Collagen/Chitosan Functionalization of Complex 3D Structures Fabricated by Laser Direct Writing via Two-Photon Polymerization for Enhanced Osteogenesis
Abstract
1. Introduction
2. Results and Discussion
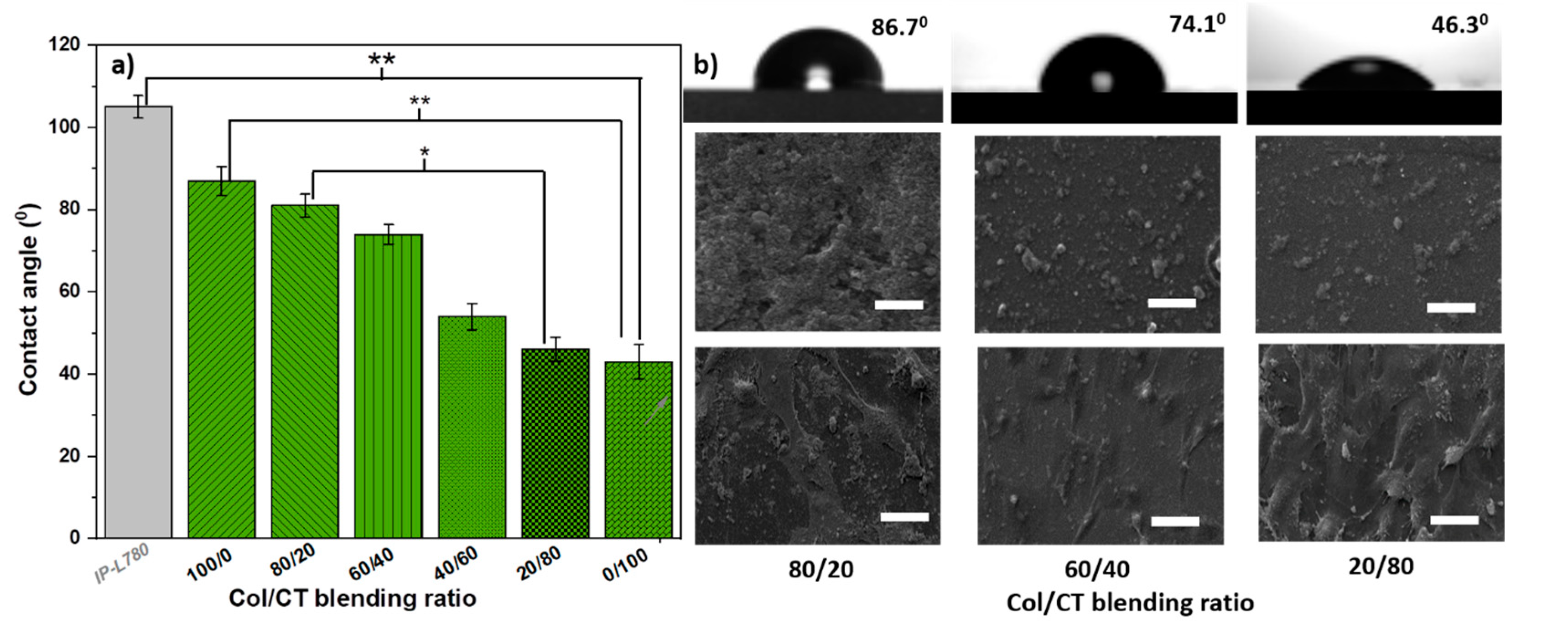
2.1. Design, Fabrication and Characterization of Functionalized 3D Structures
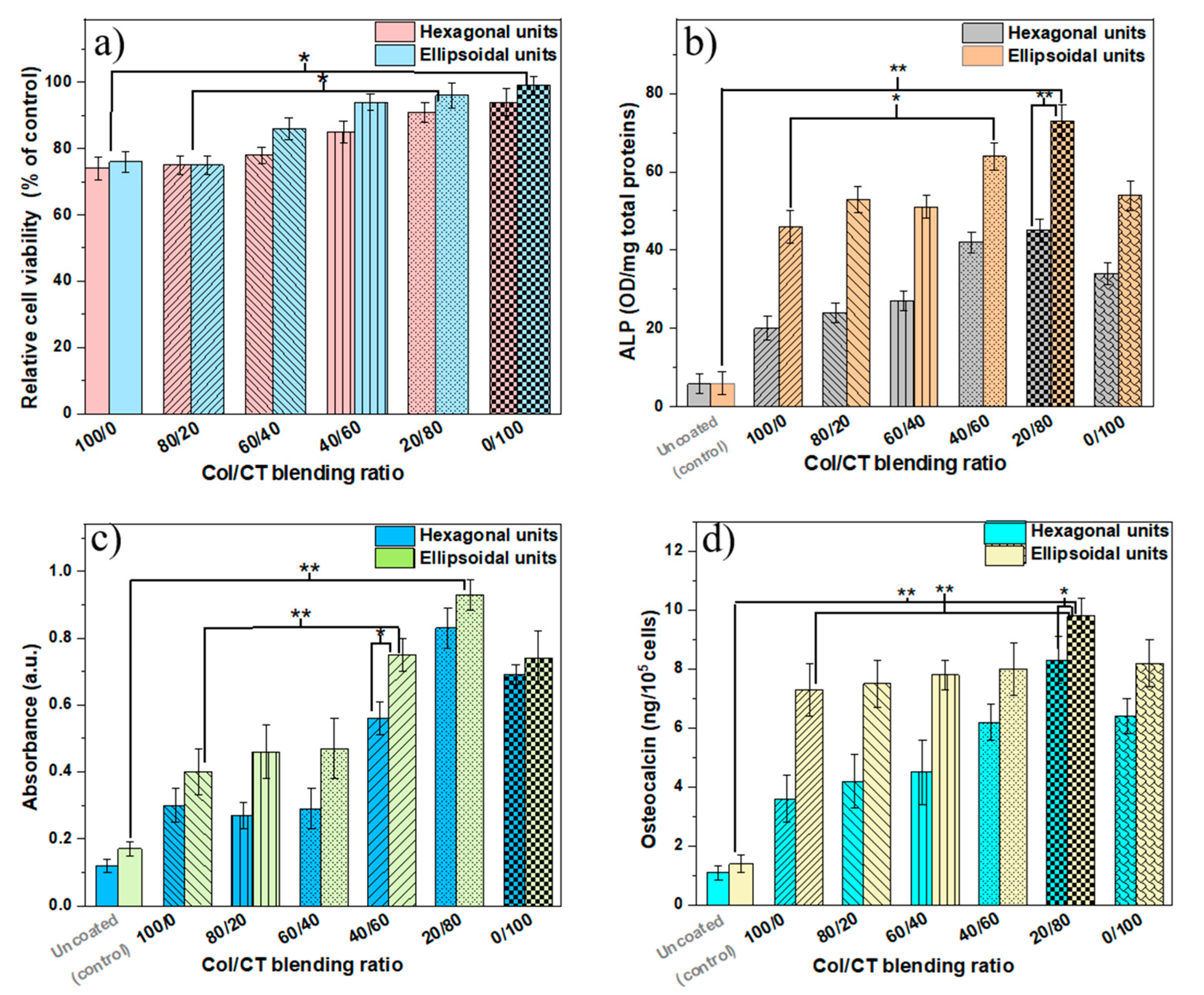
2.2. Biological Assessments of Functionalized 3D Structures
3. Materials and Methods
3.1. Materials
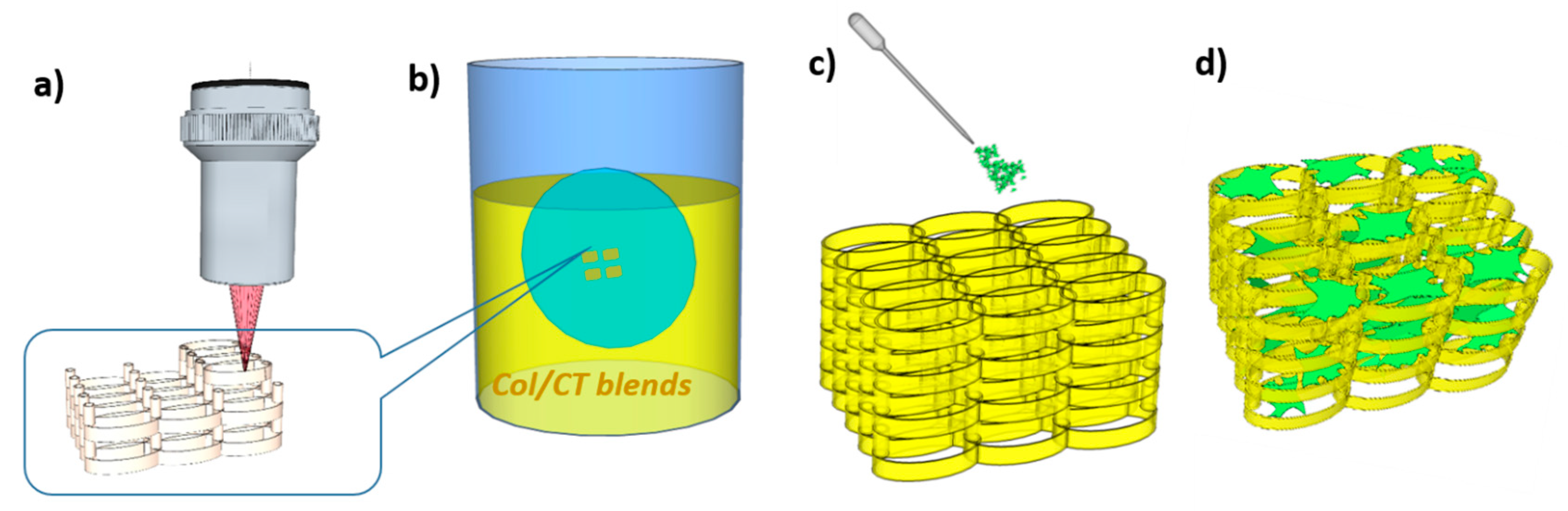
3.2. Fabrication of 3D Structures
3.2.1. Laser Direct Writing via Two-Photon Polymerization (LDW via TPP) of 3D Structures
3.2.2. Functionalization of 3D Structures
3.3. Characterization of 3D Structures
3.3.1. Morphological Investigations
3.3.2. Wettability
3.4. Biological Assessments
3.4.1. Cell Culture
3.4.2. Morphological Studies on Cells
3.4.3. Cells Viability
3.4.4. ALP Activity
3.4.5. Osteocalcin Secretion
3.4.6. Alizarin Red (ARS) Assay
3.4.7. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Col | Collagen |
| CT | Chitosan |
| ALP | Alkaline Phosphatase |
| AR | Alizarin Red |
| ARS | Alizarin Red Staining |
| LDW via TPP | Laser Direct Writing via Two-Photon Polymerization |
| SEM | Scanning Electron Microscopy |
| PBS | Phosphate Buffered Saline |
| EtOH | Ethanol |
| PNPP | P-Nitrophenyl Phosphate |
References
- Fisher, M.B.; Mauck, R.L. Tissue engineering and regenerative medicine: Recent innovations and the transition to translation. Tissue Eng. Part B Rev. 2013, 19, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Sola, A.; Bertacchini, J.; D’Avella, D.; Anselmi, L.; Maraldi, T.; Marmiroli, S.; Messori, M. Development of solvent-casting particulate leaching (SCPL) polymer scaffolds as improved three-dimensional supports to mimic the bone marrow niche. Mater. Sci. Eng. C 2019, 96, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Brougham, C.M.; Levingstone, T.J.; Shen, N.; Cooney, G.M.; Jockenhoevel, S.; Flanagan, T.C.; O’brien, F.J. Freeze-Drying as a Novel Biofabrication Method for Achieving a Controlled Microarchitecture within Large, Complex Natural Biomaterial Scaffolds. Adv. Healthc. Mater. 2017, 6, 1700598. [Google Scholar] [CrossRef] [PubMed]
- Song, P.; Zhou, C.; Fan, H.; Zhang, B.; Pei, X.; Fan, Y.; Jiang, Q.; Bao, R.; Yang, Q.; Dong, Z. Novel 3D porous biocomposite scaffolds fabricated by fused deposition modeling and gas foaming combined technology. Compos. Part B Eng. 2018, 152, 151–159. [Google Scholar] [CrossRef]
- Kumar, T.S.; Chakrapani, V.Y. Electrospun 3D Scaffolds for Tissue Regeneration. In Cutting-Edge Enabling Technologies for Regenerative Medicine; Chun, J.C., Park, C.H., Kwon, I.K., Khang, G., Eds.; Springer: Berlin, Germany, 2018; pp. 29–47. [Google Scholar] [CrossRef]
- Gay, S.; Lefebvre, G.; Bonnin, M.; Nottelet, B.; Boury, F.; Gibaud, A.; Calvignac, B. PLA scaffolds production from Thermally Induced Phase Separation: Effect of process parameters and development of an environmentally improved route assisted by supercritical carbon dioxide. J. Supercrit. Fluids 2018, 136, 123–135. [Google Scholar] [CrossRef]
- Tamay, D.G.; Usal, T.D.; Alagoz, A.S.; Yucel, D.; Hasirci, N.; Hasirci, V. 3D and 4D Printing of Polymers for Tissue Engineering Applications. Front. Bioeng. Biotechnol. 2019, 7, 164. [Google Scholar] [CrossRef]
- Peltola, S.M.; Melchels, F.P.; Grijpma, D.W.; Kellomäki, M. A review of rapid prototyping techniques for tissue engineering purposes. Ann. Med. 2008, 40, 268–280. [Google Scholar] [CrossRef]
- Raimondi, M.T.; Eaton, S.M.; Nava, M.M.; Lagana, M.; Cerullo, G.; Osellame, R. Two-photon laser polymerization: From fundamentals to biomedical application in tissue engineering and regenerative medicine. J. Appl. Biomater. Function. Mater. 2012, 10, 56–66. [Google Scholar] [CrossRef]
- Merkininkaitė, G.; Gailevičius, D.; Šakirzanovas, S.; Jonušauskas, L. Polymers for Regenerative Medicine Structures Made via Multiphoton 3D Lithography. Int. J. Polym. Sci. 2019, 2019, 3403548. [Google Scholar] [CrossRef]
- Baldacchini, T. Three-Dimensional Microfabrication Using Two-Photon Polymerization; Elsevier: Amsterdam, The Netherlands, 2020; ISBN 978-0-12-817827-0. [Google Scholar] [CrossRef]
- Cheng, C.; Gupta, M. Surface functionalization of 3D-printed plastics via initiated chemical vapor deposition. Beilstein J. Nanotechnol. 2017, 8, 1629–1636. [Google Scholar] [CrossRef]
- Păun, I.A.; Popescu, R.C.; Mustăciosu, C.C.; Zamfirescu, M.; Călin, B.S.; Mihăilescu, M.; Dinescu, M.; Popescu, A.; Chioibaşu, D.; Soproniy, M.; et al. Laser-direct writing by two-photon polymerization of 3D honeycomb-like structures for bone regeneration. Biofabrication 2018, 10, 025009. [Google Scholar] [CrossRef] [PubMed]
- Paun, I.A.; Popescu, R.C.; Calin, B.S.; Mustaciosu, C.C.; Dinescu, M.; Luculescu, C.R. 3D Biomimetic Magnetic Structures for Static Magnetic Field Stimulation of Osteogenesis. Int. J. Mol. Sci. 2018, 19, 495. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.K.; Narayan, R. Two-photon polymerization for biological applications. Mater. Today 2017, 20, 314–322. [Google Scholar] [CrossRef]
- Hong, J.M.; Kim, B.J.; Shim, J.-H.; Kang, K.S.; Kim, K.-J.; Rhie, J.W.; Cha, H.J.; Cho, D.-W. Enhancement of bone regeneration through facile surface functionalization of solid freeform fabrication-based three-dimensional scaffolds using mussel adhesive proteins. Acta Biomater. 2012, 8, 2578–2586. [Google Scholar] [CrossRef]
- Wang, X.; Guo, Q.; Cai, X.; Zhou, S.; Kobe, B.; Yang, J. Initiator-Integrated 3D Printing Enables the Formation of Complex Metallic Architectures. ACS Appl. Mater. Interfaces 2014, 6, 2583–2587. [Google Scholar] [CrossRef]
- Mano, J.; Silva, G.; Azevedo, H.; Malafaya, P.; Sousa, R.; Silva, S.; Reis, R. Natural origin biodegradable systems in tissue engineering and regenerative medicine: Present status and some moving trends. J. R. Soc. Interface 2007, 4, 999–1030. [Google Scholar] [CrossRef]
- Weinberg, C.B.; Bell, E. A blood vessel model constructed from collagen and cultured vascular cells. Science 1986, 231, 397–400. [Google Scholar] [CrossRef]
- Hoemann, C.D.; Sun, J.; Legare, A.; Mckee, M.D.; Buschmann, M.D. Tissue engineering of cartilage using an injectable and adhesive chitosan-based cell-delivery vehicle. Osteoarth. Cartil. 2005, 13, 318–329. [Google Scholar] [CrossRef]
- Nguyen, D.T.; McCanless, J.D.; Mecwan, M.M.; Noblett, A.P.; Haggard, W.O.; Smith, R.A.; Bumgardner, J.D. Balancing mechanical strength with bioactivity in chitosan-calcium phosphate 3D microsphere scaffolds for bone tissue engineering: Air- vs. freeze-drying processes. J. Biomater. Sci. Polym. Ed. 2013, 24, 1071–1083. [Google Scholar] [CrossRef]
- Seol, Y.J.; Lee, J.Y.; Park, Y.J.; Lee, Y.M.; Young, K.; Rhyu, I.C.; Lee, S.J.; Han, S.B.; Chung, C.P. Chitosan sponges as tissue engineering scaffolds for bone formation. Biotechnol. Lett. 2004, 26, 1037–1041. [Google Scholar] [CrossRef]
- Salasznyk, R.M.; Williams, W.A.; Boskey, A.; Batorsky, A.; Plopper, G.E. Adhesion to vitronectin and collagen I promotes osteogenic differentiation of human mesenchymal stem cells. J. Biomed. Biotechnol. 2004, 24–34. [Google Scholar] [CrossRef]
- Panduranga, K.R. Recent developments of collagen-based materials for medical applications and drug delivery systems. J. Biomat. Sci. Polym. Ed. 1996, 7, 623–645. [Google Scholar] [CrossRef] [PubMed]
- Paun, I.A.; Calin, B.S.; Mustaciosu, C.C.; Mihailescu, M.; Moldovan, A.; Crisan, O.; Leca, A.; Luculescu, C.R. 3D superparamagnetic Scaffolds for Bone Mineralization under Static Magnetic Field Stimulation. Materials 2019, 12, 2834. [Google Scholar] [CrossRef] [PubMed]
- Ebnesajjad, S.; Landrock, A.H. Adhesives Technology Handbook, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Molavi, A.M.; Avalshahr, A.S.; Nokhasteh, S.; Meshkin, H.N. Enhanced biological properties of collagen/chitosan coated poly(ε caprolactone) scaffold by surface modification with GHK Cu peptide and 58S bioglass. Prog. Biomater. 2020, 9, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Elliott, J.T.; Woodward, J.T.; Umarji, A.; Mei, Y.; Tona, A. The effect of surface chemistry on the formation of thin films of native fibrillar collagen. Biomaterials 2007, 28, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Dowling, D.P.; Miller, I.S.; Ardhaoui, M.; Gallagher, W.M. Effect of Surface Wettability and Topography on the Adhesion of Osteosarcoma Cells on Plasma-modified Polystyrene. J. Biomater. Appl. 2011, 26, 327–347. [Google Scholar] [CrossRef]
- Menzies, K.L.; Jones, L. The impact of contact angle on the biocompatibility of biomaterials. Optom. Vis. Sci. 2010, 87, 387–399. [Google Scholar] [CrossRef]
- Meng, J.; Yang, G.; Liu, L.; Son, Y.; Jiang, L.; Wang, S. Cell adhesive spectra along surface wettability gradient from superhydrophilicity to superhydrophobicity. Sci. China Chem. 2017, 60, 614–620. [Google Scholar] [CrossRef]
- Bumgardner, J.; Wiser, R.; Elder, S.H.; Jouett, R.; Yang, Y.; Ong, J.L. Contact angle, protein adsorption and osteoblast precursor cell attachment to chitosan coatings bonded to titanium. J. Biomater. Sci. Polym. Ed. 2003, 14, 1401–1409. [Google Scholar] [CrossRef]
- Gittens, R.A.; Scheideler, L.; Rupp, F.; Hyzy, S.L.; Geis-Gerstorfer, J.; Schwartz, Z.; Boyan, B.D. A review on the wettability of dental implant surfaces II: Biological and clinical aspects. Acta Biomater. 2014, 10, 2907–2918. [Google Scholar] [CrossRef]
- International Organization for Standardization. ISO 10993-12:2012 (E)—Biological Evaluation of Medical Devices, Part 12: Sample Preparation and Reference Materials; ISO: Geneva, Switzerland, 2012. [Google Scholar]
- Zernik, J.; Twarog, K.; Upholt, W.B. Regulation of alkaline phosphatase and alpha2(I) procollagen synthesis during early intramembranous bone formation in the rat mandible. Differentiation 1990, 44, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Kim, M.G.; Byun, J.H.; Kim, G.C.; Ro, J.-H.; Hwang, D.-S.; Choi, B.-B.; Park, G.-C.; Kim, U.-K. The effect of biomechanical stimulation on osteoblast differentiation of human jaw periosteum-derived stem cells. Maxillofac. Plast. Reconstr. Surg. 2017, 39, 7. [Google Scholar] [CrossRef]
- Delmas, P.; Eastell, R.; Garnero, P.; Seibel, M.J.; Stepan, J. The Use of Biochemical Markers of Bone Turnover in Osteoporosis. Osteoporos. Int. 2000, 11, S2–S17. [Google Scholar] [CrossRef]
- Tsao, Y.T.; Huang, Y.J.; Wu, H.H.; Liu, Y.A.; Liu, Y.S.; Lee, O.K. Osteocalcin Mediates Biomineralization during Osteogenic Maturation in Human Mesenchymal Stromal Cells. Int. J. Mol. Sci. 2017, 18, 159. [Google Scholar] [CrossRef] [PubMed]
- Muzzarelli, R.A.; Mattioli-Belmonte, M.; Tietz, C.; Biagini, R.; Ferioli, G.; Brunelli, M.A.; Fini, M.; Giardino, R.; Ilari, P.; Biagini, G. Stimulatory effect on bone formation exerted by a modified chitosan. Biomaterials 1994, 15, 1075–1081. [Google Scholar] [CrossRef]
- Amidi, M.; Mastrobattista, E.; Jiskoot, W.; Hennink, W.E. Chitosan-based delivery systems for protein therapeutics and antigens. Adv. Drug Deliv. Rev. 2010, 62, 59–82. [Google Scholar] [CrossRef]
- Dai, T.; Tanaka, M.; Huang, Y.Y.; Hamblin, M.R. Chitosan preparation for wounds and burns: Antimicrobial and wound-healing effects. Expert Ref. Anti Infect. Ther. 2011, 9, 857–879. [Google Scholar] [CrossRef]
- Levengood, S.L.; Zhang, M. Chitosan-based scaffolds for bone tissue engineering. J. Mater. Chem. B Mater. Biol. Med. 2014, 2, 3161–3184. [Google Scholar] [CrossRef]
- Zugravu, M.V.; Smith, R.A.; Reves, B.T.; Jennings, J.A.; Cooper, J.O.; Haggard, W.O.; Bumgardner, J.D. Physical properties and in vitro evaluation of collagen chitosan-calcium phosphate microparticle-based scaffolds for bone tissue regeneration. J. Biomater. Appl. 2013, 28, 566–579. [Google Scholar] [CrossRef]
- Fonseca, M.J.; Alsina, M.A.; Reig, F. Coating liposomes with collagen (Mr 50000) increases uptake into liver. Biochim. Biophys. Acta 1996, 1279, 259–265. [Google Scholar] [CrossRef][Green Version]
- Maeda, M.; Tani, S.; Sano, A.; Fujioka, K. Microstructure and release characteristics of the minipellet, a collagen based drug delivery system for controlled release of protein drugs. J. Control. Release 1999, 62, 313–324. [Google Scholar] [CrossRef]
- Friess, W. Collagen-biomaterial for drug delivery. Eur. J. Pharm. Biopharm. 1998, 45, 113–136. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Păun, I.A.; Mustăciosu, C.C.; Popescu, R.C.; Călin, B.Ş.; Mihăilescu, M. Collagen/Chitosan Functionalization of Complex 3D Structures Fabricated by Laser Direct Writing via Two-Photon Polymerization for Enhanced Osteogenesis. Int. J. Mol. Sci. 2020, 21, 6426. https://doi.org/10.3390/ijms21176426
Păun IA, Mustăciosu CC, Popescu RC, Călin BŞ, Mihăilescu M. Collagen/Chitosan Functionalization of Complex 3D Structures Fabricated by Laser Direct Writing via Two-Photon Polymerization for Enhanced Osteogenesis. International Journal of Molecular Sciences. 2020; 21(17):6426. https://doi.org/10.3390/ijms21176426
Chicago/Turabian StylePăun, Irina Alexandra, Cosmin Cătălin Mustăciosu, Roxana Cristina Popescu, Bogdan Ştefăniţă Călin, and Mona Mihăilescu. 2020. "Collagen/Chitosan Functionalization of Complex 3D Structures Fabricated by Laser Direct Writing via Two-Photon Polymerization for Enhanced Osteogenesis" International Journal of Molecular Sciences 21, no. 17: 6426. https://doi.org/10.3390/ijms21176426
APA StylePăun, I. A., Mustăciosu, C. C., Popescu, R. C., Călin, B. Ş., & Mihăilescu, M. (2020). Collagen/Chitosan Functionalization of Complex 3D Structures Fabricated by Laser Direct Writing via Two-Photon Polymerization for Enhanced Osteogenesis. International Journal of Molecular Sciences, 21(17), 6426. https://doi.org/10.3390/ijms21176426




