The Central Role of Cadherins in Gonad Development, Reproduction, and Fertility
Abstract
:1. Introduction
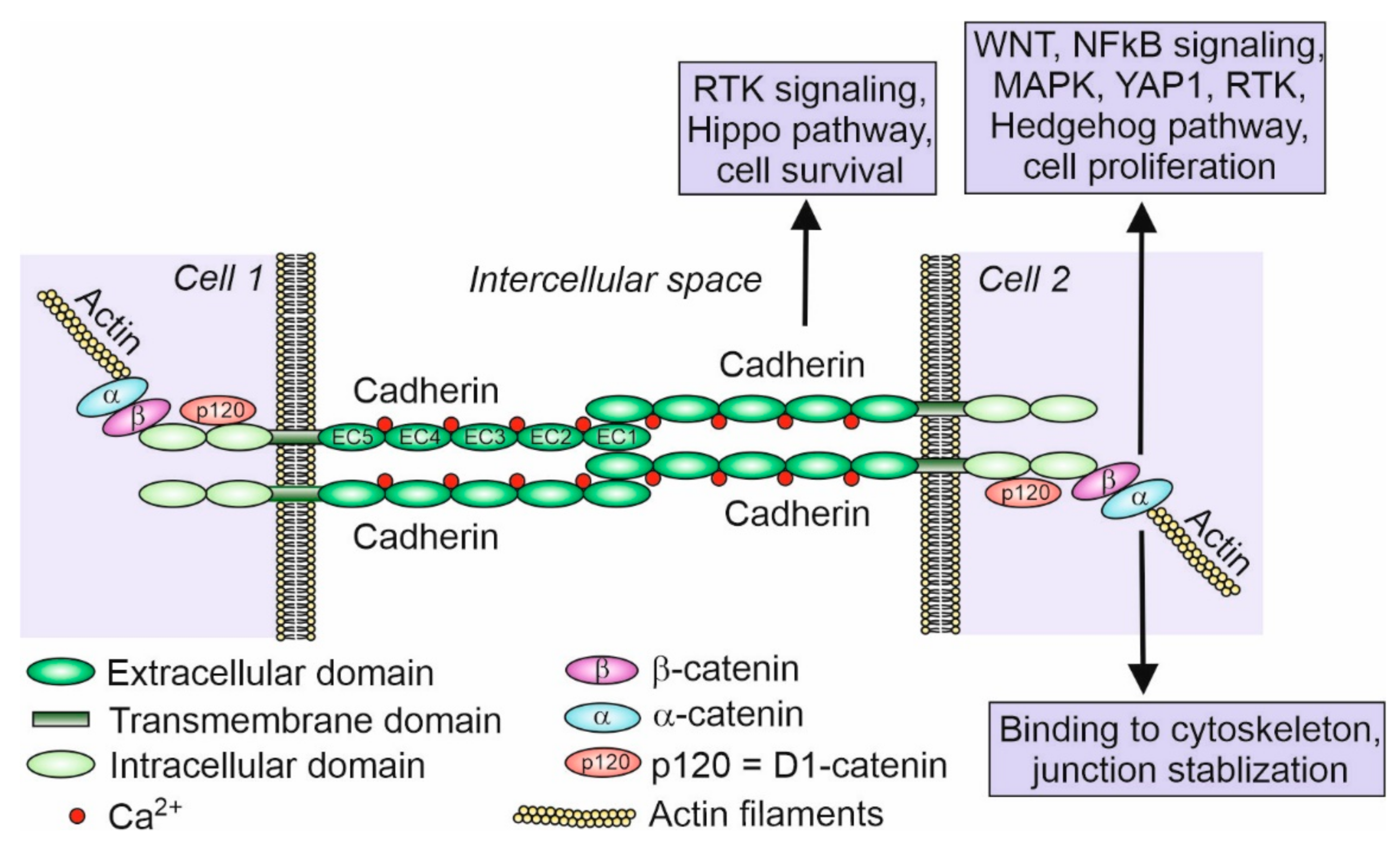
2. Cadherins—History and Function
3. Members of the Cadherin Superfamily
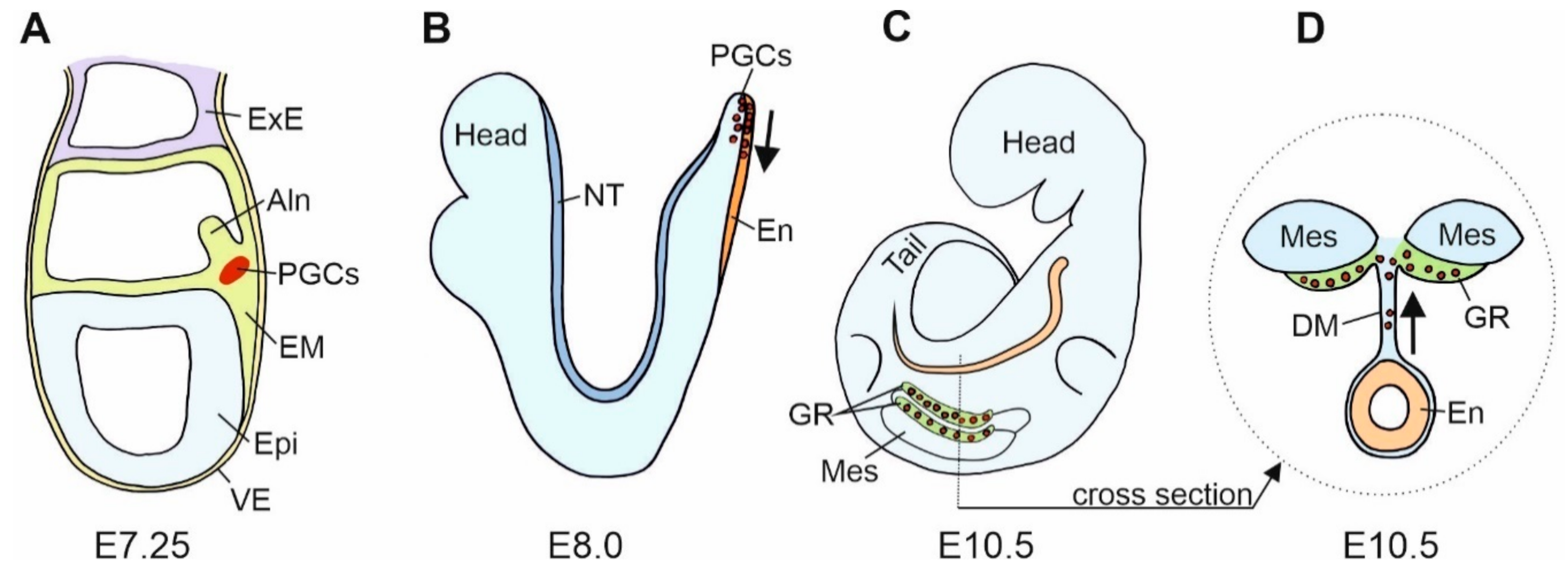
4. Cadherins and the Primordial Germ Cells (PGCs)
5. Cadherins and PGCs Migration
6. Cadherins in Gonad Development
7. Cadherins in Adult Testis
8. Cadherins in Adult Ovary
9. Cadherins in Gametes and Fertilization
10. Cadherins in Drosophila Gonad Development
11. Conclusions
- Which molecular mechanisms hide behind the requirement of cadherins, and other cell adhesion molecules (CAMs), for germ cells survival, and how do the cadherins prevent apoptosis of these cells?
- Is the properly executed adhesion between germ cells and somatic cells sufficient to prevent their apoptosis, or are the more complex cadherin-related signaling pathways involved?
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Holtfreter, J. Gewebeaffinitüt, ein Mittel der embryonalen Formbildung. Arch. Exp. Zellforsch. Besonders Gewebezücht 1939, 23, 169–209. [Google Scholar]
- Takeichi, M. Functional correlation between cell adhesive properties and some cell surface proteins. J. Cell Biol. 1977, 75, 464–474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoshida-Noro, C.; Suzuki, N.; Takeichi, M. Molecular nature of the calciumdependent cell-cell adhesion system in mouse teratocarcinoma and embryonic cells studied with a monoclonal antibody. Dev. Biol. 1984, 101, 19–27. [Google Scholar] [CrossRef]
- Hatta, K.; Okada, T.S.; Takeichi, M. A monoclonal antibody disrupting calcium-dependent cell-cell adhesion of brain tissues: Possible role of its target antigen in animal pattern formation. Proc. Natl. Acad. Sci. USA 1985, 82, 2789–2793. [Google Scholar] [CrossRef] [Green Version]
- Nose, A.; Takeichi, M. A novel cadherin cell adhesion molecule: Its expression patterns associated with implantation and organogenesis of mouse embryos. J. Cell Biol. 1986, 103, 2649–2658. [Google Scholar] [CrossRef] [Green Version]
- Inuzuka, H.; Miyatani, S.; Takeichi, M. R-cadherin: A novel Ca(2+)-dependent cell-cell adhesion molecule expressed in the retina. Neuron 1991, 7, 69–79. [Google Scholar] [CrossRef]
- Lampugnani, M.G.; Resnati, M.; Raiteri, M.; Pigott, R.; Pisacane, A.; Houen, G.; Ruco, L.P.; Dejana, E. A novel endothelial-specific membrane-protein is a marker of cell-cell contacts. J. Cell Biol. 1992, 118, 1511–1522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gul, I.S.; Hulpiau, P.; Saeys, Y.; van Roy, F. Evolution and diversity of cadherins and catenins. Exp. Cell Res. 2017, 358, 3–9. [Google Scholar] [CrossRef]
- Hulpiau, P.; van Roy, F. Molecular evolution of the cadherin superfamily. Int. J. Biochem. Cell Biol. 2009, 41, 349–369. [Google Scholar] [CrossRef]
- Lutz, K.L.; Siahaan, T.J. Modulation of the cellular junction protein E-cadherin in bovine brain microvessel endothelial cells by cadherin peptides. J. Drug Deliv. 1997, 4, 187–193. [Google Scholar] [CrossRef]
- Burden-Gulley, S.M.; Gates, T.J.; Craig, S.E.; Lou, S.F.; Oblander, S.A.; Howell, S.; Gupta, M.; Brady-Kalnay, S.M. Novel peptide mimetic small molecules of the HAV motif in N-cadherin inhibit N-cadherin-mediated neurite outgrowth and cell adhesion. Peptides 2009, 30, 2380–2387. [Google Scholar] [CrossRef]
- Williams, E.; Williams, G.; Gour, B.J.; Blaschuk, O.W.; Doherty, P. A novel family of cyclic peptide antagonists suggests that N-cadherin specificity is determined by amino acids that flank the HAV motif. J. Biol. Chem. 2000, 275, 4007–4012. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, Y.; Chang, R.; Xu, F.; Gao, Y.; Yang, F.; Wang, C.; Xiao, J.; Su, Z.; Bi, Y.; Wang, L.; et al. N-glycosylation at Asn 402 stabilizes N-cadherin and promotes cell-cell adhesion of glioma cells. J. Cell Biochem. 2017, 118, 1423–1431. [Google Scholar] [CrossRef]
- Langer, M.D.; Guo, H.; Shashikanth, N.; Pierce, J.M.; Leckband, D.E. N-glycosylation alters cadherin-mediated intercellular binding kinetics. J. Cell Sci. 2012, 125, 2478–24785. [Google Scholar] [CrossRef] [Green Version]
- Biswas, B.; Batista, F.; Sundaram, S.; Stanley, P. MGAT1 and complex N-glycans regulate ERK signaling during spermatogenesis. Sci. Rep. 2018, 8, 2022. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McEwen, A.E.; Maher, M.T.; Mo, R.; Gottardi, C.J. E-cadherin phosphorylation occurs during its biosynthesis to promote its cell surface stability and adhesion. Mol. Biol. Cell. 2014, 25, 2365–2374. [Google Scholar] [CrossRef] [PubMed]
- Stappert, J.; Kemler, R. A short core region of E-cadherin is essential for catenin binding and is highly phosphorylated. Cell Adhes. Commun. 1994, 2, 319–327. [Google Scholar] [CrossRef]
- Chen, C.; Wang, S.; Chan, P.; Shen, M.; Chen, H. Phosphorylation of E-cadherin at threonine 790 by protein kinase Cδ reduces β-catenin binding and suppresses the function of E-cadherin. Oncotarget 2016, 7, 37260–37276. [Google Scholar] [CrossRef] [Green Version]
- Sato, K.; Watanabe, T.; Wang, S.; Kakeno, M.; Matsuzawa, K.; Matsui, T.; Yokoi, K.; Murase, K.; Sugiyama, I.; Ozawa, M.; et al. Numb controls E-cadherin endocytosis through p120 catenin with aPKC. Mol. Biol. Cell. 2011, 22, 3103–3119. [Google Scholar] [CrossRef]
- Chen, Y.T.; Tai, C.Y. μ2-Dependent endocytosis of N-cadherin is regulated by β-catenin to facilitate neurite outgrowth. Traffic 2017, 18, 287–303. [Google Scholar] [CrossRef] [Green Version]
- Halbleib, J.M.; Nelson, W.J. Cadherins in development: Cell adhesion, sorting, and tissue morphogenesis. Genes Dev. 2006, 20, 3199–3214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maître, J.L.; Heisenberg, C.P. Three functions of cadherins in cell adhesion. Curr. Biol. 2013, 23, R626–R633. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, W.; Yang, L.; Li, T.; Zhang, Y. Cadherin Signaling in Cancer: Its Functions and Role as a Therapeutic Target. Front Oncol. 2019, 9, 989. [Google Scholar] [CrossRef]
- St Croix, B.; Sheehan, C.; Rak, J.W.; Flørenes, V.A.; Slingerland, J.M.; Kerbel, R.S. E-Cadherin-dependent growth suppression is mediated by the cyclin-dependent kinase inhibitor p27(KIP1). J. Cell Biol. 1998, 142, 557–571. [Google Scholar] [CrossRef]
- Klezovitch, O.; Vasioukhin, V. Cadherin signaling: Keeping cells in touch. F1000Research 2015, 4, 550. [Google Scholar] [CrossRef]
- Loh, C.Y.; Chai, J.Y.; Tang, T.F.; Wong, W.F.; Sethi, G.; Shanmugam, M.K.; Chong, P.P.; Looi, C.Y. The E-Cadherin and N-Cadherin Switch in Epithelial-to-Mesenchymal Transition: Signaling, Therapeutic Implications, and Challenges. Cells 2019, 8, 1118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lien, W.H.; Klezovitch, O.; Fernandez, T.E.; Delrow, J.; Vasioukhin, V. alphaE-catenin controls cerebral cortical size by regulating the hedgehog signaling pathway. Science 2006, 311, 1609–1612. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cole, B.K.; Curto, M.; Chan, A.W.; McClatchey, A.I. Localization to the cortical cytoskeleton is necessary for Nf2/merlin-dependent epidermal growth factor receptor silencing. Mol. Cell Biol. 2008, 28, 1274–1284. [Google Scholar] [CrossRef] [Green Version]
- Kim, N.G.; Koh, E.; Chen, X.; Gumbiner, B.M. E-cadherin mediates contact inhibition of proliferation through Hippo signaling-pathway components. Proc. Natl. Acad. Sci. USA 2011, 108, 11930–11935. [Google Scholar] [CrossRef] [Green Version]
- Silvis, M.R.; Kreger, B.T.; Lien, W.H.; Klezovitch, O.; Rudakova, G.M.; Camargo, F.D.; Lantz, D.M.; Seykora, J.T.; Vasioukhin, V. α-catenin is a tumor suppressor that controls cell accumulation by regulating the localization and activity of the transcriptional coactivator Yap1. Sci. Signal. 2011, 4, ra33. [Google Scholar] [CrossRef] [Green Version]
- Stepniak, E.; Radice, G.L.; Vasioukhin, V. Adhesive and signaling functions of cadherins and catenins in vertebrate development. Cold Spring Harb. Perspect. Biol. 2009, 1, a002949. [Google Scholar] [CrossRef] [Green Version]
- Piprek, R.P. Molecular mechanisms underlying female sex determination—Antagonism between female and male pathway. Folia Biol. 2009, 57, 105–113. [Google Scholar] [CrossRef]
- Piprek, R.P.; Kolasa, M.; Podkowa, D.; Kloc, M.; Kubiak, J.Z. Cell adhesion molecules expression pattern indicates that somatic cells arbitrate gonadal sex of differentiating bipotential fetal mouse gonad. Mech. Dev. 2017, 147, 17–27. [Google Scholar] [CrossRef]
- Johnson, A.D.; Alberio, R. Primordial germ cells: The first cell lineage or the last cells standing? Development 2015, 142, 2730–2739. [Google Scholar] [CrossRef] [Green Version]
- Martinho, R.G.; Kunwar, P.S.; Casanova, J.; Lehmann, R. A noncoding RNA is required for the repression of RNApolII-dependent transcription in primordial germ cells. Curr. Biol. 2004, 14, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Ginsburg, M.; Snow, M.H.L.; McLaren, A. Primordial germ cells in the mouse embryo during gastrulation. Development 1990, 110, 521–528. [Google Scholar]
- Okamura, D.; Kimura, T.; Nakano, T.; Matsui, Y. Cadherin-mediated cell interaction regulates germ cell determination in mice. Development 2003, 130, 6423–6430. [Google Scholar] [CrossRef] [Green Version]
- Piprek, R.P.; Kloc, M.; Kubiak, J.Z. Early development of the gonads: Origin and differentiation of the somatic cells of the genital ridges. Results Probl. Cell Differ. 2016, 58, 1–22. [Google Scholar]
- Weidinger, G.; Wolke, U.; Köprunner, M.; Klinger, M.; Raz, E. Identification of tissues and patterning events required for distinct steps in early migration of zebrafish primordial germ cells. Development 1999, 126, 5295–5307. [Google Scholar]
- Fox, M.; Damjanov, I.; Martinez-Hernandez, A.; Knowles, B.B.; Solter, D. Immunohistochemical localization of the early embryonic antigen (SSEA-1) in postimplantation mouse embryos and fetal and adult tissues. Dev. Biol. 1981, 83, 391–398. [Google Scholar] [CrossRef]
- Di Carlo, A.; De Felici, M. A role for E-cadherin in mouse primordial germ cell development. Dev. Biol. 2000, 226, 209–219. [Google Scholar] [CrossRef] [Green Version]
- Bendel-Stenzel, M.R.; Gomperts, M.; Anderson, R.; Heasman, J.; Wylie, C. The role of cadherins during primordial germ cell migration and early gonad formation in the mouse. Mech. Dev. 2000, 91, 143–152. [Google Scholar] [CrossRef]
- Baronsky, T.; Dzementsei, A.; Oelkers, M.; Melchert, J.; Pieler, T.; Janshoff, A. Reduction in E-cadherin expression fosters migration of Xenopus laevis primordial germ cells. Integr. Biol. 2016, 3, 8. [Google Scholar] [CrossRef]
- Weidinger, G.; Stebler, J.; Slanchev, K.; Dumstrei, K.; Wise, C.; Lovell-Badge, R.; Thisse, C.; Thisse, B.; Raz, E. Dead end, a novel vertebrate germ plasm component, is required for zebrafish primordial germ cell migration and survival. Curr. Biol. 2003, 13, 1429–1434. [Google Scholar] [CrossRef] [Green Version]
- Dzementsei, A.; Schneider, D.; Janshoff, A.; Pieler, T. Migratory and adhesive properties of Xenopus laevis primordial germ cells in vitro. Biol. Open. 2013, 2, 1279–1287. [Google Scholar] [CrossRef] [Green Version]
- Kunwar, P.S.; Sano, H.; Renault, A.D.; Barbosa, V.; Fuse, N.; Lehmann, R. Tre1 GPCR initiates germ cell transepithelial migration by regulating Drosophila melanogaster E-cadherin. J. Cell Biol. 2008, 183, 157–168. [Google Scholar] [CrossRef] [Green Version]
- Kardash, E.; Reichman-Fried, M.; Maître, J.; Boldajipour, B.; Papusheva, E.; Messerschmidt, E.M.; Heisenberg, C.P.; Raz, E. A role for Rho GTPases and cell–cell adhesion in single-cell motility in vivo. Nat. Cell Biol. 2010, 12, 47–53. [Google Scholar] [CrossRef]
- Blaser, H.; Eisenbeiss, S.; Neumann, M.; Reichman-Fried, M.; Thisse, B.; Thisse, C.; Raz, E. Transition from non-motile behaviour to directed migration during early PGC development in zebrafish. J. Cell Sci. 2005, 118, 4027–4038. [Google Scholar] [CrossRef] [Green Version]
- Hartwig, J.; Tarbashevich, K.; Seggewiß, J.; Stehling, M.; Bandemer, J.; Grimaldi, C.; Paksa, A.; Groß-Thebing, T.; Meyen, D.; Raz, E. Temporal control over the initiation of cell motility by a regulator of G-protein signaling. Proc. Natl. Acad. Sci. USA 2014, 111, 11389–11394. [Google Scholar] [CrossRef] [Green Version]
- Yu, M.; Guan, K.; Zhang, C. The promoting effect of retinoic acid on proliferation of chicken primordial germ cells by increased expression of cadherin and catenins. Amino Acids 2011, 40, 933–941. [Google Scholar] [CrossRef]
- Culty, M. Gonocytes, the forgotten cells of the germ cell lineage. Birth Defects Res. C Embryo Today 2009, 87, 1–26. [Google Scholar] [CrossRef]
- Lin, L.H.; DePhilip, R.M. Differential expression of placental (P)-cadherin in sertoli cells and peritubular myoid cells during postnatal development of the mouse testis. Anat. Rec. 1996, 244, 155–164. [Google Scholar] [CrossRef]
- Fleming, A.; Ghahramani, N.; Zhu, M.X.; Délot, E.C.; Vilain, E. Membrane β-catenin and adherens junctions in early gonadal patterning. Dev. Dyn. 2012, 241, 1782–1798. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krentz, A.D.; Murphy, M.W.; Kim, S.; Cook, M.S.; Capel, B.; Zhu, R.; Matin, A.; Sarver, A.L.; Parker, K.L.; Griswold, M.D.; et al. The DM domain protein DMRT1 is a dose-sensitive regulator of fetal germ cell proliferation and pluripotency. Proc. Natl. Acad. Sci. USA 2009, 106, 22323–22328. [Google Scholar] [CrossRef] [Green Version]
- Song, X.; Xie, T. DE-cadherin-mediated cell adhesion is essential for maintaining somatic stem cells in the Drosophila ovary. Proc. Natl. Acad. Sci. USA 2002, 99, 14813–14818. [Google Scholar] [CrossRef] [Green Version]
- Song, X.; Zhu, C.H.; Doan, C.; Xie, T. Germline stem cells anchored by adherens junctions in the Drosophila ovary niches. Science 2002, 296, 1855–1857. [Google Scholar] [CrossRef]
- Niu, W.; Wang, Y.; Wang, Z.; Xin, Q.; Wang, Y.; Feng, L.; Zhao, L.; Wen, J.; Zhang, H.; Wang, C.; et al. JNK signaling regulates E-cadherin junctions in germline cysts and determines primordial follicle formation in mice. Development 2016, 143, 1778–1787. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Roy, S.K. Expression of E-cadherin and N-cadherin in perinatal hamster ovary: Possible involvement in primordial follicle formation and regulation by follicle-stimulating hormone. Endocrinology 2010, 151, 2319–2330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Machell, N.H.; Farookhi, R. E- and N-cadherin expression and distribution during luteinization in the rat ovary. Reproduction 2003, 125, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Mora, J.M.; Fenwick, M.A.; Castle, L.; Baithun, M.; Ryder, T.A.; Mobberley, M.; Carzaniga, R.; Franks, S.; Hardy, K. Characterization and significance of adhesion and junction-related proteins in mouse ovarian follicles. Biol. Reprod. 2012, 86, 153. [Google Scholar] [CrossRef]
- Sundfeldt, K.; Piontkewitz, Y.; Billig, H.; Hedin, L. E-cadherin-catenin complex in the rat ovary: Cell-specific expression during folliculogenesis and luteal formation. J. Reprod. Fertil. 2000, 118, 375–385. [Google Scholar] [CrossRef] [Green Version]
- Piprek, R.P.; Kolasa, M.; Podkowa, D.; Kloc, M.; Kubiak, J.Z. Tissue-specific knockout of E-cadherin (Cdh1) in developing mouse gonads causes germ cells loss. Reproduction 2019, 158, 147–157. [Google Scholar] [CrossRef]
- Yan, H.H.; Mruk, D.D.; Lee, W.M.; Cheng, C.Y. Blood-testis barrier dynamics are regulated by testosterone and cytokines via their differential effects on the kinetics of protein endocytosis and recycling in Sertoli cells. FASEB J. 2008, 22, 1945–1959. [Google Scholar] [CrossRef] [Green Version]
- Larue, L.; Ohsugi, M.; Hirchenhain, J.; Kemler, R. E-cadherin null mutant embryos fail to form a trophectoderm epithelium. Proc. Natl. Acad. Sci. USA 1994, 91, 8263–8267. [Google Scholar] [CrossRef] [Green Version]
- Radice, G.L.; Ferreira-Cornwell, M.C.; Robinson, S.D.; Rayburn, H.; Chodosh, L.A.; Takeichi, M.; Hynes, R.O. Precocious mammary gland development in P-cadherin-deficient mice. J. Cell Biol. 1997, 139, 1025–1032. [Google Scholar] [CrossRef]
- Piprek, R.P.; Kolasa, M.; Podkowa, D.; Kloc, M.; Kubiak, J.Z. N-cadherin is critical for the survival of germ cells, the formation of steroidogenic cells, and the architecture of developing mouse gonads. Cells 2019, 8, 1610. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.C.; Gregory, C.W.; DePhilip, R.M. Expression of E-cadherin in immature rat and mouse testis and in rat Sertoli cell cultures. Biol. Reprod. 1993, 49, 1353–1361. [Google Scholar] [CrossRef]
- Combes, A.N.; Wilhelm, D.; Davidson, T.; Dejana, E.; Harley, V.; Sinclair, A.; Koopman, P. Endothelial cell migration directs testis cord formation. Dev. Biol. 2009, 326, 112–120. [Google Scholar] [CrossRef] [Green Version]
- Newton, S.C.; Blaschuk, O.W.; Millette, C.F. N-cadherin mediates Sertoli cell-spermatogenic cell adhesion. Dev. Dyn. 1993, 197, 1–13. [Google Scholar] [CrossRef]
- Andersson, A.M.; Edvardsen, K.; Skakkebaek, N.E. Expression and localization of N- and E-cadherin in the human testis and epididymis. Int. J. Androl. 1994, 17, 174–180. [Google Scholar] [CrossRef]
- Byers, S.; Jegou, B.; MacCalman, C.; Blaschuk, O. Sertoli cell adhesion molecules and the collective organization of the testis. In The Sertoli Cell; Russel, L.D., Griswold, M.D., Eds.; Cache River Press: Clearwater, IL, USA, 1993; pp. 461–476. [Google Scholar]
- Lie, P.P.; Xia, W.; Wang, C.Q.; Mruk, D.D.; Yan, H.H.; Wong, C.H.; Lee, W.M.; Cheng, C.Y. Dynamin II interacts with the cadherin- and occludin-based protein complexes at the blood-testis barrier in adult rat testes. J. Endocrinol. 2006, 191, 571–586. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kopera, I.A.; Bilinska, B.; Cheng, C.Y.; Mruk, D.D. Sertoli-germ cell junctions in the testis: A review of recent data. Philos. Trans. R Soc. Lond. B Biol. Sci. 2010, 365, 1593–1605. [Google Scholar] [CrossRef] [Green Version]
- Perryman, K.J.; Stanton, P.G.; Loveland, K.L.; McLachlan, R.I.; Robertson, D.M. Hormonal dependency of neural cadherin in the binding of round spermatids to Sertoli cells in vitro. Endocrinology 1996, 137, 3877–3883. [Google Scholar] [CrossRef] [Green Version]
- Wine, R.N.; Chapin, R.E. Adhesion and signaling proteins spatiotemporally associated with spermiation in the rat. J. Androl. 1999, 20, 198–213. [Google Scholar]
- Jiang, X.; Ma, T.; Zhang, Y.; Zhang, H.; Yin, S.; Zheng, W.; Wang, L.; Wang, Z.; Khan, M.; Sheikh, S.W.; et al. Specific deletion of Cdh2 in Sertoli cells leads to altered meiotic progression and subfertility of mice. Biol. Reprod. 2015, 92, 79. [Google Scholar] [CrossRef]
- Cyr, D.G.; Blaschuk, O.W.; Robaire, B. Identification and developmental regulation of cadherin messenger ribonucleic acids in the rat testis. Endocrinology 1992, 131, 139–145. [Google Scholar] [CrossRef]
- Tokuda, M.; Kadokawa, Y.; Kurahashi, H.; Marunouchi, T. CDH1 is a specific marker for undifferentiated spermatogonia in mouse testes. Biol. Reprod. 2007, 76, 130–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Su, H.; Luo, F.; Wu, S.; Liu, L.; Liu, T.; Yu, B.; Wu, Y. E-cadherin can be expressed by a small population of rat undifferentiated spermatogonia in vivo and in vitro. In Vitro Cell. Dev. Biol. Anim. 2011, 47, 593. [Google Scholar] [CrossRef] [PubMed]
- Tolkunova, E.N.; Malashicheva, A.B.; Chikhirzhina, E.V.; Kostyleva, E.I.; Zeng, W.; Luo, J.; Dobrinski, I.; Hierholzer, A.; Kemler, R.; Tomilin, A.N. E-cadherin as a novel surface marker of spermatogonial stem cells. Cell Tissue Biol. 2009, 3, 103–109. [Google Scholar] [CrossRef] [Green Version]
- Johnson, K.J.; Patel, S.R.; Boekelheide, K. Multiple cadherin superfamily members with unique expression profiles are produced in rat testis. Endocrinology 2000, 141, 675–683. [Google Scholar] [CrossRef]
- Edson, M.A.; Nagaraja, A.K.; Matzuk, M.M. The mammalian ovary from genesis to revelation. Endocr. Rev. 2009, 30, 624–712. [Google Scholar] [CrossRef] [Green Version]
- Makrigiannakis, A.; Coukos, G.; Christofidou-Solomidou, M.; Gour, B.J.; Radice, G.L.; Blaschuk, O.; Coutifaris, C. N-cadherin-mediated human granulosa cell adhesion prevents apoptosis: A role in follicular atresia and luteolysis? Am. J. Pathol. 1999, 154, 1391–1406. [Google Scholar] [CrossRef]
- Peluso, J.J.; Pappalardo, A.; Fernandez, G. 2001 E-cadherin-mediated cell contact prevents apoptosis of spontaneously immortalized granulosa cells by regulating Akt kinase activity. Biol. Reprod. 2001, 64, 1183–1190. [Google Scholar] [CrossRef] [Green Version]
- Nakhuda, G.S.; Pauli, S.A.; Liao, F.; Bohlen, P.; Sauer, M.V.; Zimmermann, R.C. Inhibition of the vascular endothelial cell specific adhesion molecule VE-cadherin blocks gonadotropin-dependent folliculogenesis and corpus luteum formation. Fertli. Steril. 2003, 80 (Suppl. S3), 78–79. [Google Scholar] [CrossRef]
- Nakhuda, G.S.; Zimmermann, R.C.; Bohlen, P.; Liao, F.; Sauer, M.V.; Kitajewski, J. Inhibition of the vascular endothelial cell (VE)-specific adhesion molecule VE-cadherin blocks gonadotropin-dependent folliculogenesis and corpus luteum formation and angiogenesis. Endocrinology 2005, 146, 1053–1059. [Google Scholar] [CrossRef] [Green Version]
- Cyr, D.G.; Hermo, L.; Blaschuk, O.W.; Robaire, B. Distribution and regulation of epithelial cadherin messenger ribonucleic acid and immunocytochemical localization of epithelial cadherin in the rat epididymis. Endocrinology 1992, 130, 353–363. [Google Scholar] [CrossRef]
- Cyr, D.G.; Gregory, M.; Dubé, E.; Dufresne, J.; Chan, P.T.; Hermo, L. Orchestration of occludins, claudins, catenins and cadherins as players involved in maintenance of the blood-epididymal barrier in animals and humans. Asian J. Androl. 2007, 9, 463–475. [Google Scholar] [CrossRef] [PubMed]
- Suarez, S.S. Mammalian sperm interactions with the female reproductive tract. Cell Tissue Res. 2016, 363, 185–194. [Google Scholar] [CrossRef] [Green Version]
- Tsuchiya, B.; Sato, Y.; Kameya, T.; Okayasu, I.; Mukai, K. Differential expression of N-cadherin and E-cadherin in normal human tissues. Arch. Histol. Cytol. 2006, 69, 135–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vazquez-Levin, M.H.; Marín-Briggiler, C.I.; Caballero, J.N.; Veiga, M.F. Epithelial and neural cadherin expression in the mammalian reproductive tract and gametes and their participation in fertilization-related events. Dev. Biol. 2015, 401, 2–16. [Google Scholar] [CrossRef] [Green Version]
- Caballero, J.N.; Gervasi, M.G.; Veiga, M.F.; Dalvit, G.C.; Perez-Martínez, S.; Cetica, P.D.; Vazquez-Levin, M.H. Epithelial cadherin is present in bovine oviduct epithelial cells and gametes, and is involved in fertilization-related events. Theriogenology 2014, 81, 1189–1206. [Google Scholar] [CrossRef]
- Marín-Briggiler, C.I.; Lapyckyj, L.; González Echeverría, M.F.; Rawe, V.Y.; Alvarez Sedó, C.; Vazquez-Levin, M.H. Neural cadherin is expressed in human gametes and participates in sperm-oocyte interaction events. Int. J. Androl. 2010, 33, e228–e239. [Google Scholar] [CrossRef]
- DeFalco, T.J.; Verney, G.; Jenkins, A.B.; McCaffery, J.M.; Russell, S.; Van Doren, M. Sex-specific apoptosis regulates sexual dimorphism in the Drosophila embryonic gonad. Dev. Cell. 2003, 5, 205–216. [Google Scholar] [CrossRef] [Green Version]
- Jenkins, A.B.; McCaffery, J.M.; Van Doren, M. Drosophila E-cadherin is essential for proper germ cell-soma interaction during gonad morphogenesis. Development 2003, 130, 4417–4426. [Google Scholar] [CrossRef] [Green Version]
- Niewiadomska, P.; Godt, D.; Tepass, U. DE-Cadherin is required for intercellular motility during Drosophila oogenesis. J. Cell Biol. 1999, 144, 533–547. [Google Scholar] [CrossRef]
- Epstein, Y.; Perry, N.; Volin, M.; Zohar-Fux, M.; Braun, R.; Porat-Kuperstein, L.; Toledano, H. miR-9a modulates maintenance and ageing of Drosophila germline stem cells by limiting N-cadherin expression. Nat. Commun. 2017, 8, 600. [Google Scholar] [CrossRef] [Green Version]
- Loyer, N.; Kolotuev, I.; Pinot, M.; Le Borgne, R. Drosophila E-cadherin is required for the maintenance of ring canals anchoring to mechanically withstand tissue growth. Proc. Natl. Acad. Sci. USA 2015, 112, 12717–12722. [Google Scholar] [CrossRef] [Green Version]
- Cai, D.; Chen, S.C.; Prasad, M.; He, L.; Wang, X.; Choesmel-Cadamuro, V.; Sawyer, J.K.; Danuser, G.; Montell, D.J. Mechanical feedback through E-cadherin promotes direction sensing during collective cell migration. Cell 2014, 157, 1146–1159. [Google Scholar] [CrossRef] [Green Version]
- Florecki, M.M.; Hartfelder, K. Unconventional cadherin localization in honey bee gonads revealed through domain-specific Apis mellifera E- and N-cadherin antibodies indicates alternative functions. Insects 2012, 3, 1200–1219. [Google Scholar] [CrossRef] [Green Version]


| Family | Type | Examples |
|---|---|---|
| Classical cadherins (CDH)—contain at least five conservative cadherin repeats in ectodomain, bind actin microfilaments via catenins | Type I classical cadherins—contain HAV (His-Ala-Val) motif in the first extracellular repeat | 5 cadherins: E-, N-, P- R-, M-cadherin (CDH1, 2, 3, 4, 15) |
| Type II classical cadherins—contain no HAV motif in the first extracellular repeat | 13 cadherins including e.g., VE-cadherin (CDH5, 6, 7, 8, 9, 10, 11, 12, 18, 19, 20, 22, 24) | |
| 7D cadherins—contain seven extracellular cadherin repeats and only a portion of the Ca2+-binding motif at the interface between cadherin repeats 2 and 3 | 2 cadherins: CDH16, CDH17 | |
| Desmosomal cadherins—act as transmembrane proteins in desmosomes and bind intermediate filaments via desmoplakin | 3 desmocollins (DSC1-3) and 4 desmogleins (DSG1-4) | |
| CELSR cadherins —Cadherin EGF LAG seven-pass G-type receptors | 3 receptors: CELSR1,2,3 | |
| Protocadherins (PCDH)—contain ectodomain comprising six or seven cadherin repeats, transmembrane domain, and distinct, protocadherin-specific cytoplasmic domain | Clustered protocadherins | 12 PCDHs, such as PCDH1, PCDH7 |
| Non-clustered protocadherins | 64 PCDHs, such as PCDHA1, PCDHB1 | |
| Related cadherins (CDHR)—comprise at least two consecutive typical cadherin motifs | — | 17 cadherins, such as CDHR1, FAT1-4, calsyntenins CLSTN1-3, RET |
| CadherinType | Gene Symbol | Site of Expression |
|---|---|---|
| Type I cadherins | Cdh1 (E-cadherin) | germ cells in XX and XY gonads |
| Cdh2 (N-cadherin) | high expression in XX and XY supporting cells, lower in interstitial/stromal cells | |
| Cdh3 (P-cadherin) | expression in all gonadal cells, higher in XY supporting and XY germ cells | |
| Type II cadherins | Cdh5 (VE-cadherin) | interstitial/stromal cells |
| Cdh6 (K-cadherin) | interstitial/stromal cells | |
| Cdh9 (T1-cadherin) | XX and XY supporting cells | |
| Cdh10 (T2-cadherin) | interstitial/stromal cells | |
| Cdh11 (OB-cadherin) | high expression in interstitial/stromal cell, lower in supporting cells | |
| Cdh12 (N2-cadherin) | residual expression | |
| Cdh13 | residual expression | |
| Cdh18 | high expression in all gonadal cells | |
| Cdh23 | residual expression, higher in XY supporting cells | |
| Cdh24 | residual expression | |
| Desmosomal cadherins | Dsg2 (desmoglein 2) | supporting and germ cells |
| Dsc2 (desmocollin 2) | somatic gonadal cells, higher in supporting than interstitial/stromal cells | |
| Clustered protocadherins | Pcdh1 | residual expression |
| Pcdh7 | residual expression | |
| Pcdh9 | germ cells | |
| Pcdh11x | all studied gonadal somatic cells | |
| Pcdh12 | residual expression | |
| Pcdh17 | residual expression | |
| Pcdh18 | high expression in gonadal somatic cells | |
| Pcdh19 | residual expression | |
| Non-clustered protocadherins | Pcdhb3 | residual expression |
| Pcdhb16 | residual expression | |
| Pcdhb17 | interstitial/stromal cells | |
| Pcdhb18 | residual expression | |
| Pcdhb19 | interstitial/stromal cells | |
| Pcdhb20 | interstitial/stromal cells | |
| Pcdhb21 | residual expression | |
| Pcdhb22 | residual expression | |
| Pcdhga12 | residual expression | |
| Other cadherins | Celsr1 | residual expression |
| Clstn1 | all studied gonadal cells | |
| Dchs1 (Dachsous) | interstitial/stromal cells | |
| Fat1 | all studied gonadal cells, higher in XY supporting cells, lower in XX supporting cells | |
| Fat3 | interstitial/stromal cells | |
| Fat4 | somatic gonadal cells, higher in XX supporting cells |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piprek, R.P.; Kloc, M.; Mizia, P.; Kubiak, J.Z. The Central Role of Cadherins in Gonad Development, Reproduction, and Fertility. Int. J. Mol. Sci. 2020, 21, 8264. https://doi.org/10.3390/ijms21218264
Piprek RP, Kloc M, Mizia P, Kubiak JZ. The Central Role of Cadherins in Gonad Development, Reproduction, and Fertility. International Journal of Molecular Sciences. 2020; 21(21):8264. https://doi.org/10.3390/ijms21218264
Chicago/Turabian StylePiprek, Rafał P., Malgorzata Kloc, Paulina Mizia, and Jacek Z. Kubiak. 2020. "The Central Role of Cadherins in Gonad Development, Reproduction, and Fertility" International Journal of Molecular Sciences 21, no. 21: 8264. https://doi.org/10.3390/ijms21218264
APA StylePiprek, R. P., Kloc, M., Mizia, P., & Kubiak, J. Z. (2020). The Central Role of Cadherins in Gonad Development, Reproduction, and Fertility. International Journal of Molecular Sciences, 21(21), 8264. https://doi.org/10.3390/ijms21218264








