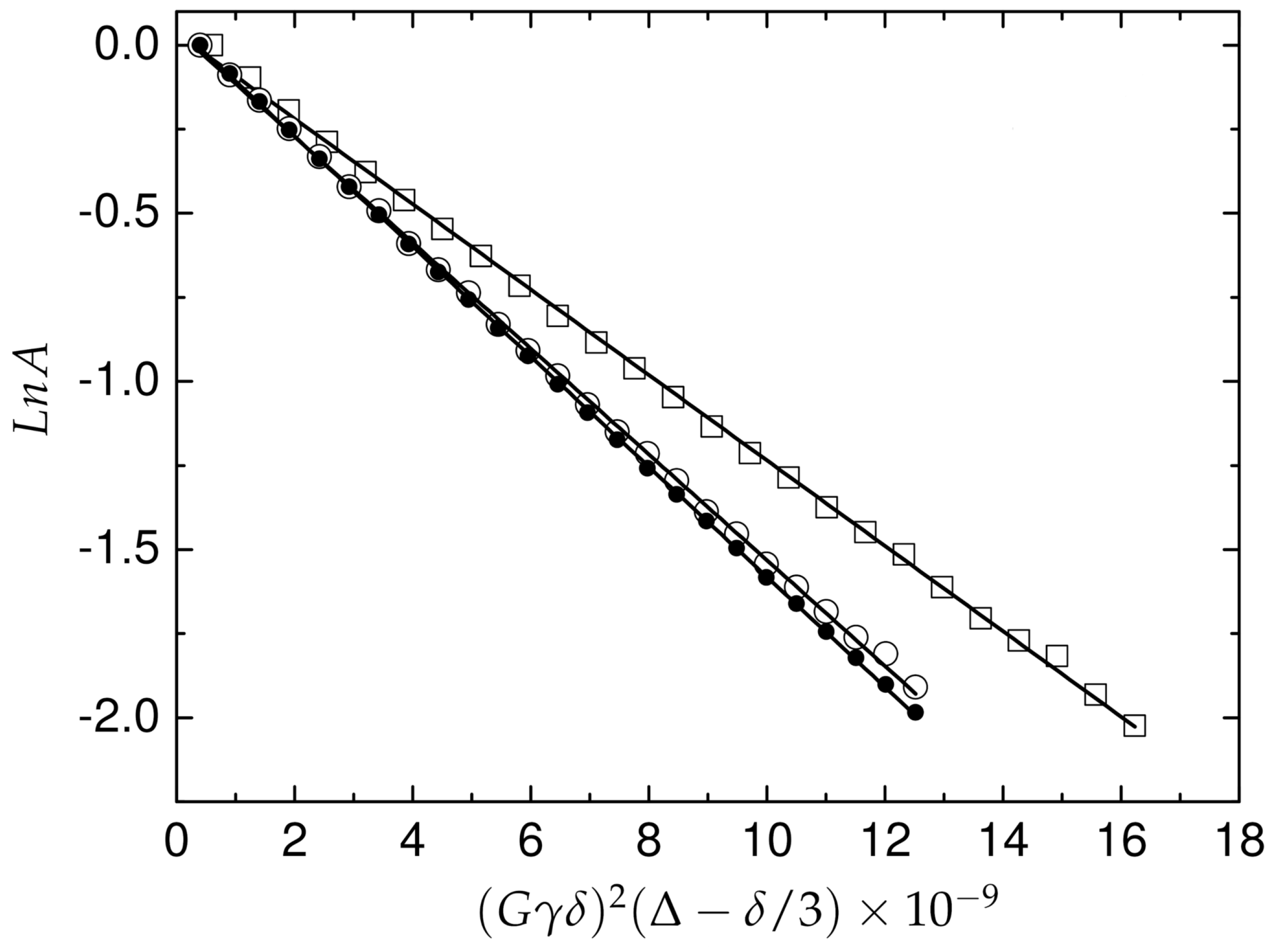
2.4. Translational Mobility of A and Pd(A) in Solution
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Sample Preparation
4.3. Mass Spectrometry
4.4. Circular Dichroism
4.5. Spectrofluorimetry
4.6. NMR Spectroscopy
4.7. Assignment of the H, C and N Resonances and 3D Structure Evaluation of A Peptide and Pd(A) Complex in Solution
4.8. C Relaxation Measurements
4.9. Diffusion Measurements
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CD | Circular dichroism |
| ESI-MS | ElectroSpray Ionisation Mass Spectrometry |
| IMS-MS | Ion-Mobility Spectrometry Mass Spectrometry |
| DSS | sodium 2,2-dimethyl-2-silapentane-5-sulfonate |
| PGSE | Pulsed Gradient Spin Echo |
| NOESY | Nuclear Overhauser SpectroscopY |
| HSQC | Heteronuclear Single Quantum Correlation spectroscopy |
| TOCSY | TOtal Correlation SpectroscopY |
| ROS | Reactive Oxygen Species |
| AD | Alzheimer’s Disease |
References
- Masters, C.L.; Simms, G.; Weinman, N.A.; Multhaup, G.; McDonald, B.L.; Beyreuther, K. Amyloid plaque core protein in Alzheimer disease and Down syndrome. Proc. Natl. Acad. Sci. USA 1985, 82, 4245–4249. [Google Scholar] [CrossRef]
- Masters, C.L.; Multhaup, G.; Simms, G.; Pottgiesser, J.; Martins, R.; Beyreuther, K. Neuronal origin of a cerebral amyloid: Neurofibrillary tangles of Alzheimer’s disease contain the same protein as the amyloid of plaque cores and blood vessels. EMBO J. 1985, 4, 2757–2763. [Google Scholar] [CrossRef]
- Lewis, H.; Beher, D.; Cookson, N.; Oakley, A.; Piggott, M.; Morris, C.; Jaros, E.; Perry, R.; Ince, P.; Kenny, R.; et al. Quantification of Alzheimer pathology in ageing and dementia: Age-related accumulation of amyloid-β (42) peptide in vascular dementia. Neuropathol. Appl. Neurobiol. 2006, 32, 103–118. [Google Scholar] [CrossRef]
- Portelius, E.; Bogdanovic, N.; Gustavsson, M.K.; Volkmann, I.; Brinkmalm, G.; Zetterberg, H.; Winblad, B.; Blennow, K. Mass spectrometric characterization of brain amyloid beta isoform signatures in familial and sporadic Alzheimer’s disease. Acta Neuropathol. 2010, 120, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Bouter, Y.; Dietrich, K.; Wittnam, J.L.; Rezaei-Ghaleh, N.; Pillot, T.; Papot-Couturier, S.; Lefebvre, T.; Sprenger, F.; Wirths, O.; Zweckstetter, M.; et al. N-truncated amyloid β (Aβ) 4-42 forms stable aggregates and induces acute and long-lasting behavioral deficits. Acta Neuropathol. 2013, 126, 189–205. [Google Scholar] [CrossRef] [PubMed]
- Wirths, O.; Walter, S.; Kraus, I.; Klafki, H.W.; Stazi, M.; Oberstein, T.J.; Ghiso, J.; Wiltfang, J.; Bayer, T.A.; Weggen, S. N-truncated Aβ 4–x peptides in sporadic Alzheimer’s disease cases and transgenic Alzheimer mouse models. Alzheimer’s Res. Ther. 2017, 9, 80. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, E.; Mathews, P.; Mezhericher, E.; Beach, T.G.; Deng, J.; Neubert, T.A.; Rostagno, A.; Ghiso, J. Aβ truncated species: Implications for brain clearance mechanisms and amyloid plaque deposition. Biochim. Biophys. Acta BBA Mol. Basis Dis. 2018, 1864, 208–225. [Google Scholar] [CrossRef] [PubMed]
- Wirths, O.; Zampar, S.; Weggen, S. N-Terminally Truncated Aβ Peptide Variants in Alzheimer’s Disease. Exon Publ. 2019, 107–122. [Google Scholar] [CrossRef]
- Zampar, S.; Klafki, H.W.; Sritharen, K.; Bayer, T.A.; Wiltfang, J.; Rostagno, A.; Ghiso, J.; Miles, L.A.; Wirths, O. N-terminal heterogeneity of parenchymal and vascular amyloid-β deposits in Alzheimer‘s disease. Neuropathol. Appl. Neurobiol. 2020, 46, 673–685. [Google Scholar] [CrossRef]
- Wirths, O.; Zampar, S. Emerging roles of N-and C-terminally truncated Aβ species in Alzheimer’s disease. Expert Opin. Ther. Targets 2019, 23, 991–1004. [Google Scholar] [CrossRef]
- Alies, B.; Renaglia, E.; Rózga, M.; Bal, W.; Faller, P.; Hureau, C. Cu(II) affinity for the Alzheimer’s peptide: Tyrosine fluorescence studies revisited. Anal. Chem. 2013, 85, 1501–1508. [Google Scholar] [CrossRef] [PubMed]
- Drew, S.C.; Barnham, K.J. The heterogeneous nature of Cu2+ interactions with Alzheimer’s amyloid-β peptide. Accounts Chem. Res. 2011, 44, 1146–1155. [Google Scholar] [CrossRef] [PubMed]
- Arrigoni, F.; Prosdocimi, T.; Mollica, L.; De Gioia, L.; Zampella, G.; Bertini, L. Copper reduction and dioxygen activation in Cu–amyloid beta peptide complexes: Insight from molecular modelling. Metallomics 2018, 10, 1618–1630. [Google Scholar] [CrossRef] [PubMed]
- Atrián-Blasco, E.; del Barrio, M.; Faller, P.; Hureau, C. Ascorbate oxidation by Cu (amyloid-β) complexes: Determination of the intrinsic rate as a function of alterations in the peptide sequence revealing key residues for reactive oxygen species production. Anal. Chem. 2018, 90, 5909–5915. [Google Scholar] [CrossRef]
- Huang, H.; Lou, X.; Hu, B.; Zhou, Z.; Chen, J.; Tian, Y. A comprehensive study on the generation of reactive oxygen species in Cu-Aβ-catalyzed redox processes. Free Radic. Biol. Med. 2019, 135, 125–131. [Google Scholar] [CrossRef]
- Mital, M.; Wezynfeld, N.E.; Frączyk, T.; Wiloch, M.Z.; Wawrzyniak, U.E.; Bonna, A.; Tumpach, C.; Barnham, K.J.; Haigh, C.L.; Bal, W.; et al. A functional role for Aβ in metal homeostasis? N-truncation and high-affinity copper binding. Angew. Chem. 2015, 127, 10606–10610. [Google Scholar] [CrossRef]
- Esmieu, C.; Ferrand, G.; Borghesani, V.; Hureau, C. N-truncated Aβ peptides impact on Cu and Cu (Aβ)-generated ROS: Cu(I) matters! Chem. Eur. J. 2020, 26. [Google Scholar] [CrossRef]
- Bossak-Ahmad, K.; Mital, M.; Płonka, D.; Drew, S.C.; Bal, W. Oligopeptides generated by neprilysin degradation of β-amyloid have the highest Cu(II) affinity in the whole Aβ family. Inorg. Chem. 2018, 58, 932–943. [Google Scholar] [CrossRef]
- Wezynfeld, N.E.; Stefaniak, E.; Stachucy, K.; Drozd, A.; Płonka, D.; Drew, S.C.; Krężel, A.; Bal, W. Resistance of Cu (Aβ4–16) to Copper Capture by Metallothionein-3 Supports a Function for the Aβ4–42 Peptide as a Synaptic CuII Scavenger. Angew. Chem. Int. Ed. 2016, 55, 8235–8238. [Google Scholar] [CrossRef]
- Santoro, A.; Wezynfeld, N.E.; Vašák, M.; Bal, W.; Faller, P. Cysteine and glutathione trigger the Cu–Zn swap between Cu(II)-amyloid-β 4-16 peptide and Zn 7-metallothionein-3. Chem. Commun. 2017, 53, 11634–11637. [Google Scholar] [CrossRef]
- Stefaniak, E.; Bal, W. CuII Binding Properties of N-Truncated Aβ Peptides: In Search of Biological Function. Inorg. Chem. 2019, 58, 13561–13577. [Google Scholar] [CrossRef] [PubMed]
- Stefaniak, E.; Płonka, D.; Szczerba, P.; Wezynfeld, N.E.; Bal, W. Copper Transporters? Glutathione Reactivity of Products of Cu–Aβ Digestion by Neprilysin. Inorg. Chem. 2020, 59, 4186–4190. [Google Scholar] [CrossRef] [PubMed]
- Atrián-Blasco, E.; Gonzalez, P.; Santoro, A.; Alies, B.; Faller, P.; Hureau, C. Cu and Zn coordination to amyloid peptides: From fascinating chemistry to debated pathological relevance. Coord. Chem. Rev. 2018, 371, 38–55. [Google Scholar] [CrossRef] [PubMed]
- Sigel, H.; Martin, R.B. Coordinating properties of the amide bond. Stability and structure of metal ion complexes of peptides and related ligands. Chem. Rev. 1982, 82, 385–426. [Google Scholar] [CrossRef]
- Harford, C.; Sarkar, B. Amino terminal Cu (II)-and Ni (II)-binding (ATCUN) motif of proteins and peptides: Metal binding, DNA cleavage, and other properties. Accounts Chem. Res. 1997, 30, 123–130. [Google Scholar] [CrossRef]
- Gonzalez, P.; Bossak, K.; Stefaniak, E.; Hureau, C.; Raibauta, L.; Bal, W.; Faller, P. N-terminal Cu binding motifs Xxx-Zzz-His (ATCUN) and Xxx-His and their derivatives: Chemistry, biology and medicinal applications. Chem. Eur. J. 2018, 24, 8029–8041. [Google Scholar] [CrossRef]
- Hureau, C.; Eury, H.; Guillot, R.; Bijani, C.; Sayen, S.; Solari, P.L.; Guillon, E.; Faller, P.; Dorlet, P. X-ray and Solution Structures of CuIIGHK and CuIIDAHK Complexes: Influence on Their Redox Properties. Chem. Eur. J. 2011, 17, 10151–10160. [Google Scholar] [CrossRef]
- Camerman, N.; Camerman, A.; Sarkar, B. Molecular design to mimic the copper (II) transport site of human albumin. The crystal and molecular structure of copper (II)–glycylglycyl-L-histidine-N-methyl amide monoaquo complex. Can. J. Chem. 1976, 54, 1309–1316. [Google Scholar] [CrossRef]
- Donaldson, L.W.; Skrynnikov, N.R.; Choy, W.Y.; Muhandiram, D.R.; Sarkar, B.; Forman-Kay, J.D.; Kay, L.E. Structural characterization of proteins with an attached ATCUN motif by paramagnetic relaxation enhancement NMR spectroscopy. J. Am. Chem. Soc. 2001, 123, 9843–9847. [Google Scholar] [CrossRef]
- Nair, N.G.; Perry, G.; Smith, M.A.; Reddy, V.P. NMR studies of zinc, copper, and iron binding to histidine, the principal metal ion complexing site of amyloid-β peptide. J. Alzheimer’s Dis. 2010, 20, 57–66. [Google Scholar] [CrossRef]
- Bal, W.; Djuran, M.I.; Margerum, D.W.; Gray, E.T.; Mazid, M.A.; Tom, R.T.; Nieboer, E.; Sadler, P.J. Dioxygen-induced decarboxylation and hydroxylation of [NiII (glycyl-glycyl-L-histidine)] occurs via Ni III: X-ray crystal structure of [NiII (glycyl-glycyl-α-hydroxy-D,L-histamine)]·3H2O. J. Chem. Soc. Chem. Commun. 1994, 16, 1889–1890. [Google Scholar] [CrossRef]
- Bal, W.; Chmurny, G.N.; Hilton, B.D.; Sadler, P.J.; Tucker, A. Axial hydrophobic fence in highly-stable Ni(II) complex of des-angiotensinogen N-terminal peptide. J. Am. Chem. Soc. 1996, 118, 4727–4728. [Google Scholar] [CrossRef]
- Best, S.L.; Chattopadhyay, T.K.; Djuran, M.I.; Palmer, R.A.; Sadler, P.J.; Sóvágó, I.; Varnagy, K. Gold(III) and Palladium(II) Complexes of Glycylglycyl-L-Histidine: Crystal Structures of [AuIII (Gly-Gly-L-His-H−2)] Cl·H2O and [PdII (Gly-Gly-L-His-H−2)]·1.5H2O and HisεNH deprotonation. J. Chem. Soc. Dalton Trans. 1997, 2587–2596. [Google Scholar] [CrossRef]
- Klein, A.; Tsiveriotis, P.; Malandrinos, G.; Hadjiliadis, N. Platinum (II) and palladium (II) complexes with histidine and histidyl containing peptides: Structure and reactivity. Rev. Inorg. Chem. 2000, 20, 305–338. [Google Scholar] [CrossRef]
- Frias, E.C.; Pitsch, H.; Ly, J.; Poitrenaud, C. Palladium complexes in concentrated nitrate and acid solutions. Talanta 1995, 42, 1675–1683. [Google Scholar] [CrossRef]
- Van Middlesworth, J.M.; Wood, S.A. The stability of palladium (II) hydroxide and hydroxy–chloride complexes: An experimental solubility study at 25–85 C and 1 bar. Geochim. Cosmochim. Acta 1999, 63, 1751–1765. [Google Scholar] [CrossRef]
- Milić, N.B.; Bugarčić, Ž.D. Hydrolysis of the palladium (II) ion in a sodium chloride medium. Transit. Met. Chem. 1984, 9, 173–176. [Google Scholar] [CrossRef]
- Torapava, N.; Elding, L.I.; Mändar, H.; Roosalu, K.; Persson, I. Structures of polynuclear complexes of palladium (II) and platinum (II) formed by slow hydrolysis in acidic aqueous solution. Dalton Trans. 2013, 42, 7755–7760. [Google Scholar] [CrossRef]
- Boily, J.F.; Seward, T.M.; Charnock, J.M. The hydrolysis and precipitation of Pd (II) in 0.6 mol kg- 1 NaCl: A potentiometric, spectrophotometric, and EXAFS study. Geochim. Cosmochim. Acta 2007, 71, 4834–4845. [Google Scholar] [CrossRef]
- Krieger, E.; Koraimann, G.; Vriend, G. Increasing the precision of comparative models with YASARA NOVA–a self-parameterizing force field. Proteins Struct. Funct. Bioinform. 2002, 47, 393–402. [Google Scholar] [CrossRef]
- Shen, Y.; Bax, A. Protein structural information derived from NMR chemical shift with the neural network program TALOS-N. In Artificial Neural Networks; Springer: Berlin/Heidelberg, Germany, 2015; pp. 17–32. [Google Scholar]
- Stejskal, E.O.; Tanner, J.E. Spin diffusion measurements: Spin echoes in the presence of a time-dependent field gradient. J. Chem. Phys. 1965, 42, 288–292. [Google Scholar] [CrossRef]
- Macchioni, A.; Ciancaleoni, G.; Zuccaccia, C.; Zuccaccia, D. Determining accurate molecular sizes in solution through NMR diffusion spectroscopy. Chem. Soc. Rev. 2008, 37, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Taube, M.; Pietralik, Z.; Szymanska, A.; Szutkowski, K.; Clemens, D.; Grubb, A.; Kozak, M. The domain swapping of human cystatin C induced by synchrotron radiation. Sci. Rep. 2019, 9, 8548. [Google Scholar] [CrossRef] [PubMed]
- Bal, W.; Wójcik, J.; Maciejczyk, M.; Grochowski, P.; Kasprzak, K.S. Induction of a secondary structure in the N-terminal pentadecapeptide of human protamine HP2 through Ni(II) coordination. An NMR study. Chem. Res. Toxicol. 2000, 13, 823–830. [Google Scholar] [CrossRef]
- Kozłowski, H. Spectroscopic and magnetic resonance studies on Ni(II), Cu(II) and Pd(II) complexes with Gly-Leu-Tyr and Tyr-Gly-Gly tripeptides. Inorganica Chim. Acta 1978, 31, 135–140. [Google Scholar] [CrossRef]
- Kozłowski, H.; Jeżowska, M.; Szyszuk, H. PMR conformational studies of Pd(II) complexes with Ala-Tyr and d-Leu-Tyr depeptides. J. Mol. Struct. 1978, 50, 73–80. [Google Scholar] [CrossRef]
- Yamauchi, O. Noncovalent interactions in biocomplexes. Phys. Sci. Rev. 2016, 1. [Google Scholar] [CrossRef]
- Stefaniak, E.; Atrian-Blasco, E.; Goch, W.; Sabater, L.; Hureau, C.; Bal, W. The aggregation pattern of Aβ1-40 is altered by the presence of N-truncated Aβ4-40 and/or Cu(II) ions in a similar way via ionic interactions. Chem. Eur. J. 2020, in press. [Google Scholar] [CrossRef]
- Wishart, D.S.; Bigam, C.G.; Yao, J.; Abildgaard, F.; Dyson, H.J.; Oldfield, E.; Markley, J.L.; Sykes, B.D. 1H, 13C and 15N chemical shift referencing in biomolecular NMR. J. Biomol. NMR 1995, 6, 135–140. [Google Scholar] [CrossRef]
- Delaglio, F.; Grzesiek, S.; Vuister, G.W.; Zhu, G.; Pfeifer, J.; Bax, A. NMRPipe: A multidimensional spectral processing system based on UNIX pipes. J. Biomol. NMR 1995, 6, 277–293. [Google Scholar] [CrossRef]
- Lee, W.; Tonelli, M.; Markley, J.L. NMRFAM-SPARKY: Enhanced software for biomolecular NMR spectroscopy. Bioinformatics 2015, 31, 1325–1327. [Google Scholar] [CrossRef] [PubMed]
- Schwieters, C.D.; Kuszewski, J.J.; Clore, G.M. Using Xplor–NIH for NMR molecular structure determination. Prog. Nucl. Magn. Reson. Spectrosc. 2006, 48, 47–62. [Google Scholar] [CrossRef]
- Koradi, R.; Billeter, M.; Wüthrich, K. MOLMOL: A program for display and analysis of macromolecular structures. J. Mol. Graph. 1996, 14, 51–55. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera–a visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef]
- Wu, D.; Chen, A.; Johnson, C.S. An improved diffusion-ordered spectroscopy experiment incorporating bipolar-gradient pulses. J. Magn. Reson. Ser. A 1995, 115, 260–264. [Google Scholar] [CrossRef]
- Hwang, T.L.; Shaka, A. Water suppression that works. Excitation sculpting using arbitrary wave-forms and pulsed-field gradients. J. Magn. Reson. Ser. A 1995, 112, 275–279. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mital, M.; Szutkowski, K.; Bossak-Ahmad, K.; Skrobecki, P.; Drew, S.C.; Poznański, J.; Zhukov, I.; Frączyk, T.; Bal, W. The Palladium(II) Complex of Aβ4−16 as Suitable Model for Structural Studies of Biorelevant Copper(II) Complexes of N-Truncated Beta-Amyloids. Int. J. Mol. Sci. 2020, 21, 9200. https://doi.org/10.3390/ijms21239200
Mital M, Szutkowski K, Bossak-Ahmad K, Skrobecki P, Drew SC, Poznański J, Zhukov I, Frączyk T, Bal W. The Palladium(II) Complex of Aβ4−16 as Suitable Model for Structural Studies of Biorelevant Copper(II) Complexes of N-Truncated Beta-Amyloids. International Journal of Molecular Sciences. 2020; 21(23):9200. https://doi.org/10.3390/ijms21239200
Chicago/Turabian StyleMital, Mariusz, Kosma Szutkowski, Karolina Bossak-Ahmad, Piotr Skrobecki, Simon C. Drew, Jarosław Poznański, Igor Zhukov, Tomasz Frączyk, and Wojciech Bal. 2020. "The Palladium(II) Complex of Aβ4−16 as Suitable Model for Structural Studies of Biorelevant Copper(II) Complexes of N-Truncated Beta-Amyloids" International Journal of Molecular Sciences 21, no. 23: 9200. https://doi.org/10.3390/ijms21239200
APA StyleMital, M., Szutkowski, K., Bossak-Ahmad, K., Skrobecki, P., Drew, S. C., Poznański, J., Zhukov, I., Frączyk, T., & Bal, W. (2020). The Palladium(II) Complex of Aβ4−16 as Suitable Model for Structural Studies of Biorelevant Copper(II) Complexes of N-Truncated Beta-Amyloids. International Journal of Molecular Sciences, 21(23), 9200. https://doi.org/10.3390/ijms21239200







