Gene Expression Regulation and Secretory Activity of Mesenchymal Stem Cells upon In Vitro Contact with Microarc Calcium Phosphate Coating
Abstract
1. Introduction
2. Results and Discussion
2.1. Cell Viability and Cellular Immunophenotype
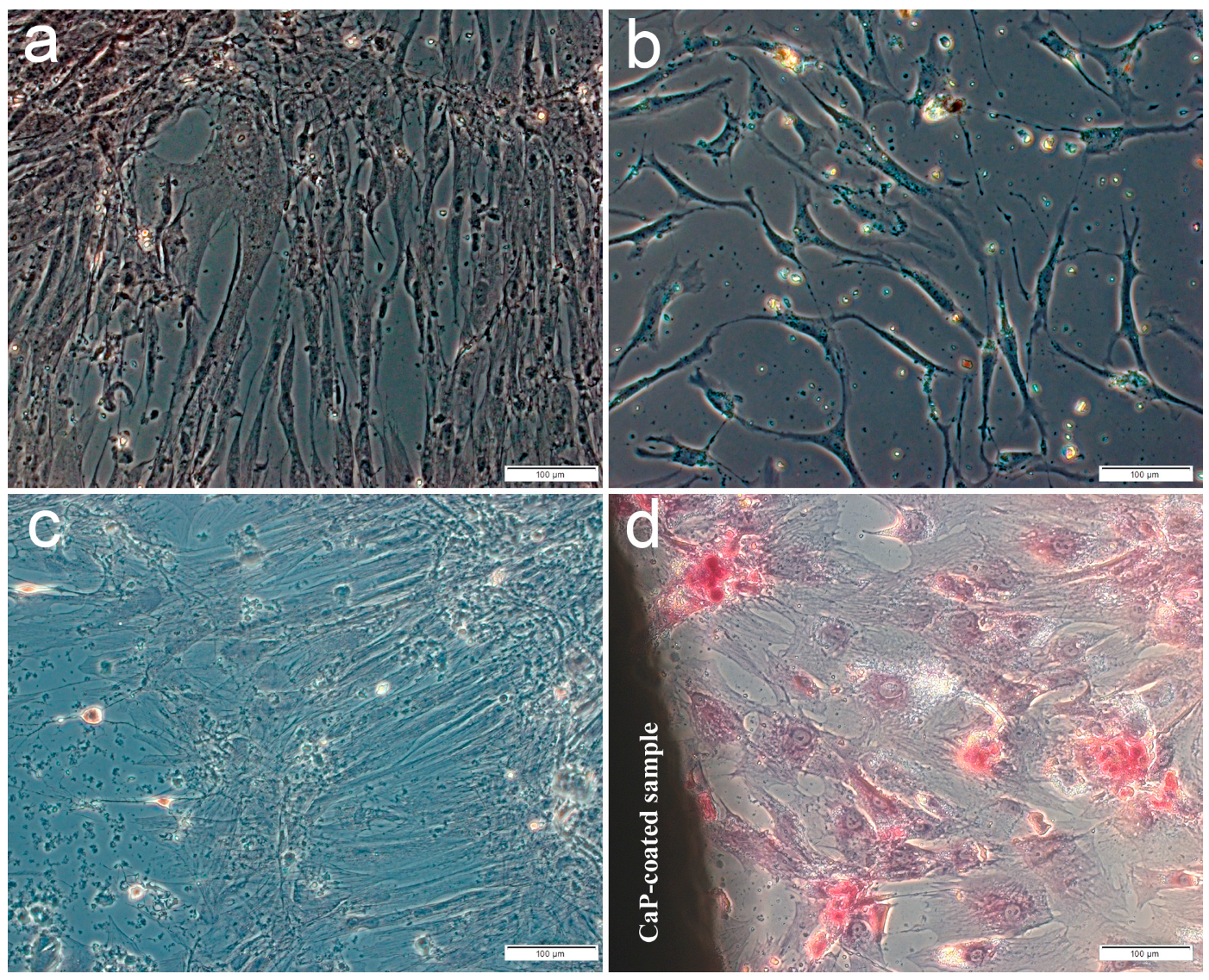
2.2. In Vitro Osteogenic Differentiation
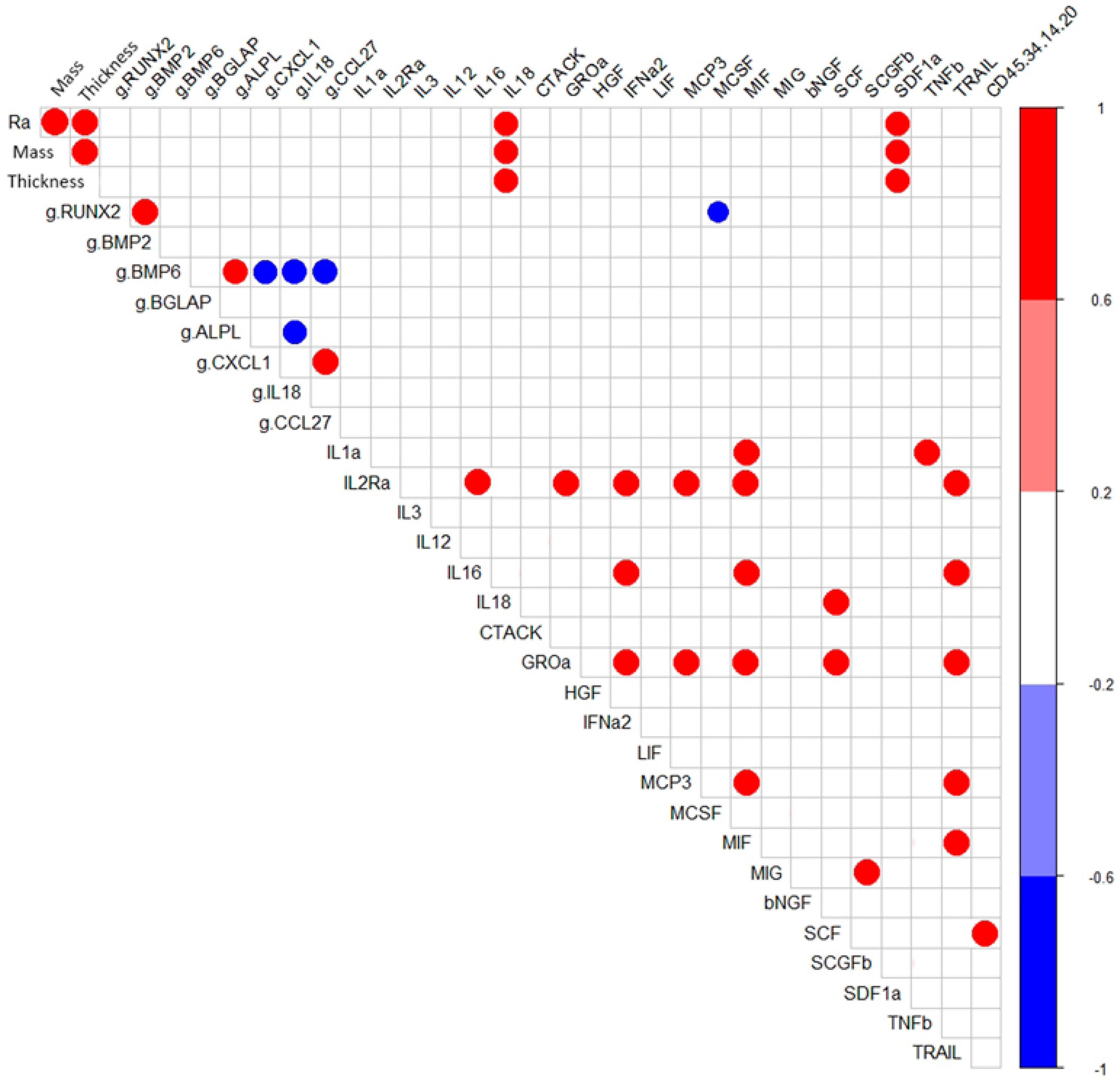
2.3. Expression of Osteogenic, Cytokine, and Chemokine Genes
2.4. Cytokine and Chemokine Secretion
3. Material and Methods
3.1. Samples of Naterial with a CaP Coating
3.2. Human Cell Isolation
3.3. Human Cell Culture
3.4. Cytokine Assay
3.5. Cellular Viability and Immunophenotype Analysis
3.6. Gene Expression Analysis
3.7. Alizarin Red Staining
3.8. Statistical Analysis
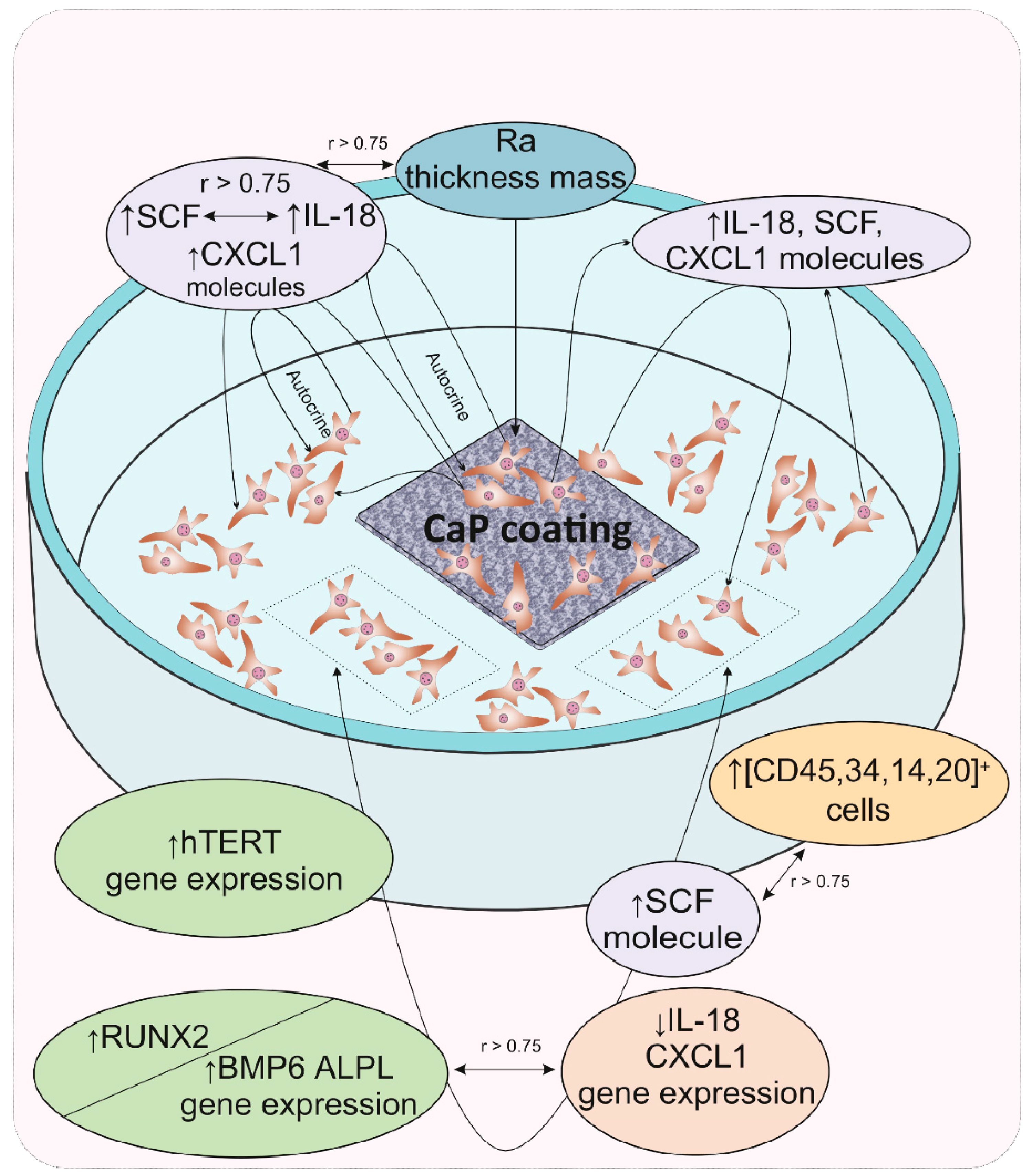
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Arron, J.R.; Choi, Y. Bone versus immune system. Nature 2000, 408, 535–536. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Bleek, K.; Kwee, B.J.; Mooney, D.J.; Duda, G.N. Boon and Bane of Inflammation in Bone Tissue Regeneration and Its Link with Angiogenesis. Tissue Eng. Part B Rev. 2015, 21, 354–364. [Google Scholar] [CrossRef] [PubMed]
- Kolar, P.; Schmidt-Bleek, K.; Schell, H.; Gaber, T.; Toben, D.; Schmidmaier, G.; Perka, C.; Buttgereit, F.; Duda, G.N. The early fracture hematoma and its potential role in fracture healing. Tissue Eng. Part B Rev. 2010, 16, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Kon, T.; Cho, T.J.; Aizawa, T.; Yamazaki, M.; Nooh, N.; Graves, D.; Gerstenfeld, L.C.; Einhorn, T.A. Expression of osteoprotegerin, receptor activator of NF-kappaB ligand (osteoprotegerin ligand) and related proinflammatory cytokines during fracture healing. J. Bone Miner. Res. 2001, 16, 1004–1014. [Google Scholar] [CrossRef]
- Litvinova, L.S.; Shupletsova, V.V.; Yurova, K.A.; Khaziakhmatova, O.G.; Todosenko, N.M.; Malashchenko, V.V.; Shunkin, E.O.; Melashchenko, E.S.; Khlusova, M.Y.; Komarova, E.G.; et al. Secretion of niche signal molecules under conditions of osteogenic differentiation of multipotent mesenchymal stromal cells induced by relief calcium phosphate coating. Biochem. Suppl. Ser. B 2019, 13, 341–348. [Google Scholar] [CrossRef]
- Theman, T.A.; Collins, M.T. The role of the calcium-sensing receptor in bone biology and pathophysiology. Curr. Pharm. Biotechnol. 2009, 10, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Bolander, J.; Chai, Y.C.; Geris, L.; Schrooten, J.; Lambrechts, D.; Roberts, S.J.; Luyten, F.P. Early BMP, Wnt and Ca (2+)/PKC pathway activation predicts the bone forming capacity of periosteal cells in combination with calcium phosphates. Biomaterials 2016, 86, 106–118. [Google Scholar] [CrossRef]
- Ali Akbari Ghavimi, S.; Allen, B.N.; Stromsdorfer, J.L.; Kramer, J.S.; Li, X.; Ulery, B.D. Calcium and phosphate ions as simple signaling molecules with versatile osteoinductivity. Biomed. Mater. 2018, 13, 055005. [Google Scholar] [CrossRef]
- Kubo, H.; Shimizu, M.; Taya, Y.; Kawamoto, T.; Michida, M.; Kaneko, E.; Igarashi, A.; Nishimura, M.; Segoshi, K.; Shimazu, Y.; et al. Identification of mesenchymal stem cell (MSC)-transcription factors by microarray and knockdown analyses, and signature molecule-marked MSC in bone marrow by immunohistochemistry. Genes Cells 2009, 14, 407–424. [Google Scholar] [CrossRef]
- Liu, T.M. Stemness of Mesenchymal Stem Cells. J. Stem Cell Ther. Transplant. 2017, 1, 71–73. [Google Scholar] [CrossRef]
- Koch, H.; Jadlowiec, J.A.; Campbell, P.G. Insulin-like growth factor-I induces early osteoblast gene expression in human mesenchymal stem cells. Stem Cells Dev. 2005, 14, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.S.; Song, Y.M.; Cho, T.H.; Park, Y.D.; Lee, K.B.; Noh, I.; Weber, F.; Hwang, S.J. In vitro response of primary human bone marrow stromal cells to recombinant human bone morphogenic protein-2 in the early and late stages of osteoblast differentiation. Dev. Growth Differ. 2008, 50, 553–564. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Ghali, O.; Lencel, P.; Broux, O.; Chauveau, C.; Devedjian, J.C.; Hardouin, P.; Magne, D. TNF-alpha and IL-1beta inhibit RUNX2 and collagen expression but increase alkaline phosphatase activity and mineralization in human mesenchymal stem cells. Life Sci. 2009, 84, 499–504. [Google Scholar] [CrossRef] [PubMed]
- Loi, F.; Córdova, L.A.; Pajarinen, J.; Lin, T.; Yao, Z.; Goodman, S.B. Inflammation, fracture and bone repair. Bone 2016, 86, 119–130. [Google Scholar] [CrossRef]
- Bryington, M.; Mendonça, G.; Nares, S.; Cooper, L.F. Osteoblastic and cytokine gene expression of implant-adherent cells in humans. Clin. Oral Implant. Res. 2014, 25, 52–58. [Google Scholar] [CrossRef]
- Zheng, Z.-W.; Chen, Y.-H.; Wu, D.-Y.; Wang, J.-B.; Lv, M.-M.; Wang, X.-S.; Sun, J.; Zhang, Z.-Y. Development of an Accurate and Proactive Immunomodulatory Strategy to Improve Bone Substitute Material-Mediated Osteogenesis and Angiogenesis. Theranostics 2018, 8, 5482–5500. [Google Scholar] [CrossRef]
- Yang, W.; Han, W.; He, W.; Li, J.; Wang, J.; Feng, H.; Qian, Y. Surface topography of hydroxyapatite promotes osteogenic differentiation of human bone marrow mesenchymal stem cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 60, 45–53. [Google Scholar] [CrossRef]
- Sedelnikova, M.B.; Komarova, E.G.; Sharkeev, Y.P.; Tolkacheva, T.V.; Khlusov, I.A.; Litvinova, L.S.; Yurova, K.A.; Shupletsova, V.V. Comparative investigations of structure and properties of micro-arc wollastonite-calcium phosphate coatings on titanium and zirconium-niobium alloy. Bioact. Mater. 2017, 2, 177–184. [Google Scholar] [CrossRef]
- Qadir, M.; Li, Y.; Munir, K.; Wen, C. Calcium phosphate-based composite coating by micro-arc oxidation (MAO) for biomedical application: A review. Crit. Rev. Solid State Mater. Sci. 2018, 43, 392–416. [Google Scholar] [CrossRef]
- Krząkała, A.; Kazek-Kęsik, A.; Simka, W. Application of plasma electrolytic oxidation to bioactive surface formation on titanium and its alloys. RSC Adv. 2013, 3, 19725. [Google Scholar] [CrossRef]
- Khlusov, I.A.; Dekhtyar, Y.; Sharkeev, Y.P.; Pichugin, V.F.; Khlusova, M.Y.; Polyaka, N.; Tjulkins, F.; Vendinya, V.; Legostaeva, E.V.; Litvinova, L.S.; et al. Nanoscale electrical potential and roughness of a calcium phosphate surface promotes the osteogenic phenotype of stromal cells. Materials 2018, 11, 978. [Google Scholar] [CrossRef] [PubMed]
- Litvinova, L.S.; Shupletsova, V.V.; Khaziakhmatova, O.G.; Yurova, K.A.; Malashchenko, V.V.; Melashchenko, E.S.; Todosenko, N.M.; Khlusova, M.Y.; Sharkeev, Y.P.; Komarova, E.G.; et al. Behavioral changes of multipotent mesenchymal stromal cells in contact with synthetic calcium phosphates in vitro. Cell Tissue Biol. 2018, 12, 112–119. [Google Scholar] [CrossRef]
- Sharkeev, Y.; Komarova, E.; Sedelnikova, M.; Khlusov, I.A.; Eroshenko, A.; Litvinova, L.; Shupletsova, V. Bioactive micro-arc calcium phosphate coatings on nanostructured and ultrafine-grained bioinert metals and alloys. In Bioceramics and Biocomposites; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2019; pp. 191–231. ISBN 978-1-119-37209-7. [Google Scholar]
- Chung, C.J.; Su, R.T.; Chu, H.J.; Te Chen, H.; Tsou, H.K.; He, J.L. Plasma electrolytic oxidation of titanium and improvement in osseointegration. J. Biomed. Mater. Res. Part B Appl. Biomater. 2013, 101, 1023–1030. [Google Scholar] [CrossRef] [PubMed]
- Rizwan, M.; Alias, R.; Zaidi, U.Z.; Mahmoodian, R.; Hamdi, M. Surface modification of valve metals using plasma electrolytic oxidation for antibacterial applications: A review. J. Biomed. Mater. Res. Part A. 2018, 106, 590–605. [Google Scholar] [CrossRef] [PubMed]
- Litvinova, L.S.; Shupletsova, V.V.; Yurova, K.A.; Khaziakhmatova, O.G.; Todosenko, N.M.; Khlusova, M.Y.; Slepchenko, G.B.; Cherempey, E.G.; Sharkeev, Y.P.; Komarova, E.G.; et al. Cell-IQ visualization of motility, cell mass, and osteogenic differentiation of multipotent mesenchymal stromal cells cultured with relief calcium phosphate coating. Dokl. Biochem. Biophys. 2017, 476, 310–315. [Google Scholar] [CrossRef]
- Bourin, P.; Bunnell, B.A.; Casteilla, L.; Dominici, M.; Katz, A.J.; March, K.L.; Redl, H.; Rubin, J.P.; Yoshimura, K.; Gimble, J.M. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: A joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International Society for Cellular Therapy (ISCT). Cytotherapy 2013, 15, 641–648. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef]
- Kaivosoja, E.; Suvanto, P.; Barreto, G.; Aura, S.; Soininen, A.; Franssila, S.; Konttinen, Y.T. Cell adhesion and osteogenic differentiation on three-dimensional pillar surfaces. J. Biomed. Mater. Res. A 2013, 101, 842–852. [Google Scholar] [CrossRef]
- Chai, Y.C.; Roberts, S.J.; Desmet, E.; Kerckhofs, G.; van Gastel, N.; Geris, L.; Carmeliet, G.; Schrooten, J.; Luyten, F.P. Mechanisms of ectopic bone formation by human osteoprogenitor cells on CaP biomaterial carriers. Biomaterials 2012, 33, 3127–3142. [Google Scholar] [CrossRef]
- Zhang, J.; Niu, C.; Ye, L.; Huang, H.; He, X.; Tong, W.-G.; Ross, J.; Haug, J.; Johnson, T.; Feng, J.Q.; et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature 2003, 425, 836–841. [Google Scholar] [CrossRef]
- Khlusov, I.A.; Karlov, A.V.; Pozhen’ko, N.S.; Sukhodolo, I.V.; Khlusova, M.Y. Relationship between osteogenic characteristics of bone marrow cells and calcium phosphate surface relief and solubility. Bull. Exp. Biol. Med. 2006, 141, 99–103. [Google Scholar] [CrossRef]
- Sedelnikova, M.B.; Komarova, E.G.; Sharkeev, Y.P.; Ugodchikova, A.V.; Mushtovatova, L.S.; Karpova, M.R.; Sheikin, V.V.; Litvinova, L.S.; Khlusov, I.A. Zn-, Cu- or Ag-incorporated micro-arc coatings on titanium alloys: Properties and behavior in synthetic biological media. Surf. Coat. Technol. 2019, 369, 52–68. [Google Scholar] [CrossRef]
- Liu, Y.K.; Lu, Q.Z.; Pei, R.; Ji, H.J.; Zhou, G.S.; Zhao, X.L.; Tang, R.K.; Zhang, M. The effect of extracellular calcium and inorganic phosphate on the growth and osteogenic differentiation of mesenchymal stem cells in vitro: Implication for bone tissue engineering. Biomed. Mater. 2009, 4, 025004. [Google Scholar] [CrossRef] [PubMed]
- Tian, T.; Xie, W.; Gao, W.; Wang, G.; Zeng, L.; Miao, G.; Lei, B.; Lin, Z.; Chen, X. Micro-Nano Bioactive Glass Particles Incorporated Porous Scaffold for Promoting Osteogenesis and Angiogenesis in vitro. Front. Chem. 2019, 7. [Google Scholar] [CrossRef]
- Zheng, J.; Zhao, F.; Zhang, W.; Mo, Y.; Zeng, L.; Li, X.; Chen, X. Sequentially-crosslinked biomimetic bioactive glass/gelatin methacryloyl composites hydrogels for bone regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 89, 119–127. [Google Scholar] [CrossRef]
- Hrubi, E.; Imre, L.; Robaszkiewicz, A.; Virág, L.; Kerényi, F.; Nagy, K.; Varga, G.; Jenei, A.; Hegedüs, C. Diverse effect of BMP-2 homodimer on mesenchymal progenitors of different origin. Hum. Cell 2018, 31, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Ducy, P.; Zhang, R.; Geoffroy, V.; Ridall, A.L.; Karsenty, G. Osf2/Cbfa1: A transcriptional activator of osteoblast differentiation. Cell 1997, 89, 747–754. [Google Scholar] [CrossRef]
- Carlier, A.; Chai, Y.C.; Moesen, M.; Theys, T.; Schrooten, J.; Van Oosterwyck, H.; Geris, L. Designing optimal calcium phosphate scaffold–cell combinations using an integrative model-based approach. Acta Biomater. 2011, 7, 3573–3585. [Google Scholar] [CrossRef] [PubMed]
- zur Nieden, N.I.; Kempka, G.; Ahr, H.J. In vitro differentiation of embryonic stem cells into mineralized osteoblasts. Differentiation 2003, 71, 18–27. [Google Scholar] [CrossRef]
- Huang, W.; Rudkin, G.H.; Carlsen, B.; Ishida, K.; Ghasri, P.; Anvar, B.; Yamaguchi, D.T.; Miller, T.A. Overexpression of BMP-2 Modulates Morphology, Growth, and Gene Expression in Osteoblastic Cells. Exp. Cell Res. 2002, 274, 226–234. [Google Scholar] [CrossRef]
- Ponader, S.; Vairaktaris, E.; Heinl, P.; Wilmowsky, C.V.; Rottmair, A.; Körner, C.; Singer, R.F.; Holst, S.; Schlegel, K.A.; Neukam, F.W.; et al. Effects of topographical surface modifications of electron beam melted Ti-6Al-4V titanium on human fetal osteoblasts. J. Biomed. Mater. Res. A 2008, 84, 1111–1119. [Google Scholar] [CrossRef] [PubMed]
- Uggeri, J.; Guizzardi, S.; Scandroglio, R.; Gatti, R. Adhesion of human osteoblasts to titanium: A morpho-functional analysis with confocal microscopy. Micron 2010, 41, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Faia-Torres, A.B.; Guimond-Lischer, S.; Rottmar, M.; Charnley, M.; Goren, T.; Maniura-Weber, K.; Spencer, N.D.; Reis, R.L.; Textor, M.; Neves, N.M. Differential regulation of osteogenic differentiation of stem cells on surface roughness gradients. Biomaterials 2014, 35, 9023–9032. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaka, K.; Yoshinari, M.; Shimono, M.; Inoue, T. Effects of multigrooved surfaces on osteoblast-like cells in vitro: Scanning electron microscopic observation and mRNA expression of osteopontin and osteocalcin. J. Biomed. Mater. Res. A 2004, 68, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Dalby, M.J.; McCloy, D.; Robertson, M.; Agheli, H.; Sutherland, D.; Affrossman, S.; Oreffo, R.O.C. Osteoprogenitor response to semi-ordered and random nanotopographies. Biomaterials 2006, 27, 2980–2987. [Google Scholar] [CrossRef]
- Sutherland, J.; Denyer, M.; Britland, S. Contact guidance in human dermal fibroblasts is modulated by population pressure. J. Anat. 2005, 206, 581–587. [Google Scholar] [CrossRef]
- McCafferty, M.M.; Burke, G.A.; Meenan, B.J. Mesenchymal stem cell response to conformal sputter deposited calcium phosphate thin films on nanostructured titanium surfaces. J. Biomed. Mater. Res. Part A 2014, 102, 3585–3597. [Google Scholar] [CrossRef]
- Thomas, C.H.; Collier, J.H.; Sfeir, C.S.; Healy, K.E. Engineering gene expression and protein synthesis by modulation of nuclear shape. Proc. Natl. Acad. Sci. USA 2002, 99, 1972–1977. [Google Scholar] [CrossRef]
- Shafrir, Y.; Forgacs, G. Mechanotransduction through the cytoskeleton. Am. J. Physiol. Cell Physiol. 2002, 282, C479–C486. [Google Scholar] [CrossRef]
- McBeath, R.; Pirone, D.M.; Nelson, C.M.; Bhadriraju, K.; Chen, C.S. Cell Shape, Cytoskeletal Tension, and RhoA Regulate Stem Cell Lineage Commitment. Dev. Cell 2004, 6, 483–495. [Google Scholar] [CrossRef]
- Prowse, P.D.H.; Elliott, C.G.; Hutter, J.; Hamilton, D.W. Inhibition of Rac and ROCK Signalling Influence Osteoblast Adhesion, Differentiation and Mineralization on Titanium Topographies. PLoS ONE 2013, 8, e58898. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lüthen, F.; Lange, R.; Becker, P.; Rychly, J.; Beck, U.; Nebe, J.G.B. The influence of surface roughness of titanium on beta1- and beta3-integrin adhesion and the organization of fibronectin in human osteoblastic cells. Biomaterials 2005, 26, 2423–2440. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, P.S.; Loboa, E.G. Cytoskeletal and focal adhesion influences on mesenchymal stem cell shape, mechanical properties, and differentiation down osteogenic, adipogenic, and chondrogenic pathways. Tissue Eng. Part B Rev. 2012, 18, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.-H.; Hwang, E.S.; McManus, M.T.; Amsterdam, A.; Tian, Y.; Kalmukova, R.; Mueller, E.; Benjamin, T.; Spiegelman, B.M.; Sharp, P.A.; et al. TAZ, a Transcriptional Modulator of Mesenchymal Stem Cell Differentiation. Science 2005, 309, 1074–1078. [Google Scholar] [CrossRef]
- DuPont, S.; Morsut, L.; Aragona, M.; Enzo, E.; Giulitti, S.; Cordenonsi, M.; Zanconato, F.; Le Digabel, J.; Forcato, M.; Bicciato, S.; et al. Role of YAP/TAZ in mechanotransduction. Nature 2011, 474, 179–183. [Google Scholar] [CrossRef]
- Cui, C.B.; Cooper, L.F.; Yang, X.; Karsenty, G.; Aukhil, I. Transcriptional coactivation of bone-specific transcription factor Cbfa1 by TAZ. Mol. Cell. Biol. 2003, 23, 1004–1013. [Google Scholar] [CrossRef]
- Stiehler, M.; Bünger, C.; Baatrup, A.; Lind, M.; Kassem, M.; Mygind, T. Effect of dynamic 3-D culture on proliferation, distribution, and osteogenic differentiation of human mesenchymal stem cells. J. Biomed. Mater. Res. A 2009, 89, 96–107. [Google Scholar] [CrossRef]
- Lienau, J.; Schmidt-Bleek, K.; Peters, A.; Weber, H.; Bail, H.J.; Duda, G.N.; Perka, C.; Schell, H. Insight into the molecular pathophysiology of delayed bone healing in a sheep model. Tissue Eng. Part A 2010, 16, 191–199. [Google Scholar] [CrossRef]
- Greenblatt, M.B.; Shim, J.-H. Osteoimmunology: A Brief Introduction. Immune Netw. 2013, 13, 111–115. [Google Scholar] [CrossRef]
- Van den Broek, L.J.; Kroeze, K.L.; Waaijman, T.; Breetveld, M.; Sampat-Sardjoepersad, S.C.; Niessen, F.B.; Middelkoop, E.; Scheper, R.J.; Gibbs, S. Differential response of human adipose tissue-derived mesenchymal stem cells, dermal fibroblasts, and keratinocytes to burn wound exudates: Potential role of skin-specific chemokine CCL27. Tissue Eng. Part A 2014, 20, 197–209. [Google Scholar] [CrossRef]
- Cornish, J.; Gillespie, M.T.; Callon, K.E.; Horwood, N.J.; Moseley, J.M.; Reid, I.R. Interleukin-18 is a novel mitogen of osteogenic and chondrogenic cells. Endocrinology 2003, 144, 1194–1201. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Zhang, Y.; Feng, W.; Chen, R.; Chen, J.; Touyz, R.M.; Wang, J.; Huang, H. Interleukin-18 Enhances Vascular Calcification and Osteogenic Differentiation of Vascular Smooth Muscle Cells Through TRPM7 Activation. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 1933–1943. [Google Scholar] [CrossRef] [PubMed]
- Pu, Y.; Wang, M.; Hong, Y.; Wu, Y.; Tang, Z. Adiponectin promotes human jaw bone marrow mesenchymal stem cell chemotaxis via CXCL1 and CXCL8. J. Cell Mol. Med. 2017, 21, 1411–1419. [Google Scholar] [CrossRef] [PubMed]
- Hardaway, A.L.; Herroon, M.K.; Rajagurubandara, E.; Podgorski, I. Marrow adipocyte-derived CXCL1 and CXCL2 contribute to osteolysis in metastatic prostate cancer. Clin. Exp. Metastasis 2015, 32, 353–368. [Google Scholar] [CrossRef]
- He, N.; Zhang, L.; Cui, J.; Li, Z. Bone marrow vascular niche: Home for hematopoietic stem cells. Bone Marrow Res. 2014, 2014, 128436. [Google Scholar] [CrossRef]
- Gadelmawla, E.S.; Koura, M.M.; Maksoud, T.M.A.; Elewa, I.M.; Soliman, H.H. Roughness parameters. J. Mater. Process. 2002, 123, 133–145. [Google Scholar] [CrossRef]
- Gnedenkov, S.V.; Scharkeev, Y.P.; Sinebryukhov, S.L.; Khrisanfova, O.A.; Legostaeva, E.V.; Zavidnaya, A.G.; Puz’, A.V.; Khlusov, I.A. Formation and properties of bioactive surface layers on titanium. Inorg. Mater. Appl. Res. 2011, 2, 474–481. [Google Scholar] [CrossRef]
- Association (WMA), W.M. Declaration of Helsinki. Ethical Principles for Medical Research Involving Human Subjects. Jahrb. Wiss. Ethik 2009, 14, 233–238. [Google Scholar] [CrossRef]
- Zuk, P.A.; Zhu, M.; Mizuno, H.; Huang, J.; Futrell, J.W.; Katz, A.J.; Benhaim, P.; Lorenz, H.P.; Hedrick, M.H. Multilineage cells from human adipose tissue: Implications for cell-based therapies. Tissue Eng. 2001, 7, 211–228. [Google Scholar] [CrossRef]
| Parameters of One Surface of a Bilateral CaP Coating on a Titanium Substrate, n = 7 | Number of Viable Cells % | Number of Apoptotic cells % | Number of Necrotic cells % | Stem Cell Markers, % | Hemato-poietic Cell Markers% | ||||
|---|---|---|---|---|---|---|---|---|---|
| Ra µm | Thickness µm | Weight mg | CD73 | CD90 | CD105 | [CD45,34,14,20] | |||
| Mesenchymal stem cell (MSC) culture on plastic (2D control), n = 3 1 | |||||||||
| − | − | − | 92.54 (91.02–95.39) | 1.54 (0.84–2.68) | 5.02 (4.01–6.3) | 95.31 (91.74–95.73) | 98.57 (98.09–98.79) | 98.86 (98.66–99.14) | 0.36 (0.33–0.37) |
| hAMSC culture on plastic in contact with the CaP-coated titanium substrates | |||||||||
| 3.1 (2.4–3.3) | 46.0 (35–53.5) | 13.0 (11.2–14.0) | 91.77 (91.47–93.14) | 3.22 (1.43–4.20) | 4.33 (3.64–6.8) | 93.70 (85.71–93.73) | 96.80 (92.83–97.31) | 97.05 (93.60–97.74) | 0.63 2 (0.56–0.63) |
| Parameters of One Surface of a Bilateral CaP Coating on a Titanium Substrate, n = 3 | Indices of Alizarin Red S Staining | |||
|---|---|---|---|---|
| Ra µm | Thickness µm | Weight mg | The Number of the Sites of Cell Culture Mineralization per Well | An Average Area of the Mineralization Sites, mm2 |
| MSC culture on plastic (2D control), n = 4 1 | ||||
| − | − | − | 0 | 0 |
| hAMSC culture on plastic in contact with the CaP-coated titanium substrates | ||||
| 2.49 (2.20; 2.90) | 45.5 (33.0; 56.5) | 11.8 (9.6; 13.8) | 156 (155–208) 2 | 0.0037 (0.0034–0.0043) 2 |
| Parameters of One Surface of a Bilateral CaP Coating on a Titanium Substrate n = 7 1 | Relative Expression of Osteogenic Genes | |||||||
|---|---|---|---|---|---|---|---|---|
| Ra µm | Thickness µm | Weight mg | hTERT | RUNX2 | BMP2 | BMP6 | BGLAP | ALPL |
| 3.1 (2.4; 3.3) | 51.5 (37.0; 53.5) | 13.1 (11.0; 14.4) | 6.9 2 (6.4; 56.5) | 1.44 2 (1.07; 1.69) | 1.05 (−1.40; 1.57) | 1.40 2 (1.04; 1.69) | −0.92 (−1.41; 1.69) | 1.43 2 (1.03; 1.63) |
| Relative expression of cytokine and chemokine genes | ||||||||
| IL-18 | CXCL1 | CCL27 | CCL7 | CXCL12 | CLEC11A | |||
| −1.47 2 (−3.52; −1.22) | −1.16 2 (−1.24; −1.13) | −3.10 2 (−3.28; −1.84) | 3.11 2 (2.33; 3.51) | 2.24 (−3.52; 4.31) | −2.10 (−2.44; 3.79) | |||
| Factors | hAMSC Culture on Plastic, n = 3 | hAMSC Cultures in Contact with the CaP-coated Titanium Substrates, n = 7 | |
|---|---|---|---|
| Surface parameters of a CaP coating | Ra, µm | 0 | 3.1 (2.4–3.3) |
| Thickness, µm | 0 | 46.0 (35–53.5) | |
| Weight, mg | 0 | 13.0 (11.2–14.0) | |
| Inflammatory interleukins and cytokines | IL-1α | 0.68 (0.65–1.10) | 0.77 (0.69–0.85) |
| IL-2Ra | 17.84 (10.23–27.63) | 8.48 (8.48–12.8) | |
| IL-3 | 5. 54 (2.51–6.01) | 3.29 (2.38–7.54) | |
| IL-12 (p40) | 14.62 (14.62–54.73) | 38.79 (12.78–52.11) | |
| IL-16 | 33.19 (21.52–47.69) | 18.22 (15.87–26.61) | |
| IL-18 | 6.40 (3.43–7.21) | 8.09 * (7.53–11.13) | |
| TNFβ | 0.49 (0.42–0.82) | 0.55 (0.21–0.57) | |
| IFNα2 | 17.10 (17.10–18.41) | 16.54 (15.91–17.45) | |
| Growth factors | M-CSF | 15.55 (13.47–19.80) | 18.20 (11.42–20.03) |
| β-NGF | 8.21 (6.55–8.61) | 7.47 (5.13–8.33) | |
| HGF | 321 (299–413) | 178 * (162–192) | |
| Chemokines | LIF | 14.43 (11.03–16.03) | 10.30 * (9.33–10.32) |
| MCP-3(CCL7) | 95.57 (45.26–112.16) | 60.18 (57.82–66.99) | |
| MIF | 536 (405–966) | 492 (254–667) | |
| MIG (CXCL9) | 12.16 (8.97–12.80) | 8.97 (8.97–14.06) | |
| GROα (CXCL1) | 36.77 (35.68–38.92) | 42.34 * (39.21–49.13) | |
| SCF | 3.14 (2.18–4.45) | 11.49 * (6.82–11.59) | |
| SCGF-b | 12838 (11657–13045) | 11629 (9997–15633) | |
| SDF-1α (CXCL12) | 81.84 (43.22–101.84) | 70.37 (49.40–76.90) | |
| TRAIL | 4.04 (3.04–4.37) | 3.21 (2.71–4.20) | |
| CTACK (CCL27) | 34.49 (32.30–43.92) | 40.40 (40.40–58.65) | |
| RUNX2_for 5′-CCAGAAGGCACAGACAGAAG-3′ |
| RUNX2_rev 5′-GATGAGGAATGCGCCCTAAA-3′ |
| BMP2_for 5′-ACGAGGTCCTGAGCGAGTT-3′ |
| BMP2_rev 5′-GACCTGAGTGCCTGCGATAC-3′ |
| BMP6_for 5′-TTACAGGAGCATCAGCACAG-3′ |
| BMP6_rev 5′-GGAGTCACAACCCACAGATT-3′ |
| BGP_for5′- GAGGGTATAAACAGTGCTGGAG-3′ |
| BGP_rev5′-AATAGGGCGAGGAGTGTGA-3′ |
| ALP_for5′-GGGAAATCTGTGGGCATTGT-3′ |
| ALP_rev5′-GAGTACCAGTCCCGGTCAGC-3′ |
| RPLPO_for 5′-GGCGACCTGGAAGTCCAACT-3′ |
| RPLPO_rev 5′-CCATCAGCACCACAGCCTTC-3′ |
| RUNX2_probe 5′-FAM-AGTTTGTTCTCTGACCGCCTCAGT--BHQ1-3′ |
| BMP2_probe 5′-FAM-CTGAAACAGAGACCCACCCCCAGCA-BHQ1-3′ |
| BMP6_probe 5′-FAM-CCTCAGAAGAAGGCTGGCTGGAAT-BHQ1 -3′ |
| BGP_probe 5′-FAM-CAGCCACCGAGACACCATGAGA-BHQ1-3′ |
| ALP_probe 5′-FAM-ACCACGAGAGTGAACCATGCCA-BHQ1-3′ |
| RPLPO_probe Bgl635-5′-ATCTGCTGCATCTGCTTGGAGCCCA-3′-BHQ-2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Litvinova, L.; Yurova, K.; Shupletsova, V.; Khaziakhmatova, O.; Malashchenko, V.; Shunkin, E.; Melashchenko, E.; Todosenko, N.; Khlusova, M.; Sharkeev, Y.; et al. Gene Expression Regulation and Secretory Activity of Mesenchymal Stem Cells upon In Vitro Contact with Microarc Calcium Phosphate Coating. Int. J. Mol. Sci. 2020, 21, 7682. https://doi.org/10.3390/ijms21207682
Litvinova L, Yurova K, Shupletsova V, Khaziakhmatova O, Malashchenko V, Shunkin E, Melashchenko E, Todosenko N, Khlusova M, Sharkeev Y, et al. Gene Expression Regulation and Secretory Activity of Mesenchymal Stem Cells upon In Vitro Contact with Microarc Calcium Phosphate Coating. International Journal of Molecular Sciences. 2020; 21(20):7682. https://doi.org/10.3390/ijms21207682
Chicago/Turabian StyleLitvinova, Larisa, Kristina Yurova, Valeria Shupletsova, Olga Khaziakhmatova, Vladimir Malashchenko, Egor Shunkin, Elena Melashchenko, Natalia Todosenko, Marina Khlusova, Yurii Sharkeev, and et al. 2020. "Gene Expression Regulation and Secretory Activity of Mesenchymal Stem Cells upon In Vitro Contact with Microarc Calcium Phosphate Coating" International Journal of Molecular Sciences 21, no. 20: 7682. https://doi.org/10.3390/ijms21207682
APA StyleLitvinova, L., Yurova, K., Shupletsova, V., Khaziakhmatova, O., Malashchenko, V., Shunkin, E., Melashchenko, E., Todosenko, N., Khlusova, M., Sharkeev, Y., Komarova, E., Sedelnikova, M., & Khlusov, I. (2020). Gene Expression Regulation and Secretory Activity of Mesenchymal Stem Cells upon In Vitro Contact with Microarc Calcium Phosphate Coating. International Journal of Molecular Sciences, 21(20), 7682. https://doi.org/10.3390/ijms21207682








