The Phenomenon of Clopidogrel High On-Treatment Platelet Reactivity in Ischemic Stroke Subjects: A Comprehensive Review
Abstract
1. Introduction
2. Molecular Mechanism of Action of Clopidogrel
3. Platelet Function Testing
4. The Prevalence of Clopidogrel High On-Treatment Platelet Reactivity
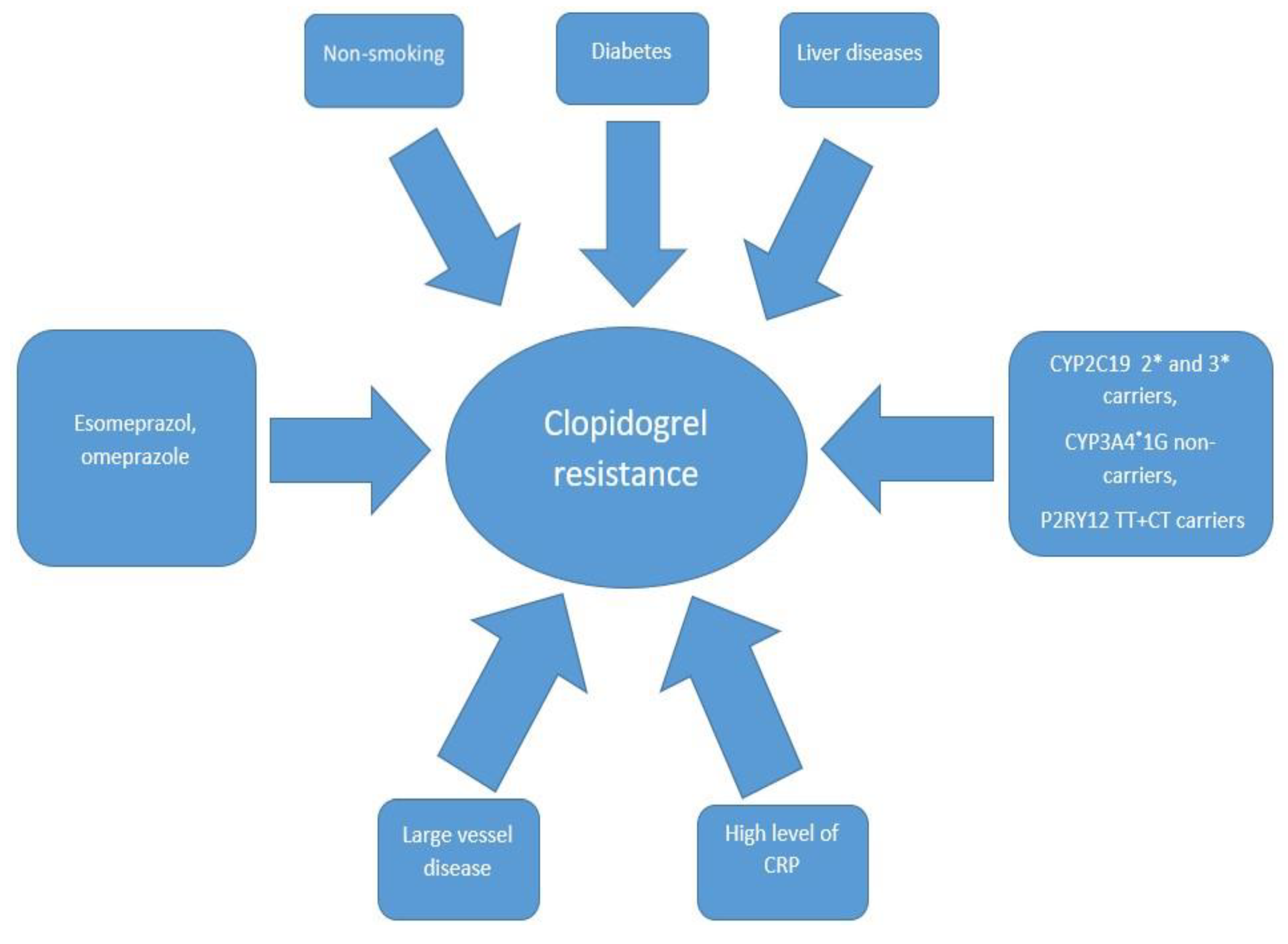
5. Factors Influencing Clopidogrel Resistance
5.1. Genetic Polymorphism
5.2. Concomitant Drugs
5.3. Risk Factors for Vascular Disease Development
5.4. Laboratory Findings
5.5. Etiology of Stroke
6. Impact of Clopidogrel Resistance on Clinical Evaluation in Ischemic Stroke
6.1. Recurrent Vascular Events
6.2. Clinical Outcomes
6.3. Intracranial Bleeding
7. Recommendations
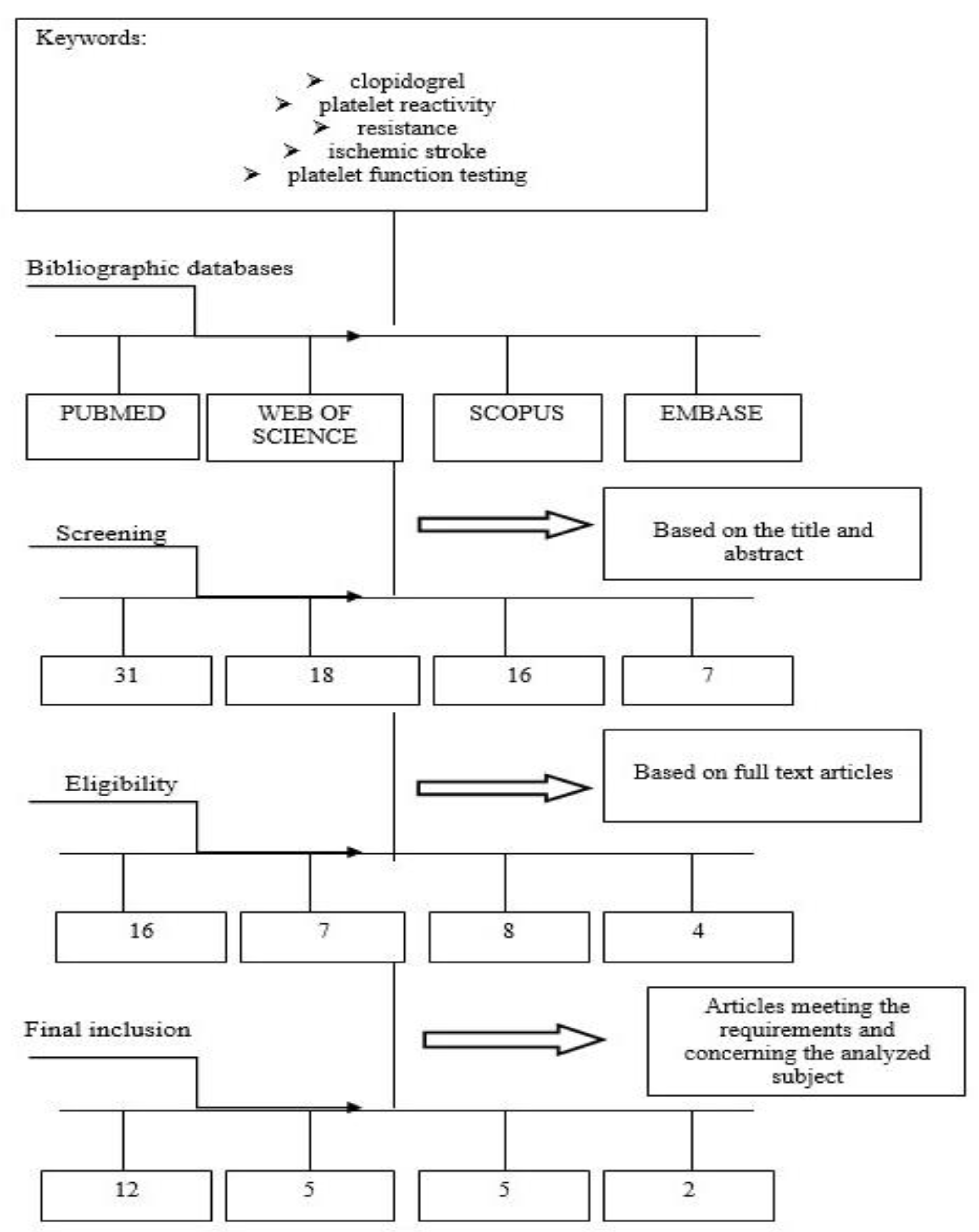
8. Materials and Methods
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ABCB1 | ABCB1 gene |
| ABCD2 | ABCD2 stroke scale |
| ADP | Adenosine diphosphate |
| CRP | C-reactive protein |
| CYP2C19 2* 3* | cytochrome P450 2C19 gene with loss-of-function 2* and 3* alleles |
| CYP3A4*1G | cytochrome P450 3A4 gene with 1G* allele |
| GPIIb/IIIa | glycoprotein IIb/IIIa complex |
| GPIIIa AA, GG, AG | glycoprotein IIIA gene with A or G alleles |
| HR | hazard ratio |
| LTA | light transmission aggregometry |
| MI | myocardial infarction |
| mRS | modified Rankin Scale |
| NIHSS | National Institute of Health Stroke Scale |
| OR | Odds ratio |
| P2RY12 H1 | P2Y12 receptor gene with H1 haplotype |
| P2RY12 H2 | P2Y12 receptor gene with H2 haplotype |
| P2RY12 TT+CT | P2Y12 receptor gene with T and C alleles |
| PFA | platelet function analyzer |
| PRU | platelet P2Y12 reactive units |
| TEG | thromboelastography |
| TIA | transient ischemic attack |
| VASP | vasodilator-stimulated phosphoprotein phosphorylation assay |
References
- Powers, W.J.; Rabinstein, A.A.; Ackerson, T.; Adeove, O.M.; Bambakidis, N.C.; Becker, K.; Biller, J.; Brwon, M.; Demaerschalk, B.M.; Hoh, B.; et al. Guidelines for the Early Management of Patients with Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke 2018, 49, e46–e110. [Google Scholar] [CrossRef] [PubMed]
- Kourlaba, G.; Fragoulakis, V.; Maniadakis, N. Clopidogrel versus Aspirin in Patients with Atherothrombosis. A CAPRIE-Based Cost-Effectiveness Model for Greece. Appl. Health Econ. Health Policy 2012, 10, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Fiolaki, A.; Katsanos, A.H.; Kyristis, A.P.; Papadaki, S.; Kosmidou, M.; Moschonas, I.C.; Tselepis, A.D.; Giannopoulos, S. High on treatment platelet reactivity to aspirin and clopidogrel in ischemic stroke: A systematic review and meta-analysis. J. Neurol. Sci. 2017, 376, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Hvas, A.-M.; Grove, E.L. Platelet function tests: Preanalytical variables, clinical utility, advantages, and disadvantages. Methods Mol. Biol. 2017, 1646, 305–320. [Google Scholar] [CrossRef] [PubMed]
- Leunissen, T.C.; Janssen, P.W.; Berg, J.M.T.; Moll, F.L.; Korporaal, S.J.; de Borst, G.J.; Pasterkamp, G.; Urbanus, R.T. The use of platelet reactivity testing in patients on antiplatelet therapy for prediction of bleeding events after cardiac surgery. Vasc. Pharmacol. 2016, 77, 19–27. [Google Scholar] [CrossRef]
- Ostrowska, M.; Kubica, J.; Adamski, P.; Kubica, A.; Eyileten, C.; Postula, M.; Toma, A.; Hengstenberg, C.; Siller-Matula, J.M. Stratified Approaches to Antiplatelet Therapies Based on Platelet Reactivity Testing. Front. Cardiovasc. Med. 2019, 6, 176. [Google Scholar] [CrossRef]
- Siller-Matula, J.M.; Petre, A.; Delle-Karth, G.; Huber, K.; Ay, C.; Lordkipanidzé, M.; de Ceterina, R.; Kolh, P.; Mahla, E.; Gersh, B.J. Impact of preoperative use of P2Y12 receptor inhibitors on clinical outcomes in cardiac and non-cardiac surgery: A systematic review and meta-analysis. Eur. Heart J. Acute Cardiovasc. Care 2017, 6, 753–770. [Google Scholar] [CrossRef]
- Mega, J.L.; Simon, T. Pharmacology of antithrombotic drugs: An assessment of oral antiplatelet and anticoagulant treatments. Lancet 2015, 386, 281–291. [Google Scholar] [CrossRef]
- Liu, R.; Zhou, Z.-Y.; Chen, Y.-B.; Li, J.-L.; Yu, W.-B.; Chen, X.-M.; Zhao, M.; Zhao, Y.-Q.; Cai, Y.-F.; Jin, J.; et al. Associations of CYP3A4, NR1I2, CYP2C19 and P2RY12 polymorphisms with clopidogrel resistance in Chinese patients with ischemic stroke. Acta Pharmacol. Sin. 2016, 37, 882–888. [Google Scholar] [CrossRef]
- Jiang, X.-L.; Samant, S.; Lesko, L.J.; Schmidt, S. Clinical pharmacokinetics and pharmacodynamics of clopidogrel. Clin. Pharmacokinet. 2015, 54, 147–166. [Google Scholar] [CrossRef]
- Hasan, M.S.; Basri, H.B.; Hin, L.P.; Stanslas, J. Genetic polymorphisms and drug interactions leading to clopidogrel resistance: Why the Asian population requires special attention. Int. J. Neurosci. 2012, 123, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Su, J.-F.; Hu, X.-H.; Li, C.-Y. Risk factors for clopidogrel resistance in patients with ischemic cerebral infarction and the correlation with ABCB1 gene rs1045642 polymorphism. Exp. Ther. Med. 2014, 9, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Kazui, M.; Nishiya, Y.; Ishizuka, T.; Hagihara, K.; Farid, N.A.; Okazaki, O.; Ikeda, T.; Kurihara, A. Identification of the human cytochrome P450 enzymes involved in the two oxidative steps in the bioactivation of clopidogrel to its pharmacologically active metabolite. Drug Metab. Dispos. 2009, 38, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Yi, X.; Wang, Y.; Lin, J.; Cheng, W.; Zhou, Q.; Wang, C. Interaction of CYP2C19, P2Y12, and GPIIIa Variants Associates With Efficacy of Clopidogrel and Adverse Events on Patients With Ischemic Stroke. Clin. Appl. Thromb. Hemost. 2016, 23, 761–768. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Chen, W.; Xu, Y.; Yi, X.; Han, Y.; Yang, Q.-W.; Li, X.; Huang, L.; Johnston, S.C.; Zhao, X.; et al. Genetic Polymorphisms and Clopidogrel Efficacy for Acute Ischemic Stroke or Transient Ischemic Attack: A Systematic Review and Meta-analysis. Circulation 2017, 135, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Jover, E.; Rodriguez, J.M.; Bernal, A.; Arroyo, A.B.; Iniesta, J.; Guiu, I.S.; Martinez, C.; Vicente, V.; Lozano, M.; Rivera, J. High on-treatment platelet reactivity in patients with ischemic cerebrovascular disease: Assessment of prevalence and stability over time using four platelet function tests. Blood Coagul. Fibrinolysis 2014, 25, 604–611. [Google Scholar] [CrossRef]
- Fu, H.; Hu, P.; Ma, C.; Peng, F.; He, Z. Association of clopidogrel high on-treatment reactivity with clinical outcomes and gene polymorphism in acute ischemic stroke patients. An observational study. Medicine 2020, 99, e19472. [Google Scholar] [CrossRef]
- Qiu, L.-N.; Wang, L.; Li, X.; Han, R.-F.; Xia, X.-S.; Liu, J. Predictive Value of High Residual Platelet Reactivity by Flow Cytometry for Outcomes of Ischemic Stroke Patients on Clopidogrel Therapy. J. Stroke Cerebrovasc. Dis. 2015, 24, 1145–1152. [Google Scholar] [CrossRef]
- Rao, Z.; Zheng, H.; Wang, F.; Wang, A.; Liu, L.; Dong, K.; Zhao, X.; Cao, Y.; Wang, Y. High On-Treatment Platelet Reactivity to Adenosine Diphosphate Predicts Ischemic Events of Minor Stroke and Transient Ischemic Attack. J. Stroke Cerebrovasc. Dis. 2017, 26, 2074–2081. [Google Scholar] [CrossRef]
- Orme, R.; Judge, H.M.; Storey, R.F. Monitoring Antiplatelet Therapy. Semin. Thromb. Hemost. 2017, 43, 311–319. [Google Scholar] [CrossRef]
- Yi, X.; Lin, J.; Wang, Y.; Zhou, J.; Zhou, Q.; Wang, C. Response to clopidogrel is associated with early neurological deterioration after acute ischemic stroke. Oncotarget 2018, 9, 19900–19910. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rath, C.L.; Joergensen, N.R.; Wienecke, T. High On-Treatment Platelet Reactivity in Danish Hyper-Acute Ischaemic Stroke Patients. Front. Neurol. 2018, 9, 712. [Google Scholar] [CrossRef] [PubMed]
- Gurbel, P.A.; Bliden, K.P.; Logan, D.K.; Kereiakes, D.J.; Lasseter, K.C.; White, A.; Angiolillo, D.J.; Nolin, T.D.; Maa, J.-F.; Bailey, W.L.; et al. The influence of smoking status on the pharmacokinetics and pharmacodynamics of clopidogrel and prasugrel: The PARADOX study. J. Am. Coll. Cardiol. 2013, 62, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Gwon, J.; Kim, M. Abstract WMP39: Clopidogrel Resistance Predicts Early Neurological Worsening In Patients With Acute Large Artery Atherosclerotic Stroke. Stroke 2018, 49, AWMP39. [Google Scholar] [CrossRef]
- Kinsella, J.A.; Tobin, W.O.; Cox, D.; Coughlan, T.; Collins, R.; O’Neill, D.; Murphy, R.P.; Mccabe, D.J.H. Prevalence of Ex Vivo High On-treatment Platelet Reactivity on Antiplatelet Therapy after Transient Ischemic Attack or Ischemic Stroke on the PFA-100® and VerifyNow. J. Stroke Cerebrovasc. Dis. 2013, 22, e84–e92. [Google Scholar] [CrossRef]
- Meves, S.H.; Schroeder, K.D.; Endres, H.G.; Krogias, C.; Krueger, J.C.; Neubauer, H. Clopidogrel high-on-treatment platelet reactivity in acute ischemic stroke Patients. Thromb. Res. 2014, 133, 396–401. [Google Scholar] [CrossRef]
- Wiśniewski, A.; Filipska, K.; Sikora, J.; Ślusarz, R.; Kozera, G. The Prognostic Value of High Platelet Reactivity in Ischemic Stroke Depends on the Etiology: A Pilot Study. J. Clin. Med. 2020, 9, 859. [Google Scholar] [CrossRef]
- Jia, D.-M.; Chen, Z.-B.; Zhang, M.-J.; Yang, W.-J.; Jin, J.-L.; Xia, Y.-Q.; Zhang, C.-L.; Shao, Y.; Chen, C.; Xu, Y. CYP2C19 polymorphisms and antiplatelet effects of clopidogrel in acute ischemic stroke in China. Stroke 2013, 44, 1717–1719. [Google Scholar] [CrossRef]
- Bhatt, D.L.; Pare, G.; Eikelboom, J.W.; Simonsen, K.L.; Emison, E.S.; Fox, K.A.A.; Steg, P.G.; Montalescot, G.; Bhakta, N.; Hacke, W.; et al. The relationship between CYP2C19 polymorphisms and ischaemic and bleeding outcomes in stable outpatients: The CHARISMA genetics study. Eur. Heart J. 2012, 33, 2143–2150. [Google Scholar] [CrossRef]
- Pare, G.; Mehta, S.R.; Yusuf, S.; Anand, S.S.; Connolly, S.J.; Hirsch, J.; Simonsen, K.; Bhatt, D.L.; Fox, K.A.A.; Eikelboom, J.W. Effects of CYP2C19genotype on outcomes of clopidogrel treatment. N. Engl. J. Med. 2010, 363, 1704–1714. [Google Scholar] [CrossRef]
- Holmes, M.W.V.; Perel, P.; Shah, T.; Hingorani, A.D.; Casas, J.P. CYP2C19 genotype, clopidogrel metabolism, platelet function, and cardiovascular events: A systematic review and meta-analysis. JAMA 2011, 306, 2704–2714. [Google Scholar] [CrossRef] [PubMed]
- Mega, J.L.; Close, S.L.; Wiviott, S.D.; Shen, L.; Walker, J.R.; Simon, T.; Antman, E.M.; Braunwald, E.; Sabatine, M.S.; Simone, T. Genetic variants in ABCB1 and CYP2C19 and cardiovascular outcomes after treatment with clopidogrel and prasugrel in the TRITON–TIMI 38 trial: A pharmacogenetic analysis. Lancet 2010, 376, 1312–1319. [Google Scholar] [CrossRef]
- Wallentin, L.; James, S.; Storey, R.F.; Armstrong, M.; Barratt, B.J.; Horrow, J.; Husted, S.; Katus, H.; Steg, P.G.; Shah, S.H.; et al. Effect of CYP2C19 and ABCB1 single nucleotide polymorphisms on outcomes of treatment with ticagrelor versus clopidogrel for acute coronary syndromes: A genetic substudy of the PLATO trial. Lancet 2010, 376, 1320–1328. [Google Scholar] [CrossRef]
- Galic, E.; Vrbanic, L.; Kapitanovic, S.; Ivkovic, T.C.; Petro, D.; Vukovic, I.; Bsharat, R.S.; Milicevic, Z.; Vcew, A.; Mirat, J. P2RY12 gene polymorphisms and effect of clopidogrel on platelet aggregation. Coll. Antropol. 2013, 37, 491–498. [Google Scholar]
- Staritz, P.; Kurz, K.; Stoll, M.; Giannitsis, E.; Katus, H.A.; Ivandic, B.T. Platelet reactivity and clopidogrel resistance are associated with the H2 haplotype of the P2Y12-ADP receptor gene. Int. J. Cardiol. 2009, 133, 341–345. [Google Scholar] [CrossRef]
- Lev, E.I.; Patel, R.T.; Guthikonda, S.; Lopez, D.; Bray, P.F.; Kleiman, N.S. Genetic polymorphisms of the platelet receptors P2Y(12), P2Y(1) and GP IIIa and response to aspirin and clopidogrel. Thromb. Res. 2007, 119, 355–360. [Google Scholar] [CrossRef]
- Yi, X.; Zhou, Q.; Zhang, Y.; Zhou, J.; Lin, J. Variants in clopidogrel-relevant genes and early neurological deterioration in ischemic stroke patients receiving clopidogrel. BMC Neurol. 2020, 20, 159. [Google Scholar] [CrossRef]
- Saw, J.; Steinhubl, S.R.; Berger, P.B.; Kereiakes, D.J.; Serebruany, V.L.; Brennan, D.; Topol, E.J. Lack of Adverse Clopidogrel–Atorvastatin Clinical Interaction From Secondary Analysis of a Randomized, Placebo-Controlled Clopidogrel Trial. Circulation 2003, 108, 921–924. [Google Scholar] [CrossRef]
- Lau, W.C.; Waskell, L.A.; Watkins, P.B.; Neer, C.J.; Horowitz, K.; Hopp, A.S.; Tait, A.R.; Carville, D.; Guyer, K.E.; Bates, E.R. Atorvastatin Reduces the Ability of Clopidogrel to Inhibit Platelet Aggregation: A New Drug-Drug Interaction. Circulation 2003, 107, 32–37. [Google Scholar] [CrossRef]
- Yi, X.; Lin, J.; Zhou, Q.; Wu, L.; Cheng, W.; Wang, C. Clopidogrel Resistance Increases Rate of Recurrent Stroke and Other Vascular Events in Chinese Population. J. Stroke Cerebrovasc. Dis. 2016, 25, 1222–1228. [Google Scholar] [CrossRef]
- Siller-Matula, J.M.; Spiel, A.O.; Lang, I.M.; Kreiner, G.; Christ, G.; Jilma, B. Effects of pantoprazole and esomeprazole on platelet inhibition by clopidogrel. Am. Heart J. 2009, 157, e1–e5. [Google Scholar] [CrossRef] [PubMed]
- Ho, P.M.; Maddox, T.M.; Wang, L.; Fihn, S.D.; Jesse, R.L.; Peterson, E.D.; Rumsfeld, J.S. Risk of adverse outcomes associated with concomitant use of clopidogrel and proton pump inhibitors following acute coronary syndrome. JAMA 2009, 301, 937–944. [Google Scholar] [CrossRef] [PubMed]
- Juurlink, D.N.; Gomes, T.; Ko, D.T.; Szmitko, P.E.; Austin, P.C.; Tu, J.V.; Henry, D.A.; Kopp, A.; Mamdani, M.M. A population-based study of the drug interaction between proton pump inhibitors and clopidogrel. CMAJ 2009, 180, 713–718. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.-G.; Chen, M.; Peng, Y.; Chai, H.; Liu, W.; Li, Q.; Ren, X.; Wang, X.-Q.; Luo, W.-L.; Zhang, C.; et al. The impact of smoking on clinical efficacy and pharmacodynamic effects of clopidogrel: A systematic review and meta-analysis. Heart 2013, 100, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.G.; Shin, Y.Y.; Heo, S.H.; Chang, D.I.; Kim, B.J. Smoking and Clopidogrel resistance in ischemic stroke. Neurology 2019, 92, P1.3-007. [Google Scholar]
- Maruyama, H.; Fakuoka, T.; Degushi, I.; Ohe, Y.; Horiuchi, Y.; Kato, Y.; Sehara, Y.; Nagamine, Y.; Sano, H.; Hayashi, T.; et al. Relationship between smoking and responsiveness to clopidogrel in non-cardiogenic ischemic stroke patients. Intern. Med. 2014, 53, 2575–2579. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, Y.; Song, H.; Hou, C.; Cao, Q.; Dong, K.; Huang, X.; Feng, W.; Ovbiagele, B.; Wang, M.; et al. Clopidogrel and ischemic stroke outcomes by smoking status: Smoker’s paradox? J. Neurol. Sci. 2017, 373, 41–44. [Google Scholar] [CrossRef]
- Jeon, S.M. Clopidogrel Resistance in Acute Ischemic Stroke. J. Korean Neurol. Assoc. 2006, 24, 318–322. [Google Scholar]
- Xanmemmedov, E.; Coban, E.; Ciftci-Kavaklıoglu, B.; Acar, E.; Eren, F.; Kale, N.; Soysal, A.; Ciftci-Kavaklioglu, B. The role of platelet count and mean platelet volume in clopidogrel resistance in ischemic stroke patients. J. Neurol. Sci. 2015, 357, e412. [Google Scholar] [CrossRef]
- Patel, S.; Arya, V.; Saraf, A.; Bhargava, M.; Agrawal, C.S. Aspirin and clopidogrel resistance in indian patients with ischemic stroke and its associations with gene polymorphisms: A pilot study. Ann. Indian Acad. Neurol. 2019, 22, 147–152. [Google Scholar] [CrossRef]
- Wiśniewski, A.; Filipska, K.; Sikora, J.; Kozera, G. Aspirin Resistance Affects Medium-Term Recurrent Vascular Events after Cerebrovascular Incidents: A Three-Year Follow-up Study. Brain Sci. 2020, 10, 179. [Google Scholar] [CrossRef] [PubMed]
- Depta, J.P.; Fowler, J.; Novak, E.; Katzan, I.; Bakdash, S.; Kottke-Marchant, K.; Bhatt, D.L. Clinical outcomes using a platelet function-guided approach for secondary prevention in patients with ischemic stroke or transient ischemic attack. Stroke 2012, 43, 2376–2381. [Google Scholar] [CrossRef] [PubMed]
- Yi, X.; Lin, J.; Wang, C.; Huang, R.; Han, Z.; Li, J. Platelet function-guided modification in antiplatelet therapy after acute ischemic stroke is associated with clinical outcomes in patients with aspirin nonresponse. Oncotarget 2017, 8, 106258–106269. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wiśniewski, A.; Filipska, K. The Phenomenon of Clopidogrel High On-Treatment Platelet Reactivity in Ischemic Stroke Subjects: A Comprehensive Review. Int. J. Mol. Sci. 2020, 21, 6408. https://doi.org/10.3390/ijms21176408
Wiśniewski A, Filipska K. The Phenomenon of Clopidogrel High On-Treatment Platelet Reactivity in Ischemic Stroke Subjects: A Comprehensive Review. International Journal of Molecular Sciences. 2020; 21(17):6408. https://doi.org/10.3390/ijms21176408
Chicago/Turabian StyleWiśniewski, Adam, and Karolina Filipska. 2020. "The Phenomenon of Clopidogrel High On-Treatment Platelet Reactivity in Ischemic Stroke Subjects: A Comprehensive Review" International Journal of Molecular Sciences 21, no. 17: 6408. https://doi.org/10.3390/ijms21176408
APA StyleWiśniewski, A., & Filipska, K. (2020). The Phenomenon of Clopidogrel High On-Treatment Platelet Reactivity in Ischemic Stroke Subjects: A Comprehensive Review. International Journal of Molecular Sciences, 21(17), 6408. https://doi.org/10.3390/ijms21176408





