Effect of the Cationic Head Group on Cationic Surfactant-Based Surfactant Mediated Gelation (SMG)
Abstract
:1. Introduction
2. Results
2.1. Gelation Tests
2.2. Confirmation of the Hydrogel Structure and Orthogonality
2.3. Sol-Gel Transition Temperature
2.4. Viscoelasticity
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Sample Preparation
4.3. Gelation Test and Sol-Gel Transition Temperatures
4.4. Optical Microscopy
4.5. Differential Scanning Calorimetry (DSC)
4.6. Rheological Measurements
4.7. SWAXS Measurements
4.8. Ultraviolet-Visible (UV-vis) Spectroscopy
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| LMG | low-molecular-weight gelator |
| SAFiNs | self-assembled fibrillar networks |
| 12-HOA | 12-hydroxyoctadecanoic acid |
| BME | bicontinuous microemulsion |
| Lα | lamellar liquid crystal |
| SMG | surfactant-mediated gelation |
| CTAB | hexadecyltrimethylammonium bromide |
| CTAC | hexadecyltrimethylammonium chloride |
| CPC | hexadecylpyridinium chloride |
| cgc | critical gelation concentration |
| SWAXS | small- and wide-angle X-ray scattering |
| CMC | critical micellar concentration |
| DSC | differential scanning calorimetry |
| LVE | linear viscoelastic |
References
- Weiss, R.G.; Terech, P. Molecular Gels. Materials with Self-Assembled Fibrillar Networks; Springer: Heidelberg, Germany, 2006. [Google Scholar]
- Caran, K.L.; Lee, D.-C.; Weiss, R.G. Molecular Gels and their Fibrillar Networks. In Soft Fibrillar Materials: Fabrication and Applications, 1st ed.; Liu, X.Y., Li, J.-L., Eds.; Wiley-VCH Verlag: Weinheim, Germany, 2013; pp. 3–75. [Google Scholar]
- Tachibana, T.; Kambara, H. Studies of Helical Aggregates of Molecules. I. Enantiomorphism in the Helical Aggregates of Optically Active 12-Hydroxystearic Acid and Its Lithium Salt. Bull. Chem. Soc. Jpn. 1969, 42, 3422–3424. [Google Scholar] [CrossRef] [Green Version]
- Tachibana, T.; Mori, T.; Hori, K. Chiral Mesophase of 12-Hydroxyoctadecanoic Acid in Jelly and in the Solid State. I. A New Type of Lyotropic Mesophase in Jelly with Organic Solvents. Bull. Chem. Soc. Jpn. 1980, 53, 1714–1719. [Google Scholar] [CrossRef]
- Tachibana, T.; Mori, T.; Hori, K. Chiral Mesophases of 12-Hydroxyoctadecanoic Acid in Jelly and in the Solid State. II. A New Type of Mesomorphic Solid State. Bull. Chem. Soc. Jpn. 1981, 54, 73–80. [Google Scholar] [CrossRef] [Green Version]
- Sato, H.; Hori, K.; Sakurai, T.; Yamagishi, A. Long Distance Chiral Transfer in a Gel: Experimental and Ab Initio Analyses of Vibrational Circular Dichroism Spectra of R- and S-12-Hydroxyoctadecanoic Acid Gels. Chem. Phys. Lett. 2008, 467, 140–143. [Google Scholar] [CrossRef]
- Sakurai, T.; Masuda, Y.; Sato, H.; Yamagishi, A.; Kawaji, H.; Atake, T.; Hori, K. A Comparative Study on Chiral and Racemic 12-Hydroxyoctadecanoic Acids in the Solutions and Aggregation States: Does the Racemic Form Really Form a Gel? Bull. Chem. Soc. Jpn. 2010, 83, 145–150. [Google Scholar] [CrossRef]
- Terech, P. Small-Angle-Scattering Study of 12-Hydroxystearic Physical Organogels and Lubricating Greases. Colloid Polym. Sci. 1991, 269, 490–500. [Google Scholar] [CrossRef]
- Terech, P. 12-D-Hydroxyoctadecanoic Acid Organogels: A Small Angle Neutron Scattering Study. J. Phys. II France 1992, 2, 2181–2195. [Google Scholar] [CrossRef]
- Terech, P.; Rodriguez, V.; Barnes, J.D.; McKenna, G.B. Organogels and Aerogels of Racemic and Chiral 12-Hydroxyoctadecanoic Acid. Langmuir 1994, 10, 3406–3418. [Google Scholar] [CrossRef]
- Terech, P.; Pasquier, D.; Bordas, V.; Rossat, C. Rheological Properties and Structural Correlations in Molecular Organogels. Langmuir 2000, 16, 4485–4494. [Google Scholar] [CrossRef]
- Wu, S.; Gao, J.; Emge, T.J.; Rogers, M.A. Solvent-Induced Polymorphic Nanoscale Transitions for 12-Hydroxyoctadecanoic Acid Molecular Gels. Cryst. Growth Des. 2013, 13, 1360–1366. [Google Scholar] [CrossRef]
- Gao, J.; Wu, S.; Emge, T.J.; Rogers, M.A. Nanoscale and Microscale Structural Changes Alter the Critical Gelator Concentration of Self-assembled Fibrillar Networks. Cryst. Eng. Comm. 2013, 15, 4507–4515. [Google Scholar] [CrossRef]
- Laupheimer, M.; Preisig, N.; Stubenrauch, C. The Molecular Organogel n-Decane/12-Hydroxyoctadecanoic Acid: Sol-Gel Transition, Rheology, and Microstructure. Colloids. Surf. A 2015, 469, 315–325. [Google Scholar] [CrossRef]
- Koitani, S.; Dieterich, S.; Preisig, N.; Aramaki, K.; Stubenrauch, C. Gelling Lamellar Phases of the Binary System Water−Didodecyldimethylammonium Bromide with an Organogelator. Langmuir 2017, 33, 12171–12179. [Google Scholar] [CrossRef]
- Aramaki, K.; Koitani, S.; Takimoto, E.; Kondo, M.; Stubenrauch, C. Hydrogelation with a Water-Insoluble Organogelator–Surfactant Mediated Gelation (SMG). Soft Matter 2019, 15, 8896–8904. [Google Scholar] [CrossRef]
- Douliez, J.-P.; Gaillard, C.; Navailles, L.; Nallet, F. Novel Lipid System Forming Hollow Microtubes at High Yields and Concentration. Langmuir 2006, 22, 2942–2945. [Google Scholar] [CrossRef] [PubMed]
- Fameau, A.-L.; Cousin, F.; Navailles, L.; Nallet, F.; Boué, F.; Douliez, J.-P. Multiscale Structural Characterizations of Fatty Acid Multilayered Tubes with a Temperature-Tunable Diameter. J. Phys. Chem. B 2011, 115, 9033–9039. [Google Scholar] [CrossRef]
- Nakagawa, M.; Kawai, T. Chirality-Controlled Syntheses of Double-Helical Au Nanowires. J. Am. Chem. Soc. 2018, 140, 4991–4994. [Google Scholar] [CrossRef]
- Nakagawa, M.; Kawai, T. Tuning Gel Sol Transition Behavior of a Hydrogel Based on 12-Hydroxystearic Acid and a Long-Chain Amidoamine Derivative. Bull. Chem. Soc. Jpn. 2019, 92, 435–440. [Google Scholar] [CrossRef]
- Fameau, A.-L.; Rogers, M.A. The Curious Case of 12-Hydroxystearic Acid—The Dr. Jekyll & Mr. Hyde of Molecular Gelators. Curr. Opin. Colloid Interface Sci. 2020, 45, 68–82. [Google Scholar]
- Stubenrauch, C.; Tessendorf, R.; Strey, R.; Lynch, I.; Dawson, K.A. Gelled Polymerizable Microemulsions. 1. Phase Behavior. Langmuir 2007, 23, 7730–7737. [Google Scholar] [CrossRef]
- Stubenrauch, C.; Tessendorf, R.; Salvati, A.; Topgaard, D.; Sottmann, T.; Strey, R.; Lynch, I. Gelled Polymerizable Microemulsions. 2. Microstructure. Langmuir 2008, 24, 8473–8482. [Google Scholar] [CrossRef]
- Magno, M.; Tessendorf, R.; Medronho, B.; da Graça Martins Miguel, M.; Stubenrauch, C. Gelled Polymerizable Microemulsions. Part 3 Rheology. Soft Matter 2009, 5, 4763–4772. [Google Scholar] [CrossRef]
- Laupheimer, M.; Jovic, K.; Antunes, F.E.; da Graça Martins Miguel, M.; Stubenrauch, C. Studying Orthogonal Self-Assembled Systems: Phase Behaviour and Rheology of Gelled Microemulsions. Soft Matter 2013, 9, 3661–3670. [Google Scholar] [CrossRef]
- Laupheimer, M.; Sottmann, T.; Schweins, R.; Stubenrauch, C. Studying Orthogonal Self-Assembled Systems: Microstructure of Gelled Bicontinuous Microemulsions. Soft Matter 2014, 10, 8744–8757. [Google Scholar] [CrossRef]
- Xu, Y.; Laupheimer, M.; Preisig, N.; Sottmann, T.; Schmidt, C.; Stubenrauch, C. Gelled Lyotropic Liquid Crystals. Langmuir 2015, 31, 8589–8598. [Google Scholar] [CrossRef]
- Peng, K.; Sottmann, T.; Stubenrauch, C. Gelled Non-Toxic Microemulsions: Phase Behavior & Rheology. Soft Matter 2019, 15, 8361–8371. [Google Scholar] [PubMed]
- Steck, K.; Schmidt, C.; Stubenrauch, C. The Twofold Role of 12-Hydroxyoctadecanoic Acid (12-HOA) in a Ternary Water—Surfactant—12-HOA System: Gelator and Co-Surfactant. Gels 2018, 4, 78–91. [Google Scholar] [CrossRef] [Green Version]
- Boekhoven, J.; Brizard, A.M.; Stuart, M.C.A.; Florusse, L.; Raffy, G.; Del Guerzo, A.; van Esch, J.H. Bio-Inspired Supramolecular Materials by Orthogonal Self-Assembly of Hydrogelators and Phospholipids. Chem. Sci. 2016, 7, 6021–6031. [Google Scholar] [CrossRef] [Green Version]
- Stubenrauch, C.; Gießelmann, F. Gelled Complex Fluids: Combining Unique Structures with Mechanical Stability. Angew. Chem. Int. Ed. 2016, 55, 3268–3275. [Google Scholar] [CrossRef]
- Carey, M.C.; Small, D.M. Micellar Properties of Dihydroxy and Trihydroxy Bile Salts: Effects of Counterion and Temperature. J. Colloid Interface Sci. 1969, 31, 382–396. [Google Scholar] [CrossRef]
- Ross-Murphy, S.B.; Shatwell, K.P. Polysaccharide Strong and Weak Gels. Biorheology 1993, 30, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Dieterich, S.; Sottmann, T.; Giesselmann, F. Gelation of Lyotropic Liquid-Crystal Phases–The Interplay between Liquid Crystalline Order and Physical Gel Formation. Langmuir 2019, 35, 16793–16802. [Google Scholar] [CrossRef] [PubMed]
- Rogers, M.A.; Marangoni, A.G. Solvent-Modulated Nucleation and Crystallization Kinetics of 12-Hydroxystearic Acid: A Nonisothermal Approach. Langmuir 2009, 25, 8556–8566. [Google Scholar] [CrossRef] [PubMed]
- Matzinger, S.; Hussey, D.M.; Fayer, M.D. Fluorescent Probe Solubilization in the Headgroup and Core Regions of Micelles: Fluorescence Lifetime and Orientational Relaxation Measurements. J. Phys. Chem. B 1998, 102, 7216–7224. [Google Scholar] [CrossRef] [Green Version]
- Gunaseelan, K.; Dev, S.; Ismail, K. Estimation of Micellization Parameters of Cetylpyridinium Chloride in Water Using the Mixed Electrolyte Model for Conductance. Indian J. Chem. A 2000, 39, 761–764. [Google Scholar]



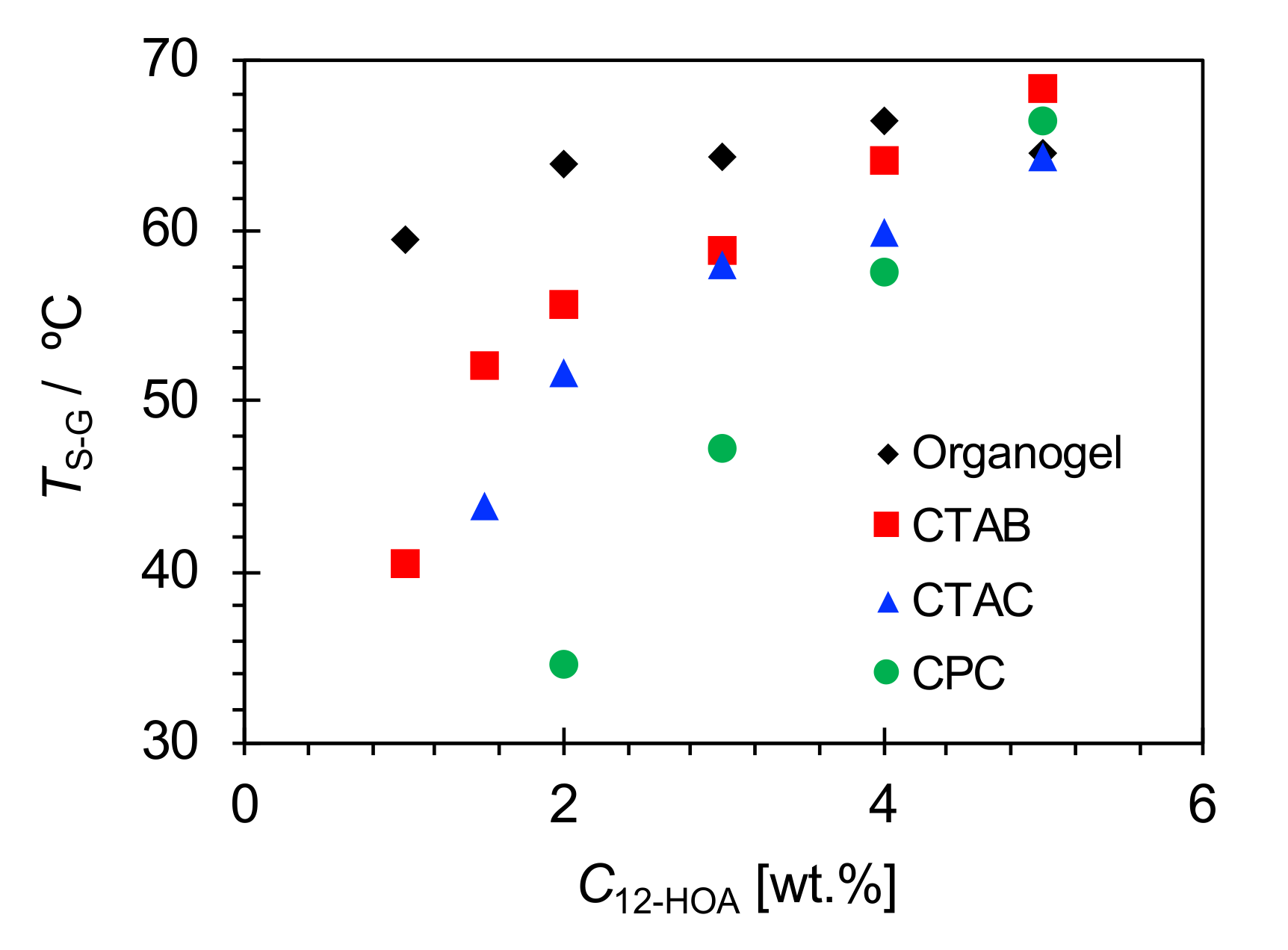

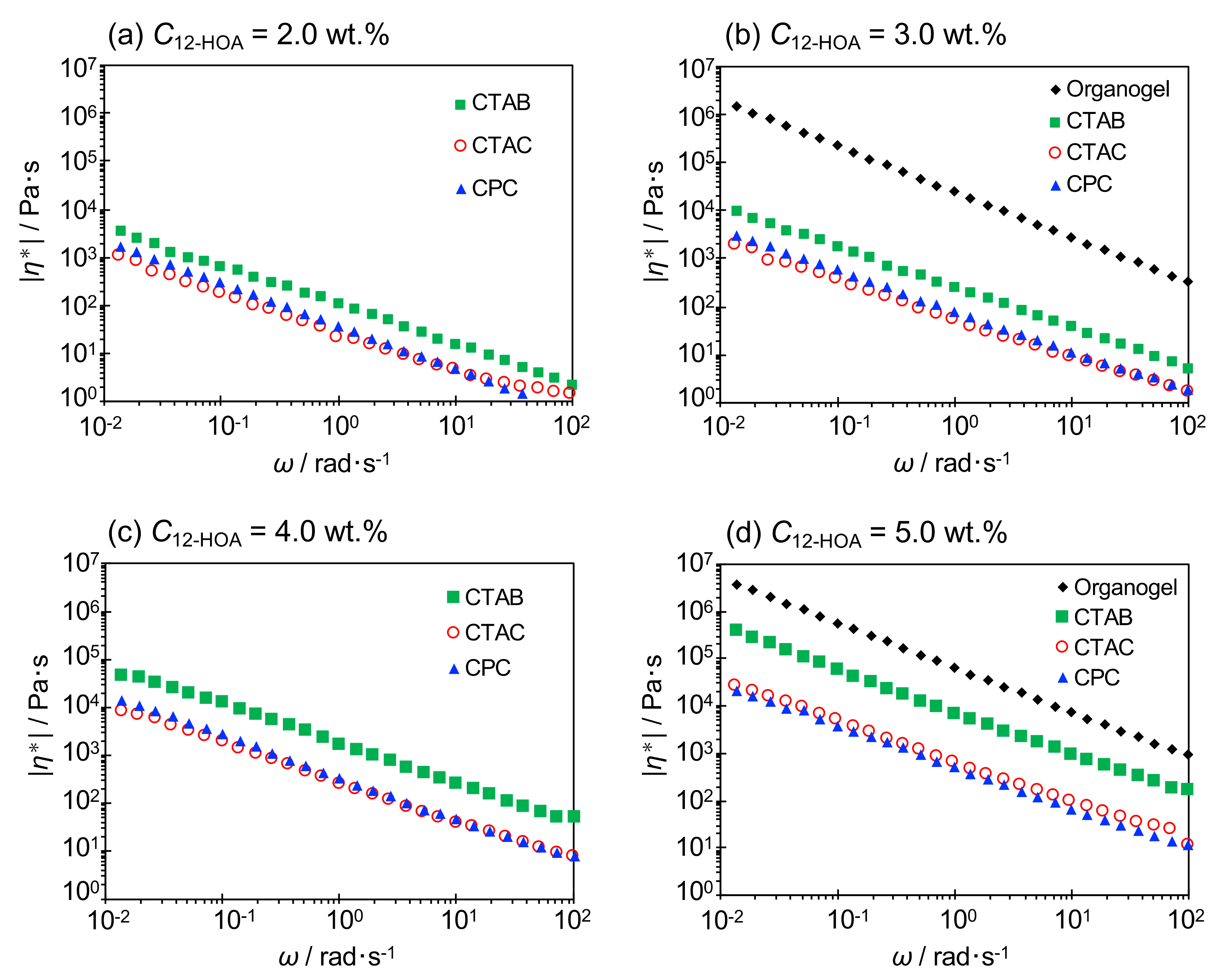
| C12-HOA [wt.%] | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Surfactant 1 | 0.5 | 0.75 | 1.0 | 1.25 | 1.5 | 1.75 | 2.0 | 3.0 | 4.0 | 5.0 |
| CTAB | sol | sol | gel | - | gel | - | gel | gel | gel | gel |
| CTAC | sol | - | sol | sol | sol | gel | gel | gel | gel | gel |
| CPC | sol | - | sol | - | sol | sol | gel | gel | gel | gel |
| CMC/mM | α/- | |
|---|---|---|
| CTAB | 0.91 | 0.28 |
| CTAC | 1.32 | 0.42 |
| CPC | 0.99 | 0.49 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aramaki, K.; Takimoto, E.; Yamaguchi, T. Effect of the Cationic Head Group on Cationic Surfactant-Based Surfactant Mediated Gelation (SMG). Int. J. Mol. Sci. 2020, 21, 8046. https://doi.org/10.3390/ijms21218046
Aramaki K, Takimoto E, Yamaguchi T. Effect of the Cationic Head Group on Cationic Surfactant-Based Surfactant Mediated Gelation (SMG). International Journal of Molecular Sciences. 2020; 21(21):8046. https://doi.org/10.3390/ijms21218046
Chicago/Turabian StyleAramaki, Kenji, Eriko Takimoto, and Takumi Yamaguchi. 2020. "Effect of the Cationic Head Group on Cationic Surfactant-Based Surfactant Mediated Gelation (SMG)" International Journal of Molecular Sciences 21, no. 21: 8046. https://doi.org/10.3390/ijms21218046





