Seminal Plasma Analysis of Oxidative Stress in Different Genitourinary Topographical Regions Involved in Reproductive Tract Disorders Associated with Genital Heat Stress
Abstract
1. Introduction
2. Results
2.1. Comparative Analysis of Biochemical Parameters among the Studied Groups
2.2. Comparative Analysis of Oxidative Stress Markers among the Studied Groups
2.3. Spearman Rank Order Correlations between Seminal Biochemical/Oxidative Stress Parameters and Standard Semen Characteristics in the Studied Groups
2.4. Spearman Rank Order Correlations between Oxidative Stress and Biochemical Parameters in Studied Groups
3. Discussion
4. Materials and Methods
4.1. Male Participants
4.2. Semen Collection and Processing
4.3. Determination of Biochemical Parameters in Semen Samples
4.3.1. Neutral Alpha-Glucosidase Activity Measurement in Seminal Plasma
4.3.2. Fructose Concentration Measurement in Seminal Plasma
4.3.3. Citric Acid Concentration Measurement in Seminal Plasma
4.4. Determination of Oxidative Stress Parameters in Semen Samples
4.4.1. Total Antioxidant Capacity (TAC) Measurement in Seminal Plasma
4.4.2. Superoxide Dismutase (SOD) Activity Measurement in Seminal Plasma
4.4.3. Catalase Activity Measurement in Seminal Plasma
4.4.4. Malondialdehyde (MDA) Concentration Measurement in Seminal Plasma
4.5. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mieusset, R.; Bujan, L. Testicular heating and its possible contributions to male infertility: A review. Int. J. Androl. 1995, 18, 169–184. [Google Scholar] [CrossRef] [PubMed]
- Durairajanayagam, D.; Agarwal, A.; Ong, C. Causes, effects and molecular mechanisms of testicular heat stress. Reprod. Biomed. Online 2015, 30, 14–27. [Google Scholar] [CrossRef] [PubMed]
- Fraczek, M.; Kamieniczna, M.; Budzinska, M.; Kurpisz, M. Hyperthermia and sperm quality—A risk factor for male infertility or contraceptive target. Adv. Androl. Online 2018, 5, 48–61. [Google Scholar]
- Sheehan, M.M.; Ramasamy, R.; Lamb, D.J. Molecular mechanisms involved in varicocele-associated infertility. J. Assist. Reprod. Genet. 2014, 31, 521–526. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cho, C.L.; Esteves, S.C.; Agarwal, A. Novel insights into the pathophysiology of varicocele and its association with reactive oxygen species and sperm DNA fragmentation. Asian J. Androl. 2016, 18, 186–193. [Google Scholar] [PubMed]
- Van Brakel, J.; Kranse, R.; de Muinck Keizer-Schrama, S.M.; Hendriks, A.E.; de Jong, F.H.; Bangma, C.H.; Hazebroek, F.W.; Dohle, G.R. Fertility potential in men with a history of congenital undescended testes: A long-term follow-up study. Andrology 2013, 1, 100–108. [Google Scholar] [CrossRef]
- Barthold, J.S.; Reinhardt, S.; Thorup, J. Genetic, Maternal, and Environmental Risk Factors for Cryptorchidism: An Update. Eur. J. Pediatr. Surg. 2016, 26, 399–408. [Google Scholar]
- Moretti, E.; Di Cairano, G.; Capitani, S.; Scapigliati, G.; Baccetti, B.; Collodel, G. Cryptorchidism and semen quality: A TEM and molecular study. J. Androl. 2007, 28, 194–199. [Google Scholar] [CrossRef]
- Rao, M.; Zhao, X.L.; Yang, J.; Hu, S.F.; Lei, H.; Xia, W.; Zhu, C.H. Effect of transient scrotal hyperthermia on sperm parameters, seminal plasma biochemical markers, and oxidative stress in men. Asian J. Androl. 2015, 17, 668–675. [Google Scholar]
- Sakamoto, Y.; Ishikawa, T.; Kondo, Y.; Yamaguchi, K.; Fujisawa, M. The assessment of oxidative stress in infertile patients with varicocele. BJU Int. 2008, 101, 1547–1552. [Google Scholar] [CrossRef]
- Abo El-Khair, S.M.; Gaballah, M.A.; Abdel-Gawad, M.M.; Ismail, S.R.M.; Elsamanoudy, A.Z. Spermatozoal fractalkine signaling pathway is upregulated in subclinical varicocele patients with normal seminogram and low-level leucospermia. Adv. Urol. 2017, 5674237. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, T.; Anis, T.; El Nashar, A.; Imam, H.; Osman, I. Seminal plasma reactive oxygen species-antioxidants relationship with varicocele grade. Andrologia 2012, 44, 66–69. [Google Scholar] [CrossRef] [PubMed]
- Misro, M.M.; Chaki, S.P.; Gautam, D.K. Germ cell death and their removal during initial stages of testicular ischemia and cryptorchidism: A comparative analysis. Indian J. Exp. Biol. 2005, 43, 1080–1087. [Google Scholar] [PubMed]
- Smith, R.; Kaune, H.; Parodi, D.; Madariaga, M.; Morales, I.; Ríos, R.; Castro, A. Extent of sperm DNA damage in spermatozoa from men examined for infertility. Relationship with oxidative stress. Rev. Med. Chil. 2007, 135, 279–286. [Google Scholar] [PubMed]
- Ishii, T.; Matsuki, S.; Iuchi, Y.; Okada, F.; Toyosaki, S.; Tomita, Y.; Ikeda, Y.; Fujii, J. Accelerated impairment of spermatogenic cells in SOD1-knockout mice under heat stress. Free Radic. Res. 2005, 39, 697–705. [Google Scholar] [CrossRef]
- Li, Y.C.; Hu, X.Q.; Xiao, L.J.; Hu, Z.Y.; Guo, J.; Zhang, K.Y.; Song, X.X.; Liu, Y.X. An oligonucleotide microarray study on gene expression profile in mouse testis of experimental cryptorchidism. Front. Biosci. 2006, 11, 2465–2482. [Google Scholar] [CrossRef]
- Vivas-Acevedo, G.; Lozano-Hernández, R.; Camejo, M.I. Varicocele decreases epididymal neutral α-glucosidase and is associated with alteration of nuclear DNA and plasma membrane in spermatozoa. BJU Int. 2014, 113, 642–649. [Google Scholar] [CrossRef]
- Zöpfgen, A.; Priem, F.; Sudhoff, F.; Jung, K.; Lenk, S.; Loening, S.A.; Sinha, P. Relationship between semen quality and the seminal plasma components carnitine, alpha-glucosidase, fructose, citrate and granulocyte elastase in infertile men compared with a normal population. Hum. Reprod. 2000, 15, 840–845. [Google Scholar] [CrossRef]
- Peña, P.; Risopatrón, J.; Villegas, J.; Miska, W.; Schill, W.B.; Sánchez, R. Alpha-glucosidase in the human epididymis: Topographic distribution and clinical application. Andrologia 2004, 36, 315–320. [Google Scholar] [CrossRef]
- Said, L.; Galeraud-Denis, I.; Carreau, S.; Saâd, A. Relationship between semen quality and seminal plasma components: Alpha-glucosidase, fructose and citrate in infertile men compared with a normospermic population of Tunisian men. Andrologia 2009, 41, 150–156. [Google Scholar] [CrossRef]
- Collodel, G.; Nerucci, F.; Signorini, C.; Iacoponi, F.; Moretti, E. Associations between biochemical components of human semen with seminal conditions. Syst. Biol. Reprod. Med. 2019, 65, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.Y.; Qiu, S.D.; Ma, X.N.; Yu, H.M.; Wu, Y.W. Effect of experimental varicocele on structure and function of epididymis in adolescent rats. Asian J. Androl. 2003, 5, 108–112. [Google Scholar] [PubMed]
- Trsinar, B.; Muravec, U.R. Fertility potential after unilateral and bilateral orchidopexy for cryptorchidism. World J. Urol. 2009, 27, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Lei, B.; Lv, D.; Zhou, X.; Zhang, S.; Shu, F.; Ding, Y.; Zhang, Y.; Mao, X. Biochemical hormone parameters in seminal and blood plasma samples correlate with histopathologic properties of testicular biopsy in azoospermic patients. Urology 2015, 85, 1074–1078. [Google Scholar] [CrossRef] [PubMed]
- Henkel, R.; Maass, G.; Schuppe, H.C.; Jung, A.; Schubert, J.; Schill, W.B. Seasonal changes of neutral alpha-glucosidase activity in human semen. J. Androl. 2006, 27, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Elzanaty, S.; Richthoff, J.; Malm, J.; Giwercman, A. The impact of epididymal and accessory sex gland function on sperm motility. Hum. Reprod. 2002, 17, 2904–2911. [Google Scholar] [CrossRef]
- Elzanaty, S.; Malm, J. Effects of ejaculation-to-analysis delay on levels of markers of epididymal and accessory sex gland functions and sperm motility. J. Androl. 2007, 28, 847–852. [Google Scholar] [CrossRef]
- Andrade-Rocha, F.T. Seminal fructose levels in male infertility: Relationship with sperm characteristics. Int. Urol. Nephrol. 1999, 31, 107–111. [Google Scholar] [CrossRef]
- Lay, M.F.; Richardson, M.E.; Boone, W.R.; Bodine, A.B.; Thurston, R.J. Seminal plasma and IVF potential. Biochemical constituents of seminal plasma of males from in vitro fertilization couples. J. Assist. Reprod. Genet. 2001, 18, 144–150. [Google Scholar] [CrossRef]
- Roychoudhury, S.; Sharma, R.; Sikka, S.; Agarwal, A. Diagnostic application of total antioxidant capacity in seminal plasma to assess oxidative stress in male factor infertility. J. Assist. Reprod. Genet. 2016, 33, 627–635. [Google Scholar] [CrossRef]
- Micheli, L.; Cerretani, D.; Collodel, G.; Menchiari, A.; Moltoni, L.; Fiaschi, A.I.; Moretti, E. Evaluation of enzymatic and non-enzymatic antioxidants in seminal plasma of men with genitourinary infections, varicocele and idiopathic infertility. Andrology 2016, 4, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Abd-Elmoaty, M.A.; Saleh, R.; Sharma, R.; Agarwal, A. Increased levels of oxidants and reduced antioxidants in semen of infertile men with varicocele. Fertil. Steril. 2010, 94, 1531–1534. [Google Scholar] [CrossRef] [PubMed]
- Zini, A.; Schlegel, P.N. Cu/Zn superoxide dismutase, catalase and glutathione peroxidase mRNA expression in the rat testis after surgical cryptorchidism and efferent duct ligation. J. Urol. 1997, 158, 659–663. [Google Scholar] [CrossRef]
- Fei, Q.J.; Zheng, X.M.; Li, S.W.; Zheng, H. The effect of antioxidant enzyme on germ cell apoptosis in cryptorchidism. Zhonghua Nan Ke Xue 2002, 8, 255–257. [Google Scholar] [PubMed]
- Shiraishi, K. Heat and oxidative stress in the germ line. In Studies on Men’s Health and Fertility. Oxidative Stress in Applied Basic Research and Clinical Practice; Agarwal, A., Aitken, R.J., Alvarez, J.G., Eds.; Humana Press: New York, NY, USA, 2012; pp. 149–178. [Google Scholar]
- Diemer, T.; Allen, J.A.; Hales, K.H.; Hales, D.B. Reactive oxygen disrupts mitochondria in MA-10 tumor Leydig cells and inhibits steroidogenic acute regulatory (StAR) protein and steroidogenesis. Endocrinology 2003, 144, 2882–2891. [Google Scholar] [CrossRef] [PubMed]
- Ren, L.; Medan, M.S.; Ozu, M.; Li, C.; Watanabe, G.; Taya, K. Effects of experimental cryptorchidism on sperm motility and testicular endocrinology in adult male rats. J. Reprod. Dev. 2006, 52, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.S.; Zhang, Z.H.; Jin, X.; Wei, P.; Hu, X.Q.; Chen, M.; Lu, C.L.; Lue, Y.H.; Hu, Z.Y.; Sinha Hikim, A.P.; et al. Dedifferentiation of adult monkey sertoli cells through activation of extracellularly regulated kinase 1/2 induced by heat treatment. Endocrinology 2006, 147, 1237–1245. [Google Scholar] [CrossRef]
- Shen, H.; Fan, X.; Zhang, Z.; Xi, H.; Ji, R.; Liu, Y.; Yue, M.; Li, Q.; He, J. Effects of elevated ambient temperature and local testicular heating on the expressions of heat shock protein 70 and androgen receptor in boar testes. Acta Histochem. 2019, 121, 297–302. [Google Scholar] [CrossRef]
- Damsgaard, J.; Joensen, U.N.; Carlsen, E.; Erenpreiss, J.; Blomberg Jensen, M.; Matulevicius, V.; Zilaitiene, B.; Olesen, I.A.; Perheentupa, A.; Punab, M.; et al. Varicocele is associated with impaired semen quality and reproductive hormone levels: A study of 7035 healthy young men from six european countries. Eur. Urol. 2016, 70, 1019–1029. [Google Scholar] [CrossRef]
- Redmon, J.B.; Drobnis, E.Z.; Sparks, A.; Wang, C.; Swan, S.H. Semen and reproductive hormone parameters in fertile men with and without varicocele. Andrologia 2019, 51, e13407. [Google Scholar] [CrossRef]
- Lima, T.F.N.; Frech, F.S.; Patel, P.; Blachman-Braun, R.; Ramasamy, R. Effect of microsurgical varicocelectomy on semen parameters, serum, and intratesticular testosterone levels. BJUI Compass. 2020, 1, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Collodel, G.; Moretti, E.; Micheli, L.; Menchiari, A.; Moltoni, L.; Cerretani, D. Semen characteristics and molonodialdehyde levels in men with different reproductive problems. Andrology 2015, 3, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, T.; Rashed, L.A.; Osman, I.; Marawan, M. Seminal plasma oxytocin and oxidative stress levels in infertile men with varicocele. Andrologia 2015, 47, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Koksal, I.T.; Usta, M.; Orhan, I.; Abbasoglu, S.; Kadioglu, A. Potential role of reactive oxygen species on testicular pathology associated with infertility. Asian J. Androl. 2003, 5, 95–99. [Google Scholar] [PubMed]
- Imamoğlu, M.; Bülbül, S.S.; Kaklikkaya, N.; Sarihan, H. Oxidative, inflammatory and immunologic status in children with undescended testes. Pediatr. Int. 2012, 54, 816–819. [Google Scholar] [CrossRef]
- Fraczek, M.; Hryhorowicz, M.; Gill, K.; Zarzycka, M.; Gaczarzewicz, D.; Jedrzejczak, P.; Bilinska, B.; Piasecka, M.; Kurpisz, M. The effect of bacteriospermia and leukocytospermia on conventional and nonconventional semen parameters in healthy young normozoospermic males. J. Reprod. Immunol. 2016, 118, 18–27. [Google Scholar] [CrossRef]
- Ahmad, G.; Agarwal, A.; Esteves, S.C.; Sharma, R.; Almasry, M.; Al-Gonaim, A.; AlHayaza, G.; Singh, N.; Al Kattan, L.; Sannaa, W.M.; et al. Ascorbic acid reduces redox potential in human spermatozoa subjected to heat-induced oxidative stress. Andrologia 2017, 49. [Google Scholar] [CrossRef]
- Zhao, J.; Dong, X.; Hu, X.; Long, Z.; Wang, L.; Liu, Q.; Sun, B.; Wang, Q.; Wu, Q.; Li, L. Zinc levels in seminal plasma and their correlation with male infertility: A systematic review and meta-analysis. Sci. Rep. 2016, 6, 22386. [Google Scholar] [CrossRef]
- Cao, Y.; Li, Y.S.; Li, Z.J.; Wang, F.; Li, C.M. Dietary zinc may attenuate heat-induced testicular oxidative stress in mice via up-regulation of Cu-Zn SOD. Genet. Mol. Res. 2015, 14, 16616–16626. [Google Scholar] [CrossRef]
- Li, Z.; Li, Y.; Zhou, X.; Cao, Y.; Li, C. Preventive effects of supplemental dietary zinc on heat-induced damage in the epididymis of boars. J. Therm. Biol. 2017, 64, 58–66. [Google Scholar] [CrossRef]
- Ghasemi, N.; Babaei, H.; Azizallahi, S.; Kheradmand, A. Effect of long-term administration of zinc after scrotal heating on mice spermatozoa and subsequent offspring quality. Andrologia 2009, 41, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Murawski, M.; Saczko, J.; Marcinkowska, A.; Chwiłkowska, A.; Gryboś, M.; Banaś, T. Evaluation of superoxide dismutase activity and its impact on semen quality parameters of infertile men. Folia Histochem. Cytobiol. 2007, 45, S123–S126. [Google Scholar] [PubMed]
- Yan, L.; Liu, J.; Wu, S.; Zhang, S.; Ji, G.; Gu, A. Seminal superoxide dismutase activity and its relationship with semen quality and SOD gene polymorphism. J. Assist. Reprod. Genet. 2014, 31, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.Y.; Sun, Y.L.; Chang, C.C.; Lee, Y.S.; Tsai, H.D.; Lin, C.S. Superoxide dismutase activities of spermatozoa and seminal plasma are not correlated with male infertility. J. Clin. Lab. Anal. 2002, 16, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Sanocka, D.; Miesel, R.; Jedrzejczak, P.; Chełmonska-Soyta, A.C.; Kurpisz, M. Effect of reactive oxygen species and the activity of antioxidant systems on human semen; association with male infertility. Int. J. Androl. 1997, 20, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Yeung, C.H.; Cooper, T.G.; De Geyter, M.; De Geyter, C.; Rolf, C.; Kamischke, A.; Nieschlag, E. Studies on the origin of redox enzymes in seminal plasma and their relationship with results of in-vitro fertilization. Mol. Hum. Reprod. 1998, 4, 835–839. [Google Scholar] [CrossRef]
- Zini, A.; Fischer, M.A.; Mak, V.; Phang, D.; Jarvi, K. Catalase-like and superoxide dismutase-like activities in human seminal plasma. Urol. Res. 2002, 30, 321–323. [Google Scholar]
- World Health Organization. WHO Laboratory Manual for the Examination and Processing of Human Semen, 5th ed.; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Eertmans, F.; Bogaert, V.; Van Poecke, T.; Puype, B. An improved neutral a-glucosidase assay for assessment of epididymal function-validation and comparison to the WHO Method. Diagnostics 2014, 4, 1–11. [Google Scholar] [CrossRef]
- World Health Organization. WHO Laboratory Manual for the Examination of Human Semen and Sperm-Mucus Interaction, 3rd ed.; Cambridge Press: Cambridge, UK, 1992. [Google Scholar]
- Marzec-Wróblewska, U.; Kamiński, P.; Lakota, P.; Szymański, M.; Wasilow, K.; Ludwikowski, G.; Kuligowska-Prusińska, M.; Odrowąż-Sypniewska, G.; Stuczyński, T.; Michałkiewicz, J. Zinc and iron concentration and SOD activity in human semen and seminal plasma. Biol. Trace Elem. Res. 2011, 143, 167–177. [Google Scholar] [CrossRef]
- Johansson, L.H.; Borg, L.A. A spectrophotometric method for determination of catalase activity in small tissue samples. Anal. Biochem. 1988, 174, 331–336. [Google Scholar] [CrossRef]
- Fraczek, M.; Wiland, E.; Piasecka, M.; Boksa, M.; Gaczarzewicz, D.; Szumala-Kakol, A.; Kolanowski, T.; Beutin, L.; Kurpisz, M. Fertilizing potential of ejaculated human spermatozoa during in vitro semen bacterial infection. Fertil. Steril. 2014, 102, 711–719.e1. [Google Scholar] [CrossRef] [PubMed]
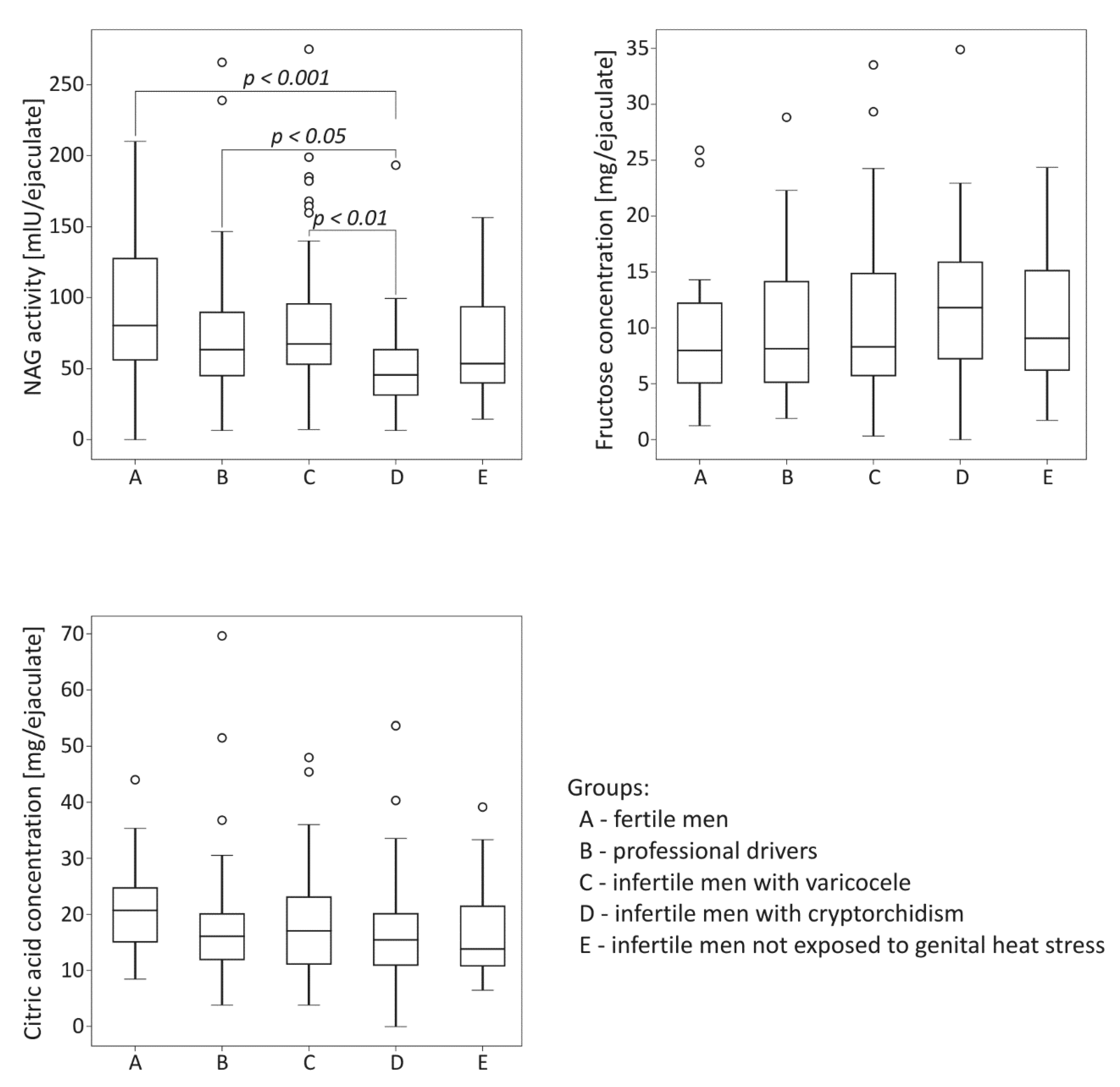
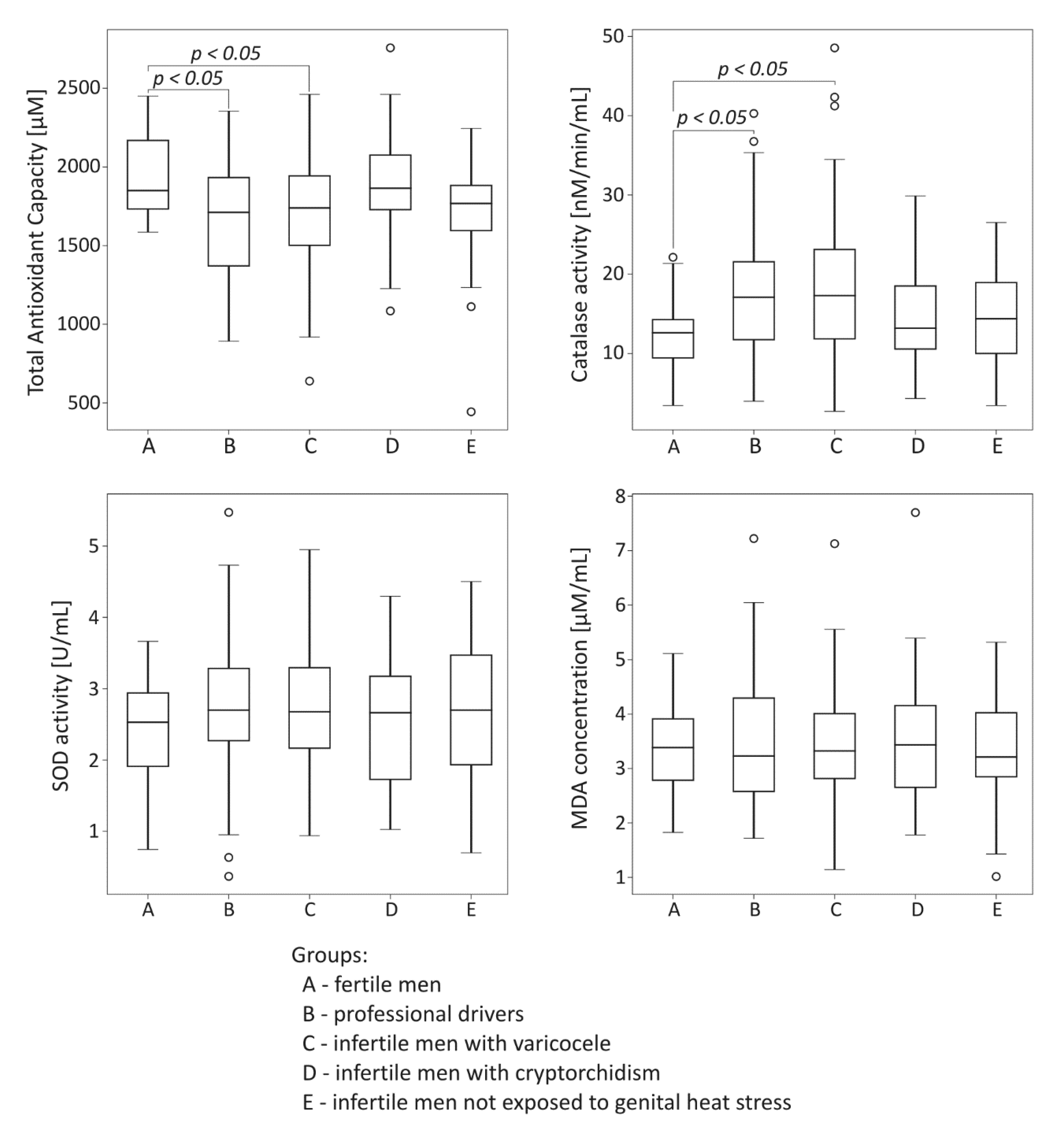
| Variables | R (Spearman) | p Level |
|---|---|---|
| Group of professional drivers | ||
| NAG vs. sperm count/mL | 0.322 | <0.05 |
| NAG vs. sperm count/ejaculate | 0.440 | <0.01 |
| NAG vs. HOS test | −0.344 | <0.05 |
| NAG vs. round cells | 0.330 | <0.05 |
| NAG vs. semen volume | 0.420 | <0.01 |
| Fructose vs. semen volume | 0.767 | <0.001 |
| Citric acid vs. semen volume | 0.596 | <0.001 |
| Group of infertile men with varicocele | ||
| NAG vs. sperm count/mL | 0.401 | <0.001 |
| NAG vs. sperm count/ejaculate | 0.419 | <0.001 |
| Fructose vs. semen volume | 0.686 | <0.001 |
| Citric acid vs. semen volume | 0.689 | <0.001 |
| Group of infertile men with cryptorchidism | ||
| Fructose vs. semen volume | 0.491 | <0.001 |
| Citric acid vs. semen volume | 0.415 | <0.01 |
| Group of infertile men not exposed to genital heat stress | ||
| NAG vs. sperm count/mL | 0.565 | <0.01 |
| NAG vs. sperm count/ejaculate | 0.580 | <0.001 |
| Fructose vs. semen volume | 0.836 | <0.001 |
| Citric acid vs. semen volume | 0.524 | <0.01 |
| Variables | R (Spearman) | p Level |
|---|---|---|
| Group of professional drivers | ||
| TAC vs. citric acid | −0.309 | <0.05 |
| NAG vs. fructose | 0.500 | <0.001 |
| NAG vs. citric acid | 0.379 | <0.01 |
| Fructose vs. citric acid | 0.342 | <0.05 |
| Group of infertile men with varicocele | ||
| TAC vs. NAG | −0.371 | <0.01 |
| TAC vs. citric acid | −0.307 | <0.01 |
| Catalase vs. MDA | 0.353 | <0.01 |
| Fructose vs. citric acid | 0.364 | <0.01 |
| Group of infertile men with cryptorchidism | ||
| TAC vs. citric acid | −0.383 | <0.01 |
| Fructose vs. citric acid | 0.366 | <0.05 |
| Group of infertile men not exposed to genital heat stress | ||
| SOD vs. NAG | −0.582 | <0.001 |
| Standard Semen Parameter | Control Group (Fertile Men) (n = 21) | Group of Professional Drivers (n = 52) | Group of Infertile Men with Varicocele (n = 50) | Group of Infertile Men with Cryptorchidism (n = 71) | Group of Infertile Men not Exposed to Genital Heat Stress (n = 32) |
|---|---|---|---|---|---|
| Volume [mL] | 2.95 ± 1.09 | 3.00 ± 1.05 | 3.40 ± 1.38 | 3.50 ± 1.40 | 3.30 ± 0.99 |
| pH | 8.00 ± 0.32 | 8.00 ± 0.31 | 8.00 ± 0.27 | 8.00 ± 0.29 | 8.00 ± 0.25 |
| Sperm concentration [×106/mL] | 90.98 ± 52.30 | 20.90 ± 21.12 | 18.00 ± 19.82 | 3.25 ± 15.42 | 18.15 ± 30.66 |
| Total number of spermatozoa [×106/ejaculate] | 272.10 ± 137.29 | 51.65 ± 78.83 | 52.50 ± 65.66 | 15.00 ± 49.75 | 57.20 ± 87.35 |
| Progressive motility of spermatozoa [%] | 52.50 ± 9.15 | 39.00 ± 14.54 | 32.00 ± 14.22 | 37.00 ± 13.98 | 33.00 ± 12.32 |
| Total sperm motility [%] | 63.00 ± 8.75 | 48.50 ± 13.95 | 46.00 ± 14.54 | 44.00 ± 13.71 | 44.00 ± 13.65 |
| Sperm viability [%] | 73.50 ± 8.50 | 68.50 ± 9.75 | 70.00 ± 12.76 | 65.00 ± 10.61 | 65.00 ± 15.58 |
| Spermatozoa with normal morphology [%] | 5.00 ± 2.59 | 1.50 ± 1.45 | 2.00 ± 1.44 | 1.00 ± 1.19 | 2.00 ± 1.17 |
| Peroxidase-positive leukocytes [×106/mL] | 0.14 ± 0.40 | 0.17 ± 0.32 | 0.14 ± 0.73 | 0.10 ± 0.31 | 0.08 ± 0.59 |
| Other round cells [×106/mL] | 0.80 ± 1.23 | 0.75 ± 1.43 | 0.74 ± 1.00 | 0.41 ± 0.49 | 0.63 ± 0.87 |
| HOS test [%] | 74.00 ± 5.00 | 68.00 ± 12.60 | 65.00 ± 12.69 | 64.00 ± 12.31 | 62.50 ± 14.00 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fraczek, M.; Wojnar, L.; Kamieniczna, M.; Piasecka, M.; Gill, K.; Kups, M.; Chopyak, V.; Havrylyuk, A.; Nakonechnyy, J.; Nakonechnyy, A.; et al. Seminal Plasma Analysis of Oxidative Stress in Different Genitourinary Topographical Regions Involved in Reproductive Tract Disorders Associated with Genital Heat Stress. Int. J. Mol. Sci. 2020, 21, 6427. https://doi.org/10.3390/ijms21176427
Fraczek M, Wojnar L, Kamieniczna M, Piasecka M, Gill K, Kups M, Chopyak V, Havrylyuk A, Nakonechnyy J, Nakonechnyy A, et al. Seminal Plasma Analysis of Oxidative Stress in Different Genitourinary Topographical Regions Involved in Reproductive Tract Disorders Associated with Genital Heat Stress. International Journal of Molecular Sciences. 2020; 21(17):6427. https://doi.org/10.3390/ijms21176427
Chicago/Turabian StyleFraczek, Monika, Lukasz Wojnar, Marzena Kamieniczna, Malgorzata Piasecka, Kamil Gill, Michal Kups, Valentina Chopyak, Anna Havrylyuk, Jozef Nakonechnyy, Andrij Nakonechnyy, and et al. 2020. "Seminal Plasma Analysis of Oxidative Stress in Different Genitourinary Topographical Regions Involved in Reproductive Tract Disorders Associated with Genital Heat Stress" International Journal of Molecular Sciences 21, no. 17: 6427. https://doi.org/10.3390/ijms21176427
APA StyleFraczek, M., Wojnar, L., Kamieniczna, M., Piasecka, M., Gill, K., Kups, M., Chopyak, V., Havrylyuk, A., Nakonechnyy, J., Nakonechnyy, A., Wozniak, T., & Kurpisz, M. (2020). Seminal Plasma Analysis of Oxidative Stress in Different Genitourinary Topographical Regions Involved in Reproductive Tract Disorders Associated with Genital Heat Stress. International Journal of Molecular Sciences, 21(17), 6427. https://doi.org/10.3390/ijms21176427







